- Infectious Diseases of Livestock
- Part 2
- Enzootic bovine leukosis
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Enzootic bovine leukosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Enzootic bovine leukosis (EBL) is the most common economically important retroviral disease of cattle. It is induced by an exogenous bovine (ruminant) retrovirus, bovine leukaemia virus (BLV). Three different manifestations of the disease are recognized:
- covert infection characterized by seroconversion but no clinical signs,
- persistent B lymphocytosis, and
- the development of solid lymphoid tumours.
As with other retroviral infections, the disease is characterized by a long incubation period that may terminate in the development of persistent lymphocytosis and lymphosarcoma. Enzootic bovine leukosis should be distinguished from sporadic bovine leukosis (SBL), which occurs mainly in young animals aged between four months and two years and presents as juvenile multicentric, thymic or skin forms of the disease. Sporadic bovine leukosis is not associated with BLV and is thought to be non-contagious. Both EBL and SBL occur in southern Africa. The latter disease is discussed at the end of this chapter.
Aetiology
The first descriptions of EBL as a disease entity date back to the end of the nineteenth century. However, it took more than half a century before the causative agent was identified by Malmquist and Miller in 1969.50, 62 Using electron microscopy, they succeeded in detecting retrovirus-like particles in short-term cultures of lymphocytes derived from cattle with persistent lymphocytosis and stimulated with phytohaemagglutinin. The aetiological agent has been grouped together with human T-cell leukaemia virus (HTLV) in the ‘BLV-HTLV’ group of the family Retroviridae.19
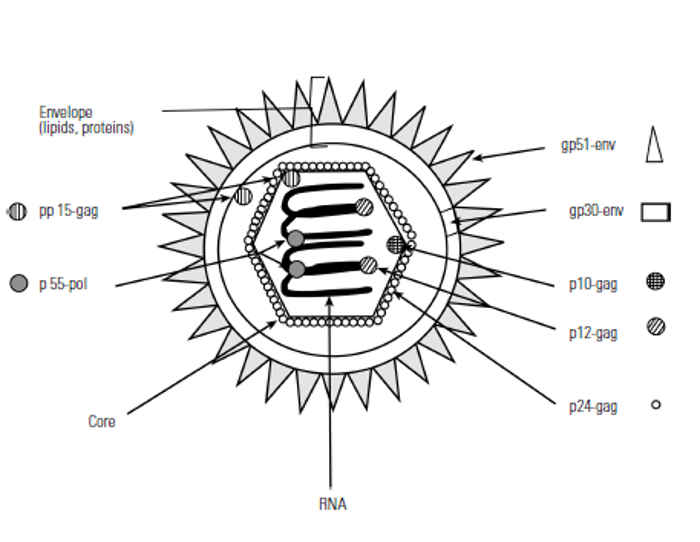
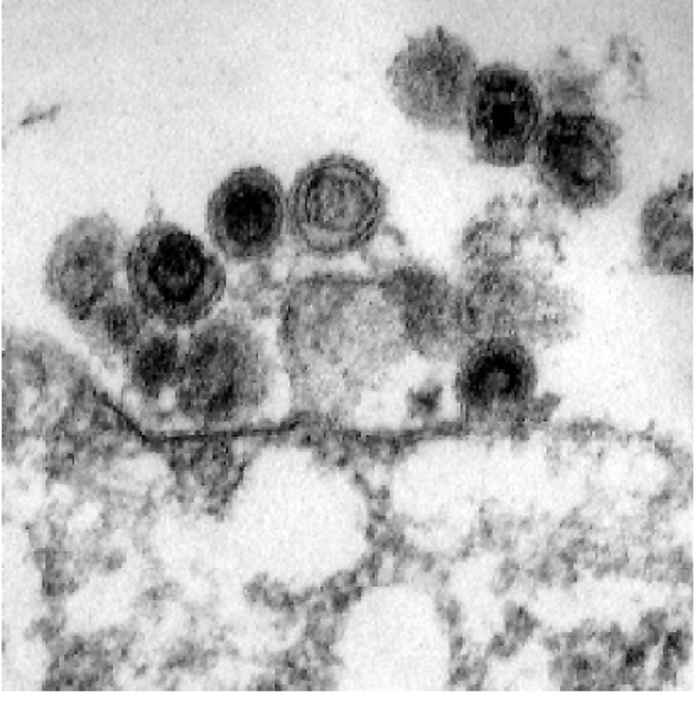
The spherical enveloped virion is approximately 90 to 125 nm in diameter. Its core ranges from 40 to 90 nm in diameter. At the surface of the outer envelope membrane, BLV possesses spikes with an approximate size of 8 nm, which consist of the two glycoproteins gp30 and gp51.16 A schematic drawing as well as an electron micrograph of BLV is shown in Figure Figure 58.1.
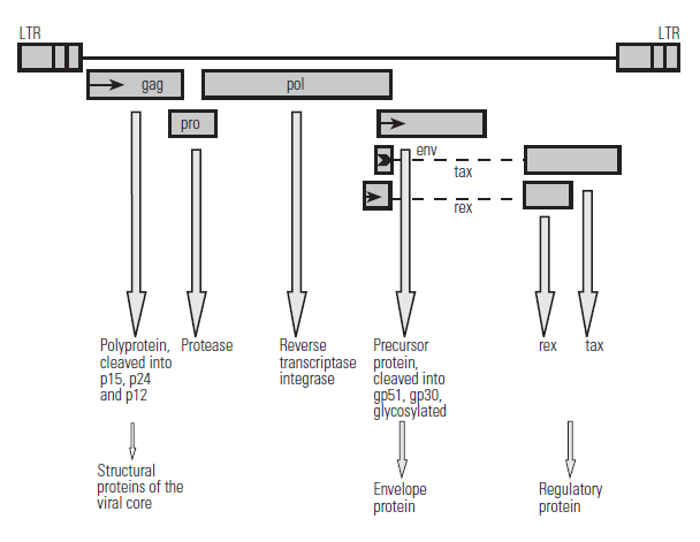
Glycoproteins gp30 and gp51 are both encoded by the env (envelope) gene. The inner nucleocapsid is formed by four proteins (p24, p15, p12, p10) encoded by the gag (group specific antigen) gene. Furthermore, three enzymatic activities are located in the viral core, the reverse transcriptase (RNA-dependent DNA-polymerase for the reverse transcription of the viral RNA into proviral DNA), an integrase (for the integration of the proviral DNA into the cellular DNA), and a protease. The viral genome is diploid, i.e. it consists of two identical single-stranded positive-sense RNAs with a sedimentation constant of 60 to 70S and a length of 8 400 nucleotides.19 The genomic RNA carries a ‘cap’ structure at the 5’end of the molecule and is polyadenylated at the 3’end. Each 5’end is associated with a proline tRNA that is used as a primer for DNA synthesis enabled by reverse transcriptase.19 Further analysis of the BLV genome reveals a striking homology to HTLV-I in the gene arrangement, although their nucleotide sequence homology is limited.78, 79 The BLV genes, their arrangement, the protein products and their functions are schematically shown in Figure Figure 58.2.
Epidemiology
Bovine leukosis virus has a worldwide distribution, but the prevalence of infection is higher in certain regions such as eastern Europe, North and South America, Africa, and Australasia. Economic losses due to culling of BLV-infected animals and reduced milk production are estimated to reach $42 million per year in the USA,74 but may be even higher considering the accompanying losses caused by reduced numbers of animals exported. The incidence of BLV in a single herd can range from 1 to 100 per cent. Interestingly, although all infected animals seroconvert, only 30 per cent of them develop persistent lymphocytosis, and of these, only 30 per cent develop tumours.
Animals remain infected for life and have persistent antibody responses to various BLV antigens. Cattle (Bos indicus and Bos taurus breeds), sheep and also the South American capybara (Hydrochoerus hydrochaeris), a rodent, are susceptible to natural BLV infection,53 but experimentally nonruminants such as pigs, monkeys, rabbits, rats and chickens also seroconvert after BLV inoculation.80 The natural transmission of BLV to sheep is of some relevance where the two species are kept in close contact.51 Experimental infection of sheep with BLV has provided a useful model, as sheep progress to the tumour stage far more quickly than cattle and the response is dose-dependent. Interestingly, infected sheep develop high antibody titres but persistent lymphocytosis does not precede the tumour formation.
As the virus is strictly cell-associated,85 a direct exchange of blood cells between infected and uninfected animals is necessary for transmission, and less than 1 μl of blood from a persistently lymphocytotic animal suffices to initiate infection. 91 The main modes of transmission for BLV are iatrogenic (syringes, needles and instruments contaminated with blood) and direct animal contact. Mechanical transmission by blood-sucking insects may also occur: this has been demonstrated for the biting fly Tabanus fuscicostatus, 29, 30, 34 but could not be experimentally substantiated for the ticks Rhipicephalus appendiculatus and Amblyomma hebraeum.65 Transmission by means of milk and colostrum has been described,60 but the efficacy of this route seems to depend on the titre of maternal antibodies.91 Other body fluids and excretions (e.g. saliva and faeces) do not seem to play a role in transmission.91 Bovine leukosis virus has not as yet been isolated from oocytes and spermatozoa or embryos of infected animals, but pre- and peripartum infections have been reported.6
Based on a limited serological survey,66, 92 the situation in South Africa appears to be similar to that in the USA. In dairy herds, especially those consisting of Holstein-Friesian and Jersey breeds, up to 90 per cent of animals had antibodies to BLV; whereas in beef cattle, especially those of indigenous breeds, antibody prevalence was much lower. Limited data are available for other African countries. In a recent survey in Ugandan slaughterhouses, Azuba and coworkers found that up to 80 per cent of cattle, depending on breed and herd, were BLV seropositive.3
Pathogenesis
Limited information is available regarding the pathogenesis of BLV infection. Hybridization analysis of cellular DNA has proved that BLV is not endogenous to cattle or any other vertebrate species tested.23 Furthermore, the complete absence of hybridization indicates the absence of oncogenes in BLV that are homologous to cellular oncogenes.43 As BLV possesses no known oncogene of its own, the transformation of B cells, and subsequently the formation of tumours, is difficult to explain.
Although the persistent lymphocytosis is attributed to polyclonal B-cell expansion, individual tumours are monoclonal in origin. All BLV-induced tumour cells contain up to four copies of the provirus integrated at different sites, indicating a completely random insertion of the viral genome into the host cell DNA.31, 44, 45 Roughly one out of four of these copies contain deletions; the complete 5’end of the viral genome may be absent from leukaemic cells.31
As all provirus sequences determined so far retain an intact X region (containing the tax and rex regulatory protein), it is suggested that this region plays an important role in tumour genesis.14 In the absence of an oncogene, these observations also suggest that neoplastic transformation may be effected by the activation or suppression of a cellular gene through some mediator. Transactivation seems to be an important mechanism for induction of cellular transformation in other viruses, including HTLV.96 Similarly, whole BLV particles seem to be necessary to induce the initial steps in immortalizing and transforming host cells, whereas subsequently these processes occur independently of BLV,40, 96, 97 e.g., the transient expression of the tax protein may induce permanent expression of cellular genes possibly involved in transformation.15
Two transforming processes are possible. One involves the modification of genetic information of the host cell by BLV, leading to cellular transformation. This hypothesis is supported by the observation that BLV-induced tumours contain mutations in the area of the cellular p53 tumour suppressor gene.22 The second hypothesis argues that processes involved in cellular transformation are based on cellular regulatory mechanisms without alterations of the DNA. In this case, viral proteins (e.g. the tax protein) switch on endogenous ‘oncogenes’ in the host cell and this modification is retained after viral functions have been switched off.96 Furthermore, cells from some BLV-infected animals express a tumour-associated antigen on their surface, and these animals have the tendency to subsequently develop multiple tumours.1, 69
A genetic linkage has been established for disease progression and, very recently, also for susceptibility to BLV infection. Both slower progression and genetic resistance are based on different alleles of the bovine lymphocyte antigen genes encoding the major histocompatibility complex (MHC).99, 100 Holstein-Friesian cattle have higher tumour prevalence rates in comparison to Brown Swiss and Simmenthaler breeds.
Several reports describe a generally suppressed immune response in BLV-infected animals, especially in those in the persistent lymphocytosis stage. Animals with persistent lymphocytosis have a higher incidence of secondary infections (such as that caused by Trichophyton verrucosum), are less able to cope with secondary infections, show decreased productivity and reproductive rates, and have a shorter life span compared to healthy animals.6, 88 Main target cells for BLV in vivo are B cells, but there is also evidence for the possible infection of other immune cells, such as T cells and monocytes/macrophages.35, 64, 81, 84 The intense interaction of these immune cells in the generation of an immune response may be very important for the progression of the disease. Therefore, the currently available knowledge concerning BLV-related changes in each immune cell type is briefly summarized below.
Monocytes/macrophages
These cells play an important role as antigen presenting cells and as the first line of defence to intruding pathogens. They are known to act as ‘virus reservoirs’ in other retrovirus infections, such as those caused by human immunodeficiency virus, feline immunodeficiency virus, simian immunodeficiency virus, and caprine arthritis-encephalitis virus. In vitro investigations have recently proved that ovine24 and bovine monocytes/ macrophages2 can be infected with BLV. Ex vivo analyses on monocytes/macrophages of BLV-infected animals have shown that these cells possess a different phenotype, reduced phagocytic capacity, and altered cytokine responses to stimulation with bacterial lipopolysaccharide, which affected the cytokines interleukin 1 (IL-1) and IL-6, as well as tumour necrosis factor (TNF).57, 94, 95 In addition, the increased production of some cytokines (IL-10, IL-12) seems to be related to disease progression.76, 77
T cells
Changes in T-cell subsets (proportion of CD4+ and CD8+ T cells) and in the phenotype of T cells related to infection with BLV have been reported. These seem to depend on the stage of disease (i.e. aleukaemic versus persistent lymphocytosis), the age of the experimental animals, and when after infection the analyses are performed.32, 82, 83 Furthermore, T cells of animals in the persistent lymphocytosis and tumour stage do not respond to BLV proteins,71 suggesting a reduced capacity of these cells to cope with the virus, and possibly with other pathogens.
B cells
Less than 1,5 per cent of B cells in aleukaemic animals contain proviral DNA, and no viral RNA can be detected in B-cell tumours.41, 45 In vivo, hardly any viral proteins are expressed on the cell surface and no transcription of viral RNA is detectable, suggesting very slow replication of BLV in circulating cells, which is possibly blocked at the transcription level.20, 47, 49 Both viral transcription and expression of viral proteins are enhanced by the activation of the cells due to changes in the micro-environment.67 This led to the isolation of a non-immunoglobulin protein in the plasma of BLV-infected animals that reversibly blocks in vivo expression of the BLV genome.33, 86 This plasma-blocking factor can be irreversibly inhibited by platelet-derived factor.89 Furthermore, BLV infection has been shown to influence the phenotype of the B cells involving elevated expression of MHC class II molecules, cytokine receptors and surface IgM molecules.48, 83 Interestingly, the largest amount of proviralDNAcan be found in a relatively small subset of B cells expressing the CD5 molecule, an activation marker, in the bovine, and the CD11b molecule, an adhesion molecule, in the ovine system.17, 56, 64
Antibodies to BLV that mainly recognize the gp51 protein of the BLV envelope can be detected as early as two weeks after experimental infection.8, 9 Subsequently, antibodies to the gp30, p24, and the rex protein become detectable. These either persist or show a cyclic occurrence.75 In contrast to a ‘normal’ humoral response, the antibody response to BLV is characterized by persistently elevated levels of IgM antibodies. 58 This is interesting with respect to the short viraemia that occurs in the first two weeks after infection, and suggests a constant antigenic stimulus. Although the antibodies which are produced show no neutralizing effect in vivo, they are capable of preventing the infection of susceptible cells in vitro.
Clinical signs
Infection is followed by a clinically silent short-term viraemia which is terminated by the appearance of neutralizing antibodies within 10 to 12 days. As in other retroviral infections, the period between the time of infection and the onset of the first clinical signs is typically long, i.e. in excess of three years.
Three clinical stages have been described for BLV infection. 15 They are:
- The so-called aleukaemic and clinically inapparent stage that is the usual outcome. Infected animals seroconvert and remain infected for the rest of their lives, but develop no clinical signs.52
- After three to six years, 30 to 70 per cent of the infected animals develop persistent lymphocytosis due to the polyclonal expansion of circulating B cells.26, 27 Although this stage is not typically associated with clinical disease, animals with persistent lymphocytosis are often culled as a result of economic losses and generally suppressed immune status.21, 88
- Only a small percentage (0,1 to 10 per cent) of BLV-infected animals develop lymphosarcoma, usually at five to eight years of age,13 and approximately half of these animals have persistent lymphocytosis before developing lymphosarcoma.98 The signs associated with this stage include inappetence, emaciation, and loss in performance. The first sign is usually the swelling of one or more lymph nodes, but in the later stages of the disease lymph node and lymphoid tissue involvement may be widespread, and tumours can be found in every organ containing lymphoid tissue, such as the bone marrow, spleen, lungs, liver and uterus. Other clinical manifestations, as well as the course of the disease, depend on changes in function of the organ or organs involved, the rate of tumour growth, and the extent and distribution of metastases.5, 28
Clinical signs such as exophthalmus, posterior paresis (as a result of tumour invasion of the lumbar epidural space), cardiac failure (sudden death occurring in some animals due to involvement of the myocardium), dysphagia or dyspnoea (if lymph nodes in the pharyngeal or mediastinal regions are affected), or melaena (as a result of ulceration of the intestinal mucosa caused by infiltrating neoplasms) may be manifested. In some animals, the disease may be diagnosed by rectal palpation on the basis of severely enlarged iliac lymph nodes or the involvement of the urogenital system.12 In most animals that have reached this stage, death occurs several weeks or months after the onset of clinical signs,12 and is usually the result of cachexia, secondary infections, anaemia and mechanical encroachment of vital organs.12 In a small proportion of cases, the course may, however, be peracute or acute, particularly if the spleen ruptures, or the heart or adrenals are affected.5
Pathology
In some animals, leukaemia following bone marrow involvement may be present and, in most cases, this is a terminal event.20, 46 Interestingly, the presence of circulating neoplastic cells is not necessarily reflected in elevated total lymphocyte counts.60 In addition, anaemia, which is generally normocytic and normochromic, may develop in as many as 55 per cent of cases.54 Haemoglobin values decline steadily during the course of the disease, with values below 5 g/100 ml occurring commonly.54 Haematocrit values parallel haemoglobin levels. The anaemia is due to a reduction in erythrocyte production after invasion of the bone marrow by neoplastic cells, resulting in a reduction of haemopoietic tissue.54
Elevated serum levels of uric acid, aspartate transaminase (SAST) and lactate dehydrogenase (LDH) may be present, but other chemical pathological findings are not remarkable, or appear only in the terminal stages.54
The lesions depend on the stage of the disease. While changes in healthy animals during the pre-leukaemic stage are found only in the blood, the tumour-bearing stage is characterized macroscopically by either the development of circumscribed solid tumours or more diffuse tumour cell infiltration in the lymphoreticular and other tissues.28, 61 Histologically, the tumours can be classified as lymphoadenose, lymphosarcomatose, reticulosarcomatose, reticulose or mixed.54
In most animals, the neoplastic process becomes widespread. Lymph nodes are most prominently and frequently affected, the number varying from a few to many, if not virtually all. Neoplasms appear to originate from lymphoid tissue, or from tissues with lymphoid potential, at any site of the body. Organs or tissues such as the abomasum, intestines, myocardium, kidneys, liver, epidural tissue or uterus may be involved. 54, 97 The tumours may commence at one focus or many foci simultaneously and then metastasize. At death, the lesions are usually so widely disseminated that it is impossible to determine the site(s) of origin.54, 97 The discrete nodular lesions consist of varying sized greyish-white to yellowish or pinkish uniform masses of soft to moderately firm consistency which, if large, may contain areas of haemorrhage and/or necrosis. Diffuse, infiltrative lesions produce greatly enlarged, pale, soft or friable organs without marked changes in shape.54 Tumours in the epidural space of the spinal cord are most frequently present in the lumbar region and may vary from a few millimetres in diameter to large masses that completely encircle the spinal cord. Lesions in the myocardium occur particularly in the right atrium and often result in clinical and pathological evidence of central venous congestion, while those in the abomasum produce a diffusely thickened mucosa and submucosa, ulceration and haemorrhage.
Microscopically lymphosarcomatous tissue is made up of uniform sheets of closely packed cells, many of which are in mitosis. There is no architectural arrangement and the stroma is scanty. In a survey involving EBL-affected cattle, two basic tumour types were found; in one the majority of cells present were lymphocytes, while in the other they were mostly lymphoblastoid cells.54 In all tumours, both cell types were present but one predominated, and the cellular pattern was consistent in the tumours in all organs involved.54 In another study of lymphosarcoma in cattle, an attempt was made to classify the tumours according to cell size, nuclear cleavage and histological architecture.90 It was found that the majority of tumours in cattle were diffuse, composed of small and medium-sized cells, and contained non-cleaved nuclei.90
Neoplastic cells not only insinuate themselves between normal cells of the organ or tissue concerned and eventually displace them, but they also invade adjacent tissue, such as serosal surfaces, organ capsules and blood vessel walls. Neoplastic emboli may be found in blood and lymphatic vessels and in sinusoids.54
Diagnosis
Enzootic bovine leukosis can often be diagnosed on the clinical signs, but this diagnosis should be confirmed histologically or serologically.
Until 1976, the diagnosis of BLV was dependent on haematological, clinical and pathological examination to detect persistent lymphocytosis or BLV-induced tumours. A commonly used method was the enumeration of B lymphocytes using an age-dependent key. Today, the simplest and routinely used diagnostic method is the detection of antibodies to BLV by various serological tests, including immunodiffusion, immunofluorescence, complement fixation, radio-immuno-assay, ELISA and neutralization tests. These tests normally detect antibodies to gp51 and gp30 proteins, but can also detect antibodies to the p24 protein.38, 59
Proviral DNA can also be detected in peripheral lymphocytes by using the polymerase chain reaction. This enables fast and precise detection of even a very low viral copy number and can be performed on a variety of cells, including cells from the peripheral blood or tumour tissue.4, 25
Further diagnostic tests may involve the direct detection by transmission electron microscopy of BLV particles in cell cultures of susceptible cells following co-cultivation with cells from the buffy coat of suspected BLV-infected animals. In addition, BLV has been shown to induce syncytium formation in some cell lines, e.g. the feline fibroblast-line CC81.13, 42 Two types of syncytia occur: early polykaryocytosis is found in virus-transformed cells a few hours after infection by co-cultivation, whereas late polykaryocytosis is found in appropriate indicator cells four to eight days after infection. The latter has been developed into a quantitative assay.39 Both tests are time-consuming, demand much experience with the technology involved, and are, at least for electron microscopy, expensive. For these reasons they are not routinely used.
Differential diagnosis
Few conditions in cattle are likely to be confused with EBL, although lymphoid hyperplasia in lymph nodes and in the spleen may be observed as a result of chronic inflammation. In isolated organs, such as the kidneys, lymphoid accumulations, which appear as circumscribed white foci, also occur in East Coast fever and malignant catarrhal fever. These diseases, however, are clinically quite distinct from EBL. Macroscopically enlarged lymph nodes or tumour-like nodules in tissues that may resemble the tumours caused by BLV include the lesions associated with mycobacterial infections, actinobacillosis, actinomycosis, or hepatic necrobacillosis.
Control
Although losses due to BLV-associated lymphosarcoma can sometimes be high, the major reason for eradication is the restriction that EBL places on the export of animals to countries free of the disease. The recent demonstration of a relationship between BLV and HTLV-I has added impetus to the attempts to eradicate the disease, although none of the studies performed so far has revealed any hazard to human health caused by BLV. In several countries of the European Union, the disease has been eradicated by testing all animals in an infected herd for antibodies to gp51 at three- to six-month intervals, and eliminating seropositive animals until the herd was free of reactors (test and culling). Alternatively, in BLVendemic areas, herds containing BLV-infected animals were completely isolated (test and isolate). The subsequent sanitation of the herd is based on selecting BLV-negative offspring. Such an approach is feasible in individual herds, but would be difficult to justify on a national scale in southern Africa.
Over the past years, several attempts have been made to construct a successful vaccine against BLV. Most of the candidates were focused on the induction of a strong cellular immune response able to kill BLV-infected cells directly. Viral glycoproteins (mainly the gp51), inactivated BLV, and persistently infected BLV-cell lines were used as antigens in these trials. So far, experimental vaccines using various glycoproteins and lymphoblastic cells have proved ineffective as they did not induce protection against subsequent BLV challenges.63, 87 However, fixed infected cells, inactivated BLV and purified gp51 have produced short-term protection against BLV infection, suggesting that it may prove possible to develop an effective vaccine.31, 72, 73, 97 Effective synthetic vaccines or recombinant live vaccines based on vaccinia virus-expressed BLV glycoproteins in combination with cytokines are also possibilities. 36, 93 Furthermore, genetically modified BLV, able to replicate in the absence of the tax and rex control genes, could be used to produce a vaccine. Recently, a minimal ovine cytotoxic T lymphocyte (CTL) peptide epitope identified within gp51 of BLV consistently induced peptide-specific CTLs. Only sheep whose CTLs were also capable of recognizing BLV-infected cells were fully protected when challenged with BLV.36
Sporadic bovine leukosis
Synonyms: calf lymphoma, thymic lymphoma, juvenile lymphoma, skin lymphoma
In contrast to the relatively well-characterized enzootic bovine leukosis (EBL), little is known about sporadic bovine leukosis (SBL).11, 70 As with EBL, SBL is a fatal disease in affected animals. It occurs mainly in young animals less than two years of age, and is characterized by the development of one or more lymphosarcomas.10 It is the most frequent tumour found in cattle of that age and has a worldwide distribution. Generally, only individual animals are affected and there is no correlation between the occurrence of SBL and EBL in herds with a high prevalence of EBL.
Based on its anatomical location SBL can be divided into three forms:
- a multicentric juvenile form,
- a thymic form in calves six month to two years of age, and
- a skin form (sometimes also involving regional lymph nodes) in animals less than two years of age.70
Eighty per cent of cases of the multicentric, juvenile form occur in calves less than six months old. In 60 per cent of cases the first clinical signs can be detected in the first eight weeks after birth. While many, if not all, lymph nodes are affected, the thymus is usually unaffected. In contrast, older calves generally show the thymic form of SBL. This resulted in the classification of the disease into a calf form and an adolescent thymic form. Based on its onset soon after birth, it is speculated that SBL is initiated during foetal development. In the multicentric form there is symmetrical enlargement of lymph nodes, paleness of the mucous membranes, and dysfunction of organs in close proximity to affected internal lymph nodes. In contrast, the thymic form is restricted to an enlargement of the thymus, subsequently leading to dyspnoea and staunching of blood in the jugular vein. The regional lymph nodes are rarely affected. In both forms, there is a high prevalence of secondary infections suggesting immune suppression. Death occurs one to four weeks after onset of clinical signs.
Both the multicentric and thymic forms are characterized by leukaemic alterations of the white blood cells and the presence of immature and abnormal lymphocytes in peripheral blood.11, 70 An aplastic anaemia is present in the multicentric form, caused by lympholeukotic infiltration of the bone marrow. Approximately 50 per cent of these animals also show an additional thrombocytopenia. Pathological findings are similar to those described for tumours caused by BLV. Sporadic bovine leukosis manifests itself only in the described organs and is therefore clearly distinguishable from EBL.46 Sporadic bovine leukosis is not contagious, cannot be transmitted, and there is no known viral agent linked with its occurrence.68
Similar to the two forms described above, the aetiology of the skin form is not yet known. The disease is characterized by the development of multicentric malignant tumours of lymphoid tissue in the skin, and can also affect the same lymph nodes as described for EBL. Eighty per cent of cases of the skin form of SBL occur in animals between one and four years old, with no gender or species preference described.18, 37, 55 The first tumours usually develop as rapidly growing solid nodules in the skin of the neck, shoulder and back. From here, spread occurs to other areas of the skin, may affect regional draining lymph nodes, and, in the final stage, also affect other lymphoid tissues of internal organs. As the nodules enlarge, the affected skin sheds its hairs and may rupture if pressure is applied to the nodules, but extensive haemorrhage does not occur subsequently. Animals that develop the skin form of SBL do not show signs of general illness at the onset of the disease, but this may occur during the progression of the disease depending on the organs secondarily affected. Circulating lymphoblastoid cells can be detected in 30 per cent of animals with the skin form of SBL. Some animals may recover completely from the skin form for a certain period, but usually develop the nodules again within two years. Histologically, the skin tumours are identical to those in EBL.
References
- AIDA, Y., OKADA, K. & AMANUMA, H., 1993. Phenotype and ontogeny of cells carrying a tumor-associated antigen that is expressed on bovine leukemia virus-induced lymphosarcoma. Cancer Research, 53, 429–437.
- ALTREUTHER, G., 1999. Studien zur in vitro-Infektion boviner Monozyten und Makrophagen mit dem Bovinen Leukämievirus. Thesis at the Faculty of Veterinary Medicine, University of Zu¨rich, Switzerland.
- AZUBA, R.C., ZIEGER, U. & SCHMIDT, F.W., 1994. A prevalence study of bovine leukemia virus infection in slaughtered cattle in selected areas of Uganda. Bulletin of Animal Production for Africa, 42, 13–17.
- BALLAGI PORDANY, A., KLINTEVALL, K., MERZA, M., KLINGEBORN, B. & BELAK, S., 1992. Direct detection of bovine leukemia virus infection: Practical applicability of a double polymerase chain reaction. Zentralblatt fu¨r Veterinärmedizin B, 39, 69–77.
- BENDIXEN, H.J., 1965. Bovine enzootic leukosis. In: bradley, c.a. & cornelius, c.e., (eds). Advances in Veterinary Science, 10, 129–204. New York: Academic Press.
- BOUILLANT, A.M., RUCKERBAUER, G.M., EAGLESOME, M.D., SAMAGH, B.S., SINGH, E.L., HARE, W.C. & RANDALL, G.C., 1982. Attempts to isolate bovine leukemia and bovine syncytial viruses from blood, uterine flush fluid, unfertilized ova and embryos from infected donor cattle. Annales de Recherche Veterinaire, 12, 385–395.
- BRENNER, J., VAN HAAM, M., SAVIR, D. & TRAININ, Z., 1989. The implication of BLV infection in the productivity, reproductive capacity and survival rate of a dairy cow. Veterinary Immunology and Immunopathology, 22, 299–305.
- BRUCK, C., MATHOT, S., PORTETELLE, D., BERTE, C., FRANSSEN, J.D., HERION, P. & BURNY, A., 1982. Monoclonal antibodies define eight independent antigenic regions on the bovine leukemia virus (BLV) envelope glycoprotein gp51. Virology, 122, 342–352.
- BRUCK, C., PORTETELLE, D., MAMMERICKX, M., MATHOT, S. & BURNY, A., 1984. Epitopes of bovine leukemia virus glycoprotein gp51 recognized by sera of infected cattle and sheep. Leukemia Research, 8, 315–321.
- BUNDZA, A., GREIG, A.S., CHANDER, S. & DUKES, T.W., 1980. Sporadic bovine leukosis: A description of eight calves received at Animal Diseases Research Institute from 1974–1980. Canadian Veterinary Journal, 21, 280–283.
- BURNY, A., BEX, F., BRUCK, C., CLEUTER, Y., DEKEGEL, D., GHYSDAEL, J., KETTMANN, R., LECLERCQ, M., MAMMERICKX, M. & PORTETELLE, D., 1979. Biochemical studies on enzootic and sporadic types of bovine leukosis. In: chandra. p., (ed.). Antiviral Mechanisms in the Control of Neoplasia. 200, 83–99. New York: Plenum Press.
- BURNY, A., BEX, F., CHANTRENNE, H., CLEUTER, Y., DEKEGEL, D., GHYSDAEL, J., KETTMANN, R., LECLERCQ, M., LEUNEN, J., MAMMERICKX, M. & PORTATELLE, D., 1978. Bovine leukemia virus involvement in enzootic bovine leukosis. Advances in Cancer Research, 28, 251–311.
- BURNY, A., BRUCK, C., CLEUTER, Y., COUEZ, D., DESCHAMPS, J., GREGOIRE, D., GHYSDAEL, J., KETTMANN, R., MAMMERICKX, M., & MARBAIX, G., 1985. Bovine leukaemia virus and enzootic bovine leukosis. Onderstepoort Journal of Veterinary Research, 52, 133–144.
- BURNY, A., CLEUTER, Y., KETTMANN, R., MAMMERICKX, M., MARBAIX, G., PORTETELLE, D., VAN DEN BROEKE, A., WILLEMS, L. & THOMAS, R., 1988. Bovine leukaemia: Facts and hypotheses derived from the study of an infectious cancer. Veterinary Microbiology, 17, 197–218.
- BURNY, A., CLEUTER, Y., KETTMANN, R., MAMMERICKX, M., MARBAIX, G., PORTETELLE, D., VAN DEN BROEKE, A., WILLEMS, L. & THOMAS, R., 1987. Bovine leukaemia: Facts and hypotheses derived from the study of an infectious cancer. Cancer Survey, 6, 139–159.
- CALAFAT, J. & RESSANG, A.A., 1977. Morphogenesis of bovine leukemia virus. Virology, 80, 42–53.
- CHEVALLIER, N., BERTHELEMY, M., LE RHUN, D., LAINE, V., LEVY, D. & SCHWARTZ CORNIL, I., 1998. Bovine leukemia virus-induced lymphocytosis and increased cell survival mainly involve the CD11b+ B-lymphocyte subset in sheep. Journal of Virology, 72, 4413–4420.
- CLEGG, F.G. & MOSS, B., 1965. Skin leukosis in a heifer—an unusual clinical finding. The Veterinary Record, 77, 271–272.
- COFFIN, J.M., 1996. Retroviridae: The viruses and their replication. In: fields, b.n., knipe, d.m. & howley, p.m., (eds). Fields Virology. pp. 1767–1847. Philadelphia: Lippincott-Raven.
- CORNIL, I. & LEVY, D., 1989. In vivo inhibition of bovine leukemia virus (BLV) expression. Leukemia, 3, 159–161.
- D’ANGELINO, J.L., GARCIA, M. & BIRGEL, E.H., 1998. Productive and reproductive performance in cattle infected with bovine leukosis virus. Journal of Dairy Research, 65, 693–695.
- DEQUIEDT, F., KETTMANN, R., BURNY, A. & WILLEMS, L., 1995. Mutations in the p53 tumor-suppressor gene are frequently associated with bovine leukemia virus-induced leukemogenesis in cattle but not in sheep. Virology, 209, 676–683.
- DESCHAMPS, J., KETTMANN, R. & BURNY, A., 1981. Experiments with cloned complete tumor-derived bovine leukemia virus information prove that the virus is totally exogenous to its target animal species. Journal of Virology, 40, 605–609.
- DOMENECH, A., LLAMES, L., GOYACHE, J., SUAREZ, G. & GOMEZ LUCIA, E., 1997. Macrophages infected with bovine leukaemia virus (BLV) induce humoral response in rabbits. Veterinary Immunology and Immunopathology, 58, 309–320.
- DUBE, S., BACHMAN, S., SPICER, T., LOVE, J., CHOI, D., ESTEBAN, E., FERRER, J.F. & POIESZ, B.J., 1997. Degenerate and specific PCR assays for the detection of bovine leukaemia virus and primate T cell leukaemia/ lymphoma virus pol DNA and RNA: phylogenetic comparisons of amplified sequences from cattle and primates from around the world. Journal of General Virology, 78, 1389–1398.
- ESTEBAN, E.N., THORN, R.M. & FERRER, J.F., 1985. An amplified immunoperoxidase assay to detect bovine leukemia virus expression: Development and comparison with other assays. Cancer Research, 45, 3231–3235.
- ESTEBAN, E.N., THORN, R.M. & FERRER, J.F., 1985. Characterization of the blood lymphocyte population in cattle infected with the bovine leukemia virus. Cancer Research, 45, 3225–3230.
- FERRER, J.F., 1980. Bovine lymphosarcoma. Advances in Veterinary Science and Comparative Medicine, 24, 1–68.
- FOIL, L.D., FRENCH, D.D., HOYT, P.G., ISSEL, C.J., LEPRINCE, D.J., MCMANUS, J.M. & SEGER, C.L., 1989. Transmission of bovine leukemia virus by Tabanus fuscicostatus. American Journal of Veterinary Research, 50, 1771–1773.
- FOIL, L.D., SEGER, C.L., FRENCH, D.D., ISSEL, C.J., MCMANUS, J.M., OHRBERG, C.L. & RAMSEY, R.T., 1988. Mechanical transmission of bovine leukemia virus by horse flies (Diptera: Tabanidae). Journal of Medical Entomology, 25, 374–376.
- FUKUYAMA, S., KODAMA, K., HIRAHARA, T., NAKAJIMA, N., TAKAMURA, K., SASAKI, O. & IMANISHI, J., 1993. Protection against bovine leukemia virus infection by use of inactivated vaccines in cattle. Journal of Veterinary Medical Science, 55, 99–106.
- GATEI, M.H., BRANDON, R.B., NAIF, H.M., MCLENNAN, M.W., DANIEL, R.C. & LAVIN, M.F., 1989. Changes in B cell and T cell subsets in bovine leukaemia virus-infected cattle. Veterinary Immunology and Immunopathology, 23, 139–147.
- GUPTA, P. & FERRER, J.F., 1982. Expression of bovine leukemia virus genome is blocked by a nonimmunoglobulin protein in plasma from infected cattle. Science, 215, 405–407.
- HASSELSCHWERT, D.L., FRENCH, D.D., HRIBAR, L.J., LUTHER, D.G., LEPRINCE, D.J., VAN DER MAATEN, M.J., WHETSTONE, C.A. & FOIL, L.D., 1993. Relative susceptibility of beef and dairy calves to infection by bovine leukemia virus via tabanid (Diptera: Tabanidae) feeding. Journal of Medical Entomology, 30, 472–473.
- HEENEY, J.L., VALLI, P.J., JACOBS, R.M. & VALLI, V.E., 1992. Evidence for bovine leukemia virus infection of peripheral blood monocytes and
- HISLOP, A.D., GOOD, M.F., MATEO, L., GARDNER, J., GATEI, M.H., DANIEL, R.C., MEYERS, B.V., LAVIN, M.F. & SUHRBIER, A., 1998. Vaccine-induced cytotoxic T lymphocytes protect against retroviral challenge. Nature Medicine, 4, 1193–1196
- HOGOSON, G., 1967. A case of congenital skin leukosis in a calf. Journal of Veterinary Medicine B, 13, 748–757.
- JACOBS, R.M., SONG, Z., POON, H., HEENEY, J.L., TAYLOR, J.A., JEFFERSON, B., VERNAU, W. & VALLI, V.E., 1992. Proviral detection and serology in bovine leukemia virus-exposed normal cattle and cattle with lymphoma. Canadian Journal of Veterinary Research, 56, 339–348.
- JERABEK, L., GUPTA, P. & FERRER, J.F., 1979. An infectivity assay for bovine leukemia virus using the immunoperoxidase technique. Cancer Research, 39, 3952–3954.
- KETTMANN, R., CLEUTER, Y., GREGOIRE, D. & BURNY, A., 1985. Role of the 3’ long open reading frame region of bovine leukemia virus in the maintenance of cell transformation. Journal of Virology, 54, 899–901.
- KETTMANN, R., CLEUTER, Y., MAMMERICKX, M., MEUNIER ROTIVAL, M., BERNARDI, G., BURNY, A. & CHANTRENNE, H., 1980. Genomic integration of bovine leukemia provirus: Comparison of persistent lymphocytosis with lymph node tumor form of enzootic. Proceedings of the National Academy of Science USA, 77, 2577–2581.
- KETTMANN, R., DESCHAMPS, J., CLEUTER, Y., COUEZ, D., BURNY, A. & MARBAIX, G., 1982. Leukemogenesis by bovine leukemia virus: Proviral DNA integration and lack of RNA expression of viral long terminal repeat and 3’ proximate cellular sequences. Proceedings of the National Academy of Science USA, 79, 2465–2469.
- KETTMANN, R., DESCHAMPS, J., COUEZ, D., CLAUSTRIAUX, J.J., PALM, R. & BURNY, A., 1983. Chromosome integration domain for bovine leukemia provirus in tumors. Journal of Virology, 47, 146–150.
- KETTMANN, R., MAMMERICKX, M., PORTETELLE, D., GREGOIRE, D. & BURNY, A., 1984. Experimental infection of sheep and goat with bovine leukemia virus: Localization of proviral information on the target cells. Leukemia Research, 8, 937–944.
- KETTMANN, R., WESTIN, E.H., MARBAIX, G., DESCHAMPS, J., WONG STAAL, F., GALLO, R.C. & BURNY, A., 1983. Lack of expression of cellular homologues of retroviral onc genes in bovine tumors. Haematologie und Bluttransfusion, 28, 218–221.
- KLINTEVALL, K., BERG, A., SVEDLUND, G., BALLAGI PORDANY, A. & BELAK, S., 1993. Differentiation between enzootic and sporadic bovine leukosis by use of serological and virological methods. The Veterinary Record, 133, 272.
- LAGARIAS, D.M. & RADKE, K., 1989. Transcriptional activation of bovine leukemia virus in blood cells from experimentally infected, asymptomatic sheep with latent infections. Journal of Virology, 63, 2099–2107.
- LEVKUT, M., PONTI, W., SOLIGO, D., QUIRICI, N., ROCCHI, M. & LAMBERTENGHI DELILIERS, G., 1995. Expression and quantification of IgG and IgM molecules on the surface of lymphocytes of cattle infected with bovine leukaemia virus. Research in Veterinary Science, 59, 45–49.
- LEVY, D., KETTMANN, R., MARCHAND, P., DJILALI, S. & PARODI, A.L., 1987. Selective tropism of bovine leukemia virus (BLV) for surface immunoglobulin-bearing ovine B lymphocytes. Leukemia, 1, 463–465.
- MALMQUIST, W.A., VAN DER MAATEN, M.J. & BOOTHE, A.D., 1969. Isolation, immunodiffusion, immunofluorescence, and electron microscopy of a syncytial virus of lymphosarcomatous and apparently normal cattle. Cancer Research, 29, 188–200.
- MAMMERICKX, M., PALM, R., PORTETELLE, D. & BURNY, A., 1988. Experimental transmission of enzootic bovine leukosis to sheep: Latency period of the tumoral disease. Leukemia, 2, 103–107.
- MAMMERICKX, M., PORTETELLE, D. & BURNY, A., 1985. Application of an enzyme-linked immunosorbent assay (ELISA) involving monoclonal antibody for detection of BLV antibodies in individual or pooled bovine milk samples. Zentralblatt für Veterinärmedizin B, 32, 526–533.
- MARIN, C., DE LOPEZ, N.M., DE ALVAREZ, M., CASTANOS, H., ESPANA, W., LEON, A. & BELLO, A., 1982. Humoral spontaneous response to bovine leukaemia virus infection in zebu, sheep, buffalo, and capybara. Current Topics in Veterinary Medicine and Animal Science, 15, 310–320.
- MARSHAK, R.R., CORIELL, L.L., LAWRENCE, W.C., CROSHAW, J.E., SCHRYVER, H.F., ALTEA, K.P. & NICHOLS, W.W., 1962. Studies on bovine lymphosarcoma. I. Clinical aspects, pathological alterations and herd studies. Cancer Research, 22, 202–216.
- MARSHAK, R.R., HARA, C.D., DUTCHER, R.M., SCHWARTZMAN, R.M., SWITZER, J.W. & HUBEIN, K., 1966. Observations on a heifer with cutaneous lymphosarcoma. Cancer, 19, 724–734.
- MEIROM, R., MOSS, S. & BRENNER, J., 1997. Bovine leukemia virus-gp51 antigen expression is associated with CD5 and IgM markers on infected lymphocytes. Veterinary Immunology and Immunopathology, 59, 113–119.
- MEIROM, R., MOSS, S., BRENNER, J., HELLER, D. & TRAININ, Z., 1997. Levels and role of cytokines in bovine leukemia virus (BLV) infection. Leukemia, 3, 219–220.
- MEIRON, R., BRENNER, J., GLUCKMAN, A., AVRAHAM, R. & TRAININ, Z., 1985. Humoral and cellular responses in calves experimentally infected with bovine leukemia virus (BLV). Veterinary Immunology and Immunopathology, 9, 105–114.
- MERZA, M., SUNDQUIST, B., SOBER, J. & MOREIN, B., 1991. Immunoaffinity purification of two major proteins of bovine leukemia virus (gp51 and p24) and their use for discrimination between vaccinated and infected animals. Journal of Virological Methods, 33, 345–353.
- MILLER, J.M. & VAN DER MAATEN, M.J., 1979. Infectivity tests of secretions and excretions from cattle infected with bovine leukemia virus. Journal of the National Cancer Institute, 62, 425–428
- MILLER, J.M., 1980. Bovine lymphosarcoma. Modern Veterinary Practitioner, 61, 588–591.
- MILLER, J.M., MILLER, L.D., OLSON, C. & GILLETTE, K.G., 1969. Virus-like particles in phytohemagglutinin-stimulated lymphocyte cultures with reference to bovine lymphosarcoma. Journal of the National Cancer Institute, 43, 1297–1305.
- MILLER, J.M., VAN DER MAATEN, M.J. & SCHMERR, M.J., 1983. Vaccination of cattle with binary ethylenimine-treated bovine leukemia virus. American Journal of Veterinary Research, 44, 64–67.
- MIRSKY, M.L., OLMSTEAD, C.A., DA, Y. & LEWIN, H.A., 1996. The prevalence of proviral bovine leukemia virus in peripheral blood mononuclear cells at two subclinical stages of infection. Journal of Virology, 70, 2178–2183.
- MORRIS, S.D., BRYSON, N.R., DE WAAL, D.T., MATTHEE, O., DU PREEZ, E.R., VAN VUUREN, M. & KADISH, E.S., 1996. The possible role of two common three-host ticks, Rhipicephalus appendiculatus and Amblyomma hebraeum, in the transmission of bovine leukosis virus. Journal of the South African Veterinary Association, 67, 148–150.
- MORRIS, S.D., MYBURGH, J.G., VAN VUUREN, M. & VAN DER VYVER, F., 1996. Serological survey to determine the prevalence of bovine leukaemia virus antibodies in dairy cattle on selected farms in the Gauteng and Mpumalanga provinces. Journal of the South African Veterinary Association, 67, 146–147.
- NOTZEL, U., DRESCHER, B. & ROSENTHAL, S., 1982. Detection of bovine leukaemia virus RNA sequences in non-cultivated peripheral lymphocytes by in situ hybridization with 3H-labelled viral cDNA. Acta Virologica, 26, 33–40.
- OGAWA, Y., SAGATA, N., TSUZUKU KAWAMURA, J., ONUMA, M., IZAWA, H. & IKAWA, Y., 1986. No involvement of bovine leukemia virus in sporadic bovine lymphosarcoma. Microbiology and Immunology, 30, 697–701.
- ONUMA, M., 1989. Tumor-associated antigen on bovine leukemia virus-induced bovine lymphosarcoma. Veterinary Immunology and Immunopathology, 22, 213–221.
- ONUMA, M., HONMA, T., MIKAMI, T., ICHIJO, S. & KONISHI, T., 1979. Studies on the sporadic and enzootic forms of bovine leukosis. Journal of Comparative Pathology, 89, 159–167.
- ORLIK, O. & SPLITTER, G.A., 1996. Progression to persistent lymphocytosis and tumor development in bovine leukemia virus (BLV)-infected cattle correlates with impaired proliferation of CD4+ T cells in response to gag- and env-encoded BLV proteins. Journal of Virology, 70, 7584–7593.
- PARFANOVICH, M.I., ZHDANOV, V.M., LAZARENKO, A.A., NOMM, E.M., SIMOVART, YU A., PARAKIN, V.K. & LEMESH, V.M., 1983. The possibility of specific protection against bovine leukaemia virus infection and bovine leukaemia with inactivated BLV. British Veterinary Journal, 139, 137–146.
- PATRASCU, I.V., COMAN, S., SANDU, I., STIUBE, P., MUNTEANU, I., COMAN, T., IONESCU, M., POPESCU, D. & MIHAILESCU, D., 1980. Specific protection against bovine leukemia virus infection conferred on cattle by the Romanian inactivated vaccine BL-VACC-RO. Virologie, 31, 95–102.
- PELZER, K.D., 1997. Economics of bovine leukemia virus infection. Veterinary Clinics North America Food Animal Practitioner, 13, 129–141.
- POWERS, M.A., GROSSMAN, D., KIDD, L.C. & RADKE, K., 1991. Episodic occurrence of antibodies against the bovine leukemia virus Rex protein during the course of infection in sheep. Journal of Virology, 65, 4959– 4965.
- PYEON, D. & SPLITTER, G.A., 1998. Interleukin-12 p40 mRNA expression in bovine leukemia virus-infected animals: Increase in alymphocytosis but decrease in persistent lymphocytosis. Journal of Virology, 72, 6917– 6921.
- PYEON, D., O’REILLY, K.L. & SPLITTER, G.A., 1996. Increased interleukin-10 mRNA expression in tumor-bearing or persistently lymphocytotic animals infected with bovine leukemia virus. Journal of Virology, 70, 5706–5710.
- SAGATA, N. & IKAWA, Y., 1984. BLV and HTLV-I: Their unique genomic structures and evolutionary relationship. Princess Takamatsu Symposium, 15, 229–240.
- SAGATA, N., YASUNAGA, T., TSUZUKU KAWAMURA, J., OHISHI, K., OGAWA, Y. & IKAWA, Y., 1985. Complete nucleotide sequence of the genome of bovine leukemia virus: Its evolutionary relationship to other retroviruses. Proceedings of the National Academy of Science USA, 82, 677.
- SCHWARTZ, I. & LEVY, D., 1994. Pathobiology of bovine leukemia virus. Veterinary Research, 25, 521–536.
- SCHWARTZ, I., BENSAID, A., POLACK, B., PERRIN, B., BERTHELEMY, M. & LEVY, D., 1994. In vivo leukocyte tropism of bovine leukemia virus in sheep and cattle. Journal of Virology, 68, 4589–4596.
- SORDILLO, L.M., HICKS, C.R. & PIGHETTI, G.M., 1994. Altered interleukin-2 production by lymphocyte populations from bovine leukemia virus-infected cattle. Proceedings of the Society for Experimental Biology and Medicine, 207, 268–273.
- STONE, D.M., MCELWAIN, T.F. & DAVIS, W.C., 1994. Enhanced B-lymphocyte expression of IL-2R alpha associated with T lymphocytosis in BLV-infected persistently lymphocytotic cows. Leukemia, 8, 1057–1061.
- STOTT, M.L., THURMOND, M.C., DUNN, S.J., OSBURN, B.I. & STOTT, J.L., 1991. Integrated bovine leukosis proviral DNA in T helper and T cytotoxic/ suppressor lymphocytes. Journal of General Virology, 72, 307–315.
- TAKATORI, I., ITOHARA, S. & YONAIYAMA, K., 1982. Difficulty in detecting in vivo extracellular infective virus in cattle naturally infected with bovine leukemia virus. Leukemia Research, 6, 511–517.
- TAYLOR, J.A. & JACOBS, R.M., 1993. Effects of plasma and serum on the in vitro expression of bovine leukemia virus. Laboratory Investigation, 69, 340–346.
- THEILEN, G.H. & HILLS, D.,1982. Comparative aspects of cancer immunotherapy: Immunologic methods used for treatment of spontaneous cancer in animals. Journal of the American Veterinary Medical Association, 181, 1134–1141.
- TRAININ, Z., BRENNER, J., MEIROM, R. & UNGAR WARON, H.,1996. Detrimental effect of bovine leukemia virus (BLV) on the immunological state of cattle. Veterinary Immunology Immunopathology, 54, 293–302.
- TSUKIYAMA, K., ONUMA, M. & IZAWA, H., 1987. Effect of platelet-derived factor on expression of bovine leukemia virus genome. Archives of Virology, 96, 89–96.
- VALLI, V.E., MCSHERRY, B.J., DUNHAM, B.M., JACOBS, R.M. & LUMSDEN, J.H., 1981. Histocytology of lymphoid tumors in the dog, cat and cow. Veterinary Pathology, 18, 494–512.
- VAN DER MAATEN, M.J. & MILLER, J.M.,1978. Sites of in vivo replication of bovine leukemia virus in experimentally infected cattle. Annales de Recherche Veterinaire, 9, 831–835.
- VERWOERD, D.W., 1990. Veterinary Research Institute, Onderstepoort, South Africa. Unpublished data.
- VON BEUST, B.R., BROWN, W.C., ESTES, D.M., ZARLENGA, D.S., MCELWAIN, T.F. & PALMER, G.H., 1999. Development and in vitro characterization of recombinant vaccinia viruses expressing bovine leukemia virus gp51 in combination with bovine IL4 or IL12. Vaccine, 17, 384–395.
- WERLING, D., HOWARD, C.J., NIEDERER, E., STRAUB, O.C., SAALMULLER, A. & LANGHANS, W., 1998. Analysis of the phenotype and phagocytic activity of monocytes/macrophages from cattle infected with the bovine leukaemia virus. Veterinary Immunology and Immunopathology, 62, 185–195.
- WERLING, D., SILEGHEM, M., LUTZ, H. & LANGHANS, W., 1995. Effect of bovine leukemia virus infection on bovine peripheral blood monocyte responsiveness to lipopolysaccharide stimulation in vitro. Veterinary Immunology and Immunopathology, 48, 77–88.
- WILLEMS, L., CHEN, G., PORTETELLE, D., MAMOUN, R., BURNY, A. & KETTMANN, R., 1989. Structural and functional characterization of mutants of the bovine leukemia virus transactivator protein p34. Virology, 171, 615–618.
- WILLEMS, L., LETELLIER, C., GONZE, M., MARTIN, R., KETTMANN, R., BURNY, A. & MEULEMANS, G., 1989. Expression of the bovine leukemia virus transactivator protein p34 by a recombinant vaccinia virus. Veterinary Immunology and Immunopathology, 22, 201–211.
- WILLEMS, L., THIENPONT, E., KERKHOFS, P., BURNY, A., MAMMERICKX, M. & KETTMANN, R., 1993. Bovine leukemia virus, an animal model for the study of intrastrain variability. Journal of Virology, 67, 1086–1089.
- XU, A., VAN EIJK, M.J., PARK, C. & LEWIN, H.A., 1993. Polymorphism in BoLA-DRB3 exon 2 correlates with resistance to persistent lymphocytosis caused by bovine leukemia virus. The Journal of Immunology, 151, 6977–6985.
- ZANOTTI, M., POLI, G., PONTI, W., POLLI, M., ROCCHI, M., BOLZANI, E., LONGERI, M., RUSSO, S., LEWIN, H.A. & VAN EIJK, M.J., 1996. Association of BoLA class II haplotypes with subclinical progression of bovine leukaemia virus infection in Holstein-Friesian cattle. Animal Genetics, 27, 337–341.