- Infectious Diseases of Livestock
- Part 2
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Paramyxovirus-induced reproductive failure and congenital defects in pigs
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Paramyxovirus-induced reproductive failure and congenital defects in pigs
Previous authors: P D KIRKLAND AND R J LOVE
Current authors:
P D KIRKLAND - Senior Principal Researcher Scientist, Manager Virology Laboratory, BVSc, PhD, FAMS, Elizabeth Macarthur Agriculture Institute, Woodbridge Road, Menangle, New South Wales, 2568, Australia
A W PHILBEY - BVSc (Hon), PhD, FRC Path, MANZCVS (Path), Cert Prod Anim Path, FHEA, MRCVS, Easter Bush Pathology, Royal (Dick) School of Veterinary Studies and The Roslin Institute, University of Edinburgh, Easter Bush, Edinburgh, EH25 9RG, Scotland, United Kingdom
Introduction
Viruses in the family Paramyxoviridae have been associated with emerging diseases in a variety of species, including humans, in several countries. However, there have been few viruses from this family which cause disease in pigs. In 1997, a paramyxovirus, named Menangle virus, was identified in pigs in Australia10 in association with severe reproductive loss and the delivery of mummified foetuses and stillborn piglets, some with malformations.
Aetiology
Menangle virus belongs to the genus Rubulavirus in the subfamily Paramyxovirinae within the family Paramyxoviridae, together with human mumps viruses.
The paramyxoviruses are typically large (150 to 350 nm), pleomorphic enveloped RNA viruses with an outer fringe of ‘spikes’ protruding from the envelope. Contained within the enveloped virion is a long helically symmetrical nucleocapsid which has a herringbone pattern when examined by transmission electron microscopy. The envelope protrusions consist of attachment and fusion proteins which have both neuraminidase and haemagglutinin activity in some viruses, or only haemagglutinin activity in others. Characterization studies of Menangle virus indicate that it is a member of the genus Rubulavirus.4
Transmission electron microscopic examination shows that it has spherical to pleomorphic particles 30 nm to over 300 nm long, containing herringbone-patterned nucleocapsids with a diameter of 19±4 nm and a pitch of 5.8±0.4 nm, surrounded by an envelope with a single fringe of surface projections 17±4 nm long (Figure 1). Menangle virus grows in a wide range of cell types from many species, including cells of porcine and human origin, and induces pronounced cytopathic effects in cell culture such as vacuolation of cells and the formation of syncytia. It is non-haemadsorbing and non-haemagglutinating using erythrocytes from several species, including humans and birds.
Epidemiology
The mechanism of spread of Menangle virus has not been established although respiratory and spread through infected faeces and urine are considered to be most likely ways of transmission. Experimental transmission studies showed that virus is shed in both faeces and urine, with virus being detected for longer periods in urine.3 Studies of archival and newly collected sera indicate that, like other members of the family, the virus is highly contagious and infection spreads rapidly from pig to pig in a fully susceptible population.7 Six months after the estimated time of entry of the virus into the affected piggery, it was found that a high proportion (>90 per cent) of sera collected from pigs of all age categories (n = 88) contained high virus neutralizing antibody titres (≥256). In contrast, all samples (n = 120) collected prior to the estimated time of entry of the virus into the pig population tested negative. All serum samples (n = 50) collected at two other piggeries that had received only weaned pigs from the affected piggery were positive. Positive virus neutralizing antibody titres ranged from 16 to 4096. Testing of sera (n = 1114) from pigs in other piggeries throughout Australia, including piggeries with reproductive problems, indicated that the infection was confined to the affected piggery and the two associated piggeries.
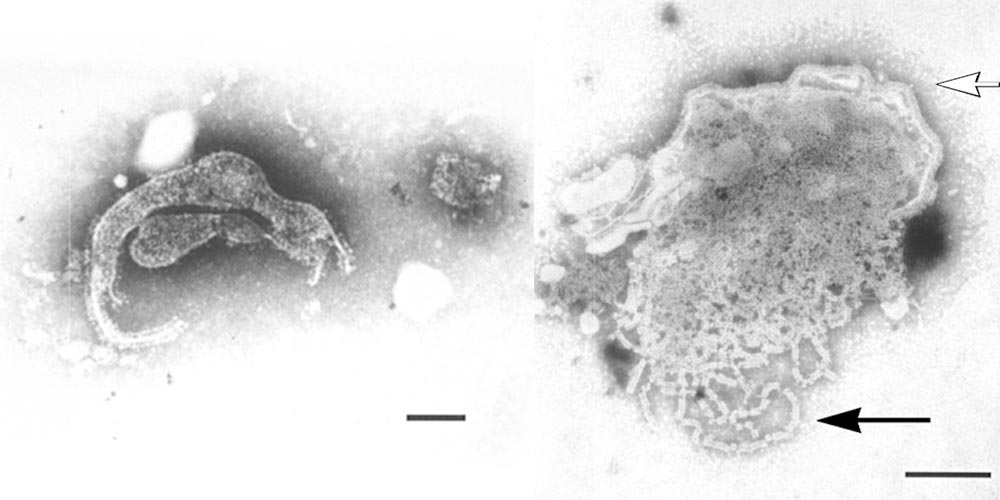
Figure 1 Transmission electron micrographs of Menangle virus negatively stained with 2 per cent phosphotungstic acid; (left) the pleomorphic nature of the virion, bar = 100 nm; (right) a disrupted virion, the virus envelope with surface projections (hollow arrow) and nucleocapsids (solid arrow), bar = 100 nm
Following the initial rapid spread of the infection through the majority of the susceptible pig population, the virus was maintained in the herd by cyclic infection in groups of young pigs as they lost their maternally derived antibody protection between 12 and 16 weeks of age. Almost all selected replacement breeding pigs were seropositive before mating at 28 weeks of age.
It appears that close contact between pigs is required for spread of infection and that the virus does not survive in the environment for any length of time. Susceptible pigs, moved into an uncleaned area that had been vacated by infected pigs three days previously, did not become infected.
Virus is only detected in pigs for 10 to 14 days3 and results in strong immunity. There is no evidence of persistent infections or of carrier pigs.
During the first outbreak of the disease there was a large breeding colony of grey-headed fruit bats (Pteropus poliocephalus) (Figure 2) as well as little red fruit bats (Pteropus scapulatus) roosting within 200 mertres of the affected piggery during the late spring to autumn months, the period when the virus was thought to have entered the piggery. A serological study of 125 sera collected from fruit bats in New South Wales and Queensland found 42 positive by the virus neutralization test, with positive titres ranging from 16 to 256. Samples which tested positive were from 26 of 79 grey-headed fruit bats, 11 of 20 black fruit bats (Pteropus alecto), 4 of 10 spectacled fruit bats (Pteropus conspicillatus), and one unidentified species. None of 15 little red fruit bats was positive. This panel of sera included positive samples collected in November 1997 from a colony of grey-headed fruit bats that were 33 km from the piggery, as well as sera collected in 1996 prior to the time of infection of pigs, supporting the hypothesis that fruit bats were the primary source of the virus. Subsequently, Menangle virus has been isolated from the urine of fruit bats.2 A range of other species in the vicinity of the affected piggery, including rodents (n = 19), birds (n = 13), cattle (n = 60), sheep (n = 70), cats (n = 25) and a dog, were all seronegative. Related viruses have been isolated from bats in South East Asia6 and Africa.1
Pathogenesis and clinical signs
Viruses from the genus Rubulavirus are often associated with systemic disease and have a tropism for the respiratory tract and brain.
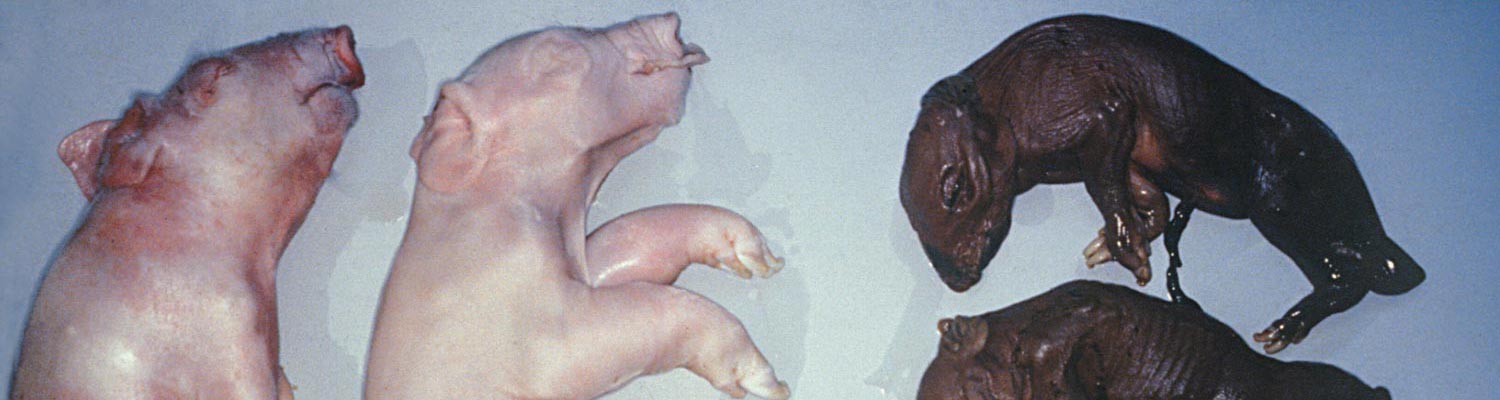
During a five-month period, from mid-April to early September 1997, the farrowing rate during some weeks in the four breeding units of the affected 3 000-sow piggery decreased from an expected 82 per cent to as low as 38 per cent.8 The disease occurred sequentially in all four breeding units of the piggery, affecting the progeny of all parity sows. In the affected weeks, up to 45 per cent of the sows farrowed litters in which there were reduced numbers of live piglets and an accompanying increase in the proportion of mummified and stillborn piglets, some with deformities. There were occasional abortions.
Individual litters characteristically contained mummified foetuses with a range of sizes and gestational ages from 30 days, together with stillborn piglets (some with malformations), and a few normal piglets (Figure 3). This indicates that, similar to parvovirus infections, the virus crosses the placenta early in gestation but only infects a few of the foetuses. Subsequent progressive spread of infection from foetus to foetus within the uterus causes foetal death at different gestational ages.
Pathology
Affected litters usually consisted of a mixture of mummified foetuses, autolysed and fresh stillborn piglets, and a few normal live piglets. Teratogenic defects such as arthrogryposis, brachygnathia and kyphosis were frequent, while cases of artiodactyla were occasionally present. The cranium of some piglets was slightly domed.
Affected stillborn piglets frequently have severe degeneration of the brain and spinal cord but, in some, this may only be slight . teratology ranging from porencephaly to hydranencephaly are most common in the cerebrum (Figure 4). Occasionally there may be fibrinous body cavity effusions and pulmonary hypoplasia.
Histological changes of affected piglets are most marked in the central nervous system. There is extensive degeneration and necrosis of grey and white matter of the brain and spinal cord associated with infiltrations of macrophages and other inflammatory cells. Intranuclear and intracytoplasmic inclusion bodies may be observed in neurones of the cerebrum and spinal cord. These bodies are eosinophilic to amphophilic, and transmission electron microscopy showed that they consist of aggregates of nucleocapsids. Non-suppurative multifocal meningitis, myocarditis and occasionally hepatitis may also be present in some cases.
Diagnosis
As Menangle virus is an infection that appears to have jumped from fruit bats to pigs on a single occasion, most porcine populations would be expected to be fully susceptible. The birth of litters in which there is a marked reduction of normal live piglets and a number of stillborn piglets showing teratological defects implicates possible Menangle virus infection. The most rapid method of excluding Menangle virus infection is to test tissues from stillborn piglets by quantitative reverse transcription polymerase chain reaction assay (qRT-PCR)3 or to test sows for the presence of specific antibody either by virus neutralization or ELISA using whole virus as antigen.
Foetal specimens should be collected for qRT-PCR, virus isolation and serology. Based on the results of experimental studies,3 it is expected that viral RNA would be readily detected by qRT-PCR in the same range of tissues from which virus has been isolated from naturally infected foetuses. Virus can be isolated from a number of organs from stillborn piglets, especially the brain, lungs and myocardium. A wide range of cell cultures support replication of Menangle virus but baby hamster kidney cells (BHK21) have been used for the isolation of the virus from field specimens. Three to sometimes up to five passages are necessary before cytopathic changes including vacuolation of cells, formation of syncytia and focal cell lysis are observed. Since the virus does not haemagglutinate, identification will depend on transmission electron microscopy and its neutralization with specific antiserum or by testing in the Menangle virus- specific qRT-PCR. Neutralizing antibodies may be detected in body cavity fluids of some stillborn piglets.
Differential diagnosis
The birth of litters containing mummified foetuses of varying size together with stillborn piglets is indicative of an in utero viral infection. By far the most common cause of this syndrome in pigs is porcine parvovirus but a variety of infections with other viruses, such as encephalomyocarditis virus, classical swine fever virus, Aujeszky’s disease virus, Japanese encephalitis virus, porcine reproductive and respiratory syndrome (Lelystad) virus and blue eye (La Piedad Michoacan) paramyxovirus11 can cause this syndrome.
A feature that distinguishes Menangle virus infection from all but Japanese encephalitis virus infection is the presence of congenital malformations in the piglets. It must be borne in mind, however, that these are only evident in approximately one third of affected litters. In addition, many of these other viral infections cause disease in both piglets and adults. The only other paramyxovirus causing this syndrome, La Piedad Michoacan paramyxovirus, also belongs to the genus Rubulavirus but differs from Menangle virus in that it agglutinates erythrocytes from mammals and birds.9
Control
Fruit bats, commonly known as ‘flying foxes’, are considered to be the wildlife reservoir of infection for pigs. Therefore, restricting direct and indirect contact with these species is important in preventing the introduction of Menangle virus into a pig population.
Flying foxes do not enter piggery buildings, but they do defaecate and urinate on and around buildings, and are known inadvertently to drop their young in flight, so roofing over all areas accessed by pigs, including outside walkways, is important in risk reduction.
In an outbreak of reproductive disease in a piggery, the infection will most likely have spread through the entire population by the time the first affected litters are farrowed. In small piggeries, infection will probably be self-limiting because the virus does not survive well in the environment (in contrast to parvovirus) and there is no carrier state. In large piggeries, infection may become endemic, with the infection being maintained in groups of pigs as they lose their maternally derived protection. In such situations, it is important to maximize the opportunity for infection of all selected replacement breeding stock prior to mating.
Eradication of Menangle virus from an endemically infected pig population can be achieved by segregating the age groups in which infection is active (e.g. pigs between 10 and 30 weeks of age) from other pigs, for example, by translocation to another site. This should be followed by cleaning and disinfection of the environment before restocking.
Although Menangle virus does not appear to be highly infectious for humans, care should be taken when working with potentially infected pigs or specimens of tissue, blood or fluid. Serological studies showed that two humans out of more than 30 piggery workers directly exposed to infected pigs were infected with the virus, probably with resultant illness.5 These two humans experienced an unexplained severe febrile illness in the weeks following the time of exposure to potentially infective material. Other signs included severe headaches and myalgia. There was no evidence of infection in those who had less direct and less protracted contact with potentially infective material including field veterinarians, and abattoir and laboratory workers.
References
- BAKER, K.S., TODD, S., MARSH, G.A., CRAMERI, G., BARR, J., KAMINS, A.O., PEEL, A.J., YU, M., HAYMAN, D.T., NADJM, B., MTOVE, G., AMOS, B., REYBURN, H., NYARKO, E., SUU-IRE, R., MURCIA, P.R., CUNNINGHAM, A.A., WOOD, J.L. & WANG, L.F., 2013. Novel, potentially zoonotic paramyxoviruses from the African straw-colored fruit bat Eidolon helvum. Journal of Virology, 87, 1348–1358.
- BARR, J.A., SMITH, C., MARSH, G.A., FIELD, H. & WANG, L., 2012. Evidence of bat origin for Menangle virus, a zoonotic paramyxovirus first isolated from diseased pigs. Journal of General Virology, 93, 2590-2594.
- BOWDEN, T.R., BINGHAM, J., HARPER, J.A. & BOYLE, D.B., 2012. Menangle virus, a pteropid bat paramyxovirus infectious for pigs and humans, exhibits tropism for secondary lymphoid organs and intestinal epithelium in weaned pigs. Journal of General Virology, 93, 1007-1016.
- BOWDEN, T.R., WESTENBERG, M., WANG, L.F., EATON, B.T. & BOYLE, D.B., 2001. Molecular characterization of Menangle virus, a novel Paramyxovirus which infects pigs, fruit bats and humans. Virology, 283, 358-373.
- CHANT, K., CHAN, R, SMITH, M., DWYER, D.E., KIRKLAND, P.D. & THE NSW EXPERT GROUP., 1998. Probable human infection with a newly described virus in the Family Paramyxoviridae. Emerging Infectious Diseases, 4, 273–275.
- CHUA, K.B., WANG, L.F., LAM, S.K., CRAMERI, G., YU, M., WISE, T., BOYLE, D., HYATT, A.D. & EATON, B.T., 2001. Tioman virus, a novel paramyxovirus isolated from fruit bats in Malaysia. Virology, 283, 215–229.
- KIRKLAND, P.D., LOVE, R.J., PHILBEY, A.W., ROSS, A.D., DAVIS, R.J. & HART, K.G., 2001. Epidemiology and control of Menangle virus in pigs. Australian Veterinary Journal, 79, 199–206.
- LOVE, R.J., PHILBEY, A.W., KIRKLAND, P.D., ROSS, A.D., DAVIS, R.J. MORRISSEY, C. & DANIELS, P.W., 2001. Reproductive disease and congenital malformations caused by Menangle virus in pigs. Australian Veterinary Journal, 79, 192–198.
- MORENO-LOPEZ, J., CORREA-GIRON, P., MARTINEZ, A. & ERICSSON. A., 1986. Characterization of a paramyxovirus isolated from the brain of a piglet in Mexico. Archives of Virology, 91, 221–231.
- PHILBEY, A.W., KIRKLAND, P.D., ROSS, A.D., DAVIS, R.J., GLEESON, A.B., LOVE, R.J., DANIELS, P.W., GOULD, A.R. & HYATT, A.D., 1998. An apparently new virus (Family Paramyxoviridae) infectious for pigs, humans and fruit bats. Emerging Infectious Diseases, 4, 269–271.
- STEPHAN, H.A., GAY, G.M. & RAMIREZ, T.C., 1988. Encephalomyelitis, reproductive failure and corneal opacity (blue eye) in pigs, associated with a paramyxovirus infection. The Veterinary Record, 122, 6–10.








