- Infectious Diseases of Livestock
- Part 2
- Bovine alphaherpesvirus 2 infections
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Bovine alphaherpesvirus 2 infections
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
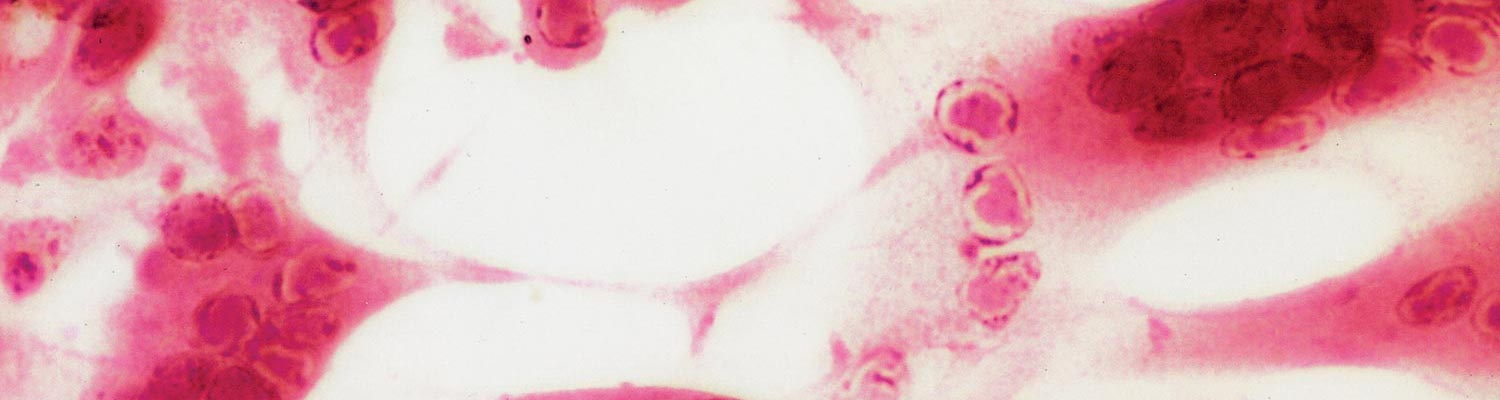
Bovine alphaherpesvirus 2 infections
Previous author: E P J GIBBS
Current author:
E P J GIBBS - Retired Emeritus Professor, BVSc, PhD, FRCVS, College of Veterinary Medicine, University of Florida, Veterinary Academic Building V3-157, 2015 SW 16th Avenue, Gainesville, Florida, 32608, USA
Introduction
Bovine alphaherpesvirus 2 (BHV-2) causes naturally occurring skin disease in cattle. In temperate areas of the world (e.g. the UK), it is generally characterized by the development of an ulcerative condition of the skin of the teats and udder of dairy cattle [bovine herpes mammillitis (BHM), bovine ulcerative mammillitis, ‘udder gangrene’], whereas in the hotter regions of the world (e.g. most parts of Africa) a generalized skin disease [pseudo-lumpy skin disease (PLSD), Allerton disease] is a more frequent manifestation of infection with the virus. Certain other domestic and laboratory species can be infected experimentally. Surveys in Africa indicate that several wildlife species may become subclinically infected.
The aetiological association of a herpesvirus with PLSD was first reported in South Africa in 1957,1 the isolate being designated the Allerton strain. In 1964, the virus was isolated from dairy herds in the British Isles affected with extensive teat infections.46 At one time, the virus causing BHM was considered to have been a recent introduction from Africa, but subsequently, in the late 1960s, 1970s and 1980s, it was isolated from cases of PLSD in the USA,72 and from teat infections in dairy cattle in the USA,69 Canada,38 Australia,67 several European countries,9, 21, 47 and Brazil.2 An Italian isolate of BHV-2 from an oral lesion in a calf has been extensively studied,7 and found to be no different from other European isolates. While there are many countries that have not reported clinical disease in their cattle populations, serological surveys indicate that the virus probably has a worldwide distribution.25, 26, 65 It is unlikely that the BHV-2, now present almost worldwide, originated from Africa.
From the perspective of the cattle industry, as a whole, the economic importance of disease caused by BHV-2 is minimal, but, to the individual farmer facing an outbreak of BHM, it may mean the difference between profit and loss for that year. Secondary bacterial mastitis is a common sequel to BHM, often necessitating drying off the cow. In severe cases of BHM casualty slaughter may be necessary for welfare reasons.30
Despite the fact that infections with BHV-2 have now been recognized for over 60 years, there are aspects related to the pathogenesis and epidemiology which are not fully understood. For example, in temperate regions of the world, outbreaks of BHM predominate while PLSD outbreaks are rare, and BHM occurs only in late summer and early autumn.
Latency has been demonstrated as a sequel to BHV-2 infections in cattle,6, 43 but how this relates to the seasonal occurrence of the disease is still unknown. For a historical perspective of the early studies on these diseases, there are several comprehensive reviews available.8, 13, 41
Aetiology
The causal virus of both BHM and PLSD is classified as bovine alphaherpesvirus 2 (BHV-2) within the family Herpesviridae, subfamily Alphaherpesvirinae, genus Simplexvirus, on the basis of the structure of its genome and general biological characteristics.27 https://talk.ictvonline.org/taxonomy/ This genus also includes human alphaherpesviruses 1 and 2 (herpes simplex viruses 1 and 2) and the B virus of macaques (macacine alphaherpesvirus 1). Bovine alphaherpesvirus 2 is included in this genus on the basis of antigenic relationships and similarities in four genes within the 15 kb U-L 23-29 cluster of the DNA genome.11, 46, 48, 50, 61, 62, 64, 73 Thus, it has been argued, from this apparently close evolutionary relationship, that the virus may represent a greater hazard to public health than has previously been appreciated11, 45 but no clinical disease has, as yet, been recorded in humans exposed to the virus.
The BHV-2 strains isolated in different parts of the world appear to be serologically and genetically similar, irrespective of whether they caused BHM or PLSD.20, 37
Foetal bovine testis and bovine kidney cell monolayers are particularly sensitive for the isolation and growth of BHV-2.13 The cytopathic effect (CPE) of BHV-2 in cell culture is characterized by the formation of large syncytia with most of the nuclei containing intranuclear inclusion bodies.42 The syncytia often enlarge to contain as many as 1 000 nuclei before they detach from the surface of the culture vessel. Cytopathic effect may be noticed within 24 hours of inoculation. Only rarely is it necessary to culture beyond four days to detect CPE. Other cell types, including cells of human origin, support the growth of the virus.45
For a herpesvirus, BHV-2 is moderately resistant to thermal decay once outside the protective environment of the cell. Even after 105 days at ambient room temperature, infective virus could still be recovered from vials (the titre decreased from 8,4 log14 tissue culture infective doses 50 per cent [TCID50]/ml to 2,5 log10 TCID50/ml).54 The virus is inactivated readily by iodophore disinfectants, less effectively by hypochlorites.39
Epidemiology
The dramatic economical impact of BHM in dairy herds in the 1960s in the UK initially led to the misconception that the milking machine played an important role in the transmission of the disease. The importance of this route of transmission was questioned once the relationship of the causative virus of BHM to that of PLSD became known. The discovery, through serological surveys, of the ubiquity of BHV-2 infection in non-dairy cattle and wildlife (particularly African), was further reason to question this assumption.
There is compelling, but circumstantial, evidence to consider that blood-sucking insects mechanically transmit BHV-2.18 Other than transmission to calves, when they suckle infected teats of their dams, infected cattle do not appear to transmit disease to susceptible cattle housed with them. Serological evidence of the subclinical transmission of BHV-2 has been reported between bulls on a stud farm, but the route of transmission was not determined.35
There is ample evidence to suggest insect transmission. In South Africa, the peak prevalence of PLSD is highest in the wet months of the summer and autumn22, 70 and it occurs most commonly in moist, low-lying areas near watercourses. In an outbreak many animals in a herd are usually more or less simultaneously affected, the highest prevalence being in the younger age group. Bovine herpes mammillitis is rarely encountered in South Africa. Bovine herpesvirus 2 was isolated from Musca confiscata trapped on cattle in the acute stage of PLSD, but attempts to transmit the disease with this fly failed.70 Similarly, localized epidemics of BHM have invariably been recorded in the late summer and early autumn in Europe.18 Many of the epidemics have involved dairy farms in river valleys and, in some of them, as many as 40 family-owned dairy and beef herds have been affected in an area as small as 160 square kilometres. As is the case with PLSD, while the epidemiology of the disease suggests spread by biting insects between farms,18, 68 experimental attempts to prove transmission of the disease between cattle by the stable fly, Stomoxys calcitrans, have been unsuccessful.16
Latent infections in cattle and sheep provide a mechanism for interepidemic survival of the virus. It is also possible that certain wildlife species may be involved in the perpetuation of the virus through latency and subclinical infection. The epidemiological significance of infected wildlife in Africa is currently unknown.
Extensive serological surveys of sera collected in nine African countries from a wide range of wildlife species (46) between 1963 and 1980 established that virus neutralizing antibodies to BHV-2 were present in 20 species.24 Fifteen of these were in the family Bovidae, and two in the family Suidae; antibodies were also found in hippopotamus (Hippopotamus amphibius), giraffe (Giraffa camelopardalis) and a green monkey (Cercopithecus aethiops). Although the significance of neutralizing antibodies in the absence of virus isolation is always questioned, the survey confirmed and extended an earlier study in East Africa.51 A similar situation exists in South Africa.4 These reports indicate that infection with BHV-2 is widespread in African wildlife. The highest virus neutralizing titres were recorded in waterbuck (Kobus ellipsiprymnus and K. defassa), reedbuck (Redunca arundinum) and African buffalo (Syncerus caffer). Infection appears to be continuous in free-living populations of buffalo and antibodies are present in the majority of animals by the age of two years. An isolate of BHV-2 was obtained from a buffalo with lesions in the mouth.59 When inoculated intravenously into a captive buffalo, the virus produced a generalized skin disease characteristic of PLSD.58 A serological survey of five species of deer in the UK revealed no evidence of infection31 but a survey of wildlife in Hungary using polymerase chain reaction (PCR) to detect characteristic DNA sequences of BHV-2 in lymph nodes indicated that red deer, fallow deer, roe deer and mouflon were infected.29 Despite the inability to demonstrate mechanical transmission of BHV-2 by insects in experiments with cattle, based on existing knowledge, the epidemiology of BHV-2 infection is believed to be as follows. It must be stressed that this is presented as a hypothesis.
Outbreaks are initiated when latently infected animals (not necessarily cattle) excrete virus. The source of the virus excretion is not known, it could be from a clinically insignificant skin lesion, or in secretions from the nares, vulva or prepuce. It is even conceivable that the virus is not excreted as such, but is present as a viraemia. Whatever the mechanism and location, it is postulated that the virus is then transmitted on the mouthparts of haematophagous biting insects such as Stomoxys calcitrans that feed on the excretions or on blood.
The virus may indeed be excreted by latently infected individuals throughout the year, but, without significant populations of biting insects to collectively introduce a minimal infective dose of virus to a susceptible animal, clinical disease does not develop. Populations of biting insects are usually largest in the late summer and early autumn, hence the seasonal occurrence of disease. In Ireland, it has been suggested that increased levels of phytoestrogens in clover and grass during wet summers may contribute to the seasonality.49
This hypothesis could explain why outbreaks of BHM in temperate areas in newly calved heifers are most likely to develop in the autumn (for example, it is common practice in some countries, such as the UK, to introduce heavily pregnant heifers to the dairy herd at this time of the year to benefit from the higher winter sale price of milk). Once cases begin to occur in newly calved cattle, particularly those with pre-parturient oedema, the extensive serous exudate from the udder skin provides an easily available source of food and a rich source of virus for insect vectors. The influence of the falling ambient temperatures of late summer and autumn on the pathogenesis of the disease (the concentration of virus in the open lesions on which the insect vectors feed is believed to be highest when the skin is cool) further amplifies the cycle to the point when an epidemic can develop. The cycle continues until the ambient temperature falls to the point where it causes a decline in the insect population and the epidemic wanes.
Pathogenesis
The early work on BHM in the UK focused on intradermal and subcutaneous injection of the virus into the skin of the teats and udder of cattle.43, 56 Such teat injections, particularly in newly calved heifers, often led to severe localized disease that spread from the teats to involve the skin of the udder. In some severely affected animals, the lesions extended to the perineum, vulva and vagina.52 In none of these experimental infections, induced by intradermal and/or subcutaneous injection of the virus, was PLSD produced. However, when high-titred inoculations of BHV-2 isolates from cases of BHM were administered intravenously to cattle in the UK, lesions typical of PLSD could be reproduced.23 It was in this way that BHV-2 was identified as the cause of PLSD in other regions of the world, apart from Africa. Nevertheless, the reason why naturally occurring PLSD has not been seen in the temperate areas of Europe remains unexplained.
The virus does not cause infection when applied to normal unabraded skin. Following local injection of BHV-2 isolated from cases of BHM into the skin of the teats and udder of cattle, skin lesions typical of the naturally occurring disease develop within three to eight days. The virus can be recovered from the lesions for approximately a week after they are first seen. The temperature of the skin is believed to be a critical factor in determining the severity of the lesions. When the surface temperature of the skin of cattle was adjusted to above or below rectal temperature by application of heat or cold packs to it, more severe lesions occurred on the cooled skin.34 The titres of virus recovered from the lesions and the period over which virus could be recovered was longer from the cooled skin. In cell culture, the virus replicates to higher titres when the cells are incubated between 30 and 37 °C than it does at 39 °C. It is possible that these observations explain why BHV-2 lesions in cattle occur exclusively in the skin and why outbreaks of BHM occur generally in the cooler months of late summer and early autumn. As mentioned earlier, insect transmission is also believed to play a role in the seasonality of disease.
Latent infections of BHV-2 can be reactivated using corticosteroids.40 The virus has been recovered several months after the initial experimental infection of calves, from nasal swabs and from skin, lymph nodes and nerve tissue collected at necropsy.6 Recrudescence could not be achieved experimentally by some workers,34, 53, 66 an indication that it may be an infrequent event in nature.
Whereas no viraemia has ever been reported following the local injection of BHV-2 isolated from cases of BHM into the skin of the teats and udder, generalized PLSD follows the local injection of BHV-2 isolated from cases of PLSD in about 50 per cent of cattle inoculated, and a low-titre viraemia is detectable.70 In the latter, a focal lesion develops at the site of inoculation within three days with disseminated skin lesions being apparent some six to eight days later. No explanation for this difference between the pathogenesis of the respective infections caused by BHM and PLSD isolates of BHV-2 is currently available. In addition to detecting a low-level viraemia, studies have shown that cattle suffering from PLSD excrete low levels of virus in saliva, semen, faeces and urine.70
Inoculation of the tongue and muzzle of calves with BHV-2 isolated from cases of BHM produced only local lesions.12 Clinical lesions similar to those of PLSD in cattle have been produced in experimentally infected sheep inoculated either intradermally or intravenously with an Australian BHM isolate.71 Virus was recovered from the skin lesions and from circulating leukocytes. Similar to sheep and cattle, goats have been found to be susceptible to intravenous inoculation, but, after intradermal inoculation, the lesions that develop at the site of inoculation resolve without rupture or scabbing.
Day-old rats develop skin infection following experimental inoculation with BHV-2,56 and when it is administered to guinea pigs, a vulvovaginitis develops.63 The latter has been used as a model system to gain comparative insight into the development of recombinant vaccines against BHV-2 (see Control). An Australian BHM isolate has been shown to partially protect guinea pigs against primary genital herpes simplex virus type 2 infections, further confirming the immunological cross-reactivity of these two viruses.36
Clinical signs and pathology
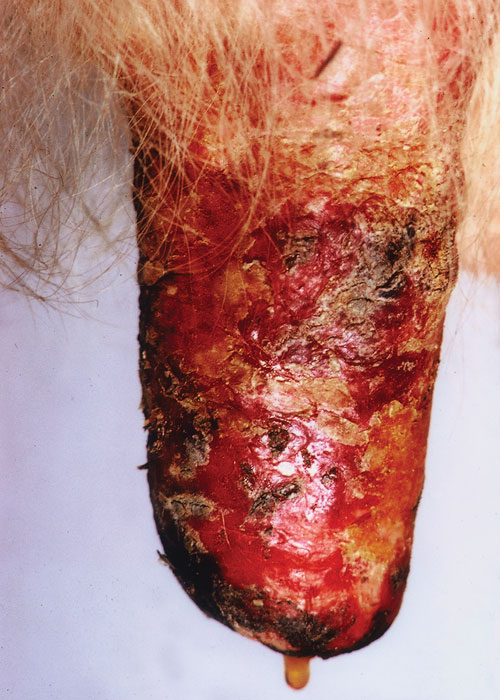
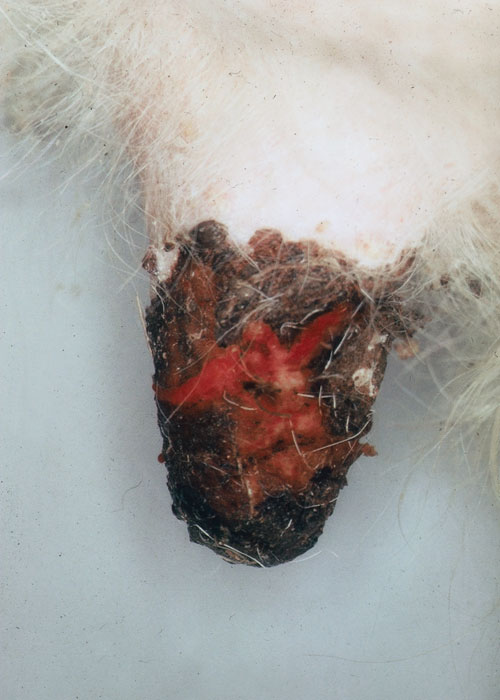
Bovine herpes mammillitis is the most alarming of all the viral diseases that affect the skin of the teats and udder of dairy cattle.15 In a newly calved heifer, the lesions begin as painful oedematous teat swellings, followed within 24 hours by occasional vesication (Figure 1). Affected cows may be impossible to machine milk and some will reject a sucking calf. All four teats may be affected. The vesicles are fragile and soon rupture, exposing an inflamed dermis (Figure 2). Severely affected heifers may have a slightly elevated temperature, but this may be due to the pain of the lesions, not the infection per se. In cows in the later stages of their lactation, the lesions are usually single, but occasionally multiple lesions, all at the same stage of development, may be seen. Scabbing follows (Figure 3), and, in the uncomplicated case, healing occurs within three weeks beneath the scab to leave no scars.
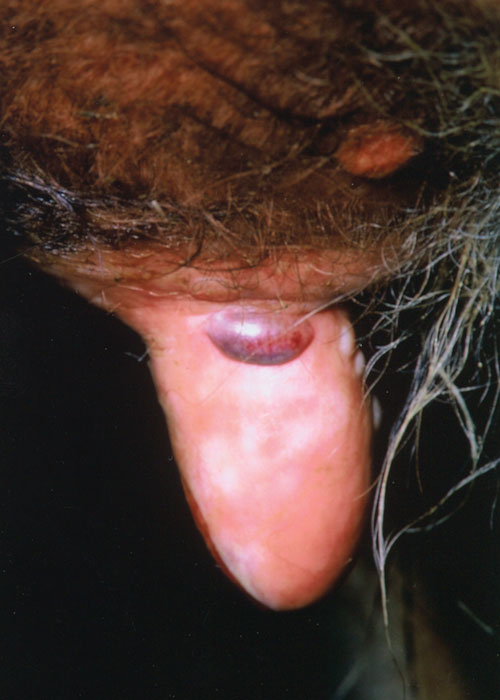
Figure 1 Vesicles of bovine herpes mammillitis in the skin of the medial surface of the teat of a newly calved heifer. Haemorrhage into one vesicle occurred

Figure 2 Ruptured epidermis of the teat skin in a newly-calved heifer suffering from bovine herpes mammillitis. Note the oedema
In many cattle, the friction of the milking machine or the sucking of the calf may prevent scab formation and a raw granulating area is exposed that heals slowly. Frequently, the stage following the oedema is a bluish-black discoloration of the surface of almost the entire teat, simulating gangrene. In such cases the epidermis of the teat usually then separates as a ‘thimble’ and healing may take up to 10 weeks. Secondary bacterial infection of the lesion is frequent (Figure 4). When the teat orifice is involved, bacterial mastitis is a common sequel.
The most severe lesions occur in newly calved heifers with pre-parturient oedema. In such animals, the lesions of BHM may be extensive and also involve the skin of the udder and extend to involve the perineum.30 Vulvovaginitis, by local extension of infection, has been recorded.52 Occasionally, BHM is seen in heavily pregnant heifers before they have entered the milking herd.12 Outbreaks have been recorded in beef herds.28
In animals affected late in their lactation cycle, the lesions may be small and relatively insignificant. Whether such animals are partially immune has never been determined. Anecdotal field observations and relatively short- term pathogenicity studies with the virus indicate that cattle are not susceptible to reinfection, certainly not within the same lactation.56
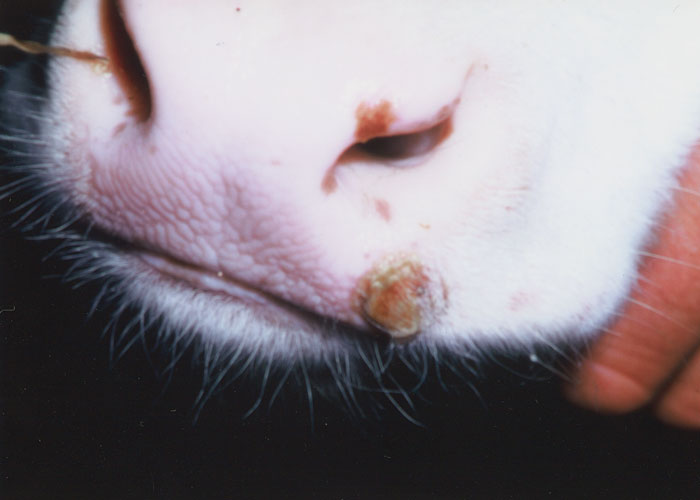
Localized lesions on the muzzle (Figure 5) and mouth have been recorded in calves.12, 30 In some of these cases, there is a relationship to cattle affected with BHM, but this is not always proven.7
Pseudo-lumpy skin disease is characterized by the sudden onset of firm, superficially situated, circular nodules in the skin, ranging in size from 10 to 25 mm in diameter.70 The nodules are elevated approximately 5 mm above the skin surface and have a characteristically flat surface and a central depression. Serous fluid may ooze from the centre of such acute lesions. A slightly elevated rectal temperature may accompany the onset of the disease. In severely affected animals, there may be several hundred nodules, with some coalescing to form irregularly shaped plaques. More commonly, only a few lesions are present and these are clustered around the face, neck, and perineum. As the lesions heal, the affected skin of the nodules dries and becomes hard and by two weeks sloughs, taking the hairs with it. At this stage the animal’s coat has a ‘scurfy, moth-eaten’ appearance. Scar tissue is not formed.
The histopathology of the lesions in BHV-2 infections, whether BHM, PLSD or the oral disease in calves, is similar.32, 57 Severe intercellular oedema of the epidermis characterizes the early lesion with a prominent perivascular infiltration of mononuclear cells present in the dermis. Intranuclear eosinophilic inclusion bodies may be found in some epidermal cells.
Diagnosis
A tentative diagnosis of PLSD or BHM is based on the presence of characteristic lesions in the skin of the body or udder and teats. However, the differentiation between PLSD and early or mild cases of lumpy skin disease may be difficult and laboratory confirmation is essential.70
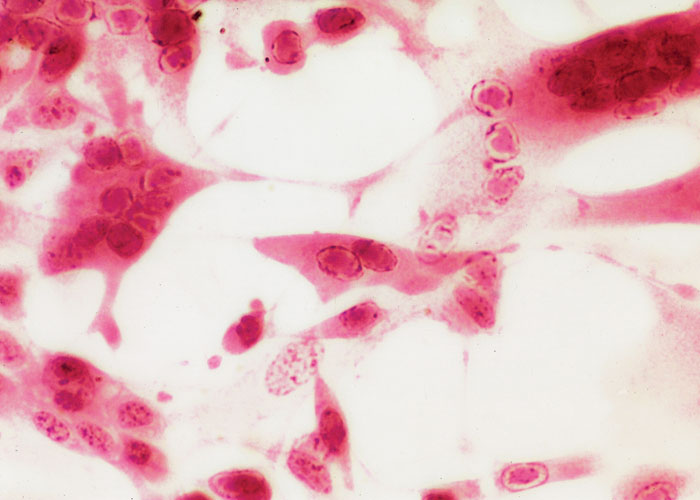
Bovine herpesvirus 2 can be readily isolated in cell culture from necrotic tissue collected from early clinical cases of BHM and PLSD17 (Figure 6). Vesicular fluid may contain as many as 7,0 log10TCID50/ml of virus but is not usually available because of the fragility of vesicular lesions. Once scabs are established and dried (from about the third day after commencement of clinical signs), the titre of virus falls considerably. Serological tests such as virus neutralization and immunofluorescence can be used to confirm the identity of the isolate. Electron microscopic examination of skin biopsies collected during the peak of the infection as well as scab material is useful to differentiate BHV-2 infections from the pox viral diseases (e.g. pseudo-cowpox, cowpox and lumpy skin disease) with which they may be clinically confused.19 A multiplex PCR is used in Brazil to differentiate pseudocowpox, vaccinia infections, and BHM.
The histopathological examination of a skin biopsy can provide useful information, but it may not be definitive, particularly for BHM. Detection of a four-fold rise in neutralizing antibody in paired serum samples collected 21 days apart from affected animals also has limited value. Apart from the difficult logistics involved in collecting the paired serum samples, the first serum sample often has a high antibody titre.
Differential diagnosis
Bovine herpes mammillitis
The dairy cow has been specifically bred for milk production. This has anatomically exposed the teats to a greater risk of physical damage and infections, whether it is the trauma of being dragged through muddy gateways, sunburn, frostbite, or incorrectly used milking machines. Apart from BHV-2 there are several other viruses (e.g. pseudocowpox and cowpox) that have a predilection for the teat skin. The making of a specific diagnosis of a disease affecting the skin of a bovine teat is therefore seldom easy, but there are several comprehensive reviews available.15, 41
While a single case may not be definitive, a clinical diagnosis can usually be established by an examination of the affected herd; the general characteristics of the different diseases usually allow a provisional diagnosis. It must be borne in mind, however, that it is not uncommon to have more than one virus condition affecting the teats of even individual animals, let alone those of several or more animals in the herd. Pseudocowpox and different types of teat warts are present in many herds as chronic infections. Where vesicles or acute scab formation are present on the teats, the possibility of foot-and-mouth disease should always be considered.
Pseudo-lumpy skin disease
Differentiation of PLSD from the early and milder cases of lumpy skin disease can be challenging and confirmatory laboratory diagnosis is often necessary.70 PCR has been successfully used to detect DNA sequences of BHV-2 in cattle in the USA with suspected PLSD.10 The generally benign nature of PLSD and the superficial lesions in the skin and the shorter course of the disease aid considerably in clinical differentiating it from lumpy skin disease.3
Control
There are few methods available to control either PLSD or BHM.15 While insect spraying and palliative teat dips can be applied in the case of BHM, these are often of limited effectiveness. An outbreak of BHM in a dairy herd can involve the majority of cows in the herd, often within a week, and most susceptible animals have already been infected by the time that control measures are implemented. Such action, however, serves to reduce the opportunity of further transmission beyond the farm. Teat dipping helps to reduce the severity of secondary bacterial infection of the lesions and, it is assumed that this reduces the risk of subsequent mastitis. Several antiviral drugs have been evaluated against BHV-2 and some have been shown to be effective in vitro;60 however, no clinical trials have been reported.
The infrequency of outbreaks of BHM or PLSD has precluded the development of a commercial vaccine. In herds affected with BHM, the intramuscular injection of unattenuated virus grown in cell culture has been used as a simple vaccine to prevent further cases developing. In experimental studies with cattle, this approach has been shown to be effective in preventing disease.55 The efficacy of the experimental vaccine was evaluated, during an epidemic in the UK in 1968, in the control of overt infection on nine farms where there were clinical cases of BHM.14 In this trial, over 450 cows in the affected milking herds and 127 heifers, about to calve, were each vaccinated with 4,7 log10 TCID50/ml of BHV-2. Apart from a local cutaneous lesion (5 to 10 mm in diameter) at the site of intramuscular inoculation in the neck, no adverse clinical disease was related to vaccination, and the cutaneous lesions healed rapidly without any loss of hair. The vaccine was considered to be highly effective by the farmers, but a retrospective serological analysis indicated that most of the unaffected cattle that were vaccinated already had antibody levels to BHV-2 prior to vaccination.
Simple unlicensed vaccines, such as that described above, are now unacceptable for use in most countries. A thymidine kinase deficient mutant of BHV-2 holds promise as a candidate for use as a vaccine.63 It has been shown to be effective in reducing the severity of infection in the guinea pig model, but there are no reports of its evaluation in cattle.
References
- ALEXANDER, R.A., PLOWRIGHT, W. & HAIG, D.A., 1957. Cytopathogenic agents associated with lumpy skin disease of cattle. Bulletin of Epizootic Diseases of Africa, 5, 489–492.
- ALICE, F.J., 1977. Isolation of bovine herpes mammillitis virus in Brazil. Revista de Microbiologia, Brazil, 8, 9–15.
- BARNARD, B.J.H., 1994. Pseudo-lumpy skin disease/bovine herpes mammillitis. In: COETZER, J.A.W., THOMSON, G.R. & TUSTIN, R.C., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press Southern Africa.
- BARNARD, B.J.H., 1997. Antibodies against some viruses of domestic animals in southern African wild animals. Onderstepoort Journal of Veterinary Research, 64, 95–110.
- Cargnelutti, J.F., Weiblen, R. & Flores, E.F., 2017. A multiplex PCR for viruses associated with exanthematic and vesicular disease in cattle. Journal of Virological Methods, 239, 38-41.
- CASTRUCCI, G., FERRARI, M., FRIGERI, F., RANUCCI, S., CILLI, V., TESEI, B. & RAMPICHINI, L., 1982. Reactivation in calves of Bovid herpesvirus 2 latent infection. Archives of Virology, 72, 75–81.
- CASTRUCCI, G., PEDINI, B., CILLI, V. & ARANCIA, G., 1972. Characterisation of a viral agent resembling bovine herpes mammillitis virus. The Veterinary Record, 90, 325–335.
- CILLI, V. & CASTRUCCI, G., 1976. Infection of cattle with bovid herpesvirus 2. Folia Veterinaria Latina, 6, 1–44.
- DILOVSKI, M., TEKERLEKOV, P. & KHADZHIEV, G., 1974. Isolation of the bovine herpes mammillitis virus in Bulgaria. Veterinarnomeditsinski Nauki, 11, 60–65.
- d'Offay, J.M., Floyd, J.G., Eberle, R., Saliki, J.T., Brock, K.V., D'Andrea, G.H. & McMillan, K.L., 2003. Use of a polymerase chain reaction assay to detect bovine herpesvirus type 2 DNA in skin lesions from cattle suspected to have pseudo-lumpy skin disease. Journal of the American Veterinary Medical Association, 222, 1404-1407.
- EHLERS, B., GOLTZ, M., EJERCITO, M.P., DASIKA, G.K. & LETCHWORTH, G.J., 1999. Bovine herpesvirus type 2 is closely related to the primate alphaherpesviruses. Virus Genes, 19, 197–203.
- GIBBS, E.P.J. & COLLINGS, D.F., 1972. Observations on bovine herpes mammillitis virus infections of heavily pregnant heifers and young calves. The Veterinary Record, 90, 66–68.
- GIBBS, E.P.J. & RWEYEMAMU, M.M., 1977. Bovine herpes viruses. II. Bovine herpes virus 2 and 3. The Veterinary Bulletin, 6, 411–425.
- GIBBS, E.P.J.,1970. A study on virus infections of the skin of the bovine teat. PhD Thesis, University of Bristol.
- GIBBS, E.P.J., 1984. Viral diseases of the skin of the bovine teat and udder. Veterinary Clinics of North America, Large Animal Practice, 6, 187–202.
- GIBBS, E.P.J., JOHNSON, R.H. & GATEHOUSE, A.G., 1973. A laboratory technique for studying the mechanical transmission of bovine herpes mammillitis virus by the stable fly (Stomoxys calcitrans L.). Research in Veterinary Science, 14, 145–147.
- GIBBS, E.P.J., JOHNSON, R.H. & OSBORNE, A.D., 1970. The differential diagnosis of viral skin infections of the bovine teat. The Veterinary Record, 87, 395–401.
- GIBBS, E.P.J., JOHNSON, R.H. & OSBORNE, A.D., 1972. Field observations on the epidemiology of bovine herpes mammillitis. The Veterinary Record, 91, 395–401.
- GIBBS, E.P.J., JOHNSON, R.H. & VOYLE, C.A., 1970. The differential diagnosis of virus infections of the bovine teat skin by electron microscopy. Journal of Comparative Pathology, 80, 455–463.
- GIGSTAD, D.C. & STONE, S.S., 1977. Clinical, serologic, and cross-challenge response and virus isolation in cattle infected with three bovine dermatotropic herpesviruses. American Journal of Veterinary Research, 38, 753–757.
- GUERIN, L.A. & CULLINANE, J.A., 1972. Bovine herpes mammillitis—a virus infection as seen in practice. Irish Veterinary Journal, 26, 10–12.
- HAIG, D.A.,1957. Lumpy skin disease. Bulletin of Epizootic Diseases of Africa, 5, 421–430.
- HAIG, D.A., 1967. Production of generalised skin lesions in calves inoculated with bovine mammillitis virus. The Veterinary Record, 80, 311–312.
- HAMBLIN, C. & HEDGER, R.S., 1982. Prevalence of neutralizing antibodies to bovid herpesvirus 2 in African wildlife. Journal of Wildlife Diseases, 18, 429–436.
- HEDGER, R.S., BARNETT, I.T. & GRAY, D.F., 1980. Some virus diseases of domestic animals in the Sultanate of Oman. Tropical Animal Health and Production, 12, 107–114.
- Imai, K., Ishihara, R. & Nishimori, T., 2005. First demonstration of bovine herpesvirus 2 infection among cattle by neutralization test in Japan. Journal of Veterinary Medical Science, 67, 317-320.
- INTERNATIONAL COMMITTEE ON TAXONOMY OF VIRUSES (ICTV)., 2016. Release http://ictv.global/report. Virus Taxonomy. Accessed November 24 2017.
- JOHNSTON, W.S., WRAY, C. & SCOTT, J.A., 1971. An outbreak of bovine herpes mammillitis in a suckler herd. The Veterinary Record, 88, 372.
- Kalman, D. & Egyed, L., 2005. PCR detection of bovine herpesviruses from nonbovine ruminants in Hungary. Journal of Wildlife Diseases, 41, 482-488.
- Kemp, R., Holliman, A. & Nettleton, P.F., 2008. A typical bovine herpes mammillitis affecting cows and calves. Veterinary Record, 163, 119-121.
- LAWMAN, M.J.P., EVANS, D., GIBBS, E.P.J., MCDIARMID, A. & ROWE, L., 1978. A preliminary survey of British deer for antibody to some virus diseases of farm animals. British Veterinary Journal, 134, 85–91.
- LEPPER, A.W., HAIG, D.A. & WILCOX, J.,1969. Cellular pathology of calves experimentally infected with bovine herpes mammillitis virus. Journal of Comparative Pathology, 79, 489–494.
- LETCHWORTH, G.J. & CARMICHAEL, L.E., 1982. Bovid herpesvirus 2 latency: Failure to recover virus from central sensory nerve ganglia. Canadian Journal of Comparative Medicine, 46, 76–79.
- LETCHWORTH, G.J. & CARMICHAEL, L.E., 1984. Local tissue temperature: A critical factor in the pathogenesis of bovid herpesvirus 2. Infection and Immunity, 43, 1072–1079.
- LETCHWORTH, G.J., CARMICHAEL, L.E. & LEIN, D.H., 1982. Bovid herpesvirus 2: Natural spread among breeding bulls. Cornell Veterinarian, 72, 200–210.
- MARAGOS, C. & MAY, J.T., 1988. Modification of the genital herpes infection in a guinea pig model, by prior immunization with bovine herpes mammillitis virus. Microbiology and Immunology, 32, 547–550.
- MARAGOS, C., SHAW, R. & MAY, J.T., 1986. Restriction endonuclease cleavage analysis of DNA from two bovine herpes mammillitis viruses, isolated in different parts of Australia. Australian Veterinary Journal, 63, 372–373.
- MARTIN, J.R., HARVEY, D. & MONTPETIT, C., 1987. Bovine herpes mammillitis in Quebec. Canadian Veterinary Journal, 28, 532.
- MARTIN, W.B. & JAMES, Z.H., 1969. Inactivation of the bovine mammillitis herpesvirus by disinfectants. The Veterinary Record, 85, 100.
- MARTIN, W.B. & SCOTT, F.M.M., 1979. Latent infection of cattle with bovid herpesvirus 2. Archives of Virology, 60, 51–58.
- MARTIN, W.B.,1990. Virus Diseases of Ruminants (DINTER, Z. & MOREIN, B., Eds.), Elsevier Science, Amsterdam, 109–116.
- MARTIN, W.B., HAY, D., CRAWFORD, L.V., LE BOUVIER, G.L. & CRAWFORD, E.M., 1966. Characteristics of bovine mammillitis virus. Journal of General Microbiology, 45, 325–332.
- MARTIN, W.B., JAMES, Z.H., LAUDER, I.M., MURRAY, M. & PIRIE, H.M., 1969. Pathogenesis of bovine mammillitis virus infection in cattle. American Journal of Veterinary Research, 30, 2151–2166.
- MARTIN, W.B., MARTIN, B., HAY, D. & LAUDER, I.M., 1966. Bovine ulcerative mammillitis caused by a herpes virus. The Veterinary Record, 78, 494–497.
- MAY, J.T. & ORDERS, G.,1993. Bovine mammillitis virus growth in human cells. The Veterinary Record, 133, 148.
- MAY, J.T., AWAD, M., REUM, A. & SHEPPARD, M., 1990. Bovine herpes mammillitis virus thymidine kinase gene. Acta Virologica, 34, 188–192.
- MULLER, R., ENGELS, M., METZLER, A.E., BOLLER, H. & WYLER, R., 1984. The first reported outbreak of bovine herpes mammillitis in Switzerland. Tierärztliche Praxis, 12, 297–305.
- NORRILD, B., LUDWIG, H. & ROTT, R., 1978. Identification of a common antigen of herpes simplex virus, bovine herpes mammillitis virus, and B virus. Journal of Virology, 26, 712–717.
- Oconnor, M., 1995. Meteorological features associated with outbreaks of bovine herpes mammillitis in Ireland. Irish Veterinary Journal, 48, 71-80.
- PIETSCHMANN, S.M., GELDERBLOM, H.R. & PAULI, G., 1989. Compartment-specific immunolocalization of conserved epitopes of the glycoprotein gB of herpes simplex virus type 1 and bovine herpes virus type 2 in infected cells. Archives of Virology, 108, 1–17.
- PLOWRIGHT, W. & JESSETT, D.M., 1971. Investigations of Allerton-type herpes virus infection in East African game animals and cattle. Journal of Hygiene, Cambridge, 69, 209–222.
- POVEY, R.C. & JAMES, Z.H., 1973. Bovine herpes mammillitis virus and vulvo-vaginitis. The Veterinary Record, 92, 231–233.
- PROBERT, M. & POVEY, R.C., 1975. Experimental studies concerning the possibility of a latent carrier state in bovine herpes mammillitis (BHM). Archives of Virology, 48, 29–38.
- RWEYEMAMU, M.M. & JOHNSON, R.H., 1967. Bovine herpes mammillitis virus 1. In vitro behaviour of the virus. British Veterinary Journal, 124, 9–15.
- RWEYEMAMU, M.M. & JOHNSON, R.H.,1969. The development of a vaccine for bovine herpes mammillitis. Research in Veterinary Science, 10, 419–427.
- RWEYEMAMU, M.M., JOHNSON, R.H. & MCCREA, M.R., 1968. Bovine herpes mammillitis virus. III. Observations on experimental infection. British Veterinary Journal, 124, 317–325.
- RWEYEMAMU, M.M., OSBORNE, A.D. & JOHNSON, R.H.,1969. Observations on the histopathology of bovine herpes mammillitis. Research in Veterinary Science, 10, 203–207.
- SCHIEMANN, B., GWAMAKA, B. & KALUNDA, M., 1972. Pathogenicity for a buffalo (Syncerus caffer) of Allerton-type herpes virus isolated from a Tanzanian buffalo. Journal of Wildlife Diseases, 8, 141–145.
- SCHIEMANN, B., PLOWRIGHT, W. & JESSETT, D.M.,1971. Allerton-type herpes virus as a cause of lesions of the alimentary tract in a severe disease of Tanzanian buffaloes (Syncerus caffer). The Veterinary Record, 89, 17–22.
- SHAW, R., MARAGOS, C. & MAY, J.T., 1986. Bovine herpes mammillitis therapy. The Veterinary Record, 119, 436.
- SHEPPARD, M. & MAY, J.T., 1989. Location and characterization of the bovine herpesvirus type 2 thymidine kinase gene. Journal of General Virology, 70, 3067–3071.
- SKINNER, G.R., BUCHAN, A., DURHAM, J., COWAN, M., DAVIES, J., BROOKES, K. & CASTRUCCI, G., 1987. Role of bovine mammillitis virus towards preparation of an alternative vaccine against herpes simplex virus infections of human subjects. Vaccine, 5, 55–59.
- SMEE, D.F. & LEONHARDT, J.A., 1994. Vaccination against bovine herpes mammillitis virus infections in guinea pigs. Intervirology, 37, 20–24.
- STERZ, H., LUDWIG, H. & ROTT, R., 1973. Immunologic and genetic relationship between herpes simplex virus and bovine herpes mammillitis virus. Intervirology, 2, 1–13.
- TABBAA, D. & LIEBERMANN, H., 1989. Detection of antibodies to bovid herpesvirus 2 infection of cattle in Syria. Archiv für Experimentelle Veterinärmedizin, 43, 929–931.
- TURNER, A.J., KOVESDY, L. & MORGAN, I.R., 1976. Isolation and characterisation of bovine herpesvirus mammillitis virus and its pathogenicity for cattle. Australian Veterinary Journal, 52, 166–169.
- TURNER, A.J., KOVESDY, L., CIANTER, M.S., NICHOLLS, W.A. & CHATHAM, R.O., 1974. Isolation of bovine herpes mammillitis virus from dairy cattle in Victoria. Australian Veterinary Journal, 50, 578–579.
- TURNER, A.J., MORGAN, I.R., SYKES, W.E. & NICHOLLS, W.A., 1976. Bovine herpes mammillitis of dairy cattle in Victoria. Australian Veterinary Journal, 52, 170–173.
- WEAVER, L.D., DELLERS, R.W. & DARDIRI, A.H., 1972. Bovine herpes mammillitis in New York. Journal of the American Veterinary Medical Association, 160, 1643–1644.
- WEISS, K.E., 1963. Lumpy skin disease. Emerging Diseases of Animals. FAO Agricultural Studies No. 61, 179–201.
- WESTBURY, H.A., 1981. Infection of sheep and goats with bovid herpesvirus 2. Research in Veterinary Science, 31, 353–357.
- YEDLOUTSCHNIG, R.J., BREESE, S.S.J., HESS, W.R., DARDIRI, A.H., TAYLOR, W.D., BARNES, D.M., PAGE, R.W. & RUEBKE, H.J., 1971. Bovine herpes mammillitis-like disease diagnosed in the United States. Proceedings 74th Annual Meeting of the United States Animal Health Association 1970, 208–212.
- YEUNG, K.C., D’OFFAY, J. & OAKES, J.E., 1988. Genomic location of bovid herpesvirus type 2 nucleotide sequences homologous to five herpes simplex virus type 1 genes. Virus Research, 10, 369–380.