- Infectious Diseases of Livestock
- Part 2
- Equine infectious anaemia
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Equine infectious anaemia
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Equine infectious anaemia
Previous author: RF COOK AND CJ ISSEL
Current authors:
C LEROUX
Research Director, Viral Infections and Comparative Pathology, UMR 754 INRAE EPHE Lyon 1 University, 50 Avenue Tony Garnier, Lyon, 69007, France
JL CADORE
Professor in internal medicine, DVM, PhD, Viral Infections and Comparative Pathology, UMR 754 INRAE EPHE Lyon 1 University, 1 Avenue Bourgelat, Marcy-L'Etoile, 69280, France
GENERAL INTRODUCTION: RETROVIRIDAE
Introduction
Equine infectious anaemia (EIA) is a disease of domestic Equidae (horses, ponies, mules, donkeys) that is caused by equine infectious anaemia virus (EIAV), a retrovirus classified in the genus Lentivirus of the family Retroviridae. Like other retroviruses, EIAV is responsible for a chronic lifelong infection; most infected equids are inapparent carriers, able to control viral replication without developing overt signs of disease. Equine infectious anaemia has an almost worldwide distribution and is a significant threat to the equine industry, evidenced by its classification as one of the eleven notifiable equine diseases listed by the OIE (the World Organisation for Animal Health).
The disease was first reported in France in 184373 and its full description, first published in 1904 included the different stages of the disease and its transmissible nature by a “filterable agent”.115 For over five decades, research in EIA suffered from the lack of any in vitro culture system. Fortunately, during the late 1960s, Japanese investigators developed leukocyte culture systems that were able to propagate EIAV.63 The discovery of insect transmission of EIAV33, 39, 40, 54 and the development of the first diagnostic test22, 23 were key steps for the management of EIAV infection. The discovery of human immunodeficiency virus (HIV), an EIAV-related lentivirus, renewed interest in the equine virus.
Clinical signs of EIA are generally non-specific and vary considerably between individual animals and possibly even between equid species,26 making the differential diagnosis often complicated. Although equids remain infected with EIAV for life, most are able to control viral replication to such an extent they are free from overt signs of disease and are therefore classified as “inapparent carriers”.29, 30, 45 These "clinically invisible" infected equids contribute to unintended spread of the infection and disease. Since the first vaccine trial in Canada in 19455 and in Japan in the early 1970s, many vaccine strategies/approaches have been developed that all failed to efficiently protect horses against infection from heterologous strains of EIAV. In contrast, however, extensive immunization of equids in China with a live attenuated EIAV strain has had a reported beneficial effect by limiting disease progression in infected equids.116 In much of the world, EIA control still relies on the identification and removal or quarantine of infected animals; EIAV-infected animals are identified using serological surveillance.
Aetiology
The infectious nature of EIA has been known since 1904,115 which places it among the first diseases proven to be caused by a virus. After their initial report of the infectious nature of this disease present in the Meuse region of eastern France, Vallée and Carré described the culture of the virus by successive passage (unfortunately lost subsequently), the blood-borne transmission of EIAV infection, the susceptibility of donkeys to infection, the fact that anaemia was not the only expression of the disease (these investigators noted that “infectious anaemia” was not an appropriate designation for the disease as it did not convey correctly the complexity of the disease entity they observed), and the cyclic nature of the disease. They anticipated that prophylaxis would be complicated by the blood-borne nature of agent transmission, and the presence of horses infected without clinical expression of disease. With the discovery of the Rous sarcoma virus in chickens,98 during the twentieth century.
Members of the family Retroviridae are ancient viruses that probably emerged during the early Palaeozoic era2, 47 more than 450 million years ago, thus they are probably as old as their jawed vertebrate hosts.2 While retroviruses are present in a wide host range, from fish to humans, lentiviruses are limited to equids, cattle, small ruminants, wild and domestic Felidae, non-human primates and humans, suggesting they arose "only" one million years ago, thus relatively recently in evolutionary terms.120
Retroviruses are enveloped RNA viruses (80–100 nm in diameter). Their genomes (~8 to 11 kb) consist of a dimer of linear, single-stranded, positive-sense RNAs. Importantly, their replication is dependent on the reverse transcriptase (RT) enzyme, which is an RNA-dependent DNA polymerase that converts positive-sense viral RNA into DNA during the early stage of infection. Equine infectious anaemia virus shares morphological, antigenic and genetic features with small ruminant lentiviruses (SRLVs)67 (see Caprine arthritis-encephalitis and Visna-maedi) and HIV. This equine retrovirus (EIAV) has been described as the “country cousin” of HIV65 because, like its more infamous relative, it is classified in the genus Lentivirus (subfamily Orthoretrovirinae) within the family Retroviridae. Early description of HIV reported on the antigenic similarities between the gag proteins of the human LAV/HTLV-III virus and that of EIAV; indeed, it was shown that sera from EIAV infected horses would cross-react with and recognize the major core protein of HIV.41
The EIAV genome is approximately 8,200 base pairs (bp) in length, making EIAV the smallest and the least genetically complex lentivirus genome discovered to date with the prototypical retroviral genome organization in which the gag, pol and env genes are framed by the long terminal repeats (LTR). The proviral genome is defined as the host-DNA integrated virus genome, and serves as template for synthesis mRNAs encoding Tat, Rev and S2 regulatory proteins, gag and Env structural proteins, protease (PR), reverse transcriptase (RT), RNase H, dUTPase (DU) and integrase (IN) enzymes (Table 1, Figure 1).
Table 1. Structural and regulatory viral proteins of equine infectious anaemia virus
| Gene | Product | Size (kDa) | Roles |
|---|---|---|---|
| gag | Matrix (MA) | 15 | Binds to the phosphoinositides of the cell membrane and initiates lentiviral particle assembly |
| Capsid (CA) | 26 | Major component of the viral core and control packing | |
| Nucleocapsid (NC) | 11 | Binds to the viral RNA genomes | |
| Late domain (p9) | 9 | Essential for assembly and budding from the host cell through ESCRT-III (Endosomal Sorting Complexes Required for Transport-III) | |
| pol | Protease (PR) | 12 | Cleavage and processing of gag and Pol polyprotein precursors |
| Reverse transcriptase (RT) | 66 | DNA-polymerase RNA dependent that converts the single-stranded viral RNA genome into double-stranded DNA in the cytoplasm of the infected cell. Its RNAse H activity degrades viral RNA during dsDNA synthesis. The lack of proof-reading activity associated to the RT promotes accumulation of error during the replication process. | |
| dUTPase (DU) | 15 | Hydrolyzes dUTP to dUMP and inorganic phosphate to minimize the misincorporation of uracil into DNA. Essential for efficient replication in non-dividing cells such as macrophages in vivo | |
| Integrase (IN) | 30 | Promotes integration of viral cDNA into host cell DNA | |
| env | Surface unit (SU) | 90 | The SU and TM glycoproteins are produced by cleavage of the envelope polyprotein by cellular endoproteinases. They constitute the virus envelope in association with component of the cell membrane acquired during virus budding at the cell surface. SU specifically interacts with the EIAV cell receptor, namely ERL1, a member of the tumor necrosis factor receptor (TNFR) |
| Transmembrane (TM) | 45 | ||
| tat | Transactivator of transcription (Tat) | 8 | Regulates transcription of the viral genome after its integration into the host-cell DNA. Tat enhances the elongation efficiency of host RNA polymerase II |
| s2 | S2 | 7 | Unique among retroviruses. Not essential for in vitro replication but important determinant of virulence in vivo. Antagonize the inhibitory activity of SERINC (Serine Incorporator) viral restriction factor. |
| rev | Rev | 18 | Regulates expression by facilitating the nuclear export of incompletely spliced viral RNA |
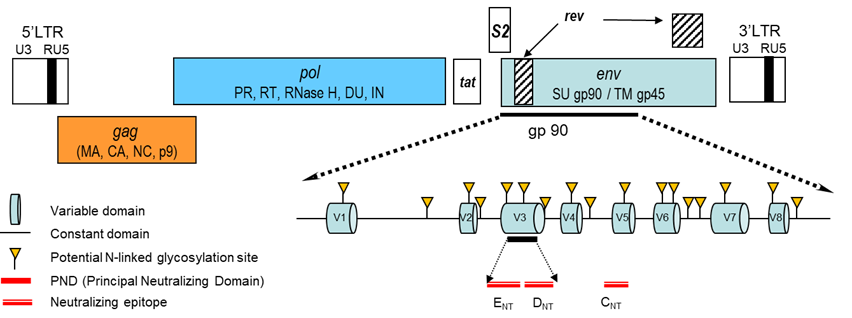
Figure 1. Genomic organization of the EIAV provirus genome. The gag and env genes encode the structural protein, the pol gene encodes the enzymatic activities and among them the RT (reverse transcriptase), converting the RNA genome into DNA before its integration into the host genome. The env gene encodes the surface SU and transmembrane TM glycoproteins. SU interacts with the cellular receptor of EIAV. It is highly glycosylated, constituted of eight variable regions (V1 to V8) and carries important neutralizing epitopes. LTR (long terminal region) are non-coding regions essential for viral replication.
The core protein p26 encoded by the gag gene and the surface (gp90) and transmembrane (gp45) glycoproteins encoded by the env gene are the three major proteins recognized by the immune system. The Tat and Rev proteins are common to all retroviruses, but the S2 small protein is unique to EIAV. While S2 is not required for in vitro viral replication, the protein encoded by this highly conserved open reading frame is required for viral replication and virulence in vivo.38, 70, 71, 102 S2 enhances proinflammatory cytokine and chemokine responses in macrophages infected in vitro and acts as an infectivity factor, counteracting serine incorporator (SERINC), a cellular restriction factor that blocks virus entry into cells.1, 20
The SU glycoproteins are responsible for the attachment of retroviruses to the cell membrane, a key element for viral replication and tropism. Equine lentivirus receptor (ELR1) is the main EIAV receptor and a member of the tumour necrosis (TNF) receptor family. Virus entry occurs through a low-pH-dependent endocytic pathway.13, 14, 15, 57, 111, 123, 124
Epidemiology
Equine infectious anaemia virus is restricted to members of the Equidae and cases of infection have been described in horses, ponies, mules and donkeys.26, 56, 90, 109, 110 Equine infectious anaemia occurs worldwide, with outbreaks being reported in Asia, Europe, North America and South America.9, 12, 17, 18, 19, 32, 36, 87, 94, 107 (Figure 2). Recent surveys did not find evidence EIAV infection in Eastern and Central Saudi Arabia or the West Indies.3, 9
Equine infectious anaemia virus is generally considered to be a "blood-borne" virus, but viral RNAs have been reported in nasal, buccal and genital swabs.94 Transmission of EIAV occurs through transfer of blood-by-blood-feeding insects such as horse flies (Tabanids), deer flies (Chrysops), and stable flies (Stomoxys calcitrans).25, 53 In natural conditions, haematophagous insects mediate transmission of EIAV by mechanically transferring blood between equids in close proximity.33, 40, 54, 55, 58 These flies need only one blood meal to complete their life cycle; therefore, when interrupted, the flies must bite a second host to feed to repletion, when they also potentially transmit the virus through residual blood on their mouthparts. The volume of blood retained in the mouthparts is very small (~10 nl) and EIAV survives only approximately 30 minutes on the mouthparts, thus the risk of mechanical transmission will depend on the amount of EIAV in the blood (which is highest during febrile episodes)54, 55 and the distance separating the equids.40 In most cases, horseflies will return to feed on the animal they just left if others are not present less than 45 metres away.54 Obviously, therefore, the mechanical transmission of EIAV requires that naïve animals be in close proximity to an acutely infected equid and a quarantine distance of 180 metres (four times 45 metres) is generally considered safe to prevent EIAV transmission. Given these realties, biting flies are quite inefficient vectors of EIAV.
In a global context, humans are probably the most successful vector of EIAV. For example, outbreaks of EIA in Ireland and Italy occurred after administration of contaminated horse blood products to thoroughbred foals.32 The re-use of needles or other unsterilized equipment continues in many countries. Importantly, amounts of residual blood volumes retained on contaminated needles is much higher than that in the mouthparts of haematophagous insects, thus EIAV may persist up to 3 days on their surface.121 Studies on working horses in Brazil, a country with high prevalence of EIAV infection, demonstrated that practices such as improper ranch management conducive to the exposure of naive equids to contaminated veterinary or riding materials were important risk factors.7, 12 Interestingly, ranches that share working horses or riding gear, those that did not require EIA tests before introducing equids, and those located in flooded areas were all positively associated with outbreaks of EIAV infection.7
Although the blood-borne transmission of EIAV is recognized, it is probably not highly effective; therefore, other routes of EIAV transmission must be considered. The expression of the viral p26 capsid protein in alveolar and bronchiolar epithelial cells of the lung parenchyma has been reported in association with an interstitial lung disease.10 Viral excretion into the air spaces potentially could result in the exhalation of EIAV leading to horizontal respiratory transmission. This suggests the hypothesis of EIAV transmission through the respiratory route as for small ruminant lentiviruses in domestic sheep and goats,93 a possibility that might be explored by keeping naive and infected horses in close contact.
In a well-documented outbreak in Ireland in 2007, aerosol infection of EIAV-free horses was implicated from the index case.85, 86 It was proposed that the virus may have been aerosolized from the infected animal during fatal haemorrhage associated with the release of several litres of EIAV-infected blood, or possibly during high pressure cleaning of the stable. Anecdotally, EIAV transmission by venereal, embryo transfer, transovarial or perinatal exposure have been suggested but never proven. 42
Pathogenesis
The disease induced by EIAV infection is characterized by recurrent febrile episodes with concomitant marked thrombocytopenia, as clearly described in experimentally infected equids. In the absence of daily monitoring of these two parameters, as frequently occurs following natural EIAV infection, the disease may be missed. Infected equids may develop splenomegaly, glomerulonephritis, a “wasting syndrome”, as well as haemorrhages. Unlike HIV, EIAV does not infect lymphocytes nor induces immune deficiency.105 Blood monocytes are permissive to EIAV, but the differentiation of monocyte to mature macrophages is required for virus replication78, 88, 105 resulting in the active replication of EIAV in macrophage-rich tissues such as spleen and liver.48 High throughput proteomic analysis has shown that EIAV infection of monocyte-derived macrophages alters several cellular processes, especially those involved in oxidative phosphorylation, protein folding, RNA splicing, or ubiquitylation.37 Infection and virus expression have been reported in epithelial cells of the lungs in association with lung disease10 (Figure 3) and in endothelial cells.79, 89
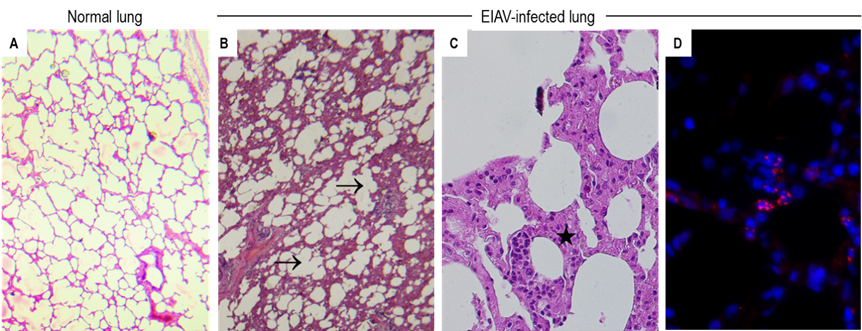
Figure 3. Lung lesions associated with EIAV infection. A. Normal lung parenchyma in a non-infected horse. B-C. Histological lesions in the lung of an EIAV-positive horse with thickening (★) or destruction (→) of the alveolar walls. D. Expression of the capsid protein (red immunofluorescent dot) in alveolar epithelial cells. Nuclei are stained with DAPI (blue)
Clinical profiles range from subclinical infection to high fever and thrombocytopenia in horses experimentally infected with the same dose of a well-characterized viral strain.24 In animals experiencing clinical signs, each febrile episode is associated with high-titre (~5.108 molecules per ml of plasma) viraemia.26, 48, 70, 72 The viral burden in tissues, especially the spleen, is highest during febrile episodes and then drastically declines with concomitant resolution of clinical signs.48 Interestingly, clinical signs develop when viral burden reaches a "pathogenic threshold", corresponding to the amount of virus necessary to switch from a subclinical to a clinically affected equid. Therefore, to be pathogenic, an EIAV strain must have a high replicative potential within its host. Following experimental infections with the same dose and strain of EIAV, it has been demonstrated that donkeys remain disease-free and control virus replication, with a virus load at least 100-fold lower as compared to those in horses.26 Passage of virulent pathogenic EIAV in donkeys and donkey cells results in its attenuation.118 Natural EIAV infections have been reported in donkeys, especially in Brazil where there are substantial populations of E. asinus.90 Although donkeys are resistant to EIAV-induced disease they may serve as reservoir hosts that contribute to dissemination of EIAV.
Thrombocytopenia is the earliest and most consistent abnormality observed in febrile animals. Despite a remarkable decrease of platelet count during the acute febrile episode, EIAV does not replicate in bone marrow megakaryocytes,21, 31 suggesting that viral infection is not directly responsible for the thrombocytopenia. Immune -mediated platelet destruction has been suggested based on increased levels of platelet-bound IgG and IgM, as well as the splenomegaly and hepatomegaly observed at necropsy in acutely-infected horses.21, 119 However, the similar levels of platelet drop in immunocompetent or in severe combined immunodeficient (SCID) horses, lacking both functional T and B-lymphocytes, suggest that immune-mediated thrombocytopenia is not a major mechanism leading to the severe platelet drop and that the shortening of platelet life span contributes moderately to the platelet drop. 31 In EIAV-infected immuno-competent as well as in SCID horses, the number of megakaryocytes in the bone marrow is not significantly altered.31 Parameters of intravascular coagulation remain normal until after the platelet number has dropped; strongly suggesting that intravascular coagulation is not a key factor responsible for EIAV-associated thrombocytopenia.31 Taken together, these results suggest that EIAV induces thrombocytopenia through an indirect, noncytocidal effect on platelet production. Platelet activation occurs during acute viral infections, leading to their degranulation, aggregation and removal from the circulation. Although production of platelets from bone marrow is not impaired in acutely EIAV-infected equids, increased fibrinogen bound to platelets associated with ultrastructural changes is consistent with their activation. In vivo activation of platelets might contribute to their removal from the circulation.99
The infection of macrophages by EIAV is responsible for a cytokine storm that contributes to the disease. Tumour necrosis factor-alpha (TNF-megakaryocytic), transforming growth factor-beta (TGF-ß) and interferon-alpha (IFN-megakaryocytic) are negative regulators of platelet production. Increased serum levels of TNF-megakaryocytic, TGF-ß and IFN-megakaryocytic have been shown in EIAV-infected foals days prior to thrombocytopenia.114 In vitro, TNF-megakaryocytic and TGF-ß significantly reduce the number of megakaryocytes colonies. Plasma taken immediately before clinical thrombocytopenia from EIAV-infected foals also reduces the number of megakaryocytic colonies in vitro and this effect can be partly reversed by neutralizing TNF megakaryocytic and TGF-ß with specific antibodies. 113 Importantly, a TNF receptor family protein serves as a cellular receptor for EIAV.123 Tumour necrosis factor-alpha directly inhibits erythropoiesis and plays a role in pro-inflammatory cytokine-mediated anaemia.84 Induction of cytokines has been shown to be correlated to the virulence of individual strains of EIAV; whereas a virulent EIAV strain significantly increased expression of IL1 megakaryocytic, IL-1ß, IL-6, IL-10, and TNF megakaryocytic in vitro in monocyte-derived macrophages, an avirulent virus failed to induce these cytokines.74, 76 The accessory protein S2 enhances expression of pro-inflammatory cytokines such as IL-1 megakaryocytic, IL-1ß, IL-8, MCP-2, MIP-1ß, IP-10 or IL-34 in EIAV-infected horses, as shown by differential cytokine expression in horses infected with a wild-type or with a S2-deleted EIAV.28
Although anaemia may occur in the initial phases of EIA, the mechanism is uncertain and both intravascular as well as extravascular haemolysis likely contribute. In early studies, it was shown that EIAV was able to haemagglutinate horse erythrocytes in vitro and that haemagglutination was inhibited by sera from horses previously infected with an homologous virus strain.106 Complement (C3)-coated erythrocytes have been shown in experimentally infected horses in all the disease stages. These complement-coated red blood cells were osmotically fragile and had a shorter life span (18-90 days) as compared to normal ones (~136 days).80
Glomerulitis characterized by mesangial and epithelial cell proliferation and glomerular membrane thickening is present in ~75 per cent of EIAV-infected horses,104 presumably due to immune complex deposition and potentially other mechanisms. Small granular deposits of IgG and C3 complement have been observed along the glomerular membrane.6
Oxidative stress, defined as an imbalance between antioxidants and oxidants upon exposure of the organism to adverse stimuli, may also be significant during EIAV infection and may contribute to tissue damage and induce inflammatory responses and possibly other adverse effects. For example, it has been showed in a large cohort of working horses that EIAV infection alters glutathione peroxidase and uric acid levels and thus modifies the oxidant/antioxidant equilibrium, especially in recently infected horses.11
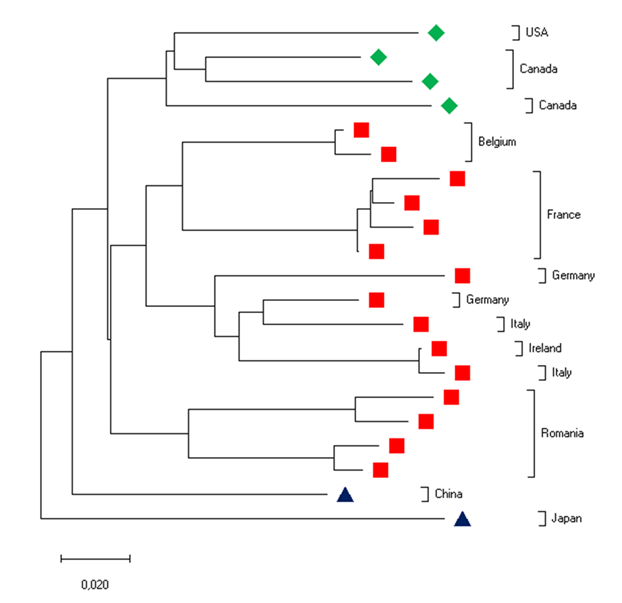
As is the case with other retroviruses, the replication of EIAV leads to genomic mutations because of the lack of proofreading activity of its reverse transcriptase.4, 97 Furthermore, the initial step of replication occurs in the cytoplasm, where host-cell proofreading activities are absent. Accumulation of nucleotide mutations results in the generation of a "viral quasispecies" or "viral swarm", defined as the co-existence of complex populations of related but distinct genomes.34, 50 This feature of EIAV confers on it a selective advantage by allowing a rapid response to any environmental selection pressure, including that exerted by the host immune response.29, 30, 68 Importantly, each febrile episode experienced by an EIAV-infected animal is associated with a new predominant virus (member of the quasispecies) with novel genomic and antigenic properties.60, 62, 68, 83, 91, 126 (Figure 4) During the course of infection, non-synonymous mutations leading to amino acid changes are not randomly distributed but accumulate in eight (V1 to V8) hypervariable domains within the surface (SU) glycoproteins.68, 92, 126 (Figure 1). The evolution of the quasispecies continues even during the asymptomatic phase, and in the absence of febrile episodes.66 (Figure 5)
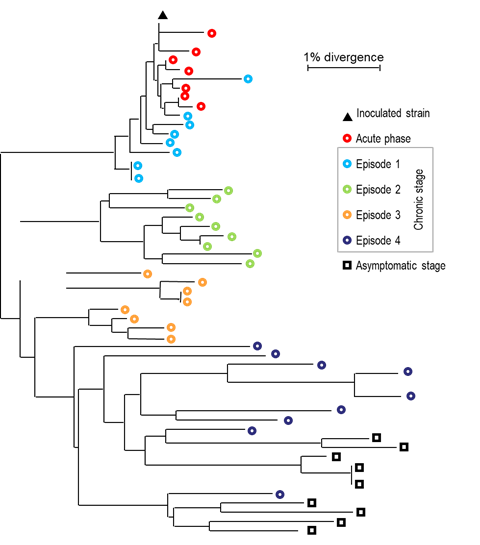
Figure 5. Evolution of the EIAV "quasispecies" during the course of the infection (adapted from (67)). The viral populations have been analyzed during the acute, chronic and asymptomatic phases and compared to the inoculated viral strain. The phylogenetic tree has been reconstructed using the Neighbor -joining method from the env sequences.
In long-term inapparent carriers, the original EIAV species that was associated with the initial infection is undetected, having been eliminated by the immune response. Nevertheless, viral persistence is maintained during the course of the disease by relentless evolution of virus populations with novel envelope proteins that emerge from the tissue reservoirs.29, 30 Interestingly, a mathematical model based on hidden Markov models has shown that constant and variable regions of EIAV, as well as of HIV-1, SRLVs, simian immunodeficiency virus (SIV) and bovine immunodeficiency virus (BIV), can be accurately predicted in silico and have a specific statistical oligonucleotide composition.8
Clinical signs
The diagnosis of EIA is often complicated because of the lack of pathognomonic clinical signs. Importantly, as with other retroviruses, infection does not necessarily correlate to disease expression and infected animals may remain subclinical for their entire life. The vast majority of EIAV-seropositive equids identified during serological surveillance do not have overt clinical signs and infection may have been ongoing for decades prior to the serological diagnosis.
Equine infectious anaemia virus is unique among lentiviruses in that it causes a dynamic and defined course of infection and recurring disease. Interestingly, descriptions of the disease in use today are virtually identical to the one presented by Carré and Vallée in 1904 in their report to the "Académie des Sciences".115 They described a tri-phasic disease characterized by an "acute phase" 3-4 weeks after infection with constant hyperthermia, weight loss, oedema, mild conjunctivitis, and heart and locomotor disorders; a "subacute phase" that evolves over ~ 2 months with the same signs as the initial phase but less severe; and finally the "chronic phase" characterized by recurring febrile peaks found to be associated with signs of laziness. The sequences of clinical events, establishment of immune responses and virus evolution are all now well documented.45, 48, 66, 88, 104 Experimental infections with defined strains helped to clearly distinguish the acute, chronic and asymptomatic phases of infection by following the evolution of the associated body temperature and platelet counts (Figure 4). While most animals recover, some highly virulent strains may induce a severe and fatal disease with unresolved high fever and haemorrhages. Recrudescence of clinical disease may occur upon stress or may be induced by immunosuppressive treatment such as dexamethasone in horses as well as in mules.29, 61
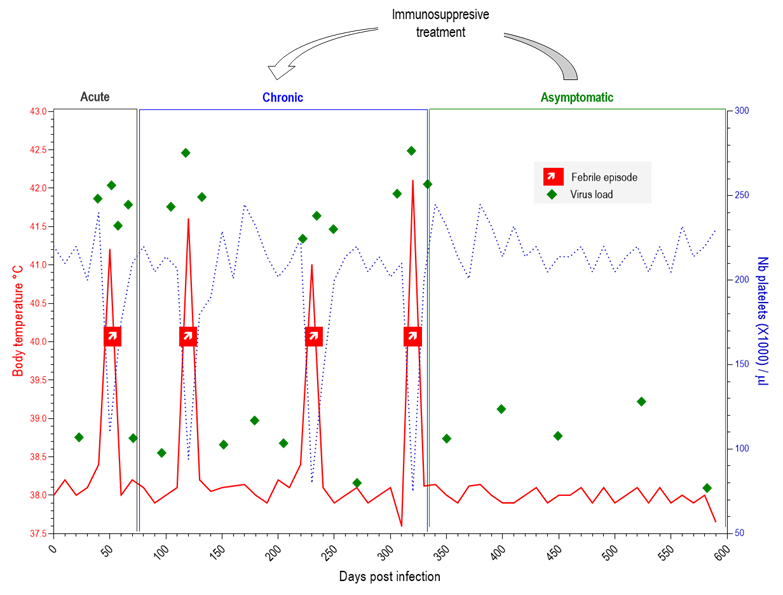
Figure 4. Clinical profile in an EIAV-infected horse. Febrile episodes (↗) are characterized by peaks (>39°C) of body temperature (red line) concomitantly with severe platelet drop (dashed blue line). Along infection (in days), the infected horse evolves from the acute "flu-like" phase weeks after the infection, to the chronic phase characterized by recurrence of febrile episodes. Eventually the horse enters the asymptomatic phase where while infected it shows no clinical signs.
Pathology
Relatively few studies (mostly dating back to the 1970s) have described the pathology of EIAV infection of horses. Acutely infected horses may exhibit marked splenomegaly, lymphadenopathy, accentuated hepatic lobular structure, anaemia, widespread haemorrhages, oedema and emaciation.104 Microscopic lesions include generalized lymphoid necrosis, perivascular infiltration of lymphocytes in the liver, lymph nodes, adrenal gland, spleen, meninges or lung.51, 104 Fatty degeneration and necrosis have been reported in the liver.59 Increased vascular permeability with associated haemorrhages and oedema is described,59 as also are haemorrhages attributed to severe thrombocytopenia associated with febrile episodes.21
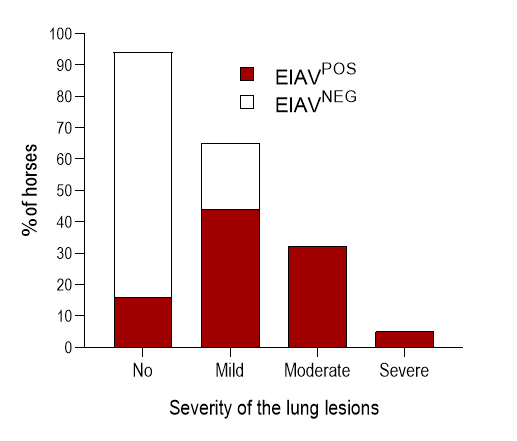
Pulmonary lesions include lymphocyte infiltration, bronchiolitis, and thickening of alveolar septae.10 (Figure 3 & Figure 6) Interestingly, these lesions resemble those associated with lymphoid interstitial pneumonia (LIP), reported in HIV-1 infected patients,108 feline immunodeficiency virus (FIV)-infected cats16 or SRLV-infected sheep.64 Based on a score previously established for lung lesions associated with SRLV infection in sheep,49 over 80 per cent of the EIAV-positive horses displayed mild to severe lesions characterized by inflammation around bronchioles and alveolar wall, changes in the thickness of the alveolar septae, epithelial bronchiolar hyperplasia, destruction of the bronchiolar epithelium and accumulation of smooth muscle cells within the pulmonary parenchyma. 10 (Figure 6) Equine infectious anaemia virus expression in alveolar and bronchiolar epithelial cells confirms virus tropism to non-mononuclear cells.10 Interestingly similar lesions have been observed in horses infected with the EIAVLN40 virulent strain but not with the EIAVDLV121 vaccine strain.76
Diagnosis
The lack of pathognomonic clinical signs and absence of clinical signs in most infected equids preclude consistent diagnosis of EIA solely based on clinical features. Thus, the identification of EIAV-infected animals is reliant on detection of antibodies directed against viral proteins. Equine infectious anaemia is one of the eleven equine notifiable diseases listed by OIE, and laboratory diagnostic testing is required to determine the infection status of individual equids or populations and to monitor the effectiveness of eradication or control policies. The gold standard for laboratory diagnosis of EIAV infection of equids is the Coggins Test, which is an agar gel immunodiffusion (AGID) test using the major core protein p26 as antigen. Developed by Coggins and colleagues in the early 1970's,22, 23 the Coggins test has received international acceptance and is recommended by OIE for the management of EIA. Although this test has been invaluable in helping to control EIAV infection worldwide, it may lack sensitivity and is prone to false-negative reactions. The interpretation of AGID results is often subjective with weakly-positive sera and inter-laboratory testing may produce discordant results. 56, 103 Several ELISA-based diagnostic tests have been developed, approved and used in North America or Europe. Furthermore, countries such as Italy and Romania have implemented extensive surveillance plans using these assays.9, 101 A three-tiered strategy has been proposed based on the results of the National Surveillance Program in Italy, 56, 103 which is an effort to address false-negative or -positive serological results; briefly, this strategy proposes to screen sera using EIAV-specific ELISA and to confirm positive results using the gold standard AGID test; any discordance between the two tests is resolved using immunoblot analysis with EIAV antigens. This strategy, although cumbersome, has proven to be very accurate and effective.56, 103
Although serological tests are valuable as screening diagnostic assays, they are an indirect detection of infection. In experimentally infected equids, EIAV-specific humoral responses are first detected 14 to 28 days post-infection using sensitive immunoblot or ELISA assays.44, 100 The delay between the exposure to the virus and the detectable humoral response, known as the immunological window, makes serological testing unable to detect newly infected animals. Recently infected equids may pose a serious threat because that is a period of high-level virus replication. Furthermore, serologically silent occult EIAV infections have been reported in Argentina; although no EIAV-specific antibodies were detected in some horses, the animals were infected as evidenced by the molecular detection of the EIAV genome.96 This suggests that not all animals exposed to EIAV eventually develop a detectable humoral response. Therefore the detection of recently infected animals is of importance and cannot be done using serological tests. To date, laboratory tests to detect EIAV particles or viral components are tedious, time-consuming and not applicable to the diagnosis of large populations. Several polymerase chain reaction (PCR) procedures have been reported for the detection of viral nucleic acids (or provirus).18, 35, 87, 94 The remarkable genetic variability of EIAV complicates the design of sensitive and specific PCR assays, as does the fact that viral loads are often very low in infected individuals. The increasing sequence data base of EIAV strains should assist in the creation of improved PCR diagnostic assays.
Differential diagnosis
The acute form of EIA can be confused with the early stages of many other conditions. For example, the haemolytic anaemia that occurs in some horses during acute EIAV infection resembles those observed in horses with Babesia caballi or Theileria equi infections. The differential diagnosis requires serological confirmation in convalescent samples 10 to 14 days after the febrile episode, as the initial sample will be negative for antibodies to EIAV. During the chronic form of the disease, a variety of other conditions associated with anaemia must be considered. To avoid any misdiagnosis, serological testing must be performed in any equid with clinical signs suggestive of EIA.
Control
In most countries, the Coggins AGID test remains the gold standard for establishing the EIAV-infection status of equids. But, as discussed earlier, it may produce a significant number of false negative results, mainly due to the large amount of antigen/antibody complexes necessary to form the visible lines of precipitation that are required to identify a positive serum sample.56 Thus, the development of a sensitive and specific ELISA is a potential improvement for systematic EIAV control programmes, such as that developed in Italy.56, 101, 103 Worldwide, the control of EIAV infection is not reliant on any consensual surveillance plan but on nationally defined strategies. Managing the risks inherent to international movement of equids and equid-derived products therefore remains challenging.52 Several outbreaks, notably in Europe, have been traced back to the introduction and use of untested horse products. Euthanasia of EIAV-seropositive animals is required in many countries; although this effectively prevents virus spread, it may not be an acceptable practice in countries where horses are essential for agriculture and when most equids are working animals (including mules and donkeys that may serve as reservoir hosts).
Since the first vaccine trial in Canada in 1945,5 and subsequently in Japan in the early 1970s, many studies have been conducted to develop EIA vaccines including strategies based on live-attenuated viruses, DNA vaccines, recombinant viral proteins, genetically attenuated virus strains, and particulate viral proteins.43, 46, 69, 75, 77, 81, 82, 95, 112, 122, 125 To date, however, widespread vaccination of equids for EIA is not practiced globally other than in China, where a live-attenuated strain of EIAV is widely used for protective immunization of horses.116 Most of the vaccines developed to date have proven unable to induce sterilizing immunity against challenge infection with heterologous viral challenges and, disconcertingly, many of them have resulted in exacerbation of clinical signs in immunized equids.27, 81, 95, 117 Therefore, no commercial vaccine is widely available to prevent EIA (other than in China), and control relies on the identification and removal or quarantine of infected animals.
Future development of an effective EIA vaccine is complicated by the remarkable genetic variability of EIAV, which results in an enormous variety of individual viral strains with potentially distinct biological properties. Between 1974 and 1980, Chinese scientists conducted a massive vaccination program in which over 60 million horses and donkeys were inoculated with a live attenuated virus produced by serial passage of the highly pathogenic EIAV Liaoning strain (EIAVLN) in donkeys and in donkey leukocyte cultures.118 This vaccine induces partial protection against experimental challenge with heterologous EIAV strains75, 125 and has apparently protected the Chinese equine population against EIA. However, it is not possible currently to differentiate naturally infected equids from those that receive this Chinese vaccine (DIVA), precluding the trade of vaccinated equids outside China.
In conclusion, although EIAV was discovered at the beginning of the twentieth century, its control remains a challenge and its continued spread remains a significant threat to equine populations worldwide. Thanks to the productive efforts of a few research teams working on this equine retrovirus in the last five decades, much has been done to clarify the interactions between the virus and its host. Furthermore, studies of EIAV have also contributed to a better understanding of related viruses such as HIV. Protection of the global equid community in the future will require the development of improved diagnostic tools and strategies to quickly and specifically identify newly infected horses. Epidemiological studies will be needed to identify all potential routes of virus transmission, and these studies should not ignore the role of reservoir hosts.
References
- AHMAD, I., LI, S., LI, R., CHAI, Q., ZHANG, L., WANG, B., YU, C. & ZHENG, Y. H., 2019. The retroviral accessory proteins S2, Nef, and glycoMA use similar mechanisms for antagonizing the host restriction factor SERINC5. Journal of Biological Chemistry. doi:10.1074/jbc.RA119.007662.
- AIEWSAKUN, P. & KATZOURAKIS, A., 2017. Marine origin of retroviruses in the early Palaeozoic Era. Nature Communications, 8, 13954.
- ALNAEEM, A. A. & HEMIDA, M. G., 2019. Surveillance of the equine infectious anemia virus in Eastern and Central Saudi Arabia during 2014-2016. Veterinary World, 12, 719-723.
- BAKHANASHVILI, M. & HIZI, A., 1993. Fidelity of DNA synthesis exhibited in vitro by the reverse transcriptase of the lentivirus equine infectious anemia virus. Biochemistry, 32, 7559-7567.
- BANKIER, J. C., 1945. Equine Infectious Anaemia: Attempted Vaccination with Crystal Violet Tissue Vaccine. Canadian Journal of Comparative Medicine and Veterinary Science, 9, 197-199.
- BANKS, K. L., HENSON, J. B. & MCGUIRE, T. C., 1972. Immunologically mediated glomerulitis of horses. I. Pathogenesis in persistent infection by equine infectious anemia virus. Laboratory Investigation, 26, 701-707.
- BARROS, M. L., BORGES, A. M. C., OLIVEIRA DE, A. C. S., LACERDA, W., O SOUZA DE, A. & AGUIAR, D. M., 2018. Spatial distribution and risk factors for equine infectious anaemia in the state of Mato Grosso, Brazil. Revue scientifique et technique, 37, 971-983.
- BOISSIN-QUILLON, A., PIAU, D. & LE ROUX, C., 2007. In silico segmentations of lentivirus envelope sequences. BMC Bioinformatics, 8, 99.
- BOLFA, P., BARBUCEANU, F., LEAU, S. E. & LE ROUX, C., 2016. Equine infectious anaemia in Europe: Time to re-examine the efficacy of monitoring and control protocols? Equine Veterinary Journal, 48, 140-142.
- BOLFA, P., NOLF, M., CADORE, J. L., CATOI, C., ARCHER, F., DOLMAZON, C., MORNEX, J. F. & LE ROUX, C., 2013. Interstitial lung disease associated with Equine Infectious Anemia Virus infection in horses. Veterinary Research, 44, 113.
- BOLFA, P. F., LE ROUX, C., PINTEA, A., ANDREI, S., CATOI, C., TAULESCU, M., TABARAN, F. & SPINU, M., 2012. Oxidant-antioxidant imbalance in horses infected with equine infectious anaemia virus. Veterinary Journal, 192, 449-454.
- BORGES, A. M., SILVA, L. G., NOGUEIRA, M. F., OLIVEIRA, A. C., SEGRI, N. J., FERREIRA, F., WITTER, R. & AGUIAR, D. M., 2013. Prevalence and risk factors for Equine Infectious Anemia in Pocone municipality, northern Brazilian Pantanal. Veterinary Science, 95, 76-81.
- BRINDLEY, M. A. & MAURY, W., 2005. Endocytosis and a low-pH step are required for productive entry of equine infectious anemia virus. Journal of Virology, 79, 14482-14488.
- BRINDLEY, M. A. & MAURY, W., 2008. Equine infectious anemia virus entry occurs through clathrin-mediated endocytosis. Journal of Virology, 82, 1628-1637.
- BRINDLEY, M. A., ZHANG, B., MONTELARO, R. C. & MAURY, W., 2008. An equine infectious anemia virus variant superinfects cells through novel receptor interactions. Journal of Virology, 82, 9425-9432.
- CADORE, J. L., STEINER-LAURENT, S., GREENLAND, T., MORNEX, J. F. & LOIRE, R., 1997. Interstitial lung disease in feline immunodeficiency virus (FIV) infected cats. Research in Veterinary Science, 62, 287-288.
- CAIJ, A. B. & TIGNON, M., 2014. Epidemiology and genetic characterization of equine infectious anaemia virus strains isolated in Belgium in 2010. Transboundary and Emerging Diseases, 61, 464-468.
- CAPOMACCIO, S., WILLAND, Z. A., COOK, S. J., ISSEL, C. J., SANTOS, E. M., REIS, J. K. & COOK, R. F., 2012. Detection, molecular characterization and phylogenetic analysis of full-length equine infectious anemia (EIAV) gag genes isolated from Shackleford Banks wild horses. Veterinary Microbiology, 157, 320-332.
- CAPPELLI, K., CAPOMACCIO, S., COOK, F. R., FELICETTI, M., MARENZONI, M. L., COPPOLA, G., VERINI-SUPPLIZI, A., COLETTI, M. & PASSAMONTI, F., 2011. Molecular detection, epidemiology, and genetic characterization of novel European field isolates of equine infectious anemia virus. Journal of Clinical Microbiology, 49, 27-33.
- CHANDE, A., CUCCURULLO, E. C., ROSA, A., ZIGLIO, S., CARPENTER, S. & PIZZATO, M., 2016. S2 from equine infectious anemia virus is an infectivity factor which counteracts the retroviral inhibitors SERINC5 and SERINC3. Proceedings of the National Academy of Sciences of the United States of America, 113, 13197-13202.
- CLABOUGH, D. L., GEBHARD, D., FLAHERTY, M. T., WHETTER, L. E., PERRY, S. T., COGGINS, L. & FULLER, F. J., 1991. Immune-mediated thrombocytopenia in horses infected with equine infectious anemia virus. Journal of Virology, 65, 6242-6251.
- COGGINS, L. & NORCROSS, N. L., 1970. Immunodiffusion reaction in equine infectious anemia. Cornell Veterinarian, 60, 330.
- COGGINS, L., NORCROSS, N. L. & NUSBAUM, S. R., 1972. Diagnosis of equine infectious anemia by immunodiffusion test. American Journal of Veterinary Research, 33, 11-18.
- COOK, R. F., COOK, S. J., BERGER, S. L., LEROUX, C., GHABRIAL, N. N., GANTZ, M., BOLIN, P. S., MOUSEL, M. R., MONTELARO, R. C. & ISSEL, C. J., 2003. Enhancement of equine infectious anemia virus virulence by identification and removal of suboptimal nucleotides. Virology, 313, 588-603.
- COOK, R. F., LE ROUX, C. & ISSEL, C. J., 2013. Equine infectious anemia and Equine Infectious Anemia Virus 2013: A Review. Veterinary Microbiology, 167, 181-204.
- COOK, S. J., COOK, R. F., MONTELARO, R. C. & ISSEL, C. J., 2001. Differential responses of Equus caballus and Equus asinus to infection with two pathogenic strains of equine infectious anemia virus. Veterinary Microbiology, 79, 93-109.
- COSTA, L. R., SANTOS, I. K., ISSEL, C. J. & MONTELARO, R. C., 1997. Tumor necrosis factor-alpha production and disease severity after immunization with enriched major core protein (p26) and/or infection with equine infectious anemia virus. Veterinary Immunology and Immunopathology, 57, 33-47.
- COVALEDA, L., FULLER, F. J. & PAYNE, S. L., 2010. EIAV S2 enhances pro-inflammatory cytokine and chemokine response in infected macrophages. Virology, 397, 217-223.
- CRAIGO, J. K., LEROUX, C., HOWE, L., STECKBECK, J. D., COOK, S. J., ISSEL, C. J. & MONTELARO, R. C., 2002. Transient immune suppression of inapparent carriers infected with a principal neutralizing domain-deficient equine infectious anaemia virus induces neutralizing antibodies and lowers steady-state virus replication. Journal of General Virology, 83, 1353-1359.
- CRAIGO, J. K., STURGEON, T. J., COOK, S. J., ISSEL, C. J., LEROUX, C. & MONTELARO, R. C., 2006. Apparent elimination of EIAV ancestral species in a long-term inapparent carrier. Virology, 344, 340-353.
- CRAWFORD, T. B., WARDROP, K. J., TORNQUIST, S. J., REILICH, E., MEYERS, K. M. & MCGUIRE, T. C., 1996. A primary production deficit in the thrombocytopenia of equine infectious anemia. Journal of Virology, 70, 7842-7850.
- CULLINANE, A., QUINLIVAN, M., NELLY, M., PATTERSON, H., KENNA, R., GARVEY, M., GILDEA, S., LYONS, P., FLYNN, M., GALVIN, P., NEYLON, M. & JANKOWSKA, K., 2007. Diagnosis of equine infectious anaemia during the 2006 outbreak in Ireland. Veterinary Record, 161, 647-652.
- CUPP, E. W. & KEMEN, M. J., 1980. The role of stable flies and mosquitoes in the transmission of equine infectious anemia virus. Proceedings, Annual Meeting of the United States Animal Health Association, 84, 362-367.
- DOMINGO, E., SHELDON, J. & PERALES, C., 2012. Viral quasispecies evolution. Microbiology and Molecular Biology Reviews, 76, 159-216.
- DONG, J. B., ZHU, W., COOK, F. R., GOTO, Y., HORII, Y. & HAGA, T., 2012. Development of a nested PCR assay to detect equine infectious anemia proviral DNA from peripheral blood of naturally infected horses. Archives of Virology. doi:10.1007/s00705-012-1406-8.
- DOREY-ROBINSON, D. L. W., LOCKER, N., STEINBACH, F. & CHOUDHURY, B., 2019. Molecular characterization of equine infectious anaemia virus strains detected in England in 2010 and 2012. Transboundary and Emerging Diseases. doi:10.1111/tbed.13286.
- DU, C., MA, J., LIU, Q., LI, Y. F., HE, X. J., LIN, Y. Z., WANG, X. F., MENG, Q. W., WANG, X. & ZHOU, J. H., 2015. Mice transgenic for equine cyclin T1 and ELR1 are susceptible to equine infectious anemia virus infection. Retrovirology, 12, 36.
- FAGERNESS, A. J., FLAHERTY, M. T., PERRY, S. T., JIA, B., PAYNE, S. L. & FULLER, F. J., 2006. The S2 accessory gene of equine infectious anemia virus is essential for expression of disease in ponies. Virology, 349, 22-30.
- FOIL, L. D., ADAMS, W. V., MCMANUS, J. M. & ISSEL, C. J., 1987. Bloodmeal residues on mouthparts of Tabanus fuscicostatus (Diptera: Tabanidae) and the potential for mechanical transmission of pathogens. Journal of Medical Entomology, 24, 613-616.
- FOIL, L. D., MEEK, C. L., ADAMS, W. V. & ISSEL, C. J., 1983. Mechanical transmission of equine infectious anemia virus by deer flies (Chrysops flavidus) and stable flies (Stomoxys calcitrans). American Journal of Veterinary Research, 44, 155-156.
- GOUDSMIT, J., HOUWERS, D. J., SMIT, L. & NAUTA, I. M., 1986. LAV/HTLV-III gag gene product p24 shares antigenic determinants with equine infectious anemia virus but not with visna virus or caprine arthritis encephalitis virus. Intervirology, 26, 169-173.
- GREGG, K. & POLEJAEVA, I., 2009. Risk of equine infectious anemia virus disease transmission through in vitro embryo production using somatic cell nuclear transfer. Theriogenology, 72, 289-299.
- HAMMOND, S. A., COOK, S. J., FALO, L. D. J., ISSEL, C. J. & MONTELARO, R. C., 1999. A particulate viral protein vaccine reduces viral load and delays progression to disease in immunized ponies challenged with equine infectious anemia virus. Virology, 254, 37-49.
- HAMMOND, S. A., COOK, S. J., LICHTENSTEIN, D. L., ISSEL, C. J. & MONTELARO, R. C., 1997. Maturation of the cellular and humoral immune responses to persistent infection in horses by equine infectious anemia virus is a complex and lengthy process. Journal of Virology, 71, 3840-3852.
- HAMMOND, S. A., LI, F., MCKEON, B. M. S., COOK, S. J., ISSEL, C. J. & MONTELARO, R. C., 2000. Immune responses and viral replication in long-term inapparent carrier ponies inoculated with equine infectious anemia virus. Journal of Virology, 74, 5968-5981.
- HAMMOND, S. A., RAABE, M. L., ISSEL, C. J. & MONTELARO, R. C., 1999. Evaluation of antibody parameters as potential correlates of protection or enhancement by experimental vaccines to equine infectious anemia virus. Virology, 262, 416-430.
- HAN, G. Z. & WOROBEY, M., 2012. An endogenous foamy-like viral element in the coelacanth genome. PLoS Pathogens, 8, e1002790.
- HARROLD, S. M., COOK, S. J., COOK, R. F., RUSHLOW, K. E., ISSEL, C. J. & MONTELARO, R. C., 2000. Tissue sites of persistent infection and active replication of equine infectious anemia virus during acute disease and asymptomatic infection in experimentally infected equids. Journal of Virology, 74, 3112-3121.
- HERRMANN-HOESING, L. M., NOH, S. M., WHITE, S. N., SNEKVIK, K. R., TRUSCOTT, T. & KNOWLES, D. P., 2009. Peripheral ovine progressive pneumonia provirus levels correlate with and predict histological tissue lesion severity in naturally infected sheep. Clinical and Vaccine Immunology, 16, 551-557.
- HOLLAND, J., SPINDLER, K., HORODYSKI, F., GRABAU, E., NICHOL, S. & VANDEPOL, S., 1982. Rapid evolution of RNA genomes. Science, 215, 1577-1585.
- ISHII, S. & ISHITANI, R., 1975. Equine infectious anemia. Advances in Veterinary Medicine, 19, 195-222.
- ISSEL, C. J., COOK, R. F., MEALEY, R. H. & HOROHOV, D. W., 2014. Equine infectious anemia in 2014: live with it or eradicate it? Veterinary Clinics of North America: Equine Practice, 30, 561-577.
- ISSEL, C. J. & FOIL, L. D., 1984. Studies on equine infectious anemia virus transmission by insects. Journal of the American Veterinary Medical Association, 184, 293-297.
- ISSEL, C. J. & FOIL, L. D., 2015. Equine infectious anaemia and mechanical transmission: man and the wee beasties. Revue Scientifique et Technique, 34, 513-523.
- ISSEL, C. J., RUSHLOW, K., FOIL, L. D. & MONTELARO, R. C., 1988. A perspective on equine infectious anemia with an emphasis on vector transmission and genetic analysis. Veterinary Microbiology, 17, 251-286.
- ISSEL, C. J., SCICLUNA, M. T., COOK, S. J., COOK, R. F., CAPRIOLI, A., RICCI, I., ROSONE, F., CRAIGO, J. K., MONTELARO, R. C. & AUTORINO, G. L., 2013. Challenges and proposed solutions for more accurate serological diagnosis of equine infectious anaemia. Veterinary Record. doi: 10.1136/vr.100735.
- JIN, S., ZHANG, B., WEISZ, O. A. & MONTELARO, R. C., 2005. Receptor-mediated entry by equine infectious anemia virus utilizes a pH-dependent endocytic pathway. Journal of Virology, 79, 14489-14497.
- KEMEN, M. J., MCCLAIN, D. S. & MATTHYSSE, J. G., 1978. Role of horse flies in transmission of wquine infectious anemia from carrier ponies. Journal of the American Veterinary Medical Association, 172, 360-362.
- KONNO, S. & YAMAMOTO, H., 1970. Pathology of equine infectious anemia. Proposed classification of pathological types of disease. Cornell Veterinarian, 60, 393-449.
- KONO, Y., 1969. Viremia and immunological responses in horses infected with equine infectious anemia virus. National Institute of Animal Health quarterly (Tokyo), 9, 1-9.
- KONO, Y., HIRASAWA, K., FUKUNAGA, Y. & TANIGUCHI, T., 1976. Recrudescence of equine infectious anemia by treatment with immunosuppressive drugs. National Institute of Animal Health quarterly (Tokyo), 16, 8-15.
- KONO, Y., KOBAYASHI, K. & FUKUNAGA, Y., 1973. Antigenic drift of equine infectious anemia virus in chronically infected horses. Arch Gesamte Virusforsch, 41, 1-10.
- KONO, Y., YOSHINO, T. & FUKANAGA, Y., 1970. Growth characteristics of equine infectious anemia virus in horse leukocyte cultures. Brief report. Arch Gesamte Virusforsch, 30, 252-256.
- LAIRMORE, M. D., ROSADIO, R. H. & DEMARTINI, J. C., 1986. Ovine lentivirus lymphoid interstitial pneumonia. Rapid induction in neonatal lambs. American Journal of Pathology, 125, 173-181.
- LEROUX, C., CADORE, J. L. & MONTELARO, R. C., 2004. Equine Infectious Anemia Virus (EIAV): what has HIV's country cousin got to tell us? Veterinary Research, 35, 485-512.
- LEROUX, C., CRAIGO, J. K., ISSEL, C. J. & MONTELARO, R. C., 2001. Equine infectious anemia virus genomic evolution in progressor and nonprogressor ponies. Journal of Virology, 75, 4570-4583.
- LEROUX, C., CRUZ, J. C. & MORNEX, J. F., 2010. SRLVs: a genetic continuum of lentiviral species in sheep and goats with cumulative evidence of cross species transmission. Current HIV Research, 8, 94-100.
- LEROUX, C., ISSEL, C. J. & MONTELARO, R. C., 1997. Novel and dynamic evolution of equine infectious anemia virus genomic quasispecies associated with sequential disease cycles in an experimentally infected pony. Journal of Virology, 71, 9627-9639.
- LI, F., CRAIGO, J. K., HOWE, L., STECKBECK, J. D., COOK, S., ISSEL, C. & MONTELARO, R. C., 2003. A live attenuated equine infectious anemia virus proviral vaccine with a modified S2 gene provides protection from detectable infection by intravenous virulent virus challenge of experimentally inoculated horses. Journal of Virology, 77, 7244-7253.
- LI, F., LEROUX, C., CRAIGO, J. K., COOK, S. J., ISSEL, C. J. & MONTELARO, R. C., 2000. The S2 gene of equine infectious anemia virus is a highly conserved determinant of viral replication and virulence properties in experimentally infected ponies. Journal of Virology, 74, 573-579.
- LI, F., PUFFER, B. A. & MONTELARO, R. C., 1998. The S2 gene of equine infectious anemia virus is dispensable for viral replication in vitro. Journal of Virology, 72, 8344-8348.
- LICHTENSTEIN, D. L., RUSHLOW, K. E., COOK, R. F., RAABE, M. L., SWARDSON, C. J., KOCIBA, G. J., ISSEL, C. J. & MONTELARO, R. C., 1995. Replication in vitro and in vivo of an equine infectious anemia virus mutant deficient in dUTPase activity. Journal of Virology, 69, 2881-2888.
- LIGNЙE, M., 1843. Mйmoire et observations sur une maladie de sang, connue sous le nom d'anhйmie hydrohйmie, cachexie acquise du cheval. Rec Med Vet Ec Alfort, 20, 30-45.
- LIM, W. S., PAYNE, S. L., EDWARDS, J. F., KIM, I. & BALL, J. M., 2005. Differential effects of virulent and avirulent equine infectious anemia virus on macrophage cytokine expression. Virology, 332, 295-306.
- LIN, Y. Z., SHEN, R. X., ZHU, Z. Y., DENG, X. L., CAO, X. Z., WANG, X. F., MA, J., JIANG, C. G., ZHAO, L. P., LV, X. L., SHAO, Y. M. & ZHOU, J. H., 2011. An attenuated EIAV vaccine strain induces significantly different immune responses from its pathogenic parental strain although with similar in vivo replication pattern. Antiviral Research, 92, 292-304.
- LIU, Q., MA, J., WANG, X. F., XIAO, F., LI, L. J., ZHANG, J. E., LIN, Y. Z., DU, C., HE, X. J., WANG, X. & ZHOU, J. H., 2016. Infection with equine infectious anemia virus vaccine strain EIAVDLV121 causes no visible histopathological lesions in target organs in association with restricted viral replication and unique cytokine response. Veterinary Immunology and Immunopathology, 170, 30-40.
- MA, J., WANG, S. S., LIN, Y. Z., LIU, H. F., WEI, H. M., DU, C., WANG, X. F. & ZHOU, J. H., 2013. An attenuated EIAV strain and its molecular clone strain differentially induce the expression of Toll-like receptors and type-I interferons in equine monocyte-derived macrophages. Veterinary Microbiology, 166, 263-269.
- MAURY, W., 1994. Monocyte maturation controls expression of equine infectious anemia virus. Journal of Virology, 68, 6270-6279.
- MAURY, W., OAKS, J. L. & BRADLEY, S., 1998. Equine endothelial cells support productive infection of equine infectious anemia virus. Journal of Virology, 72, 9291-9297.
- MCGUIRE, T. C., HENSON, J. B. & BURGER, D., 1969. Complement (C'3)-coated red blood cells following infection with the virus of equine infectious anemia. Journal of Immunology, 103, 293-299.
- MEALEY, R. H., LEIB, S. R., LITTKE, M. H., WAGNER, B., HOROHOV, D. W. & MCGUIRE, T. C., 2009. Viral load and clinical disease enhancement associated with a lentivirus cytotoxic T lymphocyte vaccine regimen. Vaccine, 27, 2453-2468.
- MENG, Q., LIN, Y., MA, J., MA, Y., ZHAO, L., LI, S., YANG, K., ZHOU, J., SHEN, R., ZHANG, X. & SHAO, Y., 2012. A pilot study comparing the development of EIAV Env-specific antibodies induced by DNA/recombinant vaccinia-vectored vaccines and an attenuated Chinese EIAV vaccine. Viral Immunology, 25, 477-484.
- MONTELARO, R. C., PAREKH, B., ORREGO, A. & ISSEL, C. J., 1984. Antigenic variation during persistent infection by equine infectious anemia virus, a retrovirus. Journal of Biological Chemistry, 259, 10539-10544.
- MORCEAU, F., DICATO, M. & DIEDERICH, M., 2009. Pro-inflammatory cytokine-mediated anemia: regarding molecular mechanisms of erythropoiesis. Inflammatory Mediators, 2009, 405016.
- MORE, S. J., AZNAR, I., BAILEY, D. C., LARKIN, J. F., LEADON, D. P., LENIHAN, P., FLAHERTY, B., FOGARTY, U. & BRANGAN, P., 2008. An outbreak of equine infectious anaemia in Ireland during 2006: investigation methodology, initial source of infection, diagnosis and clinical presentation, modes of transmission and spread in the Meath cluster. Equine Veterinary Journal, 40, 706-708.
- MORE, S. J., AZNAR, I., MYERS, T., LEADON, D. P. & CLEGG, A., 2008. An outbreak of equine infectious anaemia in Ireland during 2006: the modes of transmission and spread in the Kildare cluster. Equine Veterinary Journal, 40, 709-711.
- NAGARAJAN, M. M. & SIMARD, C., 2001. Detection of horses infected naturally with equine infectious anemia virus by nested polymerase chain reaction. Journal of Virological Methods, 94, 97-109.
- OAKS, J. L., MCGUIRE, T. C., ULIBARRI, C. & CRAWFORD, T. B., 1998. Equine infectious anemia virus is found in tissue macrophages during subclinical infection. Journal of Virology, 72, 7263-7269.
- OAKS, J. L., ULIBARRI, C. & CRAWFORD, T. B., 1999. Endothelial cell infection in vivo by equine infectious anaemia virus. Journal of General Virology, 80, 2393-2397.
- OLIVEIRA, F. G., COOK, R. F., NAVES, J. H. F., OLIVEIRA, C. H. S., DINIZ, R. S., FREITAS, F. J. C., LIMA, J. M., SAKAMOTO, S. M., LEITE, R. C., ISSEL, C. J. & REIS, J. K. P., 2017. Equine infectious anemia prevalence in feral donkeys from Northeast Brazil. Preventive Veterinary Medicine, 140, 30-37.
- PAYNE, S. L., FANG, F. D., LIU, C. P., DHRUVA, B. R., RWAMBO, P., ISSEL, C. J. & MONTELARO, R. C., 1987. Antigenic variation and lentivirus persistence: variations in envelope gene sequences during EIAV infection resemble changes reported for sequential isolates of HIV. Virology, 161, 321-331.
- PAYNE, S. L., RUSHLOW, K., DHRUVA, B. R., ISSEL, C. J. & MONTELARO, R. C., 1989. Localization of conserved and variable antigenic domains of equine infectious anemia virus envelope glycoproteins using recombinant env- encoded protein fragments produced in Escherichia coli. Virology, 172, 609-615.
- PETERHANS, E., GREENLAND, T., BADIOLA, J., HARKISS, G., BERTONI, G., AMORENA, B., ELIASZEWICZ, M., JUSTE, R. A., KRASSNIG, R., LAFONT, J. P., LENIHAN, P., PETURSSON, G., PRITCHARD, G., THORLEY, J., VITU, C., MORNEX, J. F. & PEPIN, M., 2004. Routes of transmission and consequences of small ruminant lentiviruses (SRLVs) infection and eradication schemes. Veterinary Research, 35, 257-274.
- QUINLIVAN, M., COOK, R. F. & CULLINANE, A., 2007. Real-time quantitative RT-PCR and PCR assays for a novel European field isolate of equine infectious anaemia virus based on sequence determination of the gag gene. Veterinary Record, 160, 611-618.
- RAABE, M. L., ISSEL, C. J., COOK, S. J., COOK, R. F., WOODSON, B. & MONTELARO, R. C., 1998. Immunization with a recombinant envelope protein (rgp90) of EIAV produces a spectrum of vaccine efficacy ranging from lack of clinical disease to severe enhancement. Virology, 245, 151-162.
- RICOTTI, S., GARCIA, M. I., VEAUTE, C., BAILAT, A., LUCCA, E., COOK, R. F., COOK, S. J. & SOUTULLO, A., 2016. Serologically silent, occult equine infectious anemia virus (EIAV) infections in horses. Veterinary Microbiology, 187, 41-49.
- ROBERTS, J. D., PRESTON, B. D., JOHNSTON, L. A., SONI, A., LOEB, L. A. & KUNKEL, T. A., 1989. Fidelity of two retroviral reverse transcriptases during DNA-dependent DNA synthesis in vitro. Molecular and Cellular Biology, 9, 469-476.
- ROUS, P., 1911. A sarcoma of the fowl transmissible by and agents separable from the tumor cells. Journal of Experimental Medicine, 13, 397.
- RUSSELL, K. E., PERKINS, P. C., HOFFMAN, M. R., MILLER, R. T., WALKER, K. M., FULLER, F. J. & SELLON, D. C., 1999. Platelets from thrombocytopenic ponies acutely infected with equine infectious anemia virus are activated in vivo and hypofunctional. Virology, 259, 7-19.
- RWAMBO, P. M., ISSEL, C. J., ADAMS, W. V. J., HUSSAIN, K. A., MILLER, M. & MONTELARO, R. C., 1990. Equine infectious anemia virus (EIAV) humoral responses of recipient ponies and antigenic variation during persistent infection. Archives of Virology, 111, 199-212.
- SALA, M., FERRI, G., SCICLUNA, M. T., SCARAMOZZINO, P., RICCI, I., NARDINI, R., RUOCCO, L., PAGLIALUNGA, M., FORLETTA, R. & AUTORINO, G. L., 2012. What feedback after five years from the implementation of the Italian National Surveillance Programme for Equine Infectious Anemia? Journal of Equine Veterinary Science, 32, S87.
- SCHILTZ, R. L., SHIH, D. S., RASTY, S., MONTELARO, R. C. & RUSHLOW, K. E., 1992. Equine infectious anemia virus gene expression: characterization of the RNA splicing pattern and the protein products encoded by open reading frames S1 and S2. Journal of Virology, 66, 3455-3465.
- SCICLUNA, M. T., ISSEL, C. J., COOK, F. R., MANNA, G., CERSINI, A., ROSONE, F., FRONTOSO, R., CAPRIOLI, A., ANTONETTI, V. & AUTORINO, G. L., 2013. Is a diagnostic system based exclusively on agar gel immunodiffusion adequate for controlling the spread of equine infectious anaemia? Veterinary Microbiology, doi: 10.1016/j.vetmic.2013.02.027.
- SELLON, D. C., FULLER, F. J. & MCGUIRE, T. C., 1994. The immunopathogenesis of equine infectious anemia virus. Virus Research, 32, 111-138.
- SELLON, D. C., PERRY, S. T., COGGINS, L. & FULLER, F. J., 1992. Wild-type equine infectious anemia virus replicates in vivo predominantly in tissue macrophages, not in peripheral blood monocytes. Journal of Virology, 66, 5906-5913.
- SENTSUI, H. & KONO, Y., 1981. Hemagglutination of several strains of equine infectious anemia virus. Archives of Virology, 67, 75-84.
- SHARAV, T., KONNAI, S., OCHIRKHUU, N., TS, E. O., MEKATA, H., SAKODA, Y., UMEMURA, T., MURATA, S., CHULTEMDORJ, T. & OHASHI, K., 2017. Detection and molecular characterization of equine infectious anemia virus in Mongolian horses. Journal of Veterinary Medical Science, 79, 1884-1888.
- SHARLAND, M., GIBB, D. M. & HOLLAND, F., 1997. Respiratory morbidity from lymphocytic interstitial pneumonitis (LIP) in vertically acquired HIV infection. Archives of Disease in Childhood, 76, 334-336.
- SPYROU, V., PAPANASTASSOPOULOU, M., KOUMBATI, M., NIKOLAKAKI, S. V. & KOPTOPOULOS, G., 2005. Molecular analysis of the proviral DNA of equine infectious anemia virus in mules in Greece. Virus Research, 107, 63-72.
- SPYROU, V., PAPANASTASSOPOULOU, M., PSYCHAS, V., BILLINIS, C., KOUMBATI, M., VLEMMAS, J. & KOPTOPOULOS, G., 2003. Equine infectious anemia in mules: virus isolation and pathogenicity studies. Veterinary Microbiology, 95, 49-59.
- SUN, C., ZHANG, B., JIN, J. & MONTELARO, R. C., 2008. Binding of equine infectious anemia virus to the equine lentivirus receptor-1 is mediated by complex discontinuous sequences in the viral envelope gp90 protein. Journal of General Virology, 89, 2011-2019.
- TAGMYER, T. L., CRAIGO, J. K., COOK, S. J., EVEN, D. L., ISSEL, C. J. & MONTELARO, R. C., 2008. Envelope determinants of equine infectious anemia virus vaccine protection and the effects of sequence variation on immune recognition. Journal of Virology, 82, 4052-4063
- TORNQUIST, S. J. & CRAWFORD, T. B., 1997. Suppression of megakaryocyte colony growth by plasma from foals infected with equine infectious anemia virus. Blood, 90, 2357-2363.
- TORNQUIST, S. J., OAKS, J. L. & CRAWFORD, T. B., 1997. Elevation of cytokines associated with the thrombocytopenia of equine infectious anaemia. Journal of General Virology, 78, 2541-2548.
- VALLЙE, H. & CARRЙ, H., 1904. Sur la nature infectieuse de l'anйmie du cheval. Comptes rendus de l'Acadйmie des Sciences, 139, 1239.
- WANG, H. N., RAO, D., FU, S. Q., HU, M. M. & DONG, J. D., 2018. Equine infectious anemia virus in China. Oncotarget, 9, 1356-1364.
- WANG, S. Z., RUSHLOW, K. E., ISSEL, C. J., COOK, R. F., COOK, S. J., RAABE, M. L., CHONG, Y. H., COSTA, L. & MONTELARO, R. C., 1994. Enhancement of EIAV replication and disease by immunization with a baculovirus-expressed recombinant envelope surface glycoprotein. Virology, 199, 247-251.
- WANG, X. F., LIN, Y. Z., LI, Q., LIU, Q., ZHAO, W. W., DU, C., CHEN, J., WANG, X. & ZHOU, J. H., 2016. Genetic Evolution during the development of an attenuated EIAV vaccine. Retrovirology, 13, 9.
- WARDROP, K. J., BASZLER, T. V., REILICH, E. & CRAWFORD, T. B., 1996. A morphometric study of bone marrow megakaryocytes in foals infected with equine infectious anemia virus. Veterinary Pathology, 33, 222-227.
- WERTHEIM, J. O. & WOROBEY, M., 2009. Dating the age of the SIV lineages that gave rise to HIV-1 and HIV-2. PLoS Computational Biology, 5, e1000377.
- WILLIAMS, D. L., ISSEL, C. J., STEELMAN, C. D., ADAMS, W. V. J. & BENTON, C. V., 1981. Studies with equine infectious anemia virus: transmission attempts by mosquitoes and survival of virus on vector mouthparts and hypodermic needles, and in mosquito tissue culture. American Journal of Veterinary Research, 42, 1469-1473.
- YAO, Q., MA, J., WANG, X., GUO, M., LI, Y. & WANG, X., 2017. A pilot study on interaction between donkey tetherin and EIAV stains with different virulent and replication characteristics. Microbial Pathogenesis, 106, 65-68.
- ZHANG, B., JIN, S., JIN, J., LI, F. & MONTELARO, R. C., 2005. A tumor necrosis factor receptor family protein serves as a cellular receptor for the macrophage-tropic equine lentivirus. Proceedings of the National Academy of Sciences of the United States of America, 102, 9918-9923.
- ZHANG, B., SUN, C., JIN, S., CASCIO, M. & MONTELARO, R. C., 2008. Mapping of equine lentivirus receptor 1 residues critical for equine infectious anemia virus envelope binding. Journal of Virology, 82, 1204-1213.
- ZHANG, X., WANG, Y., LIANG, H., WEI, L., XIANG, W., SHEN, R. & SHAO, Y., 2007. Correlation between the induction of Th1 cytokines by an attenuated equine infectious anemia virus vaccine and protection against disease progression. Journal of General Virology, 88, 998-1004.
- ZHENG, Y. H., SENTSUI, H., NAKAYA, T., KONO, Y. & IKUTA, K., 1997. In vivo dynamics of equine infectious anemia viruses emerging during febrile episodes: insertions/duplications at the principal neutralizing domain. Journal of Virology, 71, 5031-5039.