- Infectious Diseases of Livestock
- Part 2
- Bluetongue
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Bluetongue
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Bluetongue
Previous authors: D W VERWOERD AND B J ERASMUS
Current authors:
N J MACLACHLAN - Distinguished Professor Emeritus, BVSc, PhD, Dip ACVP, Department of Pathology, Microbiology and Immunology, VetMed 3A, School of Veterinary Medicine, University of California, One Shlelds Ave, Davis, California, 95616, USA / Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, South Africa
G SAVINI - Head of the Animal Health Department and of the National and OIE Reference Laboratory for BT, DVM, PhD, Istituto Zooprofilatico Sperimentale dell'Abruzzo e Molise "G. Caporale" Via Campo Boario, Teramo, 64100, Italy
Introduction
Bluetongue (BT) is an arthropod-borne viral disease of domestic and wild ruminants, particularly sheep amongst domestic livestock. Clinical disease is characterized by haemorrhage, excoriations and erosion, and cyanosis of the mucous membranes of the oronasal cavity, coronitis and laminitis, oedema of the head and neck, and torticollis. The name of the disease derives from distinctive cyanosis of the tongue, which may occasionally be severe.
Bluetongue was first described in South Africa,142 where the infection (but not disease) has probably been endemic in wild ruminants from antiquity. The disease has been recognized since Merino sheep were introduced into the Cape Colony in the late eighteenth century. From the outset, BT was known to be most prevalent during the summer months, especially in wet seasons. The first detailed study of BT was made by Spreull,293, 294 but Theiler312 was the first to demonstrate the filterability of the aetiological agent, thus indicating its viral nature. He also introduced the first vaccination, making use of a virus strain (subsequently shown to be serotype 4) attenuated by passage in sheep.313 This crude vaccine, which consisted of sheep blood, produced mild BT in recipient animals and, although it contained a single serotype, nevertheless induced remarkable protection. Despite its obvious shortcomings it afforded sheep farmers a reasonably effective method of control, and was used for almost 40 years.
Bluetongue was initially thought to be confined to the African continent. In 1943 the first confirmed outbreak outside Africa was reported in Cyprus,101 but the disease may have occurred there as early as 1924.284 Outbreaks of BT or presence of the causative agent, Bluetongue virus (BTV), were subsequently reported in Israel,162 the USA,118, 207 Portugal197 Spain,167 Pakistan278 and India,277 and later in the Middle East, China, Southeast Asia and Australia.84, 185, 239, 287, 289, 345 With the notable exception of transient incursions of single virus serotypes into the Iberian Peninsula and Greece, Europe was historically free of BT until 1998241 when multiple serotypes of BTV spread throughout extensive portions of the continent.86, 214, 256, 273 This recent epidemic initially involved only regions of Europe adjacent to the Mediterranean Sea but later, with the incursion of a novel strain of BTV serotype 8, spread to affect virtually the entire continent as well as portions of the United Kingdom and Scandinavia. In recent years live-attenuated vaccine-derived strains of BTV have also spread through much of Europe.67, 93, 233
Early reports incriminating blood-sucking insects as vectors were based on circumstantial evidence. The first experimental proof that BTV is transmitted in South Africa by Culicoides imicola (= C. pallidipennis) was obtained by Du Toit in 1944.77 Subsequently many other Culicoides spp. (Figure 1) were proved to be vectors of BTV in the Americas, Asia, Australia, and Europe, indeed it is now clear that different vector species are responsible for dissemination of different constellations of BTV serotypes in distinct global episystems.17, 31, 104, 108, 191, 252, 297, 309, 311, 339
The existence of a plurality of BTV serotypes, which had long been surmised, was confirmed by Neitz in 1948 by means of cross-protection studies in sheep.226 It provided an explanation for the vaccination failures that had been experienced with the monovalent Theiler vaccine and also formed the basis for later studies on the in vitro serotyping of viral isolates.127, 128
Further advances in the understanding of BT and BTV followed rapidly on the development of techniques for the study of viruses. Some of the important milestones were the isolation and propagation of BTV in embryonated chicken eggs (ECE) by yolk sac6, 201 and intravascular109 inoculation, and its adaptation to the brains of suckling mice for antigen production.162, 318 This was followed in 1956 by the successful propagation of BTV in cell cultures by Haig et al.117
The availability of cell culture systems for the propagation of BTV led to its successful purification, to the demonstration that it is composed of a segmented, double-stranded RNA genome enclosed in a double-layered capsid,326 and to its classification in the genus Orbivirus in the family Reoviridae.37, 224, 327 Most recently, rapid sequencing procedures have facilitated the genetic characterization of strains and serotypes of BTV from different global ecosystems.17, 43, 169, 231
The continued natural evolution of BTV and other orbiviruses poses a substantial challenge to research and regulatory institutions, stimulating worldwide interest and research activity. Comparative studies of the structural and functional relationships of these viruses have yielded much information, which has been reviewed elsewhere.329 Because of its economic importance and wide distribution, BTV, however, remained the focus of attention among orbivirus infections of ruminant livestock.139, 269, 295 The recent identification in Europe,170 Africa and Asia of genetically distinct BTVs that infect small ruminants (goats and sheep), potentially without requirement for an insect vector (see Epidemiology), has further complicated the taxonomic classification of BTV and understanding of the epidemiology of BTV infection.25, 123, 183, 185, 281, 283, 331, 347 A number of reviews on both early and more recent aspects of BT research have been published.19, 42, 51, 110, 126, 137, 185, 187, 191, 255, 273, 320
Aetiology
Based on its morphologic and molecular properties, BTV is classified as the typespecies of the genus Orbivirus in the family Reoviridae, subfamily Sedoreovirinae.37, 265, 327 Initially, BTV was classified as an arbovirus because of its transmission by biting midges, but its stability in the presence of lipid solvents, suggesting the absence of an envelope, as well as early electron microscopic studies304 subsequently led to its provisional classification as a reovirus.13 However, it differed in a number of respects from reoviruses (its smaller size, absence of a structured outer capsid layer and concomitantly greater sensitivity to extremes of pH and high temperature, and its requirement for an insect vector in the classical serotypes at least) and this resulted in it being classified in a separate genus.
Morphology
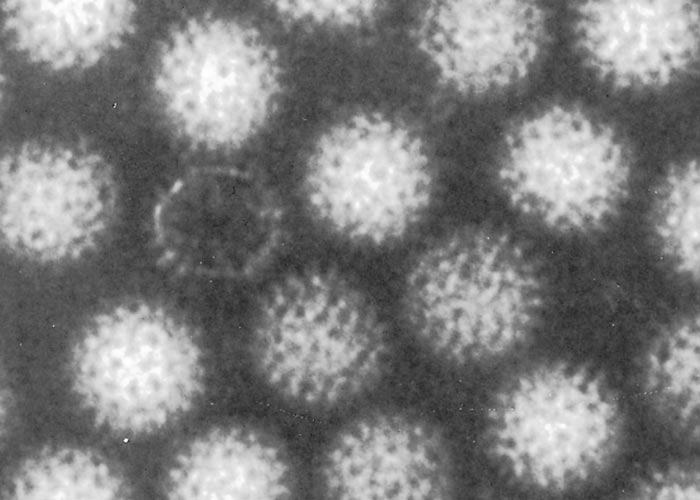
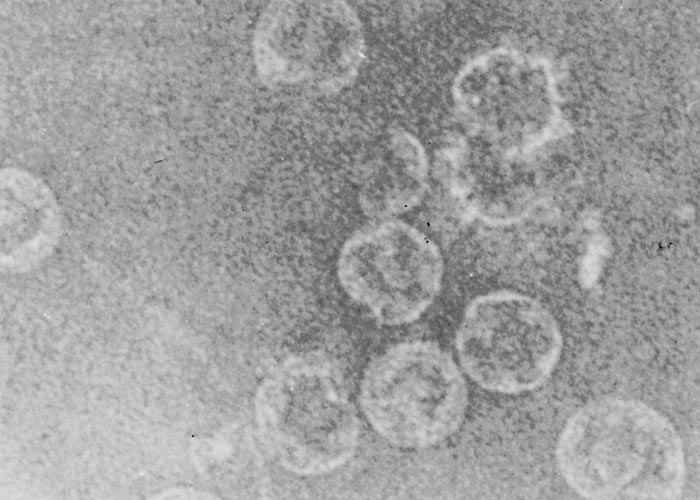
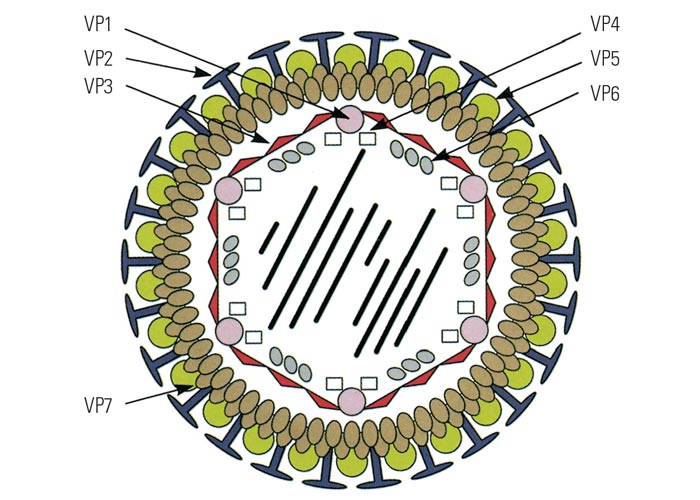
Negatively- stained BTV virions are non-enveloped, spherical and have an approximate diameter of 68 to 70 nm. When examined by electron microscopy, they have a ‘fuzzy’ appearance caused by a diffuse outer layer in the protein capsid. (Figure 2a) This layer, comprising two proteins (VP2 and VP5), can be removed enzymatically or by manipulating the salt concentration and pH of the environment220, 319 to reveal a highly structured core particle. (Figure 2b) The core particle, with a diameter of 54 nm, includes two structural proteins (VP3, VP7) and the transcriptase complex composed of VP1, VP4 and VP6. The name of the genus is derived from the capsomere structure (orbis = ring in Latin). Core particles can be further degraded by removal of VP7 to create subcore particles that include only VP3 and the transcriptase complex (Figure 2c).141 Using cryo-electron microscopy, diameters of approximately 80 and 70 nm have been obtained for virions and viral cores, respectively.120 The atomic structure of the core particle of BTV has been determined by X-ray crystallography at a resolution approaching 3.5Å and is shown in (Figure 3). It explains how approximately 1 000 protein molecules self-assemble to form transcriptionally active viral core particles.113
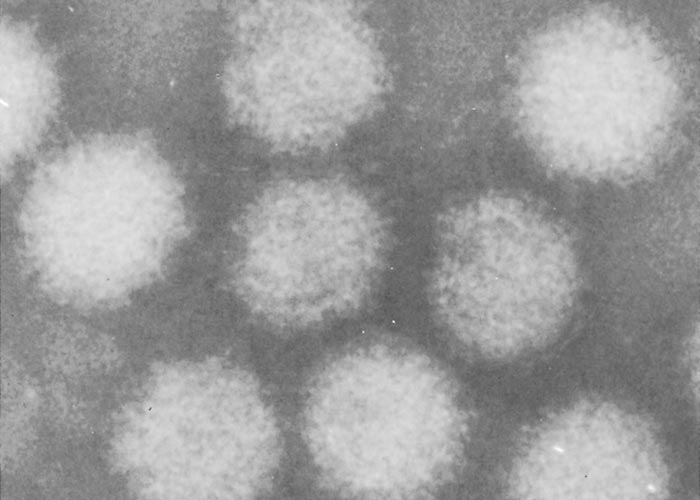
Figure 2a Electron micrographs of negatively stained bluetongue virus core particles (Courtesy of Mr H.J. Els, Faculty of Veterinary Science, and University of Pretoria, South Africa)
Genomic structure
Bluetongue virus was the first virus of domestic animals to be shown to possess a double-stranded RNA genome, initially thought to be unique to reoviruses. This finding was based on heterochromatic staining of the viral inclusion bodies with acridine orange, resistance to degradation by pancreatic RNAse, a characteristic melting curve, and a base composition suggesting A-U and G-C base pairing.326 The genome consists of ten segments which can be separated electrophoretically after disruption of the virion. These segments vary in size from 0.5 kDa to 2.7 × 103 kDa,329 and function as genes (i.e. they are translated individually into functional viral proteins). The coding relationships between the genome segments and the viral proteins are shown in Table 1.
Genome segments are labelled 1 to 10 in descending order of size (S1 – S10), whereas viral polypeptides are divided into structural proteins (VP1–VP7) and non-structural proteins (NS1–NS5).300 Nucleotide sequences have been determined for all ten segments of a number of BTV serotypes. A schematic representation of the structural components of BTV is shown in Figure 3.
Table 1 Proteins and corresponding genome segments of bluetongue virus
| PROTEIN | NUMBER OF GENOME SEGMENTa | LOCATION | MOLECULAR MASSb (kilo Dalton) | ESTIMATED NUMBER OF MOLECULES PER VIRIONc |
| VP1 | 1 | Core | 149 | 11 |
| VP2 | 2 | Outer capsid | 111 | 180 |
| VP3 | 3 | Core | 103 | 120 |
| VP4 | 4 | Core | 76 | 9 |
| VP5 | 6 | Outer capsid | 59 | 292 |
| VP6/NS4 | 9 | Core (VP6) and infected cell (NS4) | 35 | 67 |
| VP7 | 7 | Core | 38 | 780 |
| NS1 | 5 | Infected cell | 64 | 0 |
| NS2 | 8 | Infected cell | 40 | 0 |
| NS3/3A | 10 | Infected cell | 25 | 0 |
| a | Assignments as suggested by Pedley et al.246 Segments are numbered in order of decreasing size, which agrees with their order of migration in 1 per cent agarose gels |
| b | Calculated from nucleotide sequences obtained for cloned genome segments172 |
| c | Numbers in bold have been determined from X-ray data,113 or cryo-electron microscopy. Otherwise the ratios estimated by Huismans & Van Dijk137 have been normalized to 780 copies of VP7 per particle |
Structure and function of the viral proteins
The viral proteins range in size from approximately 25 000 to 149 000 Dalton.199, 220, 264, 265, 327 Four of the seven BTV proteins (VP2, VP3, VP5 and VP7) are major structural components and constitute about 94 per cent of the total virion protein. The other three (VP1, VP4 and VP6) that constitute the replicase complex are minor components, making up the other 6 per cent. Two of the major components (VP2 and VP5) form the diffuse outer capsid layer, whereas the other five are found in the core particle.111, 220, 327
VP1 is a 149 kDa minor component of the subcore particle and functions as an RNA-dependent RNA polymerase. A comparison of the amino acid sequences derived from various serotypes indicates that it is a highly conserved molecule.
VP2 is the major constituent of the outer capsid and the main antigen responsible for the induction of serotype-specific neutralizing antibodies,136, 247 although VP5 also plays a role.221 When these two proteins are removed, the infectivity of the virion for mammalian cells is greatly reduced, whereas it is essentially retained for the insect vector.219 This reflects an essential role in binding to cell receptors and penetration into the host cell. Not unexpectedly, VP2 is the most variable of the BTV proteins, although conserved regions in genome segment 2 have also been identified.
VP3 proteins form the shell of the subcore particles and act as a scaffold for the surface layer of the core particle, which consists of VP7.141 It also binds to RNA and interacts with the replicase complex of VP1, 4 and 6, indicating a fundamental role in the early stages of assembly of the BTV particle. It has a sequence that is highly conserved among BTV serotypes and other orbiviruses.310, 342
VP4 of BTV binds GTP and is a candidate guanylyl transferase of the virus.35 It is a minor component of the subcore particle, but was shown to be involved in the encapsidation process in a dimerized form. A putative leucine zipper near the carboxy terminus of the protein was found to be essential for the dimerization of VP4.257
VP5 (together with VP2, as described above) is part of the outer capsid of intact BTV particles. In contrast to VP2, VP5 is a relatively conserved protein with no clear regions of variability.79 In fact, comparing the complete nucleotide sequence of epizootic haemorrhagic disease of deer virus (EHDV) with those of various BTV serotypes, a 59 to 62 per cent homology was found.148
VP6 can often be resolved into two closely migrating bands by SDS-PAGE. Expression of a cloned genome segment 9, which codes for this minor component of the BTV core particle, showed that the two forms of the protein are derived from the initiation of protein synthesis at two distinct sites.332 Sequences within the protein are required for the retention of VP6 in the cytoplasm, and the protein was also shown to bind ATP and RNA and to possess an RNA-dependent ATPase function and helicase activity.60, 299
VP7, a major structural protein, forms the surface layer of the orbivirus core particles responsible for the ring-like ‘capsomere’ surface structures. It is the immunodominant group-specific antigen and its crystal structure has been determined by means of X-ray crystallography.113 Full sequencing of cloned genome segments 7 of various orbiviruses revealed a remarkable homology between those of BTV, EHDV and African horse sickness virus (AHSV).61, 147, 268 This close relationship is probably explained by the recent demonstration that VP7 is intimately involved in the binding of BTV to membrane proteins of the Culicoides vector, common to all of them.344
The nonstructural proteins are produced in infected cells and probably play a role in the assembly of the virion. NS1 is formed in excessive amounts and in the form of characteristic tubules in the cytoplasm of infected cells that promote synthesis of other viral proteins. A high degree of homology was found between the nucleic acid sequences of segment 6 (which encodes NS1) derived from BTV and related EHDV.227 Extensive homology was also found in the nucleotide sequences of genome segment 8 of BTV and EHDV, which encode their respective NS2 proteins. It is the only orbivirus protein that is phosphorylated by means of a cytoplasmic protein kinase. It is the major component of viral inclusion bodies and also has the ability to bind single-stranded RNA.314, 323, 332
The smallest RNA genome segment 10 encodes two closely related non-structural proteins, NS3 and NS3A. Both occur as glycosylated and non-glycosylated forms in infected cells.18 Glycosylation reflects their association with plasma membranes and suggests that they may be involved in the final stages of morphogenesis, i.e. the release of virions from infected cells.145 The NS3 protein facilitates viral egress from infected cells and it may be involved in the host innate immune response by down regulating activation of the interferon (IFN-b) promoter.58, 188 A high degree of homology and conservation was found in a comparison of proteins derived from various BTV serotypes144 but more variation between BTV, AHSV and Palyam virus.322
In addition to VP6, gene segment 9 encodes in another reading frame a small nonstructural protein (NS4) that can localize to the nucleolus of infected cells and which serves as an interferon agonist during virus replication.30, 258 Specifically, NS4 promotes replication of BTV in cells pre-treated with Type 1 interferon. An ORF in Seg 10 overlapping the larger ORF encoding NS3/3a expresses another non-structural protein S10-ORF2 (putative NS5 protein), which appears to function as an interferon antagonist.300
Physicochemical characteristics of the viral particles
The buoyant density of intact BTV virions is 1.36 g/ml, whereas the core particle has a density of 1.40 g/ml, reflecting the loss of the outer capsid proteins. When freeze-dried in buffered lactose-peptone, BTV can survive almost indefinitely at room temperature. In blood and tissue specimens it is very stable at 20 °C, 4 °C and −70 °C but not at −20 °C. The virus is unstable below pH 6.5,238, 308 and on purification, when all extraneous protein is removed, it is extremely unstable at high temperatures.129 Because of the double-stranded nature of its RNA genome, BTV is relatively resistant to ultraviolet and gamma irradiation. It is also relatively resistant to lipid solvents such as ether and chloroform, but it is readily inactivated by disinfectants containing acid, alkali, sodium hypochlorite and iodophors.126
Serological characteristics of bluetongue viruses
The existence of multiple BTV serotypes was first discovered in a series of tests in sheep when Neitz226 found that each of ten viral isolates from field cases of BT produced solid immunity against itself but only partial or no protection against heterologous strains. Although cross-protection tests in sheep proved the plurality of BTV strains they did not allow their classification into serotypes. Howell127 devised an in vitro neutralization test by which 22 BTV isolates could be grouped into 12 distinct serotypes. A further four serotypes were discovered after the introduction of the plaque inhibition test.126, 128 A total of 24 distinct serotypes were subsequently identified using conventional virus isolation and serology but, with the advent of molecular detection and identification methods, at least 3 additional types (presumed serotypes) have been described. Some of these genetically novel BTVs have never been isolated in cell culture so their precise serological characteristics remain uncertain.29, 281 The 27 distinct serotypes of BTV are all interrelated in a complex network of cross relationships,90 with common group antigens. However, analyses of the entire genomes of field strains of BTV from endemic regions of the world confirm widespread reassortment of viral genes, as well as remarkable genetic drift of individual BTV genes.17, 43, 231, 236
Epidemiology
Repeated recent incursions of BTV into previously free regions confirm that the global range of the virus is expanding, and that the epidemiology of BTV infection is changing. Historically, the virus was thought to be restricted to a geographic band between the latitudes 40°N and 35°S, coinciding with the distribution of competent vectors, but it is now clear that the virus’ range extends beyond that limit in much of the Northern Hemisphere including North America, Europe and Asia, up to at least 50°N.3, 86, 104, 153, 180, 185, 191, 233, 263, 335, 340 Both anthropogenic and natural factors might have influenced the spread and transmission of BTV into previously free areas, specifically through movement of viraemic animals (domestic and wild ruminants) or through active (local) propagation following wind-borne dissemination of infected Culicoides spp. from infected regions.80, 104, 169 Other potential routes include the translocation of live-attenuated vaccine viruses or infective animal products, or infected midges within living (plants, animals) or inanimate (e.g. airplanes, ships) objects or products. However, it is increasingly apparent that climatic changes have facilitated recent expansions in the global range of BTV infection.86, 115, 191, 255, 256, 273
With the notable exception of the recently identified, horizontally transmitted small ruminant-adapted BTVs (see Small ruminant-adapted bluetongue viruses), the establishment of BTV infection in previously virus-free areas is dependent on the presence of reservoir and amplifying hosts such as wild ungulates (game animals) and cattle, and on suitable species of Culicoides midges that serve as vectors.104, 185, 191, 309 Given that ruminant animals, wild and/or domestic, are abundant in many regions where incursions have occurred, it is the presence and activity of competent insect vectors that typically determines whether virus persists in a given area. The distribution of BT therefore closely follows the spatial and temporal distribution of its Culicoides vectors. The dynamics of vector–host relationships and their influence on the spread of the disease have been reviewed.105 Factors such as flight periodicities, biting activities, host preferences, preferred habitats for oviposition and climate have a direct influence on the incidence and spread of BT.
Even in the very early descriptions of BT it was clearly recognized that the disease is geographically limited, seasonal in its occurrence in temperate zones, most common in areas of southern Africa with high rainfall and after good rains, and that sheep can be protected by being housed at night and dipped in an insecticide. These observations, as well as the disappearance of the disease after the first frosts, were convincing evidence for the role of an insect vector in the occurrence of the disease.143, 294
In his classical experiments Du Toit77 initially transmitted BT to susceptible sheep by inoculation of homogenized pools of wild-caught C. imicola, and subsequently by the bites of these midges 10 days after they had fed on a sheep infected with BTV. This finding was confirmed under controlled conditions in the USA, but with C. sonorensis (formerly C. variipennis).98 Replication of the virus in the salivary glands of C. sonorensis (formerly C. variipennis) was demonstrated electron microscopically.41 Infected midges remain infective for the rest of their lives. Transovarial transmission of the virus from infected midge females to their F1 progeny has never been proven with any Culicoides species, rather variation in the distribution, activity and competence of the population of adult Culicoides midges can determine whether an area is endemic or BTV-free. The endemic areas for BT and BTV infection are largely defined by suitable climatological factors, which are mainly confined to the tropics and subtropics although temperate regions are increasingly affected.105, 108, 153, 285 Apart from certain high-altitude regions, such as parts of the north-eastern Eastern Cape Province in South Africa and Lesotho, the entire southern African region should be regarded as endemic. Bluetongue virus has been isolated in various parts of the world from a wide variety of Culicoides spp. (see Vectors: Culicoides spp.). The most important and widespread Asian-African vector is C. imicola whose habitat includes most of Africa, the Mediterranean Basin including southern Europe, the Middle East, and portions of Asia including Laos, Vietnam and southern portions of of China. C. sonorensis and C. insignis are important vector species in the Americas, C. brevitarsis and C. wadai in Australia, and species of the Obsoletus (including C. obsoletus and C. scoticus) and Pulicaris (including at least C. pulicaris and C. newsteadi) complexes in Europe.31, 104, 105, 108, 214, 297, 301, 309, 311
Laboratory infection of mosquitoes with BTV has been reported, and the disease itself has been transmitted experimentally by the sheep ked, Melophagus ovinus,175 and a soft tick, Ornithodoros coriaceus,303 but a biological cycle was not demonstrated in these vectors, suggesting mechanical transmission. Blood-sucking flies such as Stomoxys and Tabanus spp. should also be considered as potential mechanical vectors. However, compared to biological transmission, mechanical transmission – whether by insects or fomites - is probably of minor significance in the spread of BTV. Interestingly, transstadial and transovarial passages of BTV have been observed in hard and soft ticks, but the biological significance (if any) of this observation remains uncertain in naturally-occurring virus transmission.38
Vector independent transmission of BTV clearly can occur, although its significance is largely unknown. Although BTV infection is not generally contagious and very little virus is found in the secretions and excretions of infected animals, oral BTV infection of both ruminant livestock and wild and zoo carnivores has been described, including infection of calves via the feeding of infective colostrum.187, 203 Direct contact transmission of certain recently identified novel “small ruminant” BTV serotypes has been demonstrated in livestock, likely by aerosol, and at least some recent European strains of BTV can be transmitted directly between ruminants in close contact.25, 45, 291, 292 The mechanisms of contact transmission are uncertain but might include shared feed and water troughs, contamination of wounds with blood during fighting among red deer, and contact with infected placentas in a case in cattle. Seropositive dogs, cats and wild carnivores in Africa were thought to have eaten tissues from infected animals,5 and Eurasian lynx in a zoo had been fed ruminant foetuses and stillborn animals from outbreak areas.150
Vertical transmission of BTV in animals certainly does occur, notably with live-attenuated vaccine strains of BTV and European BTV-8, but this route is considered unlikely to be important to the epidemiology of natural BTV infections.187, 191, 274 However, some of these animals can be born infected and could therefore introduce the virus to new areas if the dam is transported during gestation. Transplacental transmission has been demonstrated in dogs, at least for serotype 11.337
BTV-8 has been detected in the semen of naturally infected bulls by both PCR and virus isolation, indicating that BTV-8 may contaminate semen in a similar way to tissue culture passaged viruses. Further work is required to investigate the duration of BTV-8 presence in semen.161 Apart from BTV-8, field/wild strains of BTV have only been identified in the semen of older bulls, most likely due to blood contamination.39 It has been demonstrated that females inseminated with cell-culture adapted BTV-contaminated semen can become infected. The vast majority of evidence shows that isolation of BTV in semen coincides with the viraemic period in bulls/rams. However, one study showed that a tissue culture passaged strain of BTV-1 was detected in semen for approximately 10 days beyond the period of detectable viraemia in the bull. No evidence was found for congenital transmission when cows were infected by various routes.187, 191, 244
The role that embryo transfer might play in the transmission of BT seems to be negligible. Embryos recovered non-surgically from infected donor cattle with high BTV titres at the time of collection failed to infect any of 39 recipients, as judged by the absence of seroconversion and the inability to isolate virus from them.40 It was also found that BTV does not infect bovine zona-intact oocytes or embryos during in vitro maturation and culture.316 In sheep, embryos from BTV-infected ewes also failed to transmit the disease.119 A quantitative risk analysis indicated that the risk of transmission of BT by embryos is 1:30 000 during the vector season and 1:1 million out of season.306 In general, embryo transfer can be regarded as a safe procedure as far as accidental BTV transfer is concerned, provided the donor animals are not viraemic at the time of semen and embryo collection,106 and that the embryos are washed extensively.290
Human activities may in some instances be important in mediating the introduction of BTV, rather than movement of either infected animals or vectors.67, 259 For example, the recent appearance of at least 3 (BTV-6, BTV-11, BTV-14) different South African live-attenuated BTV vaccine strains amongst livestock in Europe (most recently BTV-14 in western Russia, Poland and Lithuania).86 Interestingly, live attenuated vaccine strains of BTV-6 and BTV-11 first appeared in the same general region of northern Europe as did BTV-8 in 2006, again suggesting an as yet unexplained anthropogenic phenomenon. Similarly in India, strains of BTV that are genetically similar or identical to live-attenuated vaccine viruses used elsewhere in the world have also been identified as infecting local livestock, raising further concerns regarding the unauthorized international movement of vaccine viruses.184
Contamination of biological products, especially those that utilize foetal bovine serum or primary ruminant cell lines, is also well described as, for example, in canine, sheep and cattle vaccines.4, 49
Although traditionally regarded as a sheep disease because of its economic importance in this species and the occurrence of major epidemics that cause heavy economic losses in sheep-rearing countries,87 BT affects a wide range of species. The outcome of infection, however, varies among different species and breeds as well as among individuals of the same species. Indigenous breeds of sheep are generally less severely affected by BT than are exotic breeds. In sheep, the viraemia reaches a relatively high level (about 105 sheep ID50/ml) and is usually associated with severe clinical disease in fully susceptible animals. South American camelids (llamas, alpacas etc.) also may develop severe disease following BTV infection.103, 222, 234 With the notable exception of especially virulent viruses such as European BTV-8, clinical disease is rare in cattle and goats, and, when present, is much milder than in sheep.125
During a season of exceptionally high rainfall and Culicoides activity in South Africa, cattle with typical signs of stomatitis and coronitis were shown to have a double infection with BT and EHD viruses, apparently exacerbating the clinical signs.102
The susceptibility of wild ruminants was first established by experimental infection of blesbok (Damaliscus albifrons).225 Evidence of inapparent infections have subsequently been found in many other species111, 164 and it is now accepted that potentially all ruminants and camelids are probably susceptible to infection.21 African antelopes do not develop clinical disease, whereas white-tailed deer (Odocoileus virginianus), pronghorn antelope (Antilocapra americana) and desert bighorn sheep (Ovis canadensis) of the North American continent may develop severe clinical disease.121 The European strain of BTV-8 caused severe disease in all non-African ungulates, whereas infection was invariably asymptomatic in ungulates of African origin.196
Canine fatalities and abortion were recognized in pregnant dogs immunized with a canine vaccine contaminated with BTV.4, 337 Similarly, fatal infections occurred in Eurasian lynx that consumed BTV-8-infected cattle foetuses aborted during the recent European epidemic.150
Bluetongue virus may be transmitted throughout the year in areas where the winter is mild. However, overwintering of the virus in areas with long, cold winters is more difficult to explain, as transovarial transmission of BTV in Culicoides spp. apparently does not occur.157, 235 It is possible that cattle are able to act as reservoirs,228 although recent investigations only partially support this contention as viraemia in experimentally infected livestock is typically less than 60 days.187, 191 Furthermore, BTV replication in tissues is transient and, with the exception of some recently described small ruminant BTVs, truly persistent infection does not occur.189 It has also been postulated that in southern Africa the primary replicative cycle involves one or more species of African antelope and Culicoides midges, and that the role of antelopes has been largely supplanted by cattle in areas where agricultural development has displaced wild animals.78 Outbreaks of the disease in sheep usually occur simultaneously in widely separated localities in late summer or autumn, suggesting that populations of BTV-infected midges build up in the primary cycle involving cattle or wild animals during spring and early summer, and that sheep become infected in a secondary cycle as a result of ‘spill-over’.89 Importantly, recent studies in North America, and subsequently in South Africa, confirm the presence of BTV-infected adult midges during winter months in endemic temperate regions, suggesting that the virus overwinters in long- lived vector midges.202, 204, 301 It is likely that these infected midges that survive during winter could initiate a low level cycle of infection amongst domestic and/or wild ungulates, an infection cycle that would further facilitate virus overwintering.228 Other less likely mechanisms of over-wintering include vertical transmission of the virus in animals or ticks, although biological significance of these to occur is likely minimal.
In contrast to epidemic areas where incursions occurred, such as much of Europe currently, BTV serotypes are distributed apparently randomly throughout endemic areas like South Africa and North America, and the dominant serotypes at any given time are largely determined by herd immunity. Monitoring for BTV of Culicoides trapped at various sites in South Africa over a period of six years (1979 to 1984) revealed that 14 to 18 different serotypes were encountered every season, albeit at markedly varying frequencies. Usually, three to five serotypes were isolated predominantly and these were often responsible for more than 60 per cent of the total number of isolates for that particular season. These dominant serotypes were largely replaced by others the following season, only to become dominant again three to four years later. Such serotypes obviously possess a high epidemic potential. In this category were types 1 to 6, 8, 11 and 24. A second group of BTV serotypes was present at much lower levels but was encountered every season. In this category were types 9, 10, 12, 13, 16 and 19. A third group represented by types 7, 15 and 18 appeared sporadically and probably has a low epidemic potential.92
The epidemiology of the small ruminant-adapted BTVs that recently have been identified amongst goats and sheep in Europe, Africa and Asia may be quite different than that of the “conventional” 24 serotypes.25, 123, 183, 185, 281, 283, 331, 347 For some of them transmission apparently occurs without any requirement for an insect vector and may therefore be independent of climatic conditions. In further contrast to other BTV serotypes, infection of goats with BTV serotype 25 is persistent (> 2 years) (see section: Small ruminant- adapted BTVs).
Small ruminant-adapted bluetongue viruses
In the past decade, new serotypes of BTV have been described in Europe, Asia and Africa.49, 123, 170, 183, 198, 281, 305, 347 These viruses represent a novel group of BTVs that differ in their biological and genetic properties from the historical 24 serotypes (BTV 1 – 24). These novel BTVs exhibit minimal virulence or pathogenicity to ruminants; they have all been isolated from apparently healthy goats or sheep, and at least some of them can be transmitted by contact without requirement for vector insects. Although little as yet is known about these new BTV types, some have already been classified as additional serotypes (specifically BTV 25, 26, 27),123, 183, 347 whereas other apparently new serotypes have not been assigned a numerical serotype designation, only an identification designation.49, 170, 198, 281, 305
BTV-25 was initially identified as a new member of the genus Orbivirus and named as Toggenburg Orbivirus (TOV) after the region of its first detection in goats in Switzerland. The sequences of several genome segments (2, 5, 6, 7, 8, 9, and 10) of TOV clearly confirmed that it was a new BTV serotype, specifically BTV-25.123 Although first detected in 2008, subsequent retrospective analyses confirmed positive samples from Swiss goats dating back to 1998, indicating that BTV-25 has circulated in that part of Europe for at least a decade and likely considerably longer. BTV-25 produces subclinical infections in goats and is only mildly pathogenic to sheep.56 Although BTV-25 infection is highly prevalent amongst goats in certain areas of Switzerland, infection was not found in either sheep or cattle cohabiting with virus-positive goats, nor in wild animals, suggesting a highly restricted host range.54 The high prevalence of infection in goats is in part likely a consequence of the ability of BTV-25 to induce persistent infection in goats, as BTV-25 infected goats remain infective for up to 19 months and RNA-positive up to 25 months.331 Direct contact transmission of BTV-25 might also contribute.56 Experimental studies are difficult with BTV-25 because the virus is yet to be propagated in vitro using systems that are reliably used for propagation of BTV 1-24.250 A probable variant of BTV-25, BTV-Z ITA2017, was recently identified in a goat flock from North Western Italy.198 This strain is most related in Seg-2/VP-2 (83.8% nt/82.7% aa) to BTV-25. No clinical signs were observed in infected animals, which showed only low levels of RNA-aemia. Reactive antisera of goats positive by cELISA for BTV antibodies failed to neutralize a chimeric virus expressing the outermost protein of TOV.
BTV-26 was first isolated in 2010 from a blood sample of a sheep in a mixed goat and sheep flock in the Abdali region of Kuwait.183 The RNA extracted from this isolate was not amplified by a segment 2 (Seg-2) specific RT-PCR for each of BTV1-25, indicating it was a novel serotype. The sequence of Seg-2/VP2 of BTV-26 differs from all other known serotypes, but is more closely related to BTV-25 than other serotypes with a nt/aa identity of 63.9% and 61.5%, respectively. These results were then confirmed by the virus neutralization assay as none of the antisera against the existing 25 BTV serotypes neutralized BTV-26. Unlike BTV-25, BTV-26 can be propagated in vitro although its growth capability is limited to mammalian cell lines and the virus does not replicate in Culicoides-derived cell lines (Kc). Experimental BTV-26 infection results in a higher and longer-lasting RNA-aemia in goats than in sheep (28 vs. 21 days); whereas BTV-26 resulted in subclinical infection of goats, it produced mild disease in sheep.26, 27 BTV-26 can be experimentally spread between goats by direct contact transmission, with both nasal and ocular secretions as possible sources of infection.25 Horizontal transmission of BTV-26 requires close contact among animals. Serological evidence of BTV-26 infection subsequently was shown amongst cattle and dromedaries in Mauritania and in Libyan sheep.168, 195
BTV-27 was first detected among goats in Corsica in 2014.347 The infected goats were positive by BTV group-specific PCR but negative to Seg 2 specific assays specific for each of the other 26 BTV serotypes. Sequence analyses of Seg-2/VP2 of this virus showed a high identity with both BTV-25 and BTV-26, sharing 73% nt and 75% aa identity with BTV-25 and 65% nt and 60% aa identity with BTV-26. Furthermore, the virus was not neutralized by antisera raised against BTV-1 to BTV-26. Like BTV-26, in vitro propagation of BTV-27 is limited to selected cell lines; specifically, the virus was not isolated in ECE or on cell lines such as Kc, Vero and BHK that are commonly used for isolation of “traditional” BTV serotypes. Indeed, only a single isolate was obtained and that was accomplished using BSR cells. Three different variants of BTV-27, with small but detectable nt and aa differences (<8%nt/<10%aa) were described subsequently.283 All were found in healthy goats. Cattle appear to be refractory to infection as neither RNA nor antibodies were detected following experimental infection with a variant of BTV-27. It appears that BTV-27 is poorly immunogenic as virus - specific antibodies and RNA were detected at low levels and for a short period in infected goats. Analogous to BTV-26, recent studies suggest BTV-27 also can be spread by direct contact transmission.45
There are several other recently identified BTVs that likely constitute new virus serotypes and, of these, X ITL2015 is best characterized.281 X ITL2015 was identified in healthy goats in Italy, and has yet to be detected in either sheep or cattle. Although clearly a BTV in terms of reaction with group-specific RT-PCR and serological testing of infected animals, the virus is virologically and serologically distinct from the 27 recognized BTV serotypes. The Seg-2/VP2 sequences of this virus cluster with the other new BTVs (BTV-25, 26, 27) with the highest identity (75.3–75.5% nt/77.4–78.1% aa) with the recently isolated BTV-27s from Corsica and with recently discovered XJ1407 from China (75.9% nt /78.2% aa), whereas it is less related to BTV-25 (73.0% nt/75.0% aa) and BTV-26 (62.0% nt/60.5% aa). Serum of X ITL2015 infected goats failed to neutralize any of the other virus serotypes but, like BTV-25, all attempts to isolate X ITL2015 using ECE or cell cultures have been unsuccessful. Serological studies suggest that this virus, like BTV-26 and 27, may be spread by direct contact transmission.
XJ1407 was first detected in clinically normal goats and sheep in China.305 The virus is most related to BTV X ITL2015, and Seg-2/VP2 of XJ1407 shares high sequence identity with BTV-27 (75% nt/78% aa) and BTV-25 (73% nt/75% aa).123, 183, 283 The virus has not yet been serologically characterized, although it can be propagated in ECE and several mammal and insect cell lines.
Another possible new BTV serotype is a virus designated as SP vaccine-derived BTV, which was identified in 2014 in batches of sheeppox (SP) vaccine purchased in Israel.49 Sequence analyses of the genome segments 1, 4, 7 – 10 confirm that this virus is closely related to BTV-26. Like BTV-26, the virus was easily propagated in ECE and several mammal cell lines. A possible variant of SP vaccine- derived BTV (SPV BTV) was found in healthy sheep in Tunisia that were imported from Libya.170 The sequences of genome segments 4, 9, 10 of this new virus, designated BTV-Y TUN2017, have high nt sequence identity (99.3%, 96.9% and 91.1%, respectively) with SPV BTV. However, unlike the SPV BTV, attempts to isolate BTV-Y TUN2017 using ECE or several mammalian cell lines have been unsuccessful. The sequences of Seg-2 of this virus are only distantly related to other BTVs, including the small ruminant-adapted BTVs ( <62.3% nt identity), and serum from BTV-Y TUN2017 infected sheep failed to neutralize other BTV serotypes, suggesting that it is a novel serotype.
As the small ruminant- adapted BTVs cause subclinical or very mild infections and their presence currently does not lead to restrictions on animal movement or trade (like those associated with the presence of BTV 1 – 24), these viruses currently are not problematic to the livestock industries of countries in which they are endemic. However, the biological characteristics of novel and classical BTV serotypes are remarkably different,29, 281 and reassortment events between them could potentially result in progeny viruses with different properties such as the ability for contact transmission. Reassortment between classical and novel serotypes has been demonstrated to occur in vitro but has not been shown to date in animals or nature.232, 254, 321
Pathogenesis
The pathogenesis of BT reflects virus tropism and both direct cytopathic effect of the virus on vascular endothelium as well as the activities of proinflammatory and cytokine mediators produced by infected cells. Bluetongue virus first multiplies in the regional lymph nodes after virus inoculation by the bite of a midge before spreading to the rest of the body.22, 23, 24, 248 Viral replication occurs primarily in dendritic cells, macrophages and endothelial cells lining capillaries and small blood vessels.130, 165, 187, 216, 296, 336 Cytopathic changes in endothelial cells include degenerative and necrotic lesions such as cytoplasmic vesicles, nuclear and cytoplasmic hypertrophy, pyknosis and karyorrhexis. These changes, often accompanied by hyperplasia of the endothelium, lead to vascular occlusion, stasis and exudation, which eventually gives rise to hypoxia, oedema and haemorrhage as well as to secondary lesions in the overlying epithelium.165, 296
In a study of BTV infection in white-tailed deer it was concluded that viral-induced endothelial damage is the primary triggering mechanism for the disseminated intravascular coagulation that is responsible for the haemorrhagic diathesis characteristic of the disease in these animals.130 However, similar studies in sheep confirmed marked coagulopathy but without occurrence of disseminated intravascular coagulation, suggesting that the vascular injury in fulminant BT of sheep is analogous to that which occurs in human viral haemorrhagic fevers such as Ebola.186 Specifically, in these diseases, rather than direct virus-mediated endothelial injury, cytokine mediators released from virus-infected dendritic cells and macrophages cause endothelial cell retraction that results in vascular leakage and hypovolemic shock.70, 71, 72, 75, 76, 132, 186, 187, 271, 276
The severity of the secondary lesions in BTV-infected sheep is influenced by mechanical stress and abrasion, and severe lesions develop mainly in tissues exposed to the environment, such as the oral mucosa and skin of the coronary border of the hooves. There is probably a correlation between the distribution of the lesions and temperature gradients within the host because the most severe lesions occur in areas where the temperature is lower than that of circulating blood.296 There is also evidence for a highly selective involvement of endothelial cells in certain blood vessels (such as the pulmonary artery and those where the most severe lesions are found), the mechanism of which is not understood although it is abundantly clear from in vitro studies that endothelial cells from different levels of the pulmonary vascular system differ in their response to BTV infection. Exposure of affected animals to sunlight exacerbates the severity of the disease. The mortality rate can escalate dramatically when infected sheep are exposed to cold, wet conditions as often happens in late autumn.90
The innate immune response to BTV infection begins at the site of virus deposition in skin via the bite of an infected midge, and it is now clear that components of midge saliva impact this initial response.240 Infection of ruminants with most (but not all) serotypes of BTV leads to a highly blood cell-associated viraemia that may be prolonged but not persistent. Recovered animals are resistant to reinfection with the homologous virus serotype, which is the basis for vaccination strategies to prevent BTV infection and clinical disease in domestic livestock. Replication of BTV in target cells, notably mononuclear phagocytic cells (dendritic cells and macrophages) and endothelium, leads to the generation of the innate and adaptive immune responses that mediate both initial virus clearance and subsequent resistance to infection with the homologous virus serotype.53, 57, 73, 188, 262, 271, 330 The Type 1 interferon response is induced soon after infection, followed by both humoral and cellular immune responses. The non-structural NS3 and NS4 proteins of BTV have been identified as suppressors and/or agonists of the Type I interferon response.58, 258 Furthermore, there is recent evidence that BTV infection of follicular dendritic cells compromises transiently the host’s ability to develop an effective adaptive immune response.216
Following initial replication of the virus in lymphoid tissues and endothelial cells, BTV appears in the circulation three to six days after infection. The viraemia reaches a peak about seven to eight days after infection and accompanies or precedes a febrile reaction that normally lasts about four to eight days. In sheep the viraemia rarely persists for longer than 14 days7, 8, 36 and is usually present for six to eight days. Results that conflict with this statement were reported by early workers, but can probably be explained by natural reinfections with different serotypes that were unknown at that time. Biphasic viraemia has been observed in BT-infected sheep with viraemic peaks at day 5 (±1) and day 10 (±2). It was attributed to the induction of high concentrations of interferon concurrent with the first viral peak, followed by production of antibodies and a subsequent reduction in viraemia at the second peak.99, 189, 243
Despite a robust immune response, viraemia in BTV-infected cattle is often prolonged for several weeks23, 36, 191, 192 and RNA-aemia is even more extended (several months). It was found that in vitro infection of bovine erythrocytes and non-replicating lymphocytes did not progress beyond adsorption after which virus particles persisted in invaginations of the cell membrane. This could explain both the prolonged viraemia and the absence of clinical disease.22, 46 It has also been demonstrated that infected ovine endothelial cells bind more BTV initially and produce more virus than their bovine counterpart,272 although this finding was not reproduced in more extensive studies with endothelial cells cultured from different levels of the pulmonary vasculature of sheep and cattle.
Bluetongue virus in the blood is primarily associated with erythrocytes and, to a lesser extent and particularly during acute infection, with the buffy-coat fraction,122, 172 while only a small fraction is found free in the plasma.22, 23, 63, 165 Panleukopenia preceding the viraemia and a febrile reaction is consistently found in BT.165 Changes in the packed cell volume and the concentration of aspartate transaminase and creatinase in the plasma have also been observed.96
Experimental infection of dogs was only successful in pregnant bitches, leading to abortions and death from severe pulmonary oedema. Viral nucleic acid was mainly detected in lung endothelial cells.48
In cattle there is evidence of a rapid accumulation of IgE antibodies directed against the virus after infection, which may result in immediate hypersensitivity reactions involving the release of mediators such as histamine, prostaglandins and thromboxane A211, 85 However, the importance of this phenomenon in disease expression in cattle naturally infected with BTV remains uncertain as cattle infected with especially virulent strains of the virus, such as European BTV-8, may develop clinical signs similar to those in sheep.64, 82, 196, 346
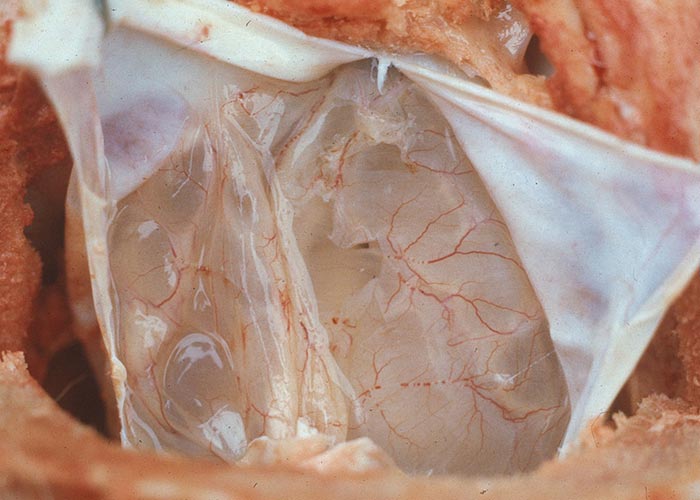
Abortion or malformation, particularly of the central nervous system (CNS), of lambs may follow vaccination of ewes with attenuated live vaccines during the first half of pregnancy.114, 237, 260, 282 Similarly, infection of bovine foetuses during the first trimester of pregnancy with laboratory-modified strains of BTV results in severe teratogenic defects in the central nervous system.20, 176, 179, 191, 193, 194, 261 The natural occurrence of similar congenital teratogenic defects in the progeny of unvaccinated animals was rare until the emergence of BTV serotype 8 in Northern Europe in 2006, when large numbers of ruminants were born with these same defects in the central nervous system (e.g. hydranencephaly) (Figures 4a and b) following infection of their dams in early gestation.196, 325, 343
Clinical signs
The extreme variability in the clinical manifestation of BT, not only between different ruminant species but also between different breeds of sheep, is a feature of the disease. Individual idiosyncrasies and environmental conditions affect the severity of the disease, as does the virulence of the infecting virus strain. Many infections in sheep are clinically inapparent and, even in fully susceptible animals, it is difficult to reproduce severe clinical signs under laboratory conditions where there is limited exposure to sunlight or ultraviolet light.
The course of the disease in sheep can vary from peracute to chronic, with a mortality rate of between 2 and 30 per cent. Peracute cases usually die within seven to nine days of infection, mainly as a result of lung oedema and eventual asphyxia. They may show very few signs of illness prior to death. In chronic cases death can result from secondary bacterial pneumonia and exhaustion, or recovery can be very protracted. Mild cases usually recover rapidly and completely.
The incubation period, following natural infection, is about seven days (judging by the length of the period during which clinical cases continue to appear after frost). Following experimental infection the incubation period is usually four to six days but may vary between 2 and 15 days. The first clinical sign is a rise in temperature over a period of 48 hours, reaching a peak of 41 to 42 °C. The febrile reaction usually lasts about six to eight days and its termination is determined by the course of the disease and the extent of secondary infection. Generally there is a fair correlation between the duration of the fever and the severity of the disease.90
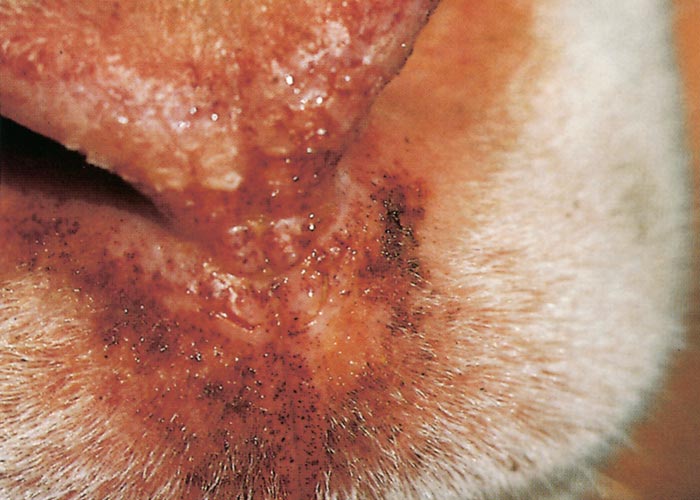
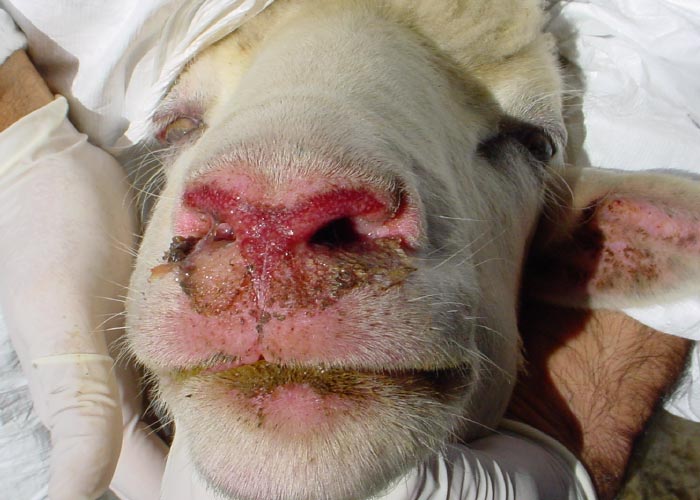
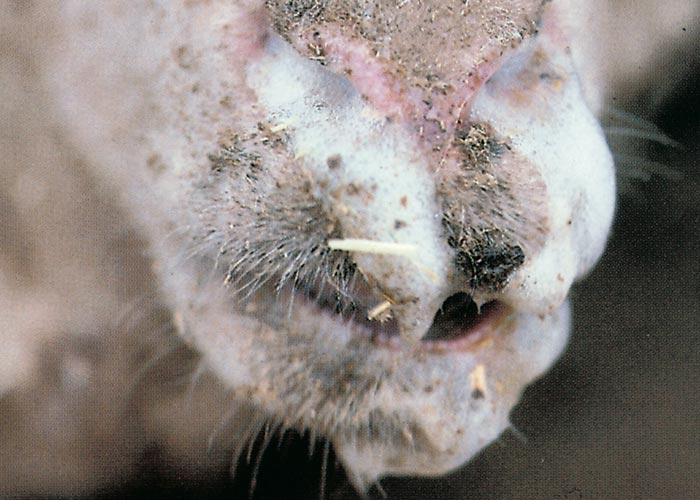
Other clinical signs may appear within one or two days of the onset of fever. Hyperaemia of the buccal and nasal mucosae usually occurs first and may also develop on more exposed parts of the skin around the muzzle and eyes and on the ears (Figure 5) increased salivation and lachrymation as well as a serous nasal discharge may soon follow. Licking movements of the tongue and smacking movements of the lips may result in froth formation at the corners of the lips.
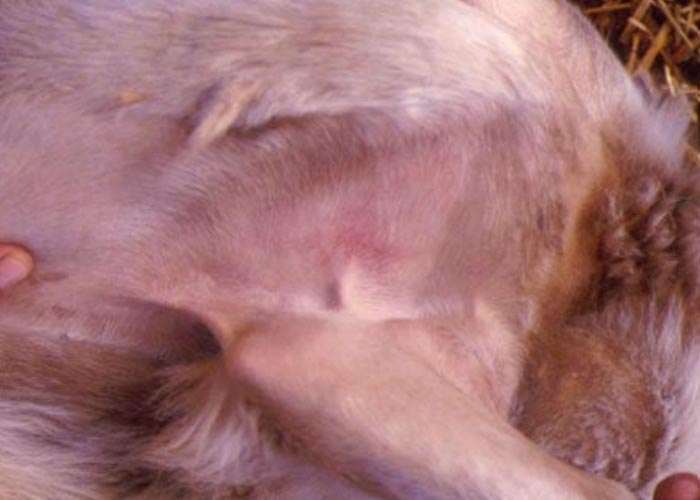
Oedema of the tongue, lips, face, eyelids and ears develop about 48 hours after the onset of fever. Submandibular oedema (Figure 6) may be marked and occasionally extends down the neck to the axillae. Petechiae may be observed on the muzzle, papillae of the lips and mucous membranes of the mouth and conjunctivae. At this stage mild cases may recover uneventfully.
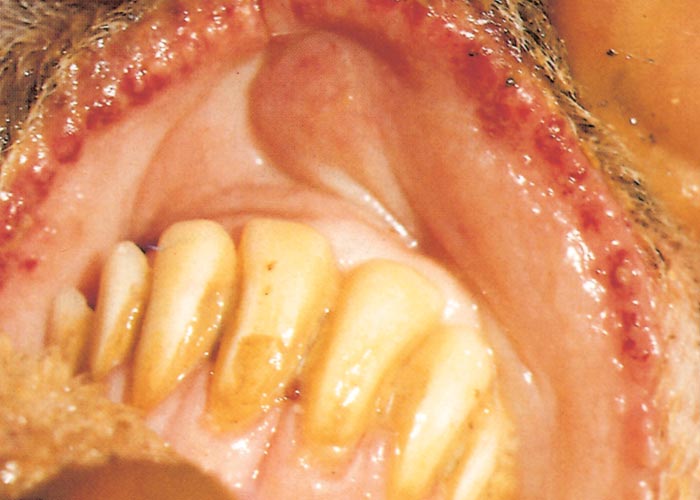
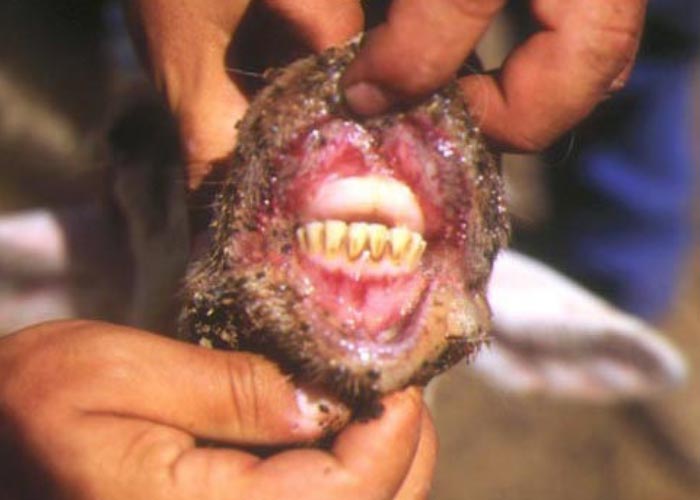
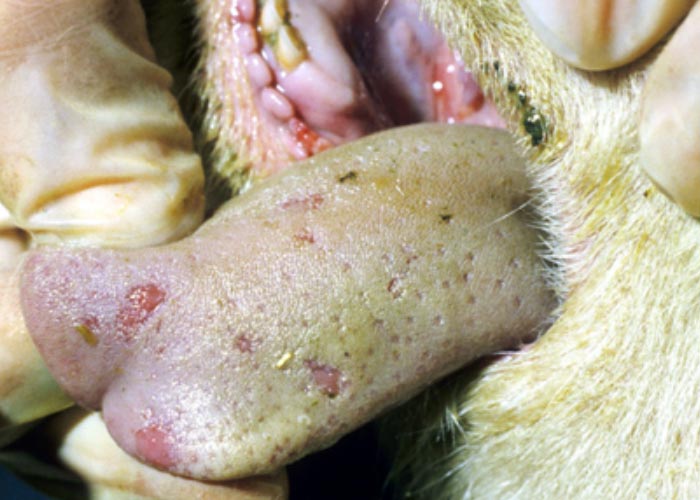
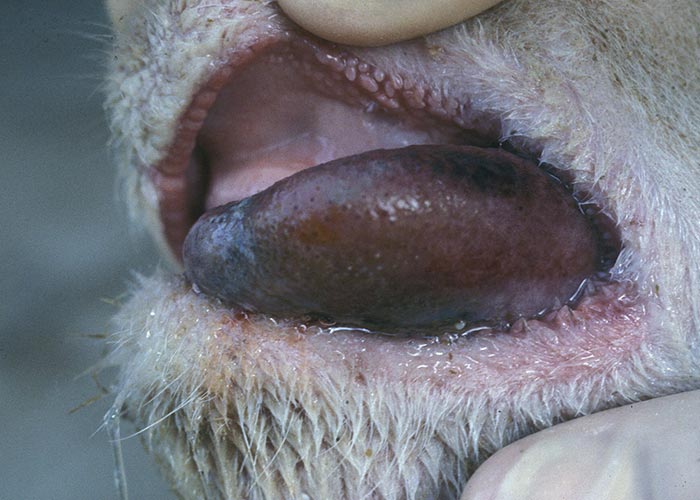
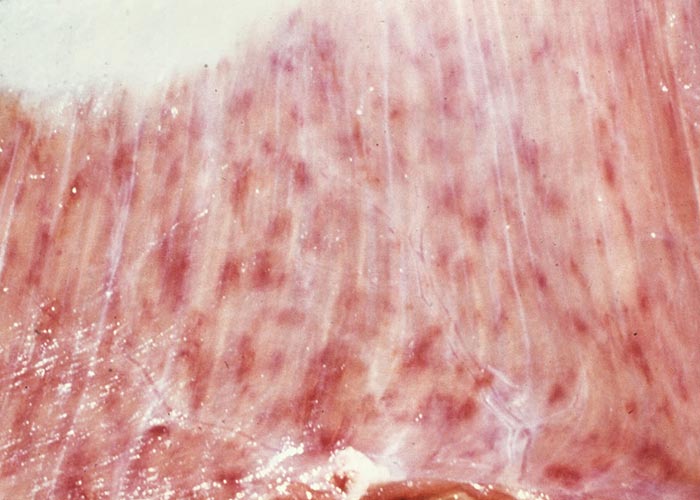
In severe cases the lesions progress and result in excoriations and erosions on the muzzle and nostrils and in the mouth (Figures 7 to 11) particularly at sites subject to friction, such as parts of the cheeks and tongue adjacent to the molar teeth and the lower lip opposite the corner incisors (Figures 12, 13a to d). Necrotizing mouth lesions result in foetid breath, which is sometimes the first clinical sign observed. They also induce pain, which may cause the animal to submerge its mouth and lips in drinking water for prolonged periods. Anorexia is common and may be exacerbated in some cases by severe swelling of the tongue, which may become cyanotic (‘blue tongue’) (Figure 14) and even protrude from the mouth. Progressive weakness and emaciation follows, accompanied by rumen stasis and occasionally haemorrhagic diarrhoea before death.
The watery nasal discharge frequently becomes mucopurulent and eventually forms crusts that interfere with breathing and cause panting. In peracute cases, severe oedema of the lungs leads to dyspnoea, frothing from the nostrils (Figure 15) and death by asphyxiation.
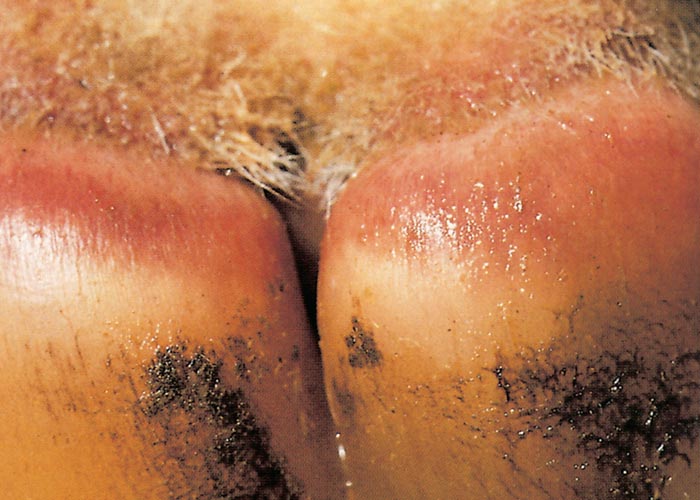
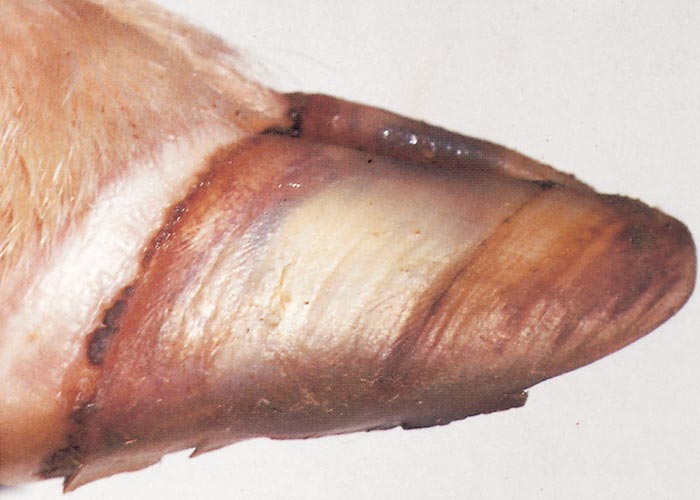
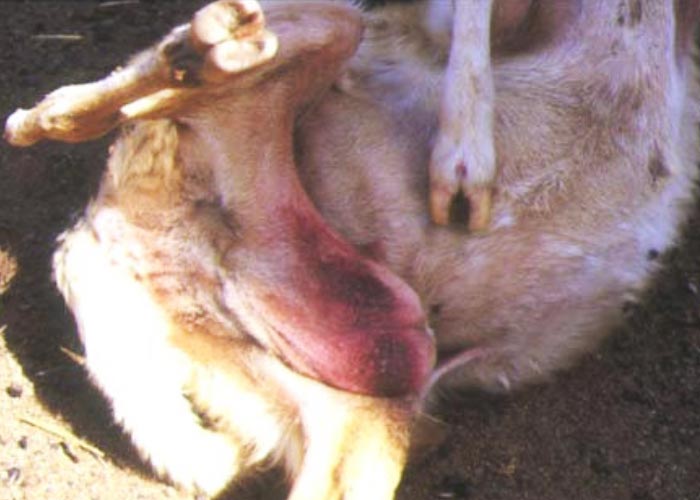
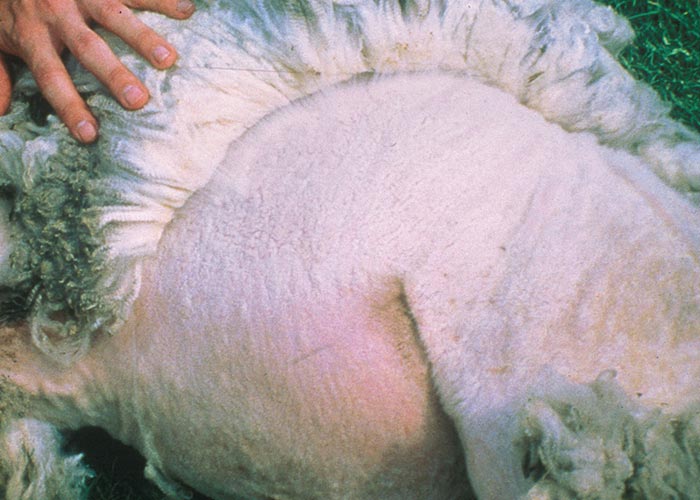
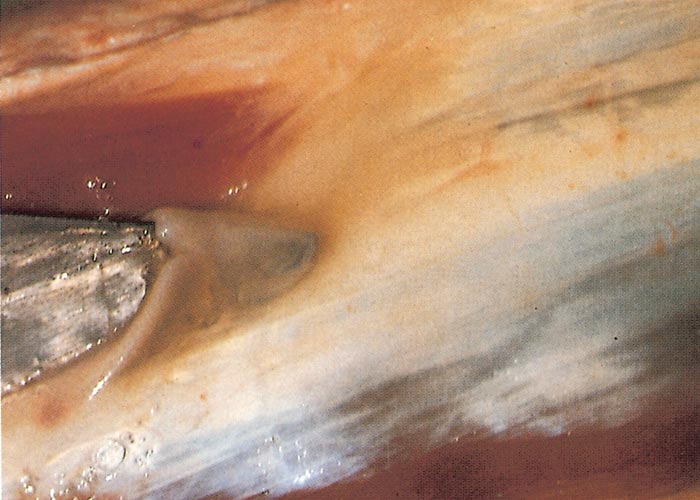
Foot lesions usually develop towards the end of the febrile reaction. Hyperaemia of the coronary bands and petechiae under the periople, which later become streaky in appearance as a result of haemorrhage into the fine medullary canals of the growing horn, give rise to a red zone or band in the horn of the hoof (Figure 16). As this lesion persists for some weeks after others have disappeared, it may constitute valuable evidence of BT during the recovery period. The lesion is most pronounced on the bulbs of the feet, and particularly of the lateral digits. The hind feet are most frequently affected. The feet are warm and painful, and affected animals are reluctant to move and often stand with arched backs or are recumbent. In affected sheep the gait is often stiff with varying degrees of lameness. Sometimes severely affected animals try to walk on their knees. In animals that recover, the bands of discoloration in the hooves grow out and a ‘break’ in the hoof (Figure 17) may develop (‘slipper formation’), with the old horn eventually sloughing off after three to four months. Inability to move and recumbency may also be exacerbated by emaciation and muscle lesions. Severe muscle degeneration and cachexia are sometimes seen in cases where buccal lesions are mild and the appetite undiminished.91 Degeneration and necrosis of skeletal muscles in the neck may lead to torticollis (Figure 18). Hyperaemia of the skin is usually most severe in those areas that are exposed to sunlight (such as, in Merino sheep, the face, ears and legs, which are not covered by wool), but may involve the whole body (Figures 19 to 21). In severely affected sheep, exanthema develops on the legs and other parts of the body subject to trauma. In such instances the slightest abrasion or handling may immediately result in extensive cutaneous haemorrhage, particularly in the groin and axilla (Figures 20 and 21). In most affected animals the wool fibres ‘break’ within the wool follicle (Figures 22 and 23) and the fleece may be shed three to six weeks later.90, 126 Normal new wool soon appears in recovered animals.
Clinical signs in cattle are rare and are usually limited to a transient febrile response, increased respiratory rate, increased lachrymation and salivation (Figure 24), stiffness and inflammatory changes in the skin.11, 155, 177, 178 Calves that were infected experimentally remained healthy throughout the experiment.189 In contrast, some cattle infected with BTV-8 during the recent European epidemic developed signs similar to those in BTV-infected sheep, with oral erosions/ulcers, conjunctivitis, nasal discharge, coronitis, and lesions of the mammary glands.64, 82, 196, 346
Recently, fatal cases of BT of camelids have been described in both Europe and the Americas. Affected llamas and alpacas developed a peracute disease course and quickly succumbed to fulminant pulmonary oedema, with few other signs or lesions at post-mortem.103, 222, 234
Pathology
The clinical pathology of BTV infection has been studied and characterized in white-tailed deer,131 and progressive prolongation of activated partial thromboplastin time and prothrombin time as well as progressive reduction of Factors VIII and XII plasma activities were observed. Haematologic changes included leukopenia, lymphopenia, and neutrophilia and low total plasma protein concentration. Mild to severe increases in serum aspartate transaminase activity were accompanied by more marked increases in creatinine kinase activity. These changes were associated with the development of disseminated intravascular coagulation, which was regarded as being important in the development of haemorrhagic diathesis — a characteristic of BT in white-tailed deer. Interestingly, however, disseminated intravascular coagulation did not occur in sheep inoculated with a highly virulent South African strain of BTV- 4, although the inoculated sheep did have marked coagulopathy and developed severe and fatal disease.186 Indeed, there is compelling evidence that the pathogenesis of fulminant BT in domestic livestock is typical of viral haemorrhagic fevers in that it reflects cytokine-mediated vascular leakage and hypovolemic shock, with or without accompanying disseminated intravascular coagulation.187 Many of the gross pathological changes, described in detail by early workers such as Spreull,294 Theiler,312 Thomas and Neitz,315 and Moulton,223 correspond largely with the clinical signs.
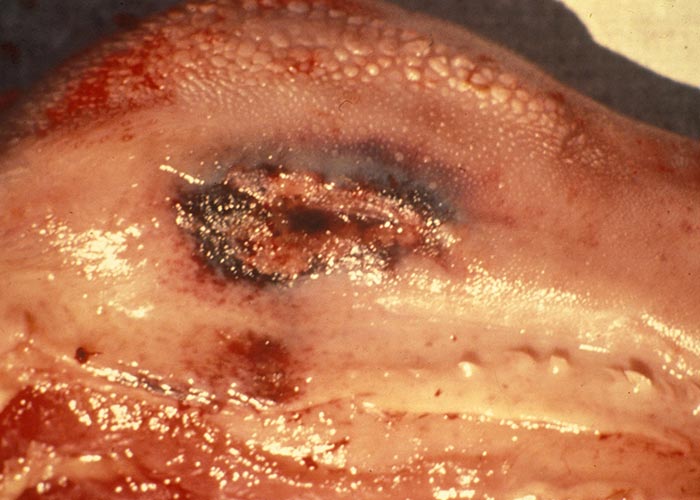
Oral lesions initially consist of hyperaemia, oedema, cyanosis and haemorrhage of the mucous membrane (Figure 14). Destruction of epithelial cells gives rise to excoriations and erosions on the inside of the lips, dental pad, cheeks and tongue (Figures 10 to 14). Many of these lesions are transient and usually disappear within days. Secondary bacterial infection is often responsible for the diphtheric necrosis of the erosions.28, 221 Microscopically, there is mononuclear cell infiltration and degeneration and necrosis of epithelial cells in which large acidophilic intracytoplasmic masses accumulate.
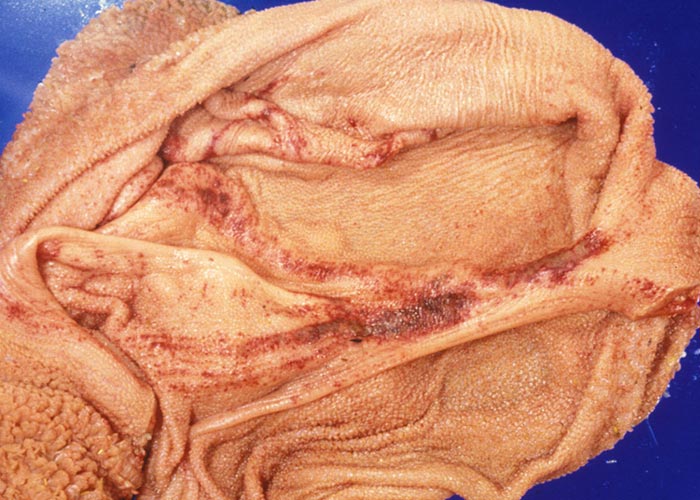
Hyperaemia, petechiation and erosions of the mucosa of the forestomachs, particularly of the papillae, rumenal pillars, reticular folds and oesophageal groove are common (Figures 25 and 26). Oedema and haemorrhages are often present in the submucosa, especially in the vicinity of the pylorus and the anterior third of the omasum. Lesions in the small intestines vary from mild localized areas of hyperaemia to severe catarrhal or haemorrhagic lesions extending into the large intestine.90
Hyperaemia, oedema and petechiae occur in the mucosae of the nasal cavity, pharynx and trachea, as well as in the lungs. Severe hyperaemia and oedema of the lungs accompanied by copious amounts of froth in the trachea and hydrothorax occur, especially in acute fatal cases. Aspiration pneumonia may occasionally be found.174
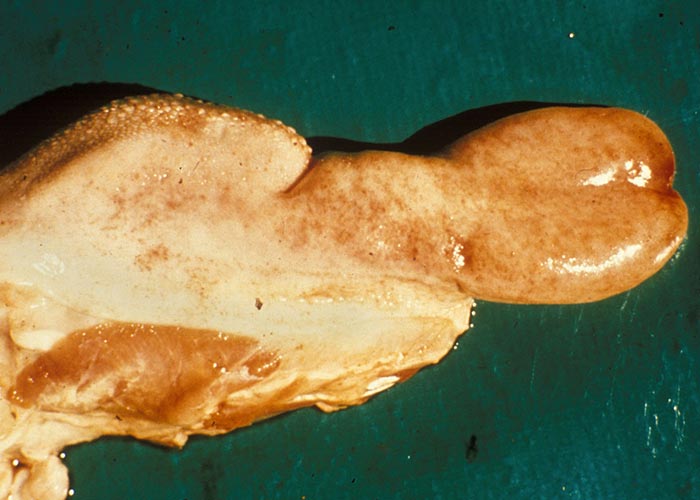
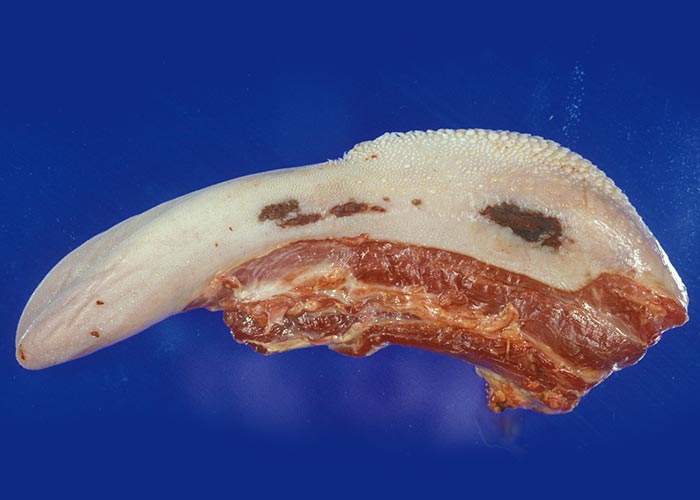
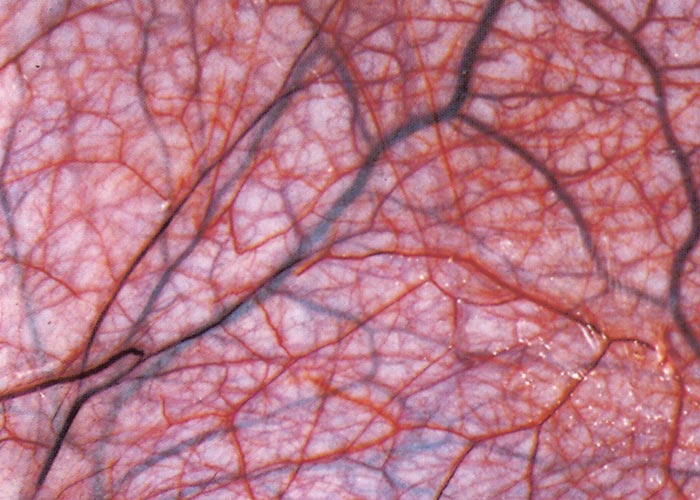
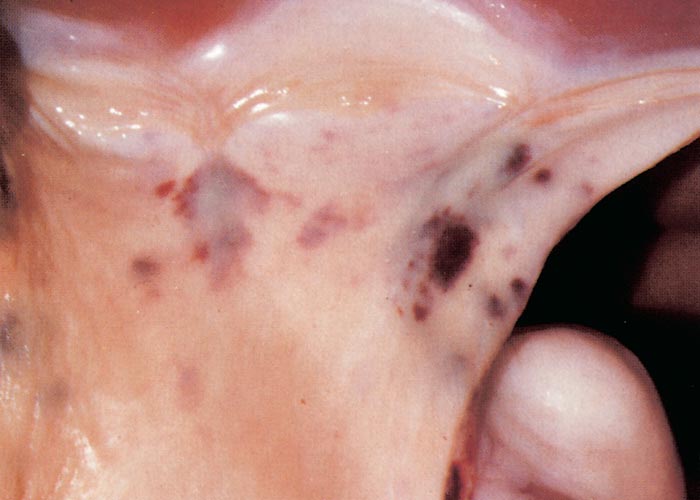
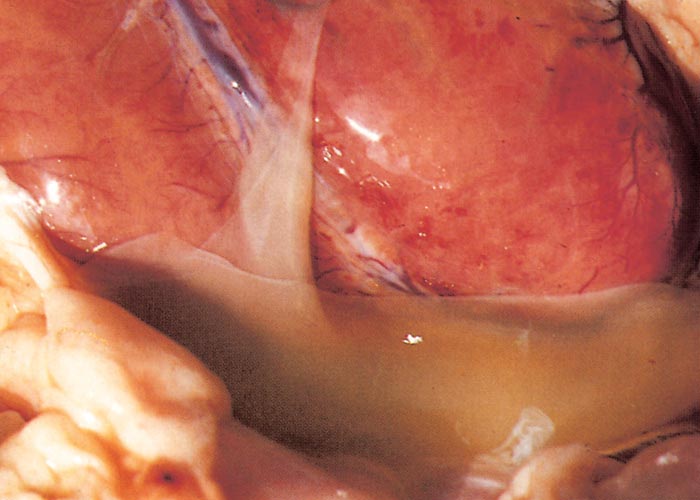
The widespread, but often localized, occurrence of hyperaemia, oedema and haemorrhages is evidence of damage to the vascular system, likely as a consequence of the activity of proinflammatory and vasoactive mediators in conjunction with virus infection of endothelium.72, 75, 132, 186, 187, 276 Petechiae, ecchymoses or even larger haemorrhages in the tunica media of the pulmonary artery near its base are characteristic of BT (Figures 27 and 28). These lesions are sometimes described as being pathognomonic for BT, but they have also been seen on rare occasions in other infections such as Rift Valley fever, heartwater and pulpy kidney disease in sheep. These haemorrhages may be difficult to see and it may be necessary to stretch the pulmonary artery and to hold it against the light. Epicardial and endocardial haemorrhages (Figure 29), as well as focal necrosis (c. 0,5 to 1,0 mm in diameter) of the papillary muscle are sometimes found in the left ventricle. Hydropericardium may be present (Figure 29), as may accumulation of oedema fluid around the nuchal ligament and in the fascial planes of the abdominal musculature (Figure 30).
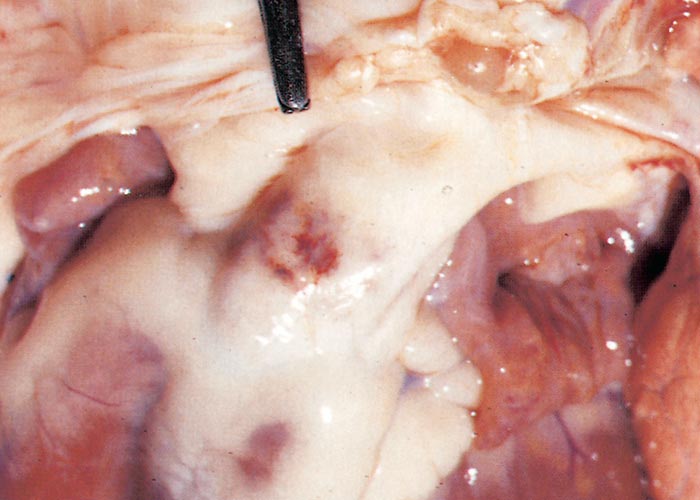
Figure 27 Haemorrhages in the wall of the pulmonary artery. (Courtesy of the Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, South Africa)
Pharyngeal, cervical and thoracic lymph nodes are commonly swollen and oedematous and the spleen may be slightly enlarged with subcapsular haemorrhages.
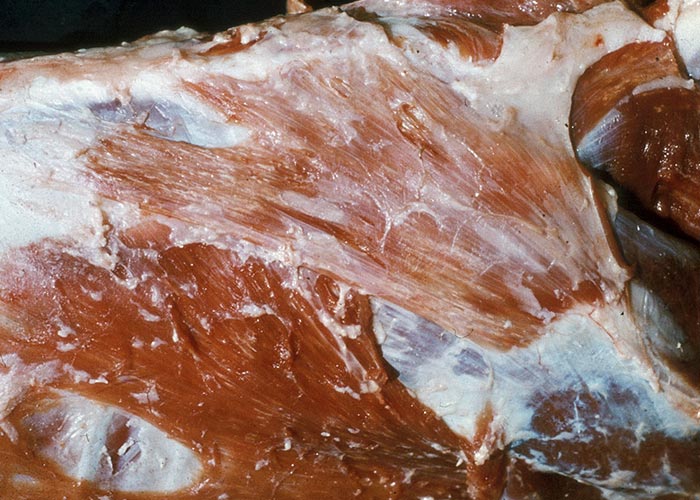
From an economic point of view, in addition to loss of wool, the pathological changes in the skeletal musculature are probably the most important lesion in BT, as they are usually associated with gross loss of condition, weakness, and a slow, protracted recovery period. Macroscopically, the muscle lesions comprise petechiae, ecchymoses and gelatinous oedema of the intermuscular connective tissues (Figures 30 and 31), particularly in the neck and the dorsal thoracic region.
Small localized areas of degeneration and necrosis appear as greyish-white foci. Extensive degeneration and necrosis involving entire muscles is occasionally seen (Figures 32 and 33). Microscopically, hyaline degeneration, necrosis and mineralization of individual muscle fibres occur together with intermuscular oedema and haemorrhage. Infiltration by neutrophils, macrophages and lymphocytes as well as evidence of regeneration of muscle fibres are present in the lesions of acute cases. In sheep that develop torticollis, the damage is most extensive on the side of the neck towards which the head is turned. At one time it was held that fibrous replacement was the cause of torticollis315 but more recent observations suggest that torticollis occurs in some cases in which there is no evidence of fibrosis.62
Diagnosis
A presumptive diagnosis of BT based on clinical signs, lesions and epidemiological assessment requires laboratory confirmation.1 Rapid detection, reliable identification and serotyping of BTV are all crucial in designing and implementing appropriate control measures, and to limiting the spread of infection.
Samples to be examined in the laboratory for virus detection should include blood (10 to 20 ml) collected as early as possible during the febrile reaction in anticoagulants such as heparin, sodium citrate, EDTA or OCG;90 in fatal cases, specimens of spleen, lymph node, and lung are useful for virus detection, and from foetuses, specimens of brain and spleen. These tissues should be collected as soon as possible after death and kept at 4°C.243 For transport, these samples should be kept on ice. Whole blood samples can be stored at 4°C for a long time. Red blood cells washed and resuspended in phosphate- buffered saline and stored at 4°C can be used for virus detection.9 Alternatively they may be frozen at −70°C in 10% dimethyl sulphoxide for storage prior to testing.
Various techniques have been used to detect the presence of infectious virus, viral antigen, or viral RNA in infected samples. The most commonly used are: real time RT-PCR, conventional RT-PCR with agarose gel electrophoresis (to identify specific amplicons), capture ELISA, viral isolation from embryonated chicken eggs (ECE), and viral isolation from insect (e.g. Kc and C6/36 cells derived from Culicoides sonorensis and Aedes albopictus, respectively) and/or mammal cells. Traditional RT-PCR and real time RT-PCR assays provide a versatile system able to give information on virus serogroup and serotype within a few hours. They are also highly sensitive and capable of detecting very low concentrations of viral RNA. In addition, the real time PCR assay is able to quantify the viral genome. Among the several existing RT-PCRs, real time RT-PCR assays based on detection and quantitation of the conserved and group-specific S10 gene (which encodes NS3) are now the most widely used14 as they are able to detect both traditional and small ruminant-adapted BTVs. Blood samples positive by this assay then can be subjected to serotype-specific real time RT-PCR assays to determine the specific virus serotype(s) involved.181, 205 Due to all these advantages, in most laboratories these new techniques are preferred to classical viral detection techniques, which require three to four weeks to be completed. However, RT-PCRs are unable to distinguish whether the RNA detected in the animal is contained in an infectious virus or merely RNA from a degraded particle no longer able to infect the vector. In other words, the mere identification of BTV nucleic acid by real time RT-PCR is not proof of disease causality, as BTV nucleic acid persists in the blood (RNA-aemia) and tissues (e.g. spleen) of infected animals for several months, long after clearance of infectious virus.74, 191, 192
The classical virus isolation techniques (inoculation of ECE or mammalian or insect cells) remain the only viable method to confirm the presence of infectious virus in an animal.
Bluetongue virus can most readily be isolated by the intravascular inoculation of 10- to 12-day-old ECE or by infection of susceptible cell cultures with processed blood or clarified tissue suspensions.97 Primary isolation can only be achieved with difficulty by intracerebral inoculation of new-born mice or in monolayer cell cultures (BHK-21, mouse L-cells, Vero, Kc or C6/36 cells). However, after a single passage in ECE or insect cells (Kc) the virus can readily be cultivated in mouse brain or in a variety of cell cultures.
Isolated BTV is now typically characterized by sequence analysis, increasingly by whole genome sequence analyses17, 29, 43, 123, 169, 182, 183, 231, 236, 281, 283, 347 that can also facilitate analyses to map the origins, movement and spread of individual virus strains. In the past, techniques like complement fixation, ELISA and immunofluorescence or immunoperoxidase staining that detect group-specific antigens were used to distinguish BTV from other orbiviruses.33, 34
Several techniques can be used to detect the presence of BTV-specific antibodies in either infected or vaccinated animals. The methods to detect serogroup- and serotype-specific antibodies in serum and milk samples of infected or vaccinated animals most commonly used are: competitive ELISA (c-ELISA), milk ELISA and serum-virus neutralisation (SN). Previously, a complement fixation test using antigen prepared from infected mouse brains160, 318 was used, and this was later replaced by the gel immunodiffusion test.245 Cross-reactions between different orbiviruses were a significant problem with these early serological techniques, but less so with more recently developed c-ELISA.52 The OIE has prescribed the c-ELISA test for serological diagnosis of BTV infection for international trade.14 Over time, the accuracy of this assay has been progressively improved through the use of antigens obtained from recombinant structural proteins of BTV. The c-ELISA incorporates a monoclonal antibody to an epitope on the core protein VP7 that is common to all BTV serotypes; thus, the assay detects the presence of antibodies to all BTV serotypes. The technique is highly sensitive and specific as the monoclonal antibody used detects an epitope common to BTV serogroup viruses that is not present in other orbiviruses. The test is relatively inexpensive, commercially available, and the preferred diagnostic assay. However, the available c-ELISA tests currently do not discriminate BTV-infected from vaccinated animals (DIVA).
Milk ELISAs are successfully used to evaluate BTV-infection in a herd. Since these tests are applied on bulk milk samples, they have the advantage of testing groups of animals in one step.163 Its specificity was recently improved by precipitating the milk protein prior to carrying out the ELISA.56
As c-ELISA does not identify which BTV serotype induced virus-specific antibodies in any given animal, other assays are required to determine serotype-specificity. Type-specific antibodies are best detected by means of virus neutralization tests, such as SN, plaque reduction, or by haemagglutination inhibition tests.173 However, because of cross-reactions between serotypes and heterotypic antibody responses, the significance of type-specific antibody titres in animals exposed to multiple serotypes may be difficult to interpret and require skilled personnel.
Differential diagnosis
In southern Africa, the clinical signs of acute BT in sheep should be differentiated from early signs of hepatogenous photosensitivity caused by a variety of plant and mycotoxin poisonings.159 In areas where the disease does not normally occur, it should be differentiated from vesicular diseases such as foot-and-mouth disease. The clinical signs could also be confused potentially with those seen in polyarthritis, foot rot and white muscle disease, orf (contagious pustular dermatitis), acute haemonchosis or liver fluke (with depression and submandibular oedema), peste des petits ruminants, laminitis and clostridial diseases.338 In sheep that die after the typical lesions of BT (oedema of the lips and tongue, and mouth and tongue erosions) have regressed, it may be difficult to differentiate BT from diseases such as heartwater and pulpy kidney disease.317
In cattle, clinical BT can easily be confused with clinical signs and lesions of epizootic haemorrhagic disease of deer and Ibaraki disease. In this species differential diagnosis should also include malignant catarrhal fever, pododermatitis, infectious bovine rhinotracheitis, bovine viral diarrhoea, bovine papular stomatitis, bovine herpesvirus 2 infection (bovine herpes mammillitis) and parainfluenza-3 infection.210, 279, 338
The teratological defects (eg. hydranencephaly and porencephaly) of the central nervous system of the foetus in sheep and cattle caused by European BTV-8 and modified BTV strains are similar to the defects caused by teratogenic bunyaviruses (Aino, Akabane, Cache Valley, Schmallenberg viruses) as well as Wesselsbron disease and palyam viruses.187, 193, 194
Control
Strategies for the control of BT differ according to whether outbreaks of the disease occur in endemic regions or in areas where the disease is not usually present.190 In the latter case the usual goal is eradication, whereas in endemic areas attempts can only be made to limit the occurrence of the disease and its economic impact. However, it is increasingly obvious that eradication of a vector-borne disease like BT is unrealistic, thus control of outbreaks, if attempted, is usually based on animal movement restrictions and vaccination. Ongoing surveillance is required to monitor the effectiveness of any control programme. In any attempt to control arthropod-borne diseases three factors should be taken into account: possible virus reservoirs, the vector, and the target animal. Where possible control actions should be integrated88 and directed towards protecting animals from insect attack and/or BTV infection.
Vector control
Bluetongue virus has now been isolated from many species of Culicoides, and the Culicoides species involved vary by region of the world.80, 105, 108, 153, 185, 211, 301, 309, 339 Elimination of Culicoides midges from the environment is generally impractical, particularly in extensive pastoral settings. Air-borne transport of infected Culicoides midges over long distances cannot be prevented and is likely to occur. The important determinant of virus spread is whether or not the midges resident in any invaded area can then acquire, spread and maintain the incursive BTV – clearly this was the case during the recent epidemic in Europe where strains of BTV vectored by C. imicola in Asia and Africa were introduced into Europe and then successfully transmitted by native C. imicola and species of midges within the Obsoletus complex (involving at least C. obsoletus and C. scoticus).
Control of Culicoides populations, or, alternatively, protection of target animals from contact with them, can make a significant contribution towards control of the disease. Methods used to control Culicoides include the use of insecticides, larvicides, and sterilization of males by irradiation,124 as well as the subcutaneous administration of ivermectin to cattle in an attempt to break the primary cycle before the infection spills over into sheep.297 Protecting sheep from exposure to Culicoides is a more practical approach and can be achieved by avoiding low-lying wet pastures, shearing in early summer to allow some wool growth before the onset of the BT season in late summer, and even the use of insect repellents.88 Screening of access points (eg. windows, doors) of stables using fine mesh capable of excluding midges, or coarser mesh impregnated with a suitable insecticide such as a synthetic pyrethroid can be used to further reduce the attack rate.215 Keeping cattle in close proximity to sheep is also effective in some areas, apparently because C. imicola, the most prevalent vector in South Africa, has a preference for cattle.229 The overall efficacy of many of these strategies is uncertain as the endo/exophagy behaviour and circadian host-seeking activity vary remarkably between individual midge species. Repellents are difficult to apply and typically have only a short duration of efficacy.190
Immunization
Vaccination remains the most effective and practical control measure against BT in endemic regions, and also to control BTV incursions into previously BT-free areas. Effective vaccination requires products that rapidly induce protection against the specific virus serotype or serotypes, especially in incursions. Vaccination of livestock in endemic areas should induce long- lasting and broad protection against all relevant virus serotypes. The main objectives of BT vaccination strategies are to (i) prevent clinical disease, (ii) reduce the spread of BTV, (iii) eradicate BT from the country or the region, and (iv) permit the safe movement of susceptible animals between BT-affected and BT-free zones.
In endemic African regions,105 a wide variety of domestic and wild ruminants (including cattle, goats and various species of antelope) can act as sources of BTV. Immunization of cattle should therefore theoretically help to limit the incidence of BT, but the presence and ubiquitous distribution of other reservoirs, both known and unknown, makes eradication of the disease impossible.
After recovery from natural infection, animals have a solid, life-long immunity to the homologous serotype but only partial or no protection against heterologous types. Protective immunity is generally associated with the presence of type-specific neutralizing antibodies that may persist for years, but it is not associated with the group-specific antibodies that are detected in standard serological tests (e.g. c ELISA). However, infection or immunization with more than one virus type usually results in protection against a wider range of serotypes, even types against which no neutralizing antibodies are present. This suggests that cellular immunity may contribute as it is less type-specific than is the humoral response.151, 152, 154
A wide variety of vaccine types have been developed for prophylactic immunization of ungulates against BTV, including inactivated, live-attenuated, recombinant and a variety of engineered molecular constructs.50, 55, 94, 190, 191, 208, 230 Currently, live-attenuated and inactivated constructs remain the only BTV vaccines produced commercially. These inactivated and live-attenuated vaccines are available in only some regions of the world.
Vaccines comprised of attenuated virus strains are highly effective, especially in epidemic situations where only one serotype of BTV is involved. Historically, vaccine strains were attenuated by serial passage in embryonated chicken eggs. These vaccine strains, now grown in BHK-21 cells, have been in use for 40 years.126 In endemic areas such as South Africa, the situation is complicated by the existence of multiple serotypes. This necessitates the use of polyvalent vaccines with attendant problems resulting from interference between virus strains, differences in immunogenicity and growth rates between various strains, as well as differences in the response of individual animals to the components of such vaccines. To overcome these problems, three pentavalent vaccines have been developed in South Africa. They are administered to sheep at three-week intervals and repeated annually. Although 21 BTV serotypes have so far been shown to occur in South Africa (types 17, 20 and 21 have not yet been isolated) only 15 serotypes are included in the present vaccine. Types 15, 16, 18, 22, 23 and 24 are not included because of their low pathogenicity for sheep and low prevalence in Culicoides.
It is anticipated that types 16 and 24 may be included in future on the basis of their increased prevalence, whilst type 7 may be omitted because of its low pathogenicity for sheep. After two or three annual immunizations, most sheep are immune to all the serotypes in the vaccine. Experience in the field has shown that the three pentavalent vaccines are far more effective when administered properly than are previous BT vaccines, which at times contained as many as 14 serotypes in a single dose.
In addition to the inconvenience of repetitive inoculations of a multivalent vaccine there are some risks involved in the use of live attenuated vaccines and some important precautions must be observed when using them. Pregnant ewes should not be immunized during the first half of pregnancy, as brain defects may result in the foetus.95 Secondly, because lambs born to immune ewes usually acquire effective colostral immunity that may last up to six months,126 during which time lambs are refractory to immunization, losses from BT may occur in spring-born lambs during the following autumn. There are also indications of temporary infertility in both ewes and rams vaccinated for the first time, necessitating immunization well before or after the mating season.8 However, this should not pose a problem if sheep are vaccinated before they reach breeding age.
It is increasingly clear that there are serious potential complications to the use of live attenuated vaccines. It is now well established that these viruses can be acquired and spread by insect vectors and reassort their genes with other vaccine or field viruses to create genetically novel reassortants with potentially deleterious properties (e.g. reversion to virulence, transplacental transmission etc.).208, 231, 233, 236, 279 In turn, these reassortant viruses are then spread by vector midges in the same manner as authentic “field strains” of the virus. The fact that animals vaccinated with live vaccines cannot be distinguished serologically from those recovered from disease poses a real problem in the international regulation of animal movement for those countries normally free from the disease.
Inactivated vaccines also suffer from the fact that vaccinated animals cannot be distinguished serologically from naturally infected animals (DIVA). However, inactivated vaccines are safe and if produced properly, they can be highly efficacious, as shown during the recent European epidemic.16 Their inherent potential disadvantages include high costs of production as vaccination requires large amounts of antigen, and the need for booster immunizations as inactivated vaccines generally induce a relatively transient immunity.
To overcome these problems, much research on new generation vaccines has been carried out during recent years aimed at the development of effective inactivated or subunit vaccines, or of constructing recombinant viruses that would combine the advantages of live vaccines with the safety of inactivated products.55, 94, 190, 191, 208, 230, 320
The demonstration that VP2 can be isolated and used to induce protective immunity140 paved the way for various studies in which the gene (segment 2) encoding VP2 was cloned, sequenced and expressed using a variety of vectors.138, 146 Again, the problem of producing sufficient quantities of the protein precluded its use as a subunit vaccine. In addition, it was soon discovered that other viral proteins also played a role in eliciting a protective immune response. A variety of recombinant vaccines have been produced and partially characterized, although none have yet been developed commercially. These include baculovirus expressed virus-like particles, recombinant VP2 expressed in plants and by various heterologous expression vectors including pox and adeno viruses, and various genetically modified or deleted BTVs that are replication defective.
References
- AFSHAR, A., 1994. Bluetongue: laboratory diagnosis. Comparative Immunology, Microbiology and Infectious Diseases, 17, 221–242.
- AFSHAR, A., ANDERSON, J., EATON, B.T. & GUSTAFSON, G.A., 1991. Serological diagnosis of bluetongue by blocking or competitive ELISA by four laboratories. Journal of Veterinary Diagnostic Investigation, 3, 255–257.
- AGREN, E.C., BURGIN, L., STERNBERG LEVERIN, S., GLOSTER, J., ELVANDER, M., 2010. Possible means of introduction of bluetongue virus serotype 8 (BTV-8) to Sweden in August 2008: comparison to results from two models for atmospheric transport of Culicoides vector. Veterinary Record, 167, 484–488.
- AKITA, G.Y., IANCONESCU, M., MACLACHLAN, N.J., OSBURN, B.I. & GREENE, R.T., 1994. Bluetongue disease in dogs associated with contaminated vaccine. The Veterinary Record, 134, 283.
- ALEXANDER, K.A., MACLACHLAN, N.J., KAT, P.W., HOUSE, C., O’BRIEN, S.J., LERCHE, N.W., SAWYER, M., FRANK, L.G., HOLEKAMP, K., SMALE, L., MCNUTT, J.W., LAURENSON, M.K., MILLS, M.G.L. & OSBURN, B.I., 1994. Evidence of natural bluetongue virus infection among African carnivores. American Journal of Tropical Medicine and Hygiene, 51, 568–576.
- ALEXANDER, R.A., 1947. The propagation of bluetongue virus in the developing chick embryo with particular reference to the temperature of incubation. Onderstepoort Journal of Veterinary Science and Animal Industry, 22, 7–26.
- ALEXANDER, R.A., 1959. Bluetongue as an international problem. Bulletin de L’Office International des Epizoties, 51, 432–439.
- ALEXANDER, R.A., HAIG, D.A. & ADELAAR, T.F., 1947. The attenuation of bluetongue virus by serial passage through fertile eggs. Onderstepoort Journal of Veterinary Science and Animal Industry, 21, 231–241.
- ALSTAD, A.D., BURGER, D., FRANK, F.W., MCCAIN, C.S., MOLL, T. & STAUBER, E.S., 1977. Localization of bluetongue virus in red blood cells of experimentally infected sheep. Proceedings of the Twentieth Meeting of the American Association of Veterinary Laboratory Diagnosticians, 273–290.
- ANDERSON, G.A., PHILLIPS, D.L., WALDVOGEL, A.S. & OSBURN, B.I., 1989. Detection of bluetongue virus in bovine fetuses using the avidin-biotin complex immunoperoxidase method. Journal of Veterinary Diagnostic Investigation, 1, 45–49.
- ANDERSON, G.A., STOTT, J.L., GERSHWIN, L.J. & OSBURN, B.I., 1985. Subclinical and clinical bluetongue disease in cattle: Clinical, pathological and pathogenic considerations. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss, 103-107.
- ANDREW, M., WHITELEY, P., JANARDHANA, V., LOBATO, Z., GOULD, A. & COUPAR, B., 1995. Antigen specificity of the ovine cytotoxic T lymphocyte response to bluetongue virus. Veterinary Immunology and Immunopathology, 47, 311–322.
- ANDREWES, C. & PEREIRA, H.G., 1967. Viruses of Vertebrates. 2nd Edition. London: Baillière, Tindal and Cox.
- ANONYMOUS., 2014. Bluetongue. Chapter 2.1.3, In: The Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. Office International des Epizooties, Paris.
- ARADAIB, I.E., SCHORE, C.E., CULLOR, J.S. & OSBURN, B.I., 1998. A nested PCR for detection of North American isolates of bluetongue virus based on NS1 genome dequence analysis of BTV-17. Veterinary Microbiology, 59, 99–108.
- BAETZA, H.J., 2014. Eradication of bluetongue disease in Germany by vaccination. Veterinary Immunology Immunopathology, 158, 116-119.
- BALASURIYA, U.B., NADLER, S.A., WILSON, W.C., PRITCHARD, L.I., SMYTHE, A.B., SAVINI, G., MONACO, F., De SANTIS, P., ZHANG, N., TABACHNICK, W.J. & MACLACHLAN, N.J., 2008. The NS3 proteins of global strains of bluetongue virus evolve into regional topotypes through negative (purifying) selection. Veterinary Microbiology, 126, 91-100.
- BANSAL, O.B., STOKES, A., BANSAL, A., BISHOP, D. & ROY, P., 1998. Membrane organization of bluetongue virus nonstructural glycoprotein NS3. Journal of Virology, 72, 3362–3369.
- BARBER, T.L. & JOCHIM, M.M., 1985. Bluetongue and Related Orbiviruses. Progress in Clinical and Biological Research. New York: Alan R. Liss,178.
- BARNARD, B.J.H. & PIENAAR, J.G., 1976. Bluetongue virus as a cause of hydranencephaly in cattle. Onderstepoort Journal of Veterinary Research, 43, 155–157.
- BARNARD, B.J.H., 1997. Antibodies against some viruses of domestic animals in southern African wild animals. Onderstepoort Journal of Veterinary Research, 64, 95–110.
- BARRATT-BOYES, S.M. & MACLACHLAN, N.J., 1994. Dynamics of viral spread in bluetongue virus infected calves. Veterinary Microbiology, 40, 361-371.
- BARRATT-BOYES, S.M. & MACLACHLAN, N.J., 1995. Pathogenesis of bluetongue virus infection of cattle. Journal of Veterinary Medical Association, 206, 1322-1329.
- BARRATT-BOYES, S.M., ROSSITTO, P.V., TAYLOR, B.C., ELLIS, J.A. & MACLACHLAN, N.J., 1995. Response of the regional lymph node to bluetongue virus infection in calves. Veterinary Immunology Immunopathology, 45, 73-84.
- BATTEN, C., DARPEL, K., HENSTOCK, M., FAY, P., VERONESI, E., GUBBINS, S., GRAVES, S., FROST, L. & OURA, C., 2014. Evidence for transmission of bluetongue virus serotype 26 through direct contact. PLoS One, 9, e96049.
- BATTEN, C., HENSTOCK, M.R., STEEDMAN, H.M., WADDINGTON, S., EDWARDS, L. & OURA, C.A., 2013. Bluetongue virus serotype 26: infection kinetics, pathogenesis and possible contact transmission in goats. Veterinary Microbiology, 162, 62-67.
- BATTEN, C.A., HENSTOCK, M.R., BIN-TARIF, A., STEEDMAN, H.M., WADDINGTON, S., EDWARDS, L. & OURA, C.A., 2012. Bluetongue virus serotype 26: infection kinetics and pathogenesis in Dorset poll sheep. Veterinary Microbiology, 157, 119-124.
- BEKKER, J.G., DE KOCK, G. & QUINLAN, J.B., 1934. The occurrence and identification of bluetongue in cattle: The so-called pseudo foot-and-mouth disease in South Africa. Onderstepoort Journal of Veterinary Science, 2, 393–507.
- BELBIS, G., ZIENTARA, S., BREARD, E., SAILLEAU, C., CAIGNARD, G., VITOUR, D. & ATTOUI, H., 2017. Bluetongue virus: From BTV-1 to BTV-27. Advances Virus Research 99, 161-197.
- BELHOUCHET, M., MOHD, J.F., FIRTH, A.E., GRIMES, J.M., MERTENS, P.P. & ATTOUI, H., 2011. Detection of a fourth orbivirus non-structural protein. PLoS One, 6, e25697.
- BELLIS, G.A., GIBSON, D.S., POLKINGHORNE, I.G., JOHNSON, S.J. & FLANAGAN, M., 1994. Infection of Culicoides brevitarsis and C. wadai (Diptera: Ceratopogonidae) with four Australian serotypes of bluetongue virus. Journal of Medical Entomology, 31, 382–387.
- BERNARD, K.A., ISRAEL, B.A., WILSON, W.C. & SCHULTZ, K.T., 1997. Sequence and cognitive analyses of two virulence-associated markers of bluetongue virus serotype 17. Intervirology, 40, 226–231.
- BLACKSELL, S.D. & LUNT, R.A., 1996. A simplified fluorescence inhibition test for the serotype determination of Australian bluetongue viruses. Australian Veterinary Journal, 73, 33–34.
- BLACKSELL, S.D., LUNT, R.A. & WHITE, J.R., 1994. A rapid indirect ELISA for the serogrouping of Australian orbiviruses. Journal of Virological Methods, 49, 67–78.
- BLOIS, H. LE., FRENCH, T., MERTENS, P.P.C., BURROUGHS, J.N. & ROY, P., 1992. The expressed VP4 protein of bluetongue virus binds GTP and is a candidate guanylyl transferase of the virus. Virology New York, 189, 757–761.
- BONNEAU, K.R., DEMAULA, C.D., MULLENS, B.A. & MACLACHLAN, N.J., 2002. Duration of viraemia infectious to Culicoides sonorensis in bluetongue virus-infected cattle and sheep. Veterinary Microbiology, 88, 115-125.
- BORDEN, E.C., SHOPE, R.E. & MURPHY, F.A., 1971. Physicochemical and morphological relationships of some arthropod-borne viruses to bluetongue virus—a new taxonomic group. Physicochemical and serological studies. Journal of General Virology, 13, 261–271.
- BOUWKNEGT, C., VAN RIJN, P.A., SCHIPPER, J.J., HOLZEL, D., BOONSTRA, J., NIJHOF, A.M., VAN ROOIJ E, M.A. & JONGEJAN, F., 2010. Potential role of ticks as vectors of bluetongue virus. Experimental and Applied Acarology, 52, 183–192.
- BOWEN, R.A., HOWARD, T.H. & PICKETT, B.W., 1985. Seminal shedding of bluetongue virus in experimentally infected bulls. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss, 91-96.
- BOWEN, R.A., HOWARD, T.H., ELSDEN, R.P. & SEIDEL, G.E., 1985. Bluetongue virus and embryo transfer in cattle. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss.
- BOWNE, J.G. & JONES, R.H., 1966. Observation on bluetongue virus in the salivary glands of an insect vector, Culicoides variipennis. Virology, 30, 127–133.
- BOWNE, J.G., 1971. Bluetongue disease. In: BRANDLY, C.A. & CORNELIUS, C.E., (eds). Advances in Veterinary Science and Comparative Medicine, 15, 1–46.
- BOYLE, D.B., AMOS-RITCHIE, R., BROZ, I., WALKER, P.J., MELVILLE, L., FLANAGAN, D., DAVIS, S., HUNT, N. & WEIR, R., 2014. Evolution of bluetongue virus serotype 1 in northern Australia over 30 years. Journal of Virology, 88, 13981-13989.
- BRAVERMAN, Y. & CHECHIK, F., 1996. Air streams and the introduction of animal diseases borne on Culicoides (Diptera, Ceratopogonidae) into Israel. Revue Scientifique et Technique Office International des Epizooties, 15, 1037–1052.
- BREARD, E., SCHULTZ, C., SAILLEAU, C., BERNELIN-COTTET, C., VIAROUGE, C., VITOUR, D., GUILLAUME, B., CAIGNARD, G., GORLIER, A., ATTOUI, H., GALLOIS, M., HOFFMANN, B., ZIENTARA, S. & BEER, M., 2017. Bluetongue virus serotype 27: Experimental infection of goats, sheep and cattle with three BTV-27 variants reveal atypical characteristics and likely direct contact transmission of BTV-27 between goats. Transboundary and Emerging Diseases, in press, 65(2), e251-e263.
- BREWER, A.W. & MACLACHLAN, N.J., 1994. The pathogenesis of bluetongue virus infection of bovine blood cells in vitro: ultrastructural characterization. Archives of Virology, 136, 287–298.
- BROWN, C.C., MEYER, R.F. & GRUBMAN, M.J., 1993. Use of a digoxigenin-labelled RNA probe to detect all 24 serotypes of bluetongue virus in cell culture. Journal of Veterinary Diagnostic Investigation, 5, 159–162.
- BROWN, C.C., RHYAN, J.C., GRUBMAN, M.J. & WILBUR, L.A., 1996. Distribution of bluetongue virus in tissues of experimentally infected pregnant dogs as determined by in situ hybridization. Veterinary Pathology, 33, 337–340.
- BUMBAROV, V., GOLENDER, N., ERSTER, O. & KHINICH, Y., 2016. Detection and isolation of Bluetongue virus from commercial vaccine batches. Vaccine, 34, 3317–3323.
- CALVO-PINILLA, E., CASTILLO-OLIVARES, J., JABBAR, T., ORTEGO, J., DE LA POZA, F. & MARIN-LOPEZ, A., 2014. Recombinant vaccines against bluetongue virus. Virus Research, 182, 78-86.
- CAMPBELL, C.H. & GRUBMAN, M.J., 1985. Current knowledge on the biochemistry and immunology of bluetongue. In: PANDEY, R., (ed). Progress in Veterinary Microbiology and Immunology, 1, 58–79.
- CAMPBELL, C.H., 1985. Immunogenicity of bluetongue virus inactivated by gamma irradiation. Vaccine, 3, 401–405.
- CAPORALE, M., Di GIALLEONORADO, L., JANOWICZ, A., WOLKIE, G., SHAW, A., SAVINI, G., VAN RIJN, P.A., MERTENS, P., Di VENTURA, M. & PALMARINI, M., 2014. Virus and host factors affecting the clinical outcome of bluetongue virus infection. Journal of Virology, 88, 10399-10411.
- CASAUBON, J., CHAIGNAT, V., VOGT, H.R., MICHEL, A.O., THUR, B. & RYSER-DEGIORGIS, M.P., 2013. Survey of bluetongue virus infection in free ranging wild ruminants in Switzerland. BMC Veterinary Research, 9, 166.
- CELMA, C.C., STEWART, M., WERNIKE, K., ESCHBAUMER, M., GONZALEZ-MOLLEDA, L., BREARD, E., SCHULTZ, C., HOFFMANN, B., HAEGEMAN, A, DE CLERCQ, K., ZIENTARA, S., VAN RIJN, P.A., BEER, M. & ROY, P., 2017. Replication-deficient particles: New insights into the next generation of bluetongue virus vaccines. Journal of Virology, 91, e01892-16.
- CHAIGNAT, V., WORWA, G., SCHERRER, N., HILBE, M., EHRENSPERGER, F., BATTEN, C., CORTYEN, M., HOFMANN, M. & THUER, B., 2009. Toggenburg Orbivirus, a new bluetongue virus: initial detection, first observations in field and experimental infection of goats and sheep. Veterinary Microbiology, 138, 11–19.
- CHAUVEAU, E., DOCEUL, V., LARA, E., ADAM, M., BREARD, E., SAILLEAU, C., VIAROUGE, C., DESPRAT, A., MEYER, G., SCHWARTZ-CORNIL, I., RUSCANU, S., CHARLEY, B., ZIENTARA, S. & VITOUR, D., 2012. Sensing and control of bluetongue virus infection in epithelial cells via RIG-I and MDA5 helicases. Journal of Virology, 86, 11789-11799.
- CHAUVEAU, E., DOCEUL, V., LARA, E., BREARD, E., SAILLEAU, C., VIDALAIN, P.O., MEURS, E.F., DABO, S., SCHWARTZ-CORNIL, I., ZIENTARA, S. & VITOUR, D., 2013. NS3 of bluetongue virus interferes with the induction of type I interferon. Journal of Virology, 87, 8241-8246.
- CHERRINGTON, J.M., GHALIB, H.W., SAWYER, M.M. & OSBURN, B.I., 1985. Detection of viral antigens in bluetongue virus-infected ovine tissues using the peroxidase-antiperoxidase technique. American Journal of Veterinary Research, 46, 2356–2359.
- CHONG-KEE-YI., BANSAL, O.B., HONG-MEI-LIU., CHATTERJEE, S., ROY, P., CHONG, K.Y. & HONG, M.L., 1996. Sequences within the VP6 molecule of bluetongue virus that determine cytoplasmic and nuclear targeting of the protein. Journal of Virology, 70, 4778–4782.
- CLOETE, M., DU PLESSIS, D.H., VAN DIJK, A.A., HUISMANS, H. & VILJOEN, G.J., 1994. Vaccinia virus expression of the VP7 protein of South African bluetongue virus serotype 4 and its use as an antigen in a capture ELISA. Archives of Virology, 135, 405–418.
- COETZER, J.A.W., 1990. Faculty of Veterinary Science, University of Pretoria, Onderstepoort 0110. Unpublished observation.
- COLLISSON, E.W. & BARBER, T.L., 1983. Blood cells associated with bluetongue virus infection in cattle. Proceedings of the Twenty-sixth Annual Meeting of the American Association of Veterinary Laboratory Diagnosticians, 287–300.
- DAL POZZO, F., DE CLERCQ, K., GUYOT, H., VANDEMEULEBROUCKE, E., SARRADIN, P., VANDENBUSSCHE, F., THIRY, E. & SAEGERMAN, C., 2009. Experimental reproduction of bluetongue virus serotype 8 clinical disease in calves. Veterinary Microbiology, 136, 352-358.
- DANGLER, C.A., DE MATTOS, C.A., DE MATTOS, C. & OSBURN, B.I., 1990. Identifying bluetongue virus ribonucleic acid sequences by the polymerase chain reaction. Journal Virological Methods, 28, 281–292.
- DE CLERCQ, K., DE LEEUW, I., VERHEYDEN, B., VANDEMEULENBROUCKE, E., VANBIST, T., HERR, C., MEROC, E., BERTELS, G., STEURBAUT, N., MIRY, C., DE BLEECKER, K., MAQUET, G., BUGHUIN, J., SAULMONT, M., LEBRUN, M., SUSTRONCK, B., DE DEKEN, R., HOOYBERGS, J., HOUDART, P., RAEMAEKERS, M., MINTIENS, K., KERHOFS, P., GORIS, N. & VANDENBUSSCHE, F., 2008. Transplacental infection and apparently immunotolerance induced by a wild type bluetongue virus serotype 8 natural infection. Transboundary and Emerging Diseases, 55, 352-359.
- DE CLERCQ, K.P., MERTENS, I., DE LEEUW, C., OURA, P., HOUDART, A.C., POTGIETER, S., MAAN, J., HOOYBERGHS, C., BATTEN, E., VANDEMEULEBROUCKE, I.M., WRIGHT, N., MAAN, F., RIOCREUX, A., SANDERS, Y., VAN DER STEDE, K., NOMIKOU, M., RAEMAEKERS, A., BIN-TARIF, A., SHAW, M., HENSTOCK, E., BREARD, E., DUBOIS, C., GASTALDI-THIERY, S., ZIENTARA, B., VERHEYDE.N. & VANDENBUSSCHE, F., 2009. Emergence of bluetongue serotypes in Europe, part 2: the occurrence of a BTV-11 strain in Belgium. Transboundary Emerging Diseases, 56, 355-361.
- DE MATTOS, C.C., DE MATTOS, C.A., MACLACHLAN, N.J., GIAVEDONI, L.D., YILMA, T. & OSBURN, B.I., 1996. Phylogenetic comparison of the S3 gene of United States prototype strains of bluetongue virus with that of field isolates from California. Journal of Virology, 70, 5735–5739.
- DE MATTOS, C.C., DE MATTOS, C.A., OSBURN, B.I., DANGLER, C.A., CHUANG, R.Y. & DOI, R.H., 1989. Recombinant DNA probe for serotype-specific identification of bluetongue virus 17. American Journal of Veterinary Research, 50, 536–541.
- DEMAULA, C.D., JUTILA, M.A., WILSON, D.W. & MACLACHLAN, N.J., 2001. Infection kinetics, prostacyclin release and cytokine-mediated modulation of the mechanism of cell death during bluetongue virus infection of cultured ovine and bovine pulmonary artery and lung microvascular endothelial cells. Journal of General Virology, 82, 787-794.
- DEMAULA, C.D., LEUTENEGGER, C.M., JUTILA, M.A. & MACLACHLAN, N.J., 2002. Bluetongue virus-induced activation of primary bovine lung microvascular endothelial cells. Veterinary Immunology Immunopathology, 86, 147-157.
- DEMAULA, C.D., LEUTENNEGGER, C.M., BONNEAU, K.R. & MACLACHLAN, N.J., 2002. The role of endothelial cell-derived inflammatory and vasoactive mediators in the pathogenesis of bluetongue. Virology, 296, 330-337.
- DHANASEKARAN, S., VIGNESH, A.R., RAJ, G.D., REDDY, Y.K., RAJA, A., TIRUMURUGAAN, K.G., 2013. Comparative analysis of innate immune response fo&llowing in vitro stimulation of sheep and goat peripheral blood mononuclear cells with bluetongue virus serotype 23. Veterinary Research Communications, 37, 319-327.
- DI GIALLEONARDO, L., MIGLIACCIO, P., TEODORI, L. & SAVINI, G., 2011. The length of BTV-8 viraemia in cattle according to infection doses and diagnostic techniques. Research in Veterinary Science, 91, 316-320.
- DREW, C.P., GARDNER, I.A., MAYO, C.E., MATSUO, E., ROY, P. & MACLACHLAN, N.J., 2010. Bluetongue virus infection alters the impedance of monolayers of bovine endothelial cells as a result of cell death. Veterinary Immunology Immunopathology, 136, 108-115.
- DREW, C.P., HELLER, M.C., MAYO, C.E., WATSON, J.L. & MACLACHLAN, N.J., 2010. Bluetongue virus infection activates bovine monocyte-derived macrophages and pulmonary artery endothelial cells. Veterinary Immunology Immunopathology, 136, 292-296.
- DU TOIT, R.M., 1944. The transmission of bluetongue and horse sickness by Culicoides. Onderstepoort Journal of Veterinary Science and Animal Industry, 19, 7–16.
- DU TOIT, R.M., 1962. The role played by bovines in the transmission of bluetongue in sheep—Preliminary communication. Journal of the South African Veterinary Medical Association, 33, 483–490.
- DUNN, S.J., HSU, D., ZEE, Y.C. & STOTT, J.L., 1992. Complete nucleotide and deduced amino acid sequence of genome segment 5 encoding the outer capsid protein, VP5, of a U.S. isolate of bluetongue virus serotype 11. Virus Research, 21, 155–162.
- EAGLES, D., MELVILLE, L., WEIR, R., DAVIS, S., BELLIS, G., ZALUCKI, M.P., WALKER, P.J. & DURR, P.A., 2014. Long-distance aerial dispersal modelling of Culicoides biting midges: case studies of incursions into Australia. BMC Veterinary Research, 10, 135.
- EL HUSSEIN, A., CALISHER, C.H., HOLBROOK, F.R., SCHOEPP, R.J. & BEATY, B.J., 1989. Detection of bluetongue virus antigens in Culicoides variipennis by enzyme immunoassay. Journal of Clinical Microbiology, 27, 1320– 1323.
- ELBERS, A.R., BACKX, A., MEROC, E., GERBIER, G., STAUBACH, C., HENDRICKX, G., VAN DER SPEK, A. & MINTIENS, K., 2008. Field observations during the bluetongue serotype 8 epidemic in 2006. I. Detection of first outbreaks and clinical signs in sheep and cattle in Belgium, France and the Netherlands. Preventive Veterinary Medicine, 87, 21-30.
- ELS, H.J. & VERWOERD, D.W., 1969. Morphology of bluetongue virus. Virology, 38, 213–219.
- ELZEIN, E.M.E.A., SANDOUKA, M.A., AL-AFALEQ, A.I., MOHAMED, O.B. & FLAMAND, J.R.B., 1998. Arbovirus infections of ruminants in Al-rub Al-Khali desert. The Veterinary Record, 142, 196–197.
- EMAU, P., GIRI, S.N., ANDERSON, G.A., STOTT, J.L. & OSBURN, B.I., 1984. Function of prostaglandins, thromboxane A2, and histamine in hypersensitivity reaction to experimental bluetongue disease in calves. American Journal of Veterinary Research, 45, 1852–1857.
- ENSERINK, M., 2006. Emerging infectious diseases: During a hot summer, bluetongue invades northern Europe. Science, 313, 1218-1219.
- ERASMUS, B.J., 1975. Bluetongue in sheep and goats. Australian Veterinary Journal, 51, 165–170.
- ERASMUS, B.J., 1975. The epizootiology of bluetongue: The African situation. Australian Veterinary Journal, 51, 196–198.
- ERASMUS, B.J., 1980. The epidemiology and control of bluetongue in South Africa. Bulletin de L’Office International des Epizooties, 92, 461–467.
- ERASMUS, B.J., 1990. Bluetongue. In: DINTER, Z. & MOREIN, B., (eds). Virus Infections of Ruminants. Amsterdam: Elsevier Science Publishers, 3.
- ERASMUS, B.J., 1991. Veterinary Research Institute, Onderstepoort, South Africa. Unpublished observations.
- ERASMUS, B.J., NEVILL, E.M. & VENTER, G.J., 1984. Veterinary Research Institute, Onderstepoort, South Africa. Unpublished observations.
- ESCHBAUMER, M., HOFFMANN, B., MOSS, A., SAVINI, G., LEONE, A., KONIG, P., ZEMKE, J., CONRATHS, F. & BEER M., 2010. Emergence of bluetongue virus serotype 6 in Europe-German field data and experimental infection in cattle. Veterinary Microbiology 143, 189–195.
- FEENSTRA, F. & VAN RIJN, P.A., 2017. Current and next-generation bluetongue vaccines: Requirements, strategies, and prospects for different field situations. Critical reviews of Microbiology, 43, 142-155.
- FLANAGAN, M. & JOHNSON, S.J., 1995. The effects of vaccination of Merino ewes with an attenuated Australian bluetongue virus serotype 23 at different stages of gestation. Australian Veterinary Journal, 72, 455–457.
- FLANAGAN, M., JOHNSON, S.J., HOFFMANN, D., POLKINGHORNE, I.G., REID, D.J. & SHEPHERD, M.A., 1993. Clinical pathology of Australian bluetongue virus serotype 16 infection in Merino sheep. Australian Veterinary Journal, 70, 101–104.
- FOSTER, N.M. & LUEDKE, A.J., 1968. The direct assay for bluetongue virus by intravascular inoculation of embryonating chicken eggs. American Journal of Veterinary Research, 29, 749–753.
- FOSTER, N.M., JONES, R.H. & MCCRORY, B.R., 1963. Preliminary investigations on insect transmission of bluetongue virus in sheep. American Journal of Veterinary Research, 24, 1195–1200.
- FOSTER, N.M., LUEDKE, A.J., PARSONSON, I.M. & WALTON, T.E., 1991. Temporal relationships of viremia, interferon activity, and antibody responses of sheep infected with several bluetongue virus strains. American Journal of Veterinary Research, 52, 192–196.
- FRENCH, T.J., MARSHALL, J.J.A. & ROY, P., 1990. Assembly of double shelled virus-like particles of bluetongue virus by the simultaneous expression of four structural proteins. Journal of Virology, 64, 5695–5700.
- GAMBLES, R.M., 1949. Bluetongue of sheep in Cyprus. Journal of Comparative Pathology, 59, 176–190.
- GERDES, G.H., NESER, J.A., BARNARD, B.J.H. & LARSEN, J., 1996. Stomatitis and coronitis in cattle—an insect-borne viral disease? Journal of the South African Veterinary Association, 67, 103–104.
- GERS, S., POTGIETER, C., WRIGHT, I. & PEYROT, B., 2016. Natural bluetongue in alpacas in South Africa. Veterinaria Italiana, 52, 291-292.
- GIBBS, E.P. & GREINER, E.C., 1994. The epidemiology of bluetongue. Comparative Immunology Microbiology and Infectious Diseases, 17, 207-220.
- GIBBS, E.P.J. & GREINER, E.C., 1988. Bluetongue and epizootic hemorrhagic disease. In: MONATH, T.P., (ed). The Arboviruses: Epidemiology and Ecology. Vol. II. Boca Raton: CRC Press, Inc, 2, 39.
- GILLESPIE, J.H., SCHLAFER, D.H., FOOTE, R.H., QUICK, S., DOUGHERTY, E., SCHIFF, E. & ALLEN, S., 1990. Comparison of persistence of seven bovine viruses on bovine embryos following in vitroexposure. Deutsche Tierärztliche Wochenschrift, 97, 65–68.
- GOFFREDO, M., CATALANI, M., FEDERICI, V., PORTANTI, O., MARINI, V., MANCINI, G., QUAGLIA, M., SANTILLI, A., TEODORI, L. & SAVINI, G., 2015. Vector species of Culicoides midges implicated in the 2012, 2014 Bluetongue epidemics in Italy. Veterinaria Italiana, 51, 131-138.
- GOFFREDO, M., MEISWINKEL, R., FEDERICI, V., Di NICOLA, F., MANCINI, G., IPPOLITI, C., Di LORENZO, A., OUAGLIA, M., SANTILLI, A., CONTE, A. & SAVINI, G., 2016. The “Culicoides obsoletus group” in Italy: relative abundance, geographic range, and role as vector for bluetongue virus. Veterinaria Italiana, 52, 235-241.
- GOLDSMIT, L. & BARZILAI, E., 1968. An improved method for the isolation and identification of bluetongue virus by intravenous inoculation of embryonating chicken eggs. Journal of Comparative Pathology, 78, 477–487.
- GORMAN, B.M., TAYLOR, J. & WALKER, P.J., 1983. Orbiviruses. In: JOKLIK, W.K., (ed). The Reoviridae. Plenum: New York.
- GOULD, A.R., 1988. Conserved and non-conserved regions of the outer coat protein, VP2, of the Australian bluetongue serotype 1 virus revealed by sequence comparison to the VP2 of North America BTV serotype 10. Virus Research, 9, 145–158.
- GOULD, A.R., 1988. The use of recombinant DNA probes to group and type orbiviruses. A comparison of Australian and South African isolates. Archives of Virology, 99, 205–220.
- GRIMES, J.M., BURROUGHS, J.N., GOUET, P., DIPROSE, J.M., MALBY, R., SIENTARA, S., MERTENS, P.P.C. & STUART, D.L., 1998. The atomic structure of the bluetongue virus core. Nature, 395, 470–478.
- GRINER, L.A. & MCCRORY, B.R., 1964. Bluetongue associated with abnormalities in newborn lambs. Journal of the American Veterinary Association, 145, 1013–1019.
- GUIS, H., CAMINADE, C., CALVETE, C., MORSE, A.P., TRAN, A. & BAYLIS, M., 2012. Modelling the effects of past and future climate on the risk of bluetongue emergence in Europe. Journal of the Royal Society Interface, 9, 339-350.
- HAIG, D.A., 1959. Bluetongue. Proceedings of the Sixteenth International Veterinary Congress, Madrid, 215–225.
- HAIG, D.A., MCKERCHER, D.G. & ALEXANDER, R.A., 1956. The cytopathogenic action of bluetongue virus on tissue cultures and its application to the detection of antibodies in the serum of sheep. Onderstepoort Journal of Veterinary Research, 27, 171–177.
- HARDY, W.T. & PRICE, D.A., 1952. Soremuzzle of sheep. Journal of the American Veterinary Medical Association, 120, 23–25.
- HARE, W.C.D., LUEDKE, A.J., THOMAS, F.C., BOWEN, R.A., SINGH, E.L., EAGLESOME, M.D., RANDALL, G.C.B. & BIELANSKI, A., 1988. Nontransmission of bluetongue virus by embryos from bluetongue virus-infected sheep. American Journal of Veterinary Research, 49, 468–472.
- HEWAT, E.A., BOOTH, T.F. & ROY, P., 1992. Structure of bluetongue virus particles by cryo-electron microscopy. Journal of Structural Biology, 10, 61–69.
- HOFF, G.L. & HOFF, D.M., 1976. Bluetongue and epizootic hemorrhagic disease: A review of these diseases in non-domestic artiodactyles. Journal of Zoo Animal Medicine, 7, 26–30.
- HOFF, G.L. & TRAINER, D.O., 1974. Observations on bluetongue and epizootic haemorrhagic disease viruses in white-tailed deer: (1) Distribution of virus in the blood (2) Crosschallenge. Journal of Wildlife Diseases, 10, 25–28.
- HOFMANN, M.A., RENZULLO, S., MADER, M., CHAIGNAT, V., WORWA, G. & THUER, B., 2008. Genetic characterization of Toggenburg Orbivirus, a new bluetongue virus, from goats, Switzerland. Emerging Infectious Diseases, 14, 1855-1861.
- HOLBROOK, F.R., 1985. Research on the control of bluetongue in livestock by vector suppression. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan Liss, Inc, 545-553.
- HOURRIGAN, J.L. & KLINGSPORN, A.L., 1975. Bluetongue: The disease in cattle. Australian Veterinary Journal, 51, 170–174.
- HOWELL, P.G. & VERWOERD, D.W., 1971. Bluetongue virus. In: GARD, S., HALLAUER, C. & MEYER, K.F., (eds). Virology Monographs, 9, 35–74.
- HOWELL, P.G., 1960. A preliminary antigenic classification of strains of bluetongue virus. Onderstepoort Journal of Veterinary Research, 28, 357–363.
- HOWELL, P.G., KÜMM, N.A. & BOTHA, M.J., 1970. The application of improved techniques to the identification of strains of bluetongue virus. Onderstepoort Journal of Veterinary Research, 37, 59–66.
- HOWELL, P.G., VERWOERD, D.W. & OELLERMANN, R.A., 1967. Plaque formation by bluetongue virus. Onderstepoort Journal of Veterinary Research, 34, 317–332.
- HOWERTH, E.W. & TYLER, D.E., 1988. Experimentally induced bluetongue virus infection in white-tailed deer: Ultrastructural findings. American Journal of Veterinary Research, 49, 1914–1922.
- HOWERTH, E.W., GREENE, C.E. & PRESTWOOD, A.K., 1988. Experimentally induced bluetongue virus infection in white-tailed deer: Coagulation, clinical pathologic, and gross pathologic changes. American Journal of Veterinary Research, 49, 1906–1913.
- HOWERTH, E.W., 2015. Cytokine release and endothelial dysfunction: a perfect storm in orbivirus pathogenesis. Veterinaria Italiana, 51, 275-281.
- HUANG, I.J., HWANG, G.Y., YANG, Y.Y., HAYAMA, E. & LI, J.K.K., 1995. Sequence analysis and antigenic epitope mapping of the putative RNA-directed RNA polymerase of five U.S. bluetongue viruses. Virology New York, 214, 280–288.
- HÜBSCHLE, O.J.B., LORENZ, R.J. & MATHEKA, K.D., 1981. Enzyme linked immunosorbent assay for detection of bluetongue virus antibodies. American Journal of Veterinary Research, 42, 61–65.
- HUISMANS, H. & CLOETE, M., 1987. A comparison of different cloned bluetongue virus genome segments as probes for the detection of virus-specific RNA. Virology, 158, 373–380.
- HUISMANS, H. & ERASMUS, B.J., 1981. Identification of the serotype-specific and group-specific antigens of bluetongue virus. Onderstepoort Journal of Veterinary Research, 48, 51–58.
- HUISMANS, H. & VAN DIJK, A.A., 1990. Bluetongue virus structural components. Current Topics in Microbiology and Immunology, 162, 2141.
- HUISMANS, H., CLOETE, M. & LE ROUX, A., 1987. The genetic relatedness of a number of individual cognate genes of viruses in the bluetongue and closely related serogroups. Virology, 161, 421–428.
- HUISMANS, H., VAN DER WALT, N.T. & ERASMUS, B.J., 1985. Immune response against the purified serotype specific antigen of bluetongue virus and initial attempts to clone the gene that codes for the synthesis of this protein. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss, 178, 347–353.
- HUISMANS, H., VAN DER WALT, N.T., CLOETE, M. & ERASMUS, B.J., 1987. Isolation of a capsid protein of bluetongue virus that induces a protective immune response in sheep. Virology, 157, 172–179.
- HUISMANS, H., VAN DIJK, A.A. & ELS, H.J., 1987. Uncoating of parental bluetongue virus to core and subcore particles in infected L-cells. Virology, 157, 180–188.
- HUTCHEON, D., 1880. Fever or epizootic catarrh. Report of the Colonial Veterinary Surgeon for 1880, 12 (1881).
- HUTCHEON, D., 1902. Malarial catarrhal fever of sheep. The Veterinary Record, 14, 629–633.
- HWANG, G.Y., YANG, Y.Y., CHIOU, J.F. & LI, J.K.K., 1992. Sequence conservation among the cognate nonstructural NS3/3A protein genes of six bluetongue viruses. Virus Research, 23, 151–161.
- HYATT, A.D., GOULD, A.R., COUPAR, B. & EATON, B.T., 1991. Localization of the non-structural protein NS3 in bluetongue virus-infected cells. Journal of General Virology, 72, 2263–2267.
- INUMARU, S. & ROY, P., 1987. Production and characterization of the neutralization antigen VP2 of bluetongue virus serotype 10 using a baculovirus expression vector. Virology, 157, 472–479.
- IWATA, H., CHUMA, T. & ROY, P., 1992. Characterization of the genes encoding two of the major capsid proteins of epizootic haemorrhagic disease virus indicates a close genetic relationship to bluetongue virus. Journal of General Virology, 73, 915–924.
- IWATA, H., HIRASAWA, T. & ROY, P., 1991. Complete nucleotide sequence of segment 5 of epizootic haemorrhagic diseases virus; the outer capsid protein VP5 is homologous to the VP5 protein of bluetongue virus. Virus Research, 20, 273–281.
- JANOWICZ, A., CAPORALE, M., SHAW, A., GULLETTA, S., Di GIALLEONARDO, L., RATINIER, M. & PALMARINI, M., 2015. Multiple genome segments determine virulence of bluetongue virus serotype 8. Journal of Virology, 89, 5238-5249.
- JAUNIAUX, T.P., DE CLERCQ, K.E., CASSART, D.E., KENNEDY, S., VANDENBUSSCHE, F.E., VANDEMEULEBROUCKE, E.L., VANBINST, T.M., VERHEYDEN, B.I., GORIS, N.E. & COIGNOUL, F.L., 2008. Bluetongue in Eurasian lynx. Emerging Infectious Diseases, 14, 1496-1498.
- JEGGO, M.H. & WARDLEY, R.C., 1985. Bluetongue vaccine: Cells and/or antibodies. Vaccine, 3, 57–58.
- JEGGO, M.H., GUMM, I.D. & TAYLOR, W.P., 1983. Clinical and serological response of sheep to serial challenge with different bluetongue virus types. Research in Veterinary Science, 34, 205–211.
- JEWISS-GAINES, A., BARELLI, L. & HUNTER, F.F., 2017. First records of Culicoides sonorensis (Diptera; Ceratopogonidae), a known vector of bluetongue virus, in southern Ontario. Journal of Medical Entomology, 54, 757-762.
- JOCHIM, M.M., 1985. Bluetongue: A review of the immune response of sheep and cattle to bluetongue virus infection. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss, 455-460.
- JOCHIM, M.M., 1986. Bluetongue in cattle. In: HOWARD, J.L., (ed). Current Veterinary Therapy: Food Animal Practice 2. Philadelphia: W.B. Saunders.
- JOCHIM, M.M., BARBER, T.L. & BANDO, B.M., 1974. Identification of bluetongue and epizootic haemorrhagic disease viruses by the indirect fluorescent antibody procedure. Proceedings of the Seventeenth Annual Meeting of the American Association for Veterinary Laboratory Diagnosticians, 91–103.
- JONES, R.H. & FOSTER, N.M., 1971. Transovarian transmission of bluetongue virus unlikely for Culicoides variipennis. Mosquito News, 31, 434–437.
- KATZ, J.B., GUSTAFSON, G.A., ALSTAD, A.D., ADLER, K.A. & MOSER, K.M., 1993. Colorimetric diagnosis of prolonged bluetongue viremia in sheep using an enzyme-linked oligonucleotide sorbent assay of amplified viral nucleic acids. American Journal of Veterinary Research, 54, 2021–2026.
- KELLERMAN, T.S., COETZER, J.A.W. & NAUDÉ, T.W., 1988. Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa. Cape Town: Oxford University Press Southern Africa, 243.
- KIPPS, A., 1956. Complement fixation with antigens prepared from bluetongue virus-infected mouse brains. Journal of Hygiene (London), 54, 79–88.
- KIRKLAND, P.D. & HAWKES, R.A., 2004. A comparison of laboratory and 'wild' strains of bluetongue virus--is there any difference and does it matter? Veterinaria Italiana, 40, 448-455.
- KOMAROV, A. & GOLDSMIT, L., 1951. A disease similar to bluetongue in cattle and sheep in Israel. Refuah Veterinarith, 8, 96–100.
- KRAMPS, J.A., VAN MAANEN, K., MARS, M.H., POPMA, J.K. & VAN RIJN, P.A., 2007. Validation of a commercial ELISA for the detection of bluetongue virus (BTV)-specific antibodies in individual milk samples of Dutch dairy cows. Veterinary Microbiology, 130, 80–87.
- LAGE, A.P., CASTRO, R.S., MELO, M.I.V., AGUIAR, P.H.P., BARRETO-FILHO, J.B. & LEITE, R.C., 1996. Prevalence of antibodies to bluetongue bovine herpesvirus 1 and bovine viral diarrhoea/mucosal disease viruses in water buffaloes in Minas Gerais State, Brazil. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 49, 195–197.
- LAWMAN, M.J.P., 1979. Observation on the pathogenesis of bluetongue virus infections in sheep. Thesis, University of Surrey.
- LOBATO, Z.I.P., COUPAR, B.E.H., GRAY, C.P., LUNT, R. & ANDREW, M.E., 1997. Antibody responses and protective immunity to recombinant vaccinia virus-expressed bluetongue virus antigens. Veterinary Immunology and Immunopathology, 59, 293–309.
- LOPEZ, A.C. & BOTIJA, C.S., 1958. Epizootie de fièvre catarrhale ovine en Espagne (bluetongue). Bulletin de L’Office International des Epizooties, 50, 65–93.
- LORUSSO, A., BADA, D., SPEDICATO, M., TEODORI, L., BONFINI, B., MARCACCI, M., DI PROVVIDO, A., ISSELMOU, K., MARINI, V., CARMINE, I., SCACCHIA, M., DI SABATINO, D., PETRINI, A., BEZEID, B.A. & SAVINI, G., 2016. Bluetongue surveillance in the Islamic Republic of Mauritania: Is serotype 26 circulating among cattle and dromedaries? Infection, Genetics and Evolution, 40, 109-112.
- LORUSSO, A., SGHAIER, S., ANCORA, M., MARCACCI, M., DI GENNARO, A., PORTANTI, O., MANGONE, I., TEODORI, L., LEONE, A., CAMMA, C., PETRINI, A., HAMMAMI, S. & SAVINI, G., 2014. Molecular epidemiology of bluetongue virus serotype 1 circulating in Italy and its connection with northern Africa. Infection, Genetics and Evolution, 28, 144-149.
- LORUSSO, A., SGHAIER, S., DI DOMENICO, M., BARBRIA, M.E., ZACCARIA, G., MEGDICH, A., PORTANTI, O., SELIMAN, I.B., SPEDICATO, M., PIZZURRO, F., CARMINE, I., TEODORI, L., MAHJOUB, M., MANGONE, I., LEONE, A., HAMMAMI, S., MARCACCI, M. & SAVINI, G., 2018. Analysis of bluetongue serotype 3 spread in Tunisia and discovery of a novel strain related to the bluetongue virus isolated from a commercial sheep pox vaccine. Infection, Genetics and Evolution, 59, 63-71.
- LOUDON, P.T., HIRASAWA, T., OLDFIELD, S., MURPHY, M. & ROY, P., 1991. Expression of the outer capsid protein VP5 of two bluetongue viruses, and synthesis of chimeric double-shelled virus-like particles using combinations of recombinant baculoviruses. Virology, 182(2), 793-801.
- LUEDKE, A.J., 1970. Distribution of virus in blood components during viremia of bluetongue. Proceedings of the Seventy- fourth Annual Meeting of the US Animal Health Association, 9, 21.
- LUEDKE, A.J., 1985. WHO/FAO Working Team Report: Virology. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss.
- LUEDKE, A.J., BOWNE, J.G., JOCHIM, M.M. & DOYLE, C., 1964. Clinical and pathologic features of bluetongue in sheep. American Journal of Veterinary Research, 25, 963–970.
- LUEDKE, A.J., JOCHIM, M.M. & BOWNE, J.G., 1965. Preliminary bluetongue transmission with the sheep ked Melophagus ovinus (L.) Canadian Journal of Comparative Medicine and Veterinary Science, 29, 229–231.
- LUEDKE, A.J., JOCHIM, M.M. & JONES, R.H., 1977. Bluetongue in cattle: Effects of Culicoides variipennis-transmitted bluetongue virus on pregnant heifers and their calves. American Journal of Veterinary Research, 38, 1687–1696.
- LUEDKE, A.J., JOCHIM, M.M. & JONES, R.H., 1977. Bluetongue in cattle: Effects of calves previously infected in utero. American Journal of Veterinary Research, 38, 1697–1700.
- LUEDKE, A.J., JOCHIM, M.M. & JONES, R.H., 1977. Bluetongue in cattle: Repeated exposure of two immunologically tolerant calves to bluetongue virus by vector bites. American Journal of Veterinary Research, 38, 1701–1703.
- LUEDKE, A.J., JOCHIM, M.M., BOWNE, J.G. & JONES, R.H., 1970. Observations on latent bluetongue virus infection in cattle. Journal of the American Veterinary Medical Association, 156, 1871–1879.
- LUNDERVOLD, M., MILNER-GULLAND, E.J., O’CALLAGHAN, C.J. & HAMBLIN, C., 2003. First evidence of bluetongue virus in Kazakhstan. Veterinary Microbiology, 92, 281-287.
- MAAN, S., MAAN, N.S., BELAGANAHALLI, M.N., POTGIETER, A.C., KUMAR, V., BATRA, K., WRIGHT, I.M., KIRKLAND, P.D. & MERTENS, P.P., 2016. Development and evaluation of real time RT-PCR assays for detection and typing of bluetongue virus. PLoS One, 11, e0163014.
- MAAN, S., MAAN, N.S., BELAGANAHALLI, M.N., RAO, P.P., SINGH, K.P., HEMADRI, D., PUTTY, K., KUMAR, A., BATRA, K., KRISHNAIVOTHI, Y., CHANDEL, B.S., REDDY, G.H., NOMIKOU, K., REDDY, Y.N., ATTOUI, H., HEGDE, N.R. & MERTENS, P.P., 2015. Full genome sequencing as a basis for molecular epidemiology studies of bluetongue virus in India. PLoS One, 10, e0131257.
- MAAN, S., MAAN, N.S., NOMIKOU, K., BATTEN, C., ANTONY, F., BELAGANAHALI, M.N., SAMY, A.M., REDA, A.A., Al-RASHID, S.A., El BATEL, M., OURA, C.A. & MERTENS, P.P., 2011. Novel bluetongue virus serotype from Kuwait. Emerging Infectious Diseases, 17, 886-889.
- MAAN, S., MAAN,, N.S., MERTENS P.P., NOMIKOU, K. & BELAGANAHALLI, M.N., 2012. Reply to Intercontinental movement of bluetongue virus and potential consequences to trade. Journal of Virology, 86, 8342-8343.
- MACLACHLAN, N.J., 2011. Bluetongue: history, global epidemiology, and pathogenesis. Preventive Veterinary Medicine, 102, 107-111.
- MACLACHLAN, N.J., CRAFFORD, J.E., VERNAU, W., GARDNER, I.A., GODDARD, A., GUTHRIE, A.J. & VENTER, E.H., 2008. Experimental reproduction of severe bluetongue in sheep. Veterinary Pathology, 45, 310-315.
- MACLACHLAN, N.J., DREW, C.P., DARPEL, K.E. & WORWA, G., 2009. The pathology and pathogenesis of bluetongue. Journal of Comparative Pathology, 141, 1-16.
- MACLACHLAN, N.J., HENDERSON, C., SCHWARTZ-CORNIL, I. & ZIENTARA, S., 2014. The immune response of ruminant livestock to bluetongue virus: from type I interferon to antibody. Virus Research, 182, 71-77.
- MACLACHLAN, N.J., JAGELS, G., ROSSITO, P.V., MOORE, P.F. & HEIDNER, H.W., 1990. The pathogenesis of experimental bluetongue virus infection of calves. Veterinary Pathology, 27, 223–229.
- MACLACHLAN, N.J. & MAYO, C.E., 2013. Potential strategies for control of bluetongue, a globally emerging, Culicoides-transmitted viral disease of ruminant livestock and wildlife. Antiviral Research, 99, 79-90.
- MACLACHLAN, N.J., MAYO, C.E., DANIELS, P.W., SAVINI, G., ZIENTARA, S. & GIBBS, E.P., 2015. Bluetongue. Review Science and Technology, 34, 329-340.
- MACLACHLAN, N.J., NUNAMAKER, R.A., KATZ, J.B., SAWYER, M.M., AKITA, G.Y., OSBURN, B.I. & TABACHNICK, W.J., 1994. Detection of bluetongue virus in the blood of inoculated calves: comparison of virus isolation, PCR assay, and in vitro feeding of Culicoides variipennis. Archives of Virology, 136, 1-8.
- MACLACHLAN, N.J. & OSBURN, B.I., 2008. Induced brain lesions in calves infected with bluetongue virus. Veterinary Record, 162, 490-491.
- MACLACHLAN, N.J. & OSBURN, B.I., 2017. Teratogenic bluetongue and related orbivirus infections in pregnant ruminant livestock: timing and pathogen genetics are critical. Current Opinions in Virology 27, 31-35.
- MAHMOUD, A., SAVINI, G., SPEDICATO, M., MONACO, F., CARMINE, I., LORUSSO, A., TOLARI, F., MAZZEI, M., FORZAN, M., ELDAGHAYES, I. & DAYHUM, A., 2018. Exploiting serological data to understand the epidemiology of bluetongue virus serotypes circulating in Libya. The Veterinary Journal. Submitted.
- MAKOSCHEY, B., MACLACHLAN, J., VAN WUIJCKHUISE, L., KIRCHVINK, N., DAL POZZO, F., PETIT, H., KAANDORP-HUBER, C., VAN RIJN, P., SELLAL, E., OURA, C., BOINAS, F., CAVIRANI, S., DE CLERCQ K., LUCIENTES, J., MEIJES, C.P., ZIENTARA, S., MEYER, G. & THIRY, E., 2009. Bluetongue control in Europe – new challenges and achievements. Berliner Und Munchener Tierarztliche Wochenschrift, 122, 314-324.
- MANSO-RIBEIRO, J., ROSA-AZEVEDO, J.A., NORONHA, F.M.O., BRACO-FORTE, M.C., GRAVE-PEREIRO, C. & VASCO-FERNANDES, M., 1957. Fievre catarrhale du mouton (bluetongue). Bulletin de L’Office International Epizooties, 48, 350–367.
- MARCACCI, M., SANT, S., MANGONE, I., GORIA, M., DONDO, A., ZOPPI, S., VAN GENNIP, R.G., RADAELLI, M.C., CAMMA, C., VAN RIJN, P.A., SAVINI, G. & LORUSSO, A., 2018. One after the other: A novel Bluetongue virus strain related to Toggenburg virus detected in the Piedmont region (North-western Italy), extends the panel of novel atypical BTV strains. Transboundary Emerging Diseases, 65(2), 370-374. doi: 10.1111/tbed.12822. Epub 2018 Feb 2.
- MARTIN, S.A. & ZWEERINK, H.J., 1972. Isolation and characterization of two types of bluetongue virus particles. Virology, 50, 495–506.
- MARTYN, J.C., GOULD, A.R. & EATON, B.T., 1991. High level expression of the major core protein VP7 and the non-structural protein NS3 of bluetongue virus in yeast: Use of expressed VP7 as a diagnostic, group-reactive antigen in a blocking ELISA. Virus Research, 18, 165–178.
- MASON, J.H., COLES, J.D.W.A. & ALEXANDER, R.A., 1940. Cultivation of bluetongue virus in fertile eggs produced on a vitamin deficient diet. Nature, 145, 1022–1023.
- MAYO, C.E, MULLENS, B.A., REISEN, W.K., OSBORNE, C.J., GIBBS, E.P., GARDNER, I.A. & MACLACHLAN, N.J., 2014. Seasonal and interseasonal dynamics of bluetongue virus infection of dairy cattle and Culicoides sonorensis midges in northern California--implications for virus overwintering in temperate zones. PLoS One, 9, e106975.
- MAYO, C.E., CROSSLEY, B.M., HIETALA, S.K., GARDNER, I.A., BREITMEYER, R.E. & MACLACHLAN, N.J., 2010. Colostral transmission of bluetongue virus nucleic acid among newborn dairy calves in California. Transboundary Emerging Diseases, 57, 277-281.
- MAYO, C.E., MULLENS, B., GIBBS, E.P. & MACLACHLAN, N.J., 2016. Overwintering of bluetongue virus in temperate zones. Veterinaria Italiana, 52, 243-246.
- MAYO, C.E., MULLENS, B.A., GERRY, A.C., BARKER, C.M., MERTENS, P.P., MAAN, N., GARDNER, I.A., GUTHRIE, A.J. & MACLACHLAN, N.J., 2012. The combination of abundance and infection rates of Culicoides sonorensis estimates risk of subsequent bluetongue virus infection of sentinel cattle on California dairy farms. Veterinary Parasitology, 187, 295-301.
- McDERMOTT, E.G., MAYO, C.E., GERRY, A.C., LAUDIER, D., MACLACHLAN, N.J. & MULLENS, B.A., 2015. Bluetongue virus infection creates light averse Culicoides vectors and serious errors in transmission risk estimates. Parasites and Vectors, 8, 460.
- MCKERCHER, D.G., MCGOWAN, B., HOWARTH, J.A. & SAITO, J.K., 1953. A preliminary report on the isolation and identification of the bluetongue virus from sheep in California. Journal of the American Veterinary Medical Association, 122, 300–301.
- MCVEY, D.S. & MACLACHLAN, N.J., 2015. Vaccines for prevention of bluetongue and epizootic hemorrhagic disease in livestock: A North American perspective. Vector Borne Zoonotic Diseases, 15, 385-396.
- MECHAM, J.O., DEAN, V.C., WIGINGTON, J.G. & NUNAMAKER, R.A., 1990. Detection of bluetongue virus in Culicoides variipennis (Diptera: Ceratopogonidae) by an antigen capture enzyme-linked immunosorbent assay. Journal of Medical Entomology, 27, 602–606.
- MEHLHORN, H., WALLDORF, V., KLIMPEL, S. & SCHMAHL, G., 2008. Outbreak of bluetongue disease (BTD) in Germany and the danger for Europe. Parasitology Research, 103, 79–86.
- MEISWINKEL, R., BALDET, T., DE DEKEN, R., TAKKEN, W., DELCOLLE, J.C. & MELLOR, P.S., 2008. The 2006 outbreak of bluetongue in northern Europe – the entomological perspective. Preventive Veterinary Medicine, 87, 55-63.
- MEISWINKEL, R., GOMULSKI, L.M., DELECOLLE, J.C., GOFFREDO, M. & GASPERI, G., 2004. The taxonomy of Culicoides vector complexes – unfinished business. Veterinaria Italiana, 40, 151-159.
- MELLOR, P.S., 1990. The replication of bluetongue virus in Culicoides vectors. In: ROY, P. & GORMAN, B.M., (eds). Bluetongue Viruses. Current Topics in Microbiology and Immunology. Berlin: Springer-Verlag, 162.
- MELLOR, P.S., CARPENTER, S., HARRUP, L., BAYLIS, M. & MERTENS, P.P., 2008. Bluetongue in Europe and the Mediterranean Basin: History of occurrence prior to 2006. Preventive Veterinary Medicine, 87, 4-20.
- MELLOR, P.S. & WITTMANN, E.J., 2002. Bluetongue virus in the Mediterranean basin 1998–2001. Veterinary Journal 164, 20–37.
- MELZI, E., CAPORALE, M., ROCCHI, M., MARTIN, V., GAMINO, V., DI PROVVIDO, A., MARRUCHELLA, G., ENTRICAN, G., SEVILLA, N. & PALMARINI, M., 2016. Follicular dendritic cell disruption as a novel mechanism of virus-induced immunosuppression. Proceedings of the National Academy of Sciences, 113, E6238-E6247.
- MENZIES, F.D., McCULLOUGH, S.J., McKEOWN, I.M., FORSTER, J.L., JESS, S., BATTEN, C., MURCHIE, A.K., GLOSTER, J., FALLOWS, J.G., PELGRIM, W., MELLOR, P.S. & OURA, C.A., 2008. Evidence for transplacental and contact transmission of bluetongue virus in cattle. Veterinary Record, 163, 203-209.
- MERTENS, P.P., BROWN, F. & SANGAR, D.V., 1984. Assignment of the genome segments of bluetongue virus type 1 to the proteins which they encode. Virology, 135, 207-217.
- MERTENS, P.P., BURROUGHS, J.N., WELLBY, M.P., FU, H., O’HARA, R.S., BROOKS, S.M. & MELLOR, P.S., 1996. Enhanced infectivity of modified bluetongue virus particles for two insect cell lines and for two Culicoides vector species. Virology, 217, 582–593.
- MERTENS, P.P.C., BURROUGHS, J.N. & ANDERSON, J., 1987. Purification and properties of virus particles, infectious subviral particles and cores of bluetongue virus serotypes 1 and 4. Virology, 157, 375–386.
- MERTENS, P.P.C., PEDLEY, S., COWLEY, J., BURROUGHS, J.N., CORTEYN, A.H., JEGGO, M.H., JENNINGS, D.M. & GORMAN, B.M., 1989. Analysis of the roles of bluetongue virus outer capsid protein VP2 and VP5 in determination of virus serotype. Virology, 170, 561–565.
- MEYER, G., LACROUX, C., LEGER, S., TOP, S., GOYEAU, K., DEPLANCHE, M. & LEMAIRE, M., 2009. Lethal bluetongue virus serotype 1 infection in llamas. Emerging Infectious Diseases, 15, 608-610.
- MOULTON, J.E., 1961. Pathology of bluetongue of sheep in California. Journal of the American Veterinary Medical Association, 138, 493–498.
- MURPHY, F.A., BORDEN, E.C., SHOPE, R.E. & HARRISON, A., 1971. Physicochemical and morphological relationships of some arthropod-borne viruses to bluetongue virus—A new taxonomic group. Electron microscopic studies. Journal of General Virology, 13, 273–288.
- NEITZ, W.O., 1933. The blesbuck (Damaliscus albifrons) as a carrier of heartwater and bluetongue. Journal of the South African Veterinary Medical Association, 4, 24–26.
- NEITZ, W.O., 1948. Immunological studies on bluetongue in sheep. Onderstepoort Journal of Veterinary Science and Animal Industry, 23, 93–136
- NEL, L.H., PICARD, L.A., HUISMANS, H., 1991. A characterization of the nonstructural protein from which the virus-specified tubules in epizootic haemorrhagic disease virus-infected cells are composed. Virus Research, 18, 219–230.
- NEVILL, E.M., 1971. Cattle and Culicoides biting midges as possible overwintering hosts of bluetongue virus. Onderstepoort Journal of Veterinary Research, 38, 65–71.
- NEVILL, E.M., 1978. The use of cattle to protect sheep from bluetongue infection. Journal of the South African Veterinary Association, 49, 129–130.
- NOAD, R. & ROY, P., 2009. Bluetongue vaccines. Vaccine, 27, Suppl 4, D86-89.
- NOMIKOU, K., HUGHES, J., WASH, R., KELLAM, P., BREARD, E., ZIENTARA, S., PALMARINI, M., BIEK, R. & MERTENS, P., 2015. Widespread reassortment shapes the evolution and epidemiology of bluetongue virus following European invasion. PloS Pathogens 11, e1005056.
- NUNES, S.F., HAMERS, C., RATINIER, M., SHAW, A., BRUNET, S., HUDELET, P. & PALMARINI, M., 2014. A synthetic biology approach for a vaccine platform against known and newly emerging serotypes of bluetongue virus. Journal of Virology, 88, 12222–12232.
- ORLOWSKA, A., TREBAS, P., SMRECZAK, M., MARZEC, A. & ZMUDZINSKI, J.F., 2016. First detection of bluetongue virus serotype 14 in Poland. Archives of Virology, 161, 1969-1972.
- ORTEGA, J., CROSSLEY, B., DECHANY, J.E., DREW, C.P. & MACLACHLAN, N.J., 2010. Fatal bluetongue virus infection in an alpaca in California. Journal of Veterinary Diagnostic Investigation, 22, 134-136.
- OSBORNE, C.J., MAYO, C.E., MULLENS, B.A., McDERMOTT, E.G., GERRY, A.C., REISEN, W.K. & MACLACHLAN, N.J., 2015. Lack of evidence for laboratory and natural vertical transmission of bluetongue virus in Culicoides sonorensis (Diptera: Ceratopogonidae). Journal of Medical Entomology, 52, 274-277.
- OSBURN, B.I., DE MATTOS, C.A., DE MATTOS, C.C. & MACLACHLAN, N.J., 1996. Bluetongue disease and the molecular epidemiology of viruses from the western United States. Comparative Immunology Microbiology of Infectious Diseases, 19, 181-190.
- OSBURN, B.I., JOHNSON, R.T., SILVERSTEIN, A.M., PRENDERGAST, R.A., JOCHIM, M.M. & LEVY, S.E., 1971. Experimental viral-induced congenital encephalopathies. II. The pathogenesis of bluetongue vaccine virus infection in fetal lambs. Laboratory Investigation, 25, 206–212.
- OWEN, N.C., 1964. Investigation into the pH stability of bluetongue virus and its survival in mutton and beef. Onderstepoort Journal of Veterinary Research, 31, 109–118.
- OZAWA, Y., 1985. Bluetongue and related orbiviruses: Overview of the world situation. In: BARBER, T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss, 13-20.
- PAGES, N., BREARD, E., URIEN, C., TALAVERA, S., VIAROUGE, C., LORCA-ORO, C., JOURNEAU, L., CHARLEY, B., ZIENTARA, S., BENSAID, A., SOLANES, D., PUJOLS, J. & SCHWARTZ-CORNIL, I., 2014. Culicoides midge bites modulate the host response and impact on bluetongue virus infection in sheep. PLoS One, 9, e83683.
- PANAGIOTATOS, D.E. 2004. Regional overview of bluetongue viruses, vectors, surveillance and unique features in Eastern Europe between 1998 and 2003. Veterinaria Italiana, 4, 61-72.
- PARKER, J., HERNIMAN, K.A.J., GIBBS, E.P.J. & SELLERS, R.F., 1975. An experimental inactivated vaccine against bluetongue. The Veterinary Record, 96, 284–287.
- PARSONSON, I.M., 1990. Pathology and pathogenesis of bluetongue infections. In: ROY, P. & GORMAN, B.M., (eds). Bluetongue Viruses. Current Topics in Microbiology and Immunology. Berlin: Springer-Verlag, 162.
- PARSONSON, I.M., THOMPSON, L.H. & WALTON, T.E., 1994. Experimentally induced infection with bluetongue virus serotype 11 in cows. American Journal of Veterinary Research, 55, 1529–1534.
- PEARSON, J.E. & JOCHIM, M.M., 1979. Protocol for the immunodiffusion test for bluetongue. Proceedings of the Twenty-second Annual Meeting of the American Association for Veterinary Laboratory Diagnosticians, 463–471.
- PEDLEY, S., MOHAMED, M.E. & MERTENS, P.P.C., 1988. Analysis of genome segments from six different isolates of bluetongue virus using RNA-RNA hybridisation: A generalised coding assignment for bluetongue viruses. Virus Research, 10, 381–390.
- PIERCE, C.M., ROSSITTO, P.V. & MACLACHLAN, N.J., 1995. Homotypic and heterotypic neutralization determinants of bluetongue virus serotype 17. Virology, 209, 263–267.
- PINI, A., 1976. A study on the pathogenesis of bluetongue: Replication of the virus in the organs of infected sheep. Onderstepoort Journal of Veterinary Research, 43, 159–164.
- PINI, A., COACKLEY, W. & OHDER, H., 1966. Concentration of bluetongue virus in experimentally infected sheep and virus identification by immune fluorescence technique. Archiv für gesamte Virusforschung, 18, 385–390.
- PLANZER, J., KAUFMANN, C., WORWA, G., GAVIER_WIDEN, D., HOFMANN, M.A., CHAIGNAT, V. & THUR, B., 2011. In vivo and in vitro propagation and transmission of Toggenburg orbivirus. Research in Veterinary Science, 91, e163-168.
- POLI, G., STOTT, J., LIU, Y.S. & MANNING, J.S., 1982. Bluetongue virus: Comparative evaluation of enzyme-linked immunosorbent assay, immunodiffusion, and serum neutralization for detection of viral antibodies. Journal of Clinical Microbiology, 15, 159–162.
- PRICE, D.A. & HARDY, W.T., 1954. Isolation of the bluetongue virus from Texas sheep: Culicoides shown to be a vector. Journal of the American Veterinary Medical Association, 124, 255–258.
- PRITCHARD, L.I. & GOULD, A.R., 1995. Phylogenetic comparison of the serotype-specific VP2 protein of bluetongue and related orbiviruses. Virus Research, 39, 207–220.
- PULLINGER, G.D., BUSQUETS, M.G., NOMIKOU, K., BOYCE, M., ATTOUI, H. & MERTENS, P.P., 2016. Identification of the genome segments of Bluetongue virus serotype 26 (Isolate KUW2010/02) that restrict replication in a Culicoides sonorensis cell line (KC cells). PLoS One 11, e0149709.
- PURSE, B.V., BROWN, H.E. HARRUP, L., MERTENS, P.P. & ROGER, D.J., 2008. Invasion of bluetongue and other Orbivirus infections into Europe: the role of biological and climatic processes. Reviews Science and Technology, 27, 427-442.
- PURSE, B.V., MELLOR, P.S., ROGER, D.J., SAMUEL, A.R., MERTEMS, P.P. & BAYLIS, M., 2005. Climate change and the recent emergence of bluetongue in Europe. Nature Reviews Microbiology, 3, 171-181.
- RAMADEVI, N., RODRIGUES, J. & ROY, P., 1998. A leucine zipper-like domain is essential for dimerization and encapsidation of bluetongue virus nucleocapsid protein VP4. Journal of Virology, 72, 2983–2990.
- RATINIER, M., SHAW, A.E., BARRY, G., GU, Q., DI GIALLEONARDO, L., JANOWICZ, A., VARELA, M., RANDALL, R.E., CAPORALE, M. & PALMARINI, M., 2016. Bluetongue virus NS4 protein is an interferon antagonist and a determinant of virus virulence. Journal of Virology, 90, 5427-5439.
- REVIRIEGO GORDEJO, F.J., 2013. Bluetongue in Europe: Lessons learned. Proceedings U.S. Animal Health Association, 117, 141-142.
- RICHARDS, W.P.C. & CORDY, D.R., 1967. Bluetongue virus infection: Pathologic responses of nervous systems in sheep and mice. Science, 156, 530–531.
- RICHARDS, W.P.C., CRENSHAW, G.L. & BUSHNELL, R.B., 1971. Hydranencephaly of calves associated with natural bluetongue virus infection. Cornell Veterinarian, 61, 336–339.
- RODRIGUEZ-CALVO, T., TOJAS, J.M., MARTIN, V. & SEVILLA, N., 2013. Type I interferon limits the capacity of bluetongue virus to infect hematopoietic precursors and dendritic cells in vitro and in vivo. Journal of Virology, 88, 859-867.
- RODRIGUEZ-SANCHEZ, B., IGLESIAS-MARTIN, I., MARTINEZ-AVILES, M. & SANCHEZ-VIZCAINO, J.M., 2008. Orbiviruses in the Mediterranean basin: updated epidemiological situation of bluetongue new methods for the detection of B.T.V. serotype 4. Transboundary Emerging Diseases, 55, 205–214.
- ROY, P., 1989. Bluetongue virus genetics and genome structure. Virus Research, 13, 179–206.
- ROY, P., 2017. Bluetongue virus structure and assembly. Current Opinions in Virology, 24, 115-123.
- ROY, P., BISHOP, D.H.L., LEBLOIS, H. & ERASMUS, B.J., 1994. Long-lasting protection of sheep against bluetongue challenge after vaccination with virus-like particles: evidence of homologous and partial heterologous protection. Vaccine, 12, 805–811.
- ROY, P., FRENCH, T. & ERASMUS, B.J., 1992. Protective efficacy of virus-like particles for bluetongue disease. Vaccine, 10, 28–32.
- ROY, P., HIRASAWA, T., FERNANDEZ, M., BLINOV, V.M. & SANCHEZ VIXCAIN RODRIQUE, J.M., 1991. The complete sequence of the group-specific antigen, VP7, of African horse sickness disease virus serotype 4 reveals a close relationship to bluetongue virus. Journal of General Virology, 72, 1237–1241.
- ROY, P., RITTER, D.G., AKASHI, H., COLLISON, E. & INABA, Y., 1985. A genetic probe for identifying bluetongue virus infections in vivo and in vitro. Journal of General Virology, 66, 1613–1619.
- ROY, P., URAKAWA, T., VAN DIJK, A.A. & ERASMUS, B.J., 1990. Recombinant virus vaccine for bluetongue disease in sheep. Journal of Virology, 64, 1998–2003.
- RUSANU, S., JOUNEAU, L., URIEN, C., BOURGE, M., LECARDONNEL, J., MOROLDO, M., LOUP, B., DALOD, M., ELHMOUZI-YOUNES, J., BEVILACQUA, C., HOPE, J., VITOUR, D., ZIENTARA, S., MEYER, G. & SCHWARTZ-CORNIL, I., 2013. Dendritic cell subtypes from lymph nodes and blood show contrasted gene expression programs upon Bluetongue virus infection. Journal of Virology, 87, 9333-9343.
- RUSSELL, H., O’TOOLE, D.T., BARDSLEY, K., DAVIS, W.C. & ELLIS, J.A., 1996. Comparative effects of bluetongue virus infection of ovine and bovine endothelial cells. Veterinary Pathology, 33, 319–331.
- SAEGERMAN, C., BERKVENS, D. & MELLOR, P.S., 2008. Bluetongue epidemiology in the European Union. Emerging Infectious Diseases, 14, 539-544.
- SAEGERMAN, C., BOLKAERTS, B., BARICALLA, C., RAES, M., WIGGERS, L., DE LEEUW, I., VANDENBUSSCHE, F., ZIMMER, J.Y., HAUBRUGE, E., CASSART, D., DE CLERCQ, K. & KIRSCHVINK, N., 2011. The impact of naturally-occurring, trans-placental bluetongue virus serotype-8 infection on reproductive performance in sheep. Veterinary Journal, 187, 72-80.
- SAMAL, S.K., LIVINGSTON, C.W. JR., MCCONNELL, S. & RAMIG, R.F., 1987. Analysis of mixed infection of sheep with bluetongue virus serotypes 10 and 17: Evidence of genetic reassortment in the vertebrate host. Journal of Virology, 61, 1086–1091.
- SANCHEZ-CORDON, P.J., PEDRERA, M., RISALDE, M.A., MOLINA, V., RODRIGUEZ-SANCHEZ, B., NUNEZ, A., SANCHEZ-VIZCAINO, J.M. & GOMEZ-VILLAMANDOS, J.C., 2013. Potential role of proinflammatory cytokines in the pathogenetic mechanisms of vascular lesions in goats naturally infected with bluetongue virus serotype 1. Transboundary and Emerging Diseases, 60, 252-260.
- SAPRE, S.N., 1964. An outbreak of bluetongue in goats and sheep in Maharashtra State, India. Veterinary Review, 15, 69–71.
- SARWAR, M.M., 1962. A note on bluetongue in sheep in West Pakistan. Pakistan Journal of Animal Science, 1, 1–2.
- SAVINI, G., AFONSO, A., MELLOR, P., ARADAIB, I., YADIN, H., SANAA, M., WILSON, W., MONACO, F. & DOMINGO, M., 2011. Epizootic haemorrhagic disease. Research in Veterinary Science, 91, 1–17.
- SAVINI, G., LORUSSO, A., PALADINI, C., MIGLACCIO, P., Di GENNARO, A., Di PROVIDO, A., SCACCHIA, M., & MONACO, F., 2014. Bluetongue serotype 2 and 9 modified live vaccine viruses as causative agents of abortion in livestock: a retrospective analysis in Italy. Transboundary Emerging Diseases, 61, 69–74.
- SAVINI, G., PUGGIONI, G., MELONI, G., MARCACCI, M., Di DOMENICO, M., ROCCHIGIANI, A.M., SPEDICATO, M., OGGIANO, A., MANUNTA, D., TEODORI, L., LEONE, A., PORTANTI, O., CITO, F., CONTE, A., ORSINI, M., CAMMA, C., CALISTRI, P., GIOVANNINI A. & LARUSSO, A., 2017. Novel putative bluetongue virus in healthy goats from Sardinia, Italy. Infection, Genetics and Evolution, 51, 108-117.
- SCHMIDT, R.E. & PANCIERA, R.J., 1973. Cerebral malformation in fetal lambs from a bluetongue-enzootic flock. Journal of the American Veterinary Medical Association, 162, 567–573.
- SCHULZ, C., BREARD, E., SAILLEAU, C., JENCKEL, M., VIAROUGE, C., VITOUR, D., PALMARINI, M., GALLOIS, M., HOPER, D., HOFFMANN, B., BEER, M. & ZIENTARA, S., 2016. Bluetongue virus serotype 27: detection and characterization of two novel variants in Corsica, France. Journal of General Virology, 97, 2073 - 2083.
- SELLERS, R.F., 1975. Bluetongue in Cyprus. Australian Veterinary Journal, 51, 198–203.
- SELLERS, R.F., 1977. Spread of African horse sickness and bluetongue in North African and Middle Eastern countries. International Symposium on Reoviridae, Guelph.
- SELLERS, R.F., 1981. Bluetongue and related diseases. In: GIBBS, E.P.J., (ed). Virus Diseases of Food Animals: A World Geography of Epidemiology and Control. London: Academic Press, 567–583.
- SENDOW, I., DANIELS, P.W., SOLEHA, E., ERASMUS, B. & RONOHARDJO, S.P., 1993. Isolation of bluetongue virus serotypes new to Indonesia from sentinel cattle in West Java. The Veterinary Record, 133, 166–168.
- SHAD, G., WILSON, W.C., MECHAM, J.O. & EVERMANN, J.F., 1997. Bluetongue virus detection: A safer reverse-transcriptase polymerase chain reaction for prediction of viremia in sheep. Journal of Veterinary Diagnostic Investigation, 9, 118–124.
- SHARIFAH, S.H., ALI, M.A., GARD, G.P. & POLKINGHORNE, I.G., 1995. Isolation of multiple serotypes of bluetongue virus from sentinel livestock in Malaysia. Tropical Animal Health and Production, 27, 37–42.
- SINGH, E.L., DULAC, G.C. & HENDERSON, J.M., 1997. Embryo transfer as a means of controlling the transmission of viral infections. XV. Failure to transmit bluetongue virus through the transfer of embryos from viraemic sheep donors. Theriogenology, 47, 1205–1214.
- SLUIJS, M., VAN DER TIMMERMANS, M., MOULIN, V., NOORDEGRAAF, C. V., VRIJENHOEK, M., DEBYSER, I., DE SMIT, A.J. & MOORMANN, R., 2011. Transplacental transmission of Bluetongue virus serotype 8 in ewes in early and mid-gestation. Veterinary Microbiology, 149, 113–125.
- SLUIJS, M.T.,VAN DER SCHROER-JOOSTEN, D.P., FID-FOURKOUR, A., VRIJENHOEK, M.P., DEBYSER, I., MOULIN, V., MOORMANN, R.J. & DE SMIT, A.J., 2013. Transplacental transmission of bluetongue virus serotype 1 and serotype 8 in sheep: virological and pathological findings. PLoS One 8, e81429.
- SPREULL, J., 1902. Report from veterinary surgeon Spreull on the result of his experiments with the malarial catarrhal fever of sheep. Agricultural Journal of the Cape of Good Hope, 20, 469–477.
- SPREULL, J., 1905. Malarial catarrhal fever (Bluetongue) of sheep in South Africa. Journal of Comparative Pathology, 18, 321–337.
- SQUIRE, K.R.E., CHUANG, R.Y., CHUANG, L.F., DOI, R.H. & OSBURN, B.I., 1985. Detecting bluetongue virus RNA in cell culture by dot hybridization with a cloned genetic probe. Journal of Virological Methods, 10, 59–68.
- STAIR, E.L., 1968. The pathogenesis of bluetongue in sheep: A study by immunofluorescence and histopathology. Dissertation, Texas A&M University, 5, 164.
- STANDFAST, H.A., DYCE, A.L. & MULLER, M.J., 1985. Vectors of bluetongue virus in Australia. In: BARBER, T.L., JOCHIM, M.M. & OSBURN, B.I., (eds). Bluetongue and Related Orbiviruses, New York: Alan R. Liss, 177‑186.
- STANISLAWEK, W.L., LUNT, R.A., BLACKSELL, S.D., NEWBERRY, K.M., HOOPER, P.T. & WHITE, J.R., 1996. Detection by ELISA of bluetongue antigen directly in the blood of experimentally infected sheep. Veterinary Microbiology, 52, 1–12.
- STAUBER, N., MARTINEZ COSTAS, J., SUTTON, G., MONASTYRSKAYA, K. & ROY, P., 1997. Bluetongue virus VP6 protein binds ATP and exhibits an RNA-dependent ATPase function and a helicase activity that catalyze the unwinding of double-stranded RNA substrates. Journal of Virology, 71, 7220–7226.
- STEWART, M., HARDY, A., BARRY, G., PINTO, R.M., CAPORALE, M., MELZI, E., HUGHES, J., TAGGART, A., JANOWICZ, A., VARELA, M., RATINIER, M. & PALMARINI, M., 2015. Characterization of a second open reading frame in genome segment 10 of bluetongue virus. Journal of General Virology, 96, 3280–3293.
- STEYN, J., VENTER, G.J., LABUSCHAGNE, K., MAJATLADI, D., BIOKANYO, S.N., LOURENS, C., EBERSOHN, K., &VENTER, E.H., 2016. Possible over-wintering of bluetongue virus in Culicoides populations in the Onderstepoort area, Guateng, South Africa. Journal South African Veterinary Medical Association, 87, e1-e5.
- STOTT, J.L., BARBER, T.L. & OSBURN, B.I., 1985. Immunologic response of sheep to inactivated and virulent bluetongue virus. American Journal of Veterinary Research, 46, 1043–1049.
- STOTT, J.L., OSBURN, B.I. & ALEXANDER, L., 1985. Ornithodoros coriaceus (Pajaroello tick) as a vector of bluetongue virus. American Journal of Veterinary Research, 46, 1197–1199.
- STUDDERT, M.J., PANGBORN, J. & ADDISON, R.B., 1966. Bluetongue virus structure. Virology, 29, 509–511.
- SUN, E.C., HUANG, L.P., XU, Q.Y., WANG, H.X., XUE, X.M., LU, P., LI, W.J., LIE, W. BU, Z.G. & WU, D.L., 2016. Emergence of a novel bluetongue virus serotype, China 2014. Transboundary and Emerging Diseases, 6, 585-589.
- SUTMOLLER, P. & WRATHALL, A.E., 1997. A quantitative assessment of the risk of transmission of foot-and-mouth disease, bluetongue and vesicular stomatitis by embryo transfer in cattle. Preventive Veterinary Medicine, 32, 111–132.
- SUTMOLLER, P. & WRATHALL, A.E., 1997. The risks of disease transmission by embryo transfer in cattle. Revue Scientifique et Technique, 16, 226–239.
- SVEHAG, S.-E., LEENDERTSEN, L. & GORHAM, J.R., 1966. Sensitivity of bluetongue virus to lipid solvents, trypsin and pH changes and its serological relationship to arboviruses. Journal of Hygiene, 64, 339–346.
- TABACHNICK, W.J., 2004. Culicoides and the global epidemiology of bluetongue virus infection. Veterinaria Italiana, 40, 145-150.
- TANAKA, S. & ROY, P., 1994. Identification of domains in bluetongue virus VP3 molecules essential for the assembly of virus cores. Journal of Virology, 68, 2795–2802.
- TANYA, V.N., GRIENER, E.C. & GIBBS, E.P.J., 1992. Evaluation of Culicoides insignis (Diptera: Ceratopogonidae) as a vector of bluetongue virus. Veterinary Microbiology, 32, 1–14.
- THEILER, A., 1906. Bluetongue in sheep. Annual Report of the Director of Agriculture, Transvaal for 1904–1905, 110–121.
- THEILER, A., 1908. The inoculation of sheep against bluetongue and the results in practice. Veterinary Journal, 64, 600–607.
- THERON, J., UITENWEERDE, J.M., HUISMANS, H. & NEL, L.H., 1994. Comparisons of the expression and phosphorylation of the non-structural protein NS2 of three different orbiviruses: Evidence for the involvement of an ubiquitous cellular kinase. Journal of General Virology, 75, 3401–3411.
- THOMAS, A.D., & NEITZ, W.O., 1947. Further observations on the pathology of bluetongue in sheep. Onderstepoort Journal of Veterinary Science and Animal Industry, 22, 27–40.
- TSUBOI, T. & IMADA, T., 1997. Effect of bovine herpes-1, bluetongue virus and Akabane virus on the in vitro development of bovine embryos. Veterinary Microbiology, 57, 135–142.
- TUSTIN, R.C., 1990. Faculty of Veterinary Science, University of Pretoria. Unpublished data.
- VAN DEN ENDE, M., LINDER, A. & KASCHULA, V.R., 1954. Experiments with the Cyprus strain of bluetongue virus: Multiplication in the central nervous system of mice and complement fixation. Journal of Hygiene (Camb.), 52, 155–164.
- VAN DIJK, A.A. & HUISMANS, H., 1980. The in vitro activation and further characterization of the bluetongue virus-associated transcriptase. Virology, 104, 347–356.
- VAN DIJK, A.A., 1993. Development of recombinant vaccines against bluetongue. Biotechnology Advances, 11, 1–12.
- VAN RIJN, P.A., VAN DE WATER, S.G., MARIS-VELDHUIS, M.A. & VAN GENNIP, R.G., 2016. Experimental infection of small ruminants with bluetongue virus expressing Toggenburg Orbivirus proteins. Veterinary Microbiology, 192, 145-151.
- VAN STADEN, V. & HUISMANS, H., 1991. A comparison of the genes which encode non-structural NS3 of different orbiviruses. Journal of General Virology, 72, 1073–1079.
- VAN STADEN, V., THERON, J., GREYLING, B.J., HUISMANS, H. & NEL, L.H., 1991. A comparison of the nucleotide sequences of cognate NS2 genes of three different orbiviruses. Virology, 185, 500–504.
- VENTER, E.H., VILJOEN, G.J., NEL, L.H., HUISMANS, H. & VAN DIJK, A.A., 1991. A comparison of different genomic probes in the detection of virus-specified RNA in Orbivirus infected cells. Journal of Virological Methods, 32, 171–180.
- VERCAUTEREN, G., MIRY, C., VANDENBUSSCHE, F., DUCATELLE, R., VAN DER HEYDEN, S., VANDEMEULEBROUCKE, E., DE LEEUS, I., DEPREZ, P., CHIERS, K. & DE CLERCQ, K., 2008. Bluetongue virus serotype 8-associated congenital hydranencephaly in calves. Transboundary and Emerging Diseases, 55, 293-298.
- VERWOERD, D.W., 1969. Purification and characterization of bluetongue virus. Virology, 38, 203–212.
- VERWOERD, D.W., 1970. Diplornaviruses: A newly recognised group of double-stranded RNA viruses. Progress in Medical Virology, 12, 192–210.
- VERWOERD, D.W., ELS, H.J., DE VILLIERS, E.M. & HUISMANS, H., 1972. Structure of the bluetongue virus capsid. Journal of Virology, 10, 783–794.
- VERWOERD, D.W., HUISMANS, H. & ERASMUS, B.J., 1979. Orbiviruses. In: FRAENKEL-CONRAT, H. & WAGNER, R.R., (eds). Comprehensive Virology, 14, 285–345.
- VITOUR, D., DOCEUL, V., RUSCANU, S., CHAUVEAU, E., SCHWARTZ-CORNIL, I. & ZIENTARA, S., 2014. Induction and control of the type I interferon pathway by Bluetongue virus. Virus Research, 182, 59-70.
- VOGTLIN, A., HOFMANN, M.A., NENNIGER, C., RENZULLO, S., STEINRIGL, A., LOITSCH, A., SCHWERMER, H., KAUFMANN, C. & THUR, B., 2013. Long-term infection of goats with bluetongue virus serotype 25. Veterinary Microbiology, 166, 165-173.
- WADE EVANS, A.M., 1992. The complete sequence of genome segment 8 of bluetongue virus, serotype 1, which encodes the non-structural protein, NS2. Gene, 118, 295–296.
- WADE EVANS, A.M., MERTENS, P.P.C. & BELSHAM, G.J., 1992. Sequence of genome segment 9 of bluetongue virus (serotype 1, South African) and expression analysis demonstrating that different forms of VP6 are derived from initiation of protein synthesis at two distinct sites. Journal of General Virology, 73, 3023–3026.
- WADE EVANS, A.M., ROMERO, C.H., MELLOR, P., TAKAMATSU, H., ANDERSON, J., THEVASAGAYAM, J., FLEMING, M.J., MERTENS, P.P.C. & BLACK, D.N., 1996. Expression of the major core structural protein (VP7) of bluetongue virus, by a recombinant capripox virus, provides partial protection of sheep against a virulent heterotypic bluetongue virus challenge. Virology, 220, 227–231.
- WALTON, T.E., 2004. The history of bluetongue and a current global overview. Veterinaria Italiana, 40, 31-38.
- WHETTER, L.E., MACLACHLAN, N.J., GEBHARD, D.H., HEIDNER, H.W. & MOORE, P.F., 1989. Bluetongue virus infection of bovine monocytes. Journal of General Virology, 70, 1663-1676.
- WILBUR, L.A., EVERMANN, J.F., LEVINGS, R.L., STOLL, I.R., STARLING, D.E., SPILLERS, C.A., GUSTAFSON, G.A. & MCKIERNAN, A.J., 1994. Abortion and death in pregnant bitches associated with a canine vaccine contaminated with bluetongue virus. Journal of the American Veterinary Medical Association, 204, 1762–1765.
- WILLIAMSON, S., WOODGER, N. & DARPEL, K., 2008. Differential diagnosis of bluetongue in cattle and sheep. Practise 30, 242–251.
- WILSON, A. & MELLOR, P., 2008. Bluetongue in Europe: vectors, epidemiology and climate change. Parasitology Research, 103 (Suppl 1), S69-77.
- WILSON, A.J. & MELLOR, P.S., 2009. Bluetongue in Europe: past, present and future. Philosophical Transactions of the Royal Society B-Biological Sciences 364, 2669–2681.
- WILSON, W.C. & CHASE, C.C.L., 1993. Nested and multiplex polymerase chain reactions for the identification of bluetongue virus infection in the biting midge, Culicoides variipennis. Journal of Virological Methods, 45, 39–47.
- WILSON, W.C., 1991. Molecular comparison of VP3 from bluetongue and epizootic haemorrhagic disease viruses. Virus Research, 21, 225–236.
- WOUDA, W., ROUMEN, M.P., PEPERKAMP, N.H., VOS, J.H., VAN GARDEREN, E. & MUSKENS, J., 2008. Hydranencephaly in calves following the bluetongue serotype 8 epidemic in the Netherlands. Veterinary Record, 162, 422-423.
- XU, G., WILSON, W., MECHAM, J., MURPHY, K., ZHOU, E.M. & TABACHNICK, W., 1997. VP7: An attachment protein of bluetongue virus for cellular receptors in Culicoides variipennis. Journal of General Virology, 78, 1617–1623.
- YAVRU, S., OZTURK, F., GURHAN, I., UNVER, G., DUMAN, R. & YAPKIC, O., 1997. Serological survey for respiratory tract viruses in sheep. Veterinarium, 8, 38–44.
- ZANELLA, G., MARTINELLE, L., GUYOT, H., MAUROY, A., DE CLERCQ, K. & SAEGERMAN, C., 2013. Clinical pattern characterization of cattle naturally infected by BTV-8. Transboundary and Emerging Diseases, 60, 231-237.
- ZIENTARA, S., SAILLEAU, C., VIAROUGE, C., HOPER, D., BEER, M., JENEKEL, M., HOFFMANN, B., ROMEY, A., BAKKALI-KASSIMI, L., FABLET, A., VITOUR, D. & BREARD, E., 2014. Identification of a novel bluetongue virus in goats, Corsica, France, 2014. Transboundary Emerging Diseases, 20, 2123-2125.
- ZIENTARA, S. & SANCHEZ-VIZCAINO, J.M., 2013. Control of bluetongue in Europe. Veterinary Microbiology, 165, 33-37.