- Infectious Diseases of Livestock
- Part 2
- Hog cholera
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Hog cholera
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Introduction
Hog cholera (HC) is a highly contagious viral disease of pigs that can run an acute, subacute, chronic, late onset or clinically inapparent course. The most typical feature of HC is that it is so atypical. Mortality figures vary from nil to almost 100 per cent. The interplay between viral and host factors largely determines the course and outcome of the infection. Under natural conditions, the domestic pig and the wild boar (Sus scrofa ferus) are the only animal species susceptible to infection with HC virus (HCV).
The first presumptive HC outbreaks were reported from Tennessee and Ohio in the USA in the early nineteenth century, whereas HCV first appeared in Europe in the middle of that century. Although the infection was first reported in South Africa in 1900,13 the country has been free of the disease since 1918. In 1903, De Schweinetz and Dorset showed that HC is caused by a filtrable agent.17
Hog cholera is classified by the Office International des Epizooties as a list A disease59 because it is a highly devastating disease that jeopardizes international trade of pigs and pig products, and leads to substantial economic losses for the industry. The disease is prevalent in Europe, Asia and South America. The African continent is free, except for Madagascar. Many countries, such as the USA, Canada, and various European countries, have eradicated the virus, while others have stringent eradication programmes in force. Nevertheless, HCV still gave rise to many outbreaks in Europe in the 1990s. Severe outbreaks plagued Belgium and Germany in 1994. An epidemic in the Netherlands in 1997 resulted in the destruction of a total of 11 million pigs and an estimated loss of two billion US dollars.
Aetiology
Hog cholera virus belongs to the family Flaviviridae, and the genus Pestivirus. The two other members of this genus are: bovine viral diarrhoea virus (BVDV) and border disease virus (BDV). Hog cholera virus has an RNA genome that is contained in a capsid of about 28 nm in diameter, which in turn is surrounded by an envelope. Fringe-like projections of 6 to 8 nm have been demonstrated on the virion surface. The virion measures between 40 and 50 nm in diameter.
The single-stranded linear RNA genome is infective and encompasses about 12,3 kilobases. There is one open reading frame that encodes one large polyprotein of 3 898 amino acids that is cleaved by proteases to yield mature viral proteins. 50, 54 The open reading frame is flanked by a 5’ noncoding region of almost 400 nucleotides and a 3’ non-coding region of about 200 nucleotides. The order of the gene products is as follows:
NH2-(Npro-C-Erns-E1-E2-p7-NS2.3-NS4A-NS4B-NS5ANS5B)- COOH
The left part of the genome codes for the capsid (C) protein, and the three envelope glycoproteins Erns, E1 and E2. The Erns protein has RNase activity,31, 66 which is unique among virus proteins, and is immunosuppressive in vitro.5 The E1 protein resides as a disulfide-linked E1-E2 heterodimer in the viral envelope, and the E2 also forms a disulfide-linked homodimer. The E2 is the most immunodominant protein and is composed of two independently formed antigenic domains.90 The p7 protein is probably a non-structural protein. The right larger part of the genome solely codes for non-structural proteins. The NS2,3 protein is the most conserved among the pestiviruses.
Hog cholera virus can be differentiated from BVDV and BDV on the basis of differences found in the 5’ non-coding region, the E2 gene and a non-structural protein.28, 34, 91, 94, 102 Although HCV is genetically more stable than BVDV, its strains have been divided into two groups on the basis of differences in nucleotide sequences of the 5’ non-coding region, the N-terminal part of E2 and a region of NS5B.41, 95, 97 Subgroups within groups 1 and 2 have also been distinguished.26,41 The amino acid similarity between HCV and BVDV is about 70 per cent, indicating that HCV shares a common evolutionary history with BVDV.60 The highest degree of homology exists between the NS3 part of the non-structural NS2,3 protein, the lowest between the E2 sequences. Antigenically, there is a close relationship of HCV with the other two pestiviruses, as can be demonstrated by various serological methods, but a clear distinction can still be made. Antigenic variation, as demonstrated by the use of monoclonal antibodies (MAbs), exists among HCV strains, but all HCV strains belong to one antigenic group.20, 21, 100, 101
The resistance of HCV to physical and chemical treatment is partly dependent on the strain of the virus and the material that contains the virus. For instance, in cell culture fluid virus was inactivated in 10 minutes at 60 °C,38 whereas it was not inactivated in defibrinated blood at 68 °C for 30 minutes.79 The viral infectivity is quickly destroyed below pH 4 and above pH 11, although the inactivation rate below pH 5 is dependent on the temperature: at pH 4 a half-life of 260 hours was found at 4 °C and 11 hours at 21 °C.14 Solvents such as chloroform and ether rapidly inactivate HCV, because its envelope contains lipids. The virus can remain infectious in pork and pork products for months, whereas in most environments outside the host or its tissues it is usually no longer infectious after a few days. However, in liquid pig manure the virus may survive for weeks.25 For disinfection, 1 to 2 per cent sodium hydroxide is considered most suitable.
Porcine cells, whether primary or secondary cell cultures, or established cell lines, are generally used for propagation of HCV. Virus replication is restricted to the cytoplasm of the cell and basically induces no or minimal cytopathology except in porcine bone marrow stroma cell cultures in which HCV produces distinct cytopathic effects. 67 Recently, HCV isolates and clones were obtained that contained defective interfering particles and which gave rise to a cytopathic effect in cell culture.37, 49, 52 Inactivation of the RNase activity of the Erns protein also resulted in the generation of a cytopathic effect in cell culture.32
The virions appear to assemble and mature at intracytoplasmic membranes and may be released via exocytosis. The major portion of infectious virus remains cell associated. The virus spreads from cell to cell and from mother to daughter cells in the presence of immune serum. A unique phenomenon is that the growth of Newcastle disease virus is enhanced in porcine testis cells infected with HCV.39
There is a wide variety of virulence among HCV strains. In vitro markers for virulence have not yet been well established and therefore the virulence of a virus can only be assessed by infecting pigs experimentally under standardized conditions. Highly virulent strains induce acute severe disease and high mortality, whereas strains of low virulence give rise to mild disease or subclinical infection. The latter strains may, however, be pathogenic for porcine foetuses. Particularly in infections with strains of moderate virulence, host factors such as breed, age, nutritional condition and immune competence appear to partially determine the course of the disease.16, 86 The virulence of HCV seems to be an unstable property because enhancement of virulence after one or multiple passages in pigs has repeatedly been reported.19 The genes responsible for the expression of virulence and the molecular basis for attenuation of the HCV vaccine strains remain to be determined.
Epidemiology
The domestic pig and wild boar are the only animals that become naturally infected with HCV, and thus are the sole source of virus spread. The oronasal route of infection is most common. Pigs infected with virulent virus that develop acute to subacute disease may shed virus before the onset of clinical signs of the disease and continue to do so until they die or recover. Pigs that develop chronic HC may shed the virus continuously or intermittently until death.46 Virus is shed in all secretions and excretions, notably in saliva, nasal and ocular discharges, urine and faeces.64, 65
Postnatal infections with strains of low virulence are characterized by short periods of virus shedding, usually until antibodies appear in the blood. Infection of pregnant sows with these strains gives rise to foetal infection. Such congenital infections may result in the birth of persistently infected healthy piglets that shed virus continuously for months until they eventually die.87 Thus, infections with HCV of low virulence may smoulder in a herd for a long period before a diagnosis is made and are therefore important in virus perpetuation. Virulent HCV infections will usually spread fast in a herd and induce high morbidity and mortality, and, as a result, will be recognized more easily and rapidly.
Direct contact between pigs is the most common mode of HCV transmission. Herd-to-herd transmission is most commonly effected by the introduction of pigs that are either shedding the virus during the incubation period or are persistently infected. Pigs can also become infected in the market-place and during transport in contaminated trucks. Boars may shed HCV in their semen which can infect sows.18 Humans appear to play a significant role in transmitting the virus within and between herds. Farmers, inseminators and veterinarians may transmit the virus by means of contaminated instruments and medicine bottles. Contaminated clothing and footwear would appear to be less important in this respect.
Although replication of HCV has not been demonstrated in arthropods, several species of flies and mosquitoes may act as mechanical vectors of the virus. In this respect, a role played by dogs, cats and birds in the spread of HCV cannot be excluded, but there is no convincing experimental evidence for this. Aerogenic transmission has been established under experimental conditions and seems to occur inside a barn,29, 40, 75 but this form of spread between herds has not yet been proved although it is not unlikely in regions with a high population density of pigs where there are many large piggeries each equipped with strong mechanical ventilation units in close proximity to each other.
Important sources of virus dissemination are pork and pork products, including ham, bacon and sausages, because the virus is not always inactivated by the various processing procedures to which they are subjected. When pork and pork products are stored, cooled or frozen, survival of the virus can be prolonged for months. Transport of such products may be over long distances and in this way HCV may cause outbreaks in hitherto free countries if swill or kitchen leftovers are not properly heat-treated before being fed to pigs. Feeding virus-containing garbage to pigs was the cause of 22 per cent of the outbreaks of HC in the USA in 1973.19
The wild boar populations of various regions in Germany, France, Italy, Austria and certain other European countries are infected with HCV and the virus can persist in them for years. This epidemiological situation constitutes a serious and permanent threat for the introduction of the virus into the domestic pig population. Most of the primary outbreaks in Germany between 1990 and 1998, which were studied in detail, were due to direct or indirect contact with infected wild boar.23
The tracing of the source of an HCV infection and the establishment of transmission routes have been markedly facilitated by the recent advances in molecular epidemiology. The genetic variability among HCV isolates and the subsequent phylogenetic analysis can be exploited to link, or exclude a connection between outbreaks in the same or different regions.26, 28, 69, 70, 93, 96
Pathogenesis
Under natural conditions, HCV enters the pig via the oral and nasal cavities. Conjunctival and genital mucous membranes and skin abrasions are uncommon portals of entry. The tonsils are the primary target organ for virus replication. The spread of highly virulent strains of HCV in a pig is characterized by lymphatic, viraemic and visceral phases. The virus initially infects epithelial cells of the tonsillar crypts and then invades the underlying lymphoreticular tissue from where it travels via lymphatic vessels to the regional lymph nodes in which it replicates and subsequently enters the efferent blood capillaries, giving rise to a viraemia. Virus proliferation in secondary target tissues, such as the spleen, visceral lymph nodes, lymphoid tissues in the intestinal wall and bone marrow results in a high level of viraemia which, in acute HC, persists until the death of the animal. A small proportion of megakaryocytes in the bone marrow appears to be infected early in the course of the infection.25 Late in the infection, immature granulocytic cells and monocytes in the blood are the major targets for HCV and T-lymphocytes may became infected.62, 71 Parenchymatous organs only become infected late in the viraemic phase. Virus titres in lymphoid tissues are generally higher than those in parenchymatous organs. The spread of highly virulent virus strains in the organs and tissues of an infected pig is usually completed within five to six days post-infection.64 The virus has a distinct affinity for vascular endothelium and cells of the immune system. A severe leukopenia and severe thrombocytopenia develop soon after infection and persist until death in acute HC.97 The leukopenia may be the result of apoptosis of activated lymphocytes.72
The widespread haemorrhages seen in acute HC are most likely caused by degeneration and necrosis of endothelial cells in conjunction with severe thrombocytopenia and disturbances in fibrinogen synthesis. The processes leading to the death of the pigs are not well understood, but the severe circulation disturbances appear to be the most likely cause.24 During the course of acute HC, alterations in immune reactivity occur. As soon as two days after infection the following changes have been reported:
- an unresponsiveness of peripheral blood and splenic lymphocytes to T- and B-cell mitogens,
- concomitant increased response of lymphocytes within lymph nodes,62, 88, 89
- B-lymphocyte deficiency and depletion in the blood,72, 73, 88 and
- depressed secondary antibody response to lysozyme.6
- Neutralizing antibodies can be detected in some pigs that die from acute to subacute HC.43, 45
Viruses of moderate or lower virulence can also induce persistent infections, which are distinguished as chronic HC or late-onset HC (Table 88.1).
In the first phase of chronic HC, the virus dissemination throughout the body resembles that of acute HC, but it occurs at a slower rate and the virus titre tends to be lower in serum and organs. In the second phase, when clinical improvement occurs, the virus titre in serum is low or absent, and viral antigen appears to be limited to epithelial cells of the tonsils, ileum, salivary glands and kidneys. In this phase there is evidence of increased plasma cell formation and increased serum immunoglobulin levels. The disappearance of virus from serum is temporary and probably the result of specific antibody production and/or of a decrease in the number of cells releasing virus. The simultaneous occurrence of virus and antibody may lead to deposition of antigen- antibody complexes in the kidneys, resulting in glomerulonephritis which occurs in many cases of this form of the disease.9 During the third phase of chronic HC, the virus again disseminates throughout the body, which may be initiated by the immune exhaustion that appears to develop. In this phase pigs seem to be more susceptible to secondary bacterial infections.8, 47 Pigs can recover from mild to severe infections with HCV strains of moderate virulence. Such animals produce neutralizing antibodies and can also elicit a cell-mediated immune response against the virus, as evidenced by lymphocyte proliferation and cytotoxic T-cell activity.12, 35, 61, 63
Late-onset HC is characterized, after infection, by an initial long period, varying from several weeks to months, of virtual absence of clinical signs. This form of the disease is the sequel of exposure of porcine foetuses to strains of low virulence during the first 40 days of gestation. Such pigs have a lifelong high level of viraemia, which may be temporarily lowered after the ingestion of colostral antibody. Hog cholera virus antigens are widespread throughout epithelial, lymphoid and reticulo-endothelial tissues, probably as a result of the absence of a specific antibody response. The factors that trigger the clinical signs that eventually appear are not known. Pigs suffering from late-onset HC are immunotolerant to the virus and have a normal to slightly depressed lymphocyte response to mitogens.84, 87
Table 88.1 General features of acute, chronic, and late-onset hog cholera
| FEATURE | ACUTE | CHRONIC | LATE-ONSET |
|---|---|---|---|
| Virulence of virus | High | Moderate | Low |
| Time of infection | Postnatal | Postnatal | Prenatal |
| Course of illness | Short incubation period, severe depression, high fever, anorexia, conjuctivitis, constipation, diarrhoea, convulsions, incoordination, haemorrhages of skin | Short incubation period, three phases of illness: (1) depression, fever, anorexia; (2) clinical improvement; (3) terminal, exacerbation of disease | Late onset of disease, gradually worsening depression and anorexia, normal to slightly elevated body temperatures, conjunctivitis, dermatitis, locomotion disturbances |
| Viraemia | High level | Temporary reduction or disappearance | Persistent high level |
| Leukopenia | Develops quickly | Develops quickly, followed by eukocytosis | Develops late during infection |
| Immune response to HCV | Absent | Present | Absent |
| Death | 10–20 days | 1–3 months | 2–11 months |
| Gross lesions | Multiple haemorrhages (especially in lymph nodes and kidneys), splenic infarcts | Ulcers in caecum and colon, splenic infarcts | Lymph node swelling, thymic atrophy, rib lesions |
| Microscopic lesions | Degeneration of endothelial cells, proliferation of reticular cells, encephalitis | Degeneration of endothelial cells severe lymphocyte depletion, histiocytic hyperplasia, glomerulonephritis | Degeneration of endothelial cells, severe lymphocyte depletion, histiocytic hyperplasia |
The multiplication of HCV strains of low virulence following postnatal infection is mainly restricted to the lymphatic phase, i.e. tonsils and their regional lymph nodes. These infections often run a subclinical course during which a transient leukopenia and lymphocyte unresponsiveness to B-cell mitogens can be found,88 but not a depressed antibody response to antigens unrelated to HCV.85 Such pigs generally elicit antibodies to HCV. In pregnant sows the virus may be transmitted via the blood to the placenta of one ormorefoetuses in which it proliferates and then spreads per continuitatum from foetus to foetus. The outcome of the foetal infection is largely determined by the age of the foetus and by the virulence of the virus. The possibility of foetal damage is higher the earlier infection takes place during pregnancy. Although foetuses gain immunocompetence at the mid-gestational stage, foetal HCV infection in the later stages of gestation may still result in them developing immunotolerance.48, 83
Two unexplained phenomena have repeatedly been observed under experimental conditions in pigs that do not mount a normal antibody response after a primary exposure to HCV. Re-exposure of them to HCV can result in a form of hyper-reactivity which is characterized by a shorter incubation period, a more severe illness and a higher mortality rate than in primarily infected pigs.86 In contrast to this sensitization- like phenomenon, an enhanced resistance to virulent HCV in persistently infected immunotolerant pigs has been described.86 It is not unlikely that both phenomena occur in the field.
The precise pathogenesis of HC is still not well understood. The disease has been interpreted as being one caused by a disorder of the chymotrypsin enzyme system.36 Certain other hypotheses have been reviewed.24, 42 The pathogenesis of HCin wild boar appears to resemble that of domestic pigs.
Clinical signs
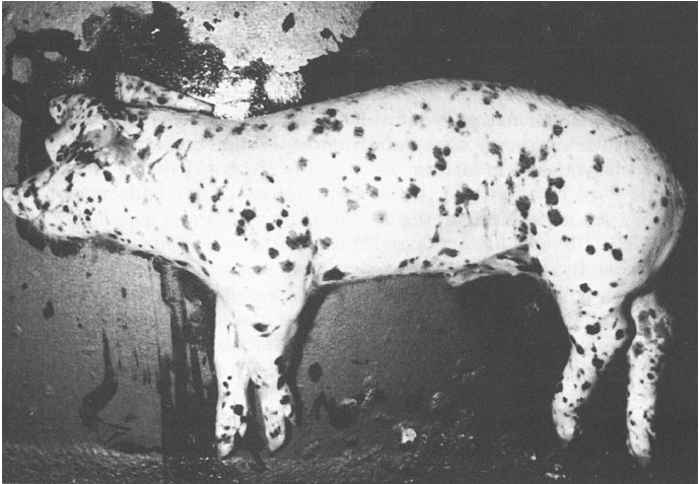
The clinical signs of HC vary from those of an acute disease with a high mortality rate to those of a disease with a subclinical course. In the acute disease, after an incubation period of two to six days, infected pigs first show fever, dullness, a reluctance to move and reduced appetite. These signs become more severe during the subsequent few days. Fever, with peak rectal temperatures above 42 °C, persists until immediately before death, when the temperature decreases to below normal. Concurrent with the fever, a leukopenia and thrombocytopenia develop that persist until death. A generalized hyperaemia of the skin is also common in acute HC. Early in the course of the disease, affected animals develop a mucopurulent conjunctivitis with a copious discharge that may cause the upper and lower eyelids to adhere to each other. Constipation usually develops during periods of high fever; this is followed by a severe watery diarrhoea which may be blood-stained. Some pigs vomit a yellowish bile-containing fluid. Affected pigs shiver and pile upon each other. Convulsions may occur in a few of them, which is usually a sign of impending death. During the terminal stages most show a typical, weaving, staggering gait which is usually followed by posterior paresis. A purplish discoloration of the skin extending over the abdomen, snout, ears and medial sides of the legs is then often seen. Most pigs suffering from acute HC die between the tenth and twentieth day of onset of clinical signs.
In subacute HC, pigs develop similar but less severe signs of illness to those of the acute disease and most succumb within 30 days.19
When pigs survive longer than 30 days, they are considered to be suffering from a persistent HCV infection.19 Persistence of infection causes several clinical forms of HC. Pigs that overcome the initial phase can develop chronic HC, during which they seldom appear healthy46 (see below). During late-onset disease, a sequel of congenital infection, infected pigs remain healthy for as long as four to six months and then develop the clinical signs typical of the acute to subacute disease characterized by the gradual development of anorexia, depression, mucopurulent conjunctivitis, diarrhoea, papulous dermatitis, leukopenia, runting, locomotion disturbances and posterior paresis. There is usually no fever. Leukocytosis may develop shortly before death.87 Intermediate forms of persistent HC exist, one of which occurs in pigs that have remained apparently healthy for long periods and have a high-titred viraemia.15 Breed-related factors can influence the clinical course of HC, as exemplified by the disease having a different course in Landrace or cross Landrace/Pietrain pigs after experimental infection.16
Chronic HC is characterized by a disease in which there are intermittent periods of anorexia, fever, diarrhoea and dermatitis, and may result in the occurrence of runts in the herd. Runts usually have large heads, small bodies, dermatitis and stand with arched backs with their hind legs positioned under the body. Three phases of chronic HC have been described.47 In the initial phase, fever, depression, anorexia and leukopenia are present; the second phase is characterized by a general clinical improvement but the leukopenia usually persists; and the third phase is one of relapse to a clinical syndrome characterized by anorexia, depression, fever and eventual death. Pigs suffering from chronic HC may survive for more than 100 days, but eventually most die.
Persistent, virtually inapparent HCV infection can also develop after postnatal infections with strains of reduced virulence.6 Low-virulent strains usually give rise to a mild disease or to subclinical infections in which leukopenia is present.
A congenital infection can result in abortion, foetal mummification or malformations of visceral organs and central nervous system as well as in stillbirths, and the birth of weak and trembling piglets and of piglets manifesting skin petechiation. Most die shortly after birth. Apparently healthy-looking piglets that are either persistently infected or have antibodies against HCV may also be born.
Pathology
The pathology of acute and subacute HC (Figure 88.1) is that of a septicaemic disease characterized by multiple haemorrhages of various sizes which are due to degeneration and necrosis op capillary endothelial cells, thrombocytopenia and disturbances in fibrinogen synthesis.42 In addition, catarrhal, fibrinous and haemorrhagic inflammatory reactions can be observed in the digestive, respiratory and urogenital tracts. The most prominent lesions are seldom concurrently present.
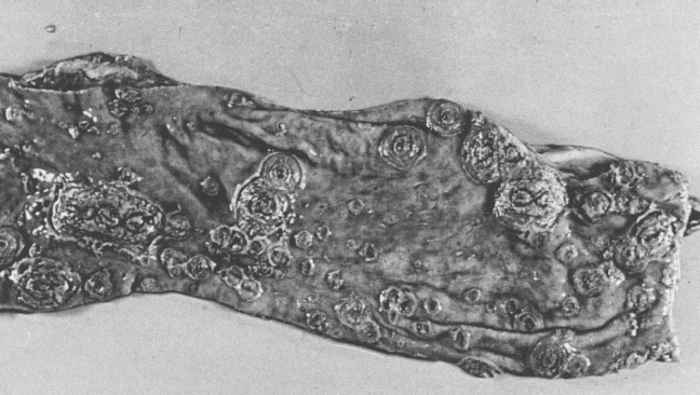
Haemorrhages are most frequently present in lymph nodes and kidneys. Lymph nodes are swollen, oedematous and show peripheral or more diffuse haemorrhages which give them a ‘marbled’ or red to near black appearance. Virtually all lymph nodes may be affected. Microscopically, lymph nodes show a lymphocytic depletion and reticular hyperplasia. Renal haemorrhages, mainly petechiae, are more common on the surface and in the cortex of the kidneys than in the medulla and hilus. Petechial to ecchymotic haemorrhages also appear in the urinary bladder, larynx, heart, intestinal mucosa and serosa, and skin. The skin may be cyanotic. Infarctions, which are the result of occlusion of small arteries by thrombi, are characteristic in the pathology of HC. The presence of splenic infarcts up to 10 mm in size are considered to be almost pathognomonic for acute HC; infarcts may also be found in the gall bladder, tonsils and lungs. The stomach is generally almost empty of contents, except for a yellow, bilous fluid. Its fundus is often congested and haemorrhagic and may contain erosions. The intestines may be hyperaemic, with diphtheroid inflammation often associated with gut-associated lymphoid tissues. In subacute to chronic HC the large intestine may contain the characteristic ‘button’ ulcers (Figure 88.2), usually associated with solitary lymph follicles. These are round and a few millimetres in diameter.80 Most HCV-infected pigs have lesions in the brain and spinal cord, which are those of a non-suppurative meningoencephalomyelitis.80
In persistent HC, haemorrhages and infarctions are less pronounced or virtually absent. Although degeneration of endothelial cells is a consistent finding, it does not lead to circulatory disturbances. The most outstanding lesions in this form of the disease are thymic atrophy and severe depletion of lymphocytes in the tonsils, lymph nodes and spleen. Histiocytic hyperplasia with phagocytosis of lymphocytic debris is also frequently seen. The plasmacytosis and glomerulonephritis that can develop in chronic HC are not seen in late-onset HC, but in both cortical hyperplasia of the adrenals occur.8, 81 Necrosis and ulcerations, sometimes in the form of ‘button’ ulcers, in the colon and caecum are common in persistent HC. Most ribs manifest a lesion which is characterized by the presence of a conspicuous transverse line of semi-solid bone near the costochondral junction.19
Congenital HCV infections can cause foetal mummification, malformation, stillbirth and other manifestations (see Clinical signs). Malformations consist of deformities of the head and limbs, such as micrognathia and arthrogryposis, hypoplasia of the cerebellum and lungs, and hypomyelogenesis. The most pronounced lesions in stillborn pigs are generalized subcutaneous oedema, hydrothorax, and ascites. In utero-infected piglets that die shortly after birth often have petechial haemorrhages in the skin and internal organs.
Diagnosis
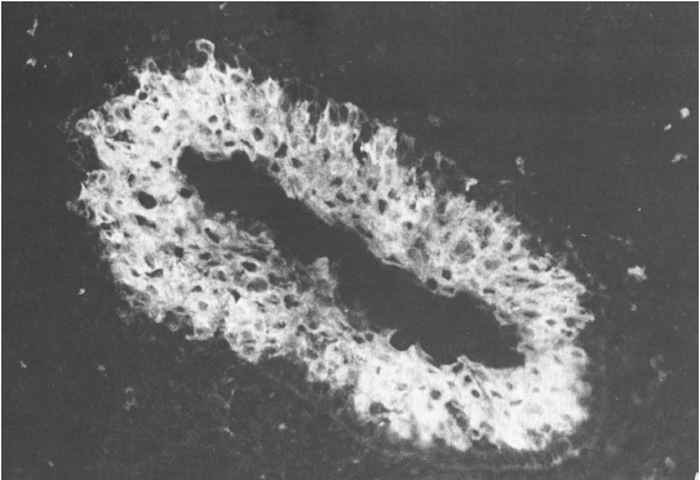
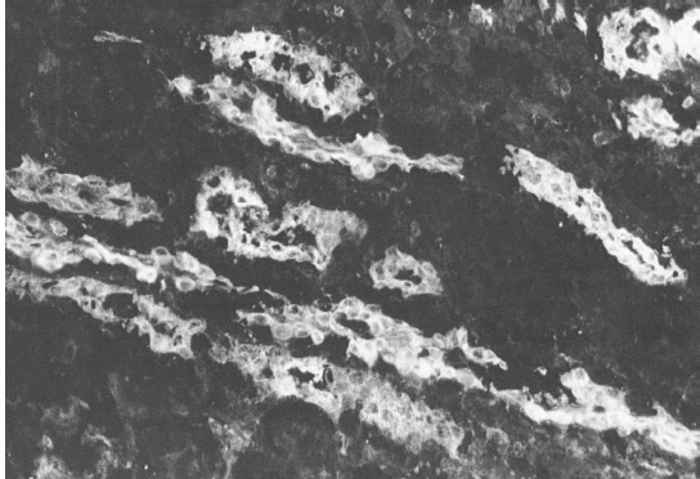
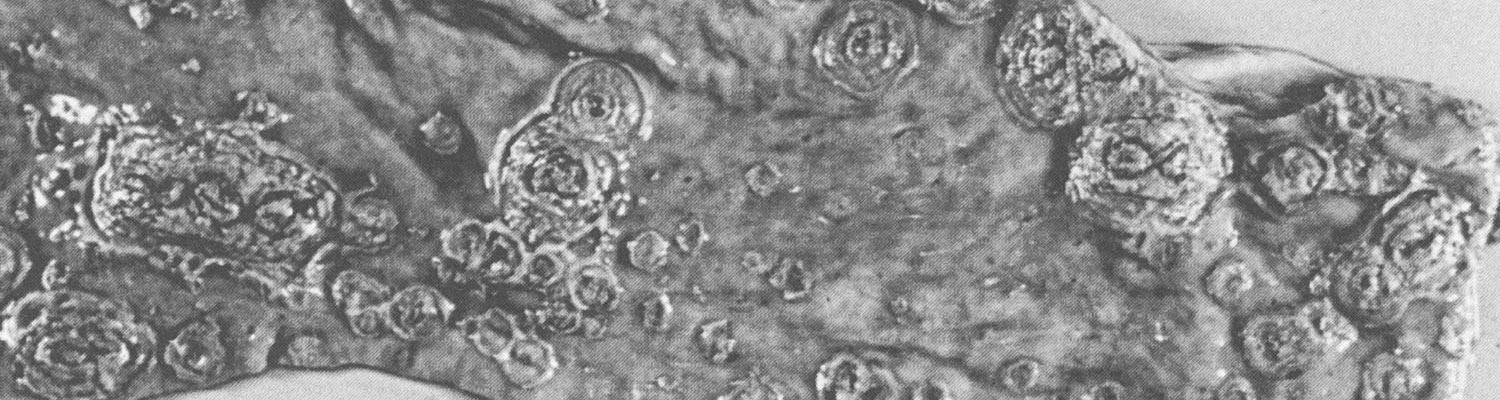
An outbreak of typical acute HC may be tentatively diagnosed in the field on the basis of an accurate anamnesis and thorough clinical and post-mortem investigations. However, it is virtually impossible to make a tentative diagnosis based on these criteria in case of subacute, chronic or lateonset HC. In all cases, when there is even just the slightest suspicion of HC, organ samples should be collected from a number of dead or moribund pigs and submitted to a laboratory. The samples should comprise tonsil, spleen, kidney and distal part of the ileum of several pigs, and should preferably be transported on ice. The direct immunofluorescence test (IFT) on frozen tissue sections is the established method of detecting viral antigen in these organs (88.3a and b). The tonsils, which are the first organs to become positive at 48 hours after oral exposure,64 are by far the most important for detecting antigen. In subacute and chronic cases the distal part of the ileum is also frequently found to be positive. The results of the IFT can be available in two hours. An absence of viral antigen does not exclude HC as the cause, and where suspicion remains additional organ samples should be examined.
Because pigs can also be infected with BVDV and BDV, and because these viruses share common antigens with HCV, the IFT can yield false-positive results. It is crucial to discriminate between HCV and the other pestivirus infections, because in many countries disease control regulations require that herds infected with HC be slaughtered out and the country concerned will lose its HC-free status. A BVDV or BDV infection erroneously diagnosed as HC can therefore have serious implications. Differentiation between these three diseases can rapidly be made by staining frozen tissue sections with conjugated monoclonal antibodies that are directed against conserved epitopes of HCV. When the MAbs bind to antigen an HCV infection is diagnosed, and when antigen cannot be detected BVDV or BDV is involved. Such non-reactive isolates, however, require confirmation that they are indeed BVDV or BDV. In countries where vaccination is practised, it is often necessary to differentiate between field virus and vaccine virus. For the differentiation of field virus from the lapinized Chinese vaccine strain, a selected pair of conjugated MAbs can be applied: a positive reaction by the first MAb and a negative by the second indicates that vaccine virus may be present.98
Lapinized vaccine strains can be further distinguished from field strains by their ability to induce fever and antibodies against HCV after intravenous inoculation into rabbits.
The method of choice of isolating HCV is by inoculation of a PK-15 cell line with a suspension comprising a 10 per cent homogenate of tonsillar and splenic tissues in phosphate buffer solution. These cell cultures are examined by the direct IFT after 24 to 72 hours. As a diagnostic tool, virus isolation is more sensitive than the direct IFT on frozen tissue sections.
Methods to detect viral genomes directly in organ samples are increasingly being used. The polymerase chain reaction (PCR) to detect portions of the HCV genome has found wide application. After RNA extraction and a reverse transcription step, the DNA is amplified and can then be detected and analysed. The PCR has already been used to differentiate between HCV and the two other pestiviruses, and has, for example, shown that an isolate from pigs with signs of HC was actually BDV.92 The PCR also enables us to quickly group the isolates on the basis of sequence homology, 28, 41, 95 which can be of importance in tracing transmission routes of viruses during epidemics. Other advantages of the PCR are that it is generally more sensitive and rapid than virus isolation, and that the test can be automated.44 A disadvantage is that it is prone to inaccuracy caused by contamination and therefore precautions have to be taken to prevent false-positive results. False-negative results may also be caused by interfering substances in the sample; it is therefore advisable that the sample be spiked with an internal control template before amplification.
Enzyme-linked immunosorbent assays (ELISAs) to detect antigen in blood or organ suspensions are relatively novel methods that are increasingly being used as diagnostic tools. Their sensitivity is, however, lower than that of virus isolation or PCR.10, 68 ELISA detection methods are particularly suited when large numbers of samples have to be screened rapidly.
Detection of HCV-specific antibodies is useful in herds suspected of being infected with strains of reduced virulence. Serological surveys are often performed during eradication programmes and to demonstrate freedom of infection in an area. Several tests to detect antibodies against HCV have been described. The virus-neutralization test in its various modifications59 is often used. This test may, however, also detect antibodies in pigs infected with BVDV or BDV. Therefore, a positive reaction should be confirmed by performing a neutralization test that measures antibodies against BVDV or BDV. Higher antibody titres against HCV than against BVDV or BDV indicate an HCV infection. Several variations of ELISAs have been developed to detect antibodies against HCV. These are much more rapid and simple to perform than neutralization tests1, 53, 99 and must detect antibodies against all HCV strains and should minimize cross-reactions with the other pestiviruses. Although some ELISAs include MAbs that only recognize HCV strains,99 positive results in ELISAs should still be confirmed in neutralization tests to differentiate between antibodies directed against HCV from those directed against BVDV or BDV.11 For reasons of efficiency and specificity, recombinant E2 antigens are increasingly being used in ELISAs.11, 57, 58
Figure 88.3a and b Virus antigen in epithelial cells of tonsillar crypts detected by direct immunofluorescence. (Reproduced with permission of Elsevier, Oxford, UK, from Virus Infections of Porcines in Atlas of Veterinary Pathology)
Differential diagnosis
Acute HC can be confused with African swine fever on clinical and pathological grounds. Differences between these two diseases are more quantitative than qualitative, except for the splenomegaly and often the haematoma-like lymph nodes that are indicative of African swine fever. Oedema of the gall bladder and major bile duct walls, and subpleural and interlobular lung oedema are rare in HC, but common in African swine fever. Septicaemic diseases, such as salmonellosis, streptococcosis, pasteurellosis and erysipelas and Haemophilus parasuis infections may be confused with HC. Generalized haemorrhages without fever may, in piglets, point to thrombocytopenia or, in all age groups, to dicumarol or other poisonings that impair blood coagulation. Runt pigs with diarrhoea as a result of chronic and late-onset HC must be differentiated from runting caused by malnutrition, enterotoxicosis by Escherichia coli or Clostridium perfringens infections, swine dysentery or campylobacteriosis. Abortions, mummifications of foetuses and still births can also be the sequelae of pseudorabies, encephalomyelocarditis virus and parvovirus infections, and the porcine reproductive and respiratory syndrome.Trembling piglets may be caused by myoclonia congenita. It is most important to differentiate HC from infections with the other pestiviruses which frequently occur in pigs.22, 77, 82
Control
Countries free of HC should take strict precautions to prevent the introduction of the disease.2 These include the banning of the importation of live pigs, pork and pork products from countries where HC is endemic or have recently suffered an outbreak or outbreaks. In addition, kitchen leftovers originating from any means of transportation that has crossed international borders and swill must either be destroyed or adequately sterilized before being fed to pigs. Nevertheless, the feeding of improperly treated swill to pigs is still often the cause of a new series of HC outbreaks. When HC is diagnosed in a country previously free of it, veterinary police and zoosanitary control measures are immediately brought into force to eradicate the virus as quickly as possible. Depending on the local situation these measures include: destruction of the infected herd (stamping out), thorough cleaning and disinfection of the premises, a ban on the movement of pigs in the neighbourhood, close surveillance of herds in the endangered area, and tracing of the source of infection and possible contacts. It usually takes one month before farms can be restocked. Countries in which sporadic HC outbreaks occur usually implement similar eradication programmes. Hog cholera is still a serious threat for many countries, because of the increasing density of pigs in many areas, the intensified transportation of pigs and their products over long distances, the HCV reservoir in wild boar populations, the dissemination of HCV in the food chain, the feeding of improperly treated swill, and outbreaks that are diagnosed too late. Vaccination is generally not applied in countries that have implemented a stamping out policy.
In countries in which HC is endemic, the introduction of HCV into a herd can be prevented by ensuring that pigs are purchased from HC-free herds and by implementing a quarantine period for incoming pigs. After four weeks, such pigs should be tested for serum antibodies and virus using isolation techniques, and if found negative can be admitted to the herd. Strict hygiene is another very important control measure. Thorough cleaning and disinfection of pens, vehicles and other objects, and, apart from the staff, strict restriction of access to the piggery by visitors and others are too often neglected as means of preventing the introduction of HCV into a herd.
In many HC-endemic countries, vaccination with live attenuated vaccines is practised on a large scale. These are based on the lapinized Chinese strain, the cell culture-adapted Japanese GPE-strain, or the French PK-15 cell adapted Thiverval strain. The Chinese strain has an insertion of 13 nucleotides in the 3’ non-coding region as compared with that of virulent HC strains.55 The GPE-strain differs in 225 nucleotides from its parental strain.33
These three vaccines are highly efficacious. They confer a rapid and long-lasting immunity, provided that each dose of vaccine contains a sufficient quantity of virus to induce an adequate level of protection. Four to five days after one vaccination, pigs will be protected against experimental challenge, and this vaccinal immunity may last two to three years, and may even be life-long. Because maternal antibodies strongly suppress the generation of active immunity, piglets that are first vaccinated at an age when they still possess maternal antibodies need to be revaccinated when these have waned (at about eight weeks of age). Vaccination does not always prevent replication of virulent HCV upon challenge.3 Vaccines based on the Chinese strain (there are several so-called Chinese strains in circulation) are most frequently used. The multiplication of this vaccine strain(s) is mainly restricted to lymphoid tissues, notably the tonsils74 and can cross the placenta but does not appear to produce abnormalities in exposed foetuses.78 All the experimental evidence to date has shown that this vaccine is safe to use. Vaccination programmes may cease to be implemented in countries in which the disease is endemic when no more outbreaks occur or when a stage is reached where stamping out and additional measures alone may lead to eradication of the residual virus.
In some countries, vaccination schemes have been implemented to prevent further spread of HCV from infected areas and thus to support the eradication programme. In the Netherlands, such a strict emergency vaccination regime with a vaccine containing the Chinese strain and which was pursued for one year has repeatedly been successful.76 A non-vaccination policy has been adopted by the European Union, because systematic vaccination will preclude the achievement of the highest health status within the Union, and because it hampers free international trade. Therefore, the use of emergency vaccinations is strongly discouraged. Vaccination has not been applied in the European Union since 1990.
A great disadvantage of vaccination with live attenuated vaccines is that it precludes the use of serological methods for diagnostic and epidemiogical purposes because the antibody response after natural infection cannot be distinguished from that after vaccination. To solve this problem, so-called diva (Differentiating Infected from Vaccinated Animals; also called marker vaccines) E2 subunit vaccines have been developed.
For this purpose, a large part of the gene encoding the E2 envelope protein of HCVis cloned in baculovirus and this recombinant virus is subsequently grown in insect cell cultures, resulting in the release of the E2 protein into the supernatant fluid. The E2 is then formulated with an adjuvant.30 These vaccines have been found to be efficacious and safe.4, 55
ELISAs have been developed that detect antibodies against the Erns protein and these can be used to detect infected pigs in vaccinated populations. A novel HCV diva vaccine may also be based on a genetically engineered live deletion mutant virus. The engineering of full-length cDNA of the genomes of the Cstrain, 56 and a virulent strain51 from which infectious RNA can be transcribed, provides the basis for the development of live diva vaccines. It may be expected that diva vaccines will have a profound impact on the control of HC.
References
- AFSHAR, AL., DULAC, G.C. & BOUFFARD, A., 1989. Application of peroxidase labelled antibody assay for detection of porcine IgG antibodies to hog cholera and bovine viral diarrhea viruses. Journal of Virological Methods, 23, 253–262.
- ANON, 2000. International Animal Health Code of the Office International des Epizooties, Paris.
- BIRONT, P., LEUNEN, J. & VANDEPUTTE, J., 1987. Inhibition of virus replication in the tonsils of pigs previously vaccinated with a Chinese strain vaccine and challenged oronasally with a virulent strain of classical swine fever virus. Veterinary Microbiology, 14, 105–113.
- BOUMA, A., DE SMIT, A.J., DE KLUIJVER, E.P., TERPSTRA, C. & MOORMANN, R.J.M., 1999. Efficacy and stability of a subunit vaccine based on glycoprotein E2 of classical swine fever virus. Veterinary Microbiology, 66, 101–114.
- BRUSCHKE, C.J.M., HULST, M.M., MOORMANN, R.J.M., VAN RIJN, P.A. & VAN OIRSCHOT, J.T., 1997. Glycoprotein Erns of pestiviruses induces apoptosis in lymphocytes of several species. Journal of Virology, 71, 6692–6696.
- CARBREY, E.A., STEWART, W.C., KRESSE, J.I. & SNYDER, M.L. 1977. Inapparent hog cholera infection following the inoculation of field isolates. In: CEC Seminar on Hog Cholera/Classical Swine Fever and African Swine Fever, Hannover, EUR 5904, pp. 214–230.
- CHARLEY, B., CORTHIER, G., HOUDAYER, M. & ROUZE, P., 1980. Modifications des reactions immunitaires au cours de la peste porcine classique. Annales des Recherches Véterinaires, 11, 27.
- CHEVILLE, N.F. & MENGELING, W.L., 1969. The pathogenesis of chronic hog cholera (swine fever). Histologic, immunofluorescent, and electron microscopic studies. Laboratory Investigation, 20, 261–274.
- CHEVILLE, N.F., MENGELING, W.L. & ZINOBER, M.R., 1970. Ultrastructural and immunofluorescent studies of glomerulonephritis in chronic hog cholera. Laboratory Investigation, 22, 458–467.
- CLAVIJO, A., ZHOU, E.M., VYDELINGUM, S. & HECKERT, R., 1998. Development and evaluation of a novel antigen capture assay for the detection of classical swine fever virus antigens. Veterinary Microbiology, 60, 155–168.
- COLIJN, E.O., BLOEMRAAD, M. & WENSVOORT, G., 1997. An improved ELISA for the detection of serum antibodies directed against classical swine fever virus. Veterinary Microbiology, 59, 15–25.
- CORTHIER, G., 1978. Cellular and humoral immune-response in pigs given vaccinal and chronic hog cholera viruses. American Journal of Veterinary Research, 39, 1841–1844.
- DE KOCK, G., ROBINSON, E.M. & KEPPEL, J.J.G., 1940. Swine fever in South Africa. Onderstepoort Journal Veterinary Science and Animal Industry, 14, 31–93.
- DEPNER, K., BAUER, T.H. & LIESS, B. 1992. Thermal and pH stability of pestiviruses. Revue science and technique Office International des Epizooties, 11, 885–893.
- DEPNER, K., RODRIGUEZ, A., PHOLENZ, J. & LIESS, B., 1996. Persistent classical swine fever virus infection in pigs infected after weaning with a virus isolated during the 1995 epidemic in Germany: Clinical, virological, serological and pathological findings. European Journal of Veterinary Pathology, 2, 61–66.
- DEPNER, K., HINRICHS, U., BICKHARDT, K., GREISER-WILKE, I., POHLENZ, J., MOENNIG, V. & LIESS, B., 1997. Influence of breed-related factors on the course of classical swine fever virus infection. The Veterinary Record, 140, 506–507.
- DE SCHWEINETZ, E.A. & DORSET M., 1903. New factors concerning the etiology of hog cholera. USDA 20th Annual Report, pp.157–162.
- DE SMIT, A.J., BOUMA, A., TERPSTRA, C. & VAN OIRSCHOT, J.T., 1999. Transmission of classical swine fever virus by artificial insemination. Veterinary Microbiology, in press.
- DUNNE, H.W., 1975. Hog cholera. In: dunne, h.w. & leman, a.d. (eds), Diseases of Swine, 4th ed. Iowa: Iowa State University Press, Ames, pp. 189–255.
- EDWARDS, S. & SANDS, J.J., 1990. Antigenic comparison of hog cholera virus isolates from Europe, America and Asia using monoclonal antibodies. Deutsche Tierärtzliche Wochenschrift, 97, 79–81.
- EDWARDS, S., MOENNIG, V. & WENSVOORT, G., 1991. The development of an international reference panel of monoclonal antibodies for the differentiation of hog cholera virus from other pestiviruses. Veterinary Microbiology, 29, 101–108.
- FERNELIUS, A.L., AMTOWER, W.C., LAMBERT, G., MCCLURKIN, A.W. & MATTHEWS, P.J., 1973. Bovine viral diarrhea virus in swine: Characteristics of virus recovered from naturally and experimentally infected swine. Canadian Journal of Comparative Medicine, 37, 13–20.
- FRITZEMEIER, J., TEUFFERT, J., GREISER-WILKE, I., STRAUBACH, C., SCHLÜTER, H. & MOENNIG, V., 2000. Epidemiology of classical swine fever in Germany in the 1900s. Veterinary Microbiology, 77, 29–41.
- FUCHS, F., 1968. Schweinepest. In: röhrer, h., (ed.). Handbuch der Virusinfektionen bei Tieren, Band III. Jena: Gustav Fischer Verlag, pp. 16–250.
- GOMEZ-VILLAMANDOS, J.C., RUIZ-VILLAMOR, E., SALGUERO, F.J., BAUTISTA, M.J., CARRASCO, L., SANCHEZ, C., QUEZADA, M. & SIERRA, M.A., 1998. Immunohistochemical and ultrastructural evidence of hog cholera virus infection of megakaryocytes in bone marrow and spleen. Journal of Comparative Pathology, 119, 111–119.
- GREISER-WILKE, I., DEPNER, K., FRITZEMEIER, J., HAAS, L. & MOENNIG. V.,1998. Application of a computer program for genetic typing of classical swine fever virus isolates from Germany. Journal of Virological Methods, 75, 141–150.
- HAAS, R., AHL, R., BÖHM, R. & STRAUCH, D., 1995. Inactivation of viruses in liquid manure. Revue science and technique Office Internationales des Epizooties ,14, 435–445.
- HOFMANN, M.A., BRECHTBÜHL, K. & STÄUBER, N., 1994. Rapid characterization of new pestivirus strains by direct sequencing of PCR-amplified cDNA from the 5’ noncoding region. Archives of Virology, 139, 217–229.
- HUGHES, R.W. & GUSTAFSON, D.P., 1960. Some factors that may influence hog cholera transmission. American Journal of Veterinary Research, 21, 464–471.
- HULST, M.M., WESTRA, D.F., WENSVOORT, G. & MOORMANN, R.J.M., 1993. Glycoprotein E1 of hog cholera virus expressed in insect cells protects swine from hog cholera. 67, 5435–5442.
- HULST, M.M., HIMES, G., NEWBIGIN, E. & MOORMANN, R.J.M., 1994. Glycoprotein E2 of classical swine fever virus: Expression in insect cells and identification as a ribonuclease. Virology, 200, 558–565.
- HULST, M.M., PANOTO, F.E., HOEKMAN, A., VAN GENNIP, H.G.P. & MOORMANN, R.J.M., 1998. Inactivation of the RNase activity of glycoprotein Erns of classical swine fever virus results in a cytopathogenic virus. Journal of Virology, 72, 151–157.
- ISHIKAWA, K., NAGAI, H., KATAYAMA, K., TSUTSUI, M., TANABAYASHI, K., TAKEUCHI, K., HISHIYAMA, M., SAITOH, A., TAKAGI, M., GOTOH, K., MARAMATSU, M. & YAMADA, A., 1995. Comparison of the entire nucleotide and deduced amino acid sequences of the attenuated hog cholera vaccine strain GPE- and the wild-type parental strain ALD. Archives of Virology, 140, 1385–1391.
- KATZ, J.B., RIDPATH, J.F. & BOLIN, S.R., 1993. Presumptive diagnostic differentiation of hog cholera virus from bovine viral diarrhea and border disease viruses by using a cDNA nested-amplification approach. Journal of Clinical Microbiology, 31, 565–568.
- KIMMAN, T.G., BIANCHI, A.T.J., WENSVOORT, G., DE BRUIN, T.G.M. & MELIEFSTE, C. 1993. Cellular immune response to hog cholera virus (HCV); T cells of immune pigs proliferate in vitro upon stimulation with live HCV, but the E1 envelope glycoprotein is not a major T-cell antigen. Journal of Virology, 67, 2922–2927.
- KORN, G. & MATTHAEUS, W., 1977. Die Schweinepestkrankheit als virus-induzierte Störung des Enzymsystems: Zur Pathogenität von Pankreassuspensionen und Chymotrypsin(ogen). Zentralblatt für Bakteriologie und Hygiene, Abteilung I Original, Reihe A, 238, 20–34.
- KOSMIDOU, A., BUTTNER, R. & MEYERS, G. 1998. Isolation and characterization of cytopathogenic classical swine fever virus (CSFV). Archives of Virology, 143, 1295–1309.
- KUBIN, G., 1967. In vitro Merkmale des Schweinepestvirus. Zentralblatt für Veterinärmedizin, B, 14, 543–552.
- KUMAGAI, T., SHIMIZU, T. & MATUMOTO, M., 1958. Detection of hog cholera virus by its effect on Newcastle disease virus in swine tissue culture. Science, 138, 336.
- LAEVENS, H., KOENEN, F., DELUYKER, H., BERKVENS, D. & DE KRUIF, A., 1998. An experimental infection with classical swine fever virus in weaner pigs. I. Transmission of the virus, course of the disease, and antibody response. Veterinary Quarterly, 20, 41–45.
- LOWINGS, P., IBATA, G., NEEDHAM, J. & PATON, D., 1996. Classical swine fever virus diversity and evolution. Journal of General Virology, 77, 1311–1321.
- MAHNEL, H. & MAYR, A., 1974. Schweinepest. Infektionskrankheiten und ihre Erreger. Band 16 VEB. Jena: Gustav Fischer Verlag.
- MATTHAEUS, W. & KORN, G., 1966. Serumproteinveränderungen und klinische Symptome bei verschiedenen Verlaufsformen der Schweinepest. Zentralblatt für Veterinärmedizin, B, 13, 558–569.
- MCGOLDRICK, A., LOWINGS, J.P., IBATA, G., SANDS, J.J., BELAK, S. & PATON, D.J., 1998. A novel approach to the detection of classical swine fever virus by RT-PCR with a fluorogenic probe (TaqMan). Journal of Virological Methods, 72, 125–135.
- MENGELING, W.L., 1970. Endogenous neutralization of virus during fatal hog cholera illness. American Journal of Veterinary Research, 31, 91–95.
- MENGELING, W.L. & CHEVILLE, N.F., 1968. Host response to persistent infection with hog cholera virus. Proceedings of the Annual Meeting of the U.S. Livestock Sanitary Association, 72, 283–295.
- MENGELING, W.L. & PACKER, R.A., 1969. Pathogenesis of chronic hog cholera: Host response. American Journal of Veterinary Research, 30, 409–417.
- MEYER, H., LIESS, B., FREY, H.-R., HERMANNS, W. & TRAUTWEIN, G., 1981. Experimental transplacental transmission of hog cholera virus in pigs. Zentralblatt für Veterinärmedizin, B, 28, 659–668.
- MEYERS, G. & THIEL, H.-J., 1995. Cytopathogenicity of classical swine fever virus caused by defective interfering particles. Journal of Virology, 69, 3683–3689.
- MEYERS, G., RUMENAPF, T. & THIEL, H.-J., 1989. Molecular cloning and nucleotide sequence of the genome of hog cholera virus. Virology, 171, 555–567.
- MEYERS, G., THIEL, H-J. & RUMENAPF, T., 1996. Classical swine fever virus: Recovery of infectious viruses from cDNA constructs and generation of recombinant cytopathogenic defective interfering particles. Journal of Virology, 70, 1588–1595.
- MITTELHOLZER, C., MOSER, C., TRATSCHIN, J.D. & HOFMANN, M.A. 1997. Generation of cytopathogenic subgenomic RNA of classical swine fever virus in persistently infected porcine cell lines. Virus Research, 51, 125–137.
- MOENNIG, V., SCHAGEMANN, G., DAHLE, J., GREISER-WILKE, I. & LEDER, L., 1990. A new approach for the diagnosis of hog cholera. Deutsche Tierärtzliche Wochenschrift, 97, 91–93.
- MOORMANN, R.J.M., WARMERDAM, P.A.M., VAN DER MEER, B., SCHAAPER, W.W.M., WENSVOORT, G. & HULST, M.M., 1990. Molecular cloning and nucleotide sequence of hog cholera virus strain Brescia and mapping of the genomic region encoding envelope protein E1. Virology, 177, 184–198.
- MOORMANN, R.J.M., VAN GENNIP, H.G.P., MIEDEMA, G.K.W., HULST, M.J. & VAN RIJN, P.A., 1996. Infectious RNA transcribed from an engineered full-length cDNA template of the genome of a pestivirus. Journal of Virology, 70, 763–770.
- MOORMANN, R.J.M., VAN RIJN, P.A., DE SMIT, A.J., WENSVOORT, G. & TERPSTRA, G., 1996. Recent developments in pig vaccinology. In: Proceedings of the 14th International Pig Veterinary Society Congress, Bologna, Italy, 7–10 July 1996. pp. 25–29.
- MOSER, C., RUGGLI, N., DURI TRATSCHIN, J. & HOFMANN, M.A., 1996. Detection of antibodies against classical swine fever virus in swine sera by indirect ELISA using recombinant envelope glycoprotein E2. Veterinary Microbiology, 51, 41–53.
- MULLER, A., DEPNER, K.R. & LIESS, B., 1996. Evaluation of a gp 55 (E2) recombinant-based ELISA for the detection of antibodies induced by classical swine fever virus. Deutsche Tierärztliche Wochenschrift, 103, 451–453.
- OFFICE INTERNATIONAL DES EPIZOOTIES, 1996. Manual of Standards for Diagnostic Tests and Vaccines, pp. 181–191.
- PATON, D.J., 1995. Pestivirus diversity. Journal of Comparative Pathology, 112, 215–236.
- PAULY, T., ELBERS, K., KÖNIG, M., LENGSFELD, T., SAALMÜLLER, A. & THIEL, H-J., 1995. Classical swine fever virus-specific cytotoxic T lymphocytes and identification of a T cell epitope. Journal of General Virology, 76, 3039–3049.
- PAULY, T., KÖNIG, M., THIEL, H-J. & SAALMULLER, A., 1998. Infection with classical swine fever virus: Effects on phenotype and immune responsiveness of porcine T lymphocytes. Journal of General Virology, 79, 31–40.
- REMOND, M., PLATEAU, E. & CRUCIERE, C., 1981. In vitro study of the cellular response of pigs vaccinated against classical swine fever. Zentralblatt für Veterinärmedizin, B, 28, 743–748.
- RESSANG, A.A., 1973. Studies on the pathogenesis of hog cholera. I, II. Zentralblatt für Veterinärmedizin, B, 20, 256–288.
- RESSANG, A.A., VAN BEKKUM, J.G. & BOOL, P.H., 1972. Virus excretion in vaccinated pigs subject to contact infection with virulent hog cholera strains. Zentralblatt für Veterinärmedizin, B, 19, 739–752.
- SCHNEIDER, R., UNGER, G., STARK, R., SCHNEIDER-SCHERZER, E. & THIEL, H-J., 1993. Identification of a structural glycoprotein of an RNA virus as a ribonuclease. Science, 261, 1169–1171.
- SHIMIZU, M., YAMADA, S. & NISHIMORI, T., 1995. Cytocidal infection of hog cholera virus in porcine bone marrow stroma cell cultures. Veterinary Microbiology, 47, 395–400.
- SHANNON, A.D., MORRISSY, C., MACKINTOSH, S.G. & WESTBURY, H.A. 1993. Detection of hog cholera virus antigens in experimentally-infected pigs using an antigen-capture ELISA. Veterinary Microbiology, 34, 233–248.
- STADEJEK, T., WARG, J. & RIDPATH, J.F., 1996. Comparative sequence analysis of the 5’noncoding region of classical swine fever virus strains from Europe, Asia, and America. Archives of Virology, 141, 771–777.
- STADEJEK, T., VILCEK, S., LOWINGS, J.P., BALLAGI-PORDANY, A., PATON, D.J. & BELAK, S., 1997. Genetic heterogeneity of classical swine fever virus in Central Europe. Virus Research, 52, 195–204.
- SUMMERFIELD, A., HOFMANN, M.A. & MCCULLOUGH, K.C., 1998. Low density blood granulocytic cells induces during classical swine fever targets for virus infection. Veterinary Immunology and Immunopathology, 63, 289–301.
- SUMMERFIELD, A., KNOTIG, S.M. & MCCULLOUGH, K.C., 1998. Lymphocyte apoptosis during classical swine fever: Implication of activation-induced cell death. Journal of Virology, 72, 1853–1861.
- SUSA, M., KÖNIG, M., SAALMÜLLER, A., REDDEHASE, M.J. & THIEL, H.-J., 1992. Pathogenesis of classical swine fever; B-lymphocyte deficiency caused by hog cholera virus. Journal of Virology, 66, 1171–1175.
- TERPSTRA, C., 1978. Detection of C-strain virus in pigs following vaccination against swine fever. Tijdschrift voor Diergeneeskunde, 103, 678–684.
- TERPSTRA, C., 1987. Epizootiology of swine fever. Veterinary Quarterly, 9, supplement, 50–60.
- TERPSTRA, C., 1994. Hog Cholera. In: coetzer, j.a.w., thomson, g.r. & tustin, r.c., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press Southern Africa.
- TERPSTRA, C. & WENSVOORT, G., 1997. A congenital persistent infection of bovine virus diarrhoea virus in pigs: Clinical, virological and immunological observations. Veterinary Quarterly, 19, 97–101.
- TESMER, S., URBANECK, D., KADEN, V., WITTMANN, D. & HAHNEFELD, H., 1973. Zur Wirkung von Schweinepest-Lebendvirusvakzine aus dem Impfvirusstam ‘C’ bei tragenden Sauen und deren Nachzucht. Monatshefte für Veterinärmedizin, I, 28, 251.
- TORREY, J.P. & PRATHER, J.K., 1963. Heat inactivation of hog cholera virus. I. Studies with defibrinated blood and serum. Proceedings of the Annual Meeting of U.S. Livestock Sanitary Association, 67, 414–418.
- TRAUTWEIN, G., 1988. Pathology and pathogenesis of the disease. In: liess, b. (ed.). Classical Swine Fever and Related Viral Infections. Boston: Nijhoff Publishing.
- VAN DER MOLEN, E.J. & VAN OIRSCHOT, J.T., 1981. Congenital persistent swine fever (hog cholera) I. Pathomorphological lesions in lymphoid tissues, kidney and adrenal. Zentralblatt für Veterinärmedizin, B, 28, 89–101.
- VANNIER, P. & LEFORBAN, Y., 1992. Bovine viral diarrhea and border disease. In: leman, a.d. et al., (eds). Diseases of Swine, 7th edn. Ames, Iowa: Iowa State University Press. pp. 242–246.
- VAN OIRSCHOT, J.T., 1979. Experimental production of congenital persistent swine fever infections. I. Clinical, pathological and virological observations. Veterinary Microbiology, 4, 117–132.
- VAN OIRSCHOT, J.T., 1979. Experimental production of congenital persistent swine fever infections. II. Effect on functions of the immune system. Veterinary Microbiology, 4, 133–147.
- VAN OIRSCHOT, J.T., 1983. Effect of infections with swine fever virus on immune functions. III. Antibody response to lipopolysaccharide and sheep red blood cells. Veterinary Microbiology, 8, 96–103.
- VAN OIRSCHOT, J.T. 1988. Description of the virus infection. In: liess, b., (ed.). Classical Swine Fever and Related Viral Infections. Boston: Martinus Nijhoff Publishing. pp. 1–25.
- VAN OIRSCHOT, J.T. & TERPSTRA, C., 1977. A congenital persistent swine fever infection. I. Clinical and virological observations. Veterinary Microbiology, 2, 121–132.
- VAN OIRSCHOT, J.T., DE JONG, D. & HUFFELS, A.D.H.N.J., 1981. Effect of infections with swine fever virus on immune functions. I. Response of lymphocytes from blood and lymphoid organs from infected and normal pigs to anti-immunoglobulin serum and protein A. Veterinary Microbiology, 6, 41–57.
- VAN OIRSCHOT, J.T., DE JONG, D. & HUFFELS, A.D.N.H.J., 1983. Effect of infections with swine fever virus on immune functions. II. Lymphocyte response to mitogens and enumeration of lymphocyte subpopulations. Veterinary Microbiology, 8, 81–95.
- VAN RIJN, P.A., MIEDEMA, G.K.W., WENSVOORT, G., VAN GENNIP, H.G.P. & MOORMANN, R.J.M., 1994. Antigenic structure of envelope glycoprotein E1 of hog cholera virus. Journal of Virology, 68, 3934–3942.
- VAN RIJN, P.A., VAN GENNIP, H.G.P., LEENDERTSE, C.H., BRUSCHKE, C.J.M., PATON, D.J., MOORMANN, R.J.M. & VAN OIRSCHOT, J.T., 1997. Subdivision of the pestivirus genes based on envelope glycoprotein E2. Virology, 237, 337–348.
- VILCEK, S. & BELAK, S., 1996. Genetic identification of pestivirus strain Frijters as a border disease virus from pigs. Journal of Virological Methods, 60, 103–108.
- VILCEK, S. & PATON, D.J., 1998. Application of genetic methods to study the relationship between classical swine fever outbreaks. Research in Veterinary Science, 65, 89–90.
- VILCEK, S., HERRING, A.J., HERRING, J.A., NETTLETON, P.F., LOWINGS, J.P. & PATON, D.J., 1994. Pestiviruses isolated from pigs, cattle and sheep can be allocated into at least three genogroups using polymerase chain reaction and restriction endonuclease analysis. Archives of Virology, 136, 309–323.
- VILCEK, S., STADEJEK, T., BALLAGI-PORDANY, A., LOWINGS, J.P., PATON, D.J. & BELAK, S., 1996. Genetic variability of classical swine fever virus. Virus Research, 43, 137–147.
- VILCEK, S., STADEJEK, T., TAKACSOVA, I., STROJNY, L. & MOJZIS, M., 1997. Genetic analysis of classical swine fever virus isolates from a small geographic area. Deutsche Tierärtzliche Wochenschrift, 104, 9–12.
- WEISS, E., TEREDSAL, A., HOFFMAN, R. & HOFMANN-FEZER, G., 1973. Distribution and ultrastructure of platelets in acute hog cholera. Thrombosis et Diathesis Haemorrhagica, 30, 371–380.
- WENSVOORT, G., TERPSTRA, C., BOONSTRA, J., BLOEMRAAD, M. & VAN ZAANE, D., 1986. Production of monoclonal antibodies against swine fever virus and their use in laboratory diagnosis. Veterinary Microbiology, 12, 101–108.
- WENSVOORT, G., BLOEMRAAD, M. & TERPSTRA, C., 1988. An enzyme immunoassay, employing monoclonal antibodies, and detecting specifically antibodies against classical swine fever virus. Veterinary Microbiology, 17, 129–140.
- WENSVOORT, G., TERPSTRA, C., DE KLUIJVER, E.P., KRAGTEN, C. & WARNAAR, J.C., 1989. Antigenic differentiation of pestivirus strains with monoclonal antibodies against hog cholera virus. Veterinary Microbiology, 21, 9–20.
- WENSVOORT, G., TERPSTRA, C. & DE KLUIJVER, E.P., 1989. Characterization of porcine and some ruminant pestivirus by cross-neutralization. Veterinary Microbiology, 20, 291–306.
- WIRZ, B., TRATSCHIN, J.-D., MÜLLER, H.K. & MITCHELL, D.B., 1993. Detection of hog cholera virus and differentiation from other pestiviruses by polymerase chain reaction. Journal of Clinical Microbiology, 31, 1148– 1154.