- Infectious Diseases of Livestock
- Part 2
- Swine vesicular disease
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Swine vesicular disease
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Introduction
Swine vesicular disease (SVD) is a vesicular condition of pigs resulting from infection with a specific enterovirus. It is classified as a List A disease by the Office International des Épizooties (OIE) not because of the severity or contagiousness of the disease itself, but because the lesions produced by SVD virus are indistinguishable from those produced by another agent on List A, foot-and-mouth disease (FMD) virus. Swine vesicular disease is a disease that has emerged relatively recently. The causative virus was first identified in Italy in 1966 as an enterovirus responsible for a vesicular condition in pigs that was not FMD.26 The same agent was detected in Hong Kong in 1971, where it has remained endemic ever since. The disease was given the name swine vesicular disease following its appearance in the UK in 1972. During the 1970s and early 1980s outbreaks occurred in several European countries, particularly Italy and the UK. By 1985 the disease had been eradicated from Western Europe with the exception of Italy. Since then it has remained endemic in Italy, despite an eradication campaign financed by the European Union. In the early 1990s, outbreaks occurred in a number of European countries due to the long-distance movement of pigs between member states of the European Union. Worldwide, the disease is also still present in Taiwan Province of China, in Hong Kong, and possibly in other countries of the Far East.
Aetiology
Swine vesicular disease virus (SVDV)is an enterovirus within the family Picornaviridae. It is antigenically related to the human coxsackie virus B5 (CB5).15 As a typical enterovirus, it is a small, non-enveloped RNA virus with an icosahedral capsid approximately 30 nm in diameter. The genome consists of a single strand of positive sense RNA, 7,4 KB in length, coding for the four capsid proteins and a number of non-structural proteins. Swine vesicular disease virus is capable of surviving a wide range of temperatures and pH: for example a pH greater than 12 or less than 2 is required to inactivate it, the time taken to achieve inactivation at a given pH is dependent on the temperature.14
This is of particular relevance to the epidemiology and control of the disease. There is only one serotype of SVDV, but antigenic and genomic differences can be recognized between isolates collected at different times and from different areas.2
Genetic analysis (Figure 122.1) shows a gradual evolution of the virus over time from the first isolation in 1966.2
Epidemiology
As the name suggests, SVD is principally a disease of pigs. Cattle do not support replication of SVDV and, although it can replicate in the pharynx of sheep exposed to infected pigs, sheep are not important in the epidemiology of the disease. 5 Infection of pigs can occur through damaged skin, and by ingestion or inhalation. Pigs that are moved or mixed often fight with each other and in this way receive small cuts and grazes, which can serve as portals of entry for the virus. The dose of virus required to initiate infection across damaged skin is as little as 101,5 PFU, which is 1 000-fold less than that needed to infect pigs orally.22 Taken together, these factors explain why recent outbreaks in Europe have been characterized by clinical disease in pigs which have recently been moved and, presumably, exposed to infection either during transport or at their destination. Airborne spread of SVDV does not occur other than by short-distance aerosol transmission between pigs in contact with each other.28 Infection can occur through ingestion of contaminated food. Whether virus enters directly across the mucosa or through damaged epithelial surfaces is not known. The virus in muscle is not inactivated by the fall in pH that occurs due to rigor mortis. Frozen meat from animals slaughtered during the viraemic phase can therefore remain as an almost indefinite source of infection and the virus can survive even in some processed meat for several months.23
The severity of the disease depends on both the strain of virus10 and the conditions under which the animals are kept. Recent European isolates of SVDV are generally less virulent than European isolates from the 1970s,19 and completely avirulent strains have been isolated in Japan.16
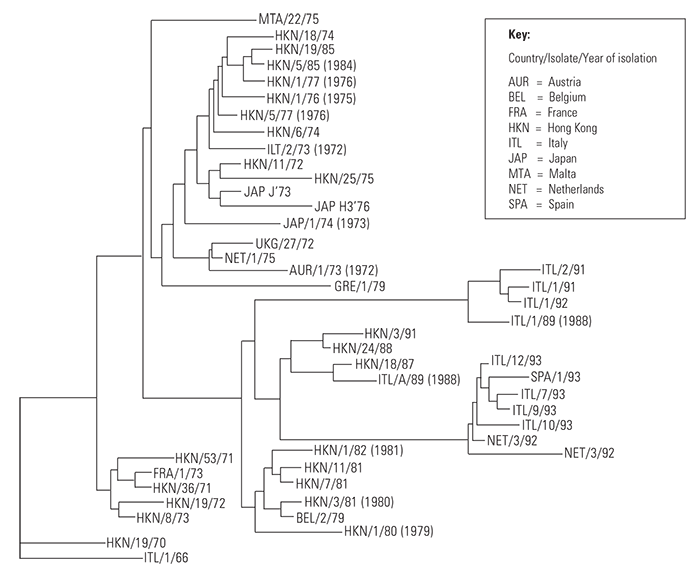
Figure 122.1 Phylogenetic analysis of the nucleotide sequence of part of the VP1 gene of SVD viruses isolated between 1966 and 1993.15 Several distinct genetic groups are seen and there is a progressive evolution of the virus with time. After 1966, each group contains representative viruses from Asia suggesting repeated introductions into Europe from an origin in the Far East
The most frequent means of transmission of SVDV between herds is the movement of infected animals. The frequently mild or subclinical nature of the disease means that apparently healthy pigs can readily be moved from an undetected source of infection. Disease only becomes apparent due to the appearance of clinical signs in animals subjected to the stress of movement or in susceptible pigs in contact with them. Shedding of virus from infected pigs starts before the first appearance of clinical signs, particularly in pigs infected by contact exposure.4 Swine vesicular disease virus is shed in oro-nasal discharges for approximately one week after the first appearance of clinical signs and virus is shed in faeces in large amounts for up to three weeks.4 Small amounts of virus have occasionally been recovered from the faeces of pigs several months after infection.13, 19 It appears that prolonged infection might be a rare sequel to exposure to the virus, but the epidemiological significance of this finding is currently unclear.
Herds that are fed waste food (‘swill’) can be readily infected if contaminated meat enters the food chain. The virus remains viable in meat for prolonged periods and can survive adverse conditions. In the early stages of the UK epidemic, swill feeding was an important means of recycling of virus and was eventually stopped by strict enforcement of legislation requiring heat treatment of waste food and tight controls on swill feeders to prevent contamination of treated food.
Another frequent means of transmission of SVDV is through exposure of pigs to an infected environment. This can occur by infected pigs contaminating transport vehicles, markets, or collection centres. Under appropriate conditions, the virus can remain viable for at least four months in transport vehicles.7 The very resistant nature of the virus to inactivation implies that only the most thorough regime of cleaning and disinfection will eliminate it from the environment. Recrudescence of infection readily occurs if re-stocking is allowed on previously contaminated premises that have been inadequately cleaned and disinfected.
Disease attributed to infection with SVDV has been reported in laboratory workers and animal handlers involved in experimental infections.3 Clinical signs are generally consistent with those of an influenza-like illness of varying severity, resembling those caused by the related CB5 virus. Despite exposure of farmers, stockmen and veterinarians to presumably similar levels of virus challenge in the field, there have been no reports of disease in humans during naturally occurring outbreaks of SVD.
Pathogenesis
Primary replication of virus occurs at the site of entry. This is most frequently damaged areas of skin, but may also be the mucosa of the pharynx or alimentary tract. There follows a period of viraemia with dissemination of the virus to sites of replication, particularly the epithelium of the coronary band of the feet and the snout. Virus replication occurs in stratified squamous epithelium resulting in localized coagulative necrosis and typical vesicle formation.17 High titres of virus are found in lymph nodes draining cutaneous sites. This is thought to be due to the presence of large amounts of virus in the afferent lymph. The extent of virus replication in lymphoid tissue is unknown.9 Recent studies using in situ hybridization suggest that dendritic cells may be involved in the early stages of infection.25 Virus is present in the tonsil from as early as the first day after exposure. As high titres of virus are required to initiate infection when applied directly to the tonsil itself, infection of this lymphoid organ is thought to be haematogenous.22 During the period of viraemia, virus can be isolated from most organs and tissues and microscopic changes can be detected in the skin, tonsils, gastrointestinal tract, kidneys, salivary glands, and pancreas.17 Virus replicates widely within the central nervous system. Neutralizing antibody develops as soon as three to four days after exposure to the virus and viraemia ceases with the onset of seroconversion. Infection by the oral route, particularly with low doses of virus, favours the development of subclinical disease.21
Clinical signs and pathology
Exposure of pigs to a contaminated environment can result in the development of viraemia within 24 hours and the appearance of clinical signs in less than two days.9
The incubation period for SVD is two to seven days but may be longer if the infecting dose is small. Both the morbidity and severity of clinical signs, when present, vary widely. The clinical situation in the field can range from a herd with virtually all animals showing vesicular lesions to a completely subclinical infection, detected only on the basis of serology.
Pigs kept in unsanitary conditions, particularly with wet or rough flooring, show more severe lesions than pigs in good husbandry systems with clean, dry and soft bedding areas.
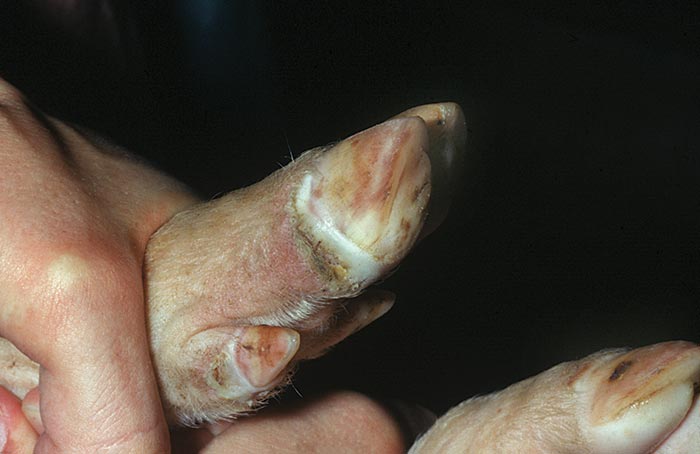
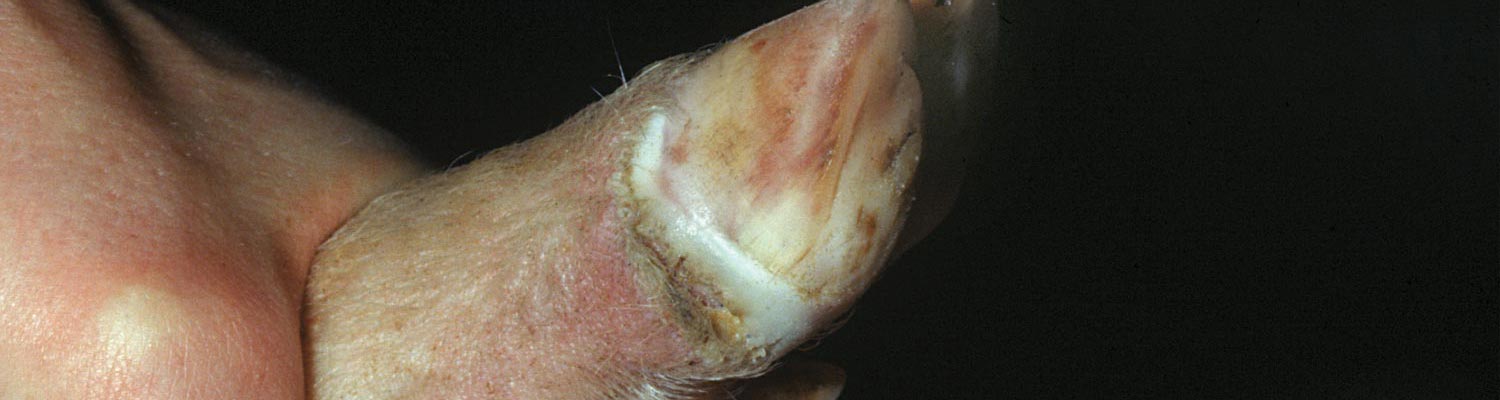
In cases of clinical disease, the end of the incubation period may be marked by a brief period of pyrexia. Pigs may be inappetant and lameness of varying degrees may be noticed. Lesions then appear at predilection sites, e.g. starting as focal whitening areas along the coronary band, particularly at the bulbs of the heel. Within 24 hours, lesions progress to fluid-filled vesicles (Figure 122.2) which rupture within a further 24 hours, releasing a virus-rich fluid and leaving raw erosions in their place. Healing of erosions is generally rapid and by the third day of clinical disease, the floor of erosions has lost its bright red colour and re-epithelialization has started at the periphery. The rate of healing depends on the degree of secondary infection. In clean conditions, lesions heal within seven to ten days, and after this time the only evidence of infection may be a dark line which progresses down the horn of the hoof at a rate of approximately 2 mm per week. Secondary infection due to wet or unsanitary conditions will delay healing and can result in complete loss of the cleat. Vesicles with subsequent erosion formation may also occur on the snout and, less often, in the mucosa of the mouth and lips or skin of the teats. Vesicles, or more often erosions, may be seen in the skin at any of the pressure points of the limbs, such as the carpus, tarsus and hocks.
When carrying out a clinical inspection for SVD, it is imperative that the pigs be caught and their feet thoroughly examined. Affected pigs may have quite extensive vesicles on their feet without being noticeably lame. Cursory inspection of such animals from a distance often fails to detect lesions.
Mortality is not a feature of the disease even in young animals. There are reports of the disease causing abortion in the field, but the virus does not cross the placenta and abortion cannot be reproduced experimentally.6
No macroscopic pathological changes are observed in internal organs. The lesions associated with viral replication in the central nervous system can only be detected microscopically and are most frequently observed in the brain stem, olfactory bulbs, cerebrum and thalamus. They are those of a non-suppurative meningitis and panencephalomyelitis, 18 which may, in some cases, result in transient nervous signs.
Diagnosis
When clinical disease is present, confirmation of the diagnosis of SVD should be based on isolation of virus from samples of epithelium collected from ruptured or unruptured vesicles. Viral antigen can be detected by ELISA11 as soon as four hours after the receipt of a sample by the diagnostic laboratory, provided sufficient material of adequate quality is submitted. Isolates of SVDV can be characterized antigenically and genomically2 for epidemiological studies. When subclinical SVD is suspected, or when suspicion is raised after clinical signs have resolved, blood samples should be collected for serology and faeces for virus isolation. Several animals from pens containing suspected cases and from adjacent pens should be sampled. Clustering of seropositive animals within pens together with evidence of seroconversion in paired samples collected from individual animals at different times are evidence of active infection with SVDV.
When screening herds as part of epidemiological surveillance, a system termed restricted randomized sampling is used. This assumes that within any pen of pigs affected with SVD, the prevalence of seropositive animals will be high but that not all pens on a farm will contain affected animals. Pigs in a high proportion of pens are therefore sampled but a low number of samples per pen are taken. Previous exposure to SVDV is most reliably detected by serology. The preferred screening test is an ELISA based on the monoclonal antibody 5B7.1 In case of doubt, sera are confirmed as positive by the virus neutralization test.12 No one test is entirely specific as approximately one in 1 000 pigs reacts positively for antibody to SVDV, despite never having been exposed to the agent. Such ‘singleton reactors’ can cause problems in serological surveys and in international trade. The problem is minimized using a combined regime of ELISA and virus neutralizing test. Where possible, the isotype of antibody should also be determined,1 as reactivity in singleton reactor sera is associated exclusively with IgM.8
Differential diagnosis
The viral vesicular diseases of pigs are FMD, SVD, vesicular stomatitis, and vesicular exanthema. These diseases cannot be distinguished reliably on clinical grounds alone. Differentiation in the laboratory relies on antigen detection by ELISA, differential growth in cell culture and, recently, by PCR and sequencing. Other non-viral causes of vesicular conditions, such as photosensitivity, have occasionally been reported in pigs.
Control
Swine vesicular disease is a notifiable disease. All suspect outbreaks should be reported to the relevant national authority, which, in turn, should report any confirmed cases to the OIE. For countries free of SVD, control of the disease relies on preventing entry of the virus. Pigs should only be accepted from SVDV-free countries or regions. If this is not possible, then pigs should be serologically tested and shown to be free of antibodies to SVDV before being accepted. Imported pigs should undergo a period of quarantine. Breeding animals supplying germ plasm for artificial insemination should be shown to be free of SVDV. Strict regulations are necessary to ensure that all waste food fed to pigs is heat-treated and that high-risk food, such as airline waste, is not fed to pigs.
Once an outbreak of SVD occurs, the control measures implemented should be the same as those used in some countries for FMD, namely ‘stamping out’. All pigs on the infected holding are slaughtered with the minimum possible delay. If possible, the carcasses are destroyed in situ by burial or incineration. If not, they are removed in sealed vehicles to a designated rendering plant. Retrospective tracing is carried out of all movements of pigs, pig products, vehicles and people onto and off the premises for 28 days before the first appearance of clinical signs in an attempt at identifying the possible origin and spread of virus. A thorough, initial cleaning and disinfection of the premises is carried out and the entire process is usually repeated not longer than 14 days later. Each cycle of cleaning and disinfection involves washing with a detergent to remove organic matter and soaking surfaces with either 1 per cent sodium hydroxide or a commercial disinfectant effective against SVDV. Final cleaning of non-flammable surfaces with a flame gun is recommended. Within the European Union, two movement-control zones are established once the disease is confirmed. A ‘Protection Zone’, usually of 3-km radius around the infected holding, and a ‘Surveillance Zone’, usually of 10-km radius. No movement of pigs is permitted within the Protection Zone. Movement is permitted under licence within the Surveillance Zone, but generally only to a designated slaughterhouse within the controlled area. Due to the mild nature of the disease it is usual to carry out serological surveillance within the movement-control zones to identify undetected spread of infection. Provided that no new foci of infection are detected, movement controls are normally lifted 21 days after initial cleansing and disinfection of the infected holding or when serological surveillance is completed, whichever is the later.
In countries where the disease has become established, eradication is achieved by a test and slaughter policy. Serological testing is the only reliable means of detecting residual, subclinical pockets of infection. The disease can be difficult to eradicate. In the UK, SVD was brought under control by a combination of licensing the movement of pigs, strict enforcement of swill feeding regulations, ensuring high standards for cleaning and disinfecting of vehicles and lairages, and by imposing the ‘21-day rule’. This prohibited the movement of pigs off premises that had received pigs within the previous 21 days. It was this regulation that slowed the spread of the disease sufficiently to allow effective tracing to be carried out.27 Final eradication of the virus from the UK was achieved in 1982, ten years after its first introduction. During the latter stages of the eradication campaign, infected premises were detected by a combination of targeted and random serological surveillance followed by slaughter of positive herds.20 In Italy during the 1990s an alternative approach was followed. Herds from which virus is isolated are slaughtered out. In herds which are serologically positive, but in which virus activity cannot be identified, only the serologically positive animals are slaughtered and the herd is subsequently re-tested. The success of this policy is not yet known.
Experimental vaccines have been shown to be effective against SVDV,25 but have never been used in the field. In situations where the disease is endemic, and husbandry practices make eradication unlikely, vaccination may present the only realistic method of control.
Although serological surveillance has never been carried out, Africa is considered to be free of SVDV. The threat to southern Africa from SVD therefore lies in the importation of infected pigs or pig products from endemic areas. The risks of incursion due to the legal movements of pigs are minimal, provided the animals are serologically tested prior to importation. Illegal movement is unlikely to be a problem due to the distances involved between southern Africa and any known focus of infection. Importation of infected pig meat, especially from the Far East where the distribution of SVD is unclear, represents the greatest threat. This risk can be controlled only by prohibiting the importation of pig products from suspect countries and by enforcing strict controls on feeding of wastefood to pigs.
References
- brocchi, e., berlinzani, e., gamba, d. & simone de, f., 1995. Development of two novel monoclonal antibody-based ELISAs for the detection of antibodies and the identification of swine isotypes against swine vesicular disease virus. Journal of Immunological Methods, 52, 155–167.
- brocchi, e., zhang, g., knowles, n.j., wilsden, g., mccauley, j.w., marquardt, o., ohlinger, v.f. & de simone, f., 1997. Molecular epidemiology of recent outbreaks of swine vesicular disease: Two genetically and antigenically distinct variants in Europe, 1987–94. Epidemiology and Infection, 118, 51–61.
- brown, f., goodridge, d. & burrows, r., 1976. Infection of man by swine vesicular disease virus. Journal of Comparative Pathology, 86, 409–414.
- burrows, r., mann, j.a. & goodridge, d., 1974. Swine vesicular disease: virological studies of experimental infections produced by the England/ 72 virus. Journal of Hygiene, 72, 135–143.
- burrows, r., mann, j.a., goodridge, d. & chapman, w.g., 1974. Swine vesicular disease: attempts to transmit infection to cattle and sheep. Journal of Hygiene, 73, 101–107.
- burrows, r., mann, j.a., goodridge, r., wrathall, a.e. & done, j.t., 1977. Swine vesicular disease: studies in pregnant sows. Veterinary Medicine B, 24, 177–182.
- dawe, p., 1974. Viability of swine vesicular disease in carcases and faeces. The Veterinary Record, 94, 430.
- de clercq, k., 1998. Reduction of singleton reactors against swine vesicular disease virus by a combination of virus neutralisation test, monoclonal antibody-based competitive ELISA and isotype specific ELISA. Journal of Virological Methods, 70, 7–18.
- dekker, a., moonen, p., boer-luijtze, e.d. & terpstra, c., 1995. Pathogenesis of swine vesicular disease after exposure of pigs to an infected environment. Veterinary Microbiology, 45, 243–250.
- donaldson, a.i.d., ferris, n.p., knowles, n.j. & barnett, i.t.r., 1983. Comparative studies of United Kingdom isolates of swine vesicular disease virus. Research in Veterinary Science, 35, 295–300.
- ferris, n.p. & dawson, m., 1988. Routine Application of Enzyme-Linked Immunosorbent Assay in Comparison with Complement Fixation for the Diagnosis of Foot-and-Mouth and Swine Vesicular Disease. Veterinary Microbiology, 16, 201–209.
- golding, s.m., hedger, r.s., talbot, p. & watson, j., 1976. Radial immuno-diffusion and serum-neutralization techniques for the assay of antibodies to SVD. Research in Veterinary Science, 20, 142–147.
- gourreau, j.m., berthaud, n., mishra, u., jacob, b. & vallet, c., 1975. Persistance of the Vesicular Disease Virus in Hogs. Recueil de Medecine Veterinaire, 151, 283–287.
- herniman, k.a.j., medhurst, p.m., wilson, j.n. & sellers, r.f., 1973. The action of heat, chemicals and disinfectants on swine vesicular disease virus. The Veterinary Record, 93, 620–624.
- knowles, n., zhang, g. & mccauley, j.w., 1994. Molecular epidemiology of swine vesicular disease. International Symposium on Porcine Picornavirus Infections, Greifswald, Germany.
- knowles, n.j. & mccauley, j.w., 1997. Coxsackievirus B5 and the relationship to swine vesicular disease virus, p. 153–167. In: tracy, s., chapman, n.m. & mahy, b.w.j., (eds). Current Topics in Microbiology and Immunology: The coxsackie B viruses. Vol. 223. Berlin: Springer.
- kodama, m., saito, t., ogawa, t., tokuda, g., sasahara, j. & kumagai, t., 1980. Swine vesicular disease viruses isolated from healthy pigs in non-epizootic period II. Vesicular formation and virus multiplication in experimentally inoculated pigs. National Institute of Animal Health Quarterly, 20, 123–130.
- lai, s.s., mckercher, p.d., moore, d.m. & gillespie, j.h., 1979. Pathogenesis of SVD in pigs. American Journal of Veterinary Research, 40, 463–468.
- lenghaus, c. & mann, j.a., 1976. General pathology of experimental swine vesicular disease. Veterinary Pathology, 13, 186–196.
- lin, f., mackay, d.k.j. & knowles, n.j., 1997. Detection of swine vesicular disease virus RNA by reverse transcription-polymerase chain reaction. Journal of Virological Methods, 65, 111–121.
- loxham, j.g. & hedger, r.s., 1983. Swine vesicular disease: Clinical signs, diagnosis, epidemiology and control. Review of Science and Technology, Office International des Epizooties, 2, 11–24.
- mann, j.a., burrows, r. & goodridge, d., 1975. Mild and sub-clinical infections with swine vesicular disease virus. Bulletin Office International des Epizooties, 83, 117–122.
- mann, j.a. & hutchings, g.h., 1980. Swine vesicular disease: Pathways of infection. Journal of Hygiene, 74, 355–363.
- mckercher, p.d., blackwell, j.h., murphy, r., callis, j.j., panina, g.f., civardi, a., bugnetti, m., simone, f.d. & scatozza, f., 1985. Survival of SVDV in ‘Prosciutto di Parma’ (Parma ham). Canadian Institute of Food Science and Technology Journal, 18, 63–167.
- mowat, g.n., prince, m.j., spier, r.e. & staple, r.f., 1974. Preliminary studies on the Development of a Swine Vesicular Disease Vaccine. Archives of Virology, 44, 350–360.
- mulder, w.a.m., poelwijk, v.f., moormann, r.j.m., reus, b., kok, g.l., pol, j.m.a. & dekker, a., 1997. Detection of early infection of swine vesicular disease virus in porcine cells and skin sections. A comparison of immunohistochemistry and in-situ hybridization. Journal of Virological Methods, 67, 169–173.
- nardelli, l., lodetti, e., gualandi, g.l., burrows, r., goodridge, d., brown, r. & cartwright, b., 1968. A foot and mouth disease syndrome in pigs caused by an enterovirus. Nature, 219, 1275–1276.
- richards, r.a., 1976. Experiences in the eradication of swine vesicular disease. OIE: XLIV General Session of the Committee, Paris. pp. 237–244.
- sellers, r.f. & herniman, k.a.j., 1974. The airborne excretion by pigs of swine vesicular disease virus. Journal of Hygiene, 72, 61–65.