- Infectious Diseases of Livestock
- Part 2
- Porcine haemagglutinating encephalomyelitis virus infection
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Porcine haemagglutinating encephalomyelitis virus infection
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Porcine haemagglutinating encephalomyelitis virus infection
Previous Authors: M B PENSAERT
Current Authors:
A N VLASOVA - Assistant Professor, PhD, DVM, Ohio State University, 1680 Madison Ave, Wooster, Ohio, OH 44691, USA
S N LANGEL - Graduate Research Assistant, MS, PhD Candidate, Ohio State University, 1680 Madison Avenue, Wooster, Ohio, OH 44691, USA
L J SAIF - Professor, MS, PhD, Food Animal Health Research Program, CFAES and CVM, OARDC, Ohio State University, 1680 Madison Ave, Wooster, Ohio, OH 44691, USA
A BOWMAN- Assistant Professor, MS, DVM, PhD, Diplomate ACVPM, 1920 Coffey Road, A110B Sisson Hall, Columbus, Ohio, OH 43210, USA
Introduction
Due to its ability to haemagglutinate erythrocytes of a variety of animals and its neurotropic characteristics in baby pigs, the virus was named porcine haemagglutinating encephalomyelitis virus (PHEV). Infection with PHEV is usually inapparent in adult pigs, but in neonatal piglets devoid of maternal antibodies, it may cause either encephalomyelitis or a condition characterized by vomiting and wasting.
The disease caused by PHEV was first reported in Canada in 1958,26 and the pathogen was first isolated in Canada in 1962 from the brains of suckling piglets with encephalomyelitis.13 Subsequently, the same virus was isolated in England and continental Europe from pigs affected with a disease called ‘vomiting and wasting disease’.7 Since then, the infection has been reported in the United States, Japan, Argentina, Belgium, South Korea, China and other pig-raising countries.12, 15, 22, 24, 25, 27
Currently the virus has a worldwide prevalence.17 Because the infection most often occurs in pigs older than three to four weeks and then remains subclinical, it is seldom of economic importance. However, there was a recent report of an uncommon respiratory (influenza-like) presentation of PHEV in exhibition swine in the USA.19
Aetiology
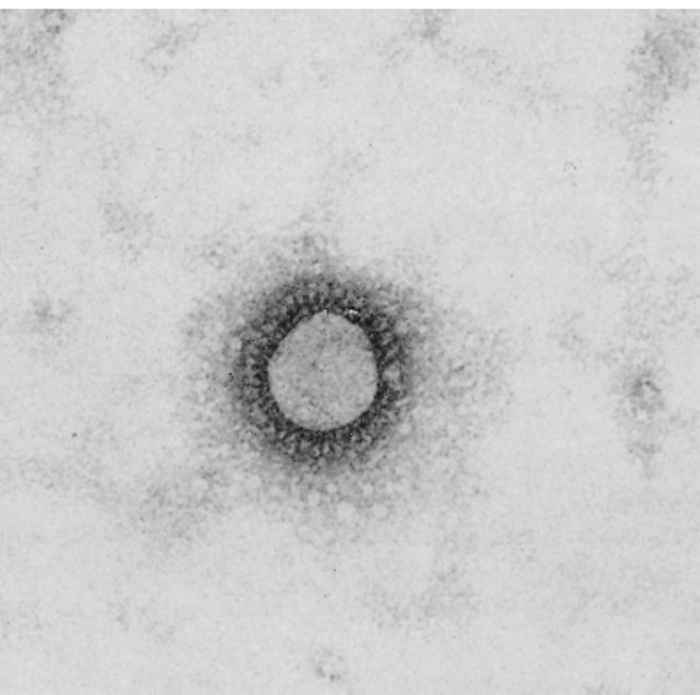
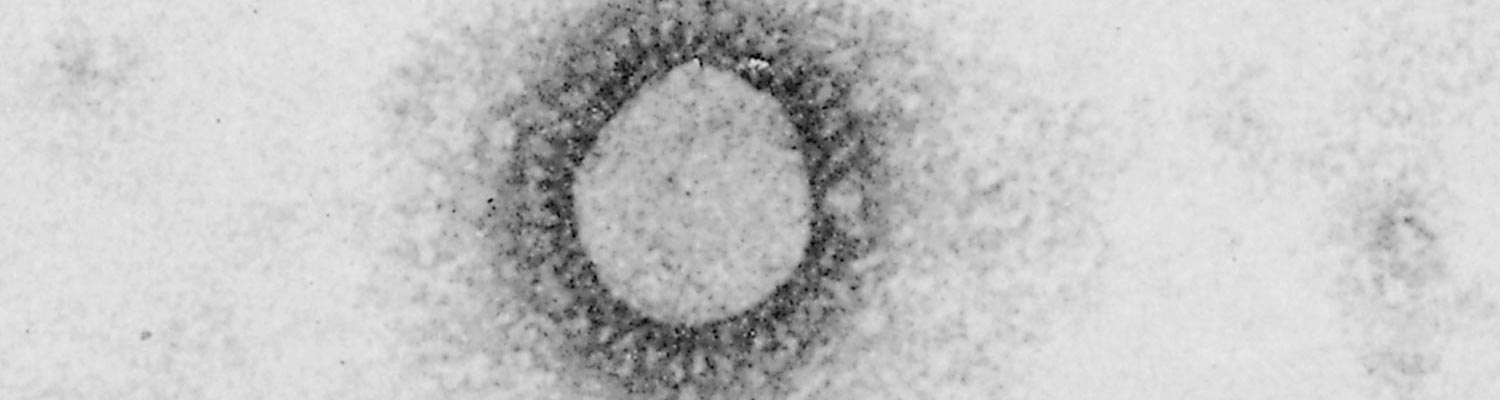
Porcine haemagglutinating encephalomyelitis virus is a clade A betacoronavirus.2, 23 Virus particles are spherical, with a diameter of 120 nm. They are covered by a double-layered fringe of club-shaped projections about 20 nm long (Figure 1).
The virus contains five structural proteins, only one of which—the nucleocapsid protein—is not glycosylated. The glycosylated proteins include the Membrane, the Haemagglutinin and the surface Spike. There is only one serotype, but PHEV is antigenically related to several other clade A betacoronaviruses, including human coronavirus OC43, bovine coronavirus, turkey coronavirus and mouse hepatitis virus.2, 9, 28
Primary cultures of pig kidney or thyroid cells are the most sensitive system for primary isolation. Syncytium formation characterizes the cytopathic effect. The virus also replicates in several continuous porcine cell lines, such as PK15, IB-RS-2 and RPD.9 It agglutinates erythrocytes of chicken, mouse, rat and several other animal species. Haemadsorption of the same spectrum of erythrocytes occurs on HEV-infected cells.22
Haemagglutinating encephalomyelitis virus is sensitive to lipid solvents, including sodium deoxycholate. Infectivity is lost after heating to 56 °C for 30 minutes but is only slightly reduced after seven days at 4 °C. The virus is stable between pH 4 and 10. The natural host is swine, but HEV is adapted to experimentally grow in mice and rats.16, 31
Epidemiology
Pigs are the only known natural hosts of PHEV. The virus is endemic in most pig populations worldwide,12, 15, 22, 24, 25, 27 including those in South Africa.11 It is also prevalent in regions or countries with relatively low pig population densities. Serological surveys have shown that between 43 and 98 per cent of pigs are serologically positive in infected regions.6 Interestingly, increased prevalence (of 38.7 per cent) of PHEV infection associated with acute respiratory illness was reported in pigs at Michigan swine fairs compared with only 4.1 per cent at swine fairs in Ohio and Indiana in 2015.19
Virus spread occurs by respiratory aerosols.
The virus is usually pathogenic only for pigs under two to three weeks of age and, because of its high prevalence in pig populations, most sows have experienced infection prior to parturition. Their offspring will be protected by colostral antibodies that remain detectable in piglets for two to three months on average.10 However, in China (where PHEV was first reported in 1985) and some other regions, the virus continues to cause vomiting and wasting disease as well as encephalomyelitis and high mortality in young (<3 weeks) piglets, resulting in substantial economic losses.10, 12, 17
By the time natural infection starts to occur, which is at four to five weeks of age or later, age-related resistance to the disease develops. Passively acquired colostral immunity is thus superseded by active immunity without the occurrence of overt disease.6, 21 When disease occurs, it is self-limiting because the sows and gilts become infected and maternal immunity protects the newborn litters. This usually occurs within three weeks after the start of an outbreak. In Taiwan, the first isolation of HEV came from pigs aged 30 to 50 days, which is rather unusual.8
Pathogenesis
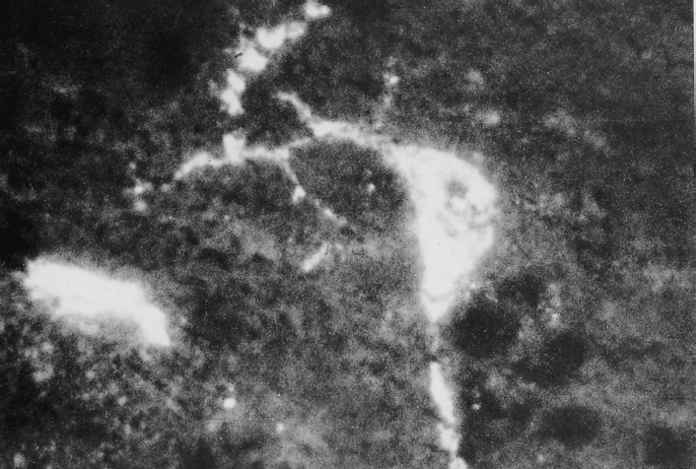
The pathogenesis of PHEV infection is studied experimentally in colostrum-deprived piglets following oronasal inoculation.4, 5 Initial infection occurs in the respiratory tract (nasal mucosa and lungs), tonsils and in a few as yet unidentified intestinal cells located between the enterocytes on the small intestinal villi. The virus spreads from the tonsillar and respiratory sites of entry along the trigeminal and vagal nerves and their respective ganglia to the corresponding sensory nuclei in the brain stem (Figure 2). From the intestinal plexi, it spreads neurally via local sensory ganglia to the spinal cord. Virus dissemination follows within the spinal cord and brain. Various combinations of central nervous motor disorders may then appear. Spread of virus from the primary sites of replication to neural tissues is efficiently halted by circulating antibodies. Recent evidence suggested that non-suppurative encephalitis was the cause of death of PHEV-infected piglets in China.17
Vomiting in the acute stage of the disease appears to be induced by viral replication in the vagal sensory ganglion, while wasting is probably the result of delayed emptying of the stomach due to virus-induced lesions in the intramural nervous plexi. Virus replication in the respiratory tract normally does not result in respiratory disease, but is responsible for the dissemination of virus in respiratory aerosols. In Japan, however, PHEV was isolated from the respiratory tract of pigs with respiratory illness.14 Additionally, a recent study evaluating acute outbreaks of respiratory disease in swine at agricultural fairs in Ohio, Michigan and Indiana, USA (2015) provided strong evidence for the role of PHEV as a respiratory pathogen. Influenza-like illness observed in swine is considered atypical for PHEV and likely reflects a unique presentation of PHEV in older naive swine populations, an atypical form of disease, or increased virulence.19
Clinical signs
There are two clinical syndromes caused by infection with PHEV: acute encephalomyelitis and so-called vomiting and wasting disease. In general, clinical signs only occur in piglets under three weeks of age that have failed to receive colostral antibodies from their dams. Disease may be present only in litters of non-immune gilts, while the litters of older (immune) sows in the same unit remain healthy.
A PHEV infection may start with sneezing and coughing but these signs usually pass unnoticed. Less common is PHEV manifestation as acute influenza-like illness in adult swine and piglets.14, 19 The vomiting and wasting disease form has an incubation period of four to seven days. The piglets continue to suckle but soon withdraw from the sow and vomit. Initially, there are no other clinical signs except for some depression and constipation. As the disease progresses, vomiting occurs after each intake of milk and results in dehydration, loss of condition and often death. Piglets that survive several days or weeks of persistent vomition become emaciated and have markedly distended abdomens. This chronic or ‘wasting’ syndrome lasts several weeks and leads to starvation and death. Mortality approaches 100 per cent in some litters and survivors remain stunted.
The enephalomyelitic form may be observed in piglets as young as three to seven days of age. Initial vomition is followed by motor disorders such as muscle tremors and hyperaesthesia. As the disease progresses, signs include walking backwards, dog-sitting and paddling movements of the legs. Piglets may develop opisthotonus and become blind. Finally, the animals become paralysed, at which time they are often dyspnoeic. Mortality is 100 per cent in pigs that are infected within a few days after birth. Older piglets may recover after a transient illness characterized by posterior paralysis.
Both clinical syndromes may occur on the same farm. Vomiting and wasting disease was most frequently observed in Europe7 when PHEV was introduced during the early 1970s, while the encephalomyelitic form was regularly observed in North America.30
An encephalomyelitic outbreak was described in Taiwan in 30- to 50-day-old pigs (disease in pigs of this age is rather unusual) and was characterized by fever, constipation, hyperaesthesia, muscular tremor, paddling movements, progressive paresis and recumbency. The morbidity rate was 4 per cent and the case fatality rate was 100 per cent, with death occurring within four to five days.8
Pathology
The only significant gross changes observed in chronically affected or wasted pigs consist of cachexia and distension of the stomach by gas. Microscopic lesions are present mainly in the central nervous system in both clinical syndromes, consisting of a non-suppurative encephalomyelitis characterized by perivascular cuffing, gliosis and neuronal degeneration. Lesions are most pronounced in the grey matter of the pons, the medulla oblongata and in the dorsal horns of the upper spinal cord.1, 17
Diagnosis and differential diagnosis
Frequent and persistent vomition in litters of piglets between four days and three weeks of age is suggestive of PHEV infection, whether or not it is accompanied by motor disorders. A definitive diagnosis requires laboratory confirmation.
Haemagglutinating encephalomyelitis virus can be isolated from suspensions of the tonsils, brain stem and lungs of piglets sacrificed in the early stages of the disease. Apart from the appearance of cytopathic effects, the presence of the virus in cell cultures can be demonstrated by haemadsorption and haemagglutination tests. Direct immunofluorescent staining of frozen sections of the brain stem is useful for the demonstration of HEV antigens during pathogenesis studies and may be helpful in diagnosis.
Antibody determination in the sera of pigs is usually achieved by virus neutralization or haemagglutination inhibition tests.29 Since PHEV-specific antibodies may be detectable in the sera of piglets within six days of the start of disease and because antibody titres in older pigs are usually the result of subclinical infection, the serological data require careful interpretation.
Pseudorabies (Aujeszky’s disease) may also cause neurological disease in young piglets, but it progresses more rapidly than HEV infection and death usually occurs within 36 to 48 hours of the first signs of illness. In pseudorabies, furthermore, older pigs may exhibit neurological and respiratory signs and sows may abort. An outbreak of the encephalomyelitic form of porcine enterovirus (Teschen-Talfan) infection may start, as does PHEV infection, with vomition commencing four to seven days after birth. However, pigs may also be affected at a later age. Also, after two to three days of vomition, piglets will exhibit various combinations of nervous signs such as tremor, hyperaesthesia, paddling limb movements and particularly posterior paralysis. The PHEV outbreak described in Taiwan8 showed many clinical similarities with Teschen-Talfan, and laboratory examination is essential for differentiation. Due to the variety of clinical manifestations associated with PHEV, RT-PCR confirmatory diagnosis should be conducted.17
Control
Gilts should ideally be exposed to the virus before the first farrowing. Their offspring will then be protected by antibodies acquired through colostrum. Seronegative sows may be present on small farms where the virus has not persisted or on farms where, for some reason, infection has not occurred before. For this reason, breeding animals should not be isolated from the rest of the population during rearing.
When an outbreak occurs, newborn piglets can be protected at birth by the injection of pooled serum collected from several sows in the slaughterhouse, as the chances are good that such pooled serum will contain specific antibodies. This procedure should be continued for litters born up to three weeks after the start of the outbreak. Thereafter, natural immunity will have developed in pregnant sows.
Recent findings on the mechanisms that PHEV utilizes to enter and traffic within neural cells may help to identify novel therapeutic targets.18
References
- ALEXANDER, T.J., 1962. Viral encephalomyelitis of swine in Ontario--experimental and natural transmission. American Journal of Veterinary Research, 23, 756-762.
- ANDREW, M.Q.K., ADAMS, M.J., CARSTENS, E.B. & LEFKOWITZ, E.J., 2011. Ninth Report of the International Committee on Taxonomy of Viruses. Virus Taxonomy, Elsevier Inc
- ANDRIES, K. & PENSAERT, M., 1980. Propagation of hemagglutinating encephalomyelitis virus in porcine cell cultures. Zentralblatt für Veterinärmedizin B, 27, 280-290.
- ANDRIES, K. & PENSAERT, M.B., 1980. Virus isolated and immunofluorescence in different organs of pigs infected with hemagglutinating encephalomyelitis virus. American Journal of Veterinary Research, 41, 215-218.
- ANDRIES, K. & PENSAERT, M.B., 1980.Immunofluorescence studies on the pathogenesis of hemagglutinating encephalomyelitis virus infection in pigs after oronasal inoculation. American Journal of Veterinary Research, 41, 1372-1378.
- ANDRIES, K., 1989. Haemagglutinating encephalomyelitis virus. In: PENSAERT, M.B., (ed). Virus Infections of Porcines. Amsterdam, Oxford, New York, Tokyo: Elsevier Scientific Publishers.
- CARTWRIGHT, S.F. & LUCAS, M., 1970.Vomiting and wasting disease in piglets. Virological and epidemiological studies. Veterinary Record, 86, 278-280.
- CHANG, G.N., CHANG, T.C., LIN, S.C., TSAI, S.S. & CHERN, R.S., 1993. Isolation and identification of hemagglutinating encephalomyelitis virus from pigs in Taiwan. Journal of Chinese Society of Veterinary Science, 19, 147–158.
- DEA, S. & TIJSSEN, P., 1989. Detection of turkey enteric coronavirus by enzyme-linked immunosorbent assay and differentiation from other coronaviruses. American Journal of Veterinary Research, 50, 226-231.
- DONG, B., LU, H., ZHAO, K., LIU, W., GAO, W., LAN, Y., ZHAO, J., TANG, B., SONG, D., HE, W. & GAO, F., 2014. Identification and genetic characterization of porcine hemagglutinating encephalomyelitis virus from domestic piglets in China. Archives of Virology, 159, 2329-2337.
- ESTERHUYSEN, J.J. & THOMSON, G.R., 1985. Unpublished data, Veterinary Research Institute, Onderstepoort.
- GAO, W., ZHAO, K., ZHAO, C., DU, C., REN, W., SONG, D., LU, H., CHEN, K., LI, Z., LAN, Y., XIE, S., HE, W. & GAO, F., 2011.Vomiting and wasting disease associated with hemagglutinating encephalomyelitis viruses infection in piglets in Jilin, China. Virology Journal, 8, 130.
- GREIG, A.S., MITCHELL, D., CORNER, A.H., BANNISTER, G.L., MEADS, E.B. & JULIAN, R.J., 1962. A Hemagglutinating Virus Producing Encephalomyelitis in Baby Pigs. Canadian Journal of Comparative Medicine and Veterinary Science, 26, 49-56.
- HIRAHARA, T., YASUHARA, H., KODAMA, K., NAKAI, M. & SASAKI, N., 1987. Isolation of hemagglutinating encephalomyelitis virus from respiratory tract of pigs in Japan. Nihon Juigaku Zasshi, 49, 85-93.
- HIRANO, N. & ONO, K., 1998. A serological survey of human coronavirus in pigs of the Tohoku District of Japan. Advances in Experimental Medicine and Biology, 440, 491-494.
- HIRANO, N., HAGA, S. & FUJIWARA, K., 1993. The route of transmission of hemagglutinating encephalomyelitis virus (HEV) 67N strain in 4-week-old rats. Advances in Experimental Medicine and Biology, 342, 333-338.
- LI, Z., HE, W., LAN, Y., ZHAO, K., LV, X., LU, H., DING, N., ZHANG, J., SHI, J., SHAN, C. & GAO, F., 2016. The evidence of porcine hemagglutinating encephalomyelitis virus induced nonsuppurative encephalitis as the cause of death in piglets. PeerJ, 4, e2443.
- LI, Z., ZHAO, K., LAN, Y., LV, X., HU, S., GUAN, J., LU, H., ZHANG, J., SHI, J., YANG, Y., SONG, D., GAI, F. & HE, W., 2017. Porcine Hemagglutinating Encephalomyelitis Virus Enters Neuro-2a Cells via Clathrin-Mediated Endocytosis in a Rab5-, Cholesterol-, and pH-Dependent Manner. Journal of Virology, 91.
- LORBACH, J.N., WANG, L., NOLTING, J.M., BENJAMIN, M.G., KILLIAN, M.L., ZHANG, Y. & BOWMAN, A.S., 2015. Porcine Hemagglutinating Encephalomyelitis Virus and Respiratory Disease in Exhibition Swine, Michigan, USA. Emerging Infectious Diseases, 23, 1168-1171.
- PAUL, P.S. & MENGELING, W.L., 1984. Persistence of passively acquired antibodies to hemagglutinating encephalomyelitis virus in swine. American Journal of Veterinary Research, 45, 932-934.
- PENSAERT, M., ANDRIES, K. & CALLEBAUT, P., 1980. A seroepizootiologic study of vomiting and wasting disease virus in pigs. Veterinary Quarterly, 2, 142-148.
- PENSAERT, M.B. & CALLEBAUT, P.E., 1974. Characteristics of a coronavirus causing vomition and wasting in pigs. Archiv für die gesamte Virusforschung, 44, 35-50.
- PHILLIP, J.I., CARTWRIGHT, S.F. & SCOTT, A.C., 1971. The size and morphology of T.G.E. and vomiting and wasting disease viruses of pigs. Veterinary Record, 88, 311-312.
- QUIROGA, M.A., CAPPUCCIO, J., PINEYRO, P., BASSO, W., MORE, G., KIENAST, M., SCHONFELD, S., CANCER, J.L., ARAUZ, S., PINTOS, M.E., NANNI, M.E., NANNI, M., MACHUCA, M., HIRANO, N. & PERFUMO, C.J., 2008. Hemagglutinating encephalomyelitis coronavirus infection in pigs, Argentina. Emerging Infectious Diseases, 14, 484-486.
- RHO, S., MOON, H.J., PARK, S.J., KIM, H.K., KEUM, H.O., HAN, J.Y., VAN NGUYEN, G. & PARK, B.K., 2011. Detection and genetic analysis of porcine hemagglutinating encephalomyelitis virus in South Korea. Virus Genes, 42, 90-96.
- ROE, C.K. & ALEXANDER, T.J., 1958. A Disease of Nursing Pigs Previously Unreported in Ontario. Canadian Journal of Comparative Medicine and Veterinary Science, 22, 305-307.
- SASSEVILLE, A.M., GELINAS, A.M., SAWYER, N., BOUTIN, M. &DEA, S., 2001. Biological and molecular characteristics of an HEV isolate associated with recent acute outbreaks of encephalomyelitis in Quebec pig farms. Advances in Experimental Medicine and Biology, 494, 57-62.
- SATO, K., INABA, Y. & MATUMOTO, M., 1980. Serological relation between calf diarrhea coronavirus and hemagglutinating encephalomyelitis virus. Archives of Virology, 66, 157-159.
- SATO, K., INABA, Y., MIURA, Y., TOKUHIS, S. & MATUMOTO, M., 1983. Inducement of cytopathic changes and plaque formation by porcine haemagglutinating encephalomyelitis virus. Veterinary Microbiology, 8, 521-530.
- WERDIN, R.E., SORENSEN, D.K. & STEWART, W.C., 1976. Porcine encephalomyelitis caused by hemagglutinating encephalomyelitis virus. Journal of the American Veterinary Medical Association, 168, 240-246.
- YAGAMI, K., IZUMI, Y., KAJIWARA, N., SUGIYAMA, F. & SUGIYAMA, Y.,1993. Neurotropism of mouse-adapted haemagglutinating encephalomyelitis virus. Journal of Comparative Pathology, 109, 21-27.