- Infectious Diseases of Livestock
- Part 2
- Equine encephalosis
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Equine encephalosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Equine encephalosis
Synonyms: Perde enkefalose (Afrik.)
Previous Authors: P G HOWELL, A J GUTHRIE AND J A W COETZER
Current Authors:
P COETZEE, PhD, Researcher, Onderstepoort Biological Products SOC Ltd, Private Bag X07, Onderstepoort, 0110, South Africa
P G HOWELL (deceased)
A J GUTHRIE, BVSc, PhD, Director, Equine Research Centre, Faculty of Veterinary Science, University of Pretoria, South Africa
J A W COETZER, BVSc, MMedVet(Path), Emeritus Professor, Faculty of Veterinary Science, University of Pretoria, South Africa
Introduction
Equine encephalosis (EE) is the name assigned to a mild or subclinical orbivirus infection of equids. The virus is transmitted by species of Culicoides, and as a result the epidemiology has much in common with African horse sickness (AHS). The designated name of the infection is unfortunately a misnomer since, by definition, neither lesions nor dysfunctions of the brain are characteristic features of the disease.
The first isolate of equine encephalosis virus (EEV) was recovered in March 1967 in South Africa from the blood and tissues of a 13-year-old Thoroughbred mare that was euthanased after showing a peracute nervous derangement. It was reported that two other mares on the same stud farm became ill during the following few days. One died, while the second recovered after a convalescence of 14 days. Although it was recorded that virus was also recovered from blood samples taken from horses that had exhibited no clinical signs of disease except for a fever, virus was also recovered from the organs of horses that had died in other parts of South Africa.7
Aetiology
Equine encephalosis virus is classified as an Orbivirus in the family Reoviridae (subfamily Sedoreovirinae) and shares many morphological, physiochemical and genomic characteristics with other orbiviruses (see Bluetongue, and African horse sickness.)
Field samples of blood and tissues from infected horses have provided seven valid serotypes (Table 1), which on cross-neutralization tests have shown insignificant cross-reactivity between heterologous antigens and antisera.8, 9, 14 Antisera produced in sheep have given unequivocal specificity in the routine identification and classification of all field isolates recovered to date.
In a transmission electron microscopic study undertaken on infected BHK21/C13 cells, it was found that the negatively stained virus particles closely resemble those of AHS and bluetongue (BT).15 Examination of thin sections of infected cells revealed a replication cycle in the cytoplasm in the perinuclear area, with progeny particles visible after 28 hours. Irregularly shaped, granular inclusion bodies without a membrane in which maturing virus particles embedded in the matrix were observed became evident in the perinuclear region. The aggregations of virus particles in the cytoplasmic matrix resemble large crystals in which sectioned particles have a diameter of 73 nm. It was concluded that the observations suggested that the capsid is composed of 32 morphological subunits, as described for bluetongue virus (BTV),6 and represent subviral particles, while the larger ill-defined particles in the crystalline bodies are the complete virion with an outer diffuse polypeptide layer.
Table 1 Classification, identification, source and origin of currently recognized serotypes of equine encephalosis virus
| SEROTYPE | IDENTIFICATION | SOURCE | ORIGIN (YEAR) | REFERENCE |
| 1 | Bryanston, M8/76 | Foetal liver/spleen | Colesberg, Western Cape (1976) | 5 |
| 2 | Cascara | Organ suspension | Kimberley, Northern Cape (1967) | 4 |
| 3 | Gamil, M9/71 | Blood | Naboomspruit, Limpopo Province (1971) | 5 |
| 4 | Kaalplaas, 7088 (7 –2) | Blood | Onderstepoort, Gauteng (1974) | 5 |
| 5 | Kyalami, 7084 (12 –3) | Blood | Johannesburg, Gauteng (1974) | 11 |
| 6 | Potchefstroom, Else EP8/91 | Blood | Potchefstroom, North West Province (1991) | 6 |
| 7 | E21/20 | Blood | St. Lucia –KwaZulu-Natal (2000) | 8 |
| 7 | Northrand | ? | Johannesburg, Gauteng Province (2008) | *, 1, 16, 25 |
* The EEV-7 Northrand isolate is given as the reference isolate in several publications, in the absence of a full genome sequence from isolate E21/20 - see GenBank accession numbers HQ630949-HQ630958 for the complete genome.
Equine encephalosis virus replicates in BHK21/C13 monolayers and produces a distinctive cytopathology when the monolayers are viewed under low-power light microscopy. Affected monolayers show a fine-textured cytopathic effect in contrast to the development of more focal refractile cells which rapidly become detached when such monolayers are infected with AHSV. Lines of Vero (ATCC CCL81) cells vary in their susceptibility to EEV. Plaque assay may be undertaken in susceptible lines: plaques become visible after five to six days’ incubation. Serotype 1 of EEV produces high yields with no cytopathic effect in C6/36 (Aedes albopictus) cell cultures.
The replication of EEV is unaffected by exposure to 5-bromodeoxyuridine at a concentration of 30 µg/ml, whereas actinomycin D at a concentration of 0,01 µg/ml reduces the yield of virus a hundred-fold. Exposure to a pH of 3 for one hour at 37 °C produces a considerable drop in the infectivity titre, whereas chloroform at a final concentration of 5 per cent has no effect on viral infectivity.7
The early molecular characterization of EEV and an attempt to develop genetic probes to distinguish between EEV and AHSV has been reported.32 The serotype 2 virus that was used in this study was partially purified by a method using Triton X-100 after which the dsRNA was extracted.31 Electrophoretic separation on a PAGE gel confirmed that the genome is composed of ten segments, which show some differences from the separation profiles of BTV and AHSV. Seven structural capsid proteins were separated on SDS PAGE gels representing four major and three minor polypeptides. These fractionation patterns closely resemble those of two other orbiviruses, namely BTV and AHSV. Incomplete clones of six of the dsRNA segments of EEV serotype 2 were obtained. From hybridization techniques it was established that segment 2 encodes for the serotype-specific antigen.
Recent advances in the sequence independent amplification of orbivirus genomes together with advances in next generation sequencing has led to a rapid increase in the number of full genome sequences available of orbiviruses on public sequence repositories. The EEV serotype 1 (Bryanston) genome was first published in 2009,21 and since then, the full genome sequences of the remaining six serotypes as well as several field isolates have been made available.25, 33, 36 The EEV genome, analogous to AHSV and BTV, is segmented, approximately 19 – 20 kb in size and contains open reading frames located on the individual genome segments, that encode the seven structural (VP1 to VP7) and at least three non-structural (NS1-NS3) proteins. 21 Bioinformatic analysis indicates that like other orbiviruses, another protein (NS4) can possibly be encoded by segment 9, however the presence of this protein in EEV infected cells has not been confirmed. The presence of NS4 in AHSV37 and BTV24 infected cells has previously been demonstrated.
Analysis of EEV full genome sequences confirms that segment 2 (VP2) encodes the virus serotype specific antigen, whereas segment 7 (VP7) is conserved within the EEV serogroup and encodes the viral group specific antigen21, 25 presence of an EEV group specific antigen was alluded to in much earlier complement fixation (CF) studies, which indicated that EEV field isolates share a common antigen, that does not cross-react with other orbiviruses including BT, AHS, Corriparta, Palyam (D’Aquilar and Abadina), Warrego (CH9935), Mitchell River (MRM10434) or epizootic haemorrhagic disease of deer viruses (EHDV).8 Reassortment of genome segments between EEV serotypes has been reported of field isolates from South Africa,25 with unknown clinical and epidemiological significance.
Epidemiology
Equine encephalosis and AHS exhibit similar epidemiological features in respect to natural hosts, vectors and the environmental conditions under which transmission occurs (see African horse sickness.). No vaccine has been developed for the control of EE, with the result that the prevalence of natural infection, whether determined by seroconversion or by recovery of virus, is not influenced by the prophylactic immunization of horses as is practised in the control of AHS in some parts of Africa.
The seven serotypes of EEV have been recovered from horses of all breeds and ages and the horse is the only equine species in which clinical signs have been recorded. The majority of the 59 isolates of EEV that were recovered from horses over a period of 14 years in South Africa were obtained incidentally.19 Neutralizing antibody has been detected in 20 to 30 per cent of yearlings in South Africa after their first seasonal exposure to the vector between the ages of four and eight months. A sero-epidemiological survey using a group-specific indirect enzyme-linked immunosorbent assay (ELISA) showed that in a random sample of 604 horse sera collected over 14 years, the mean seroprevalence of 74 per cent appeared to be uniformly high throughout South Africa.
In Africa, donkeys, although resistant to clinical disease, are ideal sentinels to determine the prevalence and distribution of both EEV and AHSV through the detection of both group- and serotype-specific antibody. A study over a 12-year period, using a group reactive single dilution ELISA, showed that 49,3 per cent of a sample of 4 875 donkeys of all ages were positive for antibody to EEV. In a similar survey of 193 healthy donkeys in Zimbabwe, an overall seroprevalence of 85 per cent was found throughout the country.30 In another study , seroprevalence rates were investigated over three consecutive rainy seasons (1999-2002) in sentinel donkey and horse herds in the Highveld region of Zimbabwe. Estimated seroprevalence rates in horses was 63 per cent and in donkeys 80 per cent over the sampling period.10 Thus, when compared with the prevalence of antibody to AHS, EE appears to have a wider distribution in the donkey population. The zebra (Equus burchelli) is also susceptible to the virus.2, 35 In an investigation into the seroprevalence of serotype-specific antibody in the sera of zebra of all ages, neutralizing antibody to all seven serotypes were identified. Seropositive sera increased from 18 per cent in six-month-old foals to 60 per cent in year-old animals, with EEV serotype 1 exhibiting the highest prevalence. Antibodies against EEV has also been detected in the African elephant (Loxodonta africana).2
The transmission of EEV is seasonal in the temperate regions of southern Africa and virus has been recovered from horses between December and July. Vector activity is terminated by the onset of winter and by low precipitation.19 The distribution of EEV appears to be determined by the distribution and abundance of competent vectors. Serotype 1 of EEV was first recovered from a pool of processed Culicoides in 1969.26 Subsequently, experimental feeding of wild caught Culicoides including C. imicola and C. bolitinos on infected blood through a chicken skin membrane, has shown that serotype 1 of EEV replicates in engorged females.28, 29 Other species of wild-caught Culicoides have not been examined in sufficient numbers to establish their competence as vectors.
A study of the prevalence of serotype-specific antibody in groups of resident brood mares in the major horse breeding areas of South Africa,14 revealed that 56,9 per cent of 518 individual donors were positive for neutralizing antibody to one or multiple serotypes of EEV. Between 19,2 and 43,6 per cent of the animals were infected with only a single serotype, while the percentage of mares showing evidence of previous infection with more than one serotype varied between 3,3 and 33,8 per cent on individual farms. It would therefore appear that prevailing conditions within a defined area influence the potential of the vectors to transmit the endemic serotypes of EEV.11
Individual serotypes are responsible for localized foci of seroconversion with an unpredictable seasonal and year-to-year distribution. Prior to 1990, and more recently, serotype 1 appears to have been the predominant serotype in South Africa, followed by serotypes 6 and 7, while antibody to the remaining serotypes is only occasionally identified. Antibody to serotype 2, which appears to have been the predominant serotype in 1967,7 was identified in only 8 of 518 mares sampled between 1995 and 1998. In a serological and virological survey that was conducted on stud farms in the Western Cape Province of South Africa from March to April 2008, serotype 4 was identified,11 while in another study (2011-2017), in which virus isolates from horses from different provinces of South Africa were analysed, serotypes 1 and 4 were identified.25
Prior to 2008, EEV was only reported from regions of southern Africa, however in 2008 through January 2009, a febrile disease resembling EE was described in horses from dozens of stable across Israel. Affected animals demonstrated clinical signs, including fever, unrest, decreased appetite, oedema of the neck, legs, lips and eyelids, accelerated pulse and breathing rates, and congested mucosae.17 It was estimated that 80 per cent of the horse population was affected during the outbreak, with 90 per cent of animals recovering without additional complication, and no deaths being reported.33 DNA array analysis17 along with sequence analysis of partial segment 10 sequences of viral isolates made from affected horses, identified the virus as EEV. In addition, partial segment 2 sequences showed 92,3 per cent nucleotide sequence identity to the Kaalplaas (serotype 4) reference strain.1 Retrospective serological analysis on samples held at the Kimron Veterinary Institute in Israel, indicated that EEV had been present in the country as early as 2001 and that it had been circulating onwards through 2008.33 Subsequent serological surveys conducted on blood samples taken from healthy horses from 2014-2015 from Israel, Palestine, and Jordan, indicated variable EEV seroprevalence rates of 58,2 per cent, 48,5 per cent and 2 per cent, respectively.27
Except for the Middle East, EEV has also been isolated from a dead horse in Pune from the Western Indian State of Maharastra, in 2008. Phylogenetic analysis of the virus segment 2 sequence grouped the isolate with the Bryanston serotype (serotype 1) of EEV.36 More recent serological surveys for EEV conducted from 2007-2010 have confirmed the presence of the virus in East and West African countries, including Ethiopia, Ghana and The Gambia. A Gambian isolate made from a donkey in 2009 demonstrated high segment 2 sequence identity (98,8 per cent) to viral isolates previously made from Israel in 2009.1, 18 In this study, serum samples from Morocco tested negative for EEV, leading the authors to conclude that the Sahara Desert acts as a natural geographical barrier to the spread of infected insects to certain North African countries.18
Pathogenesis
The pathogenesis of EE has not been studied. It may be similar to some of the other orbivirus infections, such as AHS and BT, in which vascular damage is thought to play a role in the development of disease. The pathogenicity of the different serotypes of EEV to horses has not been systematically investigated.
Clinical signs
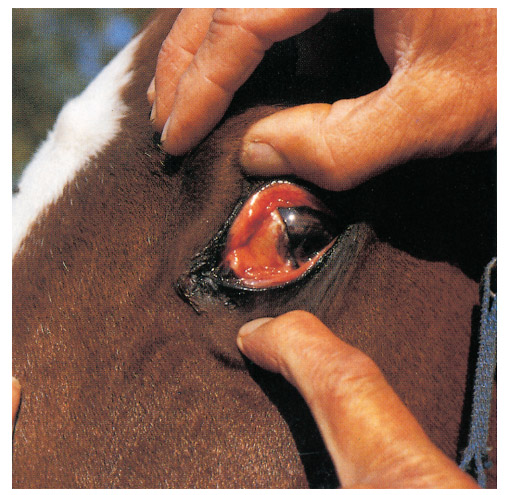
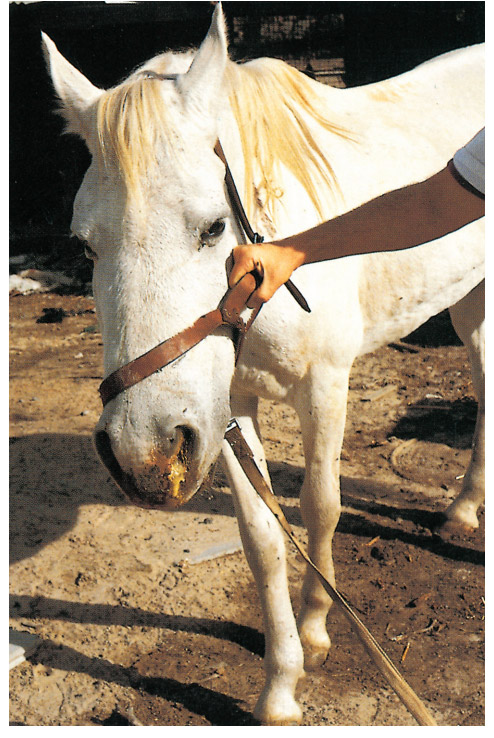
The incubation period of the disease is approximately three to six days. More than 90 per cent of animals show either no obvious signs of infection or develop only very mild clinical signs. Initially most affected horses develop either a slightly elevated rectal temperature (39 °C) for one or two days or a high fever (40 to 41 °C) for between one and five days. Fever may be accompanied by varying degrees of listlessness and inappetence. There may be increased pulse and respiratory rates, and in some animals a slight, reddish-brown discoloration of the mucous membranes as a result of congestion and mild icterus (Figure 1). Occasionally, varying degrees of swelling of the eyelids and supraorbital fossae may be observed (Figure 2). Central nervous system involvement, respiratory distress, acute heart failure and/or abortion have been observed in individual animals from which EEV has been isolated,8 but until such manifestations have been reproduced experimentally, their significance remains questionable.25
Pathology
Post-mortem examination of horse carcasses from which EEV has been isolated, have revealed varying degrees of lung oedema and hydropericardium, slight hepatomegaly and splenomegaly, petechiae in serosal surfaces (particularly of the intestines), hyperaemia of the glandular part of the stomach and, in some cases, congestion and oedema of the brain.
Diagnosis
Infection with EEV is usually subclinical and most cases can be confirmed using either molecular or serological testing. In animals showing a febrile reaction or clinical signs, the preferred method to confirm an infection is through the detection of viral nucleic acid from either EDTA blood or tissues samples (e.g. spleen, thymus, liver, lung, and brain) using the polymerase chain reaction. Sequencing of the EEV genome has led to the development of highly sensitive diagnostic (targeting segment 7, 9 or 10)16, 23, 25 and type specific (targeting segment 2)16 real-time reverse transcriptase polymerase chain reaction tests , that are able to provide a definitive diagnosis and genotype (serotype) identity, within as little as one working day of receipt of a sample in the laboratory.
An antigen capture ELISA3, 4, 12 has been developed for the direct detection of EEV antigen. For this test, the virus is usually detected either from heparinized blood or tissues.
Equine encephalosis virus can be isolated in BHK21/C13 or a line of susceptible Vero cells or, alternatively, through the intracerebral inoculation of three- to five-day-old suckling mice. Mortality may be observed in the latter, six to eight days after the development of neurological signs and terminal paralysis. The capture ELISA is then performed using material taken from either the first or subsequent passage of the virus in tissue culture or mouse brain.
Seroconversion in paired serum samples has historically been demonstrated by CF test using antigens prepared from infected mouse brain.8 More recently, several ELISAs that can be used for the detection and quantification of EEV specific antibodies have been described. A single dilution, indirect, group-reactive ELISA has been developed34, 35 and can be used in seroepidemiological surveys to detect antibody in equine species. This assay is more reliable for the detection of IgG in sera than the CF or agar gel immunodiffusion (AGID) tests. A competitive ELISA for the detection of EEV specific antibody has also been developed.3, 5 This assay can be used to determine end-point titres in individual serum samples. Both ELISAs are specific and no cross-reactions to other orbiviruses have been encountered. Additional serological tests that are of use in EEV diagnostics and sero-surveillance studies, include the serum virus neutralization test, that can be used for the quantification of neutralizing antibodies against EEV13 as well as the plaque inhibition assay that is suitable for the serotyping of specific isolates.20, 22
Differential diagnosis
Confirmation of the diagnosis of a suspected case of equine encephalosis is essential, primarily to distinguish the infection from mild forms of AHS as well as other febrile diseases. This may be achieved by the recovery and identification of the responsible virus or detection of EEV antigen in blood samples from febrile horses, or the demonstration of seroconversion in paired serum samples (see Diagnosis).
Fever and the mild, non-specific signs of illness evident in a large proportion of horses infected with EEV are features of many other infectious diseases of horses in the endemic regions. In animals manifesting fever, listlessness, inappetence, and reddish-brown mucous membranes, babesiosis should be considered. The clinical signs of those animals showing swelling of the eyelids may be confused with those of AHS. However, the mortality rate of AHS in naive unvaccinated animals is usually much higher (about 90 per cent).
In cases where EEV is isolated from aborted foetuses, other primary infectious causes of abortion should be excluded. These include infections with equid herpesvirus 1, equine viral arteritis virus, Streptococcus spp. (particularly S. zooepidemicus), Babesia spp. (particularly B. equi), Klebsiella pneumonia, Pseudomonas aeruginosa, Borrelia burgdorferi, Chlamydia psittaci and Coxiella burnetii.
Control
As infection with EEV is generally subclinical no control measures are usually implemented in endemic areas. The stabling of horses from an hour or two before the sun has set until an hour or two after it has risen the next day (the period during which Culicoides midges are particularly active) is a most useful control method. If it is practical, stables should be screened against flying insects, and no lights that could attract insects should be left burning at night in or in close proximity to the stables. Insect repellents can be sprayed on horses and in the stables.
Affected animals should be treated symptomatically. Non-steroidal, anti-inflammatory drugs can be used to combat the fever.
References
- AHARONSON-RAZ, K., STEINMAN, A., BUMBAROV, V., MAAN, S., MAAN, N. S., NOMIKOU, K., BATTEN, C., POTGIETER, C., GOTTLIEB, Y., MERTENS, P. & KLEMENT, E., 2011. Isolation and phylogenetic grouping of equine encephalosis virus in Israel. Emerging Infectious Diseases, 17(10), 1883-1886.
- BARNARD, B. J. H., 1997. Antibodies against some viruses of domestic animals in southern African wild animals. Onderstepoort Journal of Veterinary Research, 64, 95-110.
- CRAFFORD, J., 2001. Development and validation of enzyme-linked immunosorbent assays for detection of equine encephalosis virus antibody and antigen. MSc Thesis, University of Pretoria.
- CRAFFORD, J. E., GUTHRIE, A. J., VAN VUUREN, M., MERTENS, P. P. C., BURROUGHS, J. N., BATTEN, C. A. & HAMBLIN, C., 2003. A group-specific, indirect sandwich ELISA for the detection of equine encephalosis virus antigen. Journal of Virological Methods, 112(1-2), 129-135.
- CRAFFORD, J. E., GUTHRIE, A. J., VAN VUUREN, M., MERTENS, P. P. C., BURROUGHS, J. N., G., H. P., BATTEN, C. A. & HAMBLIN, C., 2011. A competitive ELISA for the detection of group-specific antibody to equine encephalosis virus. Journal of Virological Methods, 174(1-2), 60-64.
- ELS, H. J. & VERWOERD, D. W., 1969. Morphology of bluetongue virus. Virology, 38, 213-219.
- ERASMUS, B. J., ADELAAR, T. F., SMIT, J. D., LECATSAS, G. & TOMS, T., 1970. The isolation and characterization of equine encephalosis virus. Bulletin de l’Office International des Epizooties, 74, 781-789.
- ERASMUS, B. J., BOSHOFF, S. T. & PIETERSE, L. M., 1978. The isolation and characterization of equine encephalosis and serologically related orbiviruses from horses. In: BRYANS, J. T. & GERBER, H., (eds.). Equine Infectious Diseases IV. Princeton, New Jersey: Veterinary Publications Incorporated, 447-450.
- GERDES, G. H. & PIETERSE, L. M., 1993. The isolation and identification of Potchefstroom virus: A new member of the equine encephalosis group of orbiviruses. Journal of the South African Veterinary Association, 64, 131-132.
- GORDON, S. J. G., BOLWELL, C., ROGERS, C. W., MUSUKA, G., KELLY, P., GUTHRIE, A., MELLOR, P. S. & HAMBLIN, C., 2017. The sero-prevalence and sero-incidence of African horse sickness and equine encephalosis in selected horse and donkey populations in Zimbabwe. Onderstepoort Journal of Veterinary Research, 84(1), e1-e5. PMID: 28582979.
- GREWAR, J. D., THOMPSON, P. N., LOURENS, C. W. & GUTHRIE, A. J., 2015. Equine encephalosis in thoroughbred foals on a South African stud farm. Onderstepoort Journal of Veterinary Research 82(1), Art. 966, 4 pages. doi: http:// dx.doi.org/10.4102/ojvr. v82i1.966.
- HAMBLIN, C., ANDERSON, E. C., MELLOR, P. S., GRAHAM, S. D., MERTENS, P. P. & BURROUGHS, J. N., 1992. The detection of African horsesickness virus antigens and antibodies in young Equidae. Epidemiology and Infection, 108, 193-201.
- HOWELL, P. G., GROENEWALD, D., VISAGIE, C. W., BOSMAN, A. M., COETZER, J. A. W. & GUTHRIE, A. J., 2002. The classification of seven serotypes of equine encephalosis virus and the prevalence of homologous antibody in horses in South Africa. Onderstepoort Journal of Veterinary Research 69, 79-93. PMID: 12092781.
- HOWELL, P. G., GROENEWALD, M., VISAGE, C. W., BOSMAN, A.-M., COETZER, J. A. W. & GUTHRIE, A. J., 2002. The classification of seven serotypes of equine encephalosis virus and the prevalence of homologous antibody in horses in South Africa. Onderstepoort Journal of Veterinary Research, 69, 79-93.
- LECATSAS, G., ERASMUS, B. J. & ELS, H. J., 1973. Electron microscopic studies on equine encephalosis virus. Onderstepoort Journal of Veterinary Research, 40, 53-58.
- MAAN, M., BELAGANAHALLI, M. N., MAAN, S. N., POTGIETER, C. A. & C., M. P., 2019. Quantitative RT-PCR assays for identification and typing of the Equine encephalosis virus. Brazilian Journal of Microbiology, 50. 287-296. doi: https://doi.org/10.1007/s42770-018-0034-1.
- MILDENBERG, Z., WESTCOTT, D., BELLAICHE, M., DASTJERDI, A., STEINBACH, F. & DREW, T., 2009. Equine encephalosis virus in Israel. Transboundary and Emerging Diseases, 56, 291.
- OURA, C. A. L., BATTEN, C. A., IVENS, P. A. S., BALCHA, M., ALHASSAN, A., GIZAW, D., ELHARRAK, M., JALLOW, D. B., SAHLE, M., MAAN, N., MERTENS, P. C. & MAAN, S., 2012. Equine encephalosis virus: evidence for circulation beyond southern Africa. Epidemiology and Infection, 140, 1982-1986. doi: 10.1017/S0950268811002688.
- PAWESKA, J. T., GERDES, G. H., WOODS, P. S. A. & WILLIAMS, R., 1999. Equine encephalosis in southern Africa: Current situation. In: WERNERY, U., WADE, J. F., MUMFORD, J. A. & KAADEN, O.-R., (eds.). Equine Infectious Diseases VIII. Newmarket:R&W Publications (Newmarket) Ltd, 303-305.
- PORTERFIELD, J., 1960. A simple plaque-inhibition test for the study of arthropod-borne viruses. Bulletin of the World Health Organisation, 22, 373-380. PMID: 14434290
- POTGIETER, A. C., PAGE, N. A., LIEBENBERG, J., WRIGHT, I. M., LANDT, O. & VAN DIJK, A., 2009. Improved strategies for sequence-independent amplification and sequencing of viral double-stranded RNA genomes. Journal of General Virology, 90, 1423-1432.
- QUAN, M., VAN VUUREN, M., HOWELL, P. G., GROENEWALD, D. & GUTHRIE, A. J., 2008. Molecular epidemiology of the African horse sickness virus S10 gene. Journal of General Virology 89, 1159-1168. PMID: 18420793. doi: http://dx.doi.org/10.1099/ vir.0.83502-0.
- RATHOGWAA, N. M., QUAN, M., SMIT, J. Q., LOURENS, C., GUTHRIE, A. J. & VAN VUUREN, M., 2014. Development of a real time polymerase chain reaction assay for equine encephalosis virus. Journal of Virological Method, 195, 205-210.
- RATINIER, M., CAPORALE, M., GOLDER, M., FRANZONI, G., ALLAN, K., NUNES, S. F., ARMEZZANI, A., BAYOUMY, A., RIXON, F., SHAW, A. & PALMARINI, M., 2011. Identification and characterization of a novel non-Structural protein of bluetongue virus. PLoS Pathogens 7(12), e1002477. doi: 10.1371/journal.ppat.1002477.
- SNYMAN, J., KOEKEMOER, O., VAN SCHALKWYK, A., VAN VUREN, P. J., SNYMAN, L., WILLIAM, J. & VENTER, M., 2021. Epidemiology and Genomic Analysis of Equine Encephalosis Virus Detected in Horses with Clinical Signs in South Africa, 2010–2017. Viruses, 13, 398. doi: https://doi.org/10.3390/v13030398.
- THEODORIDIS, A., NEVILL, E. M., ELS, H. J. & BOSHOFF, S. T., 1979. Viruses isolated from Culicoides midges in South Africa during unsuccessful attempts to isolate bovine ephemeral fever virus. Onderstepoort Journal of Veterinary Research, 46, 191-198.
- TIROSH-LEVY, S., GELMAN, B., ZIVOTOFSKY, D., QURAAN, L., KHINICH, E., NASEREDDIN, A., ABDEEN, Z. & STEINMAN, A., 2017. Seroprevalence and risk factor analysis for exposure to equine encephalosis virus in Israel, Palestine and Jordan. Veterinary Medicine and Science, 3, 82-90.
- VENTER, G. J., GROENEWALD, D. M., PAWESKA, J. T., VENTER, E. H. & HOWELL, P. G., 1999. Vector competence of selected South African Culicoides species for the Bryanston serotype of equine encephalosis virus. Medical and Veterinary Entomology, 13, 393-400.
- VENTER, G. J., GROENEWALD, D. M., VENTER, E. H., HERMANIDES, I. & HOWELL, P. G., 2002. A comparison of the vector competence of Culicoides (Avaritia) bolitinos and C. (A.) imicola for the Bryanston serotype of Equine encephalosis virus (EEV). Medical and Veterinary Entomology, (In Press). .
- VENTER, G. J., PAWESKA, J. T., WILLIAMS, R. & NEVILL, E. M., 1999. Prevalence of antibodies against African horsesickness and equine encephalosis in donkeys in southern Africa. In: WERNERY, U., WADE, J. F., MUMFORD, J. A. & KAADEN, O. -R., (eds.). Equine Infectious Diseases VIII. Newmarket: R&W Publications (Newmarket) Ltd, 299-302.
- VERWOERD, D. W., LOUW, H. & OELLERMANN, R. A., 1970. Characterization of bluetongue virus ribonucleic acid. Journal of Virology, 5, 1-7
- VILJOEN, G. J. & HUISMANS, H., 1989. The characterization of equine encephalosis virus and the development of genomic probes. Journal of General Virology, 70, 2007-2015.
- WESTCOTT, D., MILDENBERG, Z., BELLAICHE, S. L., MCGOWAN, S. L., GRIERSON, S. S., CHOUDHURY, B. & STEINBACH, F., 2013. Evidence for the Circulation of Equine Encephalosis Virus in Israel since 2001. PLoS One, 8(8), e70532.
- WILLIAMS, R., 1987. A single dilution enzyme-linked immunosorbent assay for the quantitative detection of antibodies to African horsesickness virus. Onderstepoort Journal of Veterinary Research, 54, 67-70
- WILLIAMS, R., DU PLESSIS, D. H. & VAN WYNGAARDT, W., 1993. Group-reactive ELISAs for detecting antibodies to African horsesickness and equine encephalosis viruses in horse, donkey, and zebra sera. Journal of Veterinary Diagnostic Investigations, 5, 3-7.
- YADAV, P. D., A., C. G., NYAYANIT, D. A., GUERRERO, L., JENKS, M. H., SARKALE, P., NICHOL, S. T. & MOURYA, D. T., 2018. Equine encephalosis virus in India, 2008. Emerging Infectious Diseases, 24(5), 898-901.
- ZWART, L., POTGIETER, C. A., CLIFT, S. J. & VAN STADEN, V., 2015. Characterizing non-structural protein ns4 of African horse sickness virus. PLoS One, 10(4), e0124281. doi: 10.1371/journal.pone.0124281.