- Infectious Diseases of Livestock
- Part 2
- Peste des petits ruminants
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Peste des petits ruminants
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Peste des petits ruminants (PPR) is a disease of goats, sheep, and taxonomically related species caused by a morbillivirus in the family Paramyxoviridae.47, 48, 51, 58 It is clinically and pathologically similar to rinderpest, and is the most economically important viral disease of small ruminants in the areas where it occurs.
The disease was first described in 1942 by Gargadennec and Lalanne45 when they investigated a previously unreported syndrome in sheep and goats in Côte d’Ivoire. Because of its clinical and pathological resemblance to rinderpest (RP) or peste bovine in French, they called the disease ‘peste des petits ruminants’ (PPR). It was soon recognized in other French West African colonies such as Dahomey (now Benin) and Senegal.28, 77 More than a decade later, workers in Nigeria studied a syndrome that occurred primarily in goats and was variously named stomatitis-pneumoenteritis- complex, pseudo-rinderpest, or ‘kata’.97, 116 It was initially considered to be distinct from PPR because of its predominance in goats, as opposed to sheep, as reported in the earlier French descriptions, and because of the crusty labial lesions commonly found in convalescent cases. Further investigations, however, clearly showed that the two diseases were indistinguishable,51, 56, 96, 98 and the disease is now universally referred to by its original French name.
In Africa today, PPR is widespread in the sub-Saharan and Sahelian zones and has been confirmed in Sudan42 and Ethiopia.91 There is no clinical evidence that PPR exists south of a line from Cameroon to Ethiopia, and only limited serological evidence for infection in adjacent zones of Uganda and Kenya.114 The virus and its disease are also widespread in Asia, having been reported in epidemic form affecting both sheep and goats in India, Iran, Israel, Pakistan, Turkey and much of the Arabian peninsula.3, 9, 14, 16, 44, 52, 62, 78, 89, 105 Serological evidence indicates its presence in Jordan65 (Figure 50.1). The chronological sequence of new reports of PPR gives the impression that the disease has spread steadily eastwards from its origin in West Africa. Molecular evidence, however, shows that non-African isolates are quite distinct from African isolates,112 and it seems probable that PPR was overlooked in areas where rinderpest and contagious caprine pleuropneumonia were or are common.
Similarly, although rinderpest virus (RPV) can infect sheep and goats in Africa, most experimental studies have induced only mild or subclinical infection.90, 102 It is therefore possible that some early descriptions of severe rinderpest in small ruminants were actually PPR.17, 57 El Hag Ali and Taylor42 used specific antisera and transmission studies to confirm that virus isolates originally considered to be RPV from goats were actually the first isolates of peste des petits ruminants virus (PPRV) in eastern Africa.
The disease has been widely reviewed.13, 24, 36, 64, 68, 94, 103
Aetiology
Several early studies showed the close relationship between PPR and RP viruses48, 76, 116 and for a time PPRV was considered to be a host-adapted variant of RPV. Field observation, however, showed that the disease is independent of RP and cattle, and serology has indicated that the immunological relationship between the two viruses is as distant as that between RPV and the other morbilliviruses (including measles and canine distemper).47, 48, 107, 121 Studies with monoclonal antibodies and nucleic acid techniques confirmed the distinction between the two viruses, and that PPRV is a completely separate pathogen.32, 33, 74 It has been proposed that RPV is the direct descendent of an ancestral morbillivirus from which the others evolved, and that PPRV diverged from RPV more recently than did measles and canine distemper viruses.74 If PPR originated in West Africa in 1942, the virus must have evolved either after the 1887 to 1896 RP pandemic or from strains of RP already established in Africa long before the infamous pandemic. Since neither option seems likely, an alternative might be that PPR originated in Asia and was independently introduced into Africa much as RP was introduced through infected livestock. Nucleic acid sequencing data, however, clearly distinguishes African PPRV isolates from those outside Africa,112 and it has been suggested that PPR may have evolved from the adaptation of RPV to small ruminants on at least two separate occasions, in Africa and Asia respectively. More needs to be known about the genomic variability of the morbilliviruses before such speculation can be accepted as fact. Currently at least four main genetic lineages of PPRV are distinguishable by nucleic acid sequencing (Figure 50.2).
The virus can be isolated in vitro in primary sheep and goat kidney and Vero cells, and, once isolated, it also grows readily in some bovine kidney cell lines.42, 48, 63, 93, 111 The physico-chemical characteristics, morphology and cytopathic effects produced by PPRV are similar to those of RPV, although intranuclear inclusion bodies are more common. 22, 40, 47, 51
The N polypeptide of PPRV has a molecular weight more like those of measles and canine distemper viruses than those of RPV,33, 35 and it is antigenically distinct from the N polypeptide of RPV.73 Furthermore, African isolates can be readily distinguished from non-African isolates on the basis of the electrophoretic mobility of their N polypeptides.112Nucleotide sequencing clearly shows that PPRV is a distinct and separate morbillivirus.75
Epidemiology
In the field PPRV causes disease in goats and sheep but not in cattle or pigs, although these latter species can be infected subclinically by experimental inoculation.76, 77, 79 Goats are usually considered to be more susceptible than sheep,8, 20 but this is not always the case.19, 89 Mornet and his colleagues76, 77 described oral erosions in some young calves, suggesting that cattle might rarely transmit infection rather than always acting as dead-end hosts; the converse of the situation with RP in cattle and small ruminants in Africa. Serological surveillance in West Africa during the Pan African Rinderpest Campaign has detected antibodies to PPRV in up to 15 per cent of cattle in some herds.10 It had been postulated that these antibodies could interfere with the development of specific immunity to RPin vaccinated cattle and account for the failure to reach high prevalences of antibody in West Africa despite apparently successful immunization campaigns that have eradicated RP from the sub-region.120 Cross-protection studies in goats showed that PPR vaccine protected against RP,29 so the presence of widespread PPR antibodies in cattle may actually have assisted the development of higher levels of population immunity than were actually measured by serological tests for RP alone. Perhaps of more concern is the risk, however small, that this transfer of PPRV into the increasingly RP susceptible cattle populations of Africa and Asia might lead to a cattle-adapted form of PPR—in effect a new form of RP.
Infectious virus can be detected for at least seven days after the onset of pyrexia in the ocular and nasal secretions, urine and faeces of infected animals.2, 58 The survival of infectious virus outside the body has received little attention. In one laboratory study it was shown to have a half-life of approximately two hours at 37 °C and to be destroyed within an hour at 50 °C.64 Comparable results were obtained using the Nig 75/1 strain in liquid medium.110 The half-life of this virus at 56, 37, 4 and –20 °C was 2,2 minutes, 3,3 hours, 9,9 days and 24,2 days respectively. Between pH5,85 and 9,5 the virus was stable, but it was rapidly inactivated below pH 4,0 or above pH11,0. Lymph nodes from carcasses stored at 4 °C contain viable virus for at least eight days.64
Transmission probably occurs predominantly by the inhalation of aerosols from nearby animals, or by nuzzling and licking between infected and susceptible animals. Occasionally, bedding, feed and water troughs recently used by infected animals may be a source of infection.
As with RPV, the underlying requirement for the maintenance cycle of PPRV is a regular supply of susceptible hosts plus sufficient animal movement to allow a high rate of contact between individuals and between herds. The single most important epidemiological factor that most authors agree on, is that virtually all outbreaks can be traced to livestock movements, either migration to new areas or the introduction of new animals. Local aspects of the epidemiology of the disease require further investigation if conflicting observations on the seasonality of outbreaks and main endemic areas are to be clarified. Whereas RP in Africa is mainly confined to large populations of migratory and semi-migratory cattle, PPRV is maintained also at the village and urban level. Goats and sheep, as a readily available source of meat, are a universal commodity in villages, towns and even cities throughout Africa, the Middle East and Asia. In Oman the virus is more prevalent in unconfined urban and suburban goats than in those on fenced farms or in isolated villages.112 Whether or not the virus can be maintained in a peri-urban situation without periodic reinfection from outside must depend upon the size of the population. Most urban populations are probably too small to maintain the virus independently and it has been noted that PPR was particularly frequent in Kumasi, Ghana, after festivals when large numbers of sheep were imported for ritual consumption.18
Direct climatic effects are referred to in many reports: the onset of the rains and the humid season has been seen as a major triggering factor,21, 82, 116 as has the beginning of the cool and dusty ‘harmattan’ period in West Africa, which begins in November.86 Both are significant climatic changes that could play a role in the development of secondary bacterial pneumonias that complicate PPR. The indirect effects of climate on husbandry may also play a role. Disease flares up after the increased marketing of goats during periods of drought in the drier northern zone of Nigeria and, conversely, following marketing of goats to protect growing crops during the rains in the southern zone.41, 43, 86, 116 Although the disease occurs in animals of all ages, the highest prevalence is found in young stock usually less than two years old — sometimes in animals as young as four to eight months, in which maternal immunity would have just waned to unprotective levels. The seasonality of outbreaks might also be related to seasonal kidding and lambing patterns.82
Many authors have noted that outbreaks are more commonly reported from the humid forest and derived savannahs, often after animals have been introduced from the drier north.8, 18, 21, 38 The breeds of sheep and goats in the Sahelian regions are inherently more resistant to the disease than are the notably susceptible dwarf varieties of the southern humid zone.21, 38 This might suggest that the main endemic sources of PPR are the migratory sheep and goat herds of the Sahel, though most data show that the disease is no more prevalent there than in the humid south.82, 107, 108, 115 Nevertheless, the morbidity and case mortality rates in outbreaks in the south are often very high, suggesting sporadic epidemics rather than true endemicity. For instance, in Nigeria, case mortality rates ranging from 20 to over 90 per cent have frequently been reported,12, 84, 86, 87 whilst rates of only 4 to 5 per cent were recorded in what one author considered to be a true endemic area.72 Certainly, serological surveys have shown that infection is far more prevalent than can be assessed from clinical reports84, 107, 108 and that many, if not most, infections are subclinical or insufficiently severe to attract veterinary attention.
Whilst it is widely believed that goats are more susceptible to the disease than sheep,45, 64 outbreaks have occurred with both species being equally involved,109, 111 or mainly sheep.18, 104 In some cases this may simply reflect the proportions of the two species in the locality,18 but serology indicates that both species may suffer similar attack rates with resultant disease being more obvious in one or other species. Extrapolating from RP, in which species adaptation is a feature, it is possible that there are variants of PPRV adapted to either sheep or goats,109 and these should perhaps be looked for in future studies. Sub-Sahelian sheep are comparatively resilient to the disease,21 and several serological surveys have recorded higher prevalences of antibody in sheep than in goats.82, 107, 108, 115 Perhaps higher case mortality rates in goats account for the differences in antibody prevalences. In view of the current interest in mild RP of cattle in endemic areas, a renewed investigation of inapparently infected goats and sheep in the drier Sahelian zones might provide further insight into the epidemiology of this disease.
The last three decades have seen a considerable extension in the worldwide distribution of PPR. This is partly due to greater awareness of the disease encouraging its ‘discovery’ in new areas, but also the ease with which animals incubating the disease can be transported. In the first edition of this book94 it was predicted that PPRV would spread further south in Africa. This has not occurred, but it is difficult to understand why it has not done so and it may still only be a matter of time before it does.
Peste des petits ruminants virus has recently been isolated from camels in the Horn of Africa suffering from a widespread epidemic of serious pneumonia.
Antibiotics greatly reduced the severity of the disease, which could be fatal if left untreated. This suggests that secondary bacterial infections were the main problem, possibly as a sequel to a mild primary pneumonia caused by the virus, and further work is needed to clarify this.
A natural outbreak of PPR spread through a zoo in the United Arab Emirates killing wild sheep (Ovis orientalis laristanica), gazelles (Gazella dorcas), gemsbok (Oryx gazella) and a Nubian ibex (Capra ibex nubiana).44 Other potentially susceptible species such as Dama gazelle (Gazella dama), Arabian and scimitar-horned oryx (Oryx leucoryx and O. tao), red deer (Cervus elaphus) and blackbuck (Antilopa cervicapra) were not affected. White-tailed deer (Odocoileus virginianus), infected experimentally, developed either the acute fatal form or the subclinical form of the disease.50 After the introduction of local goats into a private zoological collection in Saudi Arabia, serological techniques confirmed PPR in unspecified deer and gazelles affected by a fatal disease.49 The comparative susceptibility of different species to PPR and RP viruses could help to illuminate the complexities of bovid taxonomy.
Fortunately, other species, such as peridomestic rats, which can be infected by experimental inoculation, do not appear to play an epidemiological role.61
Pathogenesis
The pathogenesis of PPR has not been studied but is assumed to be essentially similar to that of RP. Infection probably occurs via the oropharynx with subsequent virus multiplication in the draining lymph nodes. The ensuing viraemia transports the virus to other lymphoid tissues and susceptible epithelia throughout the body, where it multiplies, causing the cytopathology responsible for the development of lesions and disease.
Virus and viral antigens can be detected in blood, body secretions and lymphoid organs during the early stages of clinical disease. The titres of infectivity can reach 4,0 log10 TCID50 in oral, nasal, ocular and pharyngeal swabs and 5,0 log10 TCID50 per gram of faeces.2 During the later stages of disease both infectious virus and antigen are increasingly difficult to detect, probably as a result of the rising antibody levels.
Counter-immuno-electrophoresis (CIEP) can detect precipitating antibody as early as three days after infection in some animals, although most have detectable antibody only after seven or eight days.39 Precipitating antibody detected by the use of this technique seems to persist long enough for CIEP to be considered suitable for serological monitoring of vaccinated animals.69
Clinical signs
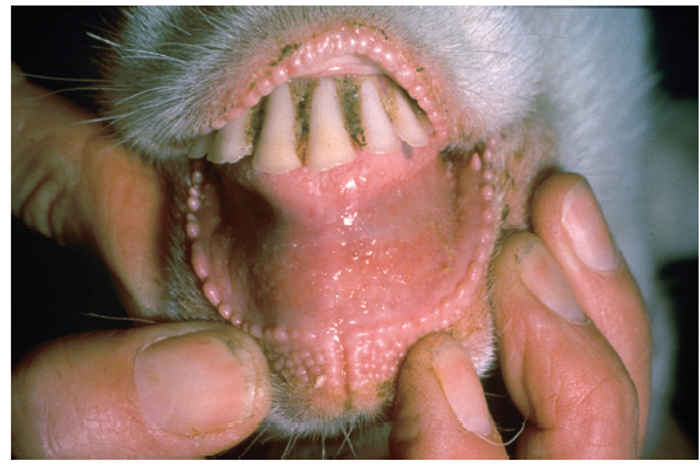
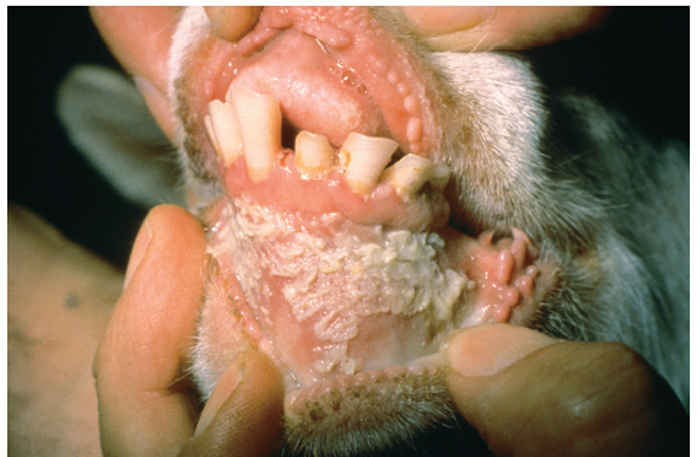
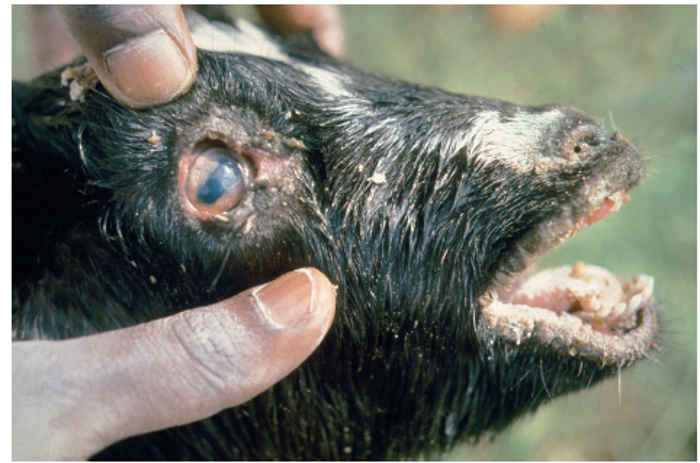
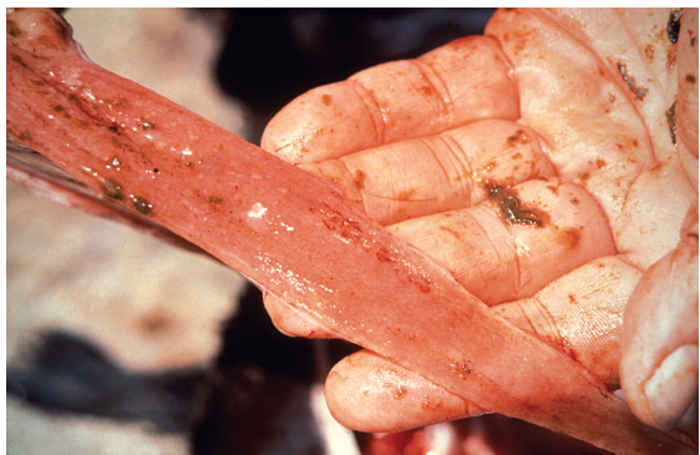
After an incubation period of between two and six days the first clinical sign is a rise in temperature that reaches 40 to 41 °C within two or three days. This is followed by depression, inappetence and development of serous nasal and ocular discharges which progressively become purulent and more profuse. The nasal discharge may block the nares and encrust the muzzle, causing the animal to snort and sneeze, while the ocular discharge may mat the eyelids together. One to two days after the start of pyrexia the buccal mucosa becomes congested and pale areas of necrosis become visible on the gums (Figure 50.3). In severe cases these increase in size and extend to involve the cheeks, dorsum of the tongue, hard palate and dental pad (Figure 50.4). The caseous epithelium easily sloughs, leaving shallow red erosions. Diarrhoea begins two or three days after the onset of pyrexia and death may occur some three to seven days later. Less severe and mild cases also occur, in which all or some of the signs develop to a lesser degree and the mortality rate is much lower.
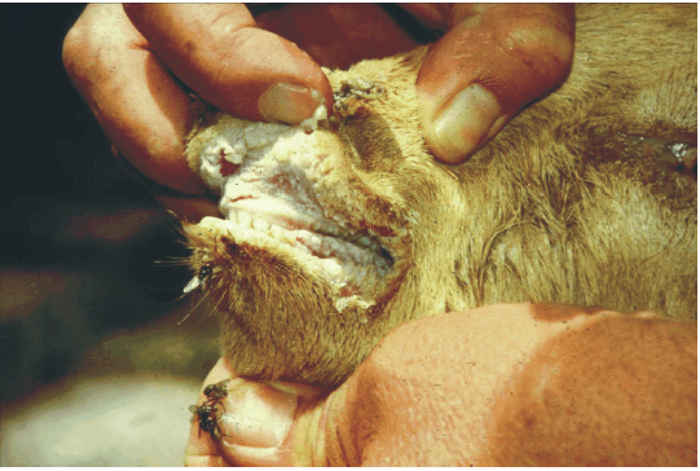
Two clinical signs that are frequently seen in PPR but not in RP are the formation of prominent crusty scabs (sometimes referred to in early descriptions as ‘proliferative’ lesions) along the outside of the lips (Figure 50.5), and the development of pneumonia in the later stages of the disease.
Some PPR-affected goats are also infected with isolates of Escherichia coli that produce a heat-labile enterotoxin that increases the clinical severity and mortality rate of PPR.4, 54
Pathology
Infected goats develop leukopenia, the degree of which may correlate with the severity of clinical disease.81, 113 Eosinopenia, monocytosis and a rise in the packed cell volume, presumably due to haemoconcentration resulting from the diarrhoea, have also been reported.81, 113
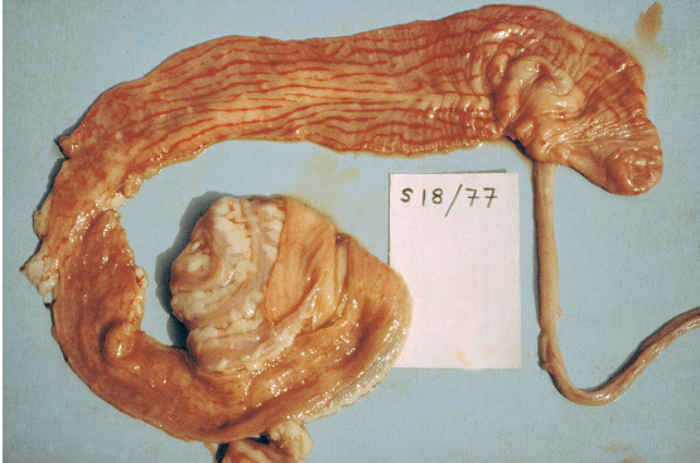
The carcass is frequently emaciated, soiled with faeces, and the eyelids, nares and lips encrusted with discharges. Erosions are found throughout the buccal cavity and pharynx, and, less frequently, the oesophagus. As in RP, erosive lesions in the forestomachs are often absent but may occasionally be found on the ruminal pillars and the leaves of the omentum.27 The abomasum and small intestine are usually dark red and congested rather than eroded, and haemorrhages have been reported. 97, 98 Peyer’s patches may be necrotic,116 and erosions and congestion can be found on the ileocaecal valve.76, 77 Marked congestion of the apices of the longitudinal folds of mucosa (zebra striping) may be seen in the large intestine and rectum (Figure 50.6).27, 76, 77, 97
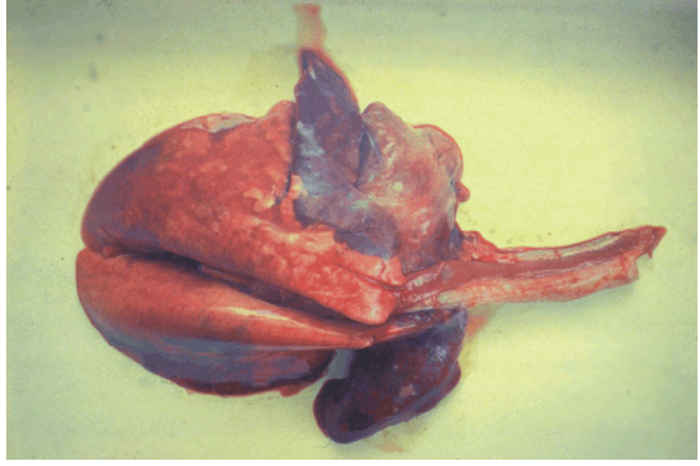
The upper respiratory tract mucosa is usually congested, erosions in the nares may be present, and tracheitis has been described.98 The lungs may be partly or diffusely congested, the affected areas appearing red and firm due to interstitial pneumonia (Figure 50.7). Changes in the trachea and lungs are more severe if there is concurrent bacterial infection.
Peripheral and visceral lymph nodes may be oedematous and friable, occasionally enlarged or haemorrhagic. The spleen is often congested and firm. The female genital mucosa may be congested and show epithelial erosions.
The erosions arise from hydropic degeneration and necrosis of epithelial cells in the stratum granulosum and do not penetrate the underlying stratum germinativum.27, 96, 97 Multinucleate giant cells, as well as eosinophilic intracytoplasmic and intranuclear inclusions are evident in the epithelium, 96 and inflammatory cells are prominent at the borders of the lesions. The ‘proliferative’ lesions on the lips contain necrotic debris, fibrin, mononuclear and polymorphonuclear leukocytes,29, 97 but there is no hyperplasia or vascularization to suggest true proliferation.
Figure 50.6 (top) Marked congestion of the apices of the longital folds of the mucosa of the large intestine and rectum (zebra striping) (top right); intestinal ulceration (right)
Lymphoid tissues are depleted of lymphocytes, with germinal centres particularly being affected. Pyknosis and karyorrhexis of lymphocytes are present in the cortex, and there is proliferation of reticuloendothelial cells along the medullary cords and in the sinuses.27 Comparable changes are seen in the Malpighian bodies of the spleen. Necrosis throughout the Peyer’s patches has been described,116 although a mere reduction in the numbers of lymphocytes without necrosis may be all that is evident in this tissue.97
Isolated areas of epithelial necrosis occur in the deep glands of the abomasum, and similar but more widespread changes are seen throughout the intestine, together with atrophy of villi and accumulation of debris in the glandular crypts. The lamina propria is infiltrated with lymphocytes, macrophages and eosinophils.54, 76, 77, 97
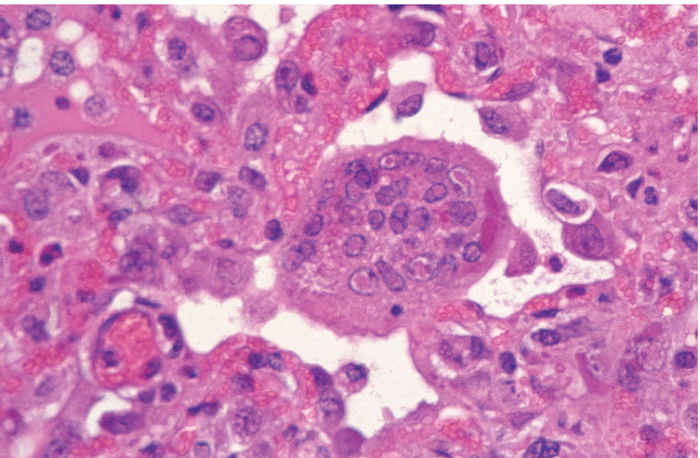
The respiratory tract mucosa may have areas of necrosis and hyperplasia, and inclusion bodies in epithelial cells.54, 97 Interstitial pneumonia characterized by the infiltration of lymphocytes and neutrophils, and proliferation of pneumocytes is common. A striking feature, not seen in RP but characteristic of measles, is severely enlarged multinucleate cells, often containing intracytoplasmic and intranuclear viral inclusions in the alveoli and terminal bronchioles (Figure 50.8). It seems probable that the virus is responsible for a primary pneumonia that is readily superinfected with secondary pathogens.
Small foci of necrotic hepatocytes have been described in the liver and there may be evidence of glomerulo-nephritis. 77
Paramyxovirus-like particles in degenerating cells in the lungs, particularly in alveolar macrophages, and intestinal epithelium have been described in ultrastructural studies,54 as have intranuclear and intracytoplasmic nucleocapsids in intestinal epithelial cells, with apparent extrusion of extracellular viral particles from microvilli.27
Diagnosis
A presumptive diagnosis of PPR can be made from clinical signs and lesions supported by epidemiological evidence such as the introduction of new and possibly diseased stock. In epidemic or virgin areas the diagnosis must be confirmed in the laboratory, either by the detection of virus-specific antigen or nucleic acid, or by the isolation of virus in cell culture. Retrospective confirmation can be made by demonstrating a rise in specific antibody in paired sera.
Specimens should be collected from up to 10 acutely affected animals with fever and early mucosal lesions. Clotted blood and whole blood in anticoagulant, together with ocular, nasal and oral swabs should be collected from live animals. If possible two or three animals should be slaughtered and 20 to 30 g each of spleen, lymph nodes, lung and gut mucosa also collected. The specimens should be chilled on ice, but not frozen, and dispatched to the laboratory as soon as possible.
Virus antigen in undiluted swab material or 30 per cent tissue suspensions is detected by hyperimmune serum to RPV in immunodiffusion or CIEP tests,51, 69, 83, 109 or, in contrast to RP, by haemagglutination.118 Hyperimmune serum to PPRV prepared in goats can also be used.5 Antigen can also be detected by immunohistochemical staining of tissues,26, 91 and by monoclonal antibody based immunocapture enzyme-linked immunosorbent assay (ELISA) techniques.66, 100
Specific nucleic acid sequences can be detected by DNA probes and by polymerase chain reaction (PCR) techniques. 34, 36, 88 The latter test has the advantage of producing large quantities of nucleic acid that can be sequenced to provide extra information about the lineage, or sub-type, of PPRV involved. This information is particularly important for tracing the origin of epidemic outbreaks.
The virus is isolated by inoculating swab material, buffy coat or 10 per cent tissue suspensions onto young monolayers of primary sheep or goat kidney cells. Vero cells, some lines of primary bovine kidney and bovine lymphoblasts93, 95 are also sensitive. Cytopathic effects develop after four days and are indistinguishable from those produced by RPV: rounding and shrinking of cells, strand formation and large flat syncytia. The identity of the virus as RPV or PPRV is confirmed by serological tests in which hyperimmune or convalescent sera to either virus is used. Distinction between the two viruses can then be made by comparative titrations against antisera to homologous and heterologous antisera, the homologous titre usually being much higher than the heterologous,107 or by their reaction with virus-specific monoclonal antibodies, or cDNA probes.32, 34, 36, 76, 88 The PCR can also be used for this purpose.
If cell culture facilities are unavailable, a pool of suspected tissue suspension should be inoculated into susceptible goats, sheep and cattle housed in suitable isolation facilities. Peste des petits ruminants usually causes severe disease in goats, milder disease in sheep and subclinical infection in cattle.42, 109 Most strains of RPV cause disease in cattle, but only subclinical or very mild disease in sheep and goats.
If neither antigen nor infectious virus can be detected in test animals, survivors should be bled two to four weeks after the first sampling and the paired sera assayed for antibody levels to the virus in neutralization tests.93, 107 A fourfold or greater increase in titre confirms the presumptive diagnosis. It is essential to use the homologous virus as titres to heterologous virus may be too low to show a four-fold rise. Other techniques for detecting and quantifying specific antibody to PPRV that have been developed include haemagglutination inhibition119 and ELISA.67, 101 Recent ELISAs take advantage of highly specific and pure antigens prepared by recombinant technology.55, 67
Differential diagnosis
The absence of clinical disease in cattle in India for nearly four years and the country’s decision to cease vaccination of all domestic bovine animals make it unlikely that small ruminant- adapted RP exists in India, or anywhere in Asia, as was once thought. Nevertheless, countries that have never reported PPR but continue to have RP should carefully examine all outbreaks of rinderpest-like disease in small ruminants for both PPR and RP viruses. All morbilliviruses isolated from goats and sheep should be further characterized with monoclonal antibodies, or by PCR and subsequent nucleotide sequencing to reveal their true identity.104
In contrast to RP, the pneumonia that frequently accompanies PPR increases the range of other conditions that can be confused with PPR. Contagious caprine pleuropneumonia (CCPP) is widespread and prevalent in many parts of sub-Saharan Africa and Asia and causes high mortality in goats. In uncomplicated cases there are no mucosal lesions or diarrhoea in CCPP, and sheep are not affected. Concurrent infections with other Mycoplasma spp. are common and can obscure a primary aetiology of PPR.85 The frequent demonstration of intranuclear inclusions in lung tissue has suggested concurrent infection with other viruses. Parainfluenza type 3 virus was thought to be involved, but serological and virological studies have not supported this.27 Adenovirus together with PPRV was recovered from the intestines of two goats that died of PPR in Nigeria.46
Peste des petits ruminants might be confused with footand- mouth disease or bluetongue, but this can be ruled out by closer examination of oral lesions, and the absence of diarrhoea. In East Africa PPR could be confused with Nairobi sheep disease (NSD), but oral lesions are minimal or absent in NSD and its distribution is restricted to areas infested with Rhipicephalus appendiculatus, and goats are rarely affected.
Nematodosis is a common cause of diarrhoea in sheep and goats, as is bacterial enteritis in kids and lambs caused particularly by E. coli and Salmonella serovars. The ‘proliferative’ labial scabs can be easily confused with the characteristic lesions of orf (contagious ecthyma),9 but this disease does not have the alimentary and pulmonary tract lesions that occur in PPR.
Control
Sick and in-contact goats and sheep should be isolated from other stock and effective quarantine imposed until at least one month after the complete recovery of the last clinical case. Strict control of animal movement is, however, often difficult to achieve. Goats and sheep, being more portable than cattle, are often transported long distances by motor vehicle, airplane and bicycle (the author once sat beside a man with a goat on his lap throughout a several hundred kilometre flight in West Africa). In these areas, vaccination is, therefore, the most effective form of control for PPR. Regrettably, it has only been sparingly applied in most affected areas.
Since the early observations of cross-immunity between PPR and RP several workers have used attenuated RPV vaccine to protect sheep and goats against PPR.18–20, 23, 70, 80, 108 This vaccine is safe in pregnant goats7 and confers clinical immunity against challenge with virulent PPRV for at least three years, although its longer-term duration is uncertain. Its regular use in Oman has already led to an increase in that country’s small ruminant population.112 Kids born to dams that have been immunized with RPV have heterologous colostral antibodies to PPRV for up to three months,15 predictably shorter than the four to eight months usually described in calves with homologous maternal immunity to RPV.25 Attenuated PPRV vaccines that protect goats against experimental challenge have now been developed and are being manufactured and marketed in Africa and Asia. One of these required 55 to 61 passages in Vero cells to achieve attenuation,37 whereas the other was still virulent after 60 passages in Vero cells and required at least 80 passages before it was suitably attenuated.6 Their efficacy in the field is still being assessed but it seems reasonable to assume that they will induce a long, probably lifetime, immunity in small ruminants. Recombinant poxvirus vaccines expressing either RPV or PPRV H and F antigens also protect against challenge with virulent PPRV,59, 71, 92 and chimaeric RPV vaccine expressing PPR glycoprotein is also available.30, 31
Infected premises should be thoroughly cleaned and disinfected with solvent, or agents with high or low pH.
There is no established therapy for affected animals. Hyperimmune serum administered during early clinical disease has been reported to help recovery,53 but antibiotics have little effect.11 Fluid replacement should be considered for valuable animals suffering from diarrhoea. Good husbandry and nursing are needed during convalescence.117
A macro-economic study in Niger concluded that the control of PPR through vaccination was cost-beneficial to the national goat industry.106
References
- ABEGUNDE, A.A., 1983. Problems connected with TCRV vaccination of sheep and goats. In: hill, d.h., (ed.). Peste des Petits Ruminants (PPR) in Sheep and Goats. Proceedings of the International Workshop, Ibadan, Nigeria, 1980. Addis Ababa: International Livestock Centre for Africa. pp. 79–81.
- ABEGUNDE, A.A. & ADU, F., 1977. Excretion of the virus of peste des petits ruminants by goats. Bulletin of Animal Health and Production in Africa,
- ABU ELZEIN, E.M.E., HASSANIEN, M.M., AL-AFALEQ, A.I., ABD ELHADI, M.A. & HOUSAWI, F.M.T., 1990. Isolation of peste des petits ruminants from goats in Saudi Arabia. The Veterinary Record, 127, 309–310. 25, 307–311.
- ADETOSOYE, A.I. & OJO, M.O., 1983. Characteristics of Escherichia coli isolated from goats suffering from ‘peste des petits ruminants’. Tropical Animal Health and Production, 16, 119–122.
- ADU, F.D. & JOANNIS, T., 1985. Goat hyperimmune serum for the diagnosis of peste des petits ruminants. The Veterinary Record, 117, 472.
- ADU, F.D., JOANNIS, T., NWOSUH, E. & ABEGUNDE, A.A., 1990. Pathogenicity of attenuated peste des petits ruminants virus in sheep and goats. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 43, 23–26.
- ADU, F.D. & NAWATHE, D.R., 1981. Safety of tissue culture rinderpest vaccine in pregnant goats. Tropical Animal Health and Production, 13, 166.
- AKLAKU, I.K., 1980. Principal causes of mortality in small ruminants in Ghana. Bulletin de l’Office International des Epizooties, 92, 1227–1231.
- AMJAD, H., QAMAR-UL-ISLAM, FORSYTH, M., BARRETT, T. & ROSSITER, P.B., 1996. Peste des petits ruminants in goats in Pakistan. The Veterinary Record, 139, 118–119.
- ANDERSON, J. & MCKAY, J.A., 1994. The detection of antibodies against peste des petits ruminants virus in cattle, sheep and goats and the possible implications to rinderpest control programmes. Epidemiology and Infection, 112, 225–231.
- ANENE, B.M., UGOCHUKWU, E.I. & OMAMEGBE, J.O., 1987. The appraisal of three different pharmaceutical regimes for the treatment of naturally occurring peste des petits ruminants (PPR) in goats. Bulletin of Animal Health and Production in Africa, 35, 1–3.
- APPELL, M.J.G., GIBBS, E.P.J., MARTIN, S.J., MEULEN, V.T., RIMA, B.K., STEPHENSON, J.R. & TAYLOR, W.P., 1981. Morbillivirus diseases of animal and man. In: kurstak, d., (ed.). Comparative Diagnosis of Virus Diseases. Vol. IV. New York: Academic Press.
- APPIAH, S.N., 1982. Peste des petits ruminants (PPR) – A review. Bulletin of Animal Health and Production in Africa, 30, 179–184.
- ASMAR, J.A., RADWAN, A.I., ABI ASSI, H. & AL RASHEID, A., 1980. A PPR-like disease in sheep in Central Saudi Arabia. Evidence of its immunological relation to rinderpest; prospects for a control method. Annual Meeting of the Saudi Arabian Society of Biological Sciences. Unpublished. 2 pp.
- ATA, F.A., AL SUMRY, H.S., KING, G.J., ISMAILI, S.I. & ATA, A.A., 1989. Duration of maternal immunity to peste des petits ruminants. The Veterinary Record, 124, 590–591.
- ATHAR, M., MUHAMMED, G., AZIM, F., SHAKOOR, A., MAQBOOL, A. & CHAUDRY,N.I., 1995. An outbreak of peste des petits ruminants-like disease among goats in Punjab (Pakistan). Pakistan Veterinary Journal, 15, 140–143.
- BEATON, W.G.,1930. Rinderpest in goats in Nigeria. Journal of Comparative Pathology, 43, 301–307.
- BONNIWELL, M.A., 1980. The use of tissue culture rinderpest vaccine (TCRV) to protect sheep and goats against peste des petits ruminants in the Ashanti region of Ghana. Bulletin de l’Office International des Epizooties, 92, 1233–1238.
- BONNIWELL, M.A., 1983. The results of TCRV in Ghana. In: hill, d.h., (ed.). Peste des Petits Ruminants (PPR) in Sheep and Goats. Proceedings of the International Workshop, Ibadan, Nigeria, 1980. Addis Ababa: International Livestock Centre for Africa. pp. 85–87.
- BOURDIN, P., 1973. La peste des petits ruminants (PPR) et sa prophylaxie au Senegal et en Afrique de l’Ouest. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 26, 71–74.
- BOURDIN, P., 1983. History, epidemiology and economic significance of PPR in West Africa and Nigeria in particular. In: hill, d.h., (ed.). Peste des Petits Ruminants (PPR) in Sheep and Goats. Proceedings of the International Workshop, Ibadan, Nigeria, 1980. Addis Ababa: International Livestock Centre for Africa.
- BOURDIN, P. & LAURENT-VAUTIER, A., 1967. Note sur la structure du virus de la peste des petits ruminants. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 20, 383–386.
- BOURDIN, P., RIOCHE, M. & LAURENT, A., 1970. Emploi d’un vaccin antibovipestique produit sur cultures cellulaires dans la prophylaxie de la peste des petits ruminants au Dahomey – note preliminaire. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 23, 295–300.
- BRAIDE, V.B.,1981. Peste des Petits Ruminants: A review. World Animal Review, 39, 25–28.
- brown, r.d., 1958. Rinderpest immunity in calves. I. The acquisition and persistence of maternally derived antibody. Journal of Hygiene, Cambridge, 56, 427–434.
- BROWN,C.C., MARINER, J.C. & OLANDER, H.J., 1991. An immunohistochemical study of the pneumonia caused by peste des petits ruminants virus. Veterinary Pathology, 28, 166–170.
- BUNDZA, A., AFSHAR, A., DUKES, T.W., MYERS, D.J., DULAC, G.C. & BECKER, S.A.W.E., 1988. Experimental peste des petits ruminants (goat plague) in goats and sheep. Canadian Journal of Veterinary Research, 52, 46–52.
- CATHOU, Y., 1944. Rapport annuel du service de l’Elevage au Dahomey. Benin: Government Printer.
- COUACY-HYMANN, E., BIDJEH, K., ANGBA, A., DOMENECH, J. & DIALLO, A., 1995. Protection of goats against rinderpest by vaccination with attenuated peste des petits ruminants virus. Research in Veterinary Science, 59, 106–109.
- DAS, S.C., BARON, M.D. & BARRETT, T., 2000. Recovery and characterization of a chimeric rinderpest virus with the glycoproteins of peste des petits ruminants virus: Homologous F and H proteins are required for virus viability. Journal of Virology, 74, 9039–9047.
- DAS, S.C., BARON, M.D. & BARRETT, T., 2000. Recombinant rinderpest viruses expressing surface glycoproteins of peste des petits ruminants virus: Poster. Eleventh International Conference on Negative Strand Viruses, Quebec City, Canada, 24–29 June 2000: abstracts MAHY, B.W.J. & KOLAKOFSKY, D. PP. 55, 31.
- DIALLO, A., BARRETT, T., BARBRON, M., SHAILA, M.S. & TAYLOR, W.P., 1989. Differentiation of rinderpest and peste des petits ruminants viruses using specific cDNA clones. Journal of Virological Methods, , 127–136.
- DIALLO, A., BARRETT, T., BARBRON, M., MEYER, G. & LEFEVRE, P.-C.,1994. Cloning of the nucleocapsid protein gene of peste des petits ruminants virus: Relationship to other morbilliviruses. Journal of General Virology, 75, 233–237.
- DIALLO, A., BARRETT, T., BARBRON, M., SHAILA, M.S. & TAYLOR, W.P., 1989. Differentiation of rinderpest and peste des petits ruminants viruses using specific cDNA clones. Journal of Virological Methods, 23, 127–136.
- DIALLO, A., BARRETT, T., LEFEVRE, P.C. & TAYLOR, W.P., 1987. Comparison of proteins induced in cells infected with rinderpest and peste des petits ruminants virus. Journal of General Virology, 68, 2033–2038.
- DIALLO, A., LIBEAU, G., COUACY-HYMANN, E. & BARBRON, M., 1995. Recent developments in the diagnosis of rinderpest and peste des petits ruminants. Veterinary Microbiology, , 307–317.
- DIALLO, A., TAYLOR, W.P., LEFEVRE, P.C. & PROVOST, A., 1989. Attenuation d’une souche de virus de la peste des petits ruminants: Candidat pour un vaccin homologue vivant. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 42, 311–319.
- DUROJAIYE, O.A., 1983. Brief notes on history, epizootiology and the economic importance of PPR in Nigeria. In: hill, d.h., (ed.). Peste des Petits Ruminants (PPR) in Sheep and Goats. Proceedings of the International Workshop, Ibadan, Nigeria, 1980. Addis Ababa: International Livestock Centre for Africa.
- DUROJAIYE, O.A. & TAYLOR, W.P., 1984. Application of countercurrent immuno-electro-osmo-phoresis to the serology of peste des petits ruminants. Revue d’Elevage et de Medicine Veterinarie des Pays Tropicaux, 37, 272–276.
- DUROJAIYE, O.A., TAYLOR, W.P. & SMALE, C.,1985. The ultrastructure of peste des petits ruminants virus. Zentralblatt für Veterinärmedizin, 32, 460–465.
- DURTNELL, R.E., 1972. A disease of Sokoto goats resembling peste des petits ruminants. Tropical Animal Health and Production, 4, 162–164.
- EL HAG ALI, B. & TAYLOR, W.P., 1984. Isolation of peste des petits ruminants virus from the Sudan. Research in Veterinary Science, 36, 1–4.
- EZEOKOLI, C.D., UMOH, J.U., CHINEME, C.N., ISITOR, G.N. & GYANG, E.O., 1986. Clinical and epidemiological features of peste des petits ruminants in Sokoto Red goats. Revue d’Elevage et de Médicine Vétérinaire des Pays Tropicaux, 39, 269–273.
- FURLEY, C., TAYLOR, W.P. & OBI, T.U., 1987. An outbreak of peste des petits ruminants in a zoological collection. The Veterinary Record, 121, 443–447.
- GARGADENNEC, L. & LALANNE, A., 1942. La peste des petits ruminants. Bulletin des Services Zootechniques et des Epizooties de l’Afrique Occidental Francaise, 5, 16–21.
- GIBBS, E.P.J., TAYLOR, W.P. & LAWMAN, M.J.P., 1977. Isolation of adenoviruses from goats affected with peste des petits ruminants in Nigeria. Research in Veterinary Science, 23, 331–335.
- GIBBS, E.P.J., TAYLOR, W.P., LAWMAN, M.J.P. & BRYANT, J., 1979. Classification of peste des petits ruminants virus as the fourth member of the genus Morbillivirus. Intervirology, 11, 268–274.
- GILBERT, Y. & MONNIER, J., 1962. Adaptation du virus de la peste des petits ruminants aux cultures cellulaires—note preliminaire. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 15, 321–335.
- HAFEZ, S. M., AL-SUKAYAN, A., DELA-CRUZ, D., BEKAIRI, S.I. & RADWAN, A.I., 1989. In: abdul aziz, abu-zinada, h., goriup, p.d. & nader, i.a., (eds). Proceedings of the First Symposium, Riyadh, 1987. Riyadh: National Commission for Wildlife Conservation and Development, Publication No. 3.
- HAMDY, F.M. & DARDIRI, A.H., 1976. Response of white-tailed deer to infection with ‘peste des petits ruminants’ virus. Journal of Wildlife Diseases, 12, 191–196.
- HAMDY, F.M., DARDIRI, A.H., NDUAKA, O., BREESE, S.S. & IHEMELANDU, E.C., 1976. Etiology of the stomatitis pneumoenteritis complex in Nigerian dwarf goats. Canadian Journal of Comparative Medicine, 40, 516–522.
- HEDGER, R.S., BARNETT, I.T.R. & GRAY, D.F., 1980. Some virus diseases of domestic animals in the Sultanate of Oman. Tropical Animal Health and Production, 17, 83–88.
- IHEMELANDU, E.C., NDUAKA, O. & OJUKWU, E.M., 1985. Hyperimmune serum in the control of peste des petits ruminants. Tropical Animal Health and Production, 17, 83–88.
- ISITOR, G.N., EZEOKOLI, C.D. & CHINEME, C.M., 1984. A histopathological and ultrastructural study of lesions of peste des petits ruminants. Tropical Veterinarian,2, 151–158.
- ISMAIL, T.M., YAMANAKA, M.K., SALIKI, J., EL-KHOLY, A., MEBUS, C. & YILMA, T., 1995. Cloning and expression of the nucleoprotein of peste des petits ruminants virus in baculovirus for use in serological diagnosis. Virology, 208, 776–778.
- ISOUN, T.T. & MANN, E.D., 1972. A stomatitis and pneumoenteritis complex of sheep in Nigeria. Bulletin of Epizootic Diseases of Africa, 20, 167–174.
- JOHNSON, R.H., 1958. An outbreak of rinderpest involving cattle and sheep. The Veterinary Record, 70, 457–461.
- JOHNSON, R.H. & RITCHIE, J.S.D., 1968. A virus associated with pseudo-rinderpest in Nigerian dwarf goats. Bulletin of Epizootic Diseases of Africa, 16, 411–417.
- JONES, L., GIAVEDONI, L., SALIKI, J., BROWN, C., MEBUS, C. & YILMA, T., 1993. Protection of goats against peste des petits ruminants with a vaccinia virus double recombinant expresing the F and H genes of rinderpest virus. Vaccine, 11, 961–964.
- KITCHING, P. & BARRETT, T., 1991. Institute of Animal Health, Pirbright, UK. Personal communication.
- KOMOLAFE, O.O., UGWU, H.O. & EBIRIM, R.A., 1987. An investigation into the possible role of peridomestic rats in the epizootiology of peste des petits ruminants in goats. Bulletin of Animal Health and Production in Africa, 35, 207–210.
- KULKARNI, D.D., BHIKANE, A.U., SHAILA, M.S., VARALAKSHMI, P., APTE, M.P. & NARLADKAR, B.W., 1996. Peste des petits ruminants in goats in India. The Veterinary Record, 138, 187–188.
- LAURENT, A., 1968. Aspects biologique de la multiplication du virus de la peste des petits ruminants ou PPR sur les cultures cellulaires. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 21, 297–308.
- LEFEVRE, P.C., 1987. Peste des petits ruminants et infection bovipestique des ovins et caprins. Etudes et syntheses de l’I.E.M.V.T., nombre 5, douzieme edition. Institut d’Elevage et de Medicine Veterinaire des Pays Tropicaux, Maison Alfort, France.
- LEFEVRE, P.C., DIALLO, A., SCHENKEL, F., HUSSEIN, S. & STAAK, G., 1991. Serological evidence of peste des petits ruminants in Jordan. The Veterinary Record, 128, 110.
- LIBEAU, G., DIALLO, A., COLAS, F., GUERRE, l., 1994. Rapid differential diagnosis of rinderpest and peste des petits ruminants using an immunocapture ELISA. The Veterinary Record, 134, 300–304.
- LIBEAU, G., PREHAUD, C., LANCELOT, R., COLAS, F., GUERRE, L., BISHOP, D.H.L., DIALLO, A., 1995. Development of a competitive ELISA for detecting antibodies to the peste des petits ruminants virus using a recombinant nucleoprotein. Research in Veterinary Science, 58, 50–55.
- LOSOS, G., 1986. Peste des petits ruminants. In: losos, g., Infectious Tropical Diseases of Domestic Animals. London: Longman.
- MAJIYAGBE, K.A., NAWATHE, D.R. & ABEGUNDE, A., 1984. Rapid diagnosis of peste des petits ruminants (PPR) infection, application of immunoelectrophoresis (IEOP) technique. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 37, 11–15.
- MARINER, J.C., HOUSE, J.A., MEBUS, C. & VAN DEN ENDE, M.C., 1993. The use of thermostable Vero cell-adapted rinderpest vaccine as heterologous vaccine against peste des petits ruminants.,em> Research in Veterinary Science, 54, 212–216.
- MARTRENCHAR, A., ZOYEM, N. & DIALLO, A., 1999. Study of a mixed vaccine against peste des petits ruminants and capripox infections in northern Cameroon. Small Ruminant Research, 8, 14–19.
- MATHEW, Z., 1983. Epidemiology of PPR in Nigeria. In: HILL, D.H., (ed.). Peste des Petits Ruminants (PPR) in Sheep and Goats. Proceedings of the International Workshop, Ibadan, Nigeria, 1980. Addis Ababa: International Livestock Centre for Africa. pp. 22–23.
- MCCULLOUGH, K.C., OBI, T.U. & SHESHBERADARAN, H., 1991. Identification of epitope(s) on the internal virion proteins of rinderpest virus which are absent from peste des petits ruminants virus. Veterinary Microbiology, 26, 313–321.
- MCCULLOUGH, K.C., SHESHBERADARAN, H., NORRBY, E., OBI, T.U. & CROWTHER, J.R., 1986. Monoclonal antibodies against morbilliviruses. Revue Scientific et Technique de l’Office Internationale des Epizooties, 5, 411–427.
- MEYER, G. & DIALLO, A., 1995. The nucleotide sequence of the fusion protein gene of the peste des petits ruminants virus: The long untranslated region in the 5 end of the F-protein gene of morbilliviruses seems to be specific to each virus. Virus Research, 37, 23–38.
- MORNET, P., ORUE, J. & GILBERT, Y., 1956. Unicité et plasticité du virus bovipestique. A propos d’un virus naturel adapté sur petits ruminants. Comptes Rendus Hebdomadaires des Seances de l’Academie des Sciences, 242, 2886–2889.
- MORNET, P., ORUE, J., GILBERT, Y., THIERY, G. & MAMADOU, S., 1956. La ‘peste des petits ruminants’ en Afrique occidentale Francaise. Ses rapports avec la peste bovine. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 9, 313–342.
- NANDA, Y.P., CHATTERJEE, A., PUROHIT, A.K., DIALLO, A., INUI, K., SHARMA, R.N., LIBEAU, G., THEVASAGAYAM, J.A., BRUNING, A. & KITCHING, R.P., 1996. The isolation of peste des petits ruminants virus from Northern India. Veterinary Microbiology, 51, 207–216.
- NAWATHE, D.R. & TAYLOR, W.P., 1979. Experimental infection of domestic pigs with the virus of peste des petits ruminants. Tropical Animal Health and Production, 11, 120–122.
- OBI, T.U., 1983. Results of TCRV vaccination in Nigeria. In: HILL, D.H., (ed.). Peste des Petits Ruminants (PPR) in Sheep and Goats. Proceedings of the International Workshop, Ibadan, Nigeria, 1980. Addis Ababa: International Livestock Centre for Africa. pp. 82–83.
- OBI, T.U. & ODUYE, O.O., 1985. Haematological changes in natural and experimental peste des petits ruminants virus infection in goats. Revue d’Elevage et de Médicine Vétérinaire des Pays Tropicaux, 39, 11–15.
- OBI, T.U., OJO, M.O., TAYLOR, W.P. & ROWE, L.W., 1983. Studies on the epidemiology of peste des petits ruminants in southern Nigeria. Tropical Veterinarian, 1, 209–217.
- OBI, T.U. & PATRICK, D., 1984. The detection of peste des petits ruminants (PPR) virus antigen by agar gel precipitation test and counter-immunoelectrophoresis. Journal of Hygiene, 93, 579–586.
- OBI, T.U., ROWE, L.W. & TAYLOR, W.P., 1984. Serological studies with peste des petits ruminants and rinderpest viruses in Nigeria. Tropical Animal Health and Production, 16, 115–118.
- ONOVIRAN, O., MAJIYAGBE, K.A., MOLOKWU, J.U., CHIMA, J.C. & ABEGBOYE, D.S., 1984. Experimental infection of goats with Mycoplasma capri and ‘peste des petits ruminants’ virus. Revue d’Elevage et de Médicine Vétérinaire des Pays Tropicaux, 37, 16–18.
- OPASINA, B.A., 1983. Epidemiology of PPR in the humid forest and the dried savannah zones. In: hill, d.h., (ed.). Peste des Petits Ruminants (PPR) in Sheep and Goats. Proceedings of the International Workshop, Ibadan, Nigeria, 1980. Addis Ababa: International Livestock Centre for Africa. pp. 14–21.
- OPASINA, B.A. & PUTT, S.N.H., 1985. Outbreaks of peste des petits ruminants in village goat flocks in Nigeria. Tropical Animal Health and Production, 17, 219–224.
- PANDEY, K.D., BARON, M.D. & BARRETT, T., 1992. Differential diagnosis of rinderpest and PPR using biotinylated cDNA probes. The Veterinary Record, 131, 199–200.
- PERL, S., ALEXANDER, A., YAKOBSON, B., NYSKA, A., HARMELIN, A., SHEIKHAT, N., SHIMSHONY, A., DAVIDSON, M., ABRAMSON, M. & RAPAPORT, E., 1994. Peste des petits ruminants (PPR) of sheep in Israel: Case report. Israel Journal of Veterinary Medicine, 49, 59–62.
- PLOWRIGHT, W., 1952. Observations on the behaviour of rinderpest virus in indigenous African sheep. British Veterinary Journal, 108, 450–457.
- ROEDER, P.L., ABRAHAM, G., KENFE, G. & BARRETT, T., 1994. Peste des petits ruminants in Ethiopian goats. Tropical Animal Health and Production, 26, 69–73.
- ROMERO, C.H., BARRETT, T., KITCHING, R.P., BOSTOCK, C. & BLACK, D.N., 1995. Protection of goats against peste des petits ruminants with recombinant capripoxviruses expressing the fusion and haemagglutinin protein genes of rinderpest virus. Vaccine, 13, 36–40.
- ROSSITER, P.B., JESSETT, D.M. & TAYLOR, W.P., 1985. Microneutralization systems for use with different strains of peste des petits ruminants virus and rinderpest virus. Tropical Animal Health and Production, 17, 75–81.
- ROSSITER, P.B. & TAYLOR, W.P., 1994. Peste des Petits Ruminants. In: coetzer, j.a.w., thomson, g.r. & tustin, r.c. (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press Southern Africa.
- ROSSITER, P.B. & WARDLEY, R.C., 1985. The differential growth of virulent and avirulent strains of rinderpest virus in bovine lymphocytes and macrophages. Journal of General Virology, 66, 969–975.
- ROWLAND, A.C. & BOURDIN, P., 1970. The histological relationship between ‘peste des petits ruminants’ and kata in West Africa. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 23, 301–307.
- ROWLAND, A.C., SCOTT, G.R. & HILL, H.D., 1969. The pathology of an erosive stomatitis and enteritis in West African dwarf goats. Journal of Pathology, 98, 83–87.
- ROWLAND, A.C., SCOTT, G.R., RAMACHANDRAN, S. & HILL, H.D., 1971. A comparative study of ‘peste des petits ruminants’ and ‘kata’ in West African dwarf goats. Tropical Animal Health and Production, 3, 241–247.
- SALIKI, J.T., BROWN, C.C., HOUSE, J.A. & DUBOVI, E.J., 1994. Differential immunohistochemical staining of peste des petits ruminants and rinderpest antigens in formalin-fixed, paraffin embedded tissues using monoclonal and polyclonal antibodies. Journal of Veterinary Diagnosis and Investigation, 6, 96–98.
- SALIKI, J.T., HOUSE, J.A., MEBUS, C. & DUBOVI, E.J., 1994. Comparison of monoclonal antibody-based sandwich enzyme-linked immunosorbent assay and virus isolation for detection of peste des petits ruminants virus in goat tissues and secretions. Journal of Clinical Microbiology, 32, 1349–1353.
- SALIKI, J.T., LIBEAU, G., HOUSE, J.A., MEBUS, C. & DUBOVI, E.J., 1993. Monoclonal antibody-based blocking enzyme-linked immunosorbent assay for specific detection and titration of peste des petits ruminants virus antibody in caprine and ovine sera. Journal of Clinical Microbiology, 31, 1075–1082.
- SCOTT, G.R., 1962. Experimental rinderpest in Red Masai sheep.Bulletin of Epizootic Diseases of Africa, 10, 423–426.
- SCOTT, G.R., 1981. Rinderpest and peste des petits ruminants. In: GIBBS, E.P.J., (ed.). Virus Diseases of Food Animals. Vol. II. Disease monographs. London: Academic Press. pp. 401–432.
- SHAILA, M.S., PURUSHOTHAMAN, V., BHAVASAR, S., VENUGOPAL, K. & VENKATESAN, R.A., 1989. Peste des petits ruminants of sheep in India. The Veterinary Record, 125, 602.
- SHAILA, M.S., SHAMAKI, D., FORSYTH, M.A., DIALLO, A., GOATLEY, L., KITCHING, R.P. & BARRETT, T., 1996. Geographic distribution and epidemiology of peste des petits ruminants viruses. Virus Research, 43, 149–153.
- STEM, C., 1993. An economic analysis of the prevention of peste des petits ruminants in Nigerian goats. Preventive Veterinary Medicine, 16, 141–150.
- TAYLOR, W.P., 1979. Serological studies with the virus of ‘peste des petits ruminants’ in Nigeria. Research in Veterinary Science, 26, 236–242.
- TAYLOR, W.P., 1979. Protection of goats against ‘peste des petits ruminants’ with attenuated rinderpest virus. Research in Veterinary Science, 27, 321–324.
- TAYLOR, W.P., 1984. The distribution and epidemiology of ‘peste des petits ruminants’. Preventive Veterinary Medicine, 2, 157–166.
- TAYLOR, W.P., 1990. National Project on Rinderpest Eradication, 16/15 West Extension Area, Karol Bagh, New Delhi 110005, India. Unpublished data.
- TAYLOR, W.P. & ABEGUNDE, A., 1979. The isolation of peste des petits ruminants virus from Nigerian sheep and goats. Research in Veterinary Science, 26, 94–96.
- TAYLOR, W.P., AL BUSAIDY, S. & BARRETT, T., 1990. The epidemiology of peste des petits ruminants in the Sultanate of Oman. Veterinary Microbiology, 22, 341–352.
- UGOCHUKWU, E.I., 1983. Haematological observations in goats suffering from natural infection of pneumonia enteritis complex (peste of small ruminants virus). Bulletin of Animal Health and Production in Africa, 31, 337–341./li>
- WAMWAYI, H.M., ROSSITER, P.B. KARIUKI, D.P., WAFULA, J.S., BARRETT, T. & ANDERSON, J., 1995. Peste des petits ruminants antibodies in East Africa. The Veterinary Record, 136, 199–200.
- WALTON, C., JEROME, C.P. & MILREE, J.M., 1977. Prevalence of neutralizing antibodies to the virus of peste des petits ruminants (PPRV) in the sera of sheep and goats in Northern Nigeria. Final report of the Royal Veterinary College, West Africa Research Team, pp. 47–54. Royal Veterinary College, London.
- WHITNEY, J.C., SCOTT, G.R. & HILL, D.H., 1967. Preliminary observations on a stomatitis and enteritis of goats in Southern Nigeria. Bulletin of Epizootic Diseases of Africa, 15, 331–341.
- WOSU, L.O., 1989. Management of clinical cases of peste des petits ruminants (PPR) disease in goats. Beitrage Tropical Landwirtschaft, Veterinar Medicin, 27, 357–361
- WOSU, L.O., 1991. Haemagglutination test for diagnosis of peste des petits ruminants disease in goats with samples from live animals. Small Ruminant Research, 27, 357–361.
- WOSU, L.O. & EZEIBE, M.C.O., 1991. Haemagglutination-inhibition technique for definitive diagnosis of peste des petits ruminants virus specific antibody. Biomedical Letters, 46, 173–177.
- ZWART, D. & MACADAM, I., 1966. Resistance of two bulls against rinderpest without detectable neutralising antibodies in their sera. Bulletin of Epizootic Diseases in Africa, 14, 53–54.
- ZWART, D. & ROWE, L.W., 1966. The occurrence of rinderpest antibodies in the sera of sheep and goats in northern Nigeria. Research in Veterinary Science, 7, 504–511.