- Infectious Diseases of Livestock
- Part 2
- Porcine transmissible gastroenteritis
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Porcine transmissible gastroenteritis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Porcine transmissible gastroenteritis
Previous Authors: M B PENSAERT AND K VAN REETH
Current Authors:
A N VLASOVA - Assistant Professor, PhD, DVM, Food Animal Health Research Program, CFAES and CVM, OARDC, Ohio State University, 1680, Madison Ave, Wooster, Ohio, OH 44691, USA
S N LANGEL - Graduate Research Assistant, MS, PhD Candidate, Food Animal Health Research Program, CFAES and CVM, OARDC, Ohio State University, 1680 Madison Avenue, Wooster, Ohio, OH 44691, USA
L J SAIF - Professor, MS, PhD, Food Animal Health Research Program, CFAES and CVM, OARDC, Ohio State University, 1680 Madison Ave, Wooster, Ohio, OH 44691, USA
Introduction
Transmissible gastroenteritis (TGE) is a highly contagious disease of swine. It is characterized by vomiting, diarrhoea and high mortality (up to 100 per cent) in seronegative piglets less than two weeks of age. The causative agent (TGEV) is an enteric coronavirus that was first detected in the United States (US) in 1946 from outbreaks of acute diarrhoea with high mortality in piglets.1 The disease has since been reported in most parts of the world where intensive pig farming is practised, including Europe, Asia (Japan, Korea, Malaysia, Taiwan), the Americas (North, Central and South) and Africa (Zaïre, Ghana). It was a major cause of disease and death in piglets in the US, Europe and elsewhere from the 1960s-1980s, notwithstanding the widespread application of vaccines. Since the emergence and rapid spread throughout Europe of porcine respiratory coronavirus (PRCV),2 a deletion mutant of TGEV with respiratory tropism, a large proportion of the pig population has become immune to that virus and, consequently, also to TGEV. The endemic status of PRCV has thus markedly decreased the clinical and economic importance of TGEV (see Porcine respiratory coronavirus infection).
Aetiology
Being a member of the family Coronaviridae and genus Alphacoronavirus,TGEV is a single-stranded RNA virus. Virions are pleomorphic (diameter of 60 to 160 nm) with a fringe of widely spaced, club-shaped projections 12 to 25 nm in length.3 They are composed of three structural proteins: the nucleocapsid protein (N), a small membrane glycoprotein (M, formerly labelled E1), and a large glycoprotein constituting the viral projections (S, formerly named E2). The S glycoprotein bears an epitope that induces neutralizing antibodies as well as several epitopes that induce non-neutralizing antibodies.4
There is only one serotype of TGEV, although some antigenic variation exists between field strains with different geographical origins, and between virulent and attenuated strains. Transmissible gastroenteritis virus is antigenically closely related to PRCV, canine coronavirus, feline enteric coronavirus and feline infectious peritonitis coronavirus,5, 6 and is distantly related to porcine epidemic diarrhoea virus (PEDV)7 and human coronavirus 229E.8, 9
The virus is sensitive to lipid solvents and to various disinfectants, such as formaldehyde, sodium hypochloride, iodine and quaternary ammonium compounds. It is inactivated within a few days at 37°C and within a few hours when exposed to sunlight. Most strains of TGEV are only moderately sensitive to trypsin and are stable at pH3. The physicochemical properties of TGEV are provide elsewhere.10, 11
Epidemiology
Pigs are the only animals for which TGEV is pathogenic. Other non-porcine hosts, such as dogs, cats and foxes, can be infected experimentally but do not develop signs of illness. Whether these or other species are natural hosts of this virus is uncertain.
Transmission of TGEV is direct, i.e. pigs excreting the virus come into contact with susceptible individuals or indirect through contact of susceptible animals with virus contained in faeces or contaminated fomites. In experimentally infected pigs, TGEV was shed for approximately 14 days in faeces.12 TGEV can also be detected in lungs of some pigs.13
Herd immunity can be accomplished by intentionally exposing the sows to virulent TGEV to boost lactogenic (milk) immunity.14, 15 On isolated small farms, herd immunity is accomplished and TGEV infection is self-limiting. Conversely, on large farms (more than 200 sows) with a continuous farrowing system and continual influx of susceptible animals, TGEV can become endemic after the primary outbreak. Subsequent groups of pigs become infected after weaning when their maternal immunity wanes and they in turn infect other groups. The only clinical evidence of infection in these groups is mild diarrhoea at weaning. Typical TGE in young piglets that is associated with high mortality occurs when gilts or sows are TGEV seronegative or they fail to provide passive protection in the colostrum and milk to their offspring.
Generaly, outbreaks of TGE occur in winter suggesting that TGEV preservation in the frozen state in faeces or at cold temperatures can facilitate its transfer. Additionally, starlings (numerous in feedlots in winter) 16 and house flies 17 were indicated as potential mechanical vectors for TGEV transmission. It is not known how the virus survives the summer, but viral persistence on endemically- infected farms (in pigs, non-porcine reservoirs such as cats 18 and dogs 19 and mechanical vectors) is likely to be the main mechanism.10
Since the emergence of PRCV, typical and severe outbreaks of TGE with high mortality in new-born piglets have become rare in many European countries and in the US. In Belgium, no classical TGE has been reported since 1992 and only in the UK have occasional TGE outbreaks been reported (ca. 1990’s).20 A 1993 serological survey revealed specific TGEV antibodies in only fifteen per cent of Belgian swine herds and in 7.6 per cent of the slaughterhouse sow sera 21 using competitive inhibition ELISA for differentiation of TGEV- and PRCV-specific antibodies 22. This contrasts with the US, where 45 per cent of swine herds in Iowa were TGEV seropositive in 1995.23
Pathogenesis
The pathogenesis of TGE in the gastrointestinal tract has been studied in detail 12, 24 and has served as a model for coronavirus-associated diarrhoea in other animal species, such as cattle and dogs.
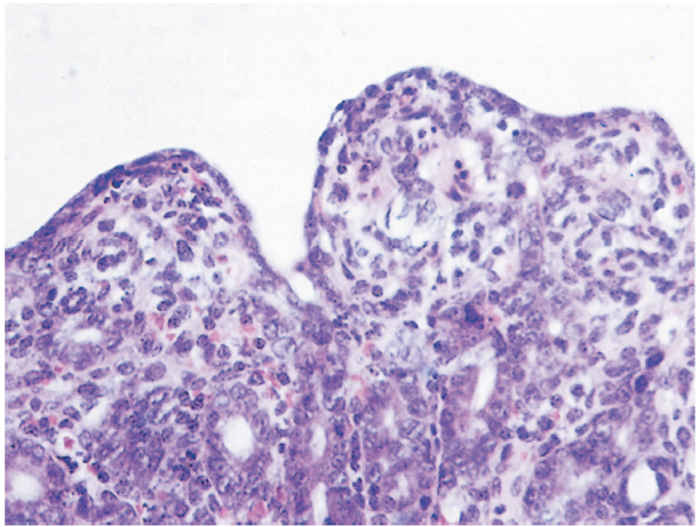
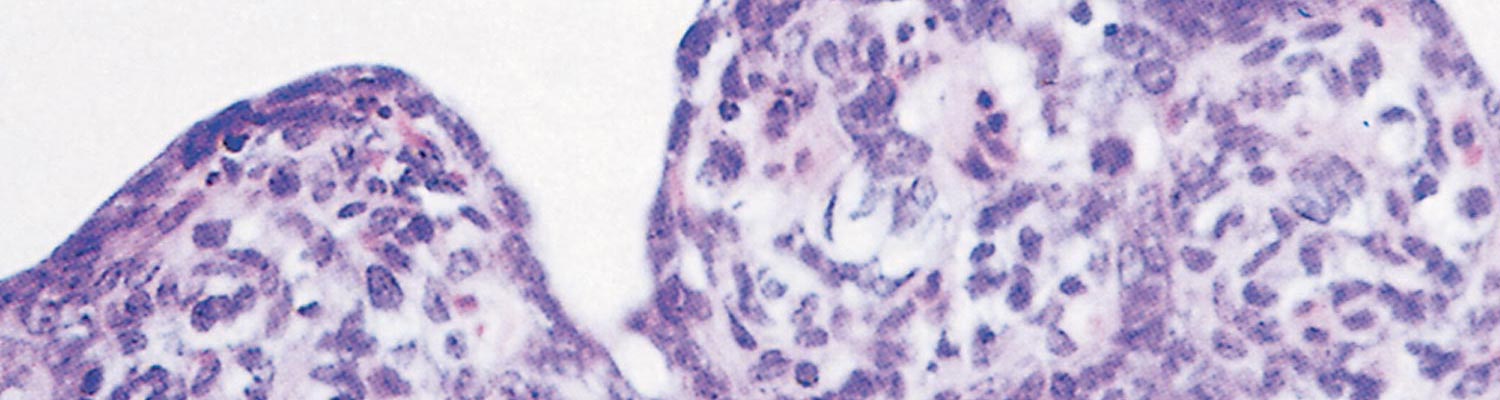
After ingestion, the virus passes intact through the stomach and replicates in enterocytes in multiple stages of differentiation along the entire small intestine, resulting in rapid and extensive epithelial cell necrosis, marked villous shortening or atrophy (reduction of the villous length/crypt depth ratio from 7:1 to 1:1) (Figure 1), accompanied by malabsorption and diarrhoea. Vomiting may be a consequence of the upper jejunum being affected. Dehydration can be severe, especially in piglets <1 week of age, and is frequently the immediate cause of death. As crypt epithelium is less affected, the villus epithelium regenerates early in the course of the disease. The lower mortality rate of older pigs following infection is likely due to the enterocytes being less susceptible to infection or to faster regenerative capacity. Additionally, older pigs may be more capable of compensation for the physiological effects of dehydration as well as having a more mature immune system. In pigs partially protected by lactogenic immunity, the proximal jejunum is usually spared from villous atrophy, explaining the milder course of the disease.
Pigs that recover from enteric infections with TGEV develop immunity manifested by the appearance of antibodies of all isotypes in the circulation and IgA [secretory IgA, (sIgA)] in intestinal secretions. Secretory IgA acquired from mother’s milk, provides protection rather than circulating antibodies (mostly IgG). Effective protection against enteric reinfection endures for at least six months after primary infection. Thereafter, if reinfection occurs, its effects are usually mild or subclinical and ephemeral. Although only indirect evidence exists concerning the role of cell mediated immunity in resistance to TGEV infection, it may play an important role in recovery or protection against reinfection.25, 26
During TGEV infection of sows, lymphocytes are stimulated by viral antigens and plasmablasts (antibody-secreting immature plasma cells) migrate to the mammary gland (MG) where they secrete IgA antibodies against TGEV in milk, initiating the gut-MG-sIgA axis. Piglets that ingest milk from an immune mother are usually protected against TGEV for as long as they continue to ingest such milk regularly and are not exposed to high amounts of infectivity. Effective passive (lactogenic) protection is afforded only if the sow has undergone intestinal infection and the gut-MG-sIgA axis has been stimulated; viral antigens inoculated parenterally induce little or no IgA antibodies against TGEV in milk.15 This mechanism which protects pigs from enteric infections is therefore different from that which confers protection in cattle. In the latter, IgG1 (that is selectively transported from circulation to the lacteal secretions) is the principal immunoglobulin for passive immunity of calves. 27, 28
Clinical signs
The clinical signs of TGE in fully susceptible pigs (epidemic situation) differ from those in swine in TGEV-endemically infected herds.
In epidemic situations, TGE spreads rapidly and is characterized by the sudden onset of acute diarrhoea in pigs of all ages, with neonatal animals being most severely affected. All the animals on a farm become infected within a week unless precautions are taken to prevent transmission of the virus. After an incubation period of 24 hours, the pigs may first vomit and then develop watery diarrhoea. After two to three days young pigs become severely dehydrated and most succumb to the infection. In suckling piglets infected during the first week of life the mortality rate may approach 100 per cent, but the rate decreases to almost zero in pigs that are older than two weeks of age. Due to the high levels of TGEV shed by infected piglets, all litters in the farrowing house will likely become infected within a few days. Sows may be sub-clinically infected or may become anorexic with diarrhoea and yield less milk or suffer agalactia. In growing and finishing pigs, watery diarrhoea usually develops, lasting five to seven days.
Sows in edemically infected herds show no signs of illness because they are immune to the virus. Piglets are also protected by antibodies in their dam’s milk as indicated previously. The advent of weaning results in the cessation of antibody supplementation with the milk, and the piglet’s intestine is again susceptible to infection (usually within one to three weeks of weaning). Infection in such animals is manifested by the development of diarrhoea and the spread of the virus to subsequent groups of recently weaned pigs. In endemic situations, diarrhoea may also occasionally develop in three to eight day-old piglets born to non-immune or partially immune sows/gilts. The mortality rate in partially protected piglets is variable.
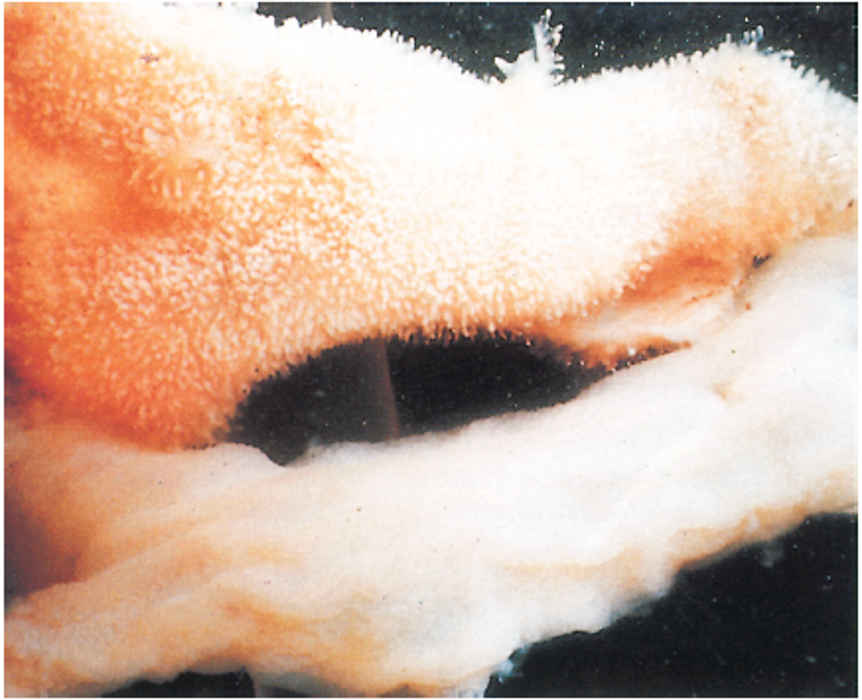
Pathology
The gross lesions in pigs that die of TGE are essentially those of dehydration accompanied by thin-walled, translucent small intestines. Histologically, villous atrophy is the main lesion, and the shortened villi are usually covered with flat to cuboidal epithelium (Figure 2), with the number of mitotic cells in the crypts markedly increased.29 In typical cases, these lesions are present throughout the small intestine, except for the first few centimetres of the duodenum. Lymphocyte infiltration is marked in the lamina propria and between the epithelial cells of the villi during recovery, when regeneration of the villous epithelium is evident.
Diagnosis
The diagnosis of TGE in neonatal piglets is best achieved by the detection of viral antigens or viral genome in enterocytes or faeces using immunofluorescence or immunohistochemistry on cryostat sections or mucosal impression smears of the small intestine or by real-time polymerase chain reaction (RT-PCR).30, 31 Ideally, specimens should be collected from pigs killed soon after the onset of diarrhoea. Faeces or intestinal contents may be tested, preferably before severe dehydration occurs, because extensive enterocyte necrosis (and therefore lack of cells susceptible to TGEV), decreased faecal output and electrolyte imbalance may interfere with viral detection.
Demonstration of TGEV in the faeces of pigs with diarrhoea is not always successful due to increased volume of the intestinal contents (and thus decreased TGEV concentration) and accelerated intestinal peristalsis. The continuous pig testis cell line ST is generally the most sensitive system for virus isolation.32 Virus replication in inoculated cell cultures is indicated by the occurrence of cytopathic effects or immunofluorescence. The latter is aimed at the detection of viral antigen although one or two passages in cell culture may be necessary to overcome toxicity of the inoculum and to increase viral replication. Coronavirus particles can be detected in negatively-stained preparations of faeces from piglets with diarrhoea by transmission electron microscopy. This technique is time and labour consuming, but has the advantage of also detecting other viruses. Additionally, TGE and porcine endemic diarrhoea (PED) coronaviruses and porcine deltacoronavirus (PDCoV) cannot be differentiated unless immunoelectron microscopy is applied.
Viral antigens can be demonstrated by ELISA systems (including those commercially available) in the faeces or intestinal contents of infected animals. Ideally, faecal samples collected from several animals at an early stage in the course of the disease should be examined.33
Serological diagnosis using TGEV- specific ELISA tests (including those commercially available 34 such as SVANOVIR® TGEV/PRCV-Ab) is often advantageous in older animals. 22 These antibody ELISA tests possess high sensitivity and specificity (>0.93) and do not cross-react with other swine coronaviruses.
Transmissible gastroenteritis virus can be propagated in primary, secondary, continuous or transformed cultures of porcine kidney (including PK-15 and SK-6), swine testis (ST) and thyroid cells. A continuous ST cell line permits the replication of most field strains. Virus neutralization tests using ST and SK-6 cells and a well-adapted virus strain in microplate systems are apt for routine serology. Cytopathic effects comprise syncytium formation and rounding of affected cells.32 Susceptible animals seroconvert within 14 days of initial infection.22, 35
However, in countries where TGEV, PRCV, PEDV or PDCoV occur, the serodiagnosis of TGE requires the use of an ELISA system that differentiates among them (see Porcine respiratory coronavirus infection).
Differential diagnosis
Typical in epidemics of TGE pigs of all ages may show acute diarrhoea with neonatal piglets being most severely affected while with outbreaks of diarrhoea caused by other enteropathogens (e.g. rotavirus, PEDV, PDCoV, Coccidia spp.and Escherichia coli) suckling or recently weaned pigs usually affected.
Epidemic and endemic TGE cannot be clinically differentiated from them,36 and laboratory tests (noted earlier) are required to confirm the diagnosis.37
Control
Multiple methods for immunizing sows with the aim of inducing lactogenic immunity and consequent protection of neonatal piglets have been attempted.15, 35, 38 A variety of viral vaccines (virulent, attenuated, inactivated and subunit) and routes of administration (oral, intranasal, intramuscular, subcutaneous, and intramammary).10, 15, 39 have been used for induction of lactogenic immunity. Intramuscular or intramammary inoculation of sows with live attenuated, inactivated or subunit vaccines during pregnancy does not affect complete protection, although vaccination reduced mortality rates. Attenuated virus does not stimulate the gut-MG-sIgA axis sufficiently for the induction of immunity similar to that observed following natural intestinal infection with virulent virus. At least two commercial vaccines against TGEV are available from Merck Animal Health (PROSYSTEM® TGE/Rota and PROSYSTEM® TREC) both contain a live-modified TGEV strain for combined oral-intramuscular administration. However, independent evaluation of the efficacy of the commercial TGEV vaccines is lacking.
Feeding virulent virus to sows is effective ininducing lactogenic immunity, but it also propagates the disease on the farm unless all the sows are immunized. This practice results in the rapid development of immunity in pregnant sows (particularly in those due to farrow two weeks or more after the start of the outbreak) and minimizes losses in new-born piglets. However, it may also result in dissemination of the virus or other pathogens to adjacent herds.
Elimination of endemic TGE in a herd can be attempted by deliberately and simultaneously exposing all the sows and weaned pigs to virulent autogenous virus, such as slurry of minced intestines of acutely infected pigs. If this is done, no weaning of piglets should occur during the following three to four weeks, so that there are no susceptible animals present in the herd while TGEV circulates on the farm.
Treatment of clinically affected new-born suckling piglets with electrolyte/glucose solutions is usually ineffective in field situations, but may reduce mortality in piglets that are one week or older.
Precautions should be taken to prevent the introduction of TGEV to susceptible herds by avoiding the introduction of animals and the access to the farm by potentially contaminated vehicles from TGEV affected farms.
Countries that are free of TGE should apply stringent measures to prevent the importation of the disease. This applies not only to the importation of live animals, but also, as has recently been shown, to the unprocessed tissues of slaughtered TGEV-infected animals.40
References
- BERNARD, S., LANTIER, I., LAUDE, H. & ET, AL., 1986. Detection of transmissible gastroenteritis coronavirus antigens by a sandwich enzyme-linked immunosorbent assay technique. American Journal of Veterinary Research, 47, 2441-2444.
- BOHL, E.H., GUPTA, R.K., OLQUIN, M.V. & ET AL., 1972. Antibody responses in serum, colostrum, and milk of swine after infection or vaccination with transmissible gastroenteritis virus. Infection and Immunity, 6, 289-301.
- BOHL, E.H. & SAIF, L.J., 1975. Passive immunity in transmissible gastroenteritis of swine: immunoglobulin characteristics of antibodies in milk after inoculating virus by different routes. Infection and Immunity, 11, 23-32.
- BOHL, E.H., 1989. Transmissible gastroenteritis virus (Classical Enteric Variant). In: PENSAERT M.B., ed. Virus Infections of Porcines. Amsterdam, Oxford, New York, Tokyo: Elsevier Science Publishers.
- BRIDGEN, A., DUARTE, M., TOBLER, K. & ET AL., 1993. Sequence determination of the nucleocapsid protein gene of the porcine epidemic diarrhoea virus confirms that this virus is a coronavirus related to human coronavirus 229E and porcine transmissible gastroenteritis virus. General Virology, 74 ( Pt 9), 1795-1804.
- BRIM, T.A., VANCOTT, J.L., LUNNEY, J.K. & ET AL., 1995. Cellular immune responses of pigs after primary inoculation with porcine respiratory coronavirus or transmissible gastroenteritis virus and challenge with transmissible gastroenteritis virus. Veterinary Immunology and Immunopathology, 48, 35-54.
- BUTLER, J.E., 1983. Bovine immunoglobulins: an augmented review. Veterinary Immunology and Immunopathology, 4, 43-152.
- CALLEBAUT, P., PENSAERT, M.B. & HOOYBERGHS, J.A., 1989. A competitive inhibition ELISA for the differentiation of serum antibodies from pigs infected with transmissible gastroenteritis virus (TGEV) or with the TGEV-related porcine respiratory coronavirus. Veterinary Microbiology, 20, 9-19.
- CARMAN, S., JOSEPHSON, G., MCEWEN, B. & ET AL., 2002. Field validation of a commercial blocking ELISA to differentiate antibody to transmissible gastroenteritis virus (TGEV) and porcine respiratory coronavirus and to identify TGEV-infected swine herds. Journal of Veterinary Diagnostic Investigation, 14, 97-105.
- DELMAS, B., GELFI, J. & LAUDE, H., 1986. Antigenic structure of transmissible gastroenteritis virus. II. Domains in the peplomer glycoprotein. General Virology, 67 ( Pt 7), 1405-1418.
- DOYLE, L.P. & HUTCHINGS, L.M., 1946. A transmissible gastroenteritis in pigs. Journal of the American Veterinary Medical Association, 108, 257-259.
- FORMAN, A.J., 1991. Infection of pigs with transmissible gastroenteritis virus from contaminated carcases. Australian Veterinary Journal, 68, 25-27.
- GOUGH, P.M. & JORGENSON, R.D., 1983. Identification of porcine transmissible gastroenteritis virus in house flies (Musca domestica Linneaus). American Journal of Veterinary Research, 4, 2078-2082.
- HAELTERMAN, E.O., 1972. On the pathogenesis of transmissible gastroenteritis of swine. Journal of the American Veterinary Medical Association, 160, 534-540.
- HOOPER, B.E. & HAELTERMAN, E.O., 1969. Lesions of the gastrointestinal tract of pigs infected with transmissible gastroenteritis. Canadian Journal of Comparative Medicine, 33, 29-36.
- HORZINEK, M.C., LUTZ, H. & PEDERSEN, N.C., 1982. Antigenic relationships among homologous structural polypeptides of porcine, feline, and canine coronaviruses. Infection and Immunity, 37, 1148-1155.
- KAJI ,T. & SHIMIZU, Y., 1978. Passive immunization against transmissible gastroenteritis virus in piglets by ingestion of milk of sows inoculated with attenuated virus. National Institute of Animal Health Quaterly, 18, 43-52.
- KEMENY, L.J., 1978. Isolation of transmissible gastroenteritis virus from pharyngeal swabs obtained from sows at slaughter. American Journal of Veterinary Research, 39, 703-705.
- KIM, L., CHANG, K.O., SESTAK, K. & ET AL., 2000. Development of a reverse transcription-nested polymerase chain reaction assay for differential diagnosis of transmissible gastroenteritis virus and porcine respiratory coronavirus from feces and nasal swabs of infected pigs. Journal of Veterinary Diagnostic Investigation, 12, 385-388.
- KIM, S.Y., SONG, D.S. & PARK, B.K., 2001. Differential detection of transmissible gastroenteritis virus and porcine epidemic diarrhea virus by duplex RT-PCR. Journal of Veterinary Diagnostic Investigation, 13, 516-520.
- LARSON, D.J., MOREHOUSE, L.G., SOLORZANO, R.F. & ET AL., 1979. Transmissible gastroenteritis in neonatal dogs: experimental intestinal infection with transmissible gastroenteritis virus. American Journal of Veterinary Research, 40, 477-486.
- LIN, C.M., GAO, X., OKA, T. & ET AL., 2015. Antigenic relationships among porcine epidemic diarrhea virus and transmissible gastroenteritis virus strains. Virology, 89, 3332-3342.
- MCCLURKIN, A.W. & NORMAN, J.O., 1966. Studies on transmissible gastroenteritis of swine. II. Selected characteristics of a cytopathogenic virus common to five isolates from transmissible gastroenteritis. Canadian Journal of Comparative Medicine, 30, 190-198.
- MORIN, M., TURGEON, D., JOLETTE, J. & ET AL., 1983. Neonatal diarrhea of pigs in Quebec: infectious causes of significant outbreaks. Canadian Journal of Comparative Medicine, 47, 11-17.
- MOXLEY, R.A. & OLSON, L.D., 1989. Clinical evaluation of transmissible gastroenteritis virus vaccines and vaccination procedures for inducing lactogenic immunity in sows. American Journal of Veterinary Research, 50, 111-118.
- PARK, S., SESTAK, K., HODGINS, D.C. & ET AL., 1998. Immune response of sows vaccinated with attenuated transmissible gastroenteritis virus (TGEV) and recombinant TGEV spike protein vaccines and protection of their suckling pigs against virulent TGEV challenge exposure. American Journal of Veterinary Research, 59, 1002-1008.
- PATON, D., IBATA, G., SANDS, J. & ET AL., 1997. Detection of transmissible gastroenteritis virus by RT-PCR and differentiation from porcine respiratory coronavirus. Journal of Virological Methods, 66, 303-309.
- PENSAERT, M., CALLEBAUT, P. & VERGOTE, J., 1986. Isolation of a porcine respiratory, non-enteric coronavirus related to transmissible gastroenteritis. Veterinary Quarterly, 8, 257-261.
- PENSAERT, M., COX, E., VAN DEUN, K. & ET AL., 1993. A sero-epizootiological study of porcine respiratory coronavirus in Belgian swine. Veterinary Quarterly, 15, 16-20.
- PENSAERT, M., HAELTERMAN, E.O. & BURNSTEIN, T.,1970. Transmissible gastroenteritis of swine: virus-intestinal cell interactions. I. Immunofluorescence, histopathology and virus production in the small intestine through the course of infection. Arch Gesamte Virusforsch, 31, 321-334.
- PHILLIP, J.I., CARTWRIGHT, S.F. & SCOTT, A.C., 1971. The size and morphology of T.G.E. and vomiting and wasting disease viruses of pigs. Veterinary Record, 88, 311-312.
- PILCHARD, E.I., 1965. Experimental transmission of transmissible gastroenteritis virus by starlings. American Journal of Veterinary Research, 26, 1177-1179.
- REYNOLDS, D.J., GARWES, D.J. & GASKELL, C.J., 1977. Detection of transmissible gastroenteritis virus neutralising antibody in cats. Archives of Virology, 55, 77-86.
- SAIF, L.J., PENSAERT, M.B., SESTAK, K. & ET AL., 2012. Coronaviruses. 10th edition. In: ZIMMERMAN. J,J., KARRIKER, L.A., RAMIREZ, A., SCHWARTZ, K.J., STEVENSON, G.W., eds. Disease of swine. New York, NY, John Wiley & Sons, Inc, 35, 501–524.
- SAIF, L.J., REDMAN, D.R., SMITH, K.L. & ET AL., 1983. Passive immunity to bovine rotavirus in newborn calves fed colostrum supplements from immunized or nonimmunized cows. Infection and Immunity, 41, 1118-1131.
- SAIF, L.J. & SESTAK, K., 2006. Transmissible gastroenteritis virus and porcine respiratory coronavirus. In: STRAW, B.L., ZIMMERMAN, J.J., D'ALLAIRE, S., TAYLOR, D.J., ed. Diseases of Swine. Ames, IA: Blackwell Publishing, 489-516.
- SANCHEZ, C.M., JIMENEZ, G., LAVIADA, M.D. & ET AL., 1990. Antigenic homology among coronaviruses related to transmissible gastroenteritis virus. Virology, 174, 410-417.
- SIRINARUMITR, T., PAUL, S.P., HALBUR, G. & ET AL., 1997. An overview of immunological and genetic methods for detecting swine coronaviruses, transmissible gastroenteritis virus, and porcine respiratory coronavirus in tissues. In: PAUL, P.S., FRANCIS, D.H., BENFIELD, D.A., eds. Mechanisms in the Pathogenesis of Enteric Diseases. New York, New York: Plenum Press, 37–45.
- VOETS, M.T., PENSAERT, M & RONDHUIS, P.R., 1980. Vaccination of pregnant sows against transmissible gastroenteritis with two attenuated virus strains and different inoculation routes. Veterinary Quaterly, 2, 211-219.
- WESLEY, R.D., WOODS, R.D., MCKEAN, J.D. & ET AL., 1997. Prevalence of coronavirus antibodies in Iowa swine. Canadian Journal of Veterinary Research, 61, 305-308.