- Infectious Diseases of Livestock
- Part 2
- Parainfluenza type 3 infection
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Parainfluenza type 3 infection
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Parainfluenza type 3 infection
M VAN VUUREN
Introduction
Parainfluenza type 3 (PI-3) virus commonly causes respiratory infection in cattle, sheep and goats with little or no clinical manifestation. However, in association with other viral and bacterial pathogens and stress-inducing situations, it causes severe pneumonia in cattle which is sometimes referred to as ‘shipping fever’ or enzootic pneumonia. It also infects humans. It is, however, uncertain whether or not PI-3 virus can be transmitted between humans and animals.
Four serotypes of parainfluenza virus have been described, but almost all infection and disease in livestock is caused by serotype 3 (PI-3). The virus was first described in the USA in 1959.23 It has a worldwide distribution in cattle, sheep and goats and is regularly isolated from clinical cases of respiratory disease in feedlot cattle. Together with other infectious agents, PI-3 virus constitutes part of the bovine respiratory disease complex28 (see Pneumonic pasteurellosis of cattle). In South Africa, serological studies have shown the virus to be widespread among feedlot cattle,3 and it has been isolated from cattle with clinical signs of respiratory disease.30
Apart from its association with disease in cattle and sheep,14 PI-3 virus may also be involved in mild respiratory disease of goats8 and it has been described in horses,10 although it is not recognized as being a common or important cause of respiratory disease in that species. The virus has been isolated from an aborted bovine foetus,25 and haemagglutination-inhibiting antibodies were found in 53 per cent of aborted foetuses in one study in the USA,11 but the significance of these findings is unclear as PI-3 is not a common or important cause of reproductive failure.2
Aetiology
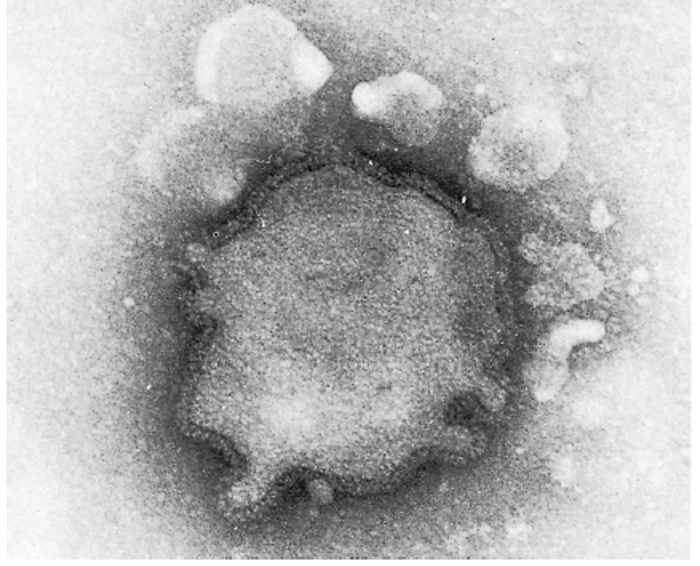
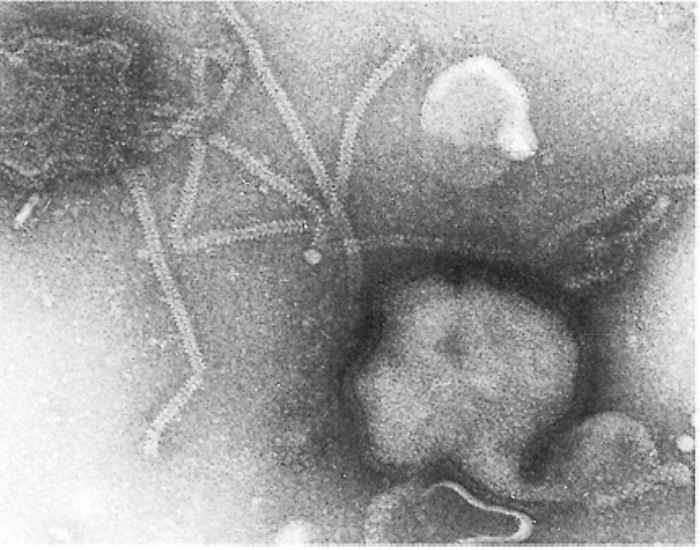
Parainfluenza viruses are classified in the genus Paramyxovirus of the family Paramyxoviridae.17 The virions are enveloped and measure between 120 and 300 nm for the round and pleomorphic forms (Figure 51.1), and up to 5 000 nm in length for filamentous forms. The single-stranded negativesense RNA genome is unsegmented. Nucleocapsids have helical symmetry and show characteristic herring-bone patterns (Figure 51.2).
The envelope contains lipids and glycosylated fusion (F) and haemagglutinin-neuraminidase (HN) proteins. These proteins are responsible for cellular fusion (syncytium formation), adsorption of the virus to cell surfaces and its subsequent release.
Human, bovine and ovine PI-3 viruses are antigenically related but are not identical.6 Differences in haemagglutinin, neuraminidase and syncytium-inducing (F protein) activities may be related to differences in pathogenicity of different isolates.4, 26
Epidemiology
Apart from livestock and humans, PI-3 virus has also been found in captive monkeys (Erythrocebus patas),7 water buffaloes (Bubalus bubalis)27 and African buffaloes (Syncerus caffer).16 There have also been reports of infection in deer, dogs, cats, guinea pigs and rats,12 indicating a wide host range. However, it is not clear whether or not these species constitute a source of infection for livestock.
The virus is shed in nasal and ocular secretions; in experimentally infected cattle, shedding of the virus may continue for eight to ten days after exposure.32 Susceptible cattle presumably become infected by means of aerosols that come into contact with the mucous membranes of the eyes, nostrils, and genital tract. Persistently infected (carrier) animals have not been reported.
The interrelationship between PI-3, other infectious agents and conditions which induce stress in the causation of the bovine respiratory complex is discussed elsewhere (see Pneumonic pasteurellosis of cattle).
Pathogenesis
Epithelial cells in the trachea, bronchi and alveoli are infected primarily. Necrosis of ciliated epithelium interferes with the mucociliary clearance of dust, fluid and cellular debris from the airways.
There is no evidence that PI-3 infection in cattle is immunosuppressive, but it does predispose lung tissue to bacterial invasion — mainly as a result of its adverse effect on alveolar macrophages,5, 9, 19, 20, 22 in which it is capable of replication,24, 29 and its influence on the interaction between macrophages and lymphocytes that is normally required for maximal lymphocyte responsiveness.15
Clinical signs and pathology
Most PI-3 infections in cattle are mild or clinically inapparent. Development of clinical disease is usually dependent upon interaction with other factors, both infectious and environmental (see Pneumonic pasteurellosis of cattle). Where PI-3 virus expresses its pathogenic potential in combination with Pasteurella and Mannheimia spp. infections, the clinical signs and lesions are typical of bovine pneumonic pasteurellosis.
After an incubation period of between 24 and 36 hours, a fever that may last six to ten days will develop in some calves. In uncomplicated cases, the fever is accompanied by lachrymation, serous to mucopurulent nasal discharge, depression and dyspnoea. Recovery is the general rule, but when secondary bacterial infection occurs, coughing, dyspnoea and depression may be severe and can be fatal, with death occurring within three to four days.
Gross lesions in fatal cases are those of bronchopneumonia, mainly in the ventral parts of the cranial and medial lobes, and consist of areas of congestion, atelectasis and consolidation. Evidence of mucopurulent inflammation of the upper respiratory tract is also usually present.
Microscopic changes include bronchitis, bronchiolitis, atelectasis, alveolitis, interstitial pneumonia, acidophilic intracytoplasmic and, more rarely, intranuclear inclusions in nasal, bronchiolar and bronchial epithelia, and the development of syncytia in affected parts of the lungs.13, 32
Clinical signs in sheep and goats are characterized by nasal and ocular discharges, coughing, rapid respiration, listlessness, anorexia and fever.8, 18
Gross lesions in sheep and goats are similar to the lesions seen in cattle, namely consolidation of cranio-ventral lung lobes. Microscopically the principal features are neutrophil and macrophage accumulation in the alveoli and bronchi. Inclusions are limited to the cytoplasm of some affected epithelial cells; only bovine strains of PI-3 have so far been shown to produce intranuclear inclusions.21
Diagnosis
The diagnosis of paramyxovirus infections is dependent on virus isolation, the detection of virus antigen in tissue specimens, or the demonstration of rising antibody levels.
In live animals the virus can be isolated from nasopharyngeal swabs or broncho-alveolar washings,andin dead animals, from the lungs and trachea and their associated lymph nodes. The virus can be grown in a variety of primary and secondary cell cultures, such as bovine and ovine foetal lung, kidney and testis, as well as in line cells, such as MDBK and RK-13. Cytopathic effects develop, and these are characterized by syncytia, intracytoplasmic and intranuclear inclusion bodies and cell destruction. These changes, however, do not always happen at the first passage, particularly when cell lines are employed. Virus antigen can be demonstrated in infected cell cultures by haemadsorption with bovine or guinea pig red blood cells. Specific identification of the antigen can be achieved by haemadsorption inhibition and immunofluorescence.
Serological diagnosis is based on haemagglutination inhibition and virus neutralization or on plaque reduction tests. As a result of the presence of serum antibodies in most cattle, paired serum samples (i.e. acute and convalescent) are essential for the demonstration of seroconversion.
Parainfluenza type 3 virus antigen can be detected in tissue specimens (e.g. cryo-preserved or paraffin-embedded lung tissue) by means of immunofluorescence and immunoperoxidase staining.31 However, these methods are dependent upon the availability of high quality antiserum that will bind to specific antigens in tissue sections.15
Control
The control of PI-3, because of its wide distribution in cattle populations, is dependent on immunization. However, because serious PI-3 disease is usually a complex condition involving interaction with other viruses, bacteria and environmental circumstances, the usual approach is to immunize cattle in those situations where respiratory disease commonly occurs (e.g. feedlots) with vaccines which also contain immunogens against other agents. Killed-virus and modified live-virus vaccines are available. Bivalent, trivalent and quadrivalent PI-3 vaccines are used in southern Africa in various combinations that include bovine herpesvirus type 1, bovine respiratory syncytial virus, bovine viral diarrhoea virus and Mannheimia (Pasteurella) haemolytica.
The use of vaccines against PI-3 virus for the control of respiratory tract disease in sheep became popular in North America during the early 1980s, but vaccination of small ruminants is not currently practised in southern Africa.
References
- ADAIR, B.M., BRADFORD, H.E.L., MACKIE, D.P. & MCNULTY, M.S., 1992. Effect of macrophages and in vitro infection with parainfluenza type 3 and respiratory syncytial viruses on the mitogenic response of bovine lymphocytes. American Journal of Veterinary Research, 53, 225–229.
- ANDERSON, M.L., BLANCHARD, P.C., BARR, B.C. & HOFFMAN, R.L., 1990. A survey of causes of bovine abortion occurring in the San Joaquin Valley, California. Journal of Veterinary Diagnostic Investigation, 2, 283–287.
- BARNARD, B.J.H., 1977. Virus diseases in feedlot cattle in South Africa. Bulletin de L’Office International des Epizooties, 88, 153–160.
- BERGMAN, R., MORENO-LOPEZ, J., MÖLLERBERG, L. & MOREIN, B., 1978. Parainfluenza-3 virus: Difference in capacity of neuraminidase-weak and strong strains to infect young calves and to elicit cellular immune response. Research in Veterinary Science, 25, 193–199.
- BROWN, T.T. & ANANABA, G., 1988. Effect of respiratory infections caused by bovine herpesvirus-1 or parainfluenza-3 virus on bovine alveolar macrophage functions. American Journal of Veterinary Research, 49, 1447–1451.
- BUXTON, A. & FRASER, G., 1977. Animal Microbiology. London, Oxford, Edinburgh, Melbourne: Blackwell Scientific Publications.
- CHURCHILL, A.E., 1963. The isolation of parainfluenza 3 virus from fatal cases of pneumonia in Erythrocebus patas monkeys. British Journal of Experimental Pathology, 45, 529–537.
- DAFT, B.M. & WEIDENBACH, S., 1987. Isolation of parainfluenza type 3 virus associated with a respiratory epizootic in goats in California. Agri-Practice, January 1987, 35–36.
- DAVIES, D.H., LONG, D.L., MCCARTHY, A.R. & HERCEG, M., 1986. The effect of parainfluenza virus type 3 on the phagocytic cell response of the ovine lung to Pasteurella haemolytica. Veterinary Microbiology, 11, 125–144.
- DITCHFIELD, J., ZBITNEW, A. & MACPHERSON, L.W., 1963. Association of Myxovirus parainfluenza 3 (RE 55) with upper respiratory infection of horses. Canadian Veterinary Journal, 4, 175
- DUNNE, H.W., AJINKA, F.M., BUBASH, G.R. & GRIEL, L.C., 1973. Parainfluenza- 3 and bovine enteroviruses as possible important causative factors in bovine abortion. American Journal of Veterinary Research, 34, 1121–1126.
- FENNER, F., BACHMANN, P.A., GIBBS, E.P.J., MURPHY, F.A., STUDDERT, M.J. & WHITE, P.O., 1987. Veterinary Virology. Orlando, San Diego, New York: Academic Press, Inc.
- FRANK, G.H. & MARSHALL, R.G., 1973. Parainfluenza-3 virus infection of cattle. Journal of the American Veterinary Medical Association, 163, 858–860.
- GOYAL, S.M., KHAN, M.A., MCPHERSON, S.W., ROBINSON, R.A. & BOYLAN, W.J., 1988. Prevalence of antibodies to seven viruses in a flock of ewes in Minnesota. American Journal of Veterinary Research, 49, 464–467.
- HAINES, D.M., KENDALL, J.C., REMENDA, B.W., BREKER-KLASEN, M.M. & CLARK,E.G., 1992. Monoclonal and polyclonal antibodies for immunohistochemical detection of bovine parainfluenza type 3 virus in frozen and formalin-fixed paraffin-embedded tissues. Journal of Veterinary Diagnostic Investigation, 4, 393–399.
- HAMBLIN, C., HEDGER, R.S. & CONDY, J.B., 1980. The isolation of parainfluenza 3 virus from free-living African buffalo (Syncerus caffer). The Veterinary Record, 107, 18.
- KINGSBURY, D.W., BRATT, M.A., CHOPPIN, P.W., HANSON, R.P., HOSAKA, Y., MEULEN, V., NORRBY, E., PLOWRIGHT, W., ROTT, R. & WUNNER, W.H., 1978. Paramyxoviridae. Intervirology, 10, 137–152.
- LEHMKUHL, H.D. & CUTLIP, R.C., 1982. Characterization of parainfluenza type 3 virus isolated from the lung of a lamb with pneumonia. American Journal of Veterinary Research, 43, 626–628.
- LIGGIT, D., HUSTON, L., SILFLOW, R., EVERMANN, J. & TRIGO, E., 1985. Impaired function of bovine alveolar macrophages infected with parainfluenza-3 virus. American Journal of Veterinary Research, 46, 1740–1744.
- LOPEZ, A., THOMSON, R.G. & SAVAN, M., 1976. The pulmonary clearance of Pasteurella haemolytica in calves infected with bovine parainfluenza-3 virus. Canadian Journal of Comparative Medicine, 40, 385–391.
- MCLEAN, A.M. & DOANE, F.W., 1971. The morphogenesis and cytopathology of bovine parainfluenza type 3 virus. Journal of General Virology, 12, 271–279.
- PERINO, L.J., 1989. Immunosuppressive effects of bovine viral pathogens: A review. The Bovine Practitioner, 24, 42–48.
- REISINGER, R.C., HEDDLESTON, K.I. & MANTEI, C.A., 1959. A myxovirus (SF-4) associated with shipping fever of cattle. Journal of the American Veterinary Medical Association, 135, 147–152.
- ROSSI, C.R. & KIESEL, G.K., 1977. Susceptibility of bovine macrophage and tracheal-ring cultures to bovine viruses. American Journal of Veterinary Research, 38, 1705–1708.
- SATTAR, S.A., BOHL, E., TRAPP, H.C. & HAMDY, A.H., 1967. Infection of bovine fetuses with myxovirus parainfluenza-3 virus. American Journal of Veterinary Research, 28, 45–49.
- SHIBUTA, H., SUZU, S. & SHIODA, T., 1986. Differences in bovine parainfluenza 3 virus variants studied by monoclonal antibodies against viral glycoproteins. Virology, 155, 688–696.
- SINGH, K.V. & BAZ, T.I., 1966. Isolation of parainfluenza-3 virus from water buffalo in Egypt. Nature, 210, 656–657.
- STOTT, E.J., THOMAS, L.H., COLLINS, A.P., CROUCH, S., JEBBETT, J., SMITH, G.S., LUTHER, P.D. & CASWELL, R., 1980. A survey of virus infections of the respiratory tract of cattle and their association with disease. Journal of Hygiene, 85, 257–270.
- TOTH, T. & HESSE, R.A., 1983. Replication of five bovine respiratory viruses in cultured bovine alveolar macrophages. Archives of Virology, 75, 219–224.
- VAN VUUREN, M., 1991. Unpublished results. Faculty of Veterinary Science, Medical University of South Africa, Medunsa, South Africa.
- watt, n.j., pirie, h.m., allan, e.m., selman, i.e., petrie, l., gibbs, h.a. & wiseman, a., 1982. Parainfluenza type III virus and respiratory syncytial virus antigens in paraffin-embedded sections of pneumonic lung. Proceedings of the Twelfth World Congress on Diseases of Cattle, Amsterdam, 1982.
- WOODS, G.T., 1968. The natural history of bovine myxovirus parainfluenza-3. Journal of the American Veterinary Medical Association, 152, 771–777.