- Infectious Diseases of Livestock
- Part 2
- Papillomavirus infection of ruminants
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Papillomavirus infection of ruminants
This content is distributed under the following licence: Attribution-NoDerivs CC BY-ND  View Creative Commons Licence details here
View Creative Commons Licence details here
Papillomavirus infection of ruminants
Current authors:
J S MUNDAY - Professor of Veterinary Pathology, BVSc, PhD, Dipl ACVP, School of Veterinary Science, Massey University, Tennent Drive, Palmerston North, Manawatu, 4410, New Zealand
S ROPERTO - Department of Veterinary Medicine and Animal Production, Naples University “Federico II”, Naples, Italy
Papillomavirus infections in bovines
There are currently 23 bovine papillomavirus (BPV) types that have been fully sequenced.3 These BPVs are classified within five genera; Deltapapillomavirus, Xipapillomavirus, Epsilonpapillomavirus, Dyoxipapillomavirus, and Dyokappapapillomavirus.15 Of these, the BPVs that have been most firmly established as a cause of disease in bovines are the DeltaPVs (especially BPV-1 and BPV-2) and the XiPVs (especially BPV-4).
The DeltaPVs demonstrate some important differences to papillomaviruses in other genera. First is their ability to infect, and cause disease in, other host species. This wider host range allows these viruses to cause disease not only in bovines including cattle, water buffalo, and yak, but the DeltaPVs also cause equine sarcoids (BPV-1, -2, and -13)14 and feline sarcoids (BPV-14).36 A second unique property of the DeltaPVs is their ability to infect and cause proliferation of both epithelial and mesenchymal cells.25 It is probable that the unique ability to influence mesenchymal cell growth is due to the proteins produced by the DeltaPVs to influence cell growth. Unlike other papillomavirus types that promote epithelial growth and differentiation primarily through E7 protein-mediated inhibition of the retinoblastoma protein (pRb),32 the predominant oncoprotein of the DeltaPVs is the E5 protein.64 This protein influences cell growth by activating the platelet derived growth factor-β receptor, phosphoinositide 3-kinase, tyrosine kinase c-Src,6 and the subunit D of the V1-ATPase proton pump.50 In addition, the DeltaPV E5 protein promotes overexpression of the transcription factor E2F3 by downregulating pRb via calpain 3.48
Transmission of papillomaviruses between animals is thought to most often occur from either direct contact or indirectly via environmental contamination. However, bovine DeltaPVs can also be found in semen of infected animals,29 suggesting that sexual transmission of these papillomaviruses could also be possible. Additionally, productive infections of DeltaPVs have been demonstrated within the trophoblasts of the placenta in cattle and water buffaloes46 and there is accumulating evidence that vertical transmission of papillomaviruses may be an important method of spread of DeltaPVs within ruminant species.
Cutaneous papillomas (warts)
Cutaneous papillomas are the most frequent clinical manifestation of papillomavirus infection in bovines and papillomas have been associated with numerous different papillomavirus types.44 It was initially suggested that each bovine papilloma subtype was caused by a specific BPV type. However, more recent studies have demonstrated that most papillomas contain numerous BPV types,55 making it difficult to determine which BPVs cause papillomas and which are present simply as a coincidental infection.
Cutaneous papillomas commonly affect cattle throughout the world and warts have been detected on up to a third of cattle at slaughter.28 Bovine papillomaviruses have also been reported to cause cutaneous papillomas in water buffalo and yak.2, 56
Papillomas typically develop on younger animals. While the epidemiology of infection remains poorly understood, this is likely due to BPV infection of an immunologically naïve animal. The lack of protective antibodies allows rapid replication by the BPV, marked epithelial hyperplasia, and the development of a visible papilloma.33 After papilloma development, the body develops protective antibodies against the BPV type and an animal will be protected against further infections by this BPV type.17, 26 The development of a cell-mediated immune response results in lesion resolution.16 As with all papillomaviruses, infection is probably due to a combination of exposure to the BPV and the presence of skin microtrauma allowing the BPV access to the basal cell layer of the epidermis.54 Bovine papillomaviruses can spread by direct contact and housed cattle develop papillomas more frequently than cattle on pasture. Indirect spread from the environment or from farming equipment is common. Genital papillomas are spread by venereal transmission.35
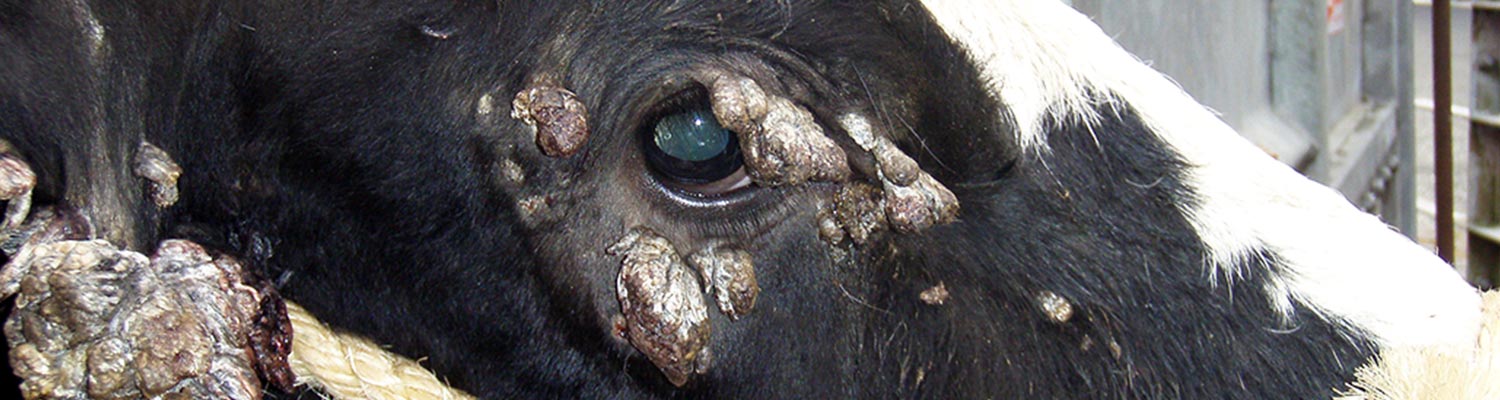
Papillomas can develop anywhere on the body but appear to be most frequent around the head (Figure 1 and Figure 2). They also commonly develop on the penis and vulva and on the teats. Most animals will have multiple papillomas. An accumulation of keratin over the surface of the papillomas can result in a grey colour and a hard roughened surface. Ulceration is rarely observed, the lesions are not painful, and generally appear to be of little concern to the animal. Rarely teat papillomas can interfere with normal milking practices and predispose to mastitis while large genital papillomas can prevent mating.
Papillomavirus-induced cutaneous papillomas in bovines can be subdivided into squamous papillomas and fibropapillomas. A squamous papilloma develops as the marked epidermal hyperplasia that is caused by the papillomavirus infection results in folding of the epidermis. The folded epidermis forms an exophytic mass that is comprised of markedly thickened epidermis supported by a narrow fibrous core. Rarely, inverted papillomas that are characterized by endophytic growth are observed. Squamous papillomas generally appear as exophytic masses that range from pedunculated, narrow-based filiform masses to larger broader-based vegetative growths.
In addition to epidermal hyperplasia, fibropapillomas also contain a proliferation of mesenchymal cells in the underlying dermis. This typically results in a more broad-based sessile clinical appearance. Histologically, thickening of the epidermis is present with the presence of narrow rete pegs of epidermis extending into the underlying dermis. Fibroblasts within the thickened dermis are well-differentiated and do not demonstrate atypia. Papillomavirus-induced cell changes are usually present in the hyperplastic epidermis of squamous papillomas and fibropapillomas. However, as viral replication does not occur in the proliferating fibroblasts of a fibropapilloma, these cells do not show any histological evidence of papillomavirus infection.25 Current evidence suggests that fibropapillomas are only caused by infection with the deltaPV types.25 Whether or not the deltaPV types can also cause squamous papillomas is currently unclear.
Most papillomas are diagnosed by clinical examination. However, if papillomas develop in an unusual location or in an older animal, surgical excision and histology may be required for definitive diagnosis. Papillomas may be mistaken for dermatophilosis. However, careful evaluation of a dermatophilosis lesion will reveal the presence of a thick multilaminar crust, a feature that is not present papillomas.
Almost all cutaneous papillomas in cattle spontaneously resolve due to the development of a cell-mediated immune response. As there appears to be significant inter-individual variation in the time taken to develop such an immune response, it is not surprising that the time taken for lesion resolution will also be variable. While there are few studies regarding the clinical course of bovine papillomas, evidence from other species suggests that papillomas may take up to 12 months to completely resolve.38 There are rare reports of cattle that develop large persistent papillomas.33
These can cause significant morbidity by interfering with vision, preventing the animal from eating, or predisposing to secondary bacterial infection and fly-strike. Cattle with persistent florid papillomas are suspected to have an underlying immunodeficiency, although these cattle will often show no other clinical evidence of immune dysfunction. While little information is available, it is likely that cutaneous papillomas in water buffalo and yak also spontaneously regress and are not associated with significant morbidity.2, 56
Multiple papillomas that are predisposed to progress to squamous cell carcinomas (SCCs) have been reported to develop on the vulva of cattle exposed to high levels of ultraviolet light.57 However, whether these papillomas are papillomavirus-induced or a manifestation of actinic keratosis is unknown and there is currently little evidence that papillomavirus-induced cutaneous papillomas in cattle are predisposed to neoplastic transformation.
Numerous treatments aimed at hastening papilloma regression have been proposed. As papillomas self-resolve after a variable length of time, the efficacy of such treatments is difficult to assess in an individual animal. Both crushing of papillomas and injecting inactivated extracts of removed papillomas (autologous vaccination) have historically been popular treatments. However, there is no evidence that either technique is effective. Injection of viral proteins may have accelerated lesion resolution in three experimentally-infected calves.24 Other proposed treatments include topical fig tree latex and salicylic acid,22 intralesional injection of suspensions of Propionibacterium acnes,20 systemic levamisole, systemic administration of an extract of tarantula venom,8 and systemic ivermectin.4 While each treatment showed some promise, there are no large double-blinded studies to confirm the results of these preliminary studies.
While vaccination has been shown to effectively prevent papilloma development,10 such vaccines are not commercially available.
Upper alimentary tract papillomas and neoplasia
Upper alimentary tract papillomas in cattle develop from the caudal aspect of the oral cavity to the rumen. They are subdivided into squamous papillomas and fibropapillomas. Squamous papillomas are caused by BPV-4 while upper alimentary tract fibropapillomas are caused by BPV-2.5, 9
While there is little information available for many geographical locations, these papillomas probably have a worldwide distribution.
Upper alimentary tract papillomas are most common in younger cattle and have been reported in 5 to 20 per cent of cattle.33 Upper alimentary tract papillomas due to BPV infection have also been reported in water buffalo, although it is currently unknown how frequently water buffalo develop these lesions.27
Most upper alimentary tract squamous papillomas of cattle are incidental findings that are discovered at slaughter (Figure 3). However, large numbers of large papillomas can develop in immunosuppressed cattle and can cause dysphagia, coughing, ruminal tympany, and weight loss.61 While infection with bovine viral diarrhea could potentially predispose to extensive papilloma development, ingestion of bracken fern (Pteridium species) appears to be the factor that most frequently results in the development of florid papillomas.23

Figure 1 A calf suffering from severe papillomatosis. Note the numerous confluent warts on its neck.
Squamous papillomas can appear pedunculated or sessile and are often covered by numerous thin projections. Histologically the papillomas appear as foci of marked epidermal thickening supported by a thin fibrovascular core. Infections are typically productive and virally-induced cell changes can be prominent within the hyperplastic epithelium.
In immunocompetent cattle, squamous papillomas remain small and spontaneously resolve, typically within a year. However, in cattle that ingest bracken fern, papillomas can undergo malignant transformation to a SCC.42 The role of the papillomavirus in promoting malignant transformation is uncertain. However, unlike in papillomavirus-induced cancers that develop in other species, papillomavirus DNA is not detectible in the SCCs.13 Additionally, while upper alimentary SCCs are common in cattle that graze bracken fern,30 they are extremely rare in cattle that do not ingest bracken fern, suggesting a primary role of bracken fern in cancer development. Bracken fern contains ptaquiloside which has been shown to be carcinogenic.43 However, as bracken fern is also immunosuppressive ingestion of this plant may also result in papilloma persistence53 and many cattle with upper alimentary cancer also have numerous squamous papillomas (Figure 4).
Fibropapillomas of the upper alimentary tract are typically asymptomatic although the development of multiple large fibropapillomas in the ruminal grove can prevent normal eructation and regurgitation.19 Fibropapillomas appear as vegetative masses or as plaque-like thickenings of the mucosa (Figure 5). Histologically, fibropapillomas consist of a proliferation of fibrous tissue covered by hyperplastic epithelium. These lesions do not support viral replication and virally-induced cell changes are not visible.34 Fibropapillomas spontaneously resolve. Interestingly, there is no evidence that the immunosuppressive properties of bracken fern significantly impair the resolution of these lesions.
As with cutaneous papillomas, treatment options for upper alimentary papillomas are limited. While vaccination using BPV-4 proteins was initially reported to have accelerated papilloma regression,11 no statistical analyses were performed and subsequent vaccination using BPV-4 virus-like particles did not significantly influence regression.26 In contrast, vaccination against BPV-4 has been shown to be an effective method of preventing papilloma formation.26 However, it remains uncertain what proportion of upper alimentary SCCs would be prevented by such a vaccine and no vaccine against BPV-4 is currently commercially available. Due to the prominent role of bracken fern in causing cancers it appears likely that preventing cattle from ingesting this plant will greatly reduce the incidence and severity of disease associated with PVs.

Figure 4 Squamous cell carcinomas of the upper alimentary tract typically appear as ulcerated masses (arrowhead). These cancers are associated with bracken fern ingestion. As well as containing carcinogens, bracken fern is also immunosuppressive and animals with upper alimentary squamous cell carcinomas often also have numerous squamous papillomas (arrows). Photograph courtesy of Dr G.D. Kommers, Universidade Federal de Santa Maria, Brazil.
Bladder neoplasia
Bladder neoplasia in cattle and water buffalo has been associated with the DeltaPVs BPV-2, -13, and -14.49, 51 Evidence linking papillomaviruses to bladder neoplasia in cattle includes the observation that injecting wart extracts into the bladder results in the formation of resolving tumors,39 the higher rate of BPV-2 and -13 infection in bladder neoplasms than in non-neoplastic samples of bladder,12, 51 and the detection of papillomavirus protein expression in bovine bladder neoplasms.7 However, as papillomavirus DNA can be detected in up to 50 per cent of non-neoplastic samples of the bovine bladder, it is difficult to differentiate between the papillomavirus being causative and the papillomavirus being more frequently present due to a more permissive environment provided by the bladder neoplasms. Furthermore, the development of bladder neoplasia is highly dependent on exposure to bracken fern and bladder neoplasms are rare in cattle that have no exposure to this plant. Bovine bladder tumors are more commonly urothelial than mesenchymal and can be classified using the same microscopic features that are used for human bladder neoplasms (Figure 6).45
Interestingly DeltaPV infections of the bladder of cattle are productive.52 How bladder infection occurs is currently unknown. However, as BPV-2 and BPV-13 DNA is detectible in the blood, haematogenous infection of the bladder mucosa could be possible.47
Animals that ingest bracken fern develop haematuria resulting in the clinical syndrome known as ‘chronic enzootic haematuria’. Enzootic haematuria is a non-specific term that includes haematuria caused by cystitis as well as haematuria caused by the development of bladder neoplasia. Due to the role of bracken fern in the disease development, it is not unusual for animals with bladder neoplasms to also have upper alimentary tract papillomas due to the immunosuppression caused by this plant.
Vaccination against BPV-2 has been shown to effectively prevent cutaneous fibropapilloma development. In contrast, a crude vaccine preparation did not decrease bladder neoplasia.40 However, some of the injected BPV-2 may have been viable and there are no definitive studies investigating whether a BPV-2 vaccine can prevent bladder cancer. Administration of a BPV-2 vaccine to cattle with bladder tumors did not result in any reduction in tumor size.41 Avoidance of bracken fern is expected to reduce the development of bladder cancers of cattle and water buffalo.
Papillomavirus infections in sheep
There are currently four Ovis aries papillomavirus (OaPV) types that have been fully sequenced. These include three DeltaPVs (OaPV-1, -2, -4) and OaPV-3 that has a proposed classification as a DyokappaPV.1, 60 The clinical diseases that are associated with papillomaviruses in sheep can be broadly subdivided into hyperplastic cutaneous warts and cutaneous SCCs. While the development of numerous ruminal papillomas has been reported in sheep, these do not appear to be caused by papillomavirus infection.37
Cutaneous papillomas
Sheep have been long recognised to develop cutaneous papillomas and an association with PV infection was first reported in 1975.18 Sheep have been reported to develop both fibropapillomas and filiform squamous papillomas.18, 21 Whether the two different types of papillomas are caused by the same or different papillomavirus types is unknown although all three ovine DeltaPVs were detected in fibropapillomas.60
Papillomas are most common on the feet, around the muzzle, and on the mammary gland (Figure 7). This may be because these areas are most likely to be become traumatized, enabling infection of the basal cells by the papillomaviruses. While little research has been performed on cutaneous papillomas in sheep, these lesions appear to undergo spontaneous resolution and there are no reports of persistent or florid papillomatosis in sheep.
Cutaneous squamous cell carcinomas
Cutaneous SCC in sheep occurs in areas of the body that are exposed to higher amounts of sunlight such as the perineum, eyelid, and pinna. In Australia, due to management practices to reduce fly- strike, the vulva, tail, and perineum can be exposed to large amounts of solar radiation and up to 7 per cent of sheep have been reported to develop SCCs in these areas.59 The potential for papillomavirus infection to act as a co-factor with solar radiation in cancer development was first proposed in 1982 when it was observed that some SCCs developed as a progression from a papilloma and that some SCCs contained papillomavirus particles.63 The presence of papillomavirus DNA in a high proportion of ovine cutaneous SCCs was subsequently reported in 1992.59 Recently, OaPV-3 was detected in a series of ovine cutaneous SCCs with additional testing confirming the presence of papillomavirus gene expression within the cancers.1, 65 Furthermore, OaPV-3 DNA could be detected in 65 per cent of SCCs the developed on sheep from Sardinia, but only in 30 per cent of non-neoplastic samples of skin from sheep in the same geographical location.65
Current evidence suggests that OaPV-3 could be a significant co-factor in the development of cutaneous SCCs in sheep. However, the frequent asymptomatic infection of sheep by OaPV-3 makes it impossible to differentiate between OaPV-3 causing the SCCs and OaPV-3 being present within the SCCs because these cancers provide a more permissive environment. If OaPV-3 does promote cancer development, it is also unknown how significant this effect is. As SCCs rarely develop in sheep in the absence of high levels of solar radiation, it appears likely that solar radiation could be the primary cause of the SCCs. Determining the precise role of OaPV-3 is important to predict the likely success of a vaccine against OaPV-3 in preventing cutaneous SCC development in sheep.
Papillomavirus infections in goats
Goats have been reported to develop papillomavirus-induced fibropapillomas on the mammary glands (Figure 8).31 While papillomas have also been described on other areas of skin and the genitals of goats, it is uncertain whether or not these papillomas are also associated with papillomavirus infection.58 The papillomas that develop on the mammary glands of goats spontaneously resolve and do not appear to cause significant clinical disease.58 Currently one papillomavirus has been fully sequenced from goats and this virus was classified as a Phipapillomavirus.62 As this papillomavirus was detected in clinically-normal skin, it is uncertain whether or not it is the cause of mammary fibropapillomas in goats.
References
- ALBERTI, A., PIRINO, S., PINTORE, F., ADDIS, M.F., CHESSA, B., CACCIOTTO, C., CUBEDDU, T., ANFOSSI, A., BENENATI, G., CORADDUZZA, E., LECIS, R., ANTUOFERMO, E., CARCANGIU, L. & PITTAU, M., 2010. Ovis aries Papillomavirus 3: A prototype of a novel genus in the family Papillomaviridae associated with ovine squamous cell carcinoma. Virology, 407, 352-359.
- BAM, J., KUMAR, P., LEISHANGTHEM, G. D., SAIKIA, A. & SOMVANSHI, R., 2013. Spontaneous cutaneous papillomatosis in yaks and detection and quantification of bovine papillomavirus-1 and -2. Transboundary and Emerging Diseases, 60, 475-480.
- BAUERMANN, F.V., JOSHI, L.R., MOHR, K.A., KUTISH, G.F., MEIER, P., CHASE, C., CHRISTOPHER-HENNINGS, J. & DIEL, D.G., 2017. A novel bovine papillomavirus type in the genus Dyokappapapillomavirus. Archives of Virology, 162, 3225-3228.
- BORKU, M.K., ATALAY, O., KIBAR, M., CAM, Y. & ATASEVER, A., 2007. Ivermectin is an effective treatment for bovine cutaneous papillomatosis. Research in Veterinary Science, 83, 360-363.
- BORZACCHIELLO, G., AMBROSIO, V., ROPERTO, S., POGGIALI, F., TSIRIMONAKIS, E., VENUTI, A., CAMPO, M.S. & ROPERTO, F., 2003. Bovine papillomavirus type 4 in oesophageal papillomas of cattle from the south of Italy. Journal of Comparative Pathology, 128, 203-206.
- BORZACCHIELLO, G., MOGAVERO, S., DE VITA, G., ROPERTO, S., DELLA SALDA, L. & ROPERTO, F., 2009. Activated platelet derived growth factor beta receptor expression, PI3K-AKT pathway molecular analysis and transforming signals in equine sarcoids. Veterinary Pathology, 46, 589-597.
- BORZACCHIELLO, G., RESENDES, A.R., ROPERTO, S. & ROPERTO, F., 2009. Co-expression of bovine papillomavirus E5 and E7 oncoproteins in naturally occurring carcinomas of the urinary bladder in cattle. Journal of Comparative Pathology, 141, 84-88.
- ÇAM, Y., KIBAR, M., ATASEVER, A., ATALAY, Ö. & BEYAZ, L., 2007. Efficacy of levamisole and Tarantula cubensis venom for the treatment of bovine cutaneous papillomatosis. Veterinary Record, 160, 486-488.
- CAMPO, M.S., 1995. Infection by bovine papillomavirus and prospects for vaccination. Trends in Microbiology, 3, 92-97.
- CAMPO, M.S., 1997. Vaccination against papillomavirus in cattle. Clinics in Dermatology, 15, 275-283.
- CAMPO, M.S., GRINDLAY, G.J., O'NEIL, B.W., CHANDRACHUD, L.M., MCGARVIE, G.M. & JARRETT, W.F., 1993. Prophylactic and therapeutic vaccination against a mucosal papillomavirus. The Journal of General Virology, 74, 945-953.
- CAMPO, M.S., JARRETT, W.F., BARRON, R., O'NEIL, B.W. & SMITH, K.T., 1992. Association of bovine papillomavirus type 2 and bracken fern with bladder cancer in cattle. Cancer Research, 52, 6898-6904.
- CAMPO, M.S., MOAR, M.H., SARTIRANA, M.L., KENNEDY, I.M. & JARRETT, W.F., 1985. The presence of bovine papillomavirus type 4 DNA is not required for the progression to, or the maintenance of, the malignant state in cancers of the alimentary canal in cattle. EMBO J, 4, 1819-1825.
- CHAMBERS, G., ELLSMORE, V.A., O'BRIEN, P.M., REID, S.W., LOVE, S., CAMPO, M.S. & NASIR, L., 2003. Association of bovine papillomavirus with the equine sarcoid. Journal of General Virology, 84, 1055-1062.
- DAUDT, C., DA SILVA, F.R.C., STRECK, A.F., WEBER, M.N., MAYER, F. Q., CIBULSKI, S.P. & CANAL, C.W., 2016. How many papillomavirus species can go undetected in papilloma lesions? Scientific Reports, 6, 36480.
- EGAWA, N. & DOORBAR, J., 2017. The low-risk papillomaviruses. Virus Research, 231, 119-127.
- GHIM, S., NEWSOME, J., BELL, J., SUNDBERG, J.P., SCHLEGEL, R. & JENSON, A.B. 2000., Spontaneously regressing oral papillomas induce systemic antibodies that neutralize canine oral papillomavirus. Experimental and Molecular Pathology, 68, 147-151.
- GIBBS, E.P., SMALE, C.J. & LAWMAN, M.J., 1975. Warts in sheep. Identification of a papilloma virus and transmission of infection to sheep. Journal of Comparative Pathology, 85, 327-334.
- GORDON, P.J., 1997. Surgical removal of a fibropapilloma from the reticulum causing apparent vagal indigestion. Veterinary Record, 140, 69-70.
- HALL, H., TEUSCHER, C., URIE, P., BODEN, B. & ROBISON, R., 1994. Induced regression of bovine papillomas by intralesional immunotherapy. Therapeutic Immunology, 1, 319-324.
- HAYWARD, M., BAIRD, P. & MEISCHKE, H., 1993. Filiform viral squamous papillomas on sheep. Veterinary Record, 132, 86-88.
- HEMMATZADEH, F., FATEMI, A. & AMINI, F., 2003. Therapeutic effects of fig tree latex on bovine papillomatosis. Journal of Veterinary Medicine, Series B, 50, 473-476.
- JARRETT, W.F., MURPHY, J., O'NEIL, B.W. & LAIRD, H.M., 1978. Virus-induced papillomas of the alimentary tract of cattle. International Journal of Cancer, 22, 323-328.
- JARRETT, W.F., SMITH, K.T., O'NEIL, B.W., GAUKROGER, J.M., CHANDRACHUD, L.M., GRINDLAY, G.J., MCGARVIE, G.M. & CAMPO, M.S., 1991. Studies on vaccination against papillomaviruses: prophylactic and therapeutic vaccination with recombinant structural proteins. Virology, 184, 33-42.
- JELINEK, F. & TACHEZY, R., 2005. Cutaneous papillomatosis in cattle. Journal of Comparative Pathology, 132, 70-81.
- KIRNBAUER, R., CHANDRACHUD, L.M., O'NEIL, B.W., WAGNER, E.R., GRINDLAY, G.J., ARMSTRONG, A., MCGARVIE, G.M., SCHILLER, J.T., LOWY, D.R. & CAMPO, M.S., 1996. Virus-like particles of bovine papillomavirus type 4 in prophylactic and therapeutic immunization. Virology, 219, 37-44.
- KUMAR, P., NAGARAJAN, N., SAIKUMAR, G., ARYA, R.S. & SOMVANSHI, R., 2015. Detection of bovine papilloma viruses in wart-like lesions of upper gastrointestinal tract of cattle and buffaloes. Transboundary and Emerging Diseases, 62, 264-271.
- LINDHOLM, I., MURPHY, J., O'NEIL, B.W., CAMPO, M.S. & JARRETT, W.F., 1984. Papillomas of the teats and udder of cattle and their causal viruses. The Veterinary Record, 115, 574-577.
- LINDSEY, C.L., ALMEIDA, M.E., VICARI, C.F., CARVALHO, C., YAGUIU, A., FREITAS, A.C., BECAK, W. & STOCCO, R.C., 2009. Bovine papillomavirus DNA in milk, blood, urine, semen, and spermatozoa of bovine papillomavirus-infected animals. Genetics and Molecular Research, 8, 310-318.
- LUCENA, R.B., RISSI, D.R., KOMMERS, G.D., PIEREZAN, F., OLIVEIRA-FILHO, J.C., MACEDO, J.T., FLORES, M.M. & BARROS, C.S., 2011. A retrospective study of 586 tumours in Brazilian cattle. Journal of Compative Pathology, 145, 20-24.
- MANNI, V., ROPERTO, F., DI GUARDO, G., GALATI, D., CONDOLEO, R.U. & VENUTI, A., 1998. Presence of papillomavirus-like DNA sequences in cutaneous fibropapillomas of the goat udder. Veterinary Microbiology, 61, 1-6.
- MCLAUGHLIN-DRUBIN, M.E. & MUNGER, K., 2009. The human papillomavirus E7 oncoprotein. Virology, 384, 335-344.
- MUNDAY, J.S., 2014. Bovine and human papillomaviruses: a comparative review. Veterinary Pathology, 51, 1063-1075.
- MUNDAY, J.S., LOHR, C.V. & KIUPEL, M., 2017. Tumors of the Alimentary Tract. In: MEUTON, D. J. (ed.) Tumors in Domestic Animals. 5th ed.: John Wiley and Sons, Inc.
- MUNDAY, J.S. & PASAVENTO, P., 2017. Papillomaviridae and Polyomaviridae. In: MACLACHLAN, N. J. & DUBOVI, E. J. (eds.) Fenner's Veterinary Virology. 5th ed. London, United Kingdom: Academic Press.
- MUNDAY, J.S., THOMSON, N., DUNOWSKA, M., KNIGHT, C.G., LAURIE, R.E. & HILLS, S., 2015. Genomic characterisation of the feline sarcoid-associated papillomavirus and proposed classification as Bos taurus papillomavirus type 14. Veterinary Microbiology, 177, 289-295.
- NORVAL, M., MICHIE, J.R., APPS, M.V., HEAD, K.W. & ELSE, R.E., 1985. Rumen papillomas in sheep. Veterinary Microbiology, 10, 219-229.
- OLSON, C., OLSON, R.O. & HUBBARD-VAN STELLE, S., 1992. Variations of response of cattle to experimentally induced viral papillomatosis. Journal of the American Veterinary Medical Association, 201, 56-62.
- OLSON, C., PAMUKCU, A.M., BROBST, D.F., KOWALCZYK, T., SATTER, E.J. & PRICE, J.M., 1959. A urinary bladder tumor induced by a bovine cutaneous papilloma agent. Cancer Research, 19, 779-782.
- PAMUKCU, A.M., OLSON, C. & GOKSOY, S.K., 1967. Influence of a papilloma vaccine on chronic bovine enzootic hematuria. Cancer Research, 27, 2197-2200.
- PATHANIA, S., KUMAR, P., DEVI, L.G., KUMAR, D., DHAMA, K. & SOMVANSHI, R., 2011. Preliminary assessment of binary ethylenimine inactivated and saponized cutaneous warts (BPV-2) therapeutic vaccine for enzootic bovine haematuria in hill cows. Vaccine, 29, 7296-7302.
- PLOWRIGHT, W., 1955. Malignant neoplasia of the oesophagus and rumen of cattle in Kenya. Journal of Comparative Pathology, 65, 108-114.
- POTTER, D.M. & BAIRD, M.S., 2000. Carcinogenic effects of ptaquiloside in bracken fern and related compounds. British Journal of Cancer, 83, 914-920.
- RECTOR, A. & VAN RANST, M., 2013. Animal papillomaviruses. Virology, 445, 213-223.
- ROPERTO, S., BORZACCHIELLO, G., BRUN, R., LEONARDI, L., MAIOLINO, P., MARTANO, M., PACIELLO, O., PAPPARELLA, S., RESTUCCI, B., RUSSO, V., SALVATORE, G., URRARO, C. & ROPERTO, F., 2010. A review of bovine urothelial tumours and tumour-like lesions of the urinary bladder. Journal of Comparative Pathology, 142, 95-108.
- ROPERTO, S., BORZACCHIELLO, G., ESPOSITO, I., RICCARDI, M., URRARO, C., LUCA, R., CORTEGGIO, A., TATE, R., CERMOLA, M., PACIELLO, O. & ROPERTO, F., 2012. Productive infection of bovine papillomavirus type 2 in the placenta of pregnant cows affected with urinary bladder tumors. PLoS One, 7, e33569.
- ROPERTO, S., BRUN, R., PAOLINI, F., URRARO, C., RUSSO, V., BORZACCHIELLO, G., PAGNINI, U., RASO, C., RIZZO, C., ROPERTO, F. & VENUTI, A., 2008. Detection of bovine papillomavirus type 2 in the peripheral blood of cattle with urinary bladder tumours: possible biological role. The Journal of General Virology, 89, 3027-3033.
- ROPERTO, S., DE TULLIO, R., RASO, C., STIFANESE, R., RUSSO, V., GASPARI, M., BORZACCHIELLO, G., AVERNA, M., PACIELLO, O., CUDA, G. & ROPERTO, F., 2010. Calpain3 is expressed in a proteolitically active form in papillomavirus-associated urothelial tumors of the urinary bladder in cattle. PLoS One, 5, e10299.
- ROPERTO, S., MUNDAY, J.S., CORRADO, F., GORIA, M. & ROPERTO, F., 2016. Detection of bovine papillomavirus type 14 DNA sequences in urinary bladder tumors in cattle. Veterinary Microbiology, 190, 1-4.
- ROPERTO, S., RUSSO, V., BORZACCHIELLO, G., URRARO, C., LUCA, R., ESPOSITO, I., RICCARDI, M.G., RASO, C., GASPARI, M., CECCARELLI, D.M., GALASSO, R. & ROPERTO, F., 2014. Bovine papillomavirus type 2 (BPV-2) E5 oncoprotein binds to the subunit D of the V(1)-ATPase proton pump in naturally occurring urothelial tumors of the urinary bladder of cattle. PLoS One, 9, e88860.
- ROPERTO, S., RUSSO, V., LEONARDI, L., MARTANO, M., CORRADO, F., RICCARDI, M.G. & ROPERTO, F., 2016. Bovine papillomavirus Type 13 expression in urothelial bladder tumours of cattle. Transboundary and Emerging Diseases, 63, 628-634.
- ROPERTO, S., RUSSO, V., OZKUL, A., CORTEGGIO, A., SEPICI-DINCEL, A., CATOI, C., ESPOSITO, I., RICCARDI, M. G., URRARO, C., LUCA, R., CECCARELLI, D.M., LONGO, M. & ROPERTO, F., 2013. Productive infection of bovine papillomavirus type 2 in the urothelial cells of naturally occurring urinary bladder tumors in cattle and water buffaloes. PLoS One, 8, e62227.
- SANTOS, C., FERREIRINHA, P., SOUSA, H., RIBEIRO, J., BASTOS, M.M.S.M., NETO, T., OLIVEIRA, P.A., MEDEIROS, R., VILANOVA, M. & GIL DA COSTA, R.M., 2016. Ptaquiloside from bracken (Pteridium spp.) inhibits tumour-infiltrating CD8+ T cells in HPV-16 transgenic mice. Food and Chemical Toxicology, 97, 277-285.
- SCHILLER, J.T., DAY, P.M. & KINES, R.C., 2010. Current understanding of the mechanism of HPV infection. Gynecologic Oncology, 118, S12-17.
- SCHMITT, M., FIEDLER, V. & MULLER, M., 2010. Prevalence of BPV genotypes in a German cowshed determined by a novel multiplex BPV genotyping assay. Journal of Virological Methods, 170, 67-72.
- SOMVANSHI, R., 2011. Papillomatosis in buffaloes: A less-known disease. Transboundary and Emerging Diseases, 58, 327-332.
- STEWART, R.J.E., JACOBS, J.J.L., KOTEN, J. W. & DEN OTTER, W., 2010. Local interleukin-2 therapy of bovine vulval papilloma and carcinoma complex. Veterinary Record, 167, 825-826.
- THEILEN, G., WHEELDON, E.B., EAST, N., MADEWELL, B., LANCASTER, W.D. & MUNN, R., 1985. Goat papillomatosis. American Journal of Veterinary Research, 46, 2519-2526.
- TILBROOK, P.A., STERRETT, G. & KULSKI, J.K., 1992. Detection of papillomaviral-like DNA sequences in premalignant and malignant perineal lesions of sheep. Veterinary Microbiology, 31, 327-341.
- TORE, G., CACCIOTTO, C., ANFOSSI, A.G., DORE, G.M., ANTUOFERMO, E., SCAGLIARINI, A., BURRAI, G.P., PAU, S., ZEDDA, M.T., MASALA, G., PITTAU, M. & ALBERTI, A., 2017. Host cell tropism, genome characterization, and evolutionary features of OaPV4, a novel Deltapapillomavirus identified in sheep fibropapilloma. Veterinary Microbiology, 204, 151-158.
- TSIRIMONAKI, E., O'NEIL, B.W., WILLIAMS, R. & CAMPO, M.S., 2003. Extensive papillomatosis of the bovine upper gastrointestinal tract. Journal of Comparative Pathology, 129, 93-99.
- VAN DOORSLAER, K., RECTOR, A., VOS, P. & VAN RANST, M., 2006. Genetic characterization of the Capra hircus papillomavirus: A novel close-to-root artiodactyl papillomavirus. Virus Research, 118, 164-169.
- VANSELOW, B.A., SPRADBROW, P.B. & JACKSON, A.R., 1982. Papillomaviruses, papillomas and squamous cell carcinomas in sheep. The Veterinary Record, 110, 561-562.
- VENUTI, A., PAOLINI, F., NASIR, L., CORTEGGIO, A., ROPERTO, S., CAMPO, M.S. & BORZACCHIELLO, G., 2011. Papillomavirus E5: the smallest oncoprotein with many functions. Molecular Cancer, 10, 140.
- VITIELLO, V., BURRAI, G.P., AGUS, M., ANFOSSI, A. G., ALBERTI, A., ANTUOFERMO, E., ROCCA, S., CUBEDDU, T. & PIRINO, S., 2017. Ovis aries Papillomavirus 3 in ovine cutaneous squamous cell carcinoma. Veterinary Pathology, 54, 775-782.










