- Infectious Diseases of Livestock
- Part 2
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Post-weaning multi-systemic wasting syndrome in swine
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). JA Ellis, EG Clark, GM Allan and GS Krakowka, Post-weaning multi-systemic wasting syndrome in swine, 2018.
Post-weaning multi-systemic wasting syndrome in swine
Previous authors: E G CLARK, J A ELLIS, G M ALLAN AND S KRAKOWKA
Current authors:
J A ELLIS - Professor, DVM, PhD, Diplomate ACVP, Diplomate ACVM, Department of Veterinary Microbiology, Western College of Veterinary Medicine, University of Saskatchewan, 52 Campus Drive, Saskatchewan, S7N5B4, Canada
E G CLARK - Prairie Diagnostic Services, Western College of Veterinary Medicine, University of Saskatchewan, 52 Campus Drive, Saskatoon, Saskatchewan, Canada
G M ALLAN - Personal Chair of Porcine Virology, Queen’s University, 51 Cabin Hill Gardens, Belfast, Northern Ireland, BT5 7AQ, United Kingdom
G S KRAKOWKA - Department of Veterinary Biosciences, College of Veterinary Medicine, Ohio State University, Columbus, OH, USA
Introduction
In 1974 a small circular, non-enveloped DNA virus was first observed as a “contaminant” in a pig kidney cell line (PK-15).85, 87 When this agent was used to experimentally infect pigs in the mid-1980s, and again in the mid-1990s, no disease was produced9, 86 and so it was considered to be avirulent. In 1994, an outbreak of subacute to chronic, progressive, systemic, inflammatory disease with a high mortality rate, restricted primarily to young weaned pigs, was first identified in several minimal disease herds of pigs in western Canada.14, 35, 37 A viral aetiology was suspected, largely on the basis of the presence of basophilic inclusion bodies within cells in the granulomatous inflammatory foci. These lesions had been recognized as distinctive among swine in Saskatchewan as long ago as 1991 but the cause was not determined.14 The disease presentation in its epidemic form in affected herds was so distinctive that the term ‘post-weaning multi-systemic wasting syndrome’ (PMWS) was coined in 1996 to emphasize its most important features, namely the occurrence in pigs eight to 12 weeks of age of a disease with multiple organ system involvement whose chief clinical manifestations were progressive wasting and failure to thrive.14, 35 During the investigation of this outbreak, and on the basis of the morphologic similarities between the ‘inclusion bodies’ in PMWS cases and similar inclusions characteristic of a circovirus disease of birds, psittacine beak and feather disease, tissue sections from paraffin-embedded formalin-fixed PMWS tissues were stained with rabbit antisera prepared against the original porcine circovirus (PCV) and strong reactivity of the inclusions with this reagent was seen. A circovirus was first isolated from diseased porcine tissues in Canada,22 and then in Europe.7, 8 Since it was shown to be similar to known PCV, but sufficiently different, the original circovirus was named PCV-1, and the new, apparently pathogenic virus was designated PCV-2.4
In addition to classical PMWS, the constellation of porcine circovirus-associated diseases (PCVAD) include: the porcine respiratory disease complex (PRDC), enteric disease, reproductive disorders and abortion, and porcine dermatitis and nephropathy (nephritis) syndrome (PDNS).12, 17, 19, 33, 40, 74, 76, 79, 80, 83, 84, 90 Porcine circovirus 2 and PCVADs are found in domestic and wild pig populations worldwide.1, 12, 17, 40, 47, 49, 63 Over the last two decades much has been learned about the epidemiology, pathogenesis and modes of transmission of this small virus, and many excellent reviews have been published.1, 4, 18, 22, 29, 41, 43, 52, 62, 76, 77 These findings and their implications for the production of pigs are summarized in this chapter, with a focus on PMWS.
Aetiology
The porcine circoviruses, PCV-1, a common avirulent contaminant of the PK-15 (ATCC-CCL 33) porcine cell line,21 and the pathogenic PCV-2 are in the genus Circovirus within the family Circoviridae. The Circoviridae 51 comprises a group of small DNA virus pathogens of plants, birds and pigs.7, 8 The viruses are unique in that they are the smallest known autonomously replicating viruses. The form of their viral DNA, as a single-stranded (ss) circularized DNA molecule, is also distinct.2, 5, 7, 66, 88 The Circoviridae family contains two genera: the Gyrovirus genus is represented by chick anaemia virus, which is the type virus for this group,1, 51, 66 and the Circovirus genus which contains both porcine circoviruses (PCV-1 and PCV-2) and several avian circiviruses including psittacine beak and feather disease virus.66 The avian members of the Circoviridae are well-known viral pathogens. Similarities between PCV-1 and plant nanoviruses have been identified and these similarities have led to the suggestion that PCV-1 may represent the most ancient mammalian DNA replicon. The circoviruses probably originated by recombination of a plant nanovirus with a vertebrate virus.61 In plants, circoviruses have long been associated with disease and the plant nanoviruses are an important group of viral pathogens in their respective host plant species.
In vitro, both porcine circoviruses (PCVs) replicate in subconfluent monolayers of porcine kidney cells (PK15 cells) without producing obvious viral cytopathic effects;1, 87 cellular foci of virus production in monolayers are monitored by immunohistochemistry with monoclonal antibodies2, 61 to the PCV proteins. Porcine circovirus-2 replicates in human monocytes1 and human transformed cell lines;39 PCV-1 does not replicate in human cells.5, 63
The PCVs are icosahedral, non-enveloped virions with a buoyant density of 1,37 gm/cm3 in caesium gradients,1, 77 and are non-haemagglutinating agents resistant to inactivation following exposure to pH 3,0, chloroform and heating to 56 and 70 °C. The genome is about 1,7kb. There has been some controversy regarding the number of predicted open reading frames (ORFs) in the PCV genome; currently 11 are predicted, four of which (ORF1-4) are translated into proteins with biological roles.11, 28, 52, 54, 55 ORF 1 encodes, through alternative splicing, for 2 proteins, Rep1 and Rep 2 comprising the replicase enzyme that is necessary for genomic replication. ORF2 encodes for the capsid protein (Cap), the only protein in the viral capsid, and major target of immune responses. ORF 3 and 4 are in the antisense region of ORF1 and encode for 2 nonstructural proteins that are thought to be involved in the regulation of viral replication and apoptosis of infected cells. The complex Rep protein is highly conserved between PCV-1 and PCV-2 (85 per cent sequence identity), accounting for immunologic cross-reactivity between them;56 whereas the Cap protein is conserved within genotypes and more variable across genotypes of PCV-2.
Cell-free infectious virus is detected at 30 hours post-infection (pi). Viral mRNAs are synthesized at 18 hours pi (capsid mRNA is most abundant) and viral proteins appear in nuclei and cytoplasm shortly thereafter.11 The capsid protein contains a nuclear localization sequence that binds to cell nucleoli, presumably facilitating encapsidation of viral DNA. The origin of PCV DNA replication is contained within a conserved single stem loop configuration at nucleotide positions 341–349.34, 56 In the case of PCV-1, viral complementary DNA is replicated by the host cell DNA polymerases recruited in the S phase of replicating cells and the viral DNA encoded replication-associated proteins through a process of ‘rolling-circle’ replication56 to create double-stranded (ds) DNA replicative form (RF) intermediates. This DNA RF contains several open reading frames (ORFs) with the potential to encode for proteins greater than 5 kDa.1, 53 For PCV-1, a distinct population of small (roughly 5S) dsDNA intermediates are produced in infected cells and are thought to be defective PCV-1 forms;57 these small DNAs have not been observed in PCV-2.
Epidemiology
Epidemiological surveys indicate that serum antibodies are widespread in pig herds of both high and low health status, in large fattening units and small back-yard herds, whether or not PMWS or other PVCADs occur in them.16, 49, 71, 86, 89 As a consequence, piglets receive varying levels of colostrum-derived antibodies after suckling. Active sero-conversion generally occurs three to four weeks after weaning, at eight to 12 weeks of age. Concurrent presence of both serum antibodies and circulating virus, as determined by PCR, is a common occurrence in asymptomatic weanling pigs.65
Post-weaning multi-systemic wasting syndrome is commonly diagnosed in herds free of the major enteric and respiratory diseases affecting pigs, including mycoplasmal pneumonia, Actinobacillus pleuropneumoniae infection, atrophic rhinitis, salmonellosis, swine dysentery and infections caused by transmissible gastroenteritis and pseudorabies viruses. Affected herds may be farrow-to-finish, off-site nursery or grower pig operations. The syndrome has been diagnosed in herds ranging in size from small (50-sow) to large (1 200-sow) operations. It most commonly affects pigs two to three weeks post-weaning (at about five to six weeks of age), although this depends somewhat on the characteristics of pig flow within the unit. Draughts, overcrowding, poor air quality and co-mingled ages are thought to exacerbate the expression and severity of the disease. On affected farms, the morbidity and mortality rates associated with PMWS appear to be dependent on the degree of compartmentalization within the unit and stage of the outbreak.38, 71
Vertical transmission has been documented by demonstration of viral antigen and DNA in dead foetuses, mummified foetuses, and aborted foetuses, as well as in neonates.71, 90 In these foetuses and neonates, the route of infection is most probably by virtue of viraemic sows, rather than through PCV-2 positive semen.71 Infection may be established in the presence of colostrum-derived antibodies and first litter sero-negative gilts are most likely to transmit the infection to foetuses. Experimentally, foetuses of varying gestational ages have been inoculated with PCV-2 and examined for disease effects 21 days later.71 The virus replicated to high titres and produced a high fatality rate in 57-day-old foetuses. Foetuses inoculated at 75 days of gestation were virus- and seropositive and stillborn at term but lacked histologic lesions in tissues. Foetuses inoculated with PCV-2 at 92 days of gestation, while seropositive and viraemic at birth, were clinically normal and also showed no histological lesions. It is unlikely that vertical transmission is a significant factor in maintenance of virus within a breeding herd. Viral DNA has been recovered from semen, which suggested that venereal transmission may occur; however, there are contradictory data relevant to the importance of this mechanism of vertical transmission in the field.71
A second porcine disease syndrome, first recognized in 1993,80 and associated with PCV-2 infection is PDNS.19, 20, 33, 40, 76, 77 While both PMWS and PDNS have been diagnosed in the same herd, in most instances PDNS developed after PMWS waned. Except in a few instances, the overall prevalence within an infected herd is sporadic, but the mortality rate may be high. Circoviruses isolated from PDNS tissues were indistinguishable from those of PMWS isolates.60 In Europe, particularly the United Kingdom, PDNS was often higher in prevalence than was PMWS.33, 84 While PDNS may occur on farms as a single entity, it most often developed in herds that had experienced an outbreak of PMWS and were in the recovery or stabilization phases of the outbreak. Its clinical presentation resembles that of both African and classical swine fever; this is of concern from the differential diagnosis viewpoint for the pig industry in Europe.19 Virtually all cases of PDNS are also systemically infected with PCV-2 and show generalized lymphadenopathy.13, 19, 20, 74, 76, 77 As indicated, although originally PDNS was associated with PCV-2 infection, its presence in typical lesions is inconsistent. Recently, a novel porcine circovirus, designated, PCV-3, has been directly associated with the lesions of naturally-occurring PDNS.70 This virus is highly divergent from PCV-2 (only 30 per cent sequence identity with the capsid protein of PVC-2) and has not been isolated in culture, but appears to have a more direct role in the pathogenesis of PDNS than PCV-2. However, the overall clinical importance of PCV-3 is currently uncertain.30
All breeds of Sus scrofa domesticus appear to be susceptible to PCV-2 infection. There has been some association between pig breeds and variable susceptibility to PCV-2 and PCVAD. Experimentally, Landrace pigs showed some increased susceptibility to infection and/or more severe lesions than Large White or Duroc pigs.67 Mixed-bred Large White and Durocs were reported to be more susceptible to PCV-2 infection than purebred Pietrain pigs.68 There is some documentation that genetic differences impact the expression of PMWS in the field.50 However, these studies were limited in numbers of animals examined and, to date, there are few data that link genetic susceptibility to pedigree or lines (versus breed) of pigs using more precise measurements such as single nucletotide polymorphisms (SNPs).22, 57 Infection and PMWS in European wild boars (Sus scrofa scrofa) in captivity in Canada and free-ranging in Europe indicate that they may serve as a source of infection for domestic pigs.22
It is still not known if warthogs (Phacochoerus africanus, P. aethiopicus) and bush pigs (Potamochoerus larvatus, P. porcus). are susceptible, but, given the susceptibility of this species and domestic pigs to African swine fever, it would not be surprising to find that these animals also carry, or be capable of carrying, PCV-2. Cattle and horses that were tested were seronegative for PCV-2 and experimental infection of small ruminants and cattle has failed to yield evidence of either subclinical infection or disease.22, 25 Human sera may contain antibodies to recently described human circoviruses that may cross-react with the replicase protein of the PCVs, but there is no evidence that humans, even those in close contact with pigs and their products (e.g. veterinarians, producers and abattoir workers) can be infected with PCV-2.22, 82 The virus has been transmitted to mice,46 but no attempts to adapt it to other laboratory animal species have been reported.
The high resistance of this agent to environmental and chemical disinfection makes it highly probable that all premises contain infectious virus. Viral DNA has been demonstrated in excretions and secretions of pigs suffering from PMWS, and infectious virus (more than 102 infectious units per gram) can be recovered from faecal slurries contained in gnotobiotic isolation units more than 45 days after the pigs in them were initially infected.71 The most probable route of infection is via the oronasal cavities. Experimentally, oronasal, parenteral and a combination of these routes have been used to transmit the virus successfully to seronegative piglets.71
The tonsillar tissues are generally positive for viral antigen and these tissues, because of their accessibility, even on the slaughter floor, are excellent sources of the virus for the detection and diagnosis of PCV-2 infection.
From a molecular epidemiologic standpoint, PCV-1 and PCV-2 are thought to have diverged in the late nineteenth or early twentieth centuries and have co-circulated and undergone independent evolution, including recombination, since then.29 The dissemination and continuing evolution of PCV-2 has undoubtedly been enhanced by extensive global trade in pig semen, and pork meat.22, 29 Porcine circovirus-2 is currently divided into five genotypes; PCV-2a, b, d being predominant, and PCV-2 c,e, being of low prevalence.41 Retrospective analyses have indicated that the original Canadian isolate on which the vaccines are based was a PCV-2a. Prior to 2003, this was the genotype that predominated in Canada and the USA; internationally both PCV-2a and PCV-2b were more equally distributed. Then, around 2003, prior to vaccine use, there was a shift globally to a predominance of PCV-2b, which was associated, by some, with an increase in virulence.41, 62 More recently, PCV-2d (previously known as “mutant PVC-2b”) has been increasing in countries/regions with large pig populations, including USA, Europe, China, Korea, and South America.41 This change, again, has been associated by some with genotype-determined changes in virulence.41 Furthermore PCV-2c, originally thought to be confined to Denmark, as well as PCV-2d, have been isolated from wild pigs including peccaries in South America.41 As with other viruses that have been increasingly divided into genotypes, equating virulence, or avirulence, with a particular genotype is probably overly simplistic and potentially misleading; virulence is probably more often a function of within genotype variation, rather than between genotype variation.22 To date potential genetic determinants of PCV-2 virulence have not been identified, except it has been shown in an examination of archival and more contemporary viruses that mutational events within a 9-bp region in the capsid can alter the virulence of PCV-2 in experimental infections.42
Pathogenesis
Porcine circovirus 2 infection begins with the interaction of the capsid protein, presumably, with one of a variety of molecules on the cell surface, including glycosaminoglycans, heparin, heparin sulphate, and chondroitin sulphate B on a variety of cell types.64 The use of this broad range of “attachment receptors” allows for a broad cellular tropism, and hence the multiple organ involvement characteristic of PMWS. A histological and consistent hallmark of PCV-2 infection is the presence of copious circoviral protein and DNA in cells of monocyte-macrophage lineage, including dendritic cells, mostly in the cytoplasm; however, the virus does not replicate in most of these cells.22, 31, 32, 62 It is probably only a small subset of these myeloid cells that are truly permissive for viral replication.22, 31, 32, 62 What is important, however, is that it appears that cells of monocyte-macrophage lineage actively phagocytose infectious virus and maintain it as infectious virus in the cytoplasm (presumably in phagosomes) for at least eight days in vitro.22, 32
As with many infectious diseases, a key determinant of virulence is probably simply the amount of infectious virus that is produced in tissues. Disseminated multi-organ infection expressed as PMWS or its clinical variants is directly correlated to the amount of infectious virus recovered from tissues. In subclinical infections, PCV-2 is restricted chiefly to lymphoid tissues and viral titres are 105 infectious units per gram or less.24, 44, 45 In contrast, infectious virus titres in the tissues of affected animals may exceed 109 infectious units per gram.45 Quantitation of viral DNA in these tissues parallels these data with more than 1012 DNA targets per gram present in them. Yet, in spite of these titration data, the cellular source(s) of new virions remains to be fully elucidated. As a DNA virus, current virologic dogma dictates that there is replication of viral DNA in the cell nucleus since this virus lacks DNA polymerase and must use this host cell enzyme to produce linearized (ultimately circularized) progeny viral DNA.
Large quantities of infectious virus can be recovered by titration of lymphoid tissue and liver homogenates from PMWS-affected gnotobiotic pigs. Sera from PCV-2 infected pigs, whether subclinically infected or overtly diseased, contain copy numbers of PCV-2 DNA as high as 1010 copies per millilitre. It has been reported that PCV-2 antigen and DNA can be detected in lymphocytes in vivo.63, 78 In these, small numbers of nuclei of B lymphocytes contained material that stained positively with a polyclonal anti-PCV-2 serum were detected and it was suggested that viral infection of B lymphocytes induces apoptosis and resultant B cell depletion. 62, 78 In other reports the depletion of T cell dependent areas of nodes and resultant CD4 lymphopenia associated with PCV-2 infection have been emphasized but viral materials in T lymphocytes were not demonstrated.62, 75 In gnotobiotic piglet studies, viral antigen in lymphocytes is rarely if ever demonstrable even though both B and T cell compartments may be depleted and/or replaced by proliferating virus-positive histiocytes and macrophages.43, 45 Unlike PCV-1,5 PCV-2 is not infectious for resting or mitogen-activated porcine lymphocytes in vitro.31, 32 Thus, it may well be that infectious virus that can be recovered in high titre from lymph nodes represents infectious virus produced elsewhere and accumulates in nodes over time chiefly by phagocytic means. This is a strong possibility since it appears that the virus can persist in infectious form in these phagocytic cell types for long periods of time in vitro and may even replicate in a small population (more than one per cent) of G-CSF-responsive monocytes.
Porcine circovirus replicates in epithelial cells (e.g. porcine kidney) in vitro.1 Evidence for replication in epithelia in vivo is fragmentary, particularly in subclinically infected PMWS-negative piglets. In field cases of PMWS with overt liver disease, some hepatocytes are viral antigen- and DNA-positive.72 In contrast, degenerative and necrotic hepatocytes in experimental PMWS in gnotobiotes are largely virus-negative,44, 45 a consistent finding that raises the question of the mechanism(s) of hepatocellular degeneration and death in the latter. The reason(s) for this discrepancy between PMWS in conventional pigs and experimental PMWS in gnotobiotes is not clear, but possibly related to the lack of environmental activation stimuli in the latter, which reaches the liver chiefly via the portal circulation from the gut.
Initially, while reproduction of subclinical PCV-2 infection in young seronegative pigs was readily accomplished by inoculation of tissue culture-propagated virus,26 cloned viral DNA27 or tissue homogenates,10 reliable reproduction of PMWS with PCV-2 alone was often difficult to achieve. Although the reason(s) for this have not yet been determined, coinfection with other porcine viral pathogens, notably porcine parvovirus (PPV) and porcine reproductive and respiratory syndrome virus (PRRSV) were associated with a high prevalence of PMWS both in field cases and experimentally.3, 9, 22, 24, 26, 44 Subsequently, a number of other co-infections/disease have been identified as co-factors in the pathogenesis of PCVAD, including Mycoplasma spp., swine influenza virus, torque tenovirus, and bacterial septicaemia or pneumonia.22, 23, 62 In addition, immunostimulation of young pigs also appears to be a risk factor for development of overt disease.22, 45, 48, 62 Both immunostimulation and coinfection with other viral pathogens share features that facilitate replication of PCV-2 in vivo. Porcine parvovirus and PRRSV replicate in cells of monocyte, dendritic, macrophage or macrophage lineages. During convalescence, these cells become hypertrophic and hyperplastic and are rendered immunologically active either because of direct viral effects or indirectly as a result of the local action of cytokines or other inflammatory mediators released from virus-infected tissues.22, 62 Immune activation with non-living stimuli such as adjuvanted antigen,22, 45 vaccination with macrophage-targeted Mycoplasma spp. vaccine formulations6, 22, 48 and Bayimmune R,48 achieve similar biological effects. Since these are non-living, their effects are most likely mediated by pro-inflammatory cytokines. Regardless of the method of ‘‘immune activation’’, it is clear that these manipulations can heighten replication of PCV-2 and ultimately PMWS.22
Beyond the identification of various infectious and non-infectious co-factors, the actual mechanisms of cell damage, including lymphoid depletion and resultant disease, remain unresolved. There have been much controversy and contradictory data regarding ORF3 and its role in apoptosis of PCV-2 infected cells in vitro and in vivo.52, 62 It has also been proposed, based primarily on in vitro studies, that autophagy plays a role in the “death” of PCV-2-infected cells.52 The prominent involvement of PCV-2 with cells of monocyte- macrophage lineage has been associated with upregulation of IL-10 and pro-inflammatory cytokines such as IL-1 and TNFa, which may mediate the catabolic state that is characteristic of PMWS.18, 62 In addition to hypersecretion of these cytokines, downregulation of IL-2 and IL-4 may be linked to depletion/atrophy of the thymus and other lymphoid organs, and explain the immunosuppression that can be a feature of PCVAD.18, 22, 62 The immunomodulatory effect of oligodeoxynucleotides (ODNs) in PVC-2 DNA containing CpG motifs may mediate, at least in part, these effects on the immune system.62
Many key events in the pathogenesis of PMWS have been identified but there are gaps in knowledge of the progression of the disease process in infected pigs and, notably, why some pigs develop only subclinical but self-limiting disease while others progress to the overt disease.
Clinical signs
In most instances PCV-2 infections are subclinical. However, PMWS-affected pigs present with striking clinical signs, the most consistent of which are wasting and unthriftiness, dyspnoea with minimal coughing, prolonged pyrexia and peripheral lymphadenopathy in that order. Less frequent signs are watery profuse diarrhoea, skin pallor, icterus and ventral oedema indicative of progressive liver failure (Figure 1).1, 35, 37, 76, 77 During an acute outbreak, the monthly calculated mortality rate associated with PMWS may reach 25 per cent whereas endemically infected herds usually experience considerably lower morbidity and mortality rates of five per cent or less. Other clinical signs of PCVAD will vary with organ involvement.
The clinical sign most apparent to producers is failure to thrive. There is often an increased prevalence of opportunistic diseases or clinical disease associated with organisms of generally low virulence for pigs such as Pneumocystis sp. pneumonia.76 Co-occurrence of these infections is compatible with the concept that PMWS-affected pigs are immunocompromized as a result of systemic lymphoid depletion43, 62, 75, 78 and generalized granulomatous inflammation.76
Pathology
While end-stage PMWS is easily recognized, it is recommended that at least five to ten pigs, six to ten weeks of age be necropsied so that the complete range of gross and histopathological lesions can be determined. The reason for this is that in an infected herd some, all or none of the characteristic clinical signs and lesions may be expressed in individuals and the diagnosis may not be definitive if the sampling process is inadequate and does not consider the individual variability of disease expression.14, 15 Many lesions of diagnostic significance are detectable only by histopathology, necessitating that a complete set of tissues, with or without grossly visible lesions, be collected in phosphate buffered formalin for examination.
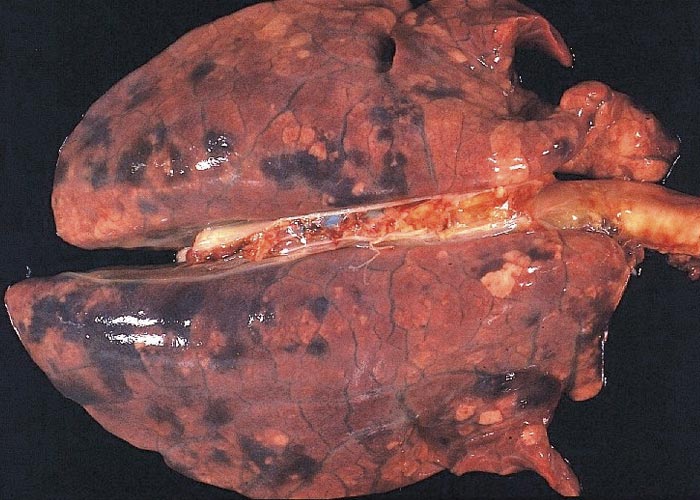
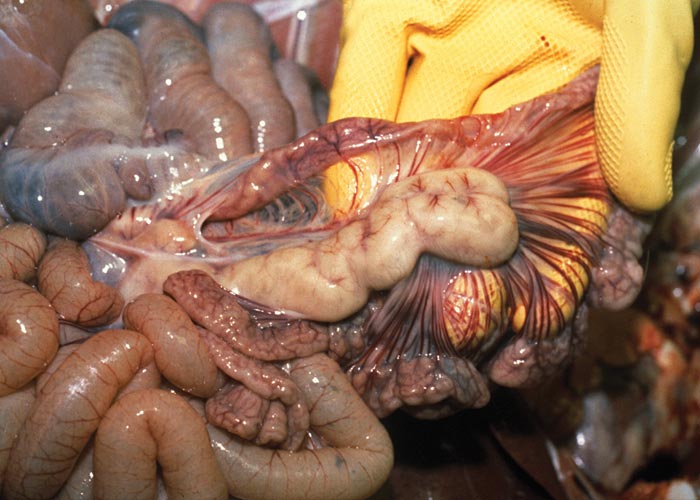
The carcasses show wasting of varying severity, skin pallor is common, and icterus may be present in severely wasted animals. Generalized oedema, most commonly the result of liver failure and hypoproteinaemia, may obscure the wastage of the carcass. The lungs show varying degrees of failure to collapse when the pleural cavity is opened, increased firmness and diffuse lobular grey to brownish mottling, particularly of the caudal lobes with interlobular oedema and focal areas of parenchymal haemorrhage. The ventral parts of the cranial lobes are sometimes consolidated (Figure 2). There is generalized lymphadenopathy: affected nodes are enlarged (often in excess of ten-fold above normal) and are firm and show loss of cortico-medullary distinction. The superficial and deep inguinal, mesenteric and bronchial nodes are most often affected, although these changes may occur in any lymph node (Figure 3). The thymus is variably reduced in size but this change is not diagnostic.
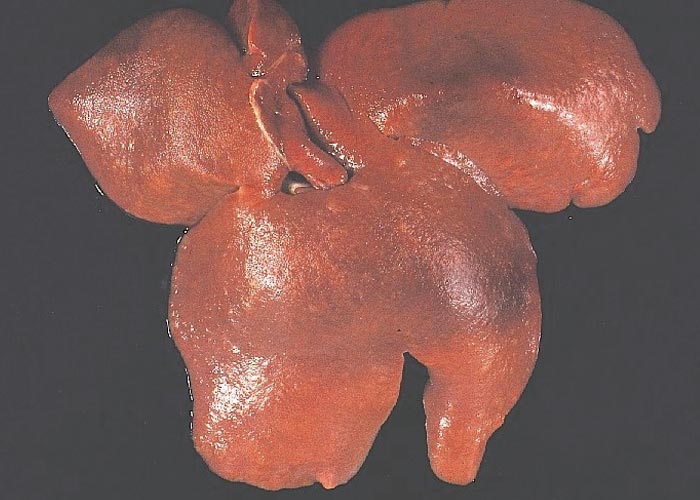
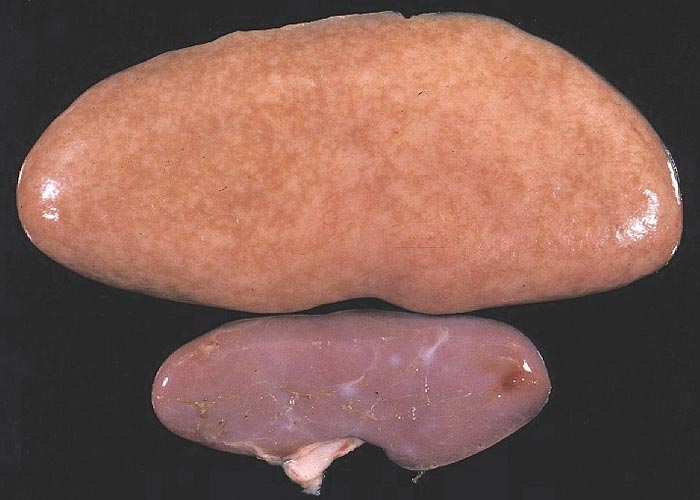
The liver may be reduced in size, firmer and paler than normal,44, 45, 72 or it may be of normal size but paler than normal or mottled with small pale foci (Figure 4). The kidneys may be normal in size and colour or very enlarged, up to eight times the normal size (Figure 5). On cut section, the renal parenchyma may be diffusely pale or studded with small pale foci or linear streaks. Oedema of the renal pelvis is frequently noticed. The spleen is often hyperplastic. Large non-bleeding erosions and ulcers of the pars oesophagus of the stomach are often present, or the stomach wall is thickened and oedematous. In cases that had manifested diarrhoea, the small intestine may be thin-walled and the caecum moderately distended with fluid content and a thickened wall.14, 15, 17, 36, 63, 76, 77
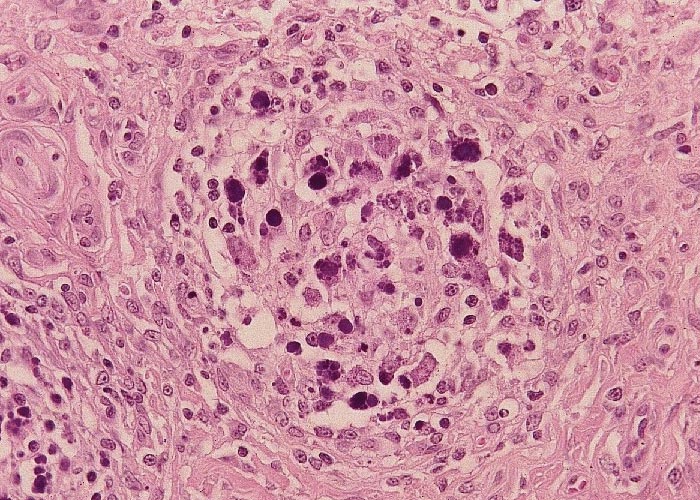
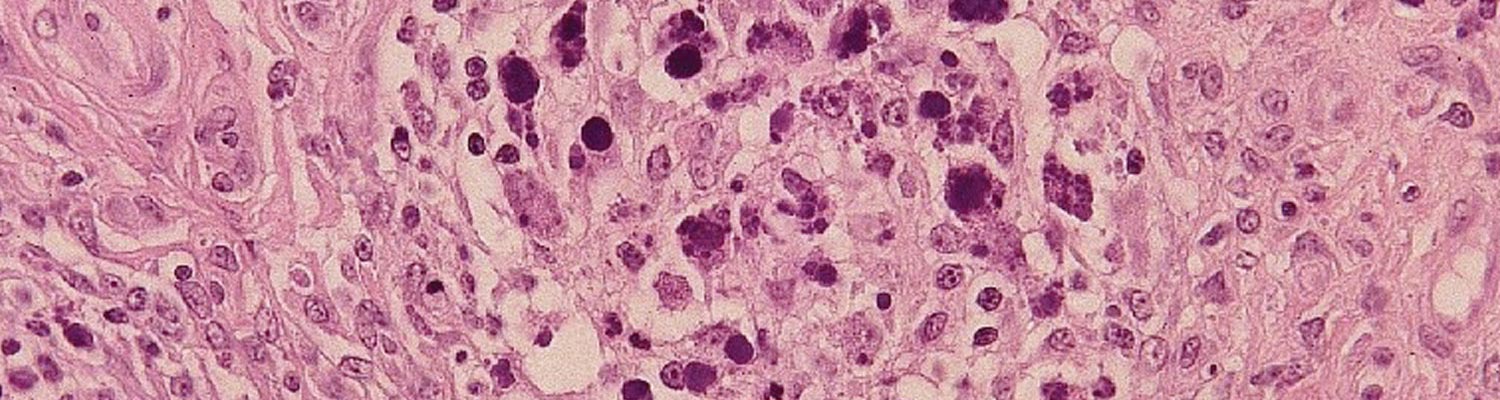
The most distinctive histopathological changes of PMWS occur in lymph nodes. Follicular B-cell zones of all lymphoid tissues are depleted of B lymphocytes and plasma cells, and the follicles are depleted of mature lymphocytes and reduced to faint remnants of primary follicles. Paracortical T cell zones are also depleted.1, 14, 15, 17, 43, 73, 75, 77 In both zones, depleted lymphocytes are replaced by large numbers of mature and immature histiocytic cells and variable numbers of multinucleated giant cells. Similar cells often almost obliterate the sinuses and T-cell dependent medullary regions. When present, large grape-like clusters of variably basophilic, cytoplasmic to seemingly extracellular inclusion bodies first appear in follicular dendritic cells of germinal centres of lymphoid tissues but may occur in histiocytes present in all tissues (Figure 6). These inclusion bodies are diagnostic of the disease but are present in less than half the number of cases. The palatine tonsils and Peyer’s patches of the ileum are important sites to examine for the inclusion bodies.
Hepatic lesions in subclinically affected pigs or in those without overt evidence of liver disease contain numerous small periportal mixed lymphoplasmacytic and histiocytic cell infiltrates44, 72 with associated single-cell necrosis of hepatocytes. As this lesion progresses in severity, the inflammatory cell infiltrates increase in extent and assume the features of multifocal to diffuse mixed granulomatous inflammation with syncytial giant cell formation. Accompanying this are proliferation of Kupffer cells and extensive panlobular hepatocellular loss and parenchymal collapse.1, 44, 45, 72, 76, 77
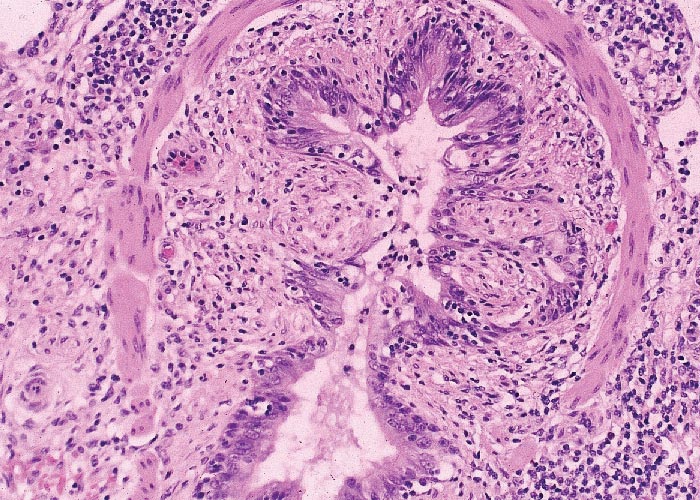
The kidneys show a mixed lymphoplasmacytic, histiocytic and occasional neutrophilic and eosinophilic interstitial nephritis accompanied by arteritis, phlebitis and lymphangitis. The tubular epithelium and glomeruli are largely intact. The lesions are frequently most prominent in the interstitial tissue of the cortex and the peripelvic connective tissue. In the gastrointestinal tract, villous atrophy and accompanying granulomatous enteritis similar to Johne’s disease in cattle are not infrequently seen. The stomach, caecum, duodenum and sometimes proximal spiral colon show necrosis and sloughing of variable severity of the glandular and crypt epithelium and mixed mononuclear cell infiltrations. Severe ulceration of the nonglandular gastric cardia is a common but not diagnostic finding. The lesions in the pancreas consist of localized areas of acinar cell atrophy and zymogen granule loss with lymphohistiocytic infiltrations. Non-suppurative myocarditis and mild perivascular leptomeningitis, particularly of the cerebral hemispheres, occur occasionally. The lungs show diffuse histiocytic-lymphocytic interstitial pneumonia. A striking feature is the apparently progressive localized to circumferential destruction and sloughing of bronchiolar and bronchial epithelium, resulting, in the terminal stages of the disease, in marked luminal stenosis (Figure 7) due to extensive submucosal histiocytic cell infiltrations. Secondary bacterial bronchopneumonia complicates the lesions in some cases.14, 15, 63, 73 In all of these extra-nodal tissues, inclusion bodies similar to those occurring in lymph nodes may be present in histiocytes within developing granulomas. However, absence of these inclusions is frequent enough to warrant the application of additional diagnostic tests on affected tissues.
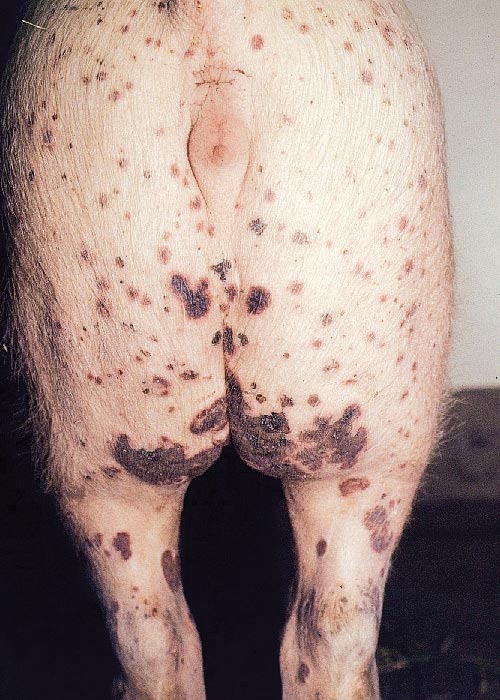
The lesions of PDNS are also distinctive. Pigs with ‘classic’ PDNS present with multifocal, firm, bright reddish to purplish to very dark reddish skin lesions that are usually prominent in the perineal, caudal thigh and ventral abdominal regions,19, 20 but may be generalized. These cutaneous lesions are 3 to 20 mm in diameter, are slightly raised and may be coalescent; large patches of coalescing lesions in severe cases are not uncommon. In most cases manifesting skin lesions, there are kidney lesions with gross changes that vary in extent and severity from case to case; not all cases of PDNS with kidney lesions will have well developed cutaneous lesions. In most affected pigs, both kidneys are markedly enlarged and diffusely paler than normal with very small red foci visible on the cortical and cut surfaces. More chronic atypical PDNS cases, such as those seen in slaughter- weight pigs, will have much paler kidneys in which the reddish foci are less apparent but the enlargement is nevertheless usually prominent. In addition to the renal lesions, generalized lymphadenopathy with histopathological features consistent with those of PMWS is a regular feature of these cases.
The histological lesions in lymphoid tissues are quite similar to those described in PMWS cases.19, 20, 76, 83 However, unlike PMWS, viral antigen is sparse in these tissues. The cutaneous lesions of PDNS appear to be mediated by fibrinoid necrosis and associated vasculitis of medium-sized arterioles in the superficial and deep dermis.76, 83 In the kidneys prominent lymphocytic interstitial nephritis is accompanied by fibrinoid degeneration and necrosis of glomerular tufts. That the filtration function of the glomeruli is compromised is manifested by the presence of proteinaceous material, fibrin, erythrocytes and, in some cases, leukocytes in Bowman’s spaces. The diagnostic feature is the abundant deposits of fibrin in Bowman’s space. In more chronic cases, there is partial to complete fibrosis of glomerular tufts as the fibrin deposits become organized and replaced by fibrous connective tissue and the renal tubules are dilated and distended with proteinaceous casts. In later stages of the disease, distinctive glomerular and periglomerular fibrosis and scarring develops. Glomeruli are reported to contain deposits of immunoglobulin and complement, compatible with an immune complex aetiology.19, 20, 40, 79, 83 Porcine circovirus capsid antigen has been demonstrated in renal tissues, specifically in tubules, mesangial cells and inflammatory cell infiltrates, but not in glomerular basement membranes.1 In some reports in piglets suffering from the PDNS syndrome there was a high prevalence of infection with PRRSV.62, 83 If the glomerular lesions are mediated by phlogistic circulating immune complexes, they do not contain PCV-2 capsid protein, but may contain PCV-3.70
Diagnosis
Given the endemicity of PCV-2, and since many if not all pigs are at least temporarily viraemic at some stage, in order to definitively establish the diagnosis of PMWS/PCVAD in individual pigs, three criteria should be met:
- observation of typical clinical signs such as wasting, increased mortality, and/or respiratory signs;
- presence of typical histological lesions, notably lymphoid deletion with granulomatous inflammation in lymphoid tissue;
- demonstration of PCV-2 antigen or DNA within histological lesions.22, 58, 81
Since lesions in individual pigs in herds experiencing a clinical outbreak of PCVAD vary considerably, it is usually prudent to examine several pigs before concluding that PCVAD is occurring. In particular, histological demonstration of the grape-like clusters of intracytoplasmic inclusion bodies, when present, and the presence of multinucleated giant cells in lymphoid organs associated with histiocytic infiltration and systemic lymphoid depletion, are almost pathognomonic for this disease complex.1, 14, 15, 76, 77 As a note of caution it should be borne in mind that well-developed viral inclusions do not develop in all PMWS cases, and the absence of them does not exclude PCV-2 as the aetiological agent of the disease.
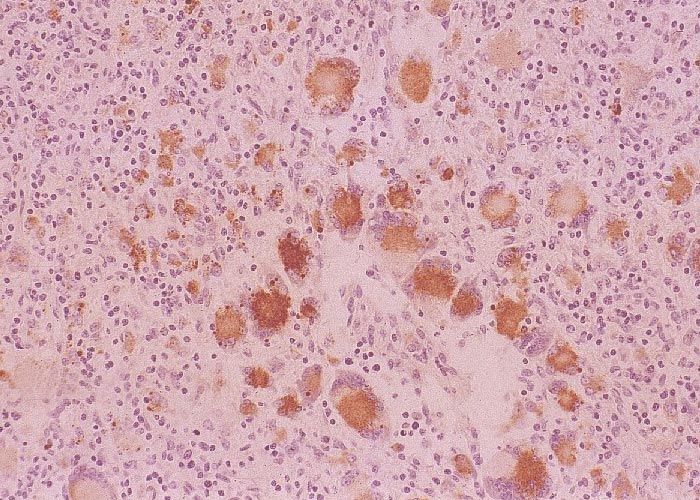
Viral isolation is possible but tedious, and is not definitive since the infection is widespread in asymptomatic pigs. Experimentally, quantitative virus titrations on infected tissues distinguish clinical from subclinical infections.43, 45 This manipulation is cumbersome and time consuming, and thus is not useful for field diagnostic procedures. Porcine circovirus 2-specific monoclonal antibodies2, 59 and polyclonal sera22 are used for demonstration of viral antigen in affected tissues by immunohistochemistry. Capsid protein is readily identified in cryostat or formalin-fixed paraffin-embedded tissue sections in the cytoplasm of monocyte-macrophages; inclusion bodies are strongly positive.1, 22 Viral antigen can also be found in the cytoplasm and occasional nuclei of hepatocytes, endothelium, renal tubular epithelium, alveolar and bronchial epithelium and poorly differentiated interstitial cells of probable histiocyte origin. A rapid diagnosis24 of PCV-2 infection/PMWS can be accomplished by immunohistochemical staining of acetone- fixed tissue impression smears made from cross sections of enlarged lymph nodes (Figure 8). Demonstration of viral DNA in tissues by in situ hybridization techniques can also be used on fresh or formalin-fixed tissues.1, 12, 13, 34, 49, 58, 65, 73 There are now commercially available antigen-capture ELISA kits to detect PCV-2 antigens, as well as PCR-based testing to detect DNA; however, the application of these methodologies can of course be limited in the absence of direct association with typical histologic changes.41, 62 Quantitative PCR (qPCR) to assess viral load can be indicative of disease status, and is currently being more widely applied in diagnostic settings.41, 62, 76 Numerous tests to detect PCV-2 antibodies are also available and have been used but should be done with the caveat that PCV-2 seropositivity is widespread.1, 22, 41, 62, 76 This is the case especially in vaccinated herds, and not necessarily indicative of causality; so that even evidence of seroconversion (acute and convalescent sera) needs to be evaluated with caution.
Differential diagnosis
Other common viral infections of pigs that can result in interstitial pneumonia and/or lymphadenopathy include, notably, PRRS and swine influenza. While the generalized lymphadenopathy characteristic of PMWS resembles, to some extent, generalized lymphoma, the epidemic nature of PMWS effectively excludes a neoplastic disease from the list of provisional diagnoses. Porcine circovirus-2 is recognized as an important pathogen of PRDC, a multifactorial (PRRSV, Mycoplasma hyopneumoniae, swine influenza virus, Pasteurella sp.) pneumonia of feeder and finisher pigs. Other causes of renal, hepatic, gastric and intestinal lesions always need to be considered in a differential diagnosis of PCVAD. Detailed macroscopic and microscopic examination of a number of suspected cases of PCVAD, together with immunohistochemistry, however, is often sufficient to allow differentiation from most other infections, or rule in PCV-2 as a co-infecting agent.14, 15, 36, 77
The diagnostic situation for PDNS (Figure 9) is complicated in Europe by the fact that the clinical signs and gross lesions strongly resemble both classical and African swine fever. Here, prompt and accurate diagnosis is critical since both of these diseases can have devastating consequences for pig production facilities as well as regulatory implications.
Control
As with many infectious disease of economic importance, PCVAD can be more or less effectively controlled, but not completely eliminated, by a combination of management/husbandry and immunoprophylaxis.41, 71 The severity and duration of PMWS and PCVAD vary from herd to herd. Since the virus is ubiquitous within pig populations and the agent itself is highly resistant to environmental degradation and commonly used disinfectants, it is unlikely that a facility can be rendered virus-free by sanitation alone. Prompt removal of affected pigs, quarantine of exposed weaners, and “all-in-all-out” systems have been promoted with some success.71 Control by management is difficult and depends largely on pig flow, improved sanitation, and early recognition and segregation of sick pigs. The association of PCVAD with pig herds of high health status and total confinement shows that good biosecurity, although always to be recommended, does not ensure freedom from PMWS.35, 37, 71
Currently, depending on the country, there are at least three types of commercial vaccines available: inactivated whole virus vaccines, a PCV-1/PCV-2 capsid (inactivated), and recombinant capsid subunit vaccines produced in a baculovirus expression system.41, 62 All of these vaccines are based on the PCV-2a strain originally isolated in Canada.22 Some vaccine protocols have focused on colostrum management, others on induction of adaptive immunity in piglets.22, 41, 62 Available vaccines have generally been shown to be highly efficacious in reducing morbidity and mortality associated with PCVAD, as well as controlling the effects of subclinical infections on production parameters such as feed conversion.22, 41, 62 Antibody responses, notably neutralizing antibodies and neutralizing epitopes, have been well characterized in vaccinated and challenged pigs and are correlated with disease-sparing. Cell-mediated responses are less well-documented, but secretion of interferon-g seems to be a correlate of protection as well.22, 41, 62
With the continuing evolution of PCV-2, there have been some controversy and contradictory data concerning cross-protection amongst antigenically disparate genotypes.22, 41, 62 Although possible vaccine failures have been reported, especially when pigs are infected with the PCV-2d strains,41 a recent study documented significant disease-sparing in PCV-2a-vaccinated pigs following experimental challenge with a virulent PCV-2d strain, indicating continued cross-protection in the face of viral evolution.69 Nevertheless, continued monitoring of the molecular epidemiology of PCV-2 infections, together with possible further vaccine development is warranted, given the nature of PCV-2, and the populations of intensively managed hosts in which it is evolving.
References
- ALLAN, G. M. & ELLIS, J. A., 2000. Porcine circoviruses: A review. Journal of Veterinary Diagnostic Investigation, 12, 3-14.
- ALLAN, G. M., KENNEDY, S. & MCNEILLY, F., 1994. Production, preliminary characterization and applications of monoclonal antibodies to porcine circovirus. Veterinary Immunology and Immunopathology, 43, 357-371.
- ALLAN, G. M., KENNEDY, S., MCNEILLY, F., FOSTER, J. C., ELLIS, H. A., KRAKOWKA, S., MEEHAN, B. M. & ADAIR, B. M., 1999. Experimental reproduction of severe wasting disease by co-infection of pigs with porcine circovirus and porcine parvovirus. Journal of Comparative Pathology, 121, 1-11.
- ALLAN, G. M., KRAKOWKA, S., ELLIS, J. A. & CHARREYRE, C., 2012. Discovery and evolving history of two genetically related but phenotypically different viruses, procine circoviruses 1 and 2. Virus Research, 164, 4-9.
- ALLAN, G. M., MCNEILLY, F., FOSTER, J. C. & ADAIR, B., 1994. Infection of leukocyte cell cultures derived from different species with porcine circovirus. Veterinary Immunology and Immunopathology, 41, 267-279.
- ALLAN, G. M., MCNEILLY, F., KENNEDY, S., MEEHAN, B. M., MOFFET, D., MALONE, F., ELLIS, J. A. & KRAKOWKA, S., 2000. Immunostimulation, PCV-2 and PMWS. The Veterinary Record, 147, 170-171.
- ALLAN, G. M., MCNEILLY, F., MEEHAN, B. M., KENNEDY, S., MACKIE, D. P., ELLIS, J. A., CLARK, E. G., ESPUNA, E., SAUBI, N., RIERA, P., BOTNER, A. & CHARREYRE, C., 1999. Isolation and characterization of circoviruses from pigs with wasting syndromes in Spain, Denmark and Northern Ireland. Veterinary Microbiology, 66, 115-123.
- ALLAN, G. M., MEEHAN, B., TODD, D., KENNEDY, S., MCNEILLY, F., ELLIS, J., CLARK, E., HARDING, J., ESPUNA, E., BOTNER, A. & CHARREYRE, C., 1998. Novel porcine circoviruses from pigs with wasting disease syndromes. The Veterinary Record, 142, 467-468.
- ALLEN, G. M., MCNEILLY, F., CASSIDY, J. P., REILLY, G. A., ADAIR, B., ELLIS, W. A. & MCNULTY, M. S., 1995. Pathogenesis of porcine; experimental infections of colostrum deprived piglets and examination of pig foetal material. Veterinary Microbiology, 44, 49-64.
- BALASCH, M., SEGALE, S. J., ROSELL, C., DOMINGO, M., MANKERTZ, A., URINIZA, A. & PLANA-DURAN, J., 1999. Experimental inoculation of conventional pigs with tissue homogenates from pigs with post weaning multisystemic wasting syndrome. Journal of Comparative Pathology, 121, 139-148.
- CHEUNG, A. K. & BOLIN, S. R., 2002. Kinetics of porcine circovirus type 2 replication. Archives of Virology, 147, 43-58.
- CHOI, C. & CHAE, C., 1999. In-situ hydrization for the detection of porcine circoviruses with post-weaning multisystemic wasting syndrome. Journal of Comparative Pathology, 121, 265-270.
- CHOI, C. & CHAE, C., 2001. Co-localization of porcine reproductive and respiratory syndrome virus and porcine circovirus 2 in porcine dermatitis and nephrology syndrome by double labeling technique. Veterinary Pathology, 38, 436-441.
- CLARK, E. G., 1996. Post-weaning multisystemic wasting syndrome. Proceedings of the American Association of Swine Practitioners, 499-501.
- CLARK, E. G. & HARDING, J. C., 1998. The pathology of post-weaning multisystemic wasting syndrome. Proceedings of the Fifteenth International Pig Veterinary Society Congress, 212.
- COTRELL, T. S., FRIENDSHIP, R. M., DEWEY, C. E., JOSEPHSON, G., ALLAN, G., F, M. & WALKER, I., 1999. Epidemiology of post-weaning multisystemic wasting syndrome in Ontario. Proceedings of the American Association of Swine Practitioners, 389-390.
- DAFT, B., NORDHAUSEN, R. W., LATIMER, K. S. & NIAGRO, F. D., 1996. Interstitial pneumonia and lymph adenopathy associated with circoviral infection in a six-week-old pig. Proceedings of the American Association of Veterinary Laboratory Technicians, 32, 39.
- DARWICH, L. & MATEU, E., 2012. Immunology of porcine circovirus type 2 (PCV2). Virus Research, 164, 61-67.
- DONE, S., GRESHAM, A., POTTER, R. & CHENNELLS, D., 2001. PMWS and PDNS—two recently recognised diseases of pigs in the UK. In Practice, 23, 14-21.
- DROLET, R., THIBAULT, S., D’ALLAIRE, S., THOMPSON, J. R. & DONE, S. H., 1999. Porcine dermatitis and nephropathy syndrome (PDNS): An overview of the disease. Swine Health and Production, 7, 283-285.
- DULAC, G. C. & FSHAR, A., 1989. Porcine circovirus antigens in PK-15 cell line (ATCC CCL-33) and evidence of antibodies in Canadian pigs. Canadian Journal of Veterinary Research, 53, 431-433.
- ELLIS, J., 2014. Porcine circovirus: A historical perspective. Veterinary Pathology, 51, 315-327.
- ELLIS, J., ALLAN, G. & KRAKOWKA, S., 2008. Effect of coinfection with genogroup 1 porcine torque teno virus on porcine circovirus type 2-associated postweaning multisystemic wasting syndrome in gnotobiotic pigs. American Journal of Veterinary Research, 69, 1608-1614.
- ELLIS, J. A., BRATANICH, A., CLARK, E. G., ALLAN, G., MEEHAN, B., HAINES, D. M., HARDING, J., WEST, K. H., KRAKOWKA, S., KONOBY, C., HASSARD, L., MARTIN, K. & MCNEILLY, F., 2000. Co-infection by porcine circoviruses and porcine parvovirus in pigs with naturally acquired post-weaning multisystemic wasting syndrome. Journal of Veterinary Diagnostic Investigation, 12, 21-27.
- ELLIS, J. A., KONOBY, C., WEST K, H., ALLAN, G. M., KRAKOWKA, S., MCNEILLY, F., B, M. & WALKER, I., 2001. Lack of antibodies to porcine circovirus type 2 virus in beef and dairy cattle and horses in western Canada. Canadian Veterinary Journal, 42, 461-464.
- ELLIS, J. A., KRAKOWKA, S., LAIRMORE, M., HAINES, D., BRANANICH, A., CLARK, E., ALLAN, G., KONOBY, C., HASSARD, L., MEEHAN, B., MARTIN, K., HARDING, J., KENNEDY, S. & F, M., 1999. Reproduction of lesions of post-weaning multisystemic wasting syndrome in gnotobiotic piglets. Journal of Veterinary Diagnostic Investigation, 11, 3-14.
- FENAUX, M., HALBUR, P. G., HAQSHENAS, G., ROYER, R., THOMAS, P., NAWAGITGUL, P., GILL M, TOTH, T. E. & MENG, X. J., 2002. Cloned genomic DNA of type 2 porcine circovirus is infectious when injected directly into the liver and lymph nodes of pigs: Characterization of clinical disease, virus distribution, and pathologic lesions. Journal of Virology, 76, 541-551.
- FINSTERBUCH, T., STEINFELDT, T., CALISKAN, R. & MANKERTZ, A., 2001. Lyses of the subcellular localization and the interaction of the rep and rep’ of porcine circovirus type 1. Proceedings: ssDNA Viruses of Plants, Birds, Pigs and Primates, St. Malo, France, 24-27 September, 37.
- FRANZO, G., CORTEY, M., SEGALES, J., HUGHES, J. & M, D., 2016. Phylodynamic analysis of porcine circovirus type -2 reveals global waves of emerging genotypes and the ciruculation of recombinant forms. Molecular Phylogenic Evolution, 100, 269-280.
- FRANZO, G., LEGNARDI, M., TUCCIANRONE, C. M., DRIGO, M., KLAUMANN, F., SOHRMANN, M. & SEGALES, J., 2018. Porcine circovirus type 3: a threat to the pig industry. Veterinary Record, 182, 83.
- GILPIN, D. F., MCCULLOUGH, K. C., MEEHAN, B. M., MCNEILLY, F., MCNAIR, I., STEVENSON, L. S., FOSTER, J. C., ELLIS, J. A., KRAKOWKA, S., ADAIR, B. M. & ALLAN, G. M., 2002. In vitro studies on the infection and replication of porcine circovirus type 2 in cells of the porcine immune system. Veterinary Immunology and Immunopathology, in press,
- GILPIN, D. F., STEVENSON, L. S., MCCULLOUGH, K., KRAKOWKA, S., MEEHAN, B. M., MCNEILLY, F., FOSTER, C., ADAIR, B., WELSCH, M. & ALLAN, G. M., 2001. Studies on the in vitro and in vivo effect of porcine circovirus type 2 infection of porcine monocytic cells. Proceedings: ssDNA Viruses of Plants, Birds, Pigs and Primates, St. Malo, France, 24-27 September, 97.
- GRESHAM, A., ALLAN, G., MCNEILLY, F. & KENNEDY, S., 2001. Links between post-weaning multiystemic wasting syndrome and porcine dermatitis nephropathy syndrome. The Pig Journal, 47, 155-159.
- HAMEL, A., L, LIN, L. L. & NAYAR, G. P. S., 1998. Nucleotide sequence of porcine circoviruses associated with post-weaning multisystemic wasting syndrome in pigs. Journal of Virology, 72, 5262-5267.
- HARDING, J. C., 1997. Post-weaning multisystemic wasting syndrome (PMWS): Preliminary epidemiology and clinical presentation. Proceedings of the American Association of Swine Practitioners, 503.
- HARDING, J. C. S. & CLARK, E. G., 1997. Recognizing and diagnosing post-weaning multisystemic wasting syndrome (PMWS). Swine Health and Production, 5, 201-203.
- HARDING, J. C. S. & CLARK, E. G., 1998. Post-weaning multisystemic wasting syndrome (PMWS): preliminary epidemiology and clinical presentation. 1384 SECTION FOUR: Viral diseases: Circoviridae Proceedings of the Fifteenth International Pig Veterinary Society Congress, 213.
- HARDING, J. C. S., CLARK, E. G., STROKAPPE, J. H., WILLSON, P. I. & ELLIS, J. A., 1998. Post-weaning multisystemic wasting syndrome: Epidemiology and clinical presentation. Swine Health and Production, 6, 249-254.
- HATTERMANN, K. & MANKERTZ, A., 2001. Infection studies of human cell-lines with porcine circovirus type 1 and type 2. Proceedings ssDNA Viruses of Plants, Birds, Pigs and Primates, St. Malo, France, 24-27 September, 50.
- HELIE, P. D. R. & GERMAIN, M. C., 1995. Systemic necrotizing vasculitis and glomerulonephritis in grower pigs in Southwestern Quebec. Canadian Veterinary Journal, 36, 150-154.
- KARUPPANNAN, A. K. & OPRIESSNIG, T., 2017. Porcine circovirus type 2 (PCV2) vaccines in the context of current molecular epidemiology. Viruses, 9, 99-103.
- KRAKOWKA, S., ALLAN, G., ELLIS J, HAMBERG, A., CHARREYRE, C., KAUFMANN, E., BROOKS, C. & B, M., 2012. A nine bas-base neucleotide sequence in the porcine circovirus type 2 (PVC-2) neucleocapsid gene determines viral replication and virulence. Virus Research, 164, 90-99.
- KRAKOWKA, S., ELLIS, J. A., MCNEILLY, F., GILPIN, D., MEEHAN, B., MCCULLOUGH, K. & ALLAN, G. A., 2002. Immunologic features of porcine circovirus type 2 infection. Virology and Immunology, 15, 567-582.
- KRAKOWKA, S., ELLIS, J. A., MEEHAN, B., KENNEDY, S., MCNEILLY, R. & ALLAN, G. A., 2000. Viral wasting syndrome of swine: Experimental reproduction of the disease by co-infection with porcine circovirus-2 (PCV 2) and porcine parvovirus (PPV). Veterinary Pathology, 37, 254-263.
- KRAKOWKA, S., ELLIS, J. A., MEEHAN, B., RINGLER, S., RINGS, M. D. & ALLAN, G. A., 2001. In vivo immune activation is the pivotal event in the production of wasting disease in pig infected with porcine circovirus-2 (PCV-2). Veterinary Pathology, 38, 31-42.
- KUIPEL, M., STEVENSON, G. W., CHOI, J., LATIMER, K. S., KANITZ, C. L. & MITTAL, S. K., 2001. Viral replication and lesions in BALB/c mice experimentally inoculated with porcine circovirus isolated from a pig with postweaning multisystemic wasting disease. Veterinary Pathology, 38, 74-82.
- KUIPEL, M., STEVENSON, G. W., MITTAL, S. K., CLARK, E. G. & HAINES, D. M., 1998. Circovirus-like disease in weaned pigs in Indiana. Veterinary Pathology, 35, 303-307.
- KYRIAKIS, S. C., SAOULIDIS, K., LEKKAS, S., MILIOTIS, C. H. C., PAPOUTIS, P. A. & KENNEDY, S., 2002. The effects of immunomodulation on the clinical and pathological expression of postweaning multisystemic wasting syndrome. Journal of Comparative Pathology, 126, 38-46.
- LAROCHELLE, R., MORIN, M., ANTAYA, M. & MAGAR, R., 1999. Identification and incidence of porcine circovirus in routine field cases in Quebec as determined by PCR. The Veterinary Record, 145, 140-142.
- LOPEZ-SORIA, S., NOFRARIAS, M., CALSAMIGLIA, M., ESPINAL, A. & VALERO, O., 2011. Post-weaning multisystemic wasting syndrome (PMWS) clinical expression under field conditions is modulated by the pig genetic background. Veterinary Microbiology, 149, 352-357.
- LUKERT, P., DE BOER, G. R., DALE, J. L., KEESE, P., MCNULTY, M. S., RANDLES, J. W. & TISCHER, I., 1995. The Circoviridae. In: MURPHY, F. A., FAUQUET, C. M., BISHOP, D. H. L, GHABRIAL, S.A., JARVIS, A. W., MARTELLI, G. P., MAYO, M. A. & SUMMERS, M. D., (eds). Virus Taxonomy. Sixth Report of the International Committee on Taxonomy of Viruses, New York: Springer, 166-168.
- LV, Q. Z., GUO, K. K. & ZHANG, Y. M., 2014. Current understanding of genomic DNA of porcine circovirus type 2. Virus Genes, 49, 1-10.
- MAHE, D., BLANCHARD, P., TRUONG, C., ARNAULD, C., LE CANN, P., CARIOLTE, R., MADEC, F., ALBINA, E. & JESTIN, A., 2000. Differential recognition of ORF2 protein from type 1 and type 2 porcine circoviruses and identification of immunorelevant epitopes. Journal of General Virology, 7, 1815-1824.
- MANKERTZ, A., FINSTERBUSCH, T., STEINFELDT, T., HILLENBRAND, B. & CALISKAN, C., 2001. Analyses of the replication proteins rep and rep’ of porcine circovirus type 1. Proceedings ssDNA Viruses of Plants, Birds, Pigs and Primates, St. Malo, France, 24-27 September, 18-19.
- MANKERTZ, A., MANKERTZ, J., WOLF, K. & BHUK, H. J., 1998. Identification of a protein essential for replication of porcine circovirus. Journal of General Virology, 79, 381-384.
- MANKERTZ, A., PERSSON, F. & MANKERTZ, J., 1997. Mapping and characterization of the origin of DNA replication of porcine circovirus. Journal of Virology, 71, 2562-2566.
- MCKNITE, A. M., et al., 2014. Genomic analysis of the differential response to experimental infection with porcine circovirus 2b. Animal Genetics, 45, 205-214.
- MCNEILLY, F., KENNEDY, S., MOFFET, T. D., MEEHAN, B. M., FOSTER, J. C., CLARK, E., G ELLIS, J. A., HAINES, D. M., ADAIR, B. M. & ALLAN, G. M., 1999. A comparison of in situ hydridization and immunocytochemistry for the detection of a new porcine circovirus in formalin-fixed tissues from pigs with post-weaning multisystemic wasting syndrome. Journal of Virological Methods, 80, 123-128.
- MCNEILLY, F., MCNAIR, F., MACKIE, D. P., MEEHAN, B. M., KENNEDY, S., MOFFETT, D., ELLIS, J. A., KRAKOWKA, S. & ALLAN, G. M., 2001. Production characterization and applications of monoclonal antibodies to porcine circovirus type 2. Archives of Virology, 146, 909-922.
- MEEHAN, B. M., MCNEILLY, F., MCNAIR, I., WALKER, I., ELLIS, J. A., KRAKOWKA, S. & ALLAN, G. M., 2001. Isolation and characterization of porcine circovirus 2 from cases of sow abortion and porcine dermatitis and nephropathy syndrome. Archives of Virology, 146, 1-8.
- MEEHAN, B. M., MCNEILLY, F., TODD, D., KENNEDY, S., JEWHURST, V. A., ELLIS, J. A., HASSARD, L. E., CLARK, E. G., HAINES, D. M. & ALLAN, G. M., 1998. Characterization of novel circovirus DNAs associated with wasting syndromes in pigs. Journal of General Virology, 79, 2171-2179.
- MENG, X. J., 2013. Porcine circovirus Type 2 (PCV2): Pathogenesis and interaction with the immune system. Annual Review of Animal Biosciences, 1, 43-64.
- MOROZOV, I., SIRINARUMITR, T., SORDEN, S. D., HALBUR, P. G., MORGAN, Y., YOON, K.-J. & PAUL, P. S., 1998. Detection of a novel strain of porcine circovirus in pigs with post-weaning multisystemic wasting syndrome. Journal of Clinical Microbiology, 36, 2535-2541.
- NAUWYNCK, H., SANCHEZ, R., MEERTS, P., LEFEBVRE, D. J., SAHA, D., HUANG, L. & MISINZO, G., 2012. Cell tropism and entry of porcine circovirus 2. Virus Research, 164, 43-45.
- NAYAR, G. P. S. H. A. & LIN, L., 1997. Detection and characterization of porcine circovirus associated with post-weaning multisystemic wasting syndrome in pigs. Canadian Veterinary Journal, 38, 385-386.
- NOTEBORN, M. H. M., 2001. Chicken anemia virus induced apoptosis: Underlying molecular mechanisms. Proceedings ssDNA Viruses of Plants, Birds, Pigs and Primates, St. Malo, France. 24-27 September, 20-22.
- OPRIESSNIG, T., FENAUX, M., THOMAS, P., HOOGLAND, M. J., ROTHSCHILD, M., MENG, X. J. & HALBUR, P. G., 2006. Evidence of breed dependent differences in susceptibility of porcine circovirus type 2-associated disease and lesions. Veterinary Pathology, 43, 281-293.
- OPRIESSNIG, T., PATTERSON, A. R., MADSON, D. M., PAL, N., ROTHSCHILD, M., KUHAR, D., LUNNEY, J. K., JUJAN, N. M., MENG, X. J. & HALBUR, P. G., 2009. Difference in severity of porcine circovirus type two-induced pathological lesions between Landrace and Pietrain pigs. Journal of Animal Science, 87, 1582-1590.
- OPRIESSNIG, T., XIAO, C.-T., HALBUR, P. G., GERBER, P. F., MATZINGER, S. R. & MENG, X.-J., 2017. A commercial porcine circovirus(PCV) type 2a-based vaccine reduces PCV2d viremia and shedding and prevents PCV2d transmission to naïve pigs under experimental conditions. Vaccine, 35, 248-254.
- PALINSKI, R., PINEYRO, P., SHANG, P., YUAN, F., GUO, R., FANG, Y., BYERS, E. & HAUSE, B. M., 2017. A novel porcine circovirus distantly related to known circoviruses is associated with porcine dermatitis and nephropathy syndrome and reproductive failure. Journal of Virology, 91, 1-13.
- ROSE, N., OPRIESSNIG, T., GRASSLAND, B. & JESTIN, A., 2012. Epidemiology and transmission of porcine circovirus type 2 (PCV2). Virus Research, 164, 78-89.
- ROSELL, C., SEGALES, J. & DOMINGO, M., 2000. Hepatitis and staging of hepatic damage in pigs naturally infected with porcine circovirus type 2. Veterinary Pathology, 37, 687-692.
- ROSELL, C., SEGALES, J., PLANA-DURAN, J., BLALSCH, M., RODRIGUEZ-ARRIOJA, G. M., KENNEDY, S., ALLAN, G. A., MCNEILLY, F., LATIMER, K. S. & DOMINGO, M., 1999. Pathological, immunohistochemical, and in-situ hybridization studies of natural cases of post-weaning multisystemic wasting syndrome (PMWS) in pigs. Journal of Comparative Pathology, 120, 59-78.
- ROSELL, C., SEGALES, J., RAMOS-VARA, J. A., FOLCH, J. M., RODRIGUEZ-ARRIOJA, G. M., DURAN, C. O., BALASCH, M., PLANA-DURAN, J. & DOMINGO, M., 2000. Identification of porcine circovirus in tissues of pigs with porcine dermatitis and nephropathy sundrome. The Veterinary Record, 146, 40-43.
- SARLI, G., MANDRIOLI, L., LAURENTI, M., SIDOLI, L., CERATI, C., ROLLA, G. & MARCATO, P. S., 2000. Immunohistochemical characterization of the lymph node reaction in pig postweaning multisystemic wasting syndrome. Veterinary Immunology and Immunopathology, 83, 53-67.
- SEGALES, J., 2012. Porcine circovirus 2 (PCV2) infections: Clinical signs, pathology, and laboratory diagnosis. Virus Research, 164, 10-19.
- SEGALES, J. & DOMINGO, M., 2002. Postweaning multisystemic wasting syndrome (PMWS) in pigs. A review. Veterinary Quarterly, 24, 109-124.
- SHIBAHARA, T., SATO, K., ISHIKAWA, Y. & KADOTA, K., 2000. Porcine circovirus Induces B lymphocyte depletion in pigs with wasting disease. Journal of Veterinary Medical Science, 62, 1125-1131.
- SIERRA, M. A., DE LAS MULAS, J. M., MOLENBEEK, R. F., VAN, MAANEN, C., QUEZADA, M. & GRUYS, E., 1997. Porcine immune complex Post-weaning multi-systemic wasting syndrome in swine 1385 glomerulonephritis dermatitis (PIGD) syndrome. European Journal of Veterinary Pathology, 3, 63-70.
- SMITH, W. J., THOMSON, J. R. & DONE, S., 1993. Dermatitis/nephropathy syndrome (Letter). The Veterinary Record, 133, 47.
- SORDEN, S., 2000. Update on porcine circovirus and postweaning multisystemic wasting syndrome. Swine Health and Production, 8, 133-136.
- TAKAHASHI, K., IWASA, Y., HIJIKATA, M. & MISHIRO, S., 2000. Identification of a new human DNA virus (TT like minivirus, TLMV) intermediately related to TT virus and chicken anemia virus. Archives of Virology, 145, 979-993.
- THIBAULT, S., DROLET, R., GERMAIN, M.-C., D’ALLAIRE, S., LAROCHELLE, R. & MAGAR, G., 1998. Cutaneous and systemic necrotizing vasculitis in swine. Veterinary Pathology, 35, 108-116.
- THOMSON, J., HENDERSON, L., MEIKLE, C. & MACINTYRE, N., 2001. Porcine dermatitis and nephropathy syndrome. The Veterinary Record, 148, 282-283.
- TISCHER, I., GELDERBLOM, H., VETTERMANN, W. & KOCH, M. A., 1982. A very small porcine virus with circular single-stranded DNA. Nature, 295, 64-66.
- TISCHER, I., MIELDS, W., WOLFF D, VAGT, M. & GRIEM, W., 1986. Studies on the epidemiology and pathogenicity of porcine circovirus. Archives of Virology, 91, 271-276.
- TISCHER, I., RASCH, R. & TOCHTERMANN, G., 1974. Characterization of papovavirus- and picornavirus-like particles in permanent piglet kidney cell lines. Zentrablat fu¨r Bakertiologie, 226, 153-167.
- TODD, D., NIAGRO, F. D., RITCHIE, B. W., CURRAN, W., ALLAN, G. M., LUKERT, P. D., LATIMER, K. S., STEFFENS, W. L. & MCNULTY, M. S., 1991. Comparison of three animal viruses with circular single-stranded DNA genomes. Archives of Virology, 117, 129-135.
- WALKER, I. W., KONOBY, C. A., JEWHURST, V. A., MCNAIR, I., MCNEILLY, F., MEEHAN, B., COTTRELL, T. S., ELLIS, J. A. & ALLAN, G. M., 2000. Development and application of a competitive ELISA for the detection of serum antibodies to porcine circovirus type 2. Journal of Veterinary Diagnostic Investigation, 12, 400-405.
- WEST, K. W., BYSTROM, J., WOJNAROWICZ, C., ELLIS, J. A., KRAKOWKA, S. & ALLAN, G. M., 1999. Myocarditis and abortion associated with intrauterine infection of sows with porcine circovirus. Journal of Veterinary Diagnostic Investigation, 11, 530-532.