- Infectious Diseases of Livestock
- Part 2
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Equid herpesvirus 1 and equid herpesvirus 4 infections
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Equid herpesvirus 1 and equid herpesvirus 4 infections
G P ALLEN, J H KYDD, J D SLATER AND K C SMITH
Introduction
Equid herpesvirus 1 (EHV-1) and equid herpesvirus 4 (EHV-4) are ubiquitous herpesviruses that infect the majority of the world’s domestic horses (Equus caballus) at some time during their lives, frequently resulting in serious clinical illness.5, 43, 44, 73, 74, 162, 174 Infections by the two herpesviruses have been recognized for over 60 years as significant and worldwide impediments to the breeding, competition and recreational horse industries.49, 82, 83, 85, 86, 87, 116, 125, 128, 129, 153, 160, 166, 167, 170, 182, 197, 233, 254The economic losses and negative impact on equine welfare caused by EHV-1 and EHV-4 occur perennially, are cumulatively immense, and are the result of morbidity or mortality from virus-induced abortion, respiratory disease, neurological disorders, or death of full-term and new-born foals.
Since the discovery of this viscerotropic subgroup of alphaherpesviruses of the horse (Table 76.1), major advances have been achieved in characterizing their physicochemical and molecular properties and in understanding the natural history of the virus–host relationship. Like all members of the Herpesviridae family of viruses, EHV-1 and EHV-4 are phylogenetically ancient and, during their protracted co-evolution with the horse, have become extremely successful viral parasites. Such success of EHV-1 and EHV-4 as permanent guests of the horse is dependent upon the possession, by the two herpesviruses, of a combination of unique biological features:
- tropism for equine respiratory mucosal epithelium that permits efficient viral entry into the horse;
- post-infection persistence of the viruses in a non-replicating, latent form in a long-lived set of equine cells to create a large, permanent reservoir of the viruses;
- virological attributes that provide a mechanism for re-emergence from their latent, cellular repository to the equine respiratory tract with shedding of infectious virions into the respiratory secretions; and
- a variety of evasive immunological mechanisms, such as latency, immunosuppression and direct cell-to-cell transfer of virus, for circumventing host antiviral defences.
From a biological viewpoint, the pathogenic potential of EHV-1 and EHV-4 for the horse and the resulting negative economic and welfare impact on equine activities can be considered as consequences of basic herpesviral attributes that, over eons of time, have been evolutionarily acquired for ensuring long-term survival of the viruses as microparasites of horses.
Aetiology
General properties of EHV-1 and EHV-4
Equid herpesvirus 1 and EHV-4 are members of the Alphaherpesvirinae subfamily of herpesviruses of the domestic horse and are now included in the genus Varicellovirus, together with several other alphaherpesviruses of veterinary importance (e.g. bovine herpesvirus 1 and 5, caprine herpesvirus 1, equid herpesvirus 3, felid herpesvirus 1 and suid herpesvirus 1).78, 196 Their size, virion architecture, and overall replicative strategy are similar to those shared by all herpesviruses. Equid herpesvirus 1 and EHV-4 are closely related to one another but are antigenically, genetically, and pathogenetically distinct from equid herpesvirus 2 (EHV-2), EHV-3, and EHV- 5.8, 20, 73, 127, 218 They share no neutralizing or protective epitopes and only minor genetic homology with these three other herpesviruses of horses.184, 185, 250
Equid herpesvirus 1 and EHV-4 are environmentally labile, and infectivity is quickly destroyed by lipid solvents, detergents, heat, and the common disinfectants available for veterinary use.94 Virus survival outside of the horse is generally thought to be of short duration, although the extracorporal viability of EHV-1 at ambient temperature has been experimentally demonstrated to be as long as seven days when dried on paper, wood or rope, and 35 days on burlap or horsehair.94
Table 76.1 Known herpesviruses of Equidae
| SUBFAMILY OF HERPESVIRIDAE | EQUUS SPECIES | |||
| DOMESTIC HORSE (Equus caballus) | DONKEY (Equus asinus) | ZEBRA (Equus grevyi) | ONAGER (Equus hemionus onager) | |
| ALPHAHERPESVIRINAE | ||||
| a. Viscerotropic subgroup | Equine herpesvirus 1a (Equid herpesvirus 1)b | Asinine herpesvirus 3 (Equid herpesvirus 8) | Zebra herpesvirus isolates | Onager herpesvirus isolates |
| Equine herpesvirus 4 (Equid herpesvirus 4) | ||||
| b. Dermatotropic subgroup | Equine herpesvirus 3 (Equid herpesvirus 3) | Asinine herpesvirus 1 (Equid herpesvirus 6) | ||
| GAMMAHERPESVIRINAE | Equine herpesvirus 2 (Equid herpesvirus 2) | Asinine herpesvirus 2 (Equid herpesvirus 7) | ||
| Equine herpesvirus 5 (Equid herpesvirus 5) | ||||
a Viruses located in the same horizontal row of the table represent closely related equid herpesviruses exhibiting minor genetic and antigenic divergence induced by natural adaptation of a common progenitor to different equid species
b Virus names in parentheses are genus designations assigned by the Herpesvirus Study Group of the International Committee on Taxonomy and Nomenclature of Viruses (ICTV).78, 196 No ICTV designations have at this date been assigned by the Study Group to the zebra or onager herpesviruses78 A neurotropic herpesvirus isolate from captive gazelle closely related to equid herpesvirus 1 has provisionally been designated equid herpesvirus 978, 111, 227
The natural host range of EHV-4 is restricted to horses, while EHV-1 occasionally infects domestic cattle and captive camelids and cervids.66, 76, 193 Equid herpesvirus 1, but not EHV-4, can infect laboratory mice and be adapted to replicate in hamsters.13, 14, 92
Genome structure and gene functions
The complete nucleotide sequences of the genomic DNAs of both EHV-1 and EHV-4 have recently been determined.229, 230 EHV-1 and EHV-4 genomes are linear, double-stranded DNA molecules 150,2 and 145,6 kbp in size with base compositions of 56,7 and 50,5 per cent G+C, respectively. The genome of both EHV-1 and EHV-4 consists of a long unique region (UL, 112,870 and 112,398 bp, respectively) flanked by a small inverted repeat sequence (TRL/IRL, 32 and 27 bp) and covalently linked to a short unique region (US, 11,861 and 12,789 bp) that is flanked by a large inverted repeat (TRS/IRS, 12,714 and 10,178 bp). By inversion, the US segment of the genome can exist in two orientations, giving rise to virion populations containing equimolar amounts of two isomeric forms of viral DNA. Both EHV-1 and EHV-4 genomes contain 76 unique open reading frames (ORFs), each with a positional and sequence counterpart in the genome of the heterologous equine herpesvirus. The spatial arrangement of the 76 ORFs is illustrated in (Table 76.2). General features of the gene layout include compactly arranged ORFs with little intervening sequence, the absence of extensive ORF overlap, and few instances of exon splicing.
Four ORFs in the EHV-1 genome (#64, 65, 66, and 67) and three in the EHV-4 genome (#64, 65, and 66) are duplicated in the repeat sequences that flank the US region. Equid herpesvirus 1 and EHV-4 share a common gene arrangement with other sequenced alphaherpesviruses. The two equine alphaherpesvirus genomes, however, encode five genes (#1, 2, 67, 71, and 75) that have no structural homologues in any of the herpesviruses sequenced to date. The functions of these unique genes are unknown, but their products are predicted to play a role in the distinctive biology of EHV-1 and EHV-4 that has allowed them to adapt to the horse as their natural host. As in other alphaherpesviruses whose genome sequences have been determined, EHV-1 and EHV-4 genomes contain several sets of short, tandemly reiterated DNA sequences. Most of these are located in non-coding regions, in which the copy number of each reiterated element varies among virus strains and clonal isolates of the same virus strain, giving rise to restriction fragment length polymorphism in those DNA restriction fragments that encompass the reiterated sequences.
The precise genomic locations of EHV-1 and EHV-4 ORFs and the predicted functions of their expressed translation products are listed in (Table 76.2). 229, 230
Most of the 76 viral gene products fall into four functional categories:
- structural polypeptides of the nucleocapsid and tegument;
- transmembrane glycoproteins associated with the virion envelope;
- transactivators and transcriptional regulatory proteins; and
- a large array of viral proteins required for DNA replication and packaging, virion morphogenesis, and the extracellular egress of nascent progeny virions.155, 169
In their natural equine host, EHV-1 and EHV-4 are genetically stable viruses. Comparisons of the genomes of viral isolates from different geographical locations over a timespan of 50 years have demonstrated the existence of only a low level of intratypic sequence variation for EHV-1, and a slightly greater genetic variability among strains of EHV-4.11, 112, 152, 168, 175, 222, 224, 225, 238, 257 Isolates of EHV-1 recovered from 148 epidemically unrelated outbreaks of equine abortion were differentiated by analysis with five restriction endonucleases into only 16 distinct electropherotypes.11 On the other hand, laboratory adaptation of EHV-1 to growth in the Syrian hamster or repetitive passage of the virus in cell lines of nonequine origin leads to the rapid appearance of multiple genomic alterations.9, 126, 132, 138, 157, 225
Individual field isolates of EHV-1 and EHV-4 exhibit wide variations in virulence for horses.164, 178 Hypervirulent EHV-1 strains, such as Ab4 and Army-183, are highly endotheliotropic and, after experimental inoculation of horses, cause high rates of abortion and neurological disease.104, 164 The genetic basis for such differences in virulence is unknown.
EHV-1 and EHV-4 are closely related, with nucleotide sequence identity within individual homologous genes ranging from 55 to 84 per cent and amino acid sequence identity ranging from 55 to 96 per cent.229, 230 Sequence information therefore substantiates the view that EHV-1 and EHV-4 are two closely related but distinct herpesviruses of the horse. The existing genetic differences between EHV-1 and EHV-4 are, however, sufficient to give rise to major biological differences in their antigenicity, restriction endonuclease cleavage patterns, host range, and level of virulence for the horse.
Viral proteins and antigenic characteristics
Purified virions of EHV-1 or EHV-4 are structurally complex and contain as many as 30 individual polypeptides.156, 180, 234, 235, 236 Six of these polypeptides form the structural architecture of the core and nucleocapsid (ORFs 22, 25, 35, 42, 43, and 56), and 12 others are located within the amorphous tegument of the virion particle (ORFs 11, 12, 13, 14, 15, 23, 24, 40, 46, 49, 51, and 76). Embedded in the virion envelope are at least 11 glycosylated polypeptides, each with a positional and presumably functional counterpart in herpes simplex virus. Among the most abundant proteins in EHV-1 and EHV-4 virions are the major capsid protein (ORF 42); the tegument protein encoded by ORF 13 (gp10); and the five highly abundant glycoproteins (gB, gC, gD, gM, and gp300).
The envelope glycoproteins of EHV-1 and EHV-4 play important roles in viral replication by mediating virus attachment to and entry into cells and are critical determinants of tropism, cell-to-cell spread, pathogenesis, and the induction of host humoral immune responses. They have been shown to play a major role in the immunogenicity of EHV-1 and EHV-4, serving as major targets for the neutralization of virus infectivity by antibodies.33, 34, 173, 177, 204 Studies have identified glycoproteins B, C, and D as immuno-dominant antigens for generating antiviral serological responses to EHV-1 and EHV-4 in infected horses.6
Five EHV-1 glycoproteins (gB, gD, gH, gL, and gK) are essential for replication of the virus. Six others (gC, gE, gG, gI, gM, and gp300) are not required for viral growth in cell culture and have been termed ‘non-essential’. Because the non-essential envelope glycoprotein genes are maintained in virulent field isolates of EHV-1 and EHV-4 and their deletion results in a strong reduction of virulence,150, 151 they are believed to be important in the viral infectious processes within their equine host. It has been demonstrated, for example, that the EHV-1 gE/gI glycoprotein complex is necessary for efficient intra-host virus transmission by cellto-cell spread150 and that virus devoid of either gM or gp300 is severely impaired in the egress of enveloped progeny virions.198
The overall electrophoretic profiles of the structural virion proteins and glycoproteins of EHV-1 and EHV-4 are similar but not identical (Figures 76.1). Stable intertypic differences exist in their viral protein electrophoretic mobilities that can be used to differentiate the two viruses.234 Thirty EHV-1 polypeptides have been identified within infected cells.60 This striking repertoire of EHV-1 encoded proteins includes a group of regulatory proteins (e.g. gene transactivators and modulators, transcriptional regulators, host shut-off factor and post-translational regulators of viral gene expression), a number of DNA replicative enzymes (including DNA polymerase, ribonucleotide reductase, helicase-primase and thymidine kinase), proteins that function in virion morphogenesis and egress, and a large group of virion structural proteins.155, 169, 229, 230 During lytic infection with EHV-1, the synthesis of infected cell polypeptides (ICPs) is temporally regulated resulting in the sequential expression of immediate-early, early, and then late viral proteins.59, 123, 143, 216 The immediate-early and early viral proteins include transcriptional regulators and replicative factors, while most of the late EHV-1 ICPs are virion structural proteins.
The product of ORF 12 of both EHV-1 and EHV-4 (alphaTIF) is a potent transactivator of immediate-early gene expression.106, 144, 190 During viral entry, alpha-TIF is carried into the infected cell as a tegument protein and is required for efficient initiation of the lytic replicative cycle of the virus.144 Equid herpesvirus 1 encodes only a single protein whose expression requires no prior de novo viral protein synthesis.216, 217 The gene for this IE protein (ORF 64) is diploid and present in each of the sequence repeats that bracket the US region of the genome. IE is synthesized immediately after infection and is a potent activator of the transcription of all other EHV-1 genes. Three additional regulatory EHV-1 proteins, homologues of herpes simplex virus ICP27, ICP0 and ICP22, and encoded by EHV-1 ORFs 5, 63, and 65, respectively, are expressed as early proteins and act co-operatively with IE to modulate early and late viral gene expression.28, 29, 256 Of the latter regulatory EHV-1 polypeptides, gene 63 is not required for replication in vitro or in vivo nor is its presence necessary for either the establishment of or reactivation from latency in the horse. 133, 191
The close antigenic relationship between EHV-1 and EHV-4 is clearly apparent by their strong antigenic cross-reactivity in any immunological assay that uses polyclonal serum as the source of antibodies (e.g. virus neutralization, ELISA, complement fixing, immunoblot, or immunoprecipitation assays). In immunoblot assays with polyclonal antiserum, antigenic cross-reactivity between EHV-1 and EHV-4 can be demonstrated for most of the immunoreactive viral proteins present on the blot.234 In young horses, a partial level of post-infection cross-protection from heterologous virus challenge has also been noted for EHV-1 and EHV-4.5, 100, 109, 220 Using monoclonal antibodies, it has been shown that most of the identified glycoproteins of EHV-1 and EHV-4 possess both type-specific and type common serological epitopes.68, 255
Table 76.2 Features of EHV-1 and EHV-4 genes and gene productsa
| GENE | EHV-1 | EHV-4 | IDENTITY (%) | CHARACTERISTIC OF GENE PRODUCT | ||||
| START | STOP | CODONS | START | STOP | CODONS | |||
| 1 | 1298 | 1906 | 202 | 944 | 1543 | 199 | 72,2 | |
| 2 | 2562 | 1945 | 205 | 2206 | 1580 | 208 | 71,7 | |
| 3 | 2841 | 3614 | 257 | 2457 | 3227 | 256 | 74,6 | |
| 4 | 4249 | 3647 | 200 | 3864 | 3262 | 200 | 87,0 | |
| 5 | 5874 | 4462 | 470 | 5484 | 4081 | 467 | 77,3 | Regulator of gene expression |
| 6 | 7042 | 6011 | 343 | 6649 | 5618 | 343 | 89,5 | Envelope glycoprotein (gK) |
| 7 | 7042 | 6011 | 343 | 6649 | 5618 | 343 | 89,5 | Envelope glycoprotein (gK) |
| 8 | 10301 | 7056 | 1081 | 9900 | 6658 | 1080 | 80,2 | DNA helicase-primase |
| 9 | 12115 | 11135 | 326 | 11702 | 10722 | 326 | 77,6 | Deoxyuridine triphosphatase |
| 10 | 12084 | 12386 | 100 | 11671 | 11973 | 100 | 88,0 | |
| 11 | 12549 | 13463 | 304 | 12128 | 13042 | 304 | 78,0 | Tegument protein |
| 12 | 13595 | 14944 | 449 | 13173 | 14519 | 448 | 86,6 | Transactivator of IE gene promoter |
| 13 | 15317 | 17932 | 871 | 14890 | 17484 | 864 | 85,3 | Tegument protein |
| 14 | 18083 | 20326 | 747 | 17633 | 19864 | 743 | 79,0 | Tegument protein |
| 15 | 21170 | 20487 | 227 | 20698 | 20018 | 226 | 69,0 | Virus egress |
| 16 | 22851 | 21445 | 468 | 22396 | 20939 | 485 | 81,4 | Envelope glycoprotein (gC) |
| 17 | 24234 | 23029 | 401 | 2378 6 | 22575 | 403 | 84,3 | |
| 18 | 25696 | 24479 | 405 | 25222 | 24002 | 406 | 83,5 | DNA polymerase |
| 19 | 26262 | 27755 | 497 | 25765 | 27255 | 496 | 84,5 | Host shut-off factor |
| 20 | 28859 | 27894 | 321 | 28324 | 27362 | 320 | 89,7 | Ribonucleotide reductase |
| 21 | 31276 | 28904 | 790 | 30732 | 28363 | 789 | 88,3 | Ribonucleotide reductase |
| 22 | 32916 | 31519 | 465 | 32355 | 30967 | 462 | 91,8 | Capsid protein |
| 23 | 33292 | 36354 | 1020 | 32712 | 35777 | 1021 | 89,1 | Tegument protein |
| 24 | 36588 | 46853 | 3421 | 36006 | 46610 | 3534 | 78,7 | Tegument protein |
| 25 | 47311 | 46952 | 119 | 47068 | 46709 | 119 | 88,2 | Capsid protein |
| 26 | 48230 | 47403 | 275 | 47980 | 47156 | 274 | 90,9 | |
| 27 | 48791 | 48369 | 140 | 48543 | 48124 | 139 | 79,1 | DNA packaging protein |
| 28 | 48763 | 50625 | 620 | 48515 | 50365 | 616 | 80,0 | DNA packaging protein |
| 29 | 50618 | 51598 | 326 | 50358 | 51338 | 326 | 91,7 | |
| 30 | 55184 | 51522 | 1220 | 54924 | 51262 | 1220 | 88,3 | DNA polymerase |
| 31 | 55453 | 59082 | 1209 | 55178 | 58804 | 1208 | 90,6 | S-stranded DNA-binding protein |
| 32 | 59243 | 61570 | 775 | 58964 | 61285 | 773 | 88,0 | DNA packaging protein |
| 33 | 61432 | 64374 | 980 | 61147 | 64074 | 975 | 89,6 | Envelope glycoprotein (gB) |
| 34 | 64578 | 65060 | 160 | 64268 | 64750 | 160 | 73,0 | |
| 35 | 67093 | 65153 | 646 | 66770 | 64827 | 647 | 87,8 | Capsid protein |
| 35,5 | 66142 | 65153 | 329 | 65822 | 64827 | 331 | 83,9 | Capsid scaffold protein |
| 36 | 68975 | 67212 | 587 | 68648 | 66885 | 587 | 90,3 | DNA packaging protein |
| 37 | 69897 | 69079 | 272 | 69567 | 68749 | 272 | 83,5 | |
| 38 | 69910 | 70968 | 352 | 69582 | 70640 | 352 | 88,9 | Thymidine kinase |
| 39 | 71192 | 73738 | 848 | 70858 | 73425 | 855 | 85,7 | Envelope glycoprotein (gH) |
| 40 | 76224 | 74632 | 530 | 75832 | 74243 | 529 | 86,6 | Tegument protein |
| 41 | 76793 | 77512 | 239 | 76399 | 77112 | 237 | 87,8 | |
| 42 | 77703 | 81832 | 1376 | 77301 | 81428 | 1375 | 96,4 | Capsid protein |
| 43 | 82083 | 83027 | 314 | 81661 | 82605 | 314 | 94,6 | Capsid protein |
| 44 | 84320 | 83148 | 734 | 83875 | 82703 | 734 | 93,7 | DNA packaging protein |
| 47 | 88917 | 87886 | 88469 | 87438 | ||||
| 45 | 84480 | 86600 | 706 | 84037 | 86157 | 706 | 83,3 | |
| 46 | 86620 | 87732 | 370 | 86176 | 87285 | 369 | 85,9 | Tegument protein |
| 48 | 88947 | 89900 | 317 | 88499 | 89464 | 321 | 74,1 | |
| 49 | 89369 | 91153 | 594 | 88921 | 90717 | 598 | 85,8 | Tegument protein |
| 50 | 91135 | 92832 | 565 | 90699 | 92396 | 565 | 88,7 | Deoxyribonuclease |
| 51 | 92784 | 93008 | 74 | 92348 | 92575 | 75 | 84,7 | Tegument protein |
| 52 | 94472 | 93120 | 450 | 94033 | 92681 | 450 | 86,7 | Envelope glycoprotein (gM) |
| 53 | 94390 | 97053 | 887 | 93951 | 96614 | 887 | 91,1 | Origin-binding protein |
| 54 | 97069 | 99324 | 751 | 96626 | 98881 | 751 | 79,2 | DNA helicase-primase |
| 55 | 100332 | 99421 | 303 | 99850 | 98942 | 302 | 84,4 | |
| 56 | 102391 | 100130 | 753 | 101891 | 99648 | 747 | 89,4 | Capsid protein |
| 57 | 102375 | 105020 | 881 | 101875 | 104517 | 880 | 92,6 | DNA helicase-primase |
| 58 | 105070 | 105747 | 225 | 104567 | 105250 | 227 | 83,9 | |
| 59 | 106416 | 105877 | 179 | 105918 | 195358 | 186 | 69,5 | |
| 60 | 107116 | 106478 | 212 | 106606 | 105971 | 211 | 89,1 | |
| 61 | 108144 | 107206 | 312 | 107640 | 106696 | 314 | 78,5 | Uracil-DNA glycosylase |
| 62 | 108843 | 108147 | 218 | 108296 | 107637 | 219 | 74,2 | Envelope glycoprotein (gL) |
| 63 | 111985 | 110387 | 532 | 111713 | 110103 | 536 | 62,3 | Regulator of gene expression |
| 64 | 118591 | 114128 | 1487 | 117422 | 113093 | 1442 | 84,2 | Regulator of gene expression |
| 144569 | 149032 | 140628 | 144956 | |||||
| 65 | 121368 | 122249 | 293 | 120069 | 120923 | 284 | 85,2 | |
| 141792 | 140911 | 137981 | 137127 | |||||
| 66 | 122862 | 123572 | 236 | 121447 | 122133 | 228 | 80,3 | |
| 140298 | 139588 | 136603 | 135917 | |||||
| 67 | 125194 | 124376 | 272 | 123491 | 122631 | 286 | 68,0 | |
| 137966 | 138784 | |||||||
| 68 | 126275 | 125019 | 418 | 124559 | 123585 | 324 | 60,0 | |
| 69 | 126411 | 127559 | 382 | 124695 | 125849 | 384 | 86,9 | Serine-threonine protein kinase |
| 70 | 127681 | 128916 | 411 | 125970 | 127277 | 435 | 72,2 | Envelope glycoprotein (gG) |
| 71 | 129097 | 131490 | 797 | 127455 | 129707 | 750 | 61,1 | Envelope glycoprotein |
| 72 | 131583 | 132791 | 402 | 129798 | 131006 | 402 | 76,6 | Envelope glycoprotein (gD) |
| 73 | 132899 | 134173 | 424 | 131111 | 132373 | 420 | 74,0 | Envelope glycoprotein (gI) |
| 74 | 134406 | 136058 | 550 | 132 593 | 134239 | 548 | 85,4 | Envelope glycoprotein (gE) |
| 75 | 136055 | 136447 | 130 | 134273 | 134605 | 110 | 59,3 | |
| 76 | 136783 | 137442 | 219 | 134911 | 135573 | 220 | 54,9 | Tegument protein |
a Data from references # 229 and # 230, with permission of the publishers

Figure 76.1 SDS-PAGE electrophoretic profiles of the proteins and glycoproteins from purified virions of EHV-1 and EHV-4. Reproduced from reference # 234 by courtesy of the author

Figure 76.2 Diagrammatic model illustrating (a) the route(s) proposed for establishment (solid lines) of EHV-1 and EHV-4 latency in either the trigeminal ganglion (green lines) or lymphocytes (lavender lines), and (b) potential routes and consequences following reactivation (broken lines) of latent virus from either of these anatomic sites
An exception is glycoprotein G, which elicits only a type-specific serological response in the horse.69, 70, 72 Immunological tests (e.g. immunofluorescence) using monoclonal antibodies that recognize type-specific viral epitopes provide a convenient method for laboratory differentiation of EHV-1 and EHV-4 isolates.255
There is no evidence for the existence of significant antigenic variants of EHV-1 or EHV-4. Isolates of both EHV-1 and EHV-4 comprise a single neutralizing serotype. The existence of very minor intratypic antigenic variability, detectable only by detailed analysis with large panels of monoclonal antibodies, among EHV-1 or EHV-4 isolates indicates that each of the herpesvirus types comprises an antigenically stable and relatively homogeneous group.5
Alphaherpesviruses of equids other than horses
Herpesviruses closely related to EHV-1 have been isolated from several members of the Equidae family other than the domestic horse, e.g. zebras (Equus grevyi), donkeys (Equus asinus) and onagers (Equus hemionus onager) (Table 76.1).24, 36, 71, 159, 179, 253 Genetic and antigenic comparisons of such non-domestic horse isolates have demonstrated their close relationship with one another and with EHV-1. Each of these non-horse, equid alphaherpesviruses reacts with a panel of EHV-1 specific monoclonal antibodies that fail to react with EHV-4. It has been surmised, on the basis of the very close genetic and antigenic relationship between EHV-1 and the alphaherpesviruses of donkeys, zebras, and onagers, and because of the increased pathogenic potential of EHV-1 for horses relative to EHV-4, that the progenitor of EHV-1 was acquired by the ancestor of the modern horse in relatively recent evolutionary times by interspecies transmission from another equine species and has not yet achieved full pathogenic equilibrium with its new equine host.71
In several instances, disease caused by a herpesvirus indistinguishable from EHV-1 has been documented in domestic cattle (abortion) and in llamas and alpacas (optic nerve neuropathy and blindness).66, 75, 131, 193 In other instances, infection and clinical disease have been reported in non-equine animal species, such as captive gazelle and antelope (encephalitis), caused by equid herpesviruses closely related to, but distinguishable from, EHV-1.66, 111, 136, 227 Monoclonal antibody characterization and restriction endonuclease analysis of the DNA of herpesvirus isolates from the latter group of non-equids suggest that such infections represent rare instances of species cross-over of the herpesvirus from its natural equine host. The true equine host species for each of these EHV-1-like virus isolates and the extent of their transmission to, and circulation within, non-equids are unknown.
Epidemiology
Surveys to estimate the seroprevalence of EHV-1 and/or EHV-4 infection in several equine populations have demonstrated the existence of antibodies in most adult horses as well as in other equine species.1, 16, 18, 25, 43, 53, 69, 79, 89, 118, 152
The epidemiological life-styles of both EHV-1 and EHV-4 are characterized by:
- the widespread infection of young, susceptible horses with overall low clinical morbidity;
- a high prevalence of latently infected carrier horses; and
- the frequent shedding of infectious virus from such carrier animals that allows efficient and uninterrupted transmission of virus to new generations of equids.
Epidemiologic reservoir for EHV-1 and EHV-4 infections
During the period immediately following primary infection by EHV-1 or EHV-4, poorly understood events occur within the horse that result in latent infection of nearly all recovered animals, which then become life-long carriers of the virus.101, 105, 206, 207, 243 This capacity of EHV-1 and EHV-4 to persist in the body of the horse in a dormant, but potentially reactivatable, state after recovery from primary infection provides a biological reservoir of the two viruses for continuous transmission of infection. In 40 horses killed in an abattoir and examined by co-cultivation, 60 per cent harboured latent EHV-1 or EHV-4 in the lymph nodes draining the respiratory tract.105 Approximately one-third of the positive animals harboured both viruses in a latent state. This ubiquitous distribution of horses latently infected with these two viruses has been confirmed by PCR-based detection of EHV-1 and/or EHV-4 DNA in the tissues of a large proportion of adult horses.27, 105, 206, 243 The world’s 80 million plus horses that carry the latent viruses thus serve as an inexhaustible reservoir of virus for the perpetuation and transmission of EHV-1 and EHV-4 infections and provide the basis for the unique epidemiology of disease caused by EHV-1 and EHV-4.
Transmission of EHV-1 and EHV-4
For both EHV-1 and EHV-4 the initial portal of viral entry into the horse is the upper respiratory tract following contact with virus-laden respiratory secretions, fomites, or aerosols. Transmission of virus to susceptible animals occurs either from virus-shedding horses with acute or reactivated EHV respiratory infections or from contact with an aborted foetus or its placenta, which are rich in infectious virus.50 Environmental shedding of infectious EHV-1 or EHV-4 from the equine respiratory tract is both efficient and prolonged. Naive horses exposed for the first time to EHV-1 or EHV-4 may release infectious progeny virus into the respiratory mucus for as long as 15 consecutive days following infection.114 In such primary infections, the magnitude of virus present in nasal mucus may be as great as 106 pfu/ swab for both EHV-1 and EHV-4.51 Shedding of virus from the respiratory tract of susceptible horses with previous virus exposure or after reactivation from latency is more transient (two to four days) and reduced in magnitude (102 to 105 pfu/swab).52 Both viruses are highly contagious, with infection rates approaching 100 per cent in susceptible incontact cohorts.
Recent type-specific sero-epidemiological investigations into the dynamics of inter-generation transmission of EHV-1 and EHV-4 have demonstrated the early and widespread acquisition of infection by foals during their first year of life.103, 117–121 Natural infection is often not accompanied by recognizable clinical signs. Foals may experience the first EHV-1 or EHV-4 infection either before or after weaning; EHV-1 infection of foals as young as 30 days has been documented.
Because the infection of unweaned foals is often accompanied by evidence of simultaneous viral replication in their dams, the most likely source of infection for foals prior to weaning is reactivated latent infection of a mare, or mares, with subsequent spread to susceptible foals. The infection rate in foals varies from year to year and among different studs but, by one year of age, 35 to 60 per cent of young horses have been exposed to one or both herpesviruses. Overall, the results suggest a cyclic and mostly silent epidemiological pattern of endemic EHV-1 and EHV-4 infection, with mares serving as the source of infectious virus for transmission to each new crop of foals during the preweaning and/or weaning periods.
Establishment of latency
After the initial lytic phase of infection in the equine respiratory tract, EHV-1 and EHV-4 enter a latent state in lymphocytes, both circulating and those in draining lymph nodes, as well as in sensory nerve-cell bodies within the trigeminal ganglia (Figures 76.2).27, 35, 53, 105, 206, 207, 243 Latent EHV-1 is harboured by CD8+ T lymphocytes, and the frequency of latently infected cells has been estimated to be 1 in 50 000 peripheral blood mononuclear cells (PBMC).62 As has been described for other alphaherpesviruses, transcription from the latent EHV-1 or EHV-4 genome is restricted. Only transcripts (LATs) antisense to either the immediate-early viral gene (ORF 64) or a regulatory early gene (ORF 63) accumulate in latently infected cells.21, 27, 62
The molecular and physiological mechanisms by which EHV-1 and EHV-4 enter into a latent relationship with their equine host cells are unknown. The infected lymphocyte population during the first two weeks following inoculation of horses with EHV-1 is dominated by ‘viraemic’ cells from which infectious virus can be readily recovered by in vitro co-cultivation with permissive cells.37, 114, 164, 202, 206 In viraemic lymphocytes, the EHV-1 genome is transcriptionally active and undergoes a lytic pattern of transcription, fusogenic viral glycoproteins are expressed and delivered to the cell surface, and the virus-containing lymphocyte is subject to immune destruction. Completely assembled and infectious EHV-1 virions, however, are not present in viraemic lymphocytes (abortive infection), but are quickly formed upon virus glycoprotein-mediated fusion of the lymphocytes with permissive cells both in vitro during co-cultivation and in vivo during lymphocyte adhesion to and fusion with endothelial cells.
Between the peak of lymphocyte-associated EHV-1 viraemia at four to ten days post-infection and the truly latent infection of lymphocytes several weeks later, a transition ensues within the virus-carrying lymphocyte population that leads to one that is dominated by latently, rather than abortively, infected cells (Figures 76.3).206 In lymphocytes harbouring latent EHV-1, viral proteins are not expressed and the latently infected cell is resistant to immune clearance mechanisms.62 Recovery of infectious virus from latently infected lymphocytes, in contrast to abortively infected lymphocytes, is difficult and requires prolonged cocultivation with numerous passages of the cultured cells.105, 206, 243 It is not known whether, during the post-infection transition into latency, there is loss of the viraemic lymphocytes with retention of the less frequent, latently infected cells or, alternatively, a small subset of viraemic cells undergoes conversion to a latent state in which the biological activity of the resident viral genome is much reduced.
Latent EHV-1 and EHV-4 can be detected by prolonged co-cultivation of permissive indicator cells with cells collected from lymph nodes draining the respiratory tract, circulating lymphocytes, and trigeminal ganglia.105, 206, 243 Latent viral DNA can be detected in these same tissues by PCR,27, 105, 206, 207, 243 and EHV-1 LAT RNA has been detected in the PBMC of latently infected horses by reverse transcription (RT)-PCR.62
Recrudescence
Periodically, horses harbouring latent EHV-1 and EHV–4 experience episodes during which infectious virus is reactivated from its quiescent state and shed into respiratory tract secretions with the potential for infecting other, susceptible horses (Figures 76.2). Reactivation of latent EHV-1 and EHV-4 infections from horses has been observed in field situations following transport, handling, re-housing and weaning, and reactivation has been achieved experimentally by treatment of horses with corticosteroids.35, 53, 101, 206 It is likely that the stresses imposed upon the horse by modern, intensive management practices, especially in racing and competition animals, result in frequent reactivation of latent EHV-1 and EHV-4 infections. Importantly, clinically apparent respiratory disease is often absent following reactivation, and such horses are therefore silent virus shedders.101This scenario presents obvious management difficulties as new cases of EHV infection and disease may develop in closed, isolated groups of horses that have had no contact with animals experiencing overt respiratory disease.53
During the process of re-emergence of EHV-1 from latency in equine T lymphocytes, the steps of the transition in the lymphocyte population that originally led to the establishment of latency are reversed (Figures 76.3). In a small subset of the lymphocytes that carry the latent EHV-1 genome, an active lytic pattern of transcription of the viral DNA is revived, fusogenic viral glycoproteins are expressed and appear on the lymphocyte surface, and the cell undergoes a transition into an abortively infected lymphocyte from which infectious virus can be easily recovered by cocultivation.35, 101, 206 The precise molecular events resulting in activation of the immediate-early viral gene (ORF 64) with subsequent conversion to a lytic pattern of viral transcription are unknown. However, it is known that the EHV-1 IE gene promoter can be trans-activated by co-infection with EHV-2.192

Figure 76.3 Illustration of the virus-related changes that occur within the EHV-1 or EHV-4 infected equine lymphocyte population during its transition from a population dominated by abortively infected (viraemic) cells (left) to one dominated by latently infected cells (right)

Figure 76.4 Equine respiratory disease caused by EHV-1 or EHV-4. (a) nasal discharge, (b) vesiculation of mucosal epithelium of trachea, (c) neutrophilic exudation, and infected epithelial cells (brown) in nasal mucosa, (d) immunoperoxidase detection of viral antigen in mononuclear leukocytes within the medullary sinus of the retropharyngeal lymph node.
Reactivation of latent EHV-1 and EHV-4 from lymphocytes and/or trigeminal ganglia may result in the delivery of infectious virus to the epithelium lining the nasopharynx.35, 101, 206 Assembly and release of infectious virions from lymphocytes in which EHV-1 has been reactivated requires fusion with permissive cells. The capacity of viraemic lymphocytes to actively adhere to and fuse with permissive vascular endothelial cells of the horse provides a mechanism for the return of reactivated virus from its latent cellular repository to the respiratory epithelium as infectious virions(Figures 76.2). Depending on the local immune status of the nasopharyngeal epithelium, a lytic infection by EHV-1 or HV-4 may become established within the respiratory tract mucosa after a reactivation event, resulting in the environmental shedding of infectious virus.206
Cell-associated viraemia, at a level that can be easily detected by co-cultivation, can also develop as a consequence of reactivation of EHV-1 from latency within lymphocytes, either prior to or following infection of the nasopharyngeal epithelium. It is probably dependent upon amplification of reactivated virus in productively infected lymphoblasts within lymphoid tissues of the respiratory tract in a sequence of events analogous to those of primary infection (see Figures 76.2, and Pathogenesis below).101
With EHV-1, the cell-associated viraemia that follows reactivation may result in dissemination of infectious virus to the uterus to induce abortion or, potentially, to the central nervous system (CNS) to cause neurological disease. Another source of reactivated EHV-1 for causing abortion is from resident lymphocytes within the local environment of the pregnant endometrium that may transfer infectious virus directly to the uterine endothelium, thereby initiating the cascade of events leading to abortion. Therefore, EHV-1 abortion may occur without the prerequisite of a lytic respiratory epithelial infection or a detectable cell-associated viraemia. Equid herpesvirus 1 is the first member of the alphaherpesviruses for which reactivation from lymphocytes, in addition to reactivation from the trigeminal ganglion, has been described. Indeed, reactivation of EHV-1 appears to occur, in vitro at least, more readily from lymphocytes than from the trigeminal ganglion.62, 105, 210 Equid herpesvirus 4, by contrast, appears at present to resemble other varicelloviruses (e.g. bovine herpesvirus 1 and suid herpesvirus 1), with functional latency being established primarily in trigeminal ganglia.27, 35
Latency and reactivation play particularly important roles in the epidemiology of EHV-1 abortion. The majority of natural EHV-1 abortions affect single mares within a group,89 implying that abortion has resulted from reactivation of latent virus rather than from a newly acquired respiratory infection. Such reactivations of latent EHV-1 may also explain abortions that occur many weeks or months after termination of the cell-associated viraemia that follows experimental intranasal inoculation of the virus.88, 122, 161, 164, 66
Pathogenesis
Respiratory tract and associated tissues
Both EHV-1 and EHV-4 infect and replicate initially in mucosal epithelial cells of the upper respiratory tract following inhalation of infectious aerosols or contact with infected fomites.178 Equid herpesvirus 1 can also infect conjunctival epithelium, presumably via aerosol contact. By both immunoperoxidase staining and virus isolation techniques, infected epithelial cells can be detected in the nasopharynx, trachea and bronchi as early as 12 hours after experimental intranasal infection with virulent EHV-1.140 Unchecked progression of respiratory epithelial infection results in the formation of multiple erosions in the nasopharyngeal mucosa, with viral antigen expression in degenerating epithelial cells, local lymphocytes and monocytes, and in endothelial cells of nasal blood vessels (Figures 76.4).
Equid herpesvirus 1 quickly breaches the respiratory epithelium, spreading to cells in the underlying lamina propria so that, within 24 hours, infected mononuclear leukocytes can be detected in the sinuses and parenchyma of the respiratory tract-associated lymph nodes (Figures 76.4). 141 A significant secondary amplification of viral infection takes place within such draining lymph nodes with discharge of infected leukocytes, via the efferent lymph, into the bloodvascular circulation to result in leukocyte-associated viraemia. The viraemia disseminates EHV-1 infection to tertiary sites of replication, including the vascular endothelium of the pregnant uterus and CNS.
Infection of epithelial cells, endothelial cells and leukocytes in the lungs can be observed from day 2 to 13 postinfection, with a peak on day nine, when occasional nonocclusive thrombi may occur in the pulmonary interstitium.141
In the initial stages of nasal and conjunctival infection, EHV-1 gains access to neurones of the trigeminal nerve and reaches the trigeminal ganglion by 48 hours post-infection.207 Equid herpesvirus 1 is generally cleared from the respiratory tract within three weeks of primary infection and one to two weeks following subsequent infections.114, 164
The detailed pathogenesis of EHV-4 infections has not been elucidated but probably parallels that demonstrated for EHV-1 with infection of the respiratory tract and its associated lymphoid system. However, the pathogenicity, extent of viral replication and tissue destruction in horses caused by EHV-4 are far lower than those that occur in EHV-1 infections. In contrast to EHV-1 infection, detectable dissemination by EHV-4 generally does not extend beyond the local lymph nodes.178 Most infections with EHV-4 do not result in the level of infected endothelial cells or cell-associated viraemia that is required to precipitate abortion and neurological disease. However, cell-associated EHV-4 viraemia can be detected,152 and EHV-4 infection of endothelial cells has been demonstrated in aborted equine foetuses as well as in a natural case of pneumonia caused by EHV-4 in a foal.23, 247 Equid herpesvirus 4 infections are generally cleared from the respiratory tract within 7 to 20 days after first infection and within two to seven days after subsequent infections.
Leukocyte-associated viraemia
The two clinically important sequelae of EHV-1 respiratory infection, namely abortion or a neurological syndrome, require a leukocyte-associated viraemia that disseminates virus to sites distant from the respiratory tract, including the reproductive tract and CNS.37, 164, 206 With EHV-1, the viraemia involves primarily CD5+ /CD8+ T lymphocytes147, 154, 206, 210 and occurs as a consequence of their discharge into the lymphatic and blood circulations from secondary sites of EHV-1 replication in lymph nodes draining the infected respiratory tract.141 Smaller numbers of viraemic lymphocytes may also result from intravascular contact between uninfected circulating lymphocytes and infected vascular endothelial cells. The susceptibility of equine lymphocytes to infection by EHV-1 is enhanced by their mitogen- or antigen-induced activation to lymphoblasts.98, 237 Free virus (i.e. a plasma viraemia) is rarely detected in the blood. Once in the bloodstream, infected leukocytes adhere to endothelial cells, by as yet undefined receptors, in a process that may be facilitated by alterations in the expression/ presence of adhesion molecules, cytokines and hormones in the micro-environment.209
Following intranasal inoculation with EHV-1, approximately 85 per cent of susceptible mares in late gestation develop lymphocyte-associated viraemia, at levels detectable by co-cultivation.37, 38, 48, 54, 104, 164, 213 The cell-associated viraemia is a prerequisite for abortion. However, abortion does not necessarily follow viraemia, even in mares in the last third of gestation. Based on a literature survey, only 42 per cent of 156 mares infected in late gestation and that subsequently developed viraemia, aborted.37, 38, 48, 54, 104, 164, 213 In experimental cases, viraemia develops from day three post-infection and may persist for up to 22 days. Most pregnant mares artificially infected with EHV-1 develop viraemia between four and ten days after inoculation. The mean duration of lymphocyte-associated viraemia in mares experimentally inoculated with EHV-1 is around five days, and the interval between onset of viraemia and subsequent abortion can range from 6 to 81 days. Virus-infected leukocytes in the blood of horses with detectable EHV-1 viraemia may range from 1 in 104 to 1 in 107 leukocytes.164, 206 The magnitude rather than the duration of viraemia is correlated with the likelihood of abortion.164
Occasional EHV-4 infections result in detectable leukocyte-associated viraemia,152 but neither the duration nor the magnitude of viraemia has been measured. The phenotype of the leukocytes involved is also unknown.
Pregnant uterus and foetus
In the case of highly virulent isolates of EHV-1, such as Ab4 and Army 183, infection of endothelial cells in the pregnant uterus causes a vasculitis that particularly affects small arteriolar branches in the glandular layer of the endometrium at the base of the microcotyledons.104, 135, 214, 215 By immunoperoxidase staining, viral antigen is first detected in endothelial cells of these blood vessels at days six to eight following experimental infection of mares with the Ab4 EHV-1 isolate. The roles of immune and inflammatory processes and of activation of the coagulation cascade in mediating EHV-1-induced vascular pathology are thought to be important but have not been fully elucidated. Endothelial cell infection is widespread over days 9 to 13 post-infection, with associated multifocal vasculitis resulting in microthrombosis of affected blood vessels (Figures 76.5). This sometimes causes thrombo-ischaemic necrosis of the overlying microcotyledons and intercotyledonary stroma (Figures 76.5c). If these vascular lesions of the endometrium are widespread, the foetus may be aborted before detectable transplacental spread of virus has occurred.214 Abortion caused by EHV-1 challenge from which the virus was not recovered from the foetus had previously been assumed to be due to maternal stress or pyrexia.57, 122 The incidence of this type of abortion following field infections is unknown.
Following either experimental or natural EHV-1 infection, virus can usually be isolated from the aborted foetuses. In experimentally infected mares examined 14 to 21 days after infection, the extent of uterine vasculitis and associated microcotyledonary necrosis was less in those mares carrying or aborting virus-positive foetuses than in those aborting virus-negative foetuses.211, 215 In the mares carrying virus-positive foetuses, there were focal areas of microcotyledonary infarction that presumably caused disruption of the physical integrity of the uteroplacental barrier and permitted egress of free virus or virus-infected cells across the placenta into the foetal circulation (Figures 76.6).104, 200, 215 Prostaglandin release at the uteroplacental interface as a consequence of thrombosis may also be important in initiating the abortion process, but functional studies to investigate this hypothesis have not been undertaken. The susceptibility of uterine endothelial cells to infection with EHV-1 is lower in early pregnancy than in late pregnancy, and association of EHV-1 infection with early embryonic death and resorption has not been investigated.213
In EHV-1 positive foetuses, infected endothelial cells occur in blood vessels of the umbilical cord and allantochorion (Figures 76.5d), although intensive searching with the aid of immunohistochemistry is generally needed to visualize foci of infection in the placenta.104, 215 Endothelial cells are consistently infected in foetuses from which EHV-1 is recovered, and it is presumed that these cells act as primary sources for spread of virus to adjacent parenchymal cells. Experiments have not been undertaken to detect the occurrence of viraemia in foetuses infected with EHV-1, although immunoperoxidase-positive intravascular leukocytes are generally demonstrable in tissue sections of EHV-1 aborted foetuses.

Figure 76.5 Endometrium of pregnant mare after inoculation with EHV-1. (a) swollen, immunoperoxidase (IP)-positive endothelial cells, (b) thrombotic vasculitis with IP-positive endothelial cells, (c) infarction of placental microcotyledon, (d) allantochorionic villus of placenta of aborted foetus with IP-positive endothelial cells in villous core. Panels B and C reproduced from reference # 7 with permission of the publisher

Figure 76.6 (Right) Cellular ultrastructure of the utero-placental interface of the mare, reproduced from reference # 200 with permission of the publisher. (Left) Diagrammatic model of the proposed route of transmission of EHV-1 infection from the viraemic mare, across the utero-placental barrier, to the foetal circulation. Endometrial and placental cellular layers in which EHV-1 antigen has been demonstrated by immunoperoxidase staining are indicated by gold shading. The proposed sequence of extension of infection, via either cell-free virus or by cell-to-cell transfer of intracellular virus, from a viraemic lymphocyte in a capillary of the mare’s endometrium to a lymphocyte in the foetal circulation, is indicated by the arrows
The pathogenesis of abortion caused by less virulent isolates of EHV-1 is not as clear, as those isolates appear to have reduced affinity for endothelial cells. It has recently been demonstrated that an isolate of EHV-1 with low abortion potential was capable of detectable replication in endothelial cells of an immunologically naive foal but not in endothelial cells of immunocompetent adults.212 This observation suggests that host maturity or immunity, in addition to viral factors, may play roles in determining the extent of EHV-1 infection in endothelial cells. It is possible that, with less virulent abortigenic strains of EHV-1, the endometrial endothelium is infected at a level sufficient to allow occasional materno–foetal transfer of virus but not detection by immunohistochemical staining of uterine tissue. An alternative hypothesis for the pathogenesis of abortion caused by less virulent EHV-1 strains, not reliant on infection of endothelial cells, is that virus crosses the placenta as a latent infection in lymphocytes and thereby evades host immune responses. Investigation of this theory awaits the application of sensitive molecular techniques, such as in situ PCR, to identify cells harbouring latent viral sequences in the uterus and elsewhere.
The rare cases of abortion that occur following infection with EHV-4 are also likely to involve the capacity of certain isolates of EHV-4 for significant replication in uterine and/or foetal endothelial cells.247
Central nervous system
Infection of endothelial cells and the accompanying vasculitis are also central to the pathogenesis of the neurological syndrome caused by EHV-1.102, 134, 135, 178 Both field cases and experimentally infected horses with CNS disease caused by EHV-1 consistently develop vasculitis, with or without local haemorrhage and thrombo-ischaemic necrosis, in the brain and spinal cord (Figures 76.7). Equid herpesvirus 1 has only occasionally been isolated from the CNS of clinically affected horses,146, 199 but, with the use of immunohistochemistry, can be demonstrated in endothelial cells at the sites of vasculitis (Figures 76.7d). It is generally believed that the neurological deficits associated with CNS disease caused by EHV-1 are the result of ischaemic death of nervous tissue consequent to infection of endothelial cells and its accompanying thrombo-ischaemic vasculitis. The lack of definitive evidence for EHV-1 replication in equine neurones contrasts with the well-established pathogenesis of infectious encephalitis caused by neurotropic herpesviruses in other animal species. It has been suggested that immunopathologic events that accompany EHV-1 infection of vascular endothelial cells may play a role in the pathogenesis of EHV-1 CNS disease.251 While the expanded levels of virusspecific antibody and T-cell populations arising in response to infection may be central to clearing viral infection from the horse, they may, at the same time, be a part of the complex immuno-inflammatory response within the vasculature of the CNS and thus be actively involved in the genesis of the thrombo-ischaemic process that follows endothelial cell infection. Thus, the horse’s immune response itself may contribute to the pathogenesis of EHV-1 neurological disease.
Clinical signs
Respiratory disease
The incubation period for respiratory signs following experimental infection of horses with EHV-1 or EHV-4 is short (one to three days),113, 114, 115 although longer incubation periods of up to 10 days have been recorded in the field.174 Such variation in the appearance of clinical respiratory disease probably reflects differences in virus strain pathogenicity, infecting dose and host immunity. Infection of horses with either EHV-1 or EHV-4 results primarily in upper respiratory tract disease (rhinopharyngitis and tracheobronchitis).3 In previously infected horses, clinical respiratory signs may be of minimal severity and of short duration.3, 140, 141, 226 Respiratory infection may be completely asymptomatic in older horses, including pregnant mares, with multiple prior immunological experiences with either EHV-1 or EHV-4. This is also the case following reactivation of latent EHV-1 or EHV-4 during which infectious virus may be shed from the nasopharynx in the absence of clinical signs.
Following primary EHV-1 or EHV-4 infection of young, immunologically naive horses, however, overt respiratory disease develops which may be clinically severe and of considerable duration. In specific pathogen-free (SPF) foals undergoing primary experimental infection with the virulent Ab4 strain of EHV-1, there is a biphasic pyrexia, peaking on days one to two post-infection and again on days six to seven post-infection, with a combined duration of pyrexia of eight to ten days.113, 114, 115

Figure 76.7 Neurological manifestations of EHV-1 infection. (a) Ataxic mare supported in sling as part of nursing care, (b) macroscopic haemorrhagic lesions in spinal cord, (c) microscopic haemorrhage and axonal swelling in spinal cord, (d ) thrombo-occlusive vasculitis with immunoperoxidase-positive endothelial cells in the spinal cord. Panel b was reproduced from reference # 145 with permission of the publisher.

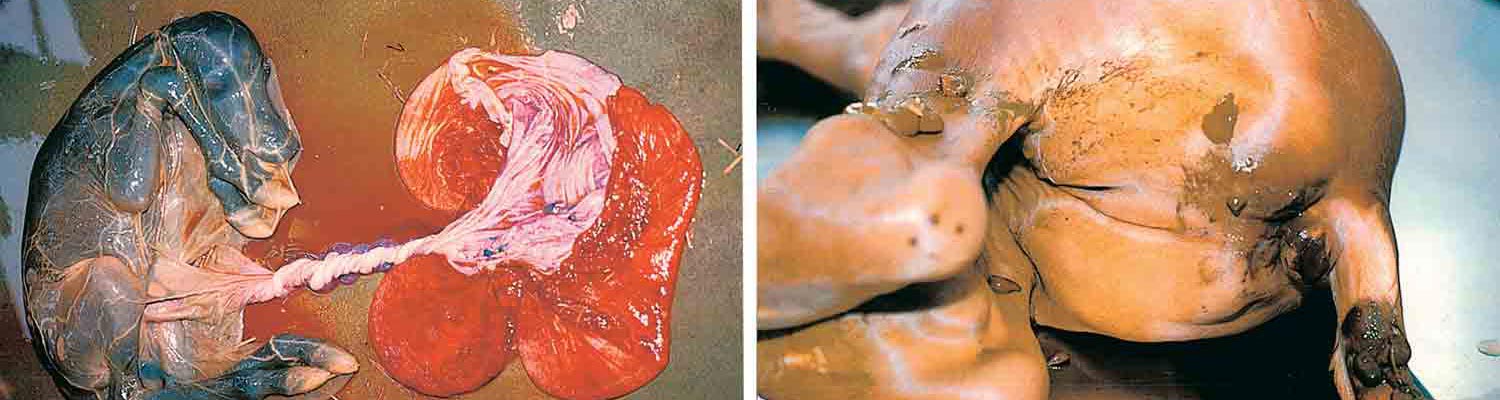
Figure 76.8 Macroscopic pathology of equine abortion caused by EHV-1 infection: (a) aborted foetus still attached to placenta and enclosed in amnion stained greenish-yellow with meconium, (b) dissection of aborted foetus showing pulmonary oedema and straw-coloured pleural effusion, (c) meconium staining of perineal region of aborted foetus, (d ) white subcapsular foci of necrosis on the hepatic surface of an aborted foetus
This is associated with moderate depression and anorexia. Initially, there is serous nasal discharge, conjunctivitis and serous ocular discharge. The character of the nasal discharge progresses rapidly to mucoid and then mucopurulent by day five to seven post-infection, which is usually attributed to secondary bacterial infection (Figures 76.4a). There is progressive lymphadenopathy, principally of the submandibular lymph nodes, although occasionally the retropharyngeal lymph nodes become sufficiently enlarged to become palpable.
Lymph nodes reach maximum size between seven to ten days postinfection and can remain enlarged for many weeks. A leukopenia, consisting of both lymphopenia and neutropenia, is present for several days after EHV-1 infection.95, 147, 154 Infected horses may occasionally cough. Experimental and field evidence suggest that the frequency, severity and duration of coughing are largely determined by management, especially stable air hygiene and whether the horse is adequately rested from training or performance activities.165 Foals that develop lower respiratory tract disease (bronchopneumonia) after virus infection are markedly depressed, tachypnoeic and dyspnoeic, lose interest in the mare and may stop suckling. Equid herpesvirus 4 causes upper respiratory tract disease which is clinically indistinguishable from that caused by EHV-167, 174 and, on occasion, can also cause viral bronchopneumonia.23
On recovery from upper respiratory tract disease caused by EHV-1 or EHV-4, some horses develop an ill-defined ‘poor performance syndrome’ which may be associated with non-specific bronchial hypersensitivity and a syndrome resembling chronic obstructive pulmonary disease.165 Thus, the economic losses associated with equine herpesvirus respiratory disease are associated, not only with the costs of veterinary care and lost training days during the acute stages of infection, but also with the longer-term detrimental effects on athletic performance.
Abortion
Mares infected with EHV-1 abort precipitously with no impending signs, and evidence of previous respiratory tract infection in the mare is usually not observed.43, 82, 85, 162, 214 The abortion may occur while the mare is still standing or very shortly after lying down. The placenta is usually expelled together with the foetus that is often still enveloped in its amniotic membrane (Figures 76.8).
At the time of abortion, the foetus has usually just died from asphyxia associated with the sudden separation of the placenta from the endometrium that precedes foetal expulsion. Some foetuses may be alive immediately after expulsion, but succumb quickly to respiratory insufficiency resulting from virus-induced pulmonary lesions. Equid herpesvirus 1 outbreaks resulting in multiple abortions (‘abortion storms’) can occur within a group of pregnant mares, and abortion case rates of up to 75 per cent have been recorded.166 However, most episodes of EHV-1 abortion within a group involve single mares only.89 Almost all EHV-1 abortions occur during the last four months of gestation89 and, in early gestation (up to 120 days), mares appear to be refractory to abortion following experimental infection.213 Once a mare has aborted, her future reproductive potential is usually unaffected; most mares conceive successfully shortly after the abortion and foal normally the following year. Mares rarely abort from EHV-1 infection in successive years,37 but may eventually become reinfected and abort again.
Occasionally, EHV-4 is the only virus isolated from an aborted foetus, and abortion can be induced by direct intrafoetal inoculation of the EHV-4 virus.11, 205, 223 The clinical presentation of such abortions is similar to that of EHV-1 abortion, but multiple abortion epidemics caused by EHV-4 have not been recognized.
Disease in neonatal foals
Occasionally, foals are born at term that are either obviously sick at birth or become ill within one to two days of parturition.55, 87, 128, 167, 182 Typically, the clinical progression of the disease is rapid and the outcome unaffected by intensive, supportive veterinary care. The foals fail to nurse, become lethargic, pyrexic, leukopenic, hypoxic, and exhibit severe respiratory distress and intractable diarrhoea. Thoracic radiographs show diffuse interstitial and alveolar densities. Congenital EHV-1 infection can be epidemic in nature and may occur in association with an outbreak of abortion or, more commonly, without concurrent abortion or respiratory disease in the dams. There is some debate about whether such foals are infected in utero with EHV-1 or acquire a rapid post-parturient infection from their dam. In either case, viral pneumonia quickly manifests, nearly always leading to respiratory failure and death within a few days. Equid herpesvirus 1 can be isolated from the foals’ lungs at necropsy. A more chronic variation of this typically acute neonatal foal syndrome has been described in which generalized lymphoid depletion is a dominant feature and death results, after a longer period (5 to 14 days), from a variety of secondary bacterial infections such as purulent bronchopneumonia, septicaemia and enteritis.45 In the latter syndrome, herpesvirus inclusion bodies were not observed and virus was not so readily isolated from the tissues.Rarely, EHV-4 may also cause neonatal disease resembling that caused by EHV-1.170
Disease in stallions
Equid herpesvirus 1 infection may also have reproductive consequences for stallions. Scrotal oedema and loss of libido have been reported in stallions during outbreaks of EHV-1 infection in the field,125, 153 and prolonged pyrexia would be expected to have a detrimental effect on spermatogenesis. Indeed, after experimental intranasal infection of a stallion, a reduction in the number of morphologically normal sperm was observed and infectious virus was shed into the semen.228 However, the importance of venereal shedding by stallions in the epidemiology of EHV-1 infections is uncertain.
Neurological disease
Neurological disease is a sporadic and uncommon, but potentially devastating, manifestation of EHV-1 infection and has been recognized clinically for many years.22, 58, 61, 65, 96, 125, 134, 139, 145, 146, 153, 199, 231, 233, 251 Neurological disease caused by EHV-4 is rare, but isolated cases have been identified.158, 238, 240 In initial reports, neurological disease associated with EHV-1 appeared to affect predominantly pregnant or lactating mares.134, 199 However, more recent observations of field outbreaks suggest that neurological disease caused by EHV-1 is not restricted by pregnancy, age or sex and can occur in foals, yearlings, geldings, stallions and both barren and in-foal mares.5, 125 The interval between initial EHV-1 infection of the respiratory tract and subsequent onset of neurological disease is usually between six and ten days, but neurological signs have been seen as early as one day following onset of pyrexia.125 Clinical signs are highly variable and depend on the extent and location of the neurological lesions, but usually appear suddenly and reach their peak intensity within two to three days of onset. The extent of neurological dysfunction ranges from temporary ataxia, proprioceptive deficiency, limb weakness, swaying, stumbling and falling, to complete paralysis. The neurological disorders affect mainly the hind limbs, although quadriplegia has been observed. Signs of bladder dysfunction or atony producing incontinence or urinary retention, and cutaneous perineal and limb sensory dysfunction have been recorded.125, 134 Some affected horses develop a head tilt. Neurological disease caused by EHV-1 often occurs as outbreaks, with up to 40 per cent of animals in the group affected. The prognosis for non-recumbent horses is favourable, but for animals that remain recumbent for longer than two days is poor. The latter cases usually develop fatal complications (e.g. pneumonia, colic or bladder rupture). Although 24-hour, intensive nursing care with the use of whole-body slings (Figures 76.7a) has been attempted in such cases, the prognosis for eventual return of severely affected horses to clinical soundness and original performance level is not particularly good.
Ocular disease
Equid herpesvirus 1 infection may also cause ocular disease, which manifests as uveitis or chorioretinal lesions. Uveitis was described in foals during an outbreak of neurological disease in mares and stallions caused by EHV-1.153 Chorioretinal lesions, in the absence of uveitis, may develop three to five weeks after respiratory tract infection,208 and three distinct types of lesions (focal, multifocal and diffuse) have recently been identified (J.D. Slater, unpublished observations). All evidence suggests that the focal and multifocal lesions do not greatly impair vision, but the diffuse lesions may result in extensive retinal destruction and blindness.
Pulmonary vasculotropic infection
Several cases of generalized, peracute disease following EHV-1 infection have recently been reported in young adult horses.24, 81 The new syndrome, termed ‘pulmonary vasculotropic EHV-1 infection’, is characterized by high fever, anorexia, severe depression, respiratory distress, and high mortality. Neurological signs have been absent. Affected horses may be found dead without prior clinical signs being noticed. Onset of the condition is sudden, and its course of progression to death is rapid. The dominant necropsy finding is a multisystemic vasculitis particularly prominent in the small blood vessels of the lungs.
Pathology
Respiratory disease
Pathological studies of field cases of respiratory disease caused by EHV-1 or EHV-4 are rare because of the low mortality of this condition. Gross pathological examination of the upper respiratory tract of foals experimentally infected with EHV-1 showed hyperaemia, vesiculation, necrosis and ulceration of the mucosa, with occasional miliary dark-red foci Figures 76.4).188 In a series of adult ponies infected experimentally with EHV-1, macroscopic pathological changes in the respiratory tract and associated lymph nodes were mild or non-specific despite widespread microscopic changes.140, 141
Microscopic lesions in the respiratory tract of foals exposed to field or experimental infections with EHV-1 generally involve a multifocal, necrotizing to exudative rhinitis and terminal bronchiolitis or alveolitis. There is neutrophilic infiltration into bronchiolar walls, mononuclear cell infiltration of peribronchiolar and perivascular regions and flooding of local alveoli by serofibrinous fluid. The pharyngeal lymphoid follicles are often hyperplastic, with foci of necrosis in which there are cells containing intranuclear inclusion bodies. Inclusion bodies may also be found in degenerate epithelial cells of the nasal mucosa, conjunctiva and airways.188
Microscopic lesions in the respiratory tract of experimentally infected adult ponies are relatively mild, with multifocal erosions in the nasal and nasopharyngeal mucosa (Figures 76.4), and there may be patchy bronchiolitis, interstitial oedema and perivascular cuffing in the lungs.140, 141 Similarly, respiratory tract lesions in adult horses with EHV-1 paresis were low-grade by comparison to foals.245 The pathology of EHV-4 respiratory disease has not been described.
Abortion
The pathological changes in foetuses aborted due to EHV-1 infection are well documented.82, 84, 244 Foetuses that are aborted after five months of gestation are generally fresh and often still enclosed in unruptured placental membranes (Figures 76.8a). There may be extensive meconium staining of the integument, amnion and hooves, consistent with foetal distress in utero. Dissection generally reveals some combination of moderate to marked pulmonary oedema, hydrothorax, mild ascites, variable icterus, petechiation in the lungs and of serosal surfaces and mucous membranes, subcutaneous and perirenal oedema, splenomegaly with prominent lymphoid follicles, softening of the thymus, moderate hepatomegaly and miliary foci of hepatic necrosis (Figures 76.8d). The relative prominence of these various macroscopic lesions varies from case to case. Only a small proportion of EHV-1 infected foetuses are aborted prior to five months of gestation, but those foetuses are often severely autolysed and lack specific macroscopic lesions.188
Microscopic lesions in foetuses that have been aborted in mid- to late gestation consist of a necrotizing bronchiolitis, pneumonitis, lymphoid depletion and necrosis in the spleen, lymph nodes and thymus, and foci of necrosis in the liver, splenic red pulp and adrenal glands (Figures 76.9 and 76.10). Intranuclear inclusion bodies are generally found in degenerated and necrotic cells, particularly in the liver, lungs, thymus and splenic red pulp. Some foetuses also show necrotizing enteritis, with viral inclusion bodies in degenerated and necrotic enterocytes.129 Histological examination of foetuses aborted in early gestation reveals a diffuse scattering of inclusion bodies in the liver, lungs and lymphoreticular tissues.93, 188 Although the placenta is usually free of both gross and microscopic lesions, inclusions can sometimes be found in chorial epithelial cells (Figures 76.9d).
Neonatal foal syndrome
Full-term new-born foals that have succumbed shortly after birth following a late-gestation, in utero infection with EHV-1 typically exhibit dark, congested, and consolidated lungs with fluid and froth in the airways.45, 167, 182 In the lungs, microscopic examination reveals an acute, focal necrotizing bronchiolitis and interstitial pneumonia with many unaerated lobules and collapsed alveoli.45, 167, 182 Intranuclear herpesvirus inclusion bodies are common in the affected areas. Scattered necrotic hepatocytes may be present in the liver, and goblet cell hyperplasia and hypertrophy have been observed in the jejunal mucosa. In primary, uncomplicated EHV-1 disease of very young neonates, in which death is rapid (one to three days), the pathological findings are dominated by acute viral lesions in the respiratory tract.128, 167, 182 In more chronically affected new-born foals that survive for longer periods (5 to 14 days), the virus-induced pulmonary pathology is usually complicated by additional lesions resulting from bacterial superinfection (purulent bronchopneumonia, necrotizing enteritis, septicaemia, and a massive necrotic destruction of the thymic and splenic lymphocyte populations).45
Neurological disease
Macroscopic lesions are often lacking in horses affected by the neurological manifestation of EHV-1 infection, although there may be focal haemorrhages or areas of malacia in the brain and spinal cord (Figures 76.7). The haemorrhages have a random distribution, but generally affect white matter, particularly in the lumbosacral segments of the spinal cord.61, 102, 134, 135, 245
The microscopic lesions of neurological disease differ in their severity and distribution, but have the common feature of a non-suppurative vasculitis affecting small arteries and veins.124, 135, 183, 245 The vasculitis is characterized by endothelial cell swelling or pyknosis, fibrinoid necrosis or neutrophilic/lymphocytic infiltration of the tunica media and lymphohistiocytic or lymphoplasmacytic perivascular cuffing (Figures 76.7d). The lumen of the affected vessel may contain fibrin or fibrino-cellular thrombi. In the central nervous system, perivascular oedema and haemorrhage or ischaemic necrosis of the adjacent neuropil are often associated with affected vessels. Examination of multiple sections of brain and spinal cord is sometimes necessary to demonstrate these typical changes. The vasculitis is not generalized, and is usually seen most commonly in the nasal mucosa, lungs, lymph nodes local to the respiratory tract, endometrium and central nervous system.102, 135, 245 Clinically significant lesions at other sites are unusual, although enterocolitis resulting in diarrhoea was described in mature standard-bred horses with paresis caused by EHV-1.56
Diagnosis
The diagnosis of EHV-1 and EHV-4 infections cannot usually be made on clinical grounds alone and requires diagnostic laboratory support. A methodical strategy for establishing a laboratory diagnosis of infection of horses is presented in Figures 76.11. 2 This shows, (1) the clinical specimens that should be submitted to the laboratory; (2) the specific diagnostic tests to be performed; and, (3) the laboratory methods currently available for confirmation of virus identity and for type-specific differentiation of EHV-1 from EHV-4 isolates. The basic approach underlying the laboratory testing of each category of clinical specimens submitted is an initial, rapid diagnostic test (e.g. PCR, immunofluorescent staining of cryostat sections, or a screen for complement fixing antibodies) for making a preliminary diagnosis followed by more lengthy confirmatory procedures (virus isolation, histopathology, immunohistochemical staining, or serology performed on paired, acute- and convalescent-phase serum samples).
Rapid laboratory tests for detection of EHV-1 and EHV-4 are most useful in explosive epidemics of disease in horses in which rapid identification of the causative agent is critical for guiding management strategies. A new generation of immunoassays and PCR-based tests have recently been developed and evaluated for use as rapid diagnostic tools for EHV-1 and EHV-4. Because of the advantages of rapidity, convenience, and the lack of a requirement for the presence of infectious virus, these new diagnostic approaches have the potential for supplementing or replacing some of the older conventional methods for laboratory diagnosis of EHV-1 and EHV-4.
Polymerase chain reaction can be used for rapid amplification and diagnostic detection of the nucleic acid of EHV-1 and EHV-4 present in clinical or pathological specimens (foetal or neonatal foal tissues, nasal mucus, brain and spinal cord, or blood leukocytes), paraffin-embedded archival tissues, or inoculated cell cultures.15, 17, 26, 137, 142, 149, 171, 194, 195, 203, 239, 242 A variety of type-specific PCR primers have been designed to detect and distinguish between the presence of these two herpesviruses. The agreement between PCR and virus isolation techniques for the diagnosis of EHV-1 or EHV-4 is in the order of 85 to 90 per cent. Polymerase chain reaction now forms an integral part of a range of diagnostic tests currently available for detection of EHV-1 and EHV-4, each with its own advantages and limitations.
Direct immunofluorescent (IF) detection of EHV-1 or EHV-4 antigens in cryostat sections of tissues freshly dissected from aborted equine foetuses provides a rapid method for making a preliminary diagnosis of herpesvirus abortion (Figures 76.9b). Side-by-side comparisons of the IF and cell culture isolation techniques on cases of equine abortion have provided evidence that the diagnostic reliability of direct IF staining of foetal tissues obtained at necropsy approaches that of virus isolation from the same tissues.
For many years, the gold standard test for making a laboratory diagnosis of EHV-1 or EHV-4 infections has been inoculation of cell cultures for isolation of the viruses. Equid herpesvirus 1 may be isolated in a variety of cell lines — such as equine dermis (ED), equine foetal kidney (EFK), equine embryonic lung (EEL), rabbit kidney (RK-13) and MadinDarby bovine kidney (MDBK) — whilst optimal primary isolation of EHV-4 requires cultures of equine-derived cells (e.g. ED, EEL and EFK). The cytopathic effects (CPE) caused by EHV-1 and EHV-4 in cell cultures develop early, progress rapidly, and are characteristically herpetic in appearance (clusters of rapidly enlarging, rounded, refractile, and partially detached cells). Under ordinary conditions, isolation of EHV-1 or EHV-4 can be achieved in two to four days (frequently within one day). Blind passage is not usually necessary to isolate the viruses.
Histopathological examination of sections of formalin- fixed, paraffin-embedded tissues from aborted foetuses or from horses affected by neurological disease is an essential part of the laboratory diagnosis of these two clinical manifestations of EHV-1 infection.
In aborted foetuses, typically herpetic, eosinophilic intranuclear inclusions present within bronchiolar epithelium or in cells at the periphery of areas of hepatic necrosis are pathognomonic lesions of EHV-1 infection (Figures 76.10c). The characteristic microscopic lesion associated with EHV-1 myeloencephalopathy is a degenerative, and often thrombotic, vasculitis of small blood vessels in the brain or spinal cord (Figures 76.7d).
Enzyme immunohistochemical (e.g. immunoperoxidase) staining methods have been developed recently as procedures for detecting EHV-1 or EHV-4 antigen in paraf- fin-embedded tissues from infected horses (Figures 76.9c).201, 246 Such ancillary, immunoenzymatic staining techniques for EHV-1 or EHV-4 antigen detection may facilitate identification of the viruses in archival tissue samples or in clinical cases for which traditional laboratory methods for virus detection have yielded inconclusive results.
Confirmation of the identity of virus isolates as equid herpesviruses and their further differentiation as EHV-1 or EHV-4 can be achieved by type-specific PCR, by restriction endonuclease analysis of isolated viral DNA, or by IF staining of cytocentrifuge preparations of infected cell cultures with type-specific monoclonal antibodies.
Serological diagnosis of recent infection by EHV-1 or EHV-4 may sometimes be achieved by the laboratory detection of a significant rise in virus-specific antibody titre between sera collected during acute illness and again two to three weeks later during convalescence. Serological assays suitable for detecting antibodies against EHV-1 or EHV-4 include the virus neutralization (VN), ELISA, and complement fixation (CF) tests.2, 232 Each of these tests detects antibodies shared by the two herpesviruses, and none allows differentiation between infection by EHV-1 and EHV-4. When serum from only a single collection of blood is available for testing, the presence of antibodies detected by the CF test provides the strongest serological evidence for recent viral infection (or vaccination) since complement fixing antibodies to EHV-1 and EHV-4 decay to undetectable levels within 60 days after exposure to viral antigen. The CF test for recent exposure to EHV-1, via infection or immunization, is a useful, rapid screen during the initial diagnostic investigation of ataxic horses.153
A type-specific ELISA that distinguishes antibodies elicited by EHV-1 from those elicited by EHV-4 infection was recently developed and made commercially available.69
It is important to remember, with each of the abovedescribed serological tests for EHV-1 or EHV-4, that interpretation of the serological results can be confounded by previous vaccination, that young foals may experience respiratory infection and clinical disease by EHV-1 or EHV-4 without mounting a detectable antibody response, and that ‘acute’ phase sera collected from aborting mares, multiple vaccinated horses, or cases of neurological disease may, at the time of collection, already contain maximal levels of antirival antibodies and, therefore, fail to exhibit further increases in titre in convalescent phase serum samples. Serological examination of mares is generally not a useful method for the diagnosis of EHV-1 abortion.

Figure 76.9 Microscopic examination of equine foetus aborted as a result of EHV-1 infection. (a) Focal necrosis in the liver, (b) immunofluorescence-positive hepatocytes and endothelial cells in the liver, (c) immunoperoxidase-positive hepatocytes and endothelial cells in the liver, (d) chorionic villus of placenta with eosinophilic, intranuclear inclusion bodies in the chorial epithelial cells (arrows). (Panel d by courtesy of Dr. H. Acland, Pennsylvania State Veterinary Laboratory, Harrisburg, USA)

Figure 76.10 Microscopic examination of equine foetus aborted as a result of EHV-1 infection. (a) Necrosis in lymphoid follicle of spleen, (b) severe pneumonitis and bronchiolitis with obliteration of bronchiolar lumen by inflammatory debris, (c) degeneration and presence of eosinophilic inclusion bodies in respiratory epithelium of bronchiole, (d) central necrosis in lymphoid follicle of mesenteric lymph node. (By courtesy of Dr. H. Acland, Pennsylvania State Veterinary Laboratory, Harrisburg, USA)
Latent EHV-1 or EHV-4 cannot be detected by the usual diagnostic methods. Post-mortem detection of latent infection in horses is most reliably achieved by nested PCR or prolonged co-cultivation techniques performed on tissues collected aseptically at necropsy from either the trigeminal ganglia or the submandibular, retropharyngeal, or bronchial lymph nodes.26, 27, 105, 207, 243
The ante-mortem diagnosis of EHV-1 or EHV-4 latency, using the same detection techniques on tissues available through non-invasive collection methods (e.g. PBMC), is more problematic, presumably due to the lower frequency of latently infected leukocytes that circulate in the blood. More sensitive tests for the laboratory demonstration of latent EHV-1 in peripheral blood leukocytes of horses include RT-PCR detection of EHV-1 LAT sequences that accumulate in latently infected leukocytes,62 and the immunohistochemical detection of EHV-1 proteins expressed in latently infected lymphocytes after in vitro cultivation in the presence of T-cell mitogens.210
Differential diagnosis
The clinical signs of EHV-1 and EHV-4 infections in horses are variable, often non-specific, and may be confused with a variety of clinically similar conditions with other aetiologies. Because of such uncertainties in making a clinical diagnosis, cases of suspected EHV-1 or EHV-4 infection should be confirmed by careful laboratory investigation.
Foetal losses in mares resulting from EHV-1 infection must be differentiated from a wide range of other infectious and non-infectious conditions that can cause equine abortion, including those due to infections by other abortigenic equine viruses (e.g. equine arteritis virus), bacteria (e.g. Leptospira spp., Streptococcus spp., and a nocardioform actinomycete), or protozoa (e.g. equine babesiosis) as well as many non-infectious causes (e.g. torsion of the umbilical cord, congenital anomalies, placental disorders, mare reproductive loss syndrome (MRLS) and perinatal asphyxia due to dystocia).116, 129
Respiratory disease resulting from infection with EHV-1 or EHV-4 can mimic disease caused by several other respiratory pathogens of horses (e.g. equine influenza virus, equine arteritis virus, equine rhinovirus, equine adenovirus, Streptococcus equi zooepidemicus, Rhodococcus equi, Mycoplasma spp.),187, 254 thus necessitating laboratory assistance in establishing a definitive aetiological diagnosis.
The neurological signs associated with EHV-1 infection are not specific and may be confused with those of many other central nervous system disorders of the horse (e.g. equine protozoal myeloencephalopathy, trauma, cervical vertebral malformation, equine motor neuron disease, equine degenerative myeloencephalopathy, arboviral encephalitides, Borna disease, Getah encephalitis, botulism, rabies, and mouldy corn poisoning).80 In stallions, early stages of EHV-1 myeloencephalopathy that are accompanied by pyrexia, scrotal oedema, and loss of libido may be confused with equine viral arteritis.
Neonatal mortality in foals is relatively common and may have a variety of causes other than EHV-1 that must be considered in the differential diagnosis (e.g. bacterial septicaemia, babesiosis and neonatal isoerythrolysis). Pulmonary vasculotropic EHV-1 infection, because of its rapid progression and severe respiratory distress, may be confused with other peracute respiratory infections of the horse (e.g., African horse sickness and Hendra virus respiratory syndrome).
Control
The practical objectives for minimizing the economic and animal welfare burdens caused by EHV-1 and EHV-4 infections are to reduce the incidence or clinical severity of disease in horses exposed to the viruses, and to contain the spread of viral infection during individual outbreaks of herpesvirus disease. The basis for achieving these objectives is the combination of a continuous programme of prophylactic immunization and the implementation of a set of empirically established stud farm management practices.4, 39, 42, 186 A common, and often disastrous, misconception among horse owners and stud managers is that vaccination alone can be relied upon to control herpesvirus disease. The EHV-1 and EHV-4 vaccines currently available only supplement and support sound management practices; they were not designed, and cannot successfully be used, for replacement of good herd management.
Control by management
From many years of collective experience with managing herpesvirus disease in horses, day-to-day operating procedures at the level of the farm, stud, or yard have emerged as being effective in preventing or limiting the spread of EHV-1 and EHV-4. In the case of abortion and/or neurological diseases caused by EHV-1, those measures have been conceptualized into discrete, precautionary actions and published as recommended herd management practices for prevention and control of epidemic disease.4, 39, 42, 153, 186 In some countries (e.g. Australia and the UK), the recommended actions have been formalized into ‘codes of practice’.12, 130, 241
In practice, the recommended stud farm management procedures for controlling epidemic EHV-1 disease are subdivided into those formulated for prevention or reduction of the likelihood of an outbreak of abortion or neurological disease, and those designed to limit the spread of disease to other groups of horses, both on and off the stud, once an outbreak has occurred.
The rationale underlying the management procedures recommended for prevention of abortion or neurological disease in brood mares can be described by the acronym ‘SISS’ (Table 76.3), which comprises:
- Segregation of the pregnant mare population from all other categories of horses on the premises, particularly weanlings, yearlings, transients and new arrivals;
- Isolation (for a period of no less than three weeks) of new mares arriving on the stud, including those that return after having left the premises for such purposes as sales and breeding;
- Early subdivision of the pregnant mares into small, physically separated foaling groups for the duration of gestation; and
- Stress reduction by avoiding or minimizing those actions known to induce physiological stress in mares, such as the disruption of established social structures, prolonged transport, relocation, poor nutritional state, heavy parasite load, other disease states, exposure to inclement weather conditions, and abrupt en masse weaning techniques.
Following an outbreak of EHV-1 abortion or neurological disease, the goal should be to limit further transmission of the virus. The actions recommended for containing the spread of an ongoing outbreak of EHV-1 abortion or neurological disease are based upon the strict observation of hygienic and quarantine principles designed to contain the infection to its original, small foaling group of mares. The recommended measures to be taken in such epidemic situations can be described by the acronym ‘DISH’ (Table 76.4), and consist of
- Disinfection of the areas contaminated by virus extruded from the aborted foetus and its placental membranes;
- The physical isolation of the aborting mare from other horses within the foaling group;
- Submission of clinical samples, including the foetus, to a diagnostic laboratory for examination; and
- Implementation of standard hygienic procedures designed for infectious disease control.
It is especially important to attempt to prevent the exposure of other pregnant mares in the group to the index abortion; i.e. the aborted foetus, its membranes, and fluids. The restriction of animal movement into and out of the infected group is essential. The single most important action to avoid, after an outbreak of abortion or neurological disease has begun, is the movement of exposed horses, including those free of clinical signs, out of the infected area and their intermingling with other groups of horses. Such mixing of animals from physically separated groups has the potential to lead to unusually high attack rates resulting from the generalized spread of infection by silently shedding animals to previously unexposed horses on the same or other premises.19 Because EHV-1 or EHV-4 may take several weeks to spread through a large group of stabled horses, subdivision of the animals into smaller, isolated groups (if the facilities permit such action) can often be beneficial in reducing the spread of infection.125, 153, 166
Table 76.3 Recommended stud farm management practices for prevention of epidemic EHV-1 abortion or neurological disease
| MANAGEMENT PRACTICE | SPECIFIC ACTION TO BE TAKEN |
| Segregation | Pregnant brood mares should be physically separated from all other horses on the premises, particularly those in which EHV-1 respiratory infections are common (e.g. weaned foals, yearlings, horses in training or purchased from sales, etc.). If handling of both pregnant mares and other horses by the same personnel cannot be avoided, the work schedules should be arranged to allow pregnant mares to be handled first. Introduced foster mares should not be mixed with pregnant mares. Segregate pregnant maiden mares from older mares. |
| Isolation | Mares or fillies arriving on the stud from sales, racetracks, or other stud farms should never be mixed with the resident brood mares prior to a period of quarantine. Isolation should be for a period of no less than three weeks in a geographically isolated facility. Ideally, staff assigned to the isolation area should have no contact with other horses on the stud farm. Pregnant mares removed and subsequently returned to the stud farm should not be reintroduced into the original foaling group without first going through a 21-day period of isolation. |
| Subdivision | As soon as possible after completion of the last pregnancy examination, pregnant mares should be divided into small foaling groups by stage of gestation. Each group should then be maintained as a closed, isolated unit until all mares in it have foaled. Delay in the formation of foaling groups until late in gestation can invite an epidemic of EHV-1 abortion. Foaling groups should be as small as the facilities will practically permit and be organized so that temporal overlap of different groups in the same foaling shed is avoided. |
| Stress reduction | Stress during late pregnancy can cause reactivation of latent EHV-1 in the mare with resultant infection of the foetus and nasal shedding of infectious virus by the mare. Management activities known to be stressful for the horse (e.g. transport, social disruption, weaning, etc.) should thus be minimized. The prolonged transport of mares in late pregnancy should be avoided. Mares should not be added to or removed from already established foaling groups. Foals at the sides of pregnant mares should be weaned by a process designed to produce the least possible stress, e.g. the removal of one mare from the group every few days |
Table 76.4 Recommended stud farm management practices for the containment of an outbreak of EHV-1 abortion
| MANAGEMENT PRACTICE | SPECIFIC ACTION TO BE TAKEN |
| Disinfection | If the foetus is found in a stall, all bedding should be sprayed with a phenolic or iodophor-type disinfectant, placed in plastic bags, removed, and burned. The stall should then be thoroughly cleaned and disinfected. The aborting mare should be cleaned and washed with an antiseptic-containing detergent. After the final abortion, the entire foaling area, horse conveyance vehicles, and all equipment and utensils used for feeding, watering, grooming, and cleaning within the infected premises should be cleaned and disinfected. |
| Isolation | Request the advice of a veterinary surgeon immediately. The aborting mare should be placed in strict isolation of 21 days and should not come into contact with mares in late pregnancy for two months after the abortion. No other mares should be removed from the foaling group in which an abortion caused by EHV-1 has occurred. Other in-contact horses, such as those in adjacent paddocks or yards, should not be moved during the outbreak. All pregnant mares must remain on the infected stud until they foal. In the infected group, the mating of mares that have already foaled should cease until the infection has been brought under control. Do not move any horse off the stud or any pregnant mare onto the stud until 21 days after the last abortion. If the foaling group in which a confirmed EHV-1 abortion has occurred is large, the remaining mares in the group should be split into smaller isolated groups as the facilities will practically permit. Breeding of mares that have aborted due to EHV-1 should be delayed until the second oestrus cycle after abortion |
| Submission of samples for laboratory examination | Aborted foetuses and their membranes should be placed in a leak-proof container and submitted to the diagnostic laboratory for post-mortem examination. Nasopharyngeal swabs and heparinized blood samples should be collected from both mares and foals in the group in which an abortion has occurred, as well as from other horses selected at random on the affected stud, and submitted to the laboratory for virological examination. Clotted blood samples collected from the same animals immediately after the first abortion and again at weekly intervals should be submitted for serological examination. Virological clearance by laboratory examination should be established before lifting the ban on movement of horses on infected premises. |
| Hygiene | A separate staff should be designated to work with the foaling group of mares in which a herpesvirus abortion has occurred. Handlers of aborted foetuses or attendants of aborting mares in isolation must have no contact with pregnant mares during the isolation period. They should wear disposable gloves, boot covers, and removable outerwear and be instructed in standard hygienic procedures for restricting the spread of infection, including the use of hand scrubbiung, antiseptics, and disinfectant footbaths. Aborted foetuses or contaminated bedding should not be dragged across the stable floor |
Control by vaccination
A robust protective immunity develops in most horses during recovery from infection with EHV-1 or EHV-4.5, 31, 37, 44, 93, 113, 176 Although fleeting in duration (three to six months), the immune response to EHV-1 and EHV-4 is vigorous and infection-induced resistance to virus reinfection during this post-recovery period is solid. Following rechallenge of horses during the three- to six-month window of post-infection protective immunity, virus cannot be recovered from nasopharyngeal secretions and detectable leukocyte-associated viraemia does not develop. In early, classic investigations by Doll and Bryans,93 it was demonstrated that 85 per cent of pregnant mares infected early in gestation (at a time when refractory to EHV-1 abortion) were rendered immune to abortion after a second challenge with EHV-1 during the later vulnerable months of gestation. Characterization of the antiviral immune effector mechanisms activated in the horse during the window of protection afforded by infection immunity has identified elevated levels of (1) virus neutralizing antibody in both serum and nasopharyngeal mucus; (2) viral antigen-driven, CD4+ T-lymphocyte proliferative activity; (3) virus-specific, CD8+ cytotoxic T-lymphocyte precursors (CTLp); (4) herpesvirus reactive natural killer (NK) lymphocyte activity; and (5) virus-specific antibodydependent, cell-mediated cytotoxic (ADCC) activity.6, 10, 31,32, 33, 34, 37, 46, 50, 63, 64, 97, 98, 99, 113, 148, 172, 173, 176, 177, 220, 221, 248, 249 Despite extensive research, no quantitative level of either serological or cellular immune responses that correlate with protection against specific EHV-1 or EHV-4 disease has been identified.164
During the past 40 years, many attempts have been made to immunize horses against both EHV-1 and EHV-4 with the objective of mimicking the resistance to disease induced by natural infection and thereby reducing the impact of disease caused by these two equine pathogens. Those vaccine efforts have been extensively described5, 41, 42, 74, 90, 91, 162, 174 and have culminated in the availability of more than a dozen commercially manufactured products that are currently marketed for use in controlling the diseases caused by EHV-1 and EHV-4. 3 Immunologically experienced, adult horses respond to vaccination by these commercial immunogens by developing high titres of serum antibodies directed against the viruses.40, 41, 54, 77, 90, 107, 181, 189 Weaker postvaccinal antibody responses are elicited in young, immunologically naive animals, and a serological response may be undetectable in immunized foals possessing maternally derived antibodies directed against the viruses at the time of vaccination.30, 252 For some of the available equine herpesvirus vaccines, cellular immune responses, both proliferative and cytotoxic, have been reported to be elicited.107, 108
The overall success of those cumulative efforts at development of efficacious vaccines has been less than hoped for,19, 47, 48, 54, 99, 110, 163, 181, 189, 219 and the two equine herpesviruses remain among the most economically significant viral pathogens of domestic horses 70 years after their discovery. This somewhat disappointing performance of vaccines against EHV-1 and EHV-4 mirrors that experienced for other animal (and human) herpesviruses, as vaccines that prevent infection and provide long-term protection to the vaccinated host have not yet been developed for most herpesviruses.
It should be clear from the foregoing comments that vaccination of horses for EHV-1 and EHV-4 should be considered as only a supplementary tool to be used together with the procedures of sound stud farm management described above. No currently registered vaccine for EHV-1 or EHV-4 is claimed to be completely effective in preventing either infection or disease that may result from exposure.
Notwithstanding these limitations, vaccination is still an important component of the overall strategy for controlling the infectious disease burden caused by EHV-1 and EHV-4. When used as an adjunct to sound management practices, vaccination can be effective in modulating the severity of EHV-1 or EHV-4 respiratory disease in young stock and in limiting the occurrence and severity of abortion storms. While stud farm management schemes for controlling equine herpesvirus disease are directed toward reducing the amount of infectious virus in the horse’s environment (reducing exposure), the goal of vaccination is to enhance the resistance of the individual horse to disease following exposure to the herpesviruses.
The expectations from current generations of vaccines for EHV-1 and EHV-4 should, realistically, be to minimize the clinical consequences of post-exposure infection, and to decrease the risk for cohort transmission of infection by reducing the extent of intrahost amplification and subsequent shedding of infectious virus. The primary beneficial effects derived from vaccination of horses against EHV-1 and EHV-4 are reduction in the severity and/or duration of respiratory disease in the young vaccinated animal and reduction of the overall incidence of foetal loss during outbreaks of abortion on stud farms. In the central Kentucky Thoroughbred horse population of approximately 12 000 brood mares mated annually, the incidence of EHV-1 abortion (i.e. abortions/1 000 pregnant mares) has declined by 75 per cent during the 20 years of intensive vaccination usage.174 Most of this reduction in EHV-1 abortigenic disease in Kentucky has been the result of a decrease in the number of single-abortion incidents that subsequently progress into multiple-case abortion storms.
The benefits of a vaccination programme in providing protection against neurological disease have not been established. The advisability of vaccination in the face of an outbreak of neurological disease caused by EHV-1 is also controversial; some believe that the immunosensitization of horses to EHV-1 antigen elicited by vaccination has the potential to exacerbate the neurological complications. Because the duration of immune resistance provided by vaccination for EHV-1 and EHV-4 is short (several weeks to a few months), annual reboosting with multiple-vaccine doses is required for optimal results. There is no evidence to suggest that vaccination is effective in blocking the annual cycle of virus transmission to young animals by preventing either the reactivation of latent herpesvirus in mares or the establishment of viral latency in foals.
The M.H. Gluck Equine Research Center (Lexington, Kentucky, USA), the Animal Health Trust (Newmarket, Suffolk, UK), and the All-Russian Institute of Experimental Veterinary Medicine (Moscow, Russia) have been designated as World Reference Laboratories of the Office International des Epizooties (OIE) for EHV-1 and EHV-4.
References
- ADEYEFA, C.A., 1992. Serological evidence of equine herpesvirus type 1 (EHV-1) activity in polo horses in Nigeria. Zentralblatt für Veterinärmedizin, Reihe B, 39, 628–630.
- ALLEN, G.P., 2000. Equine rhinopneumonitis. In: REICHARD, R., (ed.). O.I.E. Manual of Standards for Diagnostic Tests and Vaccines, 4th edition. Paris: Office International des Epizooties (O.I.E.) Press, pp. 565–575.
- ALLEN, G.P., 2002. Respiratory infections by equine herpesvirus types 1 and 4. In: LEKEUX, P., (ed). Equine Respiratory Diseases (online publication). Ithaca: International Veterinary Information Service, [http://www.ivis.org/special_books/Lekeux/allen/chapter_frm .asp?LA=1]
- ALLEN, G.P., 2002. Epidemic disease caused by equine herpesvirus-1: Recommendations for prevention and control. Equine Veterinary Education (in press).
- ALLEN, G.P. & BRYANS, J.T., 1986. Molecular epizootiology, pathogenesis, and prophylaxis of equine herpesvirus-1 infections. Progress in Veterinary Microbiology and Immunology, 2, 78–144.
- ALLEN, G.P., COOGLE, L.D., OSTLUND, E.N. & YEARGAN, M.R., 1992. Molecular dissection of two major equid herpesvirus-1 glycoprotein antigens (gB and gC) that elicit humoral immune responses in the horse. In: PLOWRIGHT, W., ROSSDALE, P.D. & WADE, J.F., (eds). Equine Infectious Diseases VI. Newmarket (Suffolk):R&W Publications, pp. 181–193.
- ALLEN, G.P., KYDD, J.K., SLATER, J.D. & SMITH, K.C., 1999. Advances in understanding of the epidemiology, pathogenesis and immunological control of equid herpesvirus-1 abortion. In: WERNERY, U., WADE, J., MUMFORD, J. & KAADEN, O.-R., (eds). Equine Infectious Diseases VIII. Newmarket (Suffolk):R&W Publications, pp.129–146
- ALLEN, G.P., O’CALLAGHAN, D.J. & RANDALL, C.C., 1977. Genetic relatedness of equine herpesvirus types 1 and 3. Journal of Virology, 24, 761–767
- ALLEN, G.P., YEARGAN, M.R. & BRYANS, J.T., 1983. Alterations in the equine herpesvirus 1 genome after in vitro and in vivo virus passage. Infection and Immunity, 40, 436–439.
- ALLEN, G.P., YEARGAN, M.R., COSTA, L.R. & CROSS, R., 1995. Major histocompatibility complex class I-restricted cytotoxic T-lymphocyte responses in horses infected with equine herpesvirus 1. Journal of Virology, 69, 606–612.
- ALLEN, G.P., YEARGAN, M.R., TURTINEN, L.W., BRYANS, J.T. & MCCOLLUM, W.H., 1983. Molecular epizootiologic studies of equine herpesvirus-1 infections by restriction endonuclease fingerprinting of viral DNA. American Journal of Veterinary Research, 44, 263–271
- AUSTRALIAN EQUINE VETERINARY ASSOCIATION, 1994. Guidelines for the Control of Equine Herpesvirus 1 Abortion
- AWAN, A.R., CHONG, Y.-C. & FIELD, H.J., 1990. The pathogenesis of equine herpesvirus type 1 in the mouse: A new model for studying host responses to the infection. Journal of General Virology, 71, 1131–1140
- AWAN, A.R., GIBSON, J.S. & FIELD, H.J., 1991. A murine model for studying EHV-1-induced abortion. Research in Veterinary Science, 51, 94–99.
- AZMI, M. & FIELD, H.J., 1993. Detection and identification of equine herpesvirus-1 and -4 by polymerase chain reaction. Veterinary Microbiology, 36, 57–67.
- BAGUST, T.J., PASCOE, R.R. & HARDEN, T.J., 1972. Studies on equine herpesviruses 3. The incidence in Queensland of three different equine herpesvirus infections. Australian Veterinary Journal, 48, 47–53.
- BALLAGI-PORDANY, A., KLINGEBORN, B., FLENSBURG, J. & BELAK, S., 1990. Equine herpesvirus type 1: Detection of viral DNA sequences in aborted fetuses with the polymerase chain reaction. Veterinary Microbiology, 22, 373–381.
- BARNARD, B.J. & PAWESKA, J.T., 1993. Prevalence of antibodies against some equine viruses in zebra (Zebra burchelli) in the Kruger National Park, 1991–1992. Onderstepoort Journal of Veterinary Research, 60, 175–179.
- BARRANDEGUY, M.E., LASCOMBES, F., LLORENTE, J., HOUSSAY, H. & FERNANDEZ, F., 2002. High case-rate equine herpesvirus 1 abortion outbreak in vaccinated polo mares in Argentina. Equine Veterinary Education (in press).
- BAUMANN, R.P., SULLIVAN, D.C., STACZEK, J. & O’CALLAGHAN, D.J., 1986. Genetic relatedness and colinearity of genomes of equine herpesvirus types 1 and 3. Journal of Virology, 57, 816–825.
- BAXI, M.K., EFSTATHIOU, S., LAWRENCE, G., WHALLEY, J.M., SLATER, J.D. & FIELD, H.J., 1995. The detection of latency-associated transcripts of equine herpesvirus 1 in ganglionic neurons. Journal of General Virology, 76, 3113–3118.
- BITSCH, V. & DAM, A., 1971. Nervous disturbances in horses in relation to infection with equine rhinopneumonitis virus. Acta Veterinaria Scandinavica, 12, 134–136
- BLUNDEN, A.S., SMITH, K.C., BINNS, M.M., ZHANG, L., GOWER, S.M. & MUMFORD, J.A., 1995. Replication of equid herpesvirus 4 in endothelial cells and synovia from a field case of viral pneumonia and synovitis in a foal. Journal of Comparative Pathology, 112, 133–140.
- BLUNDEN, A.S., SMITH, K.C., WHITWELL, K.E. & DUNN, K.A., 1998. Systemic infection by equid herpesvirus-1 in a Grevy’s zebra stallion (Equus grevyi) with particular reference to genital pathology. Journal of Comparative Pathology, 119, 485–493.
- BORCHERS, K. & FRÖLICH, K., 1997. Antibodies against equine herpesviruses in free-ranging mountain zebras from Namibia. Journal of Wildlife Diseases, 33, 812–817
- BORCHERS, K. & SLATER, J., 1993. A nested PCR for the detection and differentiation of EHV-1 and EHV-4. Journal of Virological Methods, 45, 331–336.
- BORCHERS, K., WOLFINGER, U., LAWRENZ, B., SCHELLENBACH, A. & LUDWIG, H., 1997. Equine herpesvirus 4 DNA in trigeminal ganglia of naturally infected horses detected by direct in situ PCR. Journal of General Virology, 78, 1109–1114.
- BOWLES, D.E., HOLDEN, V.R., ZHAO, Y. & O’CALLAGHAN, D.J., 1997. The ICP0 protein of equine herpesvirus 1 is an early protein that independently trans-activates expression of all classes of viral promoters. Journal of Virology, 71, 4904–4914
- BOWLES, D.E., KIM, S.K. & O’CALLAGHAN, D.J., 2000. Characterization of the trans-activation properties of equine herpesvirus 1 EICP0 protein. Journal of Virology, 74, 1200–1208.
- BREATHNACH, C.C., ALLEN, G.P., HOLLAND, R.E., CONBOY, H.S., BERRY, D.B. & CHAMBERS, T.M., 1999. Problems associated with vaccination of foals against equine herpesvirus-4 (EHV-4) and the role of anti-EHV-4 maternal antibodies. In: WERNERY, U., WADE, J.F., MUMFORD, J.A, & KAADEN, O.-R., (eds). Equine Infectious Diseases VIII. Newmarket (Suffolk):R&W Publications, pp 426–427.
- BREATHNACH, C.C., YEARGAN, M.R., SHEORAN, A.S. & ALLEN, G.p., 2001. The mucosal humoral immune response of the horse to infective challenge and vaccination with Equine herpesvirus-1 antigens. Equine Veterinary Journal, 33, 651–657.
- BRIDGES, C.G. & EDINGTON, N., 1987. Genetic restriction of cytolysis during equid herpesvirus 1 subtype 2 infections. Clinical and Experimental Immunology, 70, 276–282.
- BRIDGES, C.G. & EDINGTON, N., 1987. The proteins of equid herpesvirus 1 (EHV 1) recognised by equine antisera and their ability to promote antibody-dependent cell-mediated cytotoxicity. Tierärztliche Praxis, Supplement, 2, 47–49.
- BRIDGES, C.G., LEDGER, N. & EDINGTON, N., 1988. The characterization of equine herpes virus-1-infected cell polypeptides recognized by equine lymphocytes. Immunology, 63, 193–198.
- BROWNING, G.F., BULACH, D.M., FICORILLI, N., ROY, E.A., THORP, B.H. & STUDDERT, M.J., 1988. Latency of equine herpesvirus 4 (equine rhinopneumonitis virus). The Veterinary Record, 123, 518–519.
- BROWNING, G.F., FICORILLI, N. & STUDDERT, M.J., 1988. Asinine herpesvirus genomes: Comparison with those of the equine herpesviruses. Archives of Virology, 101, 183–190.
- BRYANS, J.T., 1969. On immunity to disease caused by equine herpesvirus-1. Journal of the American Veterinary Medical Association, 155, 294–300
- BRYANS, J.T., 1978. Immunization of pregnant mares with an inactivated equine herpesvirus 1 vaccine. In: BRYANS, J.T. & GERBER, H. (ed.). Equine Infectious Diseases IV. Princeton: Veterinary Publications, pp. 83–92.
- BRYANS, J.T., 1981. Application of management procedures and prophylactic immunization to the control of equine rhinopneumonitis. In: Proceedings of the 26th Annual Convention of the American Association of Equine Practitioners, Anaheim, 1980, pp. 259–272
- BRYANS, J.T., 1980. Serologic responses of pregnant thoroughbred mares to vaccination with an inactivated equine herpesvirus 1 vaccine. American Journal of Veterinary Research, 41, 1743–1746.
- BRYANS, J.T. & ALLEN, G.P., 1982. Application of a chemically inactivated, adjuvanted vaccine to control abortigenic infection of mares by equine herpesvirus I. Developments in Biological Standards, 52, 493–498
- BRYANS, J.T. & ALLEN, G.P., 1986. Control of abortigenic herpesviral infections. Current Therapy in Theriogenology, 2, 711–714.
- BRYANS, J.T. & ALLEN, G.P., 1986. Equine viral rhinopneumonitis. Revue Scientifique et Technique, Office International des Epizooties, 5, 837–847
- BRYANS, J.T. & ALLEN, G.P., 1989. Herpesviral diseases of the horse. In: WITTMANN, G., (ed.). Herpesvirus Diseases of Cattle, Horses and Pigs. Boston: Kluwer, pp. 176–229.
- BRYANS, J.T., SWERCZEK, T.W., DARLINGTON, R.W. & CROWE, M.W., 1977. Neonatal foal disease associated with perinatal infection by equine herpesvirus-1. Journal of Equine Medicine and Surgery, 1, 20–26.
- BUMGARDNER, M.K., DUTTA, S.K., CAMPBELL, D.L. & MYRUP, A.C., 1982. Lymphocytes from ponies experimentally infected with equine herpesvirus 1: Subpopulation dynamics and their response to mitogens. American Journal of Veterinary Research, 43, 1308–-1310.
- BÜRKI, F., NOWOTNY, N., OULEHLA, J., SCHMEHLIK, O., MÖSTL, K., PALLAN, C. & ROSSMANITH, E., 1991. Attempts to immunoprotect adult horses, specifically pregnant mares, with commercial vaccines against clinical disease induced by equine herpesvirus-1. Zentralblatt für Veterinärmedizin, Reihe B, 38, 432–440.
- BÜRKI, F., ROSSMANITH, W., NOWOTNY, N., PALLAN, C., MÖSTL, K. & LUSSY, H., 1990. Viraemia and abortions are not prevented by two commercial equine herpesvirus-1 vaccines after experimental challenge of horses. Veterinary Quarterly, 12, 80–86.
- BURRELL, M.H., WOOD, J.L.N., WHITWELL, K.E., CHANTER, N., MACKINTOSH, M.E. & MUMFORD, J.A., 1996. Respiratory disease in thoroughbred horses in training: The relationships between disease and viruses, bacteria and environment. The Veterinary Record, 139, 308–313.
- BURROWS, R., 1970. The general virology of the herpesvirus group. In: BRYANS, J.T. & GERBER, H., (eds). Equine Infectious Diseases II. Basel: Karger, pp. 1–12.
- BURROWS, R. & GOODRIDGE, D., 1972. In vivo and in vitro studies of equine rhinopneumonitis strains. In: BRYANS, J.T. & GERBER, H., (eds). Equine Infectious Diseases III. Basel: Karger, pp. 306–321.
- BURROWS, R. & GOODRIDGE, D., 1975. Experimental studies on equine herpesvirus type 1 infections. Journal of Reproduction and Fertility, Supplement, 23, 611–615.
- BURROWS, R. & GOODRIDGE, D., 1984. Studies of persistent and latent equid herpesvirus-1 and herpesvirus-3 infections in the Pirbright pony herd. In: WITTMANN, G., GASKELL, R.M. & RZIHA, H.-J., (eds). Latent Herpesvirus Infections in Veterinary Medicine. The Hague: Martinus Nijhoff, pp. 307–319
- BURROWS, R., GOODRIDGE, D. & DENYER, M.S., 1984. Trials of an inactivated equid herpesvirus 1 vaccine: Challenge with a subtype 1 virus. The Veterinary Record, 114, 369–374.
- CAMPBELL, T.M. & STUDDERT, M.J., 1983. Equine herpesvirus type 1. Veterinary Bulletin, 53, 135–146.
- CARMAN, S., NAGY, E., CALDWELL, D. & VAN DREUMEL, T.A., 1993. Equine herpesvirus type 1 neurological disease and enterocolitis in mature standardbred horses. Journal of Veterinary Diagnostic Investigation, 5, 261–265.
- CARRIGAN, M., COSGROVE, P., KIRKLAND, P. & SABINE, M., 1991. An outbreak of equid herpesvirus abortion in New South Wales. Equine Veterinary Journal, 23, 108–110.
- CARROLL, C.L. & WESTBURY, H.A., 1985. Isolation of equine herpesvirus 1 from the brain of a horse affected with paresis. Australian Veterinary Journal, 62, 345–346.
- CAUGHMAN, G.B., ROBERTSON, A.T., GRAY, W.L., SULLIVAN, D.C. & O’CALLAGHAN, D.J., 1988. Characterization of equine herpesvirus type 1 immediate early proteins. Virology, 163, 563–571
- CAUGHMAN, G.B., STACZEK, J. & O’CALLAGHAN, D.J., 1985. Equine herpesvirus type 1 infected cell polypeptides: Evidence for immediate early/early/late regulation of viral gene expression. Virology, 145, 49–61.
- CHARLTON, K.M., MITCHELL, D., GIRARD, A. & CORNER, A.H., 1976. Meningoencephalomyelitis in horses associated with equine herpesvirus 1 infection. Veterinary Pathology, 13, 59–68.
- CHESTERS, P.M., ALLSOP, R., PUREWAL, A. & EDINGTON, N., 1997. Detection of latency-associated transcripts of equid herpesvirus 1 in equine leukocytes but not in trigeminal ganglia. Journal of Virology, 71, 3437– 3443
- CHONG, Y.C. & DUFFUS, W.P., 1992. Immune responses of specific pathogen free foals to EHV-1 infection. Veterinary Microbiology, 32, 215–228
- CHONG, Y.C., DUFFUS, W.P. & HANNANT, D., 1992. Natural killer cells in normal horses and specific-pathogen-free foals infected with equine herpesvirus. Veterinary Immunology and Immunopathology, 33, 103–113.
- CHOWDHURY, S.I., KUBIN, G. & LUDWIG, H., 1986. Equine herpesvirus type 1 (EHV-1) induced abortions and paralysis in a Lipizzaner stud: A contribution to the classification of equine herpesviruses. Archives of Virology, 90, 273–288.
- CHOWDHURY, S.I., LUDWIG, H. & BUHK, H.J., 1988. Molecular biological characterization of equine herpesvirus type 1 (EHV-1) isolates from ruminant hosts. Virus Research, 11, 127–139.
- COIGNOUL, F.L., BERTRAM, T.A. & CHEVILLE, N.F., 1984. Pathogenicity of equine herpesvirus 1 subtype 2 for foals and adult pony mares. Veterinary Microbiology, 9, 533–542.
- CRABB, B.S., ALLEN, G.P. & STUDDERT, M.J., 1991. Characterization of the major glycoproteins of equine herpesviruses 4 and 1 and asinine herpesvirus 3 using monoclonal antibodies. Journal of General Virology, 72, 2075–2082
- CRABB, B.S., MACPHERSON, C.M., REUBEL, G.H., BROWNING, G.F., STUDDERT, M.J. & DRUMMER, H.E., 1995. A type-specific serological test to distinguish antibodies to equine herpesviruses 4 and 1. Archives of Virology, 140, 245–258
- CRABB, B.S., NAGESHA, H.S. & STUDDERT, M.J., 1992. Identification of equine herpesvirus 4 glycoprotein G: A type-specific, secreted glycoprotein. Virology, 190, 143–154
- CRABB, B.S. & STUDDERT, M.J., 1990. Comparative studies of the proteins of equine herpesviruses 4 and 1 and asinine herpesvirus 3: Antibody response of the natural hosts. Journal of General Virology, 71, 2033– 2041.
- CRABB, B.S. & STUDDERT, M.J., 1993. Epitopes of glycoprotein G of equine herpesviruses 4 and 1 located near the C termini elicit type-specific antibody responses in the natural host. Journal of Virology, 67, 6332– 6338
- CRABB, B.S. & STUDDERT, M.J., 1995. Equine herpesviruses 4 (equine rhinopneumonitis virus) and 1 (equine abortion virus). Advances in Virus Research, 45, 153–190.
- CRABB, B.S. & STUDDERT, M.J., 1996. Equine rhinopneumonitis (equine herpesvirus-4) and equine abortion (equine herpesvirus-1). In: STUDDERT, M.J., (ed.). Virus Infections of Vertebrates. Volume 6. Virus Infections of Equines. Amsterdam: Elsevier, pp. 11–63.
- CRANDELL, R.A., DRYSDALE, S. & STEIN, T.L., 1979. A comparative study of bovine herpesvirus 1247 and equine herpesvirus 1 in ponies. Canadian Journal of Comparative Medicine, 43, 94–97.
- CRANDELL, R.A., ICHIMURA, H. & KIT, S., 1988. Isolation and comparative restriction endonuclease DNA fingerprinting of equine herpesvirus-1 from cattle. American Journal of Veterinary Research, 49, 1807–1813
- CRANDELL, R.A., MOCK, R.E. & LOCK, T.F., 1980. Vaccination of pregnant ponies against equine rhinopneumonitis. American Journal of Veterinary Research, 41, 994–996.
- DAVISON, A., EBERLE, R., DESROSIERS, R.C., FLECKENSTEIN, B., MCGEOCH, D.J., PELLET, P.E., ROIZMAN, B. & STUDDERT, M.J., 2000. Herpesviridae. In: VAN REGENMORTEL, M.H.V., FAUQUET, C.M., BISHOP, D.H.L., CARSTENS, E.B., ESTES, M.K., LEMON, S.M., MANILOFF, J., MAYO, M.A., MCGEOCH, D.J., PRINGLE, C.R. & WICKNER, R.B. , (eds). Virus Taxonomy. Seventh Report of the International Committee on Taxonomy of Viruses. Orlando: Academic Press, pp. 114–127.
- DE BOER, G.F., OSTERHAUS, A.D., VAN OIRSCHOT, J.T. & WEMMENHOVE, R., 1979. Prevalence of antibodies to equine viruses in the Netherlands. Tijdschrift voor Diergeneeskunde, 104, Suppl, 65–74.
- DE LAHUNTA, A., 1978. Diagnosis of equine neurologic problems. Cornell Veterinarian, 68, Suppl 7, 122–132.
- DEL PIERO, F. & WILKINS, P.A., 2001. Pulmonary vasculotropic EHV-1 infection in equids. Veterinary Pathology, 38, 474–475.
- DIMOCK, W.W., 1940. The diagnosis of virus abortion in mares. Journal of the American Veterinary Medical Association, 96, 665–666.
- DIMOCK, W.W. & EDWARDS, P.R., 1933. Is there a filterable virus of abortion in mares? Kentucky Agricultural Experiment Station Bulletin, 333, 297–301.
- DIMOCK, W.W. & EDWARDS, P.R., 1936. The differential diagnosis of equine abortion with special reference to a hitherto undescribed form of epizootic abortion of mares. Cornell Veterinarian, 26, 231–240
- DIMOCK, W.W., EDWARDS, P.R. & BRUNER, D.W., 1942. Equine virus abortion. Kentucky Agricultural Experiment Station Bulletin, 426, 1–20
- DIMOCK, W.W., EDWARDS, P.R. & BRUNER, D.W., 1947. Infections observed in equine fetuses and foals. Cornell Veterinarian, 37, 90–99
- DIXON, R.J., HARTLEY, W.J., HUTCHINS, R.D., LEPHARD, E.E., FEILEN, C., JONES, R.F., LOVE, D.N., SABINE, M. & WELLS, A.L., 1978. Perinatal foal mortality associated with a herpesvirus. Australian Veterinary Journal, 54, 103–105.
- DOLL, E.R. & BRYANS, J.T., 1962. Incubation periods for abortion in equine viral rhinopneumonitis. Journal of the American Veterinary Medical Association, 141, 351–354.
- DOLL, E.R. & BRYANS, J.T., 1963. Epizootiology of equine viral rhinopneumonitis. Journal of the American Veterinary Medical Association, 142, 31–37.
- DOLL, E.R. & BRYANS, J.T., 1963. Immunization of young horses against viral rhinopneumonitis. Cornell Veterinarian, 53, 24–41.
- DOLL, E.R. & BRYANS, J.T., 1963. A planned infection program for immunizing mares against viral rhinopneumonitis. Cornell Veterinarian, 53, 249–262.
- DOLL, E.R., BRYANS, J.T., MCCOLLUM, W.H. & CROWE, E.W., 1956. Propagation of equine abortion virus in Syrian hamsters. Cornell Veterinarian, 46, 68–82
- DOLL, E.R., CROWE, M.W., BRYANS, J.T. & MCCOLLUM, W.h., 1955. Infection immunity in equine virus abortion. Cornell Veterinarian, 42, 505–509.
- DOLL, E.R., MCCOLLUM, W.H., BRYANS, J.T. & CROWE, M.W., 1959. Effect of physical and chemical environment on the viability of equine rhinopneumonitis virus propagated in hamsters. Cornell Veterinarian, 49, 75–81.
- DOLL, E.R., WALLACE, M.E. & RICHARDS, M.G., 1954. Thermal, hematological, and serological responses of weanling horses following inoculation with equine abortion virus: its similarity to equine influenza. Cornell Veterinarian, 44, 181–190.
- DONALDSON, M.T. & SWEENEY, C.R., 1997. Equine herpes myeloencephalopathy. Compendium of Continuing Education for the Practicing Veterinarian, 19, 864–871.
- DUTTA, S.K. & CAMPBELL, D.L., 1977. Cell mediated immunity in equine herpesvirus type 1 infection I. In vitro lymphocyte blastogenesis and serum neutralization antibody in normal parturient and aborting mares. Canadian Journal of Comparative Medicine, 41, 404–408.
- DUTTA, S.K., MYRUP, A. & BUMGARDNER, M.K., 1980. Lymphocyte responses to virus and mitogen in ponies during experimental infection with equine herpesvirus 1. American Journal of Veterinary Research, 41, 2066–2068.
- DUTTA, S.K. & SHIPLEY, W.D., 1975. Immunity and the level of neutralization antibodies in foals and mares vaccinated with a modified live-virus rhinopneumonitis vaccine. American Journal of Veterinary Research, 36, 445–448.
- EDINGTON, N. & BRIDGES, C.G., 1990. One way protection between equid herpesvirus 1 and 4 in vivo. Research in Veterinary Science, 48, 235–239.
- EDINGTON, N., BRIDGES, C.G. & HUCKLE, A., 1985. Experimental reactivation of equid herpesvirus 1 (EHV 1) following the administration of corticosteroids. Equine Veterinary Journal, 17, 369–372.
- EDINGTON, N., BRIDGES, C.G. & PATEL, J.R., 1986. Endothelial cell infection and thrombosis in paralysis caused by equid herpesvirus-1: Equine stroke. Archives of Virology, 90, 111–124.
- EDINGTON, N., CHESTERS, P., AZAM, H., WELCH, H., MCGLADDERY, A. & PUREWAL, A., 1994. Profiles of alphaherpesviruses in circulating leucocytes from Thoroughbred mares and foals using PCR and co-cultivation. In: NAKAJIMA, H. & PLOWRIGHT, W., (eds). Equine Infectious Diseases VII. Newmarket (Suffolk):R&W Publications, pp. 251–254
- EDINGTON, N., SMYTH, B. & GRIFFITHS, L., 1991. The role of endothelial cell infection in the endometrium, placenta and foetus of equid herpesvirus 1 (EHV-1) abortions. Journal of Comparative Pathology, 104, 379–387.
- EDINGTON, N., WELCH, H.M. & GRIFFITHS, L., 1994. The prevalence of latent Equid herpesviruses in the tissues of 40 abattoir horses. Equine Veterinary Journal, 26, 140–142.
- ELLIOTT, G. & O’HARE, P., 1995. Equine herpesvirus 1 gene 12, the functional homologue of herpes simplex virus VP16, transactivates via octamer sequences in the equine herpesvirus IE gene promoter. Virology, 213, 258–262
- ELLIS, J.A., BOGDAN, J.R., KANARA, E.W., MORLEY, P.S. & HAINES, D.M., 1995. Cellular and antibody responses to equine herpesviruses 1 and 4 following vaccination of horses with modified-live and inactivated viruses. Journal of the American Veterinary Medical Association, 206, 823–832.
- ELLIS, J.A., STEEVES, E., WRIGHT, A.K., BOGDAN, J.R., DAVIS, W.C., KANARA, E.W. & HAINES, D.M., 1997. Cell-mediated cytolysis of equine herpesvirus-infected cells by leukocytes from young vaccinated horses. Veterinary Immunology and Immunopathology, 57, 210–214.
- FITZPATRICK, D.R. & STUDDERT, M.J., 1984. Immunologic relationships between equine herpesvirus type 1 (equine abortion virus) and type 4 (equine rhinopneumonitis virus). American Journal of Veterinary Research, 45, 1947–1952.
- FRYMUS, T., KITA, J., WOYCIECHOWSKA, S. & GANOWICZ, M., 1986. Foetal and neonatal foal losses on equine herpesvirus type 1 (EHV-1) infected farms before and after EHV-1 vaccination was introduced. Polische Archiv Weter, 26, 7–14.
- FUKUSHI, H., TOMITA, T., TANIGUCHI, A., OCHIAI, Y., KIRISAWA, R., MATSUMURA, T., YANAI, T., MASEGI, T., YANAGUCHI, T. & HIRAI, K., 1997. Gazelle herpesvirus 1: A new neurotropic herpesvirus immunologically related to equine herpesvirus 1. Virology, 227, 34–44
- GALOSI, C.M., NORIMINE, J., ECHEVERRIA, M.G., OLIVA, G.A., NOSETTO, E.O., ETCHEVERRIGARAY, M.E., TOHYA, Y. & MIKAMI, T., 1998. Diversity of genomic electropherotypes of naturally occurring equine herpesvirus 1 isolates in Argentina. Brazilian Journal of Medical and Biological Research, 31, 771–774.
- GIBSON, J.S., O’NEILL, T., THACKRAY, A., HANNANT, D. & FIELD, H.J., 1992. Serological responses of specific pathogen-free foals to equine herpesvirus-1: Primary and secondary infection, and reactivation. Veterinary Microbiology, 32, 199–214.
- GIBSON, J.S., SLATER, J.D., AWAN, A.R. & FIELD, H.J., 1992. Pathogenesis of equine herpesvirus-1 in specific pathogen- free foals: Primary and secondary infections and reactivation. Archives of Virology, 123, 351– 366
- GIBSON, J.S., SLATER, J.D. & FIELD, H.J., 1992. The pathogenicity of Ab4p, the sequenced strain of equine herpesvirus-1, in specific pathogen-free foals. Virology, 189, 317–319.
- GILES, R.C., DONAHUE, J.M., HONG, C.B., TUTTLE, P.A., PETRITES MURPHY, M.B., POONACHA, K.B., ROBERTS, A.W., TRAMONTIN, R.R., SMITH, B. & SWERCZEK, T.W., 1993. Causes of abortion, stillbirth, and perinatal death in horses: 3,527 cases (1986–1991). Journal of the American Veterinary Medical Association, 203, 1170–1175.
- GILKERSON, J., JORM, L.R., LOVE, D.N., LAWRENCE, G.L. & WHALLEY, J.M., 1994. Epidemiological investigation of equid herpesvirus-4 (EHV-4) excretion assessed by nasal swabs taken from thoroughbred foals. Veterinary Microbiology, 39, 275–283.
- GILKERSON, J.R., LOVE, D.N., DRUMMER, H.E., STUDDERT, M.J. & WHALLEY, J.M., 1998. Seroprevalence of equine herpesvirus 1 in thoroughbred foals before and after weaning. Australian Veterinary Journal, 76, 677–682.
- GILKERSON, J.R., LOVE, D.N. & WHALLEY, J.M., 2000. Incidence of equine herpesvirus 1 infection in Thoroughbred weanlings on two stud farms. Australian Veterinary Journal, 78, 277–278
- GILKERSON, J.R., WHALLEY, J.M., DRUMMER, H.E., STUDDERT, M.J. & LOVE, D.N., 1999. Epidemiological studies of equine herpesvisrus 1 (EHV-1) in Thoroughbred foals: A review of studies conducted in the Hunter Valley of New South Wales between 1995 and 1997. Veterinary Microbiology, 68, 15–25.
- GILKERSON, J.R., WHALLEY, J.M., DRUMMER, H.E., STUDDERT, M.J. & LOVE, D.N., 1999. Epidemiology of EHV-1 and EHV-4 in the mare and foal populations on a Hunter Valley stud farm: Are mares the source of EHV-1 for unweaned foals? Veterinary Microbiology, 68, 27–34.
- GLEESON, L.J. & COGGINS, L., 1980. Response of pregnant mares to equine herpesvirus 1 (EHV1). Cornell Veterinarian, 70, 391–400.
- GRAY, W.L., BAUMANN, R.P., ROBERTSON, A.T., CAUGHMAN, G.B., O’CALLAGHAN, D.J. & STACZEK, J., 1987. Regulation of equine herpesvirus type 1 gene expression: Characterization of immediate early, early, and late transcription. Virology, 158, 79–87.
- GREENWOOD, R.E. & SIMSON, A.R., 1980. Pathological observations on an outbreak of paralysis in broodmares. Equine Veterinary Journal, 12, 118–126.
- GREENWOOD, R.E.S. & SIMPSON, A.R.B., 1980. Clinical report of a paralytic syndrome affecting stallions, mares and foals on a Thoroughbred studfarm. Equine Veterinary Journal, 12, 113–117
- GUPTA, A.K., SINGH, B.K. & YADAV, M.P., 1987. Changes in restriction enzyme pattern of the equine herpes virus type 1 (EHV-1) strain RacH DNA during attenuation. Zentralblatt für Veterinärmedizin, Reihe B, 34, 310–313.
- GUTEKUNST, D.E., MALMQUIST, W.A. & BECVAR, C.S., 1978. Antigenic relatedness of equine herpes virus types 1 and 3. Archives of Virology, 56, 33–45.
- HARTLEY, W.J. & DIXON, R.J., 1979. An outbreak of foal perinatal mortality due to equine herpesvirus type 1. Equine Veterinary Journal, 11, 215–218.
- HONG, C.B., DONAHUE, J.M., GILES, R.C., PETRITES MURPHY, M.B., POONACHA, K.B., ROBERTS, A.W., SMITH, B.J., TRAMONTIN, R.R., TUTTLE, P.A. & SWERCZEK, T.W., 1993. Equine abortion and stillbirth in central Kentucky during 1988 and 1989 foaling seasons. Journal of Veterinary Diagnostic Investigation, 5, 560–566.
- HORSERACE BETTING LEVY BOARD (UK), 2000. A common code of practice for the control of contagious equine metritis and other equine reproductive diseases, 1994: London.
- HOUSE, J.A., GREGG, D.A., LUBROTH, J., DUBOVI, E.J. & TORRES, A., 1991. Experimental equine herpesvirus-1 infection in llamas (Lama glama). Journal of Veterinary Diagnostic Investigation, 3, 137–143.
- HÜBERT, P.H., BIRKENMAIER, S., RZIHA, H.J. & OSTERRIEDER, N., 1996. Alterations in the equine herpesvirus type-1 (EHV-1) strain RacH during attenuation. Zentralblatt für Veterinärmedizin, Reihe B, 43, 1–14.
- IQBAL, J., PUREWAL, A.S. & EDINGTON, N., 2001. EHV-1 gene 63 is not essential for in vivo replication in horses and mice, nor does it affect reactivation in the horse. Acta Veterinaria Hungarica, 49, 473–478.
- JACKSON, T.A. & KENDRICK, J.W., 1971. Paralysis of horses associated with equine herpesvirus 1 infection. Journal of the American Veterinary Medical Association, 158, 1351–1357
- JACKSON, T.A., OSBURN, B.I., CORDY, D.R. & KENDRICK, J.W., 1977. Equine herpesvirus 1 infection of horses: Studies on the experimentally induced neurologic disease. American Journal of Veterinary Research, 38, 709–719.
- KENNEDY, M.A., RAMSEY, E., DIDERRICH, V., RICHMAN, L., ALLEN, G.P. & POTGIETER, L.N., 1996. Encephalitis associated with a variant of equine herpesvirus-1 in a Thomson’s gazelle. Journal of Zoo and Wildlife Medicine, 27, 533–538.
- KIRASAWA, R., ENDO, A., IWAI, H. & KAWAKAMI, Y., 1993. Detection and identification of equine herpesvirus-1 and -4 by polymerase chain reaction. Veterinary Microbiology, 36, 57–67.
- KIRISAWA, R., UI, S., TAKAHASHI, A., KAWAKAMI, Y. & IWAI, H., 1994. Comparison of the genomes of attenuated equine herpesvirus-1 strains with their parent virulent strain. Virology, 200, 651–660.
- KOHN, C.W. & FENNER, W.R., 1987. Equine herpes myeloencephalopathy. Veterinary Clinics of North America, Equine Practice, 3, 405–419.
- KYDD, J.H., SMITH, K.C., HANNANT, D., LIVESAY, G.J. & MUMFORD, J.A., 1994. Distribution of equid herpesvirus-1 (EHV-1) in the respiratory tract of ponies: Implications for vaccination strategies. Equine Veterinary Journal, 26, 466–469.
- KYDD, J.H., SMITH, K.C., HANNANT, D., LIVESAY, G.J. & MUMFORD, J.A., 1994. Distribution of equid herpesvirus-1 in the respiratory tract-associated lymphoid tissue: Implications for cellular immunity. Equine Veterinary Journal, 26, 470–473.
- LAWRENCE, G.L., GILKERSON, J., LOVE, D.N., SABINE, M. & WHALLEY, J.M., 1994. Rapid, single-step differentiation of equid herpesvirus 1 and 4 from clinical material using the polymerase chain reaction and virus-specific primers. Journal of Virological Methods, 47, 59–72.
- LEWIS, J.B., THOMPSON, Y.G. & CAUGHMAN, G.B., 1993. Transcriptional control of the equine herpesvirus 1 immediate early gene. Virology, 197, 788–792.
- LEWIS, J.B., THOMPSON, Y.G., FENG, X., HOLDEN, V.R., O’CALLAGHAN, D. & CAUGHMAN, G.B., 1997. Structural and antigenic identification of the ORF12 protein (alpha TIF) of equine herpesvirus 1. Virology, 230, 369–375.
- LITTLE, P.B. & THORSEN, J., 1976. Disseminated necrotizing myeloencephalitis: A herpes-associated neurological disease of horses. Veterinary Pathology, 13, 161–171.
- LITTLE, P.B., THORSEN, J. & MORAN, K., 1974. Virus involvement in equine paresis. The Veterinary Record, 95, 575.
- LUNN, D.P., HOLMES, M.A., GIBSON, J.S., FIELD, H.J., KYDD, J.H. & DUFFUS, W.P.H., 1991. Haematological changes and equine lymphocyte subpopulation kinetics during primary infection and attempted re-infection of specific pathogen free foals with EHV-1. Equine Veterinary Journal, Supplement, 12, 35–40.
- MACKIE, J.T., MACLEOD, G.A., REUBEL, G.H. & STUDDERT, M.J., 1995. Cellular and antibody responses to equine herpesviruses 1 and 4. Journal of the American Veterinary Medical Association, 207, 698–701.
- MACKIE, J.T., MACLEOD, G.A., REUBEL, G.H. & STUDDERT, M.J., 1996. Diagnosis of equine herpesvirus 1 abortion using polymerase chain reaction. Australian Veterinary Journal, 74, 390–391
- MATSUMURA, T., KONDO, T., SUGITA, S., DAMIANI, A.M., O’CALLAGHAN, D.J. & IMAGAWA, H., 1998. An equine herpesvirus type 1 recombinant with a deletion in the gE and gI genes is avirulent in young horses. Virology, 242, 68–79.
- MATSUMURA, T., O’CALLAGHAN, D.J., KONDO, T. & KAMADA, M., 1996. Lack of virulence of the murine fibroblast adapted strain, Kentucky A (KyA), of equine herpesvirus type 1 (EHV-1) in young horses. Veterinary Microbiology, 48, 353–365.
- MATSUMURA, T., SUGIURA, T., IMAGAWA, H., FUKUNAGA, Y. & KAMADA, M., 1992. Epizootiological aspects of type 1 and type 4 equine herpesvirus infections among horse populations. Journal of Veterinary Medical Science, 54, 207–211.
- MCCARTAN, C.G., RUSSELL, M.M., WOOD, J.L. & MUMFORD, J.A., 1995. Clinical, serological and virological characteristics of an outbreak of paresis and neonatal foal disease due to equine herpesvirus-1 on a stud farm. The Veterinary Record, 136, 7–12.
- MCCOLLOCH, J., WILLIAMSON, S.A., POWIS, S.J. & EDINGTON, N., 1993. The effect of EHV-1 infection upon circulating leucocyte populations in the natural equine host. Veterinary Microbiology, 37, 147–161.
- MCGEOCH, D.J., BARNETT, B.C. & MACLEAN, C.A., 1993. Emerging functions of alphaherpesvirus genes. Seminars in Virology, 4, 125–134.
- MEREDITH, D.M., STOCKS, J.M., WHITTAKER, G.R., HALLIBURTON, I.W., SNOWDEN, B.W. & KILLINGTON, R.A., 1989. Identification of the gB homologues of equine herpesvirus types 1 and 4 as disulphide-linked heterodimers and their characterisation using monoclonal antibodies. Journal of General Virology, 70, 1161–1172.
- MEYER, H., HUBERT, P.H. & EICHHORN, W., 1987. Changes in restriction enzyme pattern of the equine herpesvirus type 1 (EHV-1) strain Rac-H DNA during attenuation. Zentralblatt für Veterinärmedizin, Reihe B, 34, 310–313.
- MEYER, H., THEIN, P. & HÜBERT, P., 1987. Characterization of two equine herpesvirus (EHV) isolates associated with neurological disorders in horses. Zentralblatt für Veterinärmedizin, Reihe B, 34, 545–548.
- MONTALI, R.J., ALLEN, G.P., BRYANS, J.T., PHILLIPS, L.G. & BUSH, M., 1985. Equine herpesvirus type 1 abortion in an onager and suspected herpesvirus myelitis in a zebra. Journal of the American Veterinary Medical Association, 187, 1248–1249.
- MORLEY, P.S., 1995. Epidemiology of infectious upper respiratory tract disease in horses. Ph.D. Thesis, 1995, University of Saskatchewan: Saskatoon.
- MUMFORD, J.A., 1991. The epidemiology of equid herpesvirus abortion: a tantalizing mystery [editorial; comment]. Equine Veterinary Journal, 23, 77–78.
- MUMFORD, J.A., 1994. Equid herpesvirus 1 and 4 infections. In: COETZER, J.A.W., THOMSON, G.R. & TUSTIN, R.c., (eds). Infectious Diseases of Livestock with special reference to Southern Africa. Cape Town: Oxford University Press Southern Africa, pp. 911–925.
- MUMFORD, J.A. & BATES, J., 1984. Trials of an inactivated equid herpesvirus 1 vaccine: Challenge with a subtype 2 virus. The Veterinary Record, 114, 375–381.
- MUMFORD, J.A., HANNANT, D., JESSETT, D.M., O’NEILL, T., SMITH, K.C. & OSTLUND, E.N., 1994. Abortigenic and neurological disease caused by experimental infection with equid herpesvirus-1. In: PLOWRIGHT, W. & NAKAJIMA, H., (eds). Equine Infectious Diseases VII. Newmarket (Suffolk): R & W Publications, pp. 261–275.
- MUMFORD, J.A. & ROSSDALE, P.D., 1980. Virus and its relationship to the ‘poor performance’ syndrome. Equine Veterinary Journal, 12, 3–9.
- MUMFORD, J.A., ROSSDALE, P.D., JESSETT, D.M., GANN, S.J., OUSEY, J. & COOK, R.F., 1987. Serological and virological investigations of an equid herpesvirus 1 (EHV-1) abortion storm on a stud farm in 1985. Journal of Reproduction and Fertility Supplement, 35, 509–518
- NAGY, E., IDAMAKANTI, N. & CARMAN, S., 1997. Restriction endonuclease analysis of equine herpesvirus-1 isolates recovered in Ontario, 1986– 1992, from aborted, stillborn, and neonatal foals. Journal of Veterinary Diagnostic Investigation, 9, 143–148.
- OETTLER, D., KAADEN, O.-R. & NEUBAUER, A., 2001. The equine herpesvirus 1 UL45 homolog (ORF 15) encodes a glycosylated type II transmembrane protein and is involved in virus egress. Virology, 279, 302–312.
- OETTLER, D., KAADEN, O.-R. & NEUBAUER, A., 2001. The equine herpesvirus 1 UL45 homolog (ORF 15) encodes a glycosylated type II transmembrane protein and is involved in virus egress. Virology, 279, 302–312.
- O’KEEFE, J.S., ALLEY, M.R., JONES, D. & WILKS, C.R., 1995. Neonatal mortality due to equid herpesvirus 4 (EHV-4) in a foal. Australian Veterinary Journal, 72, 353–354.
- O’KEEFE, J.S., MURRAY, A., WILKS, C.R. & MORIARTY, K.M., 1991. Amplification and differentiation of the DNA of an abortigenic (type 1) and a respiratory (type 4) strain of equine herpesvirus by the polymerase chain reaction. Research in Veterinary Science, 50, 349–351.
- O’NEILL, T., KYDD, J.H., ALLEN, G.P., WATTRANG, E., MUMFORD, J.A. & HANNANT, D., 1999. Determination of equid herpesvirus 1-specific, CD8+, cytotoxic T lymphocyte precursor frequencies in ponies. Veterinary Immunology and Immunopathology, 70, 43–54
- OSTLUND, E.N., Antibody responses of horses to five major glycoproteins of equine herpesvirus 1 and specificity of responses to antigenic domains of glycoproteins B and C. Ph.D. Thesis, 1992, University of Kentucky: Lexington
- OSTLUND, E.N., 1993. The equine herpesviruses. Veterinary Clinics of North America, Equine Practice, 9, 283–294.
- PALFI, V. & CHRISTENSEN, L.S., 1995. Analyses of restriction fragment patterns (RFPs) and pathogenicity in baby mice of equine herpesvirus 1 and 4 (EHV-1 and EHV- 4) strains circulating in Danish horses. Veterinary Microbiology, 47, 199–204.
- PAPP VID, G. & DERBYSHIRE, J.B., 1978. the protective antigens of equine herpesvirus type 1. Canadian Journal of Comparative Medicine, 42, 219–226.
- PAPP VID, G. & DERBYSHIRE, J.B., 1979. The virus neutralizing activity of antibodies specific to the envelope and nucleocapsid of equine herpesvirus type 1. Canadian Journal of Comparative Medicine, 43, 231–233.
- PATEL, J.R., EDINGTON, N. & MUMFORD, J.A., 1982. Variation in cellular tropism between isolates of equine herpesvirus 1 in foals. Archives of Virology, 74, 41–51.
- PAWESKA, J.T., GERDES, T. & VAN HEERDEN, J., 1994. Serological relationship between a donkey alphaherpesvirus (isolate M7/91) and equid herpesvirus type 1 and 4. Journal of the South African Veterinary Association, 65, 64–66.
- PERDUE, M.L., KEMP, M.C., RANDALL, C.C. & O’CALLAGHAN, D.J., 1974. Studies on the molecular anatomy of the L-M cell strain of equine herpesvirus type 1: Proteins of the nucleocapsid and intact virion. Virology, 59, 201–216.
- PHILLIP, J.I., 1978. Equine rhinopneumonitis vaccine: Immunogenicity and safety in adult horses, including pregnant mares. American Journal of Veterinary Research, 39, 377–383.
- PLATT, H., SINGH, H. & WHITWELL, K.E., 1979. An outbreak of foal perinatal mortality due to equid herpesvirus type 1: Pathological observations. Equine Veterinary Journal, 11, 215–218.
- PLATT, H., SINGH, H. & WHITWELL, K.E., 1980. Pathological observations on an outbreak of paralysis in broodmares. Equine Veterinary Journal, 12, 118–126.
- PLUMMER, G., BOWLING, C.P. & GOODHEART, C.R., 1969. Comparison of four horse herpesviruses. Journal of Virology, 4, 738–741
- PLUMMER, G., GOODHEART, C.R. & STUDDERT, M.J., 1973. Equine herpesviruses: Antigenic relationships and deoxyribonucleic acid densities. Infection and Immunity, 8, 621–627.
- POWELL, D., 1992. Prevention, treatment of equine herpesvirus. Thoroughbred Times. January 31, 1992, p. 9.
- POWELL, D.G., 1991. Viral respiratory disease of the horse. Veterinary Clinics of North America: Equine Practice, 7, 27–52.
- PRICKETT, M.E., 1970. The pathology of disease caused by equine herpesvirus 1. In: BRYANS, J.T. & GERBER, H., (eds). Equine Infectious Diseases II. Basel: S. Karger, pp. 24–33.
- PURDY, C.W., PORTER, R.C. & FORD, S.J., 1978. Equine rhinopneumonitis vaccine: Immunogenicity and safety in foals. American Journal of Veterinary Research, 39, 745–752.
- PUREWAL, A.S., ALLSOPP, R., RIGGIO, M., TELFORD, E.A., AZAM, S., DAVISON, A.J. & EDINGTON, N., 1994. Equid herpesviruses 1 and 4 encode functional homologs of the herpes simplex virus type 1 virion transactivator protein, VP16. Virology, 198, 385–389.
- PUREWAL, A.S., IQBAL, J. & EDINGTON, N., 1998. The equid herpesvirus-1 gene 63 expressed as a leaky late gene (gamma1) transcript is non-essential for replication in vitro. Virus Research, 54, 189–195.
- PUREWAL, A.S., SMALLWOOD, A.V., KAUSHAL, A., AGEBOYE, D. & EDINGTON, N., 1992. Identification and control of the cis acting elements of the immediate early gene of equine herpesvirus type 1. Journal of General Virology, 73, 513–519.
- REBHUN, W.C., JENKINS, D.H., RIIS, R.C., DILL, S.G., DUBOVI, E.J. & TORRES, A., 1988. An epizootic of blindness and encephalitis associated with a herpesvirus indistinguishable from equine herpesvirus I in a herd of alpacas and llamas. Journal of the American Veterinary Medical Association, 192, 953–956
- RIMSTAD, E. & EVENSEN, O., 1993. The identification of equid herpesvirus 1 in paraffin-embedded tissues from aborted fetuses by polymerase chain reaction and immunohistochemistry. Journal of Veterinary Diagnostic Investigation, 5, 174–183.
- RIMSTAD, E. & HYLLSETH, B., 1994. Equine herpesviruses 1 and 4: Amplification and differentiation by polymerase chain reaction. Acta Veterinaria Scandinavica, 35, 303–306.
- ROIZMAN, B., DESROSIERS, R.C., FLECKENSTEIN, B., LOPEZ, C., MINSON, A.C. & STUDDERT, M.J., 1995. Herpesviridae. In: MURRAY, F.A., FAUCET, C.M., BISHOP, D.H.L., GHABRIAL, S.A., JARVIS, A.W., MATELLI, G.P., MAYO, M.A. & SUMMERS, M.D., (eds). Virus Taxonomy. Sixth Report of the International Committee on Taxonomy of Viruses. New York: Springer-Verlag, pp. 114–127.
- ROSSDALE, P.D., HOPES, R., WINGFIELD-DIGBY, N.J. & OFFORD, K., 1985. Epidemiological study of wastage among racehorses 1982 and 1983. The Veterinary Record, 116, 66–69.
- RUDOLPH, J. & OSTERRIEDER, N., 2002. Equine herpesvirus type 1 devoid of gM and gp2 is severely impaired in virus egress but not direct cell-to-cell spread. Virology, 293, 356–367.
- SAAXEGARD, F., 1966. Isolation and identification of equine rhinopneumonitis virus (equine abortion virus) from cases of abortion and paralysis. Nordiske Veterinaer Medicin, 18, 504–512.
- SAMUEL, C.A., ALLEN, W.R. & STEVEN, D.H., 1976. Studies on the equine placenta. II: Ultrastructure of the placental barrier. Journal of Reproduction and Fertility, 48, 257–264.
- SCHULTHEISS, P.C., COLLINS, J.K. & CARMAN, J., 1993. Use of an immunoperoxidase technique to detect equine herpesvirus- 1 antigen in formalin-fixed paraffin-embedded equine fetal tissues. Journal of Veterinary Diagnostic Investigation, 5, 12–15.
- SCOTT, J.C., DUTTA, S.K. & MYRUP, A.C., 1983. In vivo harboring of equine herpesvirus-1 in leukocyte populations and subpopulations and their quantitation from experimentally infected ponies. American Journal of Veterinary Research, 44, 1344–1348.
- SHARMA, P.C., CULLINANE, A.A., ONIONS, D.E. & NICOLSON, L., 1992. Diagnosis of equid herpesviruses 1 and 4 by polymerase chain reaction. Equine Veterinary Journal, 24, 20–25.
- SHIMIZU, M., SATOU, K. & NISHIOKA, N., 1989. Monoclonal antibodies with neutralising activity to equine herpesvirus 1. Archives of Virology, 104, 169–174.
- SHIMUZU, T., ISHIZAKI, R., ISHII, S., KAWAKAMI, Y., KAJI, R., SUGIMURA, K. & MATUMOTE, M., 1959. Isolation of equine abortion virus from natural cases of equine abortion in horse kidney cell culture. Japanese Journal of Experimental Medicine, 29, 643–649.
- SLATER, J.D., BAXI, M., TEWARI, D., GIBSON, J.S., FIELD, H.J., LUDWIG, H., STEINBACH, F. & BORCHERS, K., 1994. Experimental infection of specific pathogen free ponies with equid herpesvirus-1: Detection of infectious virus and viral DNA. In: NAKAJIMA, H. & PLOWRIGHT, W., (eds). Equine Infectious Diseases VII. Newmarket (Suffolk):R&W Publications, pp. 255–260.
- SLATER, J.D., BORCHERS, K., THACKRAY, A.M. & FIELD, H.J., 1994. The trigeminal ganglion is a location for equine herpesvirus 1 latency and reactivation in the horse. Journal of General Virology, 75, 2007–2016.
- SLATER, J.D., GIBSON, J.S., BARNETT, K.C. & FIELD, H.J., 1992. Chorioretinopathy associated with neuropathology following infection with equine herpesvirus-1. The Veterinary Record, 131, 237–239.
- SMITH, D.J., THE INTERACTIONS OF EHV-1 INFECTED LEUCOCYTES AND EQUINE ENDOTHELIAL CELLS. PH.D. THESIS, 1998, University of London: London.
- SMITH, D.J., IQBAL, J., PUREWAL, A., HAMBLIN, A.S. & EDINGTON, N., 1998. In vitro reactivation of latent equid herpesvirus-1 from CD5+/CD8+ leukocytes indirectly by IL-2 or chorionic gonadotrophin. Journal of General Virology, 79, 2997–3004.
- SMITH, K.C., MCGLADDERY, A.J., BINNS, M.M. & MUMFORD, J.A., 1997. Use of trans-abdominal ultrasound-guided aminocentesis for detection of equid herpesvirus 1-induced fetal infection in utero. American Journal of Veterinary Research, 58, 997–1002.
- SMITH, K.C., MUMFORD, J.A., HANNANT, D. & WHITWELL, K.E., 1999. A comparison between the pathogenicity of EHV-1 isolates of high and low abortigenic potential in the natural host and in the mouse model. In: WERNERY, U., WADE, J.F., MUMFORD, J.A. & KAADEN, O.-R., (eds). Equine Infectious Diseases VIII. Newmarket (Suffolk):R&W Publications, pp. 581–582.
- SMITH, K.C., MUMFORD, J.A. & LAKHANI, K., 1996. A comparison of equid herpesvirus 1 (EHV-1) vascular lesions in the early versus late pregnant equine uterus. Journal of Comparative Pathology, 114, 231–247.
- SMITH, K.C., WHITWELL, K.E., BINNS, M.M., DOLBY, C.A., HANNANT, D. & MUMFORD, J.A., 1992. Abortion of virologically negative foetuses following experimental challenge of pregnant pony mares with equid herpesvirus 1. Equine Veterinary Journal, 24, 256–259.
- SMITH, K.C., WHITWELL, K.E., MUMFORD, J.A., GOWER, S.M., HANNANT, D. & TEARLE, J.P., 1993. An immunohistological study of the uterus of mares following experimental infection by equid herpesvirus 1. Equine Veterinary Journal, 25, 36–40.
- SMITH, R.H., CAUGHMAN, G.B. & O’CALLAGHAN, D.J., 1992. Characterization of the regulatory functions of the equine herpesvirus 1 immediate-early gene product. Journal of Virology, 66, 936–945.
- SMITH, R.H., ZHAO, Y. & O’CALLAGHAN, D.J., 1994. The equine herpesvirus type 1 immediate-early gene product contains an acidic transcriptional activation domain. Virology, 202, 760–770.
- STACZEK, J., ATHERTON, S.S. & O’CALLAGHAN, D.J., 1983. Genetic relatedness of the genomes of equine herpesvirus types 1, 2, and 3. Journal of Virology, 45, 855–858.
- STEAR, R.L., BASS, E.P. & BECKENHAUER, W.H., 1976. Letter: Efficacy of modified live-virus equine rhinopneumonitis vaccine. American Journal of Veterinary Research, 37, 349–351.
- STOKES, A., CORTEYN, A.H. & MURRAY, P.K., 1991. Clinical signs and humoral immune response in horses following equine herpesvirus type-1 infection and their susceptibility to equine herpesvirus type-4 challenge. Research in Veterinary Science, 51, 141–148.
- STOKES, A. & WARDLEY, R.C., 1988. ADCC and complement-dependent lysis as immune mechanisms against EHV-1 infection in the horse. Research in Veterinary Science, 44, 295–302.
- STUDDERT, M.J., 1983. Restriction endonuclease DNA fingerprinting of respiratory, foetal and perinatal foal isolates of equine herpesvirus type 1. Archives of Virology, 77, 249–258.
- STUDDERT, M.J. & BLACKNEY, M.H., 1979. Equine herpesviruses: on differentiation of respiratory from foetal strains of type 1. Australian Veterinary Journal, 55, 488–492
- STUDDERT, M.J., CRABB, B.S. & FICORILLI, N., 1992. The molecular epidemiology of equine herpesvirus 1 (equine abortion virus) in Australasia 1975 to 1989. Australian Veterinary Journal, 69, 104–111.
- STUDDERT, M.J., FITZPATRICK, D.R., BROWNING, G.F., CULLINANE, A.A. & WHALLEY, J.M., 1986. Equine herpesvirus genomes: Heterogeneity of naturally occurring type 4 isolates and of a type 1 isolate after heterologous cell passage. Archives of Virology, 91, 375–381.
- SUTTON, G.A., VIEL, L., CARMAN, P.S. & BOAG, B.L., 1998. Pathogenesis and clinical signs of equine herpesvirus 1 in experimentally infected ponies in vivo. Canadian Journal of Veterinary Research, 62, 49–55.
- TANIGUCHI, A., FUKUSHI, H., MATSUMURA, T., YANAI, T., MASEGI, T. & HIRAI, K., 2000. Pathogenicity of a new neurotropic equine herpesvirus 9 (gazelle herpesvirus 1) in horses. Journal of Veterinary Medical Science, 62, 215–218.
- TEARLE, J.P., SMITH, K.C., BOYLE, M.S., BINNS, M.M., LIVESAY, G.J. & MUMFORD, J.A., 1996. Replication of equid herpesvirus 1 (EHV-1) in the testes and epididymes of ponies and venereal shedding of infectious virus. Journal of Comparative Pathology, 115, 385–397.
- TELFORD, E.A., WATSON, M.S., MCBRIDE, K. & DAVISON, A.J., 1992. The DNA sequence of equine herpesvirus 1. Virology, 189, 304–316.
- TELFORD, E.A., WATSON, M.S., PERRY, J., CULLINANE, A.A. & DAVISON, A.J., 1998. The DNA sequence of equine herpesvirus 4. Journal of General Virology, 79, 1197–1203.
- THEIN, P., 1981. Infection of the central nervous system of horses with equine herpesvirus serotype 1. Journal of the South African Veterinary Association, 52, 239–241.
- THOMSON, G.R., MUMFORD, J.A., CAMPBELL, J., GRIFFITHS, L. & CLAPHAM, P., 1976. Serological detection of equid herpesvirus 1 infections of the respiratory tract. Equine Veterinary Journal, 8, 58–65.
- THORSEN, J. & LITTLE, P.B., 1975. Isolation of equine herpesvirus type 1 from a horse with an acute paralytic disease. Canadian Journal of Comparative Medicine, 39, 358–359.
- TURTINEN, L., 1983. Studies on the antigenic and genetic variation between the two subtypes of equine herpesvirus 1. Ph.D. Thesis, 1983, University of Kentucky: Lexington.
- TURTINEN, L.W. & ALLEN, G.P., 1982. Identification of the envelope surface glycoproteins of equine herpesvirus type 1. Journal of General Virology, 63, 481–485.
- TURTINEN, L.W., ALLEN, G.P., DARLINGTON, R.W. & BRYANS, J.T., 1981. Serologic and molecular comparisons of several equine herpesvirus type 1 strains. American Journal of Veterinary Research, 42, 2099–2104.
- VAN DER MEULEN, K.M., NAUWYNCK, H.J., BUDDAERT, W. & PENSAERT, M.B., 2000. Replication of equine herpesvirus type 1 in freshly isolated equine peripheral blood mononuclear cells and changes in susceptibility following mitogen stimulation. Journal of General Virology, 81, 21–25
- VAN MAANEN, C., VREESWIJK, J., MOONEN, P., BRINKHOF, J., DE BOER-LUIJTZE, E. & TERPSTRA, C., 2000. Differentiation and genomic and antigenic variation among fetal, respiratory, and neurological isolates from EHV-1 and EHV-4 infections in The Netherlands. Veterinary Quarterly, 22, 88–93.
- VARRASSO, A., DYNON, K., FICORILLI, N., HARTLEY, C.A., STUDDERT, M.J. & DRUMMER, H.E., 2001. Identification of equine herpesviruses 1 and 4 by polymerase chain reaction. Australian Veterinary Journal, 79, 563–569.
- VERHEYEN, K., NEWTON, J.R., WOOD, J.L.N., BIRCH-MACHIN, I. & HANNANT, D., 1998. Possible case of EHV-4 ataxia in warmblood mare. The Veterinary Record, 143, 456.
- VICTORIAN BLOOD HORSE BREEDERS ASSOCIATION, INC., 1994. The Code of Practice for the Control of Equine Herpesvirus 1 Disease in Australia, 1994
- WAGNER, W.N., BOGDAN, J., HAINES, D., TOWNSEND, H.G. & MISRA, V., 1992. Detection of equine herpesvirus and differentiation of equine herpesvirus type 1 from type 4 by the polymerase chain reaction. Canadian Journal of Microbiology, 38, 1193–1196.
- WELCH, H.M., BRIDGES, C.G., LYON, A.M., GRIFFITHS, L. & EDINGTON, N., 1992. Latent herpesviruses 1 and 4: Detection and distinction using the polymerase chain reaction and co-cultivation from lymphoid tissues. Journal of General Virology, 73, 261–268.
- WESTERFIELD, C. & DIMOCK, W.W., 1946. The pathology of equine virus abortion. Journal of the American Veterinary Medical Association, 109, 101–111.
- WHITWELL, K.E. & BLUNDEN, A.S., 1992. Pathological findings in horses dying during an outbreak of the paralytic form of Equid herpesvirus type 1 (EHV-1) infection. Equine Veterinary Journal, 24, 13–19.
- WHITWELL, K.E., GOWER, S.M. & SMITH, K.C., 1992. An immunoperoxidase method applied to the diagnosis of equine herpesvirus abortion, using conventional and rapid microwave techniques. Equine Veterinary Journal, 24, 10–12.
- WHITWELL, K.E., SMITH, K.C., SINCLAIR, R. & MUMFORD, J.A., 1994. Foetal lesions in spontaneous EHV-4 abortions in mares. In: PLOWRIGHT, W. & NAKAJIMA, H., (eds). Equine Infectious Diseases VII. Newmarket (Suffolk): R & W Publications, pp. 354.
- WILKS, C.R., 1977. In vitro cytotoxicity of serum and peripheral blood leukocytes for equine herpesvirus type 1-infected target cells. American Journal of Veterinary Research, 38, 117–121.
- WILKS, C.R. & COGGINS, L., 1976. Immunity to equine herpesvirus type 1 (rhinopneumonitis): In vitro lymphocyte response. American Journal of Veterinary Research, 37, 486–492.
- WILKS, C.R. & STUDDERT, M.J., 1976. Equine herpesviruses. 6. Sequential infection of horses with types 2, 3 and 1. Australian Veterinary Journal, 52, 199–203.
- WILSON, W.D., 1997. Equine herpesvirus 1 myeloencephalopathy. Veterinary Clinics of North America, Equine Practice, 13, 53–72.
- WILSON, W.D. & ROSSDALE, P.D., 1999. Effect of age on the serological responses of Thoroughbred foals to vaccination with an inactivated equine herpesvirus EHV-1/EHV-4 vaccine. In: WERNERY, U., WADE, J.F., MUMFORD, J.A., & KAADEN, O.-R., (eds). Equine Infectious Diseases VIII. Newmarket (Suffolk):R&W Publications, p. 428.
- WOLFF, P.L., MEEHAN, T.P., BASGALL, E.J., ALLEN, G.P. & SUNDBERG, J.P., 1986. Abortion and perinatal foal mortality associated with equine herpesvirus type 1 in a herd of Grevy’s zebra. Journal of the American Veterinary Medical Association, 189, 1185–1186.
- WOOD, J.L.N., NEWTON, J.R., CHANTER, N., MUMFORD, J.A., TOWNSEND, H.G.G., LAKHANI, K.H., GOWER, S.M., BURRELL, M.H., PILSWORTH, R.C., SHEPHARD, M., HOPES, R., DUGDALE, D., HERINCKX, B.M.B., MAIN, J.P.M., WINDSOR, H.M. & WINDSOR, G.G., 1999. A longitudinal epidemiological study of respiratory disease in racehorses: Disease definitions, prevalence and incidence. In: WERNERY, U., WADE, J., MUMFORD, J. & KAADEN, O.-R., (eds). Equine Infectious Diseases VIII. Newmarket (Suffolk):R&W Publications, pp. 69–70.
- YEARGAN, M.R., ALLEN, G.P. & BRYANS, J.T., 1985. Rapid subtyping of equine herpesvirus 1 with monoclonal antibodies. Journal of Clinical Microbiology, 21, 694–697
- ZHAO, Y., HOLDEN, V.R., SMITH, R.H. & O’CALLAGHAN, D.J., 1995. Regulatory function of the equine herpesvirus 1 ICP27 gene product. Journal of Virology, 69, 2786–2793.
- ZIENTARA, S., PLATEAU, E., SAILLEAU, C. & CRUCIERE, C., 1993. Comparison of the restriction patterns of equine herpesvirus-1 (EHV-1) strains isolated for eight years in France. Zentralblat für Veterinärmedizin, Reihe B, 40, 501–507.






