- Infectious Diseases of Livestock
- Part 2
- Rinderpest
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Rinderpest
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Classical rinderpest is an acute to subacute, highly contagious viral disease of cattle and other artiodactyles. The main gross pathological changes are necrosis and erosion throughout the gastrointestinal tract, which can cause diarrhoea, dehydration, and death. Epidemics of the severe ‘cattle plague’ form of the disease may have morbidity and mortality rates exceeding 90 per cent. However, mild and virtually inapparent infections with very low mortality rates also occur and are perhaps the more typical form in endemic areas.
The decimation of cattle and wildlife populations by rinderpest has influenced human history,119, 180, 235 and the ecology of large areas of southern and central Africa.369, 376 So serious were the effects of the disease that its control and eradication were major stimuli for the establishment of the first veterinary schools in Europe during the eighteenth century.
Historically, the disease invaded Europe with armies from the Orient, encouraging the view that the disease originated in Asia. Antigenic comparison between morbilliviruses has suggested that rinderpest may be the archetypal morbillivirus from which measles and distemper evolved five thousand or more years ago, possibly in the river civilizations of south-west Asia where the domestication of cattle is thought to have occurred.109, 233
Introductions of rinderpest into Europe, usually as a sequel to wars, were often followed by major epidemics. By the end of the nineteenth century other forms of transport began to replace oxen as the main source of military draught power, effectively removing this source of infection. Strict application of slaughter and quarantine eradicated rinderpest from most of Europe, though a series of outbreaks occurred during and immediately after the First World War, most notably in Poland. The last reported outbreaks in Europe were in Belgium in 1920 and Rome zoo in 1949, both of which were caused by the importation of live infected animals from high-risk areas.15, 79 The only recorded outbreaks in the New World, in Brazil in 1920 and Australia in 1923, were also due to the importation of live infected animals. 294, 295, 407
After the eradication of the disease from Europe the scientific study of rinderpest continued in Asia where the disease was widespread in the first quarter of the twentieth century. The first vaccine that allowed large-scale prophylactic immunization of rinderpest was developed in India in 1925 by Edwards,106 allowing the possibility of mass control and even eradication of the disease in countries where strict control of animal movement or even slaughter were unfeasible. The vast and comparatively unconfined cattle population and even larger goat and sheep populations of India continued to pose an almost insuperable problem for vaccination and control, even with the advent of improved vaccines.
Finally, after focused programmes in the 1980s and 1990s, rinderpest was eradicated from the country in the mid-1990s, setting the example for the rest of Asia and for Africa. Unfortunately, in Asia, at the time of writing, Pakistan remains endemically infected and, although the prevalence of the disease has decreased significantly in the past decade, widespread traffic in live animals still poses a threat to southern Asia and the Middle East.219, 304 Effective control in the Near and Middle East decreased during the 1980s because of reduced vaccination and increased movement of cattle resulting from civil and military disturbances. Fortunately, the 1990s saw renewed control and it is increasingly likely that there are no endemic foci in this region.1, 20
In Africa the disease was introduced to North Africa, especially Egypt, and possibly to West Africa on a number of occasions before 1887.87, 112, 211 If it reached West Africa it apparently failed to establish itself there, and if it persisted in Egypt it was effectively isolated from the rest of Africa’s cattle. For most historians the history of rinderpest on the continent begins with the ‘Great African Pandemic’ of 1887 to 1897, which was widely described in southern Africa and Ethiopia.135, 147, 202, 387, 388 Although the source of the virus that started the pandemic is still unknown, there are several possibilities, all classically associated with military campaigns. The most frequently quoted possibility is the introduction of infected Indian or Arabian cattle to Massawa in Eritrea by the Italian army in either 1887, 1888 or 1889.194, 424 The inefficient attempts to use prophylactic and therapeutic control rather than slaughter and quarantine, 424 made it easier for the disease to establish itself. Infection was then transported throughout Ethiopia and into the Nile Valley in Sudan in 1890. Alternatively, the virus may have moved slowly up the Nile from Egypt in cattle used by British military campaigns, which reputedly brought rinderpest to Khartoum in 1884 and 1885. Whichever is true, it seems likely that the disease was not present in this area before the 1880s when the American adventurer James137 encountered numerous herds of healthy African buffalo (Syncerus caffer) as he travelled between the Nile and Massawa in 1881. All subsequent work has shown that the African buffalo is probably the most susceptible species to rinderpest. Another possible origin of the pandemic was suggested by Littlewood,194 who reported that in late 1889 a German military expedition brought cattle from Aden and Bombay against his advice, instigating a severe outbreak of rinderpest on the coast of what is today Kenya and Tanzania. It may have been virus from this outbreak, carried in cattle which the Maasai say were brought back to Kilimanjaro from the coast by their warriors in late 1890, that was responsible for the outbreak at Oloitokitok in early 1891. Suggestions202, 323 that Joseph Thompson encountered rinderpest among the Laikipia Maasai in 1883 overlook the author’s description of the severe epidemic disease he saw in cattle. His book records, as Theiler also noted,376 a convincing post-mortem description of contagious bovine pleuropneumonia, which was widespread in the area some six to seven years before the outbreak of rinderpest.133
Whatever its source, the pandemic was raging in Kenya, Uganda and Tanzania in 1891. Moving south in 1892, it had reached Karonga on Lake Nyasa by July and the Luapala River in Zambia by October. Very high mortality was reported not only in cattle but also in wildlife, though alcelaphines and waterbuck (Kobus ellipsiprymnus) seem to have been relatively insusceptible. The southerly spread of the virus was halted by the Zambezi River, where the disease, which had not yet been fully confirmed as rinderpest, came to be called Zambezi cattle fever. During this period of southerly advance the virus also may have spread westward into Angola and Zaire, whilst further north the western extension of the pandemic had penetrated right through sub- Saharan West Africa to the Atlantic Ocean.
Eventually the virus was transported across the Zambezi River. Disease was reported south of the river in February 1896 and appeared at Bulawayo in Zimbabwe in March, where it was confirmed by Gray and Theiler, and in the same month at Palapye in Botswana. Soon afterwards the mostnorthern provinces of South Africa were infected via the transport routes (Figure 49.1). stretching through Botswana and Mafikeng, and in October the disease spread south through Kimberley in the Cape Colony to the Orange (now the Gariep) River by December. There it was temporarily halted not only by the river, but by a 1 600-kilometre-long fence and rigorous quarantine measures (Figure 49.2). Unfortunately, in March 1897, a drover crossed the river reputedly wearing a pair of trousers he had found in a sack containing meat. Within a few days his cattle developed rinderpest and so the virus moved swiftly southwards. During 1897 the disease also infected the Natal Colony (present-day KwaZulu-Natal), Lesotho and the region of present-day Namibia.
Wild ungulates were reputedly also a factor in the spread of the disease. President Paul Kruger of the then Zuid Afrikaansche Republiek (now comprising the four most northerly provinces of South Africa) was of the opinion that game were the most important spreaders of infection, with bushbuck (Tragelaphus scriptus), impala (Aepycerosmelampus) and kudu (Tragelaphus strepsiceros) being the main culprits.180
Energetic control campaigns which included newly developed prophylactic inoculations eventually contained the disease and it was briefly eradicated from South Africa in 1899. A re-introduction from Namibia in 1901 was milder and easier to control, and southern Africa was reported to be free of rinderpest in 1905.383
It was the losses incurred during the pandemic — 5,25 million cattle and innumerable wildlife south of the Zambezi River — which have most dramatically illustrated the killing power of rinderpest. In the Zuid Afrikaansche Republiek alone there were 1 556 760 cattle prior to the rinderpest, of which 1 002 297 (66,5 per cent) died from or were killed as a result of the disease. A total of 205 801 (13,2 per cent) was said to have recovered from the infection while the rest (20,3 per cent) was unaffected. The direct financial losses were estimated to total 3 006 891 pounds sterling.180 In the Natal Colony, 46 per cent of the cattle population was lost, but the highest mortality rate in southern Africa occurred in Matabeleland (Zimbabwe), where it reached 90 per cent.180
The pandemic also left its mark on the continent in other ways. In East Africa dominant cattle-keeping tribes such as the Maasai were irrevocably weakened and replaced politically and economically by agricultural tribes. Further south, the disease and the stern measures that were used to fight it provoked rebellions in Matabeleland and Lesotho. In South Africa not only were stock owners, both black and white, ruined financially, but the whole transport system virtually collapsed, further contributing to the impoverishment and resultant urbanization of the Afrikaners.180
The decimation of wildlife and cattle led to the apparent disappearance of Glossina spp. from many areas of southern Africa. The comparatively swift eradication of rinderpest from the subcontinent allowed wildlife in the region to recover their numbers and distribution, whereas the persistence of the virus further north may have contributed to the formation of isolated ‘island’ communities of species such as sable (Hippotragus niger) and greater kudu (T. strepsiceros).
The pandemic set the stage for one of the keenest competitions medical science has ever seen. The southern African republics and colonies each set about finding a method of curing or preventing the disease. The race became even more significant when the Russian government offered a prize of one million roubles for the winner. Drs Arnold Theiler of the Zuid Afrikaansche Republiek and Herbert Watkins-Pitchford of the Natal Colony collaborated from October 1886 to March 1887 in developing a technique for immunizing cattle with simultaneous but separate inoculations of immune serum and infectious virus (which was used until the advent of attenuated vaccines in the 1930s). In December 1886 Dr Robert Koch, the eminent German microbiologist, arrived at the request of the government of the Cape Colony to begin work at Kimberley.
He lost no time in evaluating, among other things, the Boer farmers’ preventive remedy of inoculating bile from dead cattle into healthy animals. Koch refined the technique to a stage where it was successful in experienced hands at the laboratory, at which point it was hastily introduced throughout the affected areas. With hindsight it is easy to criticize the bile method for being a poor way of immunizing cattle and for spreading the virus, but the experiments of Theiler and Watkins-Pitchford were barely complete and immune serum was in desperately short supply and time-consuming to produce. Undoubtedly the bile method helped to reduce the mortality in the later stages of the pandemic.
The Zuid Afrikaansche Republiek government also obtained the services of the distinguished French microbiologists, Drs Jean Danysz and Jules Bordet, who arrived to begin work in January 1897. Theiler was ordered to join them and they eventually reported jointly to the government on the success of the serum-virus method of immunization, which then gradually replaced the bile method. Watkins-Pitchford was less fortunate: the government of the Natal Colony largely ignored his results and his data were destroyed in a fire at Pietermaritzburg town hall. The million roubles were never officially awarded.
Elsewhere in Africa rinderpest persisted wherever nomadic or semi-nomadic pastoralism was common. Such husbandry involves keeping large herds of cattle that may range over thousands of square kilometres and are frequently inaccessible for vaccination. Southern Africa has been under constant potential threat from the north, but the threat has assumed real proportions only occasionally. Usually the danger has arisen from a southerly spread of the disease through Tanzania along natural stock routes towards the gap between Lakes Tanganyika and Malawi. This occurred during the First and Second World Wars,59, 89, 126, 135, 196 but was contained by vigorous control of cattle movement and vaccination of all stock in the belt between the lakes. Implementation of a ‘cordon sanitaire’ involving, to varying degrees, the elimination of all domestic stock and susceptible species of wildlife from a 6- to 40-kilometre-wide strip and the erection of game-proof fencing was also carried out.379 In 1983 and 1984, this same region was again the target of co-ordinated vaccination campaigns by the governments of Tanzania, Zambia and Malawi. Their aim was to generate a cordon of immune animals that would halt the passage of disease from what can now be regarded as the second Great African Pandemic. Fortunately the effectiveness of this immune belt was never tested as the virus did not spread south of the central railway line in Tanzania, and southern Africa escaped the losses and disruption experienced by less fortunate countries further north.
The progress of the second Africanpandemic(1979 to 1984) attracted considerable attention, though accurate details of the origin of primary outbreaks and subsequent dissemination of the virus were often as elusive as they had been in the first pandemic 90 years earlier.128, 132, 228, 230, 271, 310, 314 The underlying causes, however, were easy to discern. The outstanding success of vaccination campaigns using cell-culture attenuated rinderpest virus led to an impressive fall in the number of outbreaks of rinderpest in Africa in the mid-1970s (Figure 49.3). Joint Project 15 (JP15) of the Organization of African Unity, Food and Agriculture Organization of the United Nations, and the European Community, assisted by individual donor countries including African states such as Nigeria, was especially instrumental in this control.21 The virus was eradicated throughout vast areas of the continent and by 1976 was apparently restricted to two zones where access and animal movement control had always been difficult: one in West Africa between the Mali/Mauritania border and the internal delta of the Niger, and the other in East Africa including Ethiopia and Sudan (Figure 49.3).
When JP15 ceased in 1976, it left behind a strong sense of security but no funds to maintain the high levels of herd immunity it had induced. Coinciding with rising fuel prices, the effects of a recession in the world economy on the emergent African nations were particularly hard. With rinderpest having been eradicated from many countries for over a decade, cutting back vaccination against the virus was an obvious way to save money for more pressing matters. Some countries ceased vaccination completely while others covered only a small proportion of their cattle.
The outcome was that the number of susceptible cattle started to increase as soon as JP15 ceased and, despite warnings, 271, 336 the virus began to spread from its endemic strongholds. Motor transport facilitated rapid spread of infection, and civil and military strife in many parts of the continent hindered control. The virus rapidly regained most of the territory it had lost during JP15 (Figure 49.3). Fortunately, in one of the most successful emergency campaigns ever seen, this second pandemic was controlled by vaccination carried out by national veterinary departments supported by international funds totalling well over US$ 20 million. The emergency provoked renewed calls for a campaign to eradicate rinderpest from Africa completely.19, 152, 228 From 1986 until 1999, the Pan African Rinderpest Campaign (PARC), funded by the European Community, and advised and assisted by various other donors and agencies, attempted this. Building on the successes of the national campaigns of the 1980s this regional programme facilitated further eradication in several parts of the continent, most notably Ethiopia. Unfortunately, inaccessibility of cattle populations in some of Africa’s chronically unstable, insecure and most inhospitable areas, especially southern Sudan and the southern Somali pastoral ecosystem in Somalia and Kenya (Figure 49.3), prevented PARC from achieving its objective.78, 189 New campaigns are now attempting to eradicate these remaining African foci of infection, and the one remaining Asian focus in Pakistan.
Extensive descriptions of the history and all aspects of rinderpest are covered in numerous reviews.18, 87, 119, 135, 159, 206, 252, 333, 334, 337, 338
Aetiology
In 1902 Nicolle and Adil Bey showed that rinderpest was caused by a filterable agent. It was grown in whole blood cultures by Boynton,55 and Takematsu and Morimoto367 detected increased amounts of infectivity in cultured leukocytes derived from infected animals. After earlier unsuccessful attempts to grow the agent in cell culture,76 Plowright and Ferris258–260 demonstrated cytopathic effects and new infectivity in bovine cell monolayers. Subsequent studies revealed the close antigenic relationship of rinderpest virus (RPV) to canine distemper267, 409 and measles viruses,153, 259, 404 and led to RPV being included in the measles-rinderpest-distemper subgroup of the Paramyxoviridae. This subgroup, reclassified as the genus Morbillivirus, 175 also includes the viruses that cause peste des petits ruminants (PPR) in sheep and goats, phocine and cetacean distemper,85, 203 and hedgehog distemper.386

Figure 49.4 Dendrogram of rinderpest virus. (By kind permission of T. Barrett, 2001. Morbilliviruses: dangers of old and new. In: Smith, G.L., McCauley, J.W. & Rowlands, D.J. (eds). New Challenges to Health: The Threat of Virus Infections. Society for General Microbiology. Cambridge: Cambridge University Press)
The virus is pleomorphic with roughly spherical (100 to 300 nm in diameter) and tubular forms (up to 1 × 103 nm in length).60, 257 The enveloped virus is readily inactivated by lipid solvents and is sensitive to light, ultraviolet radiation, heat and extremes of pH.252, 328, 329 Infectivity is destroyed by most disinfectants and chemicals such as β-propiolactone. 158, 363, 399 Glycerol also destroys RPV and must not be used in transport media for diagnostic samples.169, 178
The virus contains a negative strand of ribonucleic acid, approximately 15 kilo base pairs long, which codes for eight polypeptides.38, 43, 104, 127, 182, 324 The viral envelope contains the H protein (a haemagglutinin in other morbilliviruses), which binds to cell receptors, and the fusion (F) protein that induces the formation of syncytia, and is underlain by the matrix (M) protein. Within the virion the RNA is protected by a nucleocapsid (N) protein. Transcription in infected cells is mediated by polymerase (L) and polymerase- associated (P) proteins. A non-structural polypeptide (C) is translated by modification from the P polypeptide mRNA.38
The main advance in rinderpest research since the first edition of this book has been the molecular dissection of the viral genome and the benefits this has brought. The nucleotide sequences of all the genes have now been sequenced and most have been compared to their equivalents in other morbilliviruses.30, 31, 35, 37, 111, 192, 382, 414, 415 The P, N, and L genes appear to be more conserved amongst the morbilliviruses and the F andHgenes more variable. Comparison of a region of the F gene can distinguish between different strains of the virus, which has allowed the virus to be ‘taxonomically’ classified into a number of subtypes or lineages. The main contemporary lineages (Figure 49.4). are Asia, Africa-1 and Africa-2, and there are a number of ancestral lines that have been maintained in vaccines.9, 41, 400 Refined subtyping within these lineages is now allowing molecular epidemiology to make significant contributions to the understanding of the persistence and distribution of different clades of the virus in Africa and Asia.39, 41, 400 However, most of the differences are small; for instance, the difference in the sequences of the complete genome of the modified live cell culture vaccine and its highly virulent parent stock, Kabete O strain, is only 87 base pairs or 0,55 per cent.18 Fortunately, the antigenic differences between the different lineages and clades are also very small and the immunity induced by the vaccine protects against all known isolates of the virus. Determination of the gene sequences has facilitated the construction of new experimental recombinant vaccines and diagnostic reagents.40, 44, 49, 123, 155, 168, 231, 299, 364, 417, 420, 421 Most of the earlier recombinant vaccines inserted rinderpest genes into larger DNA vectors such as pox viruses. The recovery of viable virus from cloned cDNA copies of the RNA genome has facilitated the development of a new generation of RPV recombinants in which foreign genes can be introduced into the virus genome.32, 121, 122
In vitro, RPV grows relatively slowly in cells derived from bovine tissues and in cell lines such as Hela and Vero, producing titres of cell-free virus of 105 to 106 (occasionally 107) log10 tissue culture infectious doses (TCID50) per millilitre of fluid.212, 259 Cytopathic effects comprise rounding of cells which become refractive, cytomegaly, cytoplasmic stranding, the formation of small and large syncytia and eventual degeneration of the cell sheet. In culture the virus is capable of producing plaques and induces interferon.28, 145, 371, 423
Individual strains of virus vary in their pathogenicity for various host species. Experimentally this has been best shown by attenuation studies whereby adaptation of a strain to produce disease in one species can often bring about a concomitant reduction in the severity of clinical diseases for some but not all other species.70 This was the basis for the production of many early live vaccines.107, 156, 163, 224, 253, 333, 362
In contrast to the measles virus, the haemagglutinin equivalent of RPV does not readily agglutinate erythrocytes;148 328, 329 some haemagglutinating activity has been demonstrated in special circumstances,187 but the specificity of the reaction was not established. However, since RPV induces the development of antibodies which react with measles virus haemagglutinin,54, 406 it is likely that RPV bears a similar envelope antigen in a more sequestered site.
Epidemiology
Rinderpest virus infects a wide variety of vertebrates. Some of these, including rabbits, hamsters, mice, giant rats (Cricetomys gambianus), ferrets, and susliks (Citellus mongoliscus ramosus) usually are only infected experimentally, and often only by using strains of virus adapted to them.88, 154, 226, 252, 333, 346 In the field, only artiodactyles are naturally infected, although dogs fed infected meat may develop antibodies to the virus, suggesting subclinical infection.267 Amongst domestic stock, cattle and water buffaloes (Bubalus bubalis) are especially susceptible and are more frequently infected than other species. Sheep, goats and pigs are also susceptible and may develop clinical disease.56, 125, 143, 284, 326 European breeds of pigs undergo subclinical infection with African strains of virus.98, 342 Although some early reports indicated that camels are susceptible to clinical disease,101, 129, 193, 232 more recent experimental studies have shown only mild or subclinical disease in this species. 356, 368 Contact transmission from cattle to camels occurs under experimental conditions368 but is probably rare in the field.343
Infection of wild artiodactyls with strains largely maintained in cattle causes a wide spectrum of clinical disease ranging from very severe in African buffalo, giraffe (Giraffa camelopardalis), eland (Taurotragus oryx) and kudu (Tragelaphus strepsiceros and T. imberbis) through increasingly less severe syndromes in other antelopes to mild or atypical in impala341 and subclinical in hippopotami (Hippopotamus amphibius).263 There is also variation in susceptibility to clinical disease between breeds or races of a species, especially cattle.138, 171, 322, 332, 384 Most European cattle breeds (Bos taurus) are more susceptible than Bos indicus breeds. African humpless cattle, such as the Ankole in East Africa, are notoriously susceptible in comparison to East African zebus.62, 71, 83, 135, 216, 332 Japanese black cattle reacted so severely to goat-adapted vaccines, which were sufficiently attenuated for other cattle, that the virus had to be further attenuated in rabbits and embryonated chicken eggs.225, 226
Infected animals excrete virus in their ocular, nasal, oral and vaginal secretions and faeces.87, 130, 139, 333 Excretion begins one or two days before the onset of fever (the first clinical sign) and continues for up to 9 to 10 days after the start of pyrexia.113, 188 Highest titres of virus are excreted during the early stages of clinical disease when epithelial lesions, especially those in the mouth, are developing to their maximum extent. Subsequently, the titres of excreted virus wane as antibody develops.188, 340 Recovered cows may abort an infected foetus some weeks after apparent recovery, with virus excretion in their uterine and vaginal discharges. 87, 157, 390
The fragility of the virus ensures that most infectivity survives for only a few hours outside the host, though some may persist under favourable conditions for up to two to four days.353, 380 Carcass decomposition inactivates the virus within one to three days.87
The spread of RPV is effected almost exclusively by contact between infected and susceptible animals. Airborne transmission by infected aerosols probably only occurs under ideal conditions of close proximity and gentle air currents, for example among housed animals.151 Nevertheless, experimentation has proved that airborne transmission is a theoretical possibility over several hundred metres,150 and circumstantial evidence for such transmission occurred during the 1980s’ epidemics in Nigeria.
The high levels of antibody that are maintained in recovered animals for years after infection, usually for life, suggest persistent infection with RPV. A preliminary study went some way to confirming this by demonstrating viral RNA in the peripheral blood lymphocytes of cattle for up to three months after the cessation of viraemia and recovery from clinical disease.38 As a rule there is no carrier state in rinderpest and recovered animals do not excrete infectious RPV and are not involved in the maintenance and transmission of the disease. The handful of reports of chronic cases of rinderpest87, 92, 99, 124 are exceptions to the rule. The virus is not transmitted by arthropods347 and the potential for transmission through abortion390 is limited.
Consequently, RPV has a short and direct cycle of infection and is spread by close contact. Even under experimental conditions regular contact transmission can be difficult to achieve.82, 151, 373 As originally observed by Lowe,196 the spread of rinderpest is like a bushfire; it continually moves on to new uninfected animals (unburnt bush) leaving behind dead or immune animals (burnt bush). The disease stops when it runs out of susceptibles (firebreaks) and vaccination is a technique of rendering animals non-susceptible (backburning). As a result, rinderpest is maintained by large, heterogeneous populations of animals with a sufficient supply of susceptible animals. In Africa in recent times the endemic areas have been those with large cattle populations belonging to nomadic or semi-nomadic people, which ensures good mixing of the population, especially when restricted by the availability of water during dry seasons. One of the most important epidemiological features of rinderpest was summarized byHuytra et al.:149
Rinderpest causes enormous losses among cattle in regions previously free from infection and may lead to the almost complete extinction of animals in large areas. On the other hand, in permanently infected regions it may assume a milder form and cause only a slight loss.
In populations consisting mainly of susceptible animals rinderpest behaves in epidemic fashion with the virus infecting virtually all the susceptibles and causing severe clinical disease in most age groups. Endemic rinderpest, however, is much milder and is maintained by young animals usually less than two years old that have lost their maternal immunity. Intermediate patterns also exist. Examples in Africa were the enormous losses reported in southern Africa during the first Great Pandemic135, 147 compared with the mild and frequently subclinical infections seen in endemic areas such as Maasailand in the 1950s and early 1960s and the Somali pastoral ecosystem today. 197, 250, 297 Undoubtedly the selection of resistant animals in the wake of epidemics plays a part in this.135, 216, 332 Selection of milder strains of the virus, however, may be an equally or more important mechanism, particularly as the generation time of virus is so much shorter than that of cattle. Computer modelling provided insight as to how this might occur through the selection of strains with differing transmissibility.161, 305 The model showed that during an epidemicRPVis transmitted rapidly to susceptible animals and the most transmissible strains eventually predominate. For a strain to be highly transmissible it must cause the release of large quantities of infectivity from damaged epithelia. Hence transmissibility and virulence are directly related and the selection of increasingly transmissible strains in an epidemic involves an increase in virulence. In endemic areas such highly transmissible strains tend to die out because of the lack of susceptibles. However, milder, less transmissible strains of virus which are continually produced, presumably through mutation, can persist for longer in the limited supply of susceptible individuals, thus being given the opportunity to become dominant strains.305 A limited experimental study supported this hypothesis.52 When the less transmissible strain is reintroduced to an unconfined supply of susceptibles it will again transmit freely and revert to virulence. This ability of the virus to render itself less clinically obvious in endemic areas may explain those outbreaks of disease that have occurred in areas long thought to have been free of the virus.184, 197, 369, 374
Other domestic species such as small ruminants play only an accessory role in the maintenance of the disease by helping it to ‘smoulder’ for a while after epidemics have occurred in the cattle population.426, 427 They are, however, insufficiently susceptible to maintain endemic strains of RPV. In India it was thought that certain strains of RPV have become established in sheep and goats, which could pass the disease back to bovine animals.45, 96, 227, 242, 265 The origin of such strains is unknown, though the use of insufficiently attenuated goat-adapted vaccines has been implied.86 Fortunately, these viruses, if truly rinderpest and not PPR virus, appear not to have been maintained in small ruminants because rinderpest has now been eradicated from India.
Clinical rinderpest in pigs was also common in southern India before the virus was eradicated from cattle.284 European pigs can be infected by diseased cattle and by eating contaminated meat. Although clinical disease has been reported, 125 mild or subclinical infection is more common, especially with African strains of virus, making pigs a potential source of infection for other pigs and cattle.97, 342
In Asia wildlife have been described with clinical disease, and such infected animals can transmit infection to other susceptible species, including domestic stock.27, 92, 193, 245, 357, 385 However, the sizes and densities of these wildlife populations are low and they are not considered to be involved in the maintenance of the virus in Asia.
The role of wildlife in Africa has received much closer attention due to the greater population sizes and densities, the larger number of susceptible species, and the frequency with which the disease has been reported in wildlife.5, 75, 84,410 Until the 1960s a widely held view in East Africa was that wildlife, particularly the vast populations of plains game in Maasailand, could maintain the virus independently of cattle,191, 196, 249, 292 though some authorities considered cattle to be the main reservoir of infection.94, 140 However, when cell-culture attenuated vaccine led to the eradication of the disease from cattle in Maasailand in the early 1960s, clinical disease also disappeared from wildlife.361 The absence of antibodies in wildebeest and other species born after 1963 supported this,256, 264, 374 and considered opinion changed to the view that wildlife could not maintain the virus.254
Nevertheless, the virus may persist for months, perhaps years, during prolonged epidemics in wildlife. In a study of wildlife sera collected in Kenya between 1970 and 1981 significant levels of specific antibody to RPV were detected in 8 per cent of the samples.311 Whilst acknowledging that these infected animals may have been sentinels for RPV in nearby cattle or that other morbilliviruses might be responsible, it was suggested that certain strains of RPV might be maintained independently of cattle for variable periods of time.272, 311 Coincidentally, rinderpest was diagnosed in African buffalo in the Serengeti National Park in 1982, which was the first confirmation of the disease in Tanzania since 1965. A re-evaluation of mortalities in wildlife during the preceding two to three years in Tanzania attributed many of these to rinderpest.234 The 1982 outbreak then expanded to involve wildlife and cattle throughout a large area of Tanzania north of the central railway line before being controlled by the vaccination of cattle in 1983 and subsequent years.135
Subsequent serological surveillance showed that most of the African buffalo population in the area of Kenya immediately north of the Serengeti also had high levels of antibody to RPV, despite having shown no signs of clinical disease. Analysis of the birth dates of sampled animals indicated that new seroconversions had occurred during a two- to threeyear period314 but, unfortunately, no virus was recovered. Since no rinderpest was reported in cattle in this area, it appeared that the wildlife may have assisted in maintaining RPV for a period of years and that the infections were inapparent or mild enough to escape notice by wildlife authorities. Between 1994 and 1997, and again in 2001, similar outbreaks occurred in wildlife in Kenya’s national parks with only limited evidence of infection in nearby cattle.179 Fortunately, virus was isolated from these outbreaks and shown in experimental infections to cause only very mild or subclinical disease in cattle. Molecular typing revealed that the virus involved was the Africa lineage-2 virus, which had not been isolated in Africa for over 30 years.41 Since this previous isolation was from a giraffe,230 this encouraged renewed debate on the role of wildlife. At present all evidence indicates that wildlife do not maintain RPV indefinitely, though it is possible that sufficiently large populations may occasionally maintain mild or subclinical RPV for periods of a few months to perhaps one or two years. Nevertheless, wildlife do play a very important role in rinderpest. Rather than being permanent reservoirs of infection they act as sensitive sentinels for mild rinderpest in nearby cattle populations. All recent confirmed outbreaks of Africa lineage-2 virus have been detected first in wildlife. Even when rinderpest is proved to have been eradicated from cattle it is likely that final acceptance of the eradication of rinderpest from Africa will come from clinical and serological surveillance of wildlife.
Pathogenesis
Experimental infections can be established by all routes of parenteral inoculation and, more variably, by intranasal or conjunctival installation. Natural infection usually occurs via the upper respiratory tract following inhalation of virus containing aerosols, or the oropharynx after ingestion of infected material.87, 130, 139, 251, 252, 333, 380 Primary multiplication of RPV has not been demonstrated in the invaded epithelium,373 but following intranasal and contact challenge, the virus can be recovered within 24 hours from the pharyngeal lymph nodes and tonsils and, to a lesser degree, from other lymph nodes draining the head and upper respiratory tract.251, 373 In vivo infectivity is closely associated with mononuclear leukocytes and is not readily detected in plasma and other body fluids.93, 252, 325, 380
Primary multiplication in lymph nodes draining the site of entry of the virus leads to viraemia, which enables the virus to infect and replicate in lymphoid tissues throughout the body, and eventually in epithelial tissues, especially those of the alimentary tract. There, virus-induced cytopathic effects produce the typical lesions of the disease, some of which may also be due to infection of small lymphoid aggregates in the lamina propria.252
There is a direct relationship between attenuation and the degree of viral multiplication in lymph nodes of cattle.333, 418 Virulent strains of RPV have a greater ability to infect lymphoid cells and mononuclear phagocytes and may grow to higher titres in these cells than do strains which induce mild disease.317 The cell-culture attenuated variant of the Kabete ‘O’ strain of RPV, which is the most commonly used vaccine, only produces low levels of infectivity in lymphoid tissues and is barely detectable in the blood.372 These low levels of viraemia are probably one reason why attenuated and mild strains cause so little epithelial damage. The virus has a predilection for T lymphocytes and attains higher titres in the T4 and T8 subsets of T cells than in lymphoblasts of B or null cell origin.301, 312, 315, 317 Rinderpest virus grows more readily than PPR virus in bovine lymphoblasts, whereas the reverse is true in sheep and goat lymphoblasts. 347
During disease the virus is also found in non-lymphoid organs, such as the lungs, liver and kidneys.54, 57, 93, 188, 372, 373 Immunocytochemistry has shown that antigen-bearing cells in these organs are usually associated with reticuloendothelial and perivascular connective tissue.38, 73, 412, 413
Virulent strains of RPV are excreted from epithelial tissues one or two days before the appearance of fever or lesions, 188 but the amount of excreted virus increases considerably as the lesions develop and only starts to decline when the immune response becomes detectable some four to six days after the start of fever. The virus is usually undetectable by 12 to 14 days after the start of fever. At the height of virus excretion, three to six days after the start of pyrexia, virus titres of up to 105 tissue culture infectious doses (TCID50)/nasal swab and up to 106 TCID50/g of faeces can be recovered from cattle infected with virulent strains.188 This copious output explains why the disease can be so contagious despite the fragility of the virus.
The diarrhoea and oculonasal discharge probably help to increase the transmissibility of the virus by forming infectious aerosols, and by contaminating the environment.
Infected animals mount a vigorous response against the virus. Interferon is produced within two days of infection, enabling attenuated vaccines to protect cattle very rapidly against challenge by virulent virus.146, 405, 411 Viral antigens are produced in large amounts throughout the lymphoid tissues and affected epithelia224, 306, 335, 340, 418 and stimulate an effective antibody response which begins two to five days after the onset of clinical disease in virulent infections, and some 6 to 10 days after infection with mild or avirulent strains.164, 223, 260, 339, 394 The early response consists predominantly of IgM antibodies14, 241 which can be detected by virus neutralization (VN), enzyme-linked immunosorbent assay (ELISA) and also, for a period of a few months, by immunoprecipitation, complement fixation and measles virus haemagglutination inhibition.316, 345 At the same time IgG antibodies are produced, but these persist for much longer, usually for life,255, 261, 320 and are usually measured by VN or ELISA tests.13, 293, 307, 308, 339, 351 Generally, high titres (102 to 103 log10 VN50) of neutralizing antibodies are produced within two to three weeks of infection and remain high for several months, after which they may decline slowly, but usually remain at easily detectable levels (in excess of 101log10 VN50) for the rest of the animal’s life.255, 261, 266 Rarely, neutralizing antibodies may decline to very low or undetectable levels but such animals are clinically immune, although limited replication of the virus may occur in tissues such as the tonsils before the stimulation of an anamnestic response.255, 266, 281, 320, 425 The antibody responses of naturally infected cattle and those vaccinated with live tissue culture virus vaccine are indistinguishable.
A study using ELISA showed the development of serum IgA antibodies in cattle after vaccination.14 However, one other study using an indirect immunoperoxidase technique failed to detect IgA antibodies and, in contrast to most other studies in cattle, ascribed most of the IgG antibodies to the IgG2 subclass.350 Secretory antibody is found in nasal secretions of convalescent cattle269 but its persistence is presumably limited to only a few months after recovery, and the role it plays in preventing reinfection is unknown. Cell-mediated immunity to vaccine and infectious virus has received very limited study and its role is still uncertain though undoubtedly important.198, 199, 237, 238
The severity of the cytopathology caused by the virus before the onset of antibody development influences the course of the disease. Virulent strains cause severe lesions before being constrained by the immune response, and such animals, if sufficiently damaged, will still die despite high titres of antibody and low or undetectable amounts of virus.328, 340 The persistence of immunity in recovered animals and those given live virus vaccines contrasts with the short-lived immunity induced by inactivated vaccines,158, 339 thus implying that recovered animals may be immunologically re-stimulated by RPV antigen throughout their lives.
Immunohistological techniques have shown that the necrotic changes in the lymphoid tissues, epithelia and other organs are associated with large quantities of RPV antigen. 73, 85, 239, 365, 412, 418 Cells containing RPV antigen are widespread in lymphoid tissues and are also prevalent in cells in the loose connective tissue surrounding blood vessels in most tissues.85, 307, 412
Circulating immune complexes have been demonstrated in rabbits recovering from rinderpest,288 associated with the rapid elimination of the mass of viral antigen by the specific humoral response. There is a transient autoimmune response in infected rabbits which produce an IgM (19S) haemagglutinin to homologous erythrocytes and an IgG (7S) anti-nuclear antibody.116, 287
The massive destruction of lymphocytes causes immunosuppression. In rabbits, cell-mediated and humoral immunity is suppressed through the loss of both T and B lymphocytes.176, 239, 240, 416 In cattle, lymphoid necrosis is first seen in the germinal centres which are populated with B cells.378 However, in vitro, RPV grows readily in bovine T and B lymphocytes.303 Immunosuppression in cattle therefore probably involves both cell-mediated and humoral immune responses.
Possible immunosuppression by the cell-culture attenuated vaccine has been investigated experimentally by simultaneous inoculation of RPV with other immunogens such as vaccines against foot-and-mouth disease and contagious bovine pleuropneumonia. Most experimental studies have shown no diminished responses to RPV or the other immunogens, 72, 105, 134, 169, 201, 277, 278 although there is still some doubt, especially with regard to bivalent vaccines incorporating live Mycoplasma mycoides162 or inactivated foot-andmouth disease virus.170 One report also describes the development of severe bovine virus diarrhoea virus (BVDV) infection in cattle given live attenuated RPV in conjunction with live attenuated BVDV,276 another lymphotropic and known immunosuppressive agent.
There are some reports that the vaccine may exacerbate trypanosomosis or that trypanosomosis reduces the antibody response to the vaccine.356 The overwhelming evidence from the field, where several hundred million cattle have been vaccinated in the past 40 years, many of which would have had intercurrent infections, is that rinderpest vaccine does not reactivate or exacerbate trypanosomosis or other diseases as experimental studies show.360 Even when suffering from severe but non-fatal rinderpest, cattle develop a rapid and full humoral response to the virus despite the changes in their lymphoid tissues. Admittedly, the early vaccines prepared from caprinized, lapinized and avianized virus often did produce mild clinical signs and occasional flare-ups of latent or subclinical infection,65, 71, 332, 362 but they produced no lasting check on the growth rate of healthy cattle67 and did not increase the severity of a concurrent outbreak of East Coast fever.16
Rabbits inoculated with lapinized virus show depressed and delayed hypersensitivity skin responses and impaired lymphocyte responses to mitogenic stimulation.419
Clinical signs
The clinical signs of rinderpest have been extensively described. 25, 87, 88, 119, 135, 252, 274, 322, 335, 336 The following description gives the basic signs in the sequence in which they usually occur in severely affected cattle.
Acute natural infections and those induced by parenteral inoculation of RPV have an incubation period of three to five days. Incubation periods following experimental contact infection are longer, from 8 to 15 days,188, 250, 297, 373 and there are reports of extended incubation periods in natural outbreaks. 87
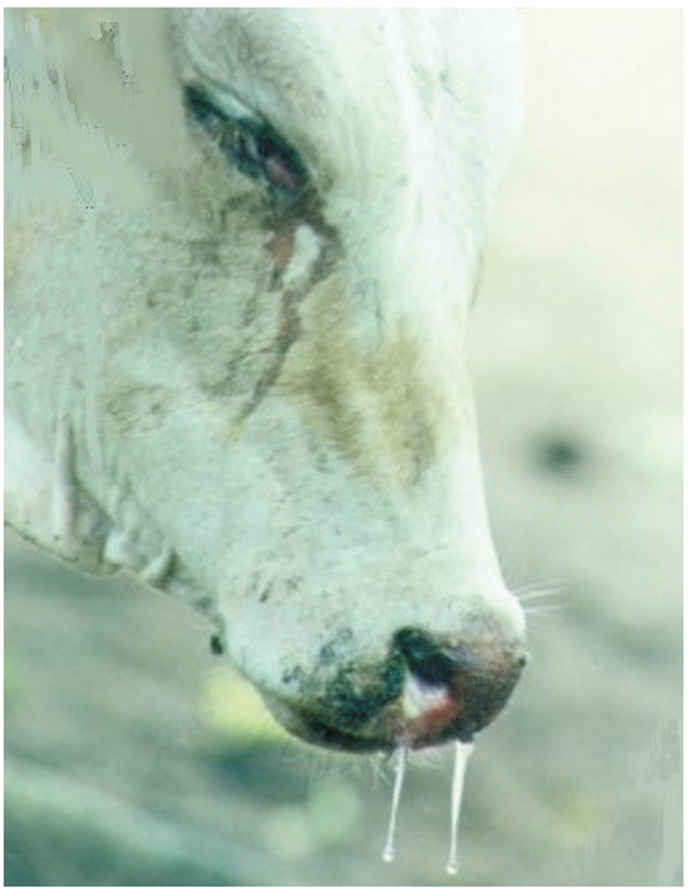
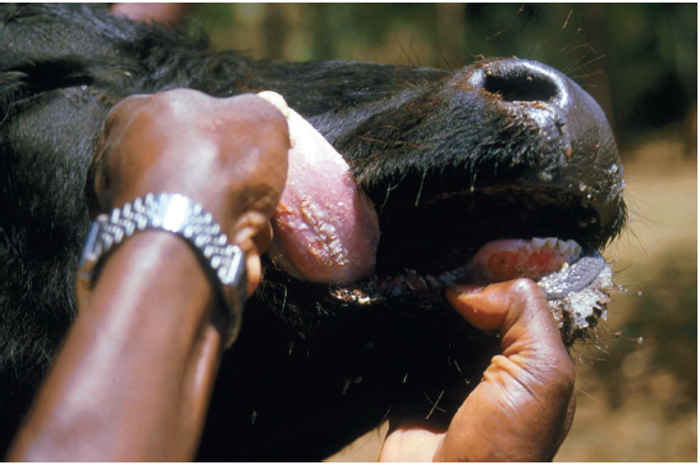
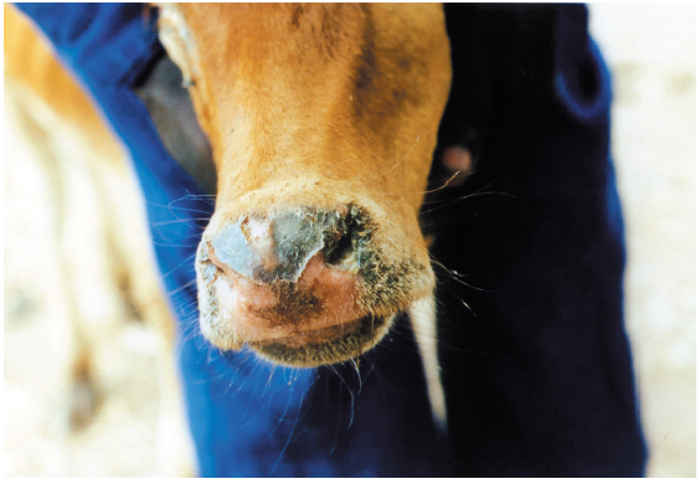
Pyrexia heralds the onset of clinical disease. The rectal temperature rises rapidly to 40 to 41,5 °C and this can be maintained for up to one week before gradually returning to normal. During the first one or two days of fever there may be depression or restlessness, inappetence and reduced milk yield. The visible mucosae are congested and a serous oculonasal discharge (Figure 49.5), one of the characteristic signs of rinderpest, becomes apparent. Between the second and fifth days of fever the mucosal lesions become obvious,initially as pin-point white foci on the gums (Figure 49.6) and lips, which spread rapidly to involve the buccal mucosa and ventral and lateral aspects of the tongue (Figure 49.7) and muzzle (Figure 49.8 and 49.9).
The foci enlarge and coalesce into plaques of caseous necrotic debris, which desquamate easily, leaving circumscribed erosions. The halitosis is memorable. Similar lesions may be seen on the palate, posterior dorsum of the tongue, pharynx and in the nares and vagina. Severe cases often drool foetid saliva, presumably because of the discomfort in their mouths and pharynges when swallowing.
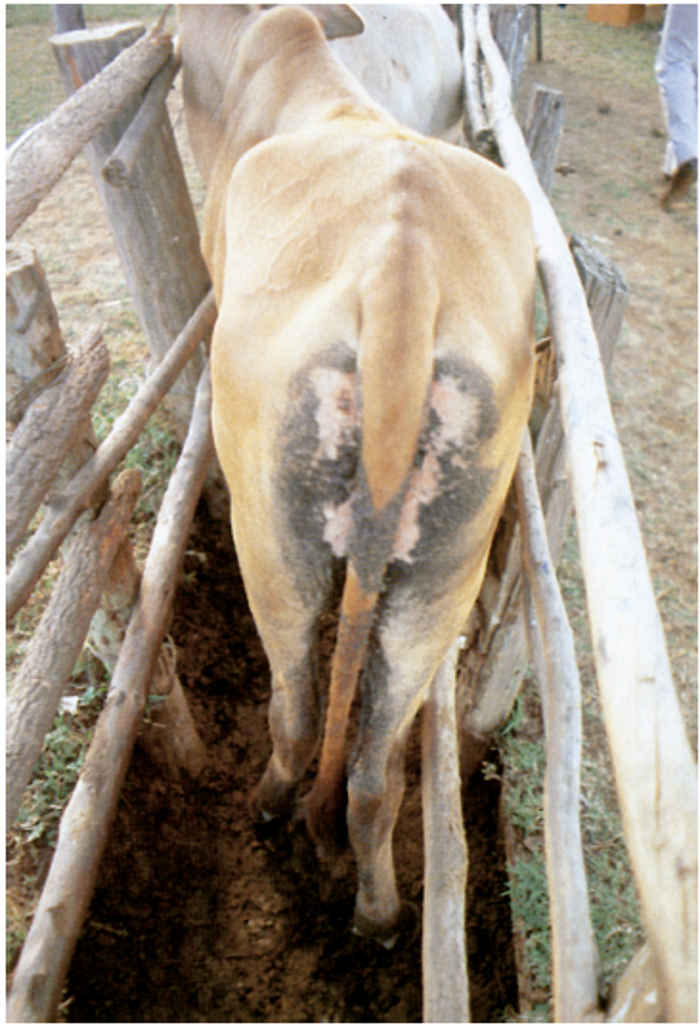
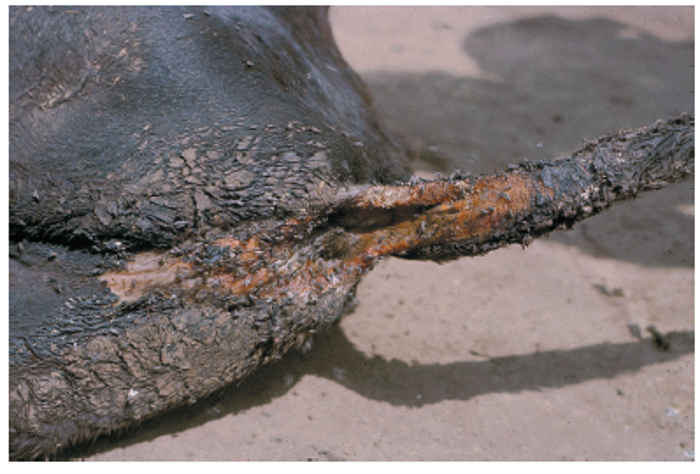
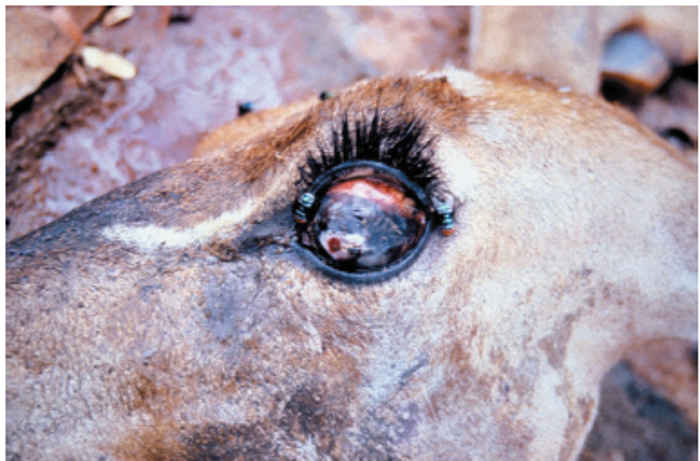
In cattle infected with strains of high virulence, diarrhoea, sometimes preceded by an absence of defaecation (there are usually no signs of constipation), starts four to five days after the onset of pyrexia, usually one to three days after mouth lesions become visible. The faeces are initially thin and dark, but may progress to contain blood, mucus and shreds of necrotic epithelium. The hindquarters are fouled (Figure 49.10 and 49.11) and tenesmus with eversion of the rectal mucosa is frequent. The diarrhoea causes dehydration (Figure 49.12), weakness and prostration, and in severe cases abdominal pain is evident. Recumbent animals face their flanks in typical ‘milk fever’ posture. The oculonasal discharge becomes increasingly purulent during the course of the disease and conjunctivitis causes photophobia. Corneal opacity is rare in cattle, but has been recorded in some species of wildlife such as giraffe, kudu (Figure 49.13 and Figure 49.14) and buffalo.179, 349 Epiphora may be evident below the medial canthus.135
Early descriptions of rinderpest frequently made mention of skin lesions.232, 322 Most recent accounts88, 252, 333 regard these as rare, though they are sometimes present in water buffaloes, sheep and goats. The lesions appear as a maculo-papular rash on areas of soft skin such as the axillae and groin, where the hair may become matted when the rash eventually becomes pustular and breaks open.25, 87, 165, 215, 218
Although respiration may be laboured during acute clinical disease, pulmonary lesions are uncommon and are mainly due to secondary bacterial infection. Emphysema of the lungs may occur terminally. Lymphadenopathy is not usually evident even on palpation of the superficial lymph nodes. Milk may become watery or dry up. Pregnant animals frequently abort, occasionally weeks or months after clinical disease.87, 157, 232, 390 Most animals that succumb to rinderpest die 5 to 14 days after the onset of pyrexia.
Convalescence is marked by the rapid healing, within two to six days of oral mucosal lesions, starting some three to six days after their appearance. In cases of mild disease the mouth lesions may only be visible for one or two days. Diarrhoea diminishes more slowly and may persist, in a less severe form, for up to two weeks. Complete recovery from severe rinderpest takes one to two months or more, depending on how much condition the animal has lost and the quality of nursing.
Since RPV destroys lymphoid tissues, infected animals suffer from a variable degree of immunosuppression. In Africa this frequently leads to the reactivation of a variety of chronic or latent diseases, especially those caused by haemoparasites such as anaplasmosis, which, if not treated therapeutically, increase the mortality caused by RPV.87, 120, 138, 141, 275, 291, 333, 335
In endemic areas the disease is milder: most of the signs are much reduced in severity and some may be absent.87, 197, 250, 274, 297 Experimental infections with virus strains recovered from these areas may cause no deaths, no diarrhoea, transient mouth lesions in only a proportion of infected Bos taurus cattle, and only transient pyrexia in Bos indicus cattle.250, 297, 369
Clinical signs vary in sheep and goats. Disease was seen in sheep during the British epidemics of 1865 to 1866322 and in the first Great African Pandemic.147 Clinical signs in naturally infected small ruminants have occasionally been reported in Africa47, 191, 200, 326, 340, 388 but experimental infections with African strains of RPV have produced only transient fever, sometimes accompanied by slight oculonasal discharges.4, 110, 248, 331, 388, 393 Sero-conversion is usually the only evidence that sheep and goats have been infected.71, 72, 309, 428
In contrast, clinical rinderpest occurred frequently in sheep and goats in India before its eradication.8, 108, 285, 286, 348, 358 The duration of the incubation period is similar to that in cattle, with fever being the first clinical sign although its onset may be more gradual than in cattle. Diarrhoea starts two to three days later and, if severe enough, causes dehydration and death. The tail and hindquarters are soiled by faeces. Sero-mucoid, oculonasal discharges that rapidly become purulent are seen within 24 hours of fever. Mouth lesions are less obvious than in cattle. Inappetence is usual. Pneumonia is commoner in small ruminants than in cattle, but is also probably due to secondary bacterial infections.95
In Asia, both local and European pig breeds develop clinical disease.153 The signs are typical: pyrexia, inappetence and depression, and prostration.56, 81, 143 Typical mouth lesions develop in one to two days, and diarrhoea within two to three days after the onset of fever. The diarrhoea lasts 5 to 10 days and in severe cases leads to dehydration and death. Pregnant sows may abort. Erythematous skin lesions may be seen. Transient low fever is all that may develop in European pigs inoculated with African strains of virus, most infections being totally inapparent.342
Mouth lesions, diarrhoea129, 19 and deaths101 have been reported in camels, but generally, in areas where rinderpest is prevalent in other species, camels do not show clinical signs194, 246 or develop antibodies which would indicate inapparent infection.343 Most of the reports of clinical disease were based only on clinical evidence without other diagnostic confirmation. Experimental infection of dromedaries (Camelus dromedarius) with cattle strains of RPV resulted in subclinical infection.280, 368
Disease in water buffalo is frequently severe, with clinical signs being the same as those in cattle, although skin lesions are reputedly more common.25, 87
Wild ungulates exhibit a wide range of clinical signs, from typical erosive stomatitis, gastroenteritis and death in African buffalo (Figure 49.15), eland (Taurotragus oryx), giraffe and warthog (Phacocoerus aethiopicus)247, 379 to mild or nonspecific signs in impala.341 As a generalization, the more bovine- like species amongst the Bovidae suffer more severe clinical disease than do the caprine-like species. Corneal opacity and skin lesions have often been described for rinderpest in wildlife such as giraffe, Grant’s gazelle (Gazella granti) and African buffalo, though not all of these outbreaks wereconfirmed in a laboratory.254, 349, 354, 379 During the 1994 to 1997 and 2001 wildlife epidemics in Kenya these same signs were seen in animals in which the disease was confirmed by laboratory tests. Corneal opacity and blindness were such regular features in these outbreaks that reports of blindness in some species such as kudu (Figure 49.13 and Figure 49.14) should always be treated as possible rinderpest.220 Affected wildlife often exhibit behavioural changes. Dehydration forces thirsty animals to migrate long distances for water, while discomfort makes them more aggressive, especially African buffalo.58, 75 In the Kenyan outbreaks, sick animals such as eland fell behind their herds and were closely approached by inquisitive members of other species, thereby providing a greater chance for interspecies transmission.
Pathology
A proportion of infected cattle shows slight lymphocytosis before the onset of pyrexia. This is followed by marked lymphopenia, caused by lymphoid necrosis, which in most cases lasts throughout the acute clinical stage of the disease. 24, 136, 208, 290, 296, 378 During convalescence, lymphocyte levels slowly return to normal over a period of days to weeks. The number of neutrophils remains relatively unaltered, though juvenile forms are not infrequent during the terminal stages of fatal infection. However, a degree of neutropenia that parallels the decline in lymphocyte levels has been reported.378
Eosinophils may also disappear from the blood during the early stages of clinical disease, returning to normal levels some two to three weeks later. In severe cases the excessive loss of water causes haemoconcentration.24
Serum aspartate transaminase and blood urea nitrogen levels increase during severe cases of disease.51, 137 Serum chloride levels fall markedly in terminal illness, and other electrolytes may decrease in absolute terms although this can be masked by haemoconcentration. Blood clotting may be impaired in severely affected animals. Serum protein levels may be lowered, especially in fatally infected animals.87, 102, 115 In cattle recovering from experimental infections a rise in serum globulins was attributed to the specific humoral response to the virus,115 but since the challenge material was citrated blood, this may need reinterpretation in the light of known responses to heterologous tissue antigens.
The lesions of rinderpest are a direct result of virus-induced cytopathology. There are many descriptions of the gross pathology,25, 61, 87, 119, 159, 333 but those of Maurer et al.207, 208 are especially valuable. Generally, the severity of the lesions is directly related to the virulence of the strain of virus involved.377, 418 Complications may arise during convalescence through reactivation of latent pathogens, especially protozoa.87, 138, 335
The overall appearance at necropsy is similar for most species that die of typical severe rinderpest. The carcass is dehydrated, sometimes emaciated, and usually soiled with fluid faeces. The eyes are sunken and often encrusted with mucopurulent discharge and the cheeks may show signs of epiphora.
Erosions with or without necrotic material may be found throughout the mouth but predilection sites are the gums, lips, buccal papillae, dorsal and ventral aspects of the tongue and the soft palate. The erosions often extend into the pharynx, cranial part of the oesophagus, rumen (especially the pillars), reticulum and omasum. Necrotic areas, some of which may penetrate the leaves of the omasum, are sometimes present.
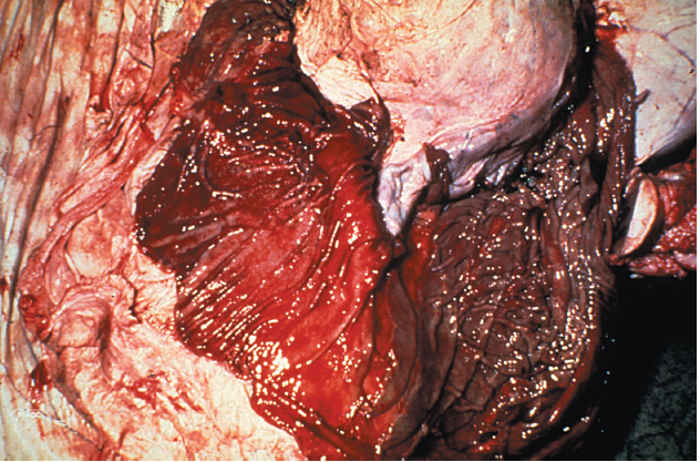
The folds of the abomasum are congested and oedematous and often show necrosis, erosions and haemorrhage along the edges (Figure 49.16). The fundus of the abomasum may have small discrete erosions that increase in size towards the pylorus where whole areas of mucosa may become desquamated. The early necrotic lesions are palegreyish, whereas the erosions are often red as a result of congestion of the underlying lamina propria. Haemorrhage may occur from the raw surfaces. The abomasum is almost invariably severely affected, whereas the small intestine frequently shows less involvement. Congestion, oedema and erosions may occur on the margins of mucosal folds of the cranial part of the duodenum and terminal ileum. The Peyer’s patches, being lymphoid tissue, are often severely affected and may be swollen, dark red to almost black as a result of haemorrhage, and may slough completely leaving deep ulcer-like areas. Large erosions are commonly found on the ileocaecal valve. In the large intestine, marked oedema and congestion accompanied by petechiae or larger haemorrhages occur, particularly along the crests of longitudinal folds of the mucosa. This can be very striking in the colon and rectum, meriting the description ‘zebra striping’. In acute cases, the gut contains little other than desquamated necrotic epithelium, blood, and fibrin exuding from exposed lamina propria.
The urinary and gall bladders are sometimes congested and haemorrhagic with occasional erosions. The vaginal mucosa may be congested and have small erosions.
The mucosa of the nasal passages, sinuses and larynx may be congested and is usually covered with mucopurulent exudate. Petechiae are frequent and necrotic; erosive lesions may extend from the nares to the larynx. The tracheal mucosa is frequently congested. Congestion and emphysema may be seen in the lungs while secondary bronchopneumonia may complicate chronic cases.
Although regularly described in early reports, skin lesions are now rarely seen, although they are reputedly still common in water buffalo. The exudative dermatitis would seem to develop from macular to pustular lesions, but the role of secondary bacterial infections such as Dermatophilus congolensis needs clarification.141, 196, 218
Although RPV has a predilection for lymphoid tissues, there are usually few visible changes in the superficial and visceral lymph nodes. These may show congestion, oedema, and a few petechiae. The nodes of animals that die after a prolonged clinical course may be shrunken and show greyish radial streaks in the cortex, presumably due to haemorrhage.25 The spleen and haemolymph nodes appear normal or slightly enlarged.
The necropsy findings in goats inoculated with caprinized virus are similar to those in cattle, although they may be less marked.
The lymphotropism of the virus is perhaps best shown in rabbits inoculated with lapinized virus. Although there are no observable epithelial lesions, white, pin-head sized foci of necrosis are easily seen in the gut-associated lymphoid tissues.117, 159
Histopathological lesions become more easily detectable with increasing severity of clinical disease, implying that the pathology is directly related to the ability of a strain to multiply rapidly in the tissues.378, 418
The essential histopathology of rinderpest was first described in infected rabbits.62, 117 Widespread necrosis of lymphocytes is apparent throughout the lymphoid tissues, together with syncytia and intracytoplasmic and, less frequently, intranuclear inclusion bodies. The histology in cattle is similar172, 208, 377 with lytic destruction of lymphoid tissues that is most evident in the germinal centres, sometimes accompanied by an increase in the numbers of macrophages. In acute cases lymph nodes are virtually devoid of cells, with just a reticular stroma containing eosinophilic material remaining.
The early epithelial lesions in the squamous epithelium of the digestive tract are associated with the formation of syncytia and eosinophilic intracytoplasmic inclusions in the stratum spinosum.173 Infected epithelial cells become necrotic and slough off, leaving clearly demarcated erosions. The erosions heal rapidly unless complicated by secondary infections which may rarely cause them to ulcerate.
Changes in other tissues are not remarkable, although small foci of necrosis in the liver have been described.174
Ultrastructurally, infected cells contain large parallel arrays of tubular nucleocapsid material which are probably the intranuclear and intracytoplasmic inclusion bodies seen by light microscopy.60, 282, 366 Virus is released from cells by some of the tubular strands budding through the cell membrane. Similar changes were described in lymphoid cells from two Japanese black cattle infected with the virulent Fusan strain of RPV,177 and in persistently infected Vero cells.177
Diagnosis
The clinical and laboratory diagnosis of rinderpest is described in detail in many handbooks and reports.6, 25, 29, 270, 279, 335, 345 A presumptive diagnosis can be made on the basis of the clinical signs and gross pathology. However, in countries where rinderpest is not prevalent it is essential to obtain laboratory confirmation of the diagnosis as soon as possible. Countries where rinderpest is either endemic or a high risk should treat any syndrome resembling rinderpest as such until proven otherwise. This will allow immediate steps to be taken to control the disease and restrict losses.
The collection of adequate quantities of appropriate specimens greatly increases the chances of an accurate laboratory diagnosis. A thorough clinical examination should be made of animals in suspected herds and six or seven animals in the early acute stage of the disease with fever, mouth lesions and lachrymation should be selected for sampling. Animals that are dead, moribund or have had diarrhoea and mucopurulent discharges for more than three days are less reliable sources of virus or antigen as the levels of these decline with the onset of antibody development. 335
From each selected animal the following specimens should be collected: whole blood for serum antibody assay, and in anticoagulant for virus isolation from leukocytes, a biopsy from a superficial lymph node, debris from oral lesions, and ocular and nasal swabs for virus isolation and antigen or nucleic acid detection. If possible, two or more animals should be killed for necropsy examination and collection of up to three universal bottles of splenic tissue and mesenteric lymph nodes. All specimens should be collected and bottled aseptically, kept cool on ice (but not frozen) and transported as rapidly as possible to a diagnostic laboratory. 335, 345, 389
Glycerol should not be used as a preservative because it inactivates RPV. The use of anti-proteases increases the survival of RPV antigens in tissue suspensions301 and reduces the degradation of RNA.
At the laboratory, suspensions of solid tissues are prepared in physiological saline or cell-culture medium, the buffy coat is removed from the whole blood and the serum separated from the clotted blood. Thirty per cent tissue suspensions (w/v) for antigen detection can be prepared by most techniques, including grinding with sand in a mortar, but 10 per cent suspensions for attempted virus isolation can best be prepared in Ten Broeck or similar grinders.
The first procedure usually carried out is to detect viral antigen using specific rabbit hyperimmune serum against RPV. The most commonly used assay is the agar-gel immunodiffusion test (AGID)340, 408 which is simple, easy to read, and highly specific. Moreover, it can be used in the field with swabs and gum debris and can give a result within two hours, especially if micro-versions are used.113 Counterimmunoelectrophoresis is quicker and more sensitive than AGID but requires more sophisticated equipment.3, 313 Immunofluorescence and immunoperoxidase staining methods are very sensitive but also need more equipment than AGID.22, 118, 269, 306, 352 Although once widely used, complement fixation and conglutinating complement absorption tests are too complicated in comparison with more recently developed tests. Various haemagglutination assays are sensitive but not widely applied,27, 355 though latex bead agglutination tests have given encouraging preliminary results213 and, if combined with monoclonal antibodies, could prove very sensitive. Recently, a monoclonal antibody-based rapid chromatographic strip test has proved useful under penside conditions.74, 144, 398 If classically prepared rabbit hyperimmune serum335 is unavailable, serum can be prepared using other immunizing techniques in rabbits214, 220, 301 or in goats or cattle.330, 344 A positive test result in any of these antigen detection tests confirms rinderpest.
Where cell-culture facilities are available, attempts should be made to isolate the virus. Suspensions prepared from swabs, gum debris, buffy coats or lymphoid tissues are inoculated onto growing monolayers of primary or secondary bovine kidney cells in tubes.258, 262 Vero cells and lymphoblastoid cell lines160, 212 are also suitable, while culture systems such as microplates can also be used403 but may be less sensitive. After 12 to 24 hours adsorption the tubes are washed, refed with maintenance medium and rolled at 37 °C. Typical cytopathic effects develop within 3 to 14 days, occasionally longer, and consist initially of foci of round and refractile cells with cytoplasmic processes and small syncytia, followed by generalization throughout the monolayer with distinct syncytium formation. Negative test cultures should be passaged at least once. The virus can be identified by inoculating sample materials into tubes containing antiserum to RPV or by examining fixed monolayers using immunofluorescent or immunoperoxidase techniques.181, 186, 268 Where cell cultures are not unavailable, the specimens can be inoculated into known immune and susceptible cattle, as long as these are isolated from other susceptible animals.
If antigen detection and virus isolation are negative, then convalescent animals should be bled again two to four weeks later. Assays for serum antibodies should demonstrate a four-fold or greater increase in antibody titre in recovered cases. Virus neutralization in microplates was most commonly used for this,12, 293, 307 although several other techniques such as measles virus haemagglutination inhibition, indirect immunofluorescence, ELISA and counterimmuno- electrophoresis are alternatives.166 A number of ELISA tests have been developed. Original indirect tests based upon whole virus antigens12, 13, 14, 308 have largely been replaced by competition ELISAs that use monoclonal antibodies to different viral antigens such as the H or N protein, and may also use purified or recombinant antigens.11, 190, 321 The ELISA has the advantage that laboratories without cell-culture can test thousands of sera, which is often required in current eradication programmes, and the sensitivity and specificity of these new tests is under validation at present. During the early antibody response, serum contains significant levels of IgM to RPV,14, 241 the detection of which confirms the diagnosis, though this approach is rarely used.
Histopathology is not sufficiently specific to confirm a diagnosis of rinderpest, but demonstration of syncytia and viral inclusions is supportive.
Nucleic acid techniques including hybridization with probes and polymerase chain reactions (PCR) are capable of detecting minute quantities of RPV RNA in tissues and secretions,38, 244 and are now often a routine choice for confirmation in reference laboratories. The PCR offers the advantage of providing amplified viral RNA for nucleotide sequencing in order to establish the virus subtype or lineage for epidemiological purposes.114
Differential diagnosis
All conditions that cause stomatitis and/or enteritis in domestic stock can be clinically confused with rinderpest. In cattle, difficulties may occasionally arise in distinguishing rinderpest from mucosal disease (MD), malignant catarrhal fever,376 infectious bovine rhinotracheitis (particularly when caused by strains that induce diarrhoea),131, 132 papular stomatitis,222 Jembrana disease375 and foot-and-mouth disease. In small ruminants, PPR and Nairobi sheep disease can resemble rinderpest. Infection with Campylobacter spp.,Brachyspira hyodysenteriae and Salmonella serovars needs to be considered when investigating possible rinderpest in pigs.
In practice, onlyMDin cattle and PPR in small ruminants present a problem.69, 334 The clinical signs and gross pathology in cattle with MD can be indistinguishable from rinderpest and diagnosis requires laboratory confirmation. However, MD usually affects very few animals in a herd, whereas morbidity rates in rinderpest are much higher. Agar-gel immunodiffusion applied to tissue suspensions can rapidly differentiate the two diseases.90 Immunohistochemical techniques can be used on frozen sections of mesenteric lymph node or on formalin-fixed tissues to distinguish between rinderpest and MD.118 Failing this, virus isolation with subsequent virus identification must be attempted, with follow-up studies to detect rising antibody titres. Nucleic acid-based techniques may also be used.
The differentiation of PPR from rinderpest is more difficult. Useful epidemiological evidence is provided by the absence of disease in cattle. The virus cross-reacts serologically with RPV and is difficult to differentiate with hyperimmune polyclonal sera. Fortunately, contemporary studies have produced monoclonal antibodies and nucleic acid techniques that clearly distinguish between PPR virus and RPV, at least for the limited number of strains tested to date.10, 103, 209, 210 In African countries that have previously been free of PPR it is unwise to assume that a rinderpest-like syndrome in small ruminants is not PPR.
Control
In countries where rinderpest is exotic, confirmed outbreaks are controlled by the slaughter and disposal of all affected and in-contact animals, as well as by appropriate quarantine and animal-movement controls. Such measures eradicated the only outbreak of rinderpest in Australia within two months407 and halted the 1866 outbreak of rinderpest in Britain within six months.383
Virtually all outbreaks of rinderpest in virgin areas have been due to the importation of live infected animals.327 Prevention in such areas is therefore largely dependent upon vigilant control of the introduction of live animals from potentially infected areas. Importation of fresh carcasses and meat products constitutes a minimal threat, although at least one epidemic has been attributed to this source2, 80, 88 and outbreaks in endemic areas have been traced to fresh infected meat. During the first Great Pandemic, the virus is reputed to have crossed the Orange River in South Africa in this way. In frozen meat the virus persists for much longer than in fresh meat and is therefore a risk to swill-fed pigs.289, 327, 329 Infectivity disappears rapidly from adequately dried infected hides48 and from decomposing carcasses held at ambient temperatures in tropical regions for more than two or three days.87, 106 A limited number of outbreaks have also been associated with the spread of the virus from laboratories. 422
Contaminated areas should be physically cleaned of all animal waste and soiled bedding and treated with disinfectant solutions of high (>10) or low (<3) pH containing solvents to destroy the virus envelope. Most disinfectants have some activity against RPV, but it has been shown that solutions of caustic soda and lysol have the highest virucidal activity against virus contaminated with organic matter.399 Such premises could be restocked after a week but the customary caution of veterinary authorities usually results in the period being longer.
Control is more difficult in endemic areas. Clinical diagnosis should be sufficient to instigate control procedures and all outbreaks of rinderpest-like disease should be treated as rinderpest until proven otherwise. Slaughter of affected and in-contact animals removes the source of virus, but may be counter-productive where owners have either a close cultural bond with their animals or are unlikely to receive compensation. In such areas, quarantine and strict movement control must be enforced immediately and the support of local cattle owners (which is usually forthcoming) is essential in this. Countries bordering high-risk zones must channel the import of domestic animals through border quarantine stations for vaccination and three weeks of observation.191
All susceptible animals in affected herds should be vaccinated immediately with subsequent ‘ring’ vaccination of herds in the surrounding districts.
The importance of vaccination in the control of rinderpest in endemic areas cannot be overemphasized, and most early research concentrated on the development of effective vaccines.87, 217, 333
Initial ‘vaccines’ in South Africa included the inoculation of infected bile, developed by Koch,178 and the simultaneous administration of convalescent serum and virulent virus developed by Danysz and Bordet, Theiler and Watkins Pitchford, and Kolle and Turner.147 Neither of these was entirely satisfactory, and Kakizaki167 subsequently developed a glycerol inactivated tissue vaccine. The immunity it induced was solid but short-lived. This encouraged the development of many other inactivated vaccines, but these required frequent administration. It was known that, where it could be produced, immunity following techniques that used live virus persisted much longer, perhaps for life. Continuing research led to the development of a series of increasingly attenuated vaccines that were produced by passage in hosts other than cattle (caprinized, lapinized, and avianized vaccines).107, 163, 226, 362
Finally, repeated passage of the virulent Kabete ‘O’ strain of RPV in bovine kidney cells produced a totally safe and effective attenuated virus,164, 261 which has been the principal vaccine used to combat rinderpest throughout the world during the past 40 years. It is inexpensive and simple to produce and test, stable in freeze-dried form,253 and highly effective even in animals with severe infections such as East Coast fever and trypanosomosis.318, 392
It induces life-long immunity. The only drawback of the vaccine is that after reconstitution in normal saline it has a working life of only a few hours in the hot climates where rinderpest still occurs, 253, 338 and there is some evidence that it should not be used more than one hour after reconstitution at ambient temperatures around or above 37 °C.283 Nevertheless, this vaccine has produced strikingly successful results such as those of the JP15 campaign in northern Tanzania where vaccination often produced herd antibody prevalences in excess of 90 per cent. The thermostability of the vaccine has been improved through more stringent freeze-drying procedures. 185, 204, 205, 275, 338 This product, ‘Thermovax’, is very stable at high ambient temperatures and is now the vaccine of choice in areas with little or no ‘cold-chain’ facilities and where it is often administered by community-based animal health workers. The vaccine is, however, still sensitive to heat and light when reconstituted.
Several laboratories have attempted to address this problem by genetically engineering recombinant vaccines in which genes expressing RPV-immunizing antigen are incorporated into more thermostable poxviruses.142, 231, 381, 382 These recombinants have proved effective against experimental challenge in the short-term, but preliminary results from most longer term studies indicate that immunity is not as long lasting as with cell-culture vaccine,49, 231, 298, 300, 417, 420, 421 though one recent study claims that lasting immunity can be achieved even after a single inoculation.236 One advantage these vaccines do possess is the possibility of distinguishing vaccinated from naturally infected animals, which could be valuable in eradicating outbreaks. Another and perhaps more promising approach has been the recent development of recombinants of RPV itself. These new chimaeras and antigenically marked rinderpest viruses also offer the potential for serological differentiation between vaccinated and unvaccinated animals but without losing the known benefits of the homologous vaccine.33, 34, 91, 396, 397
The use of dry vaccine in subcutaneous implants66 or powder for application to mucosal surfaces7 could remove the ‘diluent problem’ in hot, isolated areas.
Prevention of rinderpest in endemic areas requires annual vaccination of all calves up to two years of age. Maternal antibodies from immune dams interfere with active immunization and may persist for 9 to 10 months.63, 64 In practice, however, few animals over six months of age have antibody and it is sensible to immunize the 6- to 12-month-old animals rather than only those over a year old in an effort to economize on vaccine. Where vaccination is difficult, infrequent or inefficient, and where cattle populations are continually mobile, annual vaccination of all cattle will produce the highest levels of herd immunity.265, 305 In such areas some serological monitoring of herd antibody levels is also advisable to assess whether mass vaccination campaigns have been successful or need to be repeated. The successful eradication of endemic rinderpest from Maasailand in 1962 and 1963 was accomplished by very efficient vaccination campaigns which raised overall herd immunity levels to 90 per cent or more. Values in excess of this have been calculated to be necessary for the eradication of measles virus from human populations and rinderpest from cattle.4, 305 Lower levels of vaccination coverage may also stop virus transmission on occasion, especially milder strains.305
In endemic areas sero-surveillance of unvaccinated animals such as small ruminants and wildlife is also important. These species are sentinels, as any antibody they possess must have been induced by field strains of RPV or related agents such as PPRV.
The development of the cell-culture vaccine opened the door to a world free from rinderpest. The Global Rinderpest Eradication Programme (GREP) has set the year 2010 as the target for complete eradication of the disease from the world, and very significant progress has been made in Africa and Asia in the last decade and a half. In Africa in 1999 the virus was restricted to southern Sudan and to the southern Somali pastoral ecosystem, neither of which has ever been completely under government administration, and both of which are now effectively war zones; and in Asia to parts of southern Pakistan. The challenge now faced is to safely access the cattle populations in these remaining foci using conventional and community-based techniques, which have proved successful elsewhere, including the Afar area of Ethiopia. If human and financial resources can be maintained, 395 the goal of global freedom from rinderpest could become a reality.17, 229, 319, 333, 370
References
- ABRAHAM, A., LEBEL, E. & DAVIDSON, M., 1984. Virological and serological investigations on rinderpest in Israel. Refuah Veterinarith, 41, 112–121.
- ADEMOLLO, A., 1958. A proposito di importazione di carne dai paesi infetti di peste bovina. Veterinaria Italiana, 9, 130–138.
- ALI, B., EL, H. & LEES, G.E., 1979. The application of immunoelectroprecipitation in the diagnosis of rinderpest. Bulletin of Animal Health and Production in Africa, 27, 1–6.
- ALI, B., EL, H. & TAYLOR, W.P., 1988. An investigation on rinderpest virus transmission and maintenance by sheep, goats and cattle. Bulletin of Animal Health and Production in Africa, 36, 290–294.
- ANDERSON, E.C., 1995. Morbillivirus infections in wildlife (in relation to their population biology and disease control in domestic animals). Veterinary Microbiology, 44, 319–332.
- ANDERSON, J., BARRETT, T. & SCOTT, G.R., 1996. Manual on the Diagnosis of Rinderpest. Rome, FAO.
- ANDERSON, J., FISHBOURNE, E., CORTEYN, A. & DONALDSON, A.I., 2001. Protection of cattle against rinderpest by intranasal immunization with a dry powder tissue culture vaccine. Vaccine, 19, 840–843.
- ANDERSON, E.C., HASSAN, A., BARRETT, T., & ANDERSON, J., 1990. Observations on the pathogenicity for sheep and goats and the transmissibility of the strain of virus isolated during the rinderpest outbreak in Sri Lanka in 1987. Veterinary Microbiology, 21, 309–318.
- ANDERSON, E.C., JAGO, M., MLENGEYA, T., TIMMS, C., PAYNE, A. & HIRJI, K., 1990. A serological survey of wildlife and sheep and goats in Northern Tanzania. Epidemiology and Infection, 105, 203–214.
- ANDERSON, J. & MCKAY, J.A., 1994. The detection of antibodies against peste des petits ruminants virus in cattle, sheep and goats and the possible implications to rinderpest control programmes. Epidemiology and Infection, 112, 225–231.
- ANDERSON, J., MCKAY, J.A. & BUTCHER, R.N., 1990. The use of monoclonal antibodies in competitive ELISA for the detection of antibodies to rinderpest and peste des petits ruminants viruses. In: jeggo, m.h., (ed.). The seromonitoring of rinderpest throughout Africa—phase one. Proceedings of a Final Research Co-ordination Meeting of the FAO/IAEA/ SIDA/OAU/IBAR/PARC Co-ordinated Research Programme. Ivory Coast, 1990. IAEA-TECDOC, 623; 43–53.
- ANDERSON, J. & ROWE, L.W., 1982. The use of an enzyme-labelled assay as an aid to reading micro virus neutralisation tests. Journal of Immunological Methods, 53, 182–186.
- ANDERSON, J., ROWE, L.W. & TAYLOR, W.P., 1983. Use of an enzyme-linked immunosorbent assay for the detection of IgG antibodies to rinderpest virus in epidemiological surveys. Research in Veterinary Science, 34, 77–81.
- ANDERSON, J., ROWE, L.W., TAYLOR, W.P. & CROWTHER, J., 1982. An enzyme-linked immunosorbent assay for the detection of IgG, IgA and IgM antibodies to rinderpest virus in experimentally infected cattle. Research in Veterinary Science, 32, 242–247.
- ANDERSON, R.M. & MAY, R.H., 1982. Directly transmitted infectious diseases: Control by vaccination. Science, 215, 1953–1060.
- ANON., 1920. La peste bovine en Belgique. Revue générale de Médécine Vétérinaire, 29, 577–583.
- ANON., 1949. Rinderpest vaccines: Their production and use in the field. FAO Agricultural Studies. No. 8, Washington: Food and Agricultural Organisation.
- ANON., 1963. International veterinary preventive medicine. British Veterinary Journal, 119, 336–337.
- ANON., 1966. Connaissances acquises récemment sur la peste bovine et son virus. Revue d’Elevage et de Médecine Vétérinaire des Pays Tropicaux, 19, 365–413.
- ANON., 1983. Rinderpest in West Africa. The Veterinary Record, 113, 145.
- ATA, F.A. & SINGH, K.V., 1982. Rinderpest in Kuwait. Journal of the Egyptian Veterinary Medical Association, , 27–32.
- ATANG, P.G. & PLOWRIGHT, W., 1969. Extension of the JP-15 Rinderpest Control Campaign to eastern Africa: The epizootiological background. Bulletin of Epizootic Diseases of Africa, 17, 161–170.
- BABU, Y.H., JAMES, R.M., SREENIVAS, C.S. & KHAN, M.A., 1984. The use of the immunoperoxidase test to the rinderpest viral antigens in infected tissue smears. Indian Veterinary Journal, 61, 271–275.
- BALDREY, F.S.H., 1906. Some observations on normal and rinderpest blood. Journal of Tropical Veterinary Science, 1, 47–69.
- BANSAL, R.P.,1986. Diagnosis of Rinderpest. With Special Reference to the Application of Modern Techniques. Mukteswar: Indian Veterinary Research Institute.
- BANSAL. R.P. & JOSHI, R.C., 1980. A note on rinderpest in Gaur (Bibos gaurus). Indian Veterinary Journal, 57, 259–260.
- BANSAL, R.P., JOSHI, R.C., SHARMA, B. & CHANDRA, U., 1987. Reverse phase passive haemagglutination test for the detection of rinderpest antigen. Tropical Animal Health and Production, 19, 53–55.
- BANSAL, R.P., JOSHI, R.C., TANDON, H.K.L. & KUMAR, S., 1981. Production of interferon by rinderpest virus in calf kidney cell cultures. Acta Virologica, 25, 61.
- BANSAL, R.P., YADAV, M.P., BANDYOPADHAY, S.K., SHARMA, B. & JOSHI, R.C., 1983. Proceedings of the Workshop on Recent Advancements in Rinderpest Diagnosis. 43pp. Mukteswar. Indian Veterinary Research Institute.
- BARON, M.D. & BARRETT, T., 1995. The sequence of the N and L genes of rinderpest virus, and the 5 and 3 extra-genic sequences: the completion of the genome sequence of the virus. Veterinary Microbiology, 44, 175–186.
- BARON, M.D. & BARRETT, T., 1995. Sequencing and analysis of the nucleocapsid (N) and polymerase (L) genes and the terminal extragenic domains of the vaccine strain of rinderpest virus. Journal of General Virology, 76, 593–602.
- BARON, M.D. & BARRETT, T., 1997. Rescue of rinderpest virus from cloned cDNA. Journal of Virology, 71, 1265–1271.
- BARON, M.D. & BARRETT, T., 2000. Rinderpest viruses lacking the C and V proteins show specific defects in growth and transcription of viral RNAs. Journal of Virology, 74, 2603–2611.
- BARON, M.D., FOSTER-CUEVAS, M., BARON, J. & BARRETT, T., 1999. Expression in cattle of epitopes of a heterologous virus using a recombinant rinderpest virus. Journal of General Virology, 80, 2031–2039.
- BARON, M.D., GOATLEY, L. & BARRETT, T., 1994. Cloning and sequence analysis of the matrix (M) protein gene of rinderpest virus and evidence for another bovine morbillivirus. Virology, 200, 121–129.
- BARON, M.D., KAMATA, Y., BARRAS, V., GOATLEY, L. & BARRETT, T., 1996. The genome sequence of the virulent Kabete O strain of rinderpest virus: Comparison with the derived vaccine. Journal of General Virology, 77, 3041–3046.
- BARON, M.D., SHAILA, M.S. & BARRETT, T., 1993. Cloning and sequence analysis of the phosphoprotein gene of rinderpest virus. Journal of General Virology, 74, 299–304.
- BARRETT, T., 1987. The molecular biology of the morbillivirus (measles) group. Biochemical Society Symposia, 53, 25–37.
- BARRETT, T., AMAREL-DOEL, C., KITCHING, R.P., & GUSEV, A., 1993. Use of the polymerase chain reaction in differentiating rinderpest field virus and vaccine virus in the same animal. Revue Scientifique et Technique de l’Office International des Epizooties, 12, 865–872.
- BARRETT, T., BELSHAM, G.J., SHAILA, M.S. & EVANS, S.A., 1989. Immunisation with a vaccinia recombinant expressing the F protein protects rabbits from challenge with a lethal dose of rinderpest virus. Virology, 170, 11–18.
- BARRETT, T., FORSYTH, M.A., INUI, K., WAMWAYI, H.M., KOCK, R., WAMBUA, J. MWANZIA, J. & ROSSITER, P.B., 1998. Rediscovery of the second African lineage of rinderpest virus: Its epidemiological significance. The Veterinary Record, 142, 669–671.
- BARRETT, T. & ROSSITER, P.B., 1999. Rinderpest: the disease and its impact on humans and animals. Advances in Virus Research, 53, 89–110.
- BARRETT, T. & UNDERWOOD, B., 1985. Comparison of messenger RNAs induced in cells infected with each member of the morbillivirus group. Virology, 145, 195–199.
- BASSIRI, M., AHMAD, S., GIAVEDONI, L., JONES, L., SALIKI, J.T., MEBUS, C. & YILMA, T., 1993. Immunological responses of mice and cattle to baculovirus-expressed F and H proteins of rinderpest virus: Lack of protection in the presence of neutralising antibody. Journal of Virology, 67, 1255–1261.
- BAWA, H.S., 1940. Rinderpest in sheep and goats in Ajmer Merwara. Indian Journal of Veterinary Science and Animal Husbandry, 10, 103–112.
- BAZ, T.I., ADAWY, A.T., MOHAMED, Z.A. & AHMED, I.K., 1983. Investigations on rinderpest outbreak in Egypt. II. Isolation of rinderpest (RP) virus. Agricultural Research Review, 61, 123–131.
- BEATON, W.G., 1930. Rinderpest in goats in Nigeria. Journal of Comparative Pathology, 43, 301–307.
- BEATON, W.G., 1932. Infectivity of shade dried hides from rinderpest infected cattle. Indian Journal of Veterinary Science and Animal Husbandry, 2, 86–87.
- BELSHAM, G.J., ANDERSON, E.C., MURRAY, P.K., ANDERSON, J. & BARRETT, T., 1989. Immune response and protection of cattle and pigs generated by a vaccinia virus recombinant expressing the F protein of rinderpest virus. The Veterinary Record, 124, 655–658.
- BERHANU, A.A., 1992. The epidemiology and epidemiological modelling of rinderpest in Ethiopia. MSc Thesis, University of Edinburgh.
- BHATTACHARYA, B. & CHAKRABORTY, A.K., 1979. Serum aminotransferase activity in normal and experimentally infected Jamunapuri goats with rinderpest virus. Indian Veterinary Journal, 56, 435–436.
- BIDJEH, K., BARRETT, T., THIRY, E., TILLE, A., DIALLO, A., CHAMBERLIN, R.W., LEFEVRE, P.-C., PASTORET, P.-P. & WAMWAYI, H.M., 1993. Virulence and transmissibility of different strains of rinderpest virus. Veterinary Pathology, 30, 544–554.
- BIDJEH, K., OUAGAL, M., DIALLO, A. & BORNAREL, P., 1996. Transmissions des souches du virus bovipestique de virulence variable aux chevres tchadiennes. Annales des Médecine Vétérinaire, 141, 65–69.
- BOGEL, K., RUCKLE-ENDERS, G. & PROVOST, A., 1964. Une réaction sérologique rapide de mesure des anticorps antibovipestiques. Comptes Rendus Hebdomadaires des Séances de l’Académie des Sciences, Paris, 259, 482–484.
- BOYNTON, W.H., 1914. A preliminary report of experiments on the cultivation of the virus of rinderpest in vitro. Philippine Journal of Science, B, 9, 39–44.
- BOYNTON, W.H., 1916. Rinderpest in swine with experiments upon the transmission from cattle and carabaos to swine and vice versa. Philippine Agricultural Reviews, 9, 288–336.
- BOYNTON, W.H., 1917. Preliminary report on the virulence of certain body organs in rinderpest. Philippine Agricultural Reviews, 10, 410–433.
- BRANAGAN, D., 1966. Behaviour of buffalo infected with rinderpest. Bulletin of Epizootic Diseases of Africa, 14, 341–342.
- BRANAGAN, D. & HAMMOND, J.A., 1965. Rinderpest in Tanganyika: A review. Bulletin of Epizootic Diseases of Africa, 13, 225–246.
- BREESE, S.S. & DE BOER, C.J., 1963. Electron microscopy of rinderpest virus in bovine kidney tissue culture cells. Virology, 19, 340–348.
- BRISTOWE, J.S., 1866. Morbid anatomy of the disease. In: Third report of the commissioners into the origin and nature etc. of the cattle plague. London: Her Majesty’s Stationery Office.
- BROTHERSTONE, J.G., 1951. Lapinized rinderpest virus and a vaccine. Some observations in East Africa. I. Laboratory experiments. Journal of Comparative Pathology, 61, 263–288.
- BROWN, R.D.,1958. Rinderpest immunity in calves. I. Acquisition and persistence of maternally derived antibody. Journal of Hygiene, Cambridge, 56, 427–434.
- BROWN, R.D., 1958. Rinderpest immunity in calves. II. Active immunization. Journal of Hygiene, Cambridge, 56, 436–444.
- BROWN, R.D., 1964. Endobronchial inoculation of cattle with various strains of Mycoplasma mycoides and the effects of stress. Research in Veterinary Science,
- BROWN, R.D. & GLOSSOP, W.E., 1965. Rinderpest immunization by means of vaccine implants in tablet form. Bulletin of Epizootic Diseases of Africa, 13, 305–309.
- BROWN, R.D. & LAMPKIN, G.H., 1961. Effect of caprinized rinderpest vaccine on the growth of weaner calves. East African Agriculture and Forestry Journal, 26, 156–157.
- BROWN, C.C., OJOK, L. & MARINER, J.C., 1995. Immunohistochemical detection of rinderpest virus: Effects of autolysis and period of fixation. Research in Veterinary Science, 60, 182–184.
- BROWN, R.D. & SCOTT, G.R., 1957. Mucosal disease complex. The Veterinary Record, 12, 287–298.
- BROWN, R.D. & SCOTT, G.R., 1960. Vaccination of game with lapinized rinderpest virus. The Veterinary Record, 72, 1232–1233.
- BROWN, C.W., SCOTT, G.R. & BROTHERSTON, J.G., 1955. Lapinized rinderpest vaccine: Post inoculation reactions in high-grade Guernsey cattle. The Veterinary Record, 67, 467–468.
- BROWN, R.D. & TAYLOR, W.P., 1966. Simultaneous vaccination of cattle against rinderpest and contagious bovine pleuropneumonia. Bulletin of Epizootic Diseases of Africa, 14, 141–146.
- BROWN, C.C. & TORRES, A., 1994. Distribution of antigen in cattle infected with rinderpest virus. Veterinary Pathology, 31, 194–200.
- BRUNING, A., BELLAMY, K., TALBOT, D. & ANDERSON, J., 1999. A rapid chromatographic test for the pen-side diagnosis of rinderpest virus. Journal of Virological Methods, 81, 143–154.
- CARMICHAEL, J., 1938. Rinderpest in African game. Journal of Comparative Pathology, 51, 264–268.
- CARMICHAEL, J., 1939. Attempts to propagate the virus (rinderpest) in tissue culture. Annual Report of the Veterinary Department, Uganda, Entebbe: Government Printer.
- CHAMBERLAIN, R.W., WAMWAYI, H.M., HOCKLEY, E., SHAILA, M.S., KNOWLES, N.J., & BARRET, T. 1994. Evidence for different lineages of rinderpest virus reflecting their geographic isolation. Journal of General Virology, 74, 2775–2780.
- CHENEAU, Y., 1985. Prevalence of rinderpest and strategy of the Pan-African Campaign. Revue Scientifique et Technique de l’Office International des Epizooties, 4, 415–425.
- CILLI, V., MAZZARACHIO, V. & ROETTI, C., 1951. L’epidosio di peste bovina al Giardiono Zoologico di Roma. Archivio Italiano di Scienze Mediche Tropicali e di Parassitologia, 32, 83–94.
- CLAVERIE, J., 1931. La lutte contre la peste bovine en Guinée française. Recueil de Médecine Vétérinaire Exotique, 4, 121–140.
- CONTI, P.A. & VAN DAC, N.G., 1929. Utilisation du porcelet pour l’entretien du virus pestique servant aux pratiques d’immunisation antiseptique. Recueil de Médicine Vétérinaire Exotique, 2, 205–215.
- COOPER, H., 1932. Rinderpest. Transmission of infection by contact. Indian Journal of Veterinary Science and Animal Husbandry, 2, 284–392.
- CORNELL, R.L. & EVANS, S.A., 1937. On the values and limitations of tissue vaccines against rinderpest. Journal of Comparative Pathology, 50, 122–135.
- CORNELL, R.L. & REID, N.R., 1934. Rinderpest in wildebeest. Annual report of the Department of Veterinary Science and Animal Husbandry, Tanganyika Territory, 1933. Dar-es-Salaam: Government Printer.
- COSBY, S.L., MCQUAID, S., DUFFY, N., LYONS, C., RIMA, B.K., ALLAN, G.M., MCCULLOUGH, G.M., KENNEDY, S., SMYTH, J.A., MCNEILLY, F., CRAIG, C. & ORVELL, C., 1988. Characterisation of a seal morbillivirus. Nature, 336, 115–116.
- CRAWFORD, M., 1947. The immunology and epidemiology of some virus diseases. The Veterinary Record, 59, 537–540.
- CURASSON, G., 1932. La Peste Bovine. Paris: Vigot Frères.
- CURASSON, G., 1942. La peste bovine. In: Traité de Pathologie Exotique Vétérinaire et Comparée, Vol. I. Paris: Vigot Frères.
- CURSON, H.H., 1919. Rinderpest in the New Langenberg Province of German East Africa during 1917–1918. Journal of Comparative Pathology and Therapeutics, 32, 197–210.
- DARBYSHIRE, J.H., BROWN, R.D., SCOTT, G.R. & HUCK, R.A., 1961. A serological differentiation of rinderpest and bovine mucosal disease by agar gel diffusion. The Veterinary Record, 73, 255–256.
- DAS, S.C., BARON, M.D. & BARRETT, T., 2000. Recovery and characterization of a chimeric rinderpest virus with the glycoproteins of peste des petits ruminants virus: Homologous F and H proteins are required for virus viability. Journal of Virology, 74, 9039–9047.
- DATTA, C.A. & RAJAGOPOLAN, V.R., 1932. An unusual case of chronic rinderpest with special reference to the carrier problem in this disease. Indian Journal of Veterinary Science and Animal Husbandry, 2, 357–382.
- DAUBNEY, R., 1928. Observations on rinderpest. Journal of Comparative Pathology, 41, 228–248, 263–297.
- DAUBNEY, R., 1929. Prophylactic vaccination against rinderpest. Proceedings of the Pan-African Veterinary Conference, Pretoria.
- DAUBNEY, R., 1949. Goat-adapted virus. In: KESTEVEN, K.V.L. (ed.). Rinderpest Vaccines: Their Production and Use in the Field. FAO Agricultural Studies No 8. Washington: Food and Agriculture Organisation.
- D’COSTA, J. & SINGH, B., 1933. Rinderpest: Clinical syndrome in goats in India. Indian Journal of Veterinary Science and Animal Husbandry, 3, 122–128.
- DE LAY, P.D. & BARBER, T.L., 1962. Transmission of rinderpest virus from experimentally infected cattle to pigs. Proceedings of the United States Livestock Sanitary Association 65th Annual Meeting, 1961, Minneapolis.
- DE LAY, P.D., MOULTON, W.M. & STONE, S.S., 1962. Survival of rinderpest virus in experimentally infected swine. Proceedings of the United States Livestock Sanitary Association 65th Annual Meeting, 1961, Minneapolis.
- DELPY, L., 1930. Les formes atypiques de la peste bovine et les porteurs de virus. Revue générale de Médecine Vétérinaire, 39, 138–141.
- DHAR, P., JOSHI, R.C. & BANDYOPADHAY, S.K., 1995. Humoral antibody response in animals infected with virulent rinderpest virus. Tropical Animal Health and Production, 27, 26–30.
- HILLON, S.S., 1959. Incidence of rinderpest in camels in Hissar district. Indian Veterinary Journal, 36, 603–607.
- DHIR, R.C., DIXIT, N.K., ADAVAL, S.C. & TIWARI, G.P., 1987. Blood biochemical changes in male buffalo calves during rinderpest infection. Indian Journal of Animal Sciences, 57, 177–179.
- DIALLO, A., BARRETT, T., BARBRON, M., SHAILA, M.S. & TAYLOR, W.P., 1989. Differentiation of rinderpest and peste des petits ruminants viruses using specific cDNA clones. Journal of Virological Methods, 23, 127–136.
- DIALLO, A., BARRETT, T., LEFEVRE, P.C. & TAYLOR, W.P., 1987. Comparison of proteins induced in cells infected with rinderpest and peste des petits ruminants virus. Journal of General Virology, 68, 2033–2038.
- DOUTRE, M.P., CHAMBRON, J. & BOURDIN, P., 1972. Valeur de l’immunité conférée par un vaccin mixte antibovipestique-antipéripneumonique lyophilisé préparé à l’aide de la souche T1 (S-R). Revue d’Elevage et de Médecine Vétérinaire des Pays Tropicaux, 25, 1–14.
- EDWARDS, J.T., 1925. Rinderpest (Etiological researches). Report of the Imperial Bacteriological Institute, Mukteswar for 1922–23. pp. 30–35.
- EDWARDS, J.T., 1930. The problem of rinderpest in India. Bulletin 199 of the Imperial Institute of Agricultural Research, Pusa, Calcutta, Government of India, Central Publications Branch.
- ELANKUMARAN, S., GNANABARANAM, N.F., PALANISWAMI, K.S., CHANDRAN, N.D.J., 1992. Natural contact transmission of rinderpest from cattle to goats. Indian Veterinary Journal, 69, 1055–1056.
- EPSTEIN, H., 1971. The Origin of the Domestic Animals of Africa. New York: Africana Publishing Corporation.
- EVANS, S.A., 1932. Rinderpest in goats and sheep. Annual Report of the Department of Veterinary Science and Animal Husbandry, Tanganyika Territory, 1931. Dar-es-Salaam: Government Printer.
- EVANS, S.A., BELSHAM, G.J. & BARRETT, T., 1990. The role of the 5 non-translated regions of the fusion protein mRNAs of canine distemper virus and rinderpest viruses. Virology, 177, 317–323.
- FLEMING, G., 1882. Animal Plagues: Their History, Nature and Prevention. Vol. II. London: Baillière Tindal.
- FORMAN, A.J., ROWE, L.W. & TAYLOR, W.P., TROPICAL ANIMAL HEALTH AND PRODUCTION, 15, 83–85.
- FORSYTH, M.A. & BARRETT, T., 1995. Evaluation of polymerase chain reaction for the detection and characterisation of rinderpest and peste des petits ruminants viruses for epidemiological studies. Virus Research, 39, 151–163.
- FRENCH, M.H., 1936. The serum protein changes induced by rinderpest virus. Journal of Comparative Pathology, 49, 147–156.
- FUKUDA, A. & YAMANOUCHI, K., 1981. Comparison of autoimmunity induction with virulent and attenuated virus in rabbits. Japanese Journal of Medical Science and Biology, 34, 149–159.
- FUKUSHO, K. & NAKAMURA, J., 1940. On the experimental infection with rinderpest virus in the rabbit. II. Pathology. Japanese Journal of Veterinary Science, 2, 75–101.
- GAJENDRAGAD, M.R., SHARMA, B., PRABHUDAS, K., JOSHI, R.C. & BANSAL, R.P., 1983. Immunoperoxidase staining of rinderpest virus antigen in formalin-fixed paraffin-embedded sections. Indian Journal of Veterinary Pathology, 7, 79–81.
- GAMGEE, J., 1866. The Cattle Plague. London: Hardwick.
- GHAFFAR, S.A. & ATA, F.A., 1982. Bovine virus diarrhoea in Egypt. The Veterinary Record, 110, 566.
- GHOSH, A., JOSHI, V.D. & SHAILA, M.S., 1995. Characterisation of an in-vitro transcription system from rinderpest virus. Veterinary Microbiology, 44, 165–173.
- GHOSH, A., NAYAK, R. & SHAILA, M.S., 1996. Synthesis of leader RNA and editing of P mRNA during transcription by rinderpest virus. Virus Research, 41, 69–76.
- GIAVEDONI, L., JONES, L., MEBUS, C. & YILMA, T., 1991. A vaccinia virus double recombinant expressing the F and H genes of rinderpest virus protects cattle against rinderpest and causes no pock lesions. Proceedings of the National Academy of Science, USA, 88, 8011–8015.
- GIBBS, C.S., 1933. Filterable virus carriers. Journal of Infectious Diseases, 53, 169–174.
- GOVINDARAJAN, R., MURALIMANOHAR, B., KOTEESWARAN, A., VENUGOPALAN, A.T., VARALAKSHMI, P., SHAILA, M.S. & RAMACHANDRAN, S., 1996. Occurrence of rinderpest in European pigs in India. The Veterinary Record, 139, 473.
- GRAY, C.E., 1917. Rinderpest in East Africa. London: Colonial Office.
- GRUBMAN, M.J., MEBUS, C., DALE, B., YAMANAKA, M. & YILMA, T., 1988. Analysis of the polypeptides synthesized in rinderpest virus-infected cells. Virology, 163, 261–267.
- GUITTIN, P., 1981. Rinderpest epizootic in Niger. Action Vétérinaire, 826, 6–8.
- HAJI, C.S.G., 1932. Rinderpest in camels. Indian Veterinary Journal, 9, 13–14.
- HALL, G.N., 1933. Investigations of rinderpest immunization D.V.M. Thesis, University of Zürich.
- HASSAN, A.K.M. & EL TOM, K., 1985. Combined natural infection with infectious bovine rhinotracheitis and rinderpest viruses. Tropical Animal Health and Production, 17, 52.
- HASSAN, D.G.A., KARIM, I.A., ARAB, R.M., SABER, M.S., AMIN, M.M., OSMAN, O.A. & REDA, I.M., 1987. Studies on rinderpest and diseases like rinderpest in Egypt. Journal of the Egyptian Veterinary Medical Association, 47, 399–416.
- HAYES, J.W., 1927. Contagious bovine pleuropneumonia in Tanganyika Territory. Annual Report of the Department of Veterinary Sciences and Animal Husbandry, Tanganyika Territory, 1926. Dar-es-Salaam: Government Printer.
- HEDGER, R.S., TAYLOR, W.P., BARNETT, I.T.R., RIEK, R. & HARPHAM, D., 1986. Simultaneous vaccination of cattle against foot-and-mouth disease and rinderpest. Tropical Animal Health and Production, 18, 21–25.
- HENNING, M.W., 1956. Animal Diseases in South Africa. 3rd edn. Pretoria: Central News Agency.
- HEUSCHELLE, W.P. & BARBER, T.L., 1963. Haematologic studies of rinderpest-infected swine. Canadian Journal of Comparative Medicine and Veterinary Science, 27, 56–60.
- HEUSCHELE, W.P. & BARBER, T.L., 1966. Changes in certain blood components of rinderpest-infected cattle. American Journal of Veterinary Research, 27, 1001–1006.
- HOLMES, J.D.E., 1904. Some diseases complicating rinderpest among cattle in India. Journal of Comparative Pathology, 17, 317–326.
- HORNBY, H.E., 1926. Studies in rinderpest immunity. II. Methods of infection. The Veterinary Journal, 82, 348–355.
- HORNBY, H.E., 1932. Disease control. Rinderpest. Annual Report of the Department of Veterinary Science and Animal Husbandry. Tanganyika Territory, 1931. Dar-es-Salaam: Government Printer.
- HORNBY, H.E., 1934. Pathogenicity of Trypanosoma theileria and Streptothricosis in cattle concurrently infected with rinderpest. Annual Report of the Department of Veterinary Science and Animal Husbandry, Tanganyika Territory, 1933. Dar-es-Salaam: Government Printer.
- HSU, D., YAMANAKA, M., MILLER, J., DALE, B., GRUBMAN, M. & YILMA, T., 1988. Cloning of the fusion gene of rinderpest virus: Comparative sequence analysis with other morbilliviruses. Virology, 166, 149–153.
- HUDSON, J.R. & WONGSONGSARN, C., 1950. The utilization of pigs for the production of lapinized rinderpest virus. British Veterinary Journal, 106, 453–472.
- HUSSAIN, M., IQBAL, M., TAYLOR, W.P. & ROEDER, P.L., 2001. Pen-side test for the diagnosis of rinderpest in Pakistan. The Veterinary Record, 149, 300–302.
- HUSSAIN, S.F., RWEYEMAMU, M.M., KAMINJOLO, J.S., AKHTAR, A.S. & MUGERA, G.M., 1980. Studies on viral interference induced by rinderpest virus: Interference and interferon induction by tissue culture rinderpest vaccine (TCRV) virus in vitro. Zentralblatt für Veterinärmedizin, 27B, 181–189.
- HUSSAIN, S.F., RWEYEMAMU, M.M., KAMINJOLO, J.S., AKHTAR, A.S. & MUGERA, G.M., 1982. Studies on viral interference induced by rinderpest virus: IV. Induction of circulating interferon and antibodies in buffaloes following inoculation with tissue culture rinderpest vaccine (TCRV) virus. Bulletin of Animal Health and Production in Africa, 30, 7–10.
- HUTCHEON, D., 1902. Rinderpest in South Africa. Journal of Comparative Pathology, 15, 300–342.
- HUYGELEN, C., 1960. Failure to demonstrate agglutination of red blood cells by rinderpest virus. Bulletin of Epizootic Diseases of Africa, 8, 121–125.
- HUYTRA, F., MAREK, J. & MANNINGER, R., 1964. Rinderpest. In: Special Pathology and Therapeutics of the Diseases of Domestic Animals. 5th English edn. London: Ballière, Tindall & Cox.
- HYSLOP, N. ST. G., 1979. Observations on the survival and infectivity of airborne rinderpest virus. International Journal of Bioclimatology and Biometeorology, 23, 1–7.
- IDNANI, J.A., 1944. Transmission of rinderpest by expired air. Indian Journal of Veterinary Science and Animal Husbandry, 14, 216–220.
- IKEDE, B.O., 1984. Rinderpest epidemics in Tropical Africa. Tropical Veterinarian, 2, 1–2.
- IMAGAWA, D.T., GORET, P. & ADAMS, J.M., 1960. Immunological relationships of measles, distemper and rinderpest viruses. Proceedings of the National Academy of Sciences, 46, 1119–1123.
- INOUE, T., HARADA, S. & SHIMIZU, T., 1930. Preliminary note on the experimental infection with rinderpest virus of susliks. Selected Contributions from the Mukden Institute for Infectious Diseases of Animals, 1, 221–222. (Abstracted in Veterinary Bulletin, 1, 53).
- INUI, K., BARRETT, T., KITCHING, R.P. & YAMANOUCHI, K., 1995. Long-term immunity in cattle vaccinated with a recombinant rinderpest vaccine. The Veterinary Record, 137, 669–670.
- ISOGAI, S., 1994. On the rabbit-virus inoculation as an active immunization method against rinderpest for Mongolian cattle. Japanese Journal of Veterinary Science,6, 388–390.
- JACOTOT, H., 1931. L’infection pestique qui entraine l’avortement peut-elle être propagée par le foetus et la femelle qui l’a expulsée. Bulletin de la Société de Pathologie Exotique et de ses Filiales, 24, 74–76.
- JACOTOT, J., 1950. Rapport concernant le contrôle et la standardisation des sérums et vaccins contre la peste bovine. Bulletin de l’Office International des Epizooties, 33, 168–183.
- JACOTOT, H. & MORNET, P., 1967. La Peste Bovine. Paris: L’Expansion Scientifique Française.
- JAMES, F.L., 1883. The Wild Tribes of the Sudan. New York: Negro Universities Press.
- JAMES, A.D. & ROSSITER, P.B., 1989. An epidemiological model of rinderpest. I. Description of the model. Tropical Animal Health and Production, 21, 59–68./li>
- JEGGO, M.H., WARDLEY, R.C. & CORTEYN, A.K., 1987. A reassessment of the dual vaccine against rinderpest and contagious bovine pleuropneumonia. The Veterinary Record, 120, 131–135.
- JENKINS, D.L. & SHOPE, R.E., 1946. Rinderpest. VII. The attenuation of rinderpest virus for cattle by cultivation in embryonating eggs. American Journal of Veterinary Research, 7, 174–178.
- JOHNSON, R.H., 1962. Rinderpest in tissue culture: Use of the attenuated strain as a vaccine for cattle. British Veterinary Journal, 118, 141–150.
- JOSHI, R.C., CHAUDHARY, P.G. & BANSAL, R.P., 1977. Occurrence of cutaneous eruptions in rinderpest outbreak among bovines. Indian Veterinary Journal, 54, 871–873.
- JOSHI, R.C., SHARMA, B., BANDYOPADHAY, S.K. & BANSAL, R.P., 1984. Detection of rinderpest antibodies. Tropical Animal Health and Production, 16, 167–170.
- KAKIZAKI, C., 1918. Study on the glycerinated rinderpest vaccine. Kitasato Archives of Experimental Medicine, 2, 59–66.
- KAMATA, H., OHKUBO, S., SUGIYAMA, M., MATSUURA, Y., KAMATA, Y., TSUKIYAMA-KOHARA, K., IMAOKA, K., KAI, C., YOSHIKAWA, Y. & YAMANOUCHI, K., 1993. Expression in baculovirus vector system of the nucleocapsid protein gene of rinderpest virus. Journal of Virological Methods, 43, 159–166.
- KAPWADI, K., 1987. Trial of simultaneous vaccination against rinderpest and brucellosis in cattle in Zaïre. Revue Scientifique et Technique de l’Office International des Epizooties, 6, 1043–1049.
- KATHURIA, B.K., UPPAL, P.K. & KUMAR, S., 1976. Studies on simultaneous vaccination of cattle against rinderpest and foot-and-mouth disease. Indian Veterinary Journal, 53, 571–576.
- KEYLOCK, H.E., 1933. The control of rinderpest in a large dairy herd in Shanghai, China. Journal of Comparative Pathology, 46, 149–158.
- KHERA, K.S., 1958. Etude histologique de la peste bovine. I. Pathogenèse du virus de la peste bovine dans les ganglions lymphatiques. Revue d’Elevage et de Médecine Vétérinaire des Pays Tropicaux, 11, 399–405.
- KHERA, K.S., 1958. Etude histologique de la peste bovine. II. Lésions histologiques au niveau du tractus digestif. Revue d’Elevage et de Médecine Vétérinaire des Pays Tropicaux, 11, 406–415.
- KHERA, K.S., 1958. Etude histologique de la peste bovine. III. Lésions dans les différents organes. Revue d’Elévage et de Médecine Vétérinaire des Pays Tropicaux, 11, 416–420.
- KINGSBURY, D.W., BRATT, M.A., CHOPPIN, P.W., HANSON, R.P., HOSAKA, Y., TER MEULEN, V., NORRBY, E., PLOWRIGHT, W., ROTT, R. & WUNNER, W.H., 1978. Paramyxoviridae.Intervirology, 10, 137–152.
- KOBUNE, F., CHINO, F. & YAMANOUCHI, K., 1976. Studies on recovery mechanism from rinderpest virus infection in rabbits. I. Effect of anti-thymocyte serum and thymectomy. Japanese Journal of Medical Science and Biology, 29, 265–275.
- KOBUNE, F., YAMANOUCHI, K., NAGASHIMA, K. & SHISHIDO, A., 1981.Establishment of a persistently infected cell line with rinderpest virus. Archives of Virology, 68, 271–277.
- KOCH, R., 1897. Berichte des Herrn Prof. Dr. Koch über seine in Kimberley gemachten versuche bezuglich bekämpfung der rinderpest. Zentralblatt für Bakteriologie, 1, 21, 526–537.
- KOCK, R.A., WAMBUA, J., MWANZIA, J., WAMWAYI, H., NDUNGU, E.K., BARRETT, T., KOCK, N.D, & ROSSITER, P.B., 1999. Rinderpest epidemic in wild ruminants in Kenya 1994–7. The Veterinary Record, 145, 275–283.
- KOTZÉ, J.P., 1974. Die runderpes in die Transvaal en die onmiddellike gevolge daarvan 1896–99. Magister in die Lettere en Wysbegeerte in Geskiedenis, Randse Afrikaanse Universiteit, Johannesburg, Suid-Afrika.
- KRISHNASWAMY, S., KESHAVAMURTHY, B.S. & SUNDARARAJAM, S., 1981. The use of the direct immunoperoxidase test to detect the multiplication of rinderpest virus in bovine kidney cell culture. Veterinary Microbiology, 6, 23–29.
- KRISHNASWAMY, S. & VEERABHADRAPPA, G., 1982. Intra-cellular polypeptides in the rinderpest virus infected cells. Indian Journal of Comparative Microbiology, Immunology and Infectious Diseases, 3, 68–71.
- KULKARNI, D.D., AHER, V.D. & BANNALIKER, A.S., 1990. Rinderpest in crossbred calves weaned at birth. The Veterinary Record, 127, 479.
- KURCHENKO, F.P., NIKISHIN, I.V., KARPOV, G.M., VISHNYAKOV, I.F., BAKULOV, I.A., BUSUN, A.I., BLABANOV, V.A., MITIN, N.I., ARSHIPOV, N.I., MALAKHOVA, M.S. & KOPTEVA, A.A., 1995. Isolation and identification of cattle plague virus in Georgia. Russian Veterinary Journal, 8, 27–31.
- LANGUET, B., PRECAUSTA, P., MACKOWIAK, M., DUBOURGET, P., REYLNAUD, E. & DURET, C., 1985. Freeze-dried vaccine against rinderpest: Stability and activity study. Journal of Comparative Immunology, Microbiology and Infectious Diseases, 8, 285–295.
- lEISS, B., 1963. Fluoreszenzserologische untersuchungen an zellkulturen nach infektion mit rinderpestvirus. Zentralblatt für Bakteriologie, 190, 424–444.
- LEISS, B., 1964. Untersuchungen über des virus der rinderpest unter verwendung von zellkulturen. Archives of Experimental Veterinary Medicine, 20, 157–257.
- LEISS, B. & PLOWRIGHT, W., 1964. Studies on the pathogenesis of rinderpest in experimentally infected cattle. I. Correlation of clinical signs, viraemia and virus excretion by various routes. Journal of Hygiene, 62, 81–100.
- LEPISSIER, H.E., 1983. Pan-African Campaign against Rinderpest. Organization and Logistics. Nairobi: Inter-African Bureau of Animal Resources, Organization of African Unity.
- LIBEAU, G., DIALLO, A., CALVEZ, D. & LEFEVRE, P.C., 1992. A competitive ELISA using anti-N monoclonal antibodies for specific detection of rinderpest antibodies in cattle and small ruminants. Veterinary Microbiology, 31, 147–160.
- LIBEAU, J. & SCOTT, G.R., 1960. Rinderpest in eastern Africa today. Bulletin of Epizootic Diseases of Africa, 8, 23–26.
- LIMO, M. & YILMA, T., 1990. Molecular cloning of the rinderpest virus matrix (M) gene: Comparative sequence analysis with other paramyxoviruses. Virology, 175, 323–327.
- LINGARD, A., 1905. Report of the Imperial Institute for Bacteriology 1905–06.
- LITTLEWOOD, W., 1905. Cattle plague in Egypt in 1903–04–05. Journal of Comparative Pathology, 18, 312–321.
- LORETU, K., 1996. Studies on the lack of rinderpest virus persisting in North Tanzania. Bulletin of Animal Health and Production in Africa, 44, 91–94.
- LOWE, H.J., 1942. Rinderpest in Tanganyika Territory. Empire Journal of Experimental Agriculture, 10, 189–202.
- LOWE, J.J., WILDE, J.K.H., LEE, R.P. & STUTCHBERRY, H.M., 1947. An outbreak of an aberrant type of rinderpest in Tanganyika Territory. Journal of Comparative Pathology, 57, 175–183.
- LUND, B.T. & BARRETT, T., 2000. Rinderpest virus infection in primary bovine skin fibroblasts. Archives of Virology, 145, 1231–1237.
- LUND, B.T., TIWARI, A., GALBRAITH, S., BARON, M.D., MORRISON, W.I. & BARRETT, T., 2000. Vaccination of cattle with attenuated rinderpest virus stimulates CD4+ T cell responses with broad viral antigen specificity. Journal of General Virology, 81, 2137–2146.
- MACADAM, I., 1968. Transmission of rinderpest from goats to cattle in Tanzania. Bulletin of Epizootic Diseases of Africa, 16, 53–60.
- MACADAM, I., EZEBUIRO, E.O. & OREFFO, V.O.C., 1964. The response of zebu cattle to mixed bovine pleuropneumonia and tissue culture rinderpest vaccine. Bulletin of Epizootic Diseases of Africa, 12, 237–240.
- MACK, R., 1970. The great African cattle plague epidemic of the 1890s. Tropical Animal Health and Production,2, 210–219.
- MAHY, B.W.J., BARRETT, T., EVANS, S., ANDERSON, E.C. & BOSTOCK, C.J., 1988. Characterization of a seal morbillivirus. Nature, 336, 115.
- MARINER, J.C., HOUSE, J.A., SOLLOD, A.E., STEM, C., VAN DEN ENDE, M. & MEBUS, C.A., 1990. Comparison of the effect of various chemical stabilizers and lyophilization cycles on the thermostability of a Vero cell-adapted rinderpest vaccine. Veterinary Microbiology, 21, 195–209.
- MARINER, J.C., VAN DEN ENDE, M.C., HOUSE, J.A., MEBUS, C.A., SALIFOU, S. & STEM, C., 1990. The serological response to a thermostable Vero cell-adapted rinderpest vaccine under field conditions in Niger. Veterinary Microbiology, 22, 119–127.
- MASIGA, W., HALPIN, B. & CHENEAU, Y., 1988. Rinderpest. In: williams, a.o. & masiga, w., (eds). Viral Diseases of Animals in Africa. Lagos: Scientific, Technical and Research Committee, Organization of African Unity.
- MAURER, F.D., JONES, T.C., EASTERDAY, B. & DE TRAY, D.E., 1955. Pathology of rinderpest—an abstract. Journal of the American Veterinary Medical Association, 127, 512–514.
- MAURER, F.D., JONES, T.C., EASTERDAY, B. & DE TRAY, D.E., 1956. The pathology of rinderpest. In: Proceedings of the 92nd Annual Meeting of the American Veterinary Medical Association, 1955, Minneapolis.
- MCCULLOUGH, K.C., OBI, T.U. & SHESHBERADARAN, H., 1991. Identification of epitope(s) on the internal virion proteins of rinderpest virus which are absent from peste des petits ruminants virus. Veterinary Microbiology, 26, 313–321.
- MCCULLOUGH, K.C., SHESHBERADARAN, H., NORRBY, E., OBI, T.U. & CROWTHER, J.R., 1986. Monoclonal antibodies against morbilliviruses. Revue Scientifique et Technique de l’Office International des Épizooties, 5, 411–427.
- METTAM, R.W.M., 1937. A short history of rinderpest with special reference to Africa. The Uganda Journal, 5, 22–26.
- MIRCHAMSY, H., SHAFYI, A. & BAHRAMI, S., 1970. Use of Vero cells for titration of rinderpest virus and its neutralizing antibody. Applied Microbiology, 19, 545–546.
- MOHAMED, E.H.M., 1983. Rapid diagnosis of rinderpest. M.Sc. Thesis, University of Reading, United Kingdom.
- MOHAMMED, Z.E., HAFEZ, S.M. & OZAWA, Y., 1977. Studies on the methods of preparation of rinderpest hyperimmune serum in rabbits. Tropical Animal Health and Production, 9, 25–28.
- MOHAN, R. & BAHL, M.R., 1953. Cutaneous eruptions of rinderpest in goats. Indian Journal of Veterinary Science, 23, 39–42.
- MORNET, P., 1948. Schéma epizootiologique de la peste bovine en Afrique Occidentale Française. Bulletin du Service de l’Élevage et de l’Industrie Animal, A.O.F., 1, 7–12.
- MORNET, P. & GILBERT, Y., 1958. Les méthodes actuelles de lutte contre la peste bovine. Les Cahiers de Médecine Vétérinaire, 27, 1–52.
- MORNET, P. & GUERRET, M., 1950. Les lésions cutanées dans la peste bovine. Bulletin de l’Académie Vétérinaire de France, 23, 283–285.
- MOUSTAFA, T., 1993. Rinderpest and peste des petits ruminants-like disease in the Al-Ain region of the United Arab Emirates. Revue Scientifique et Technique de l’Office International des Epizooties, 12, 857–863.
- MULLICK, S.G., RAMACHANDRAN, S.P. & RAMACHANDRAN, S., 1982.Preparation of rinderpest precipitins using adjuvants. Indian Veterinary Journal, 59, 833–836.
- NACHIMUTHU, K., DHINAKAR RAJ, G., THANGAVELU, A., GOVINDRAJAN, R. & VENUGOPALAN, A.T., 1993. Transmission studies with rinderpest virus. Indian Journal of Animal Sciences, 63, 363–367.
- NAGINGTON, J., LANDER, I.M. & SMITH, J.S., 1967. Bovine papular stomatitis, pseudocowpox and milker’s nodules. The Veterinary Record, 81, 306–312.
- NAKAMURA, J., 1940. On the experimental infection with rinderpest virus in the rabbit. III. Neutralization experiment.,em> Japanese Journal of Veterinary Science, 2, 567–578.
- NAKAMURA, J., 1957. Peste Bovine. Bulletin de l’Office International des Épizooties, 47, 542–554.
- NAKAMURA, J. & MIYAMOTO, T., 1953. Avianisation of lapinized rinderpest virus. American Journal of Veterinary Research, 14, 307–317.
- NAKAMURA, J., WAGATUMA, S. & FUKOSHO, K., 1938. On the experimental infection with rinderpest virus in the rabbit. I. Some fundamental experiments. Journal of the Japanese Society of Veterinary Science, 17, 185–204
- NARAYANASWAMY, M. & RAMANI, K., 1973. Preliminary studies on rinderpest virus isolated from outbreaks in sheep in Mysore State. Indian Veterinary Journal, 50, 829–832.
- NAWATHE, D.R. & LAMORDE, A.G., 1982. Recurrence of rinderpest in Nigeria. The Veterinary Record, 11, 203.
- NAWATHE, D.R. & LAMORDE, A.G., 1983. Towards global eradication of rinderpest. Revue Scientifique et Technique, Office International des Epizooties, 2, 997–1011.
- NAWATHE, D.R., LAMORDE, A.G. & KUMAR, S., 1983. Recrudescence of rinderpest in Nigeria. The Veterinary Record, 113, 156–157.
- NGICHABE, C.K., WAMWAYI, H.M., BARRETT, T., NDUNGU, E.K., BLACK, D.N. & BOSTOCK, C. J., 1997. Trial of a capripox-rinderpest recombinant vaccine in African cattle. Epidemiology and Infection, 118, 63–70.
- NOCARD, E. & LECLAINCHE, E., 1896. Peste bovine. In: Les Maladies Microbiennes des Animaux. Paris: Masson.
- NORRBY, E., SHESBERADARAN, H., MCCULLOUGH, K.C., CARPENTER, W.C. & ORVELL, C., 1985. Is rinderpest the archvirus of the morbillivirus genus? Intervirology, 23, 228–232.
- NYANGE, J.F.C., OTARU, M.M.M. & MBISE, A.N., 1985. Incidences of abnormal wild animal mortality in northern Tanzania, 1979–1982. Bulletin of Animal Health and Production in Africa, 33, 55–57.
- OFCANSKY, T.P., 1981. The 1889–97 rinderpest epidemic and the rise of British and German colonialism in eastern and southern Africa. Journal of African Studies, 8, 31–38.
- OHISHI, K., INUI, K., BARRETT, T. & YAMANOUCHI, K., 2000. Long-term protective immunity to rinderpest in cattle following a single vaccination with a recombinant vaccinia virus expressing the virus haemagglutinin protein. Journal of General Virology, 81, 1439–1446.
- OHISHI, K., INUI, K., YAMANOUCHI, K. & BARRETT, T., 1999. Cell-mediated immune responses in cattle vaccinated with a vaccinia virus recombinant expressing the nucleocapsid protein of rinderpest virus. Journal of General Virology, 80, 1627–1634.
- OHISHI, K., KAMATA, H., YAMANOUCHI, K. & BARRETT, T., 2000. Identification of T-helper cell epitopes in the hypervariable region of the nucleocapsid (N) protein of rinderpest virus (RPV) in cattle. Vaccine, 18, 3077–3081.
- OKITA, M., MORI, T., SHIN, Y.-S., MIYASAKA, M., YAMANOUCHI, K., MIKAMI, T. & KAI, C., 1995. Immunohistochemical studies of lymphoid tissues of rabbits infected with rinderpest virus. Journal of Comparative Pathology, 112, 41–51.
- OKITA, M., OCHIKUBO, F., KAMATA, Y., KAMATA, H., YAMANOUCHI, K. & KAI, C., 1993. Comparison of the pathogenicity of rinderpest virus in different strains of rabbits. Journal of Veterinary Medical and Science, 55, 951– 954.
- OKUNA, N. & RWEYEMAMU, M.M., 1974. Observations on the development of serum-neutralising antibodies in rinderpest infection. Bulletin of Epizootic Diseases of Africa, 22, 185–193.
- ORR, W., 1945. Observations on rinderpest in goats imported to Malaya. Journal of Comparative Pathology, 55, 185–200.
- PADMARAJ, A., SOPHILA, R., SRITHAR, A., SUGIRTHA, P.G. & GOPLAN, V., 1994. Storage of tissue culture rinderpest virus (TCRV), (Kabete ‘O’) vaccine in liquid nitrogen: Effect of titre and antibody response in calves. Indian Veterinary Journal, 71, 539–542.
- PANDEY, K.D., BARON, M.D. & BARRETT, T., 1992. Differential diagnosis of rinderpest and PPR using biotinylated cDNA probes. The Veterinary Record, 131, 199–200.
- PEASE, H.T., 1894. Rinderpest. Colonial Veterinary Department, Ledger Series No 1.
- PÉCAUD, G., 1924. Contribution à l’étude de la pathologie vétérinaire de la colonie du Tchad. Bulletin de la Société de Pathologie Exotique et de ses Filiales, 17, 196–207.
- PERCIVAL, A.B., 1918. Game and disease. Journal of the East African and Uganda Natural History Society, 13, 302–315.
- PLOWRIGHT, W., 1952. Observations on the behaviour of rinderpest virus in indigenous African sheep. British Veterinary Journal, 108, 450–457.
- PLOWRIGHT, W., 1963. The role of game animals in the epizootiology of rinderpest and malignant catarrhal fever in East Africa. Bulletin of Epizootic Diseases in Africa, 11, 149–162.
- PLOWRIGHT, W., 1963. Some properties of strains of rinderpest recently isolated in East Africa. Research in Veterinary Science, 4, 96–108.
- PLOWRIGHT, W., 1964. Studies on the pathogenesis of rinderpest in experimental cattle. II. Proliferation of the virus in different tissues following intranasal infection. Journal of Hygiene, 62, 257–281.
- PLOWRIGHT, W., 1968. Rinderpest virus. Monographs in Virology, 3, 25–110.
- PLOWRIGHT, W., 1972. The production and use of rinderpest cell culture vaccine in developing countries. World Animal Review, 1, 14–18.
- PLOWRIGHT, W., 1982. The effect of rinderpest and rinderpest control on wildlife in Africa. Symposium of the Zoological Society of London, 50, 1–28.
- PLOWRIGHT, W., 1984. The duration of immunity in cattle following inoculation of rinderpest cell culture vaccine. Journal of Hygiene, 92, 285–296.
- PLOWRIGHT, W., 1987. Investigations of rinderpest antibody in East African Wildlife, 1967–1971. Revue Scientifique et Technique de l’Office International des Épizooties, 6, 497–513.
- PLOWRIGHT, W., CRUIKSHANK, J.G. & WATERSON, A.P., 1962. The morphology of rinderpest virus. Virology, 17, 118–122.
- PLOWRIGHT, W. & FERRIS, R.D., 1957. Cytopathogenicity of rinderpest virus in tissue cultures. Nature, 179, 316.
- PLOWRIGHT, W. & FERRIS, R.D., 1959. Studies with rinderpest virus. 1. Growth and cytopathogenicity. Journal of Comparative Pathology, 69, 152–172.
- PLOWRIGHT, W. & FERRIS, R.D., 1959. Studies with rinderpest virus in tissue culture. II. Pathogenicity for cattle of culture passaged virus. Journal of Comparative Pathology, 69, 173–184.
- PLOWRIGHT, W. & FERRIS, R.D., 1962. Studies with rinderpest virus in tissue culture. The use of attenuated virus as a vaccine for cattle. Research in Veterinary Science, 3, 172–182.
- PLOWRIGHT, W. & FERRIS, R.D., 1962. Studies with rinderpest virus in tissue culture. A technique for the detection and titration of virulent virus in cattle tissues. Research in Veterinary Science, 3, 94–103.
- PLOWRIGHT, W., LAWS, R.M. & RAMPTON, C.S., 1964. Serological evidence for the susceptibility of the hippopotamus (Hippopotamus amphibius Linnaeus) to natural infection with rinderpest virus. Journal of Hygiene, Cambridge, 62, 329–336.
- PLOWRIGHT, W. & MCCULLOUGH, B., 1967. Investigations on the incidence of rinderpest virus infection in game animals of N. Tanganyika and S. Kenya 1960/1963. Journal of Hygiene, Cambridge, 65, 343–358.
- PLOWRIGHT, W., STAAK, C. & BOTTCHER, M., 1981. The immunity following rinderpest vaccination. In: Proceedings of the scientific conference,diamond jubilee of the National Veterinary Research Laboratory, 1981, Vom, Nigeria.
- PLOWRIGHT, W. & TAYLOR, W.P.,1967. Long-term studies of the immunity in East African cattle following inoculation with rinderpest culture vaccine. Research in Veterinary Science, 8, 118–128.
- POLDING, J.B. & SIMPSON, R.M., 1957. A possible immunological relationship between canine distemper and rinderpest. The Veterinary Record, 69, 582–584.
- PRABHUDAS, K. & SAMBAMURTI, B., 1976. A note on fluorescent antibody technique for rapid diagnosis of rinderpest. Indian Journal of Animal Science, 46, 454–457.
- PROVOST, A., 1970. Observations sur les muco-anticorps nasaux des bovins. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 23, 283–293.
- PROVOST, A., 1974. Peste bovine. In: bricout, f., joubert, l. & huraux, j.m., (eds). Diagnosis Sero-Immunologique des Virus Humains et Animals. Paris: Molinie.
- PROVOST, A., 1979. La pérennité de la peste bovine en Afrique intertropicale. Bulletin de l’Office International des Épizooties, 91, 761–765.
- PROVOST, A., 1981. Queries about rinderpest in African wild animals. In: karstad, l., nestel, b. & graham, m., (eds). Wildlife Disease Research and Economic Development. Ottawa: International Development Research Centre.
- PROVOST, A., 1982. Scientific and technical bases for the eradication of rinderpest in inter-tropical Africa. Revue Scientifique et Technique de l’Office International des Épizooties, 1, 589–665.
- PROVOST, A. & BORREDON, C., 1963. Les différents aspects du diagnostic clinique et expérimental de la peste bovine. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 16, 445–526.
- PROVOST, A. & BORREDON, C., 1972. Un vaccin mixte antibovipestique-antipéripneumonique lyophilise utilisable, sur le terrain, sans réfrigération. I. Sélection de virious bovipestiques à inactivation thermique retardée. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 25, 507–520.
- PROVOST, A. & BORREDON, C., 1972. Note sur l’inopportunité de l’association vaccinale peste bovine-maladie des muqueuses. Bulletin of Epizootic Diseases of Africa, 20, 265–267.
- PROVOST, A., BORREDON, C. & BOCQUET, P., 1974. Un vaccin mixte trivalent contre la peste bovine, la péripneumonie et le charbon bactéridien. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 27, 385–395.
- PROVOST, A., BORREDON, C. & QUEVAL, R., 1970. Recherches immunologiques sur la péripneumonie. XI. Un vaccin vivant mixte antibovipestique-antipéripneumonique inoculé en un seul temps. Conception—Production—Contrôles. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 23, 143–162.
- PROVOST, A. & JOUBERT, L., 1973. Modalités et techniques modernes du diagnostic expérimental de la peste bovine. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 26, 283–396.
- PROVOST, A., MAURICE, Y. & BORREDON, C., 1968. Note sur la peste bovine experimentale du dromadaire. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 21, 293–296.
- PROVOST, A., MAURICE, Y. & BORREDON, C., 1969. Comportement clinique et immunologique, lors de contamination bovipestique, de bovins vaccinés depuis plusieurs années contre la peste bovine avec des vaccins de cultures cellulaires. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 22, 453–464.
- PROVOST, A., QUEVAL, R. & BORREDON, C., 1965. Quelques recherches fondamentales sur le virus bovipestique. Revue d’Élevage et de Médécine Vétérinaire des Pays Tropicaux, 18, 371–384.
- RAMACHANDRAN, S. & SCOTT, G.R., 1985. Potency of reconstituted rinderpest vaccine. Indian Veterinary Journal, 62, 335–336.
- RAMANI, K., CHARLES, Y.S., SRINIVAS, R.P., NARAYANASWAMY, M. & RAMACHANDRAN, S., 1974. Isolation of rinderpest virus from an outbreak in domestic pigs in Karnataka. Indian Veterinary Journal, 51, 36–41.
- RAMESH BABU, N.G. & RAJASEKHAR, M., 1992. Response of sheep to primary and repeat vaccination against rinderpest. Indian Veterinary Journal, 69, 297–300.
- RAMESH BABU, N.G. & RAJASEKHAR, M., 1988. Prevalence of rinderpest antibodies in sheep and goats in southern India. The Veterinary Record, 123, 595–597.
- RATTAN,B., SHARMA, B. & BANSAL, R.P., 1993. Detection of anti-DNA antibody responses in rabbits infected with rinderpest virus by ELISA. Acta Virologica, 37, 403–406.
- RATTAN, B., SHARMA, B. & BANSAL, R.P.,1994. Circulating immune complexes in rabbits surviving rinderpest virus infection. Acta Virologica, 38, 105–110.
- RECEVEUR, R., 1957. Risques de dispersion de la peste bovine par les viandes fraîches ou congelées provenant des pays contaminés? Bulletin de l’Office International des Épizooties, 48, 148–158.
- RÉFIK-BEY, 1902. Modifications leucocytaires dans la peste bovine. Annales de l’Institut Pasteur, 16, 163–168.
- RÉFIK-BEY & RÉFIK-BEY, 1899. La peste bovine en Turquie. Épidémiologie, formes cliniques, sérothérapie. Annales de l’Institut Pasteur, 13, 596– 608.
- REID, N.R., 1949. Annual Report of the Department of Veterinary Science and Animal Husbandry. Tanganyika Territory 1947/1948. p. 3. Dar-es-Salaam: Government Printer.
- RIOCHE, M., 1969. Adaptation en microtest de la technique de séroneutralisation par la méthode cinétique pour la recherche et le titrage des anticorps neutralisant le virus de la peste bovine. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 22, 465–471.
- ROBERTS, G.A., 1921. Rinderpest (Peste bovina) in Brazil. Journal of the American Veterinary Medical Association, 13, 177–185.
- ROBERTSON, W.A.N., 1924. Rinderpest in Western Australia 1923. Melbourne: Government Printer.
- ROBEY, T.O. & HALE, M.W., 1946. Rinderpest. XI. Morphological changes in the blood of young cattle during rinderpest and after vaccination with attenuated virus vaccine. American Journal of Veterinary Research, 7, 222–227.
- ROBSON, J., ARNOLD, R.M., PLOWRIGHT, W. & SCOTT, G.R., 1959. The isolation from an eland of a strain of virus attenuated for cattle. Bulletin of Epizootic Diseases of Africa, 7, 97–102.
- ROMERO, C.H., BARRETT, T., CHAMBERLAIN, R.W., KITCHING, R.P., FLEMING, M. & BLACK, D.N., 1994. Recombinant capripoxvirus expressing the hemagglutinin protein gene of rinderpest virus: Protection of cattle against rinderpest and lumpy skin disease viruses. Virology, 204, 425–429.
- ROMERO, C.H., BARRETT, T., EVANS, S.A., KITCHING, R.P., GERSHON, P.D., BOSTOCK, C. & BLACK, D.N., 1993. Single capripox virus recombinant vaccine for the protection of cattle against rinderpest and lumpy skin disease. Vaccine, 11, 737–742.
- ROMERO, C.H., BARRETT, T., KITCHING, R.P., CARN, V.M. & BLACK., D.N., 1994. Protection of cattle against rinderpest and lumpy skin disease with a recombinant capripoxvirus expressing the fusion protein gene of rinderpest virus. The Veterinary Record, 135, 152–154.
- ROSSITER, P.B. 1985. Notes on immunoprecipitation reactions with rinderpest virus. Tropical Animal Health and Production, 17, 55–56.
- ROSSITER, P.B., HERNIMAN, K.A.J., GUMM, I.D. & MORRISON, W.I., 1993. The growth of cell culture-attenuated rinderpest virus in bovine lymphoblasts with B cell, CD4+ and CD8+ alpha/beta T cell and gamma/delta T cell phenotypes. Journal of General Virology, 74, 305–309.
- ROSSITER, P.B., HERNIMAN, K.A.J. & WAMWAYI, H.M., 1992. Improved isolation of rinderpest virus in transformed bovine lymphoblast cell lines. Research in Veterinary Science, 53, 11–18.
- ROSSITER, P.B., HUSSAIN, M., RAJA, R.H., MOGHUL, W., KHAN, Z. & BROADBENT, D.W., 1998. Cattle plague in Shangri-La: Observations on a severe outbreak of rinderpest in northern Pakistan, 1994–1995. The Veterinary Record, 143, 39–42.
- ROSSITER, P.B. & JAMES, A.D., 1989. An epidemiological model of rinderpest. II. Simulations of the behaviour of the virus in populations. Tropical Animal Health and Production, 21, 69–84.
- ROSSITER, P.B. & JESSETT, D.M., 1982. Detection of rinderpest virus antigens in vitro and vivo by direct immunofluorescence. Research in Veterinary Science, 33, 198–204.
- ROSSITER, P.B. & JESSETT, D.M., 1982. Microtitre techniques for the assay of rinderpest virus and neutralising antibody. Research in Veterinary Science, 32, 253–256.
- ROSSITER, P.B., JESSETT, D.M. & HOLMES, P., 1981. Micro-ELISA test for detecting antibodies to rinderpest virus antigens. Tropical Animal Health and Production, 13, 113–116.
- ROSSITER, P.B., JESSETT, D.M. & TAYLOR, W.P., 1982. Neutralising antibodies to rinderpest virus in sheep and goats in western Kenya. The Veterinary Record, 111, 504–505.
- ROSSITER, P.B., JESSETT, D.M., WAFULA, J.S., KARSTAD, L., CHEMA, S., TAYLOR, W.P., ROWE, L., NYANGE, J.C., OTARU, M., MUMBALA, M. & SCOTT, G.R., 1983. Re-emergence of rinderpest as a threat in East Africa since 1979. The Veterinary Record, 113, 459–461.
- ROSSITER, P.B., KARSTAD, L., JESSETT, D.M., YAMAMOTO, T., DARDIRI, A.H. & MUSHI, E.Z., 1982. Neutralising antibodies to rinderpest virus in wild animal sera collected in Kenya between 1970 and 1981. Preventive Veterinary Medicine, 1, 257–264.
- ROSSITER, P.B. & MORRISON, W.I., 2002. Unpublished observations. Kenya Agricultural Research Institute, P.O. Box 32, Kikuyu, Kenya.
- ROSSITER, P.B. & MUSHI, E.Z., 1980. Rapid detection of rinderpest virus antigens by counter-immuno-electrophoresis. Tropical Animal Health and Production, 12, 209–216.
- ROSSITER, P.B., TAYLOR, W.P., BWANGAMOI, B., NGEREZA, A.R.H., MOORHOUSE, P.D.S., HARESNAPE, J.M., WAFULA, J.S., NYANGE, J.F.C. & GUMM, I.D., 1987. Continuing presence of rinderpest virus as a threat in East Africa 1983–1985. The Veterinary Record, 120, 59–62.
- ROSSITER, P.B., WAFULA, J.S., GUMM, I.D., STAGG, D.A., MOZARIA, S.P. & SHAW, M.K., 1988. Growth of rinderpest and bovine virus diarrhoea viruses in Theileria parva infected cell lines. The Veterinary Record, 122, 491–492.
- ROSSITER, P.B. & WAMWAYI, H.W., 1989. Surveillance and monitoring programmes in the control of rinderpest: A review. Tropical Animal Health and Production, 21, 89–99.
- ROSSITER, P.B. & WARDLEY, R.C., 1985. The differential growth of virulent and avirulent strains of rinderpest virus in bovine lymphocytes and macrophages. Journal of General Virology, 66, 969–975.
- RURANGIRWA, F.R., MUSHI, E.Z., TABEL, H., TIZARD, I.R. & LOSOS, G.J., 1980. The effect of Trypanosoma congolense and T. vivax infections on the antibody response of cattle to live rinderpest virus vaccine. Research in Veterinary Science, 28, 264–266.
- RWEYEMAMU, M.M & CHENEAU, Y., 1995. Strategy for the global rinderpest eradication programme. Veterinary Microbiology, 44, 369–376.
- RWEYEMAMU, M.M., REID, H.W. & OKUNA, N., 1974. Observations on the behaviour of rinderpest virus in immune animals challenged intranasally. Bulletin of Epizootic Diseases of Africa, 22, 1–9.
- SALIKI, J.T., BERNINGER, M.L., TORRES, A., HOUSE, J.A., MEBUS, C.A. & DUBOVI, E.J., 1993. Effect of gamma irradiation on reactivity of rinderpest viral antigen with bovine immune serum in enzyme-linked immunosorbent assays and virus neutralisation and indirect fluorescent-antibody tests. Journal of Clinical Microbiology, 31, 428–432.
- SANDERSON, J.B., 1866. Nature, propagation, progress and symptoms of the disease. Third Report of the commissioners into the origin and nature etc. of the cattle plague. London: Her Majesty’s Stationery Office.
- SANDFORD, G.R., 1920. An Administrative and Political History of the Masai Reserve, East African Protectorate. London: Waterlow and Sons Ltd.
- SATO, T.A., HAYAMI, M. & YAMANOUCHI, K., 1981. Analysis of structural proteins of measles, canine distemper and rinderpest viruses.Japanese Journal of Medical Science and Biology, 34, 355–364.
- SCHEIN, H., 1917. Études sur la peste bovine. Annales de l’Institut Pasteur, 31, 571–592.
- SCOTT, G.R., 1955. The incidence of rinderpest in sheep and goats. Bulletin of Epizootic Diseases of Africa, 3, 117–119.
- SCOTT, G.R., 1957. The risk associated with the importation of meat from countries where rinderpest control measures are still required. Bulletin of Epizootic Diseases of Africa, 5, 11–13.
- SCOTT, G.R., 1959. A précis of the characteristics of rinderpest virus. Bulletin of Epizootic Diseases of Africa, 7, 173–178.
- SCOTT, G.R., 1959. Heat inactivation of rinderpest-infected bovine tissues. Nature, 184, 1948–1949.
- SCOTT, G.R., 1962. Bovine hyperimmune serum in the diagnosis of rinderpest. The Veterinary Record, 74, 409.
- SCOTT, G.R., 1962. Experimental rinderpest in Red Maasai sheep. Bulletin of Epizootic Diseases of Africa, 10, 423–426.
- SCOTT, G.R., 1963. Adverse reaction in cattle after vaccination with lapinized rinderpest virus. Journal of Hygiene, Cambridge, 61, 193–203.
- SCOTT, G.R., 1964. Rinderpest. Advances in Veterinary Science, 9, 113–224.
- SCOTT, G.R., 1964. The new pseudorinderpests. Bulletin of Epizootic Diseases of Africa, 12, 287–298.
- SCOTT, G.R., 1967. Diagnosis of Rinderpest. FAO Agricultural Studies No. 71. Rome: Food and Agriculture Organization.
- SCOTT, G.R., 1981. Recrudescence of rinderpest in Africa. Transactions of the Royal Society for Tropical Medicine and Hygiene, 75, 892.
- SCOTT, G.R., 1982. Rinderpest and peste des petits ruminants. In: gibbs, e.p.j., (ed.). Virus Diseases of Food Animals. A World Geography of Epidemiology and Control. Vol. II. Disease monographs. London: Academic Press.
- SCOTT, G.R., 1985. Rinderpest in the 1980s. Progress in Veterinary Microbiology, 1, 145–174.
- SCOTT, G.R. & BROWN, R.D., 1958. A neutralisation test for the detection of rinderpest antibodies. Journal of Comparative Pathology, 68, 303–314.
- SCOTT, G.R. & BROWN, R.D., 1961. Rinderpest diagnosis with special reference to the agar gel double diffusion test. Bulletin of Epizootic Diseases of Africa, 9, 83–100.
- SCOTT, G.R., COWAN, K.M. & ELLIOT, R.T., 1960. Rinderpest in impala. The Veterinary Record, 72, 787–788.
- SCOTT, G.R., DE TRAY, D.E. & WHITE, G., 1962. Rinderpest in pigs of European origin. American Journal of Veterinary Research, 23, 452–456.
- SCOTT, G.R. & MACDONALD, J., 1962. Kenya camels and rinderpest. Bulletin of Epizootic Diseases of Africa, 10, 495–497.
- SCOTT, G.R. & MACLEOD, A.K. & RAMPTON, C.S., 1963. Goats as donors of rinderpest hyperimmune serum. The Veterinary Record, 75, 1221–1222.
- SCOTT, G.R., TAYLOR, W.P. & ROSSITER, P.B., 1986. Manual on the Diagnosis of Rinderpest. FAO Animal Production and Health Series No 23. Rome: Food and Agriculture Organization.
- SCOTT, G.R. & WITCOMB, M.A., 1958. Rinderpest virus in laboratory animals. Report of the East African Veterinary Research Organisation, 1957. Nairobi: Government Printer.
- SEN, S.K. & SALAM, A., 1937. Experiments on the transmission of rinderpest through the agency of Stomoxys calcitrans (Linnaeus). Indian Journal of Veterinary Science and Animal Husbandry, 7, 219–224.
- SHAILA, M.S., BHASAR, A., GOPAL, T., RAMESH BABU, N.G. & SRIPAD, K., 1990. Preliminary observations on the isolation and identification of rinderpest virus from an ovine outbreak in Karnataka State. Indian Veterinary Journal, 67, 383–384.
- SHANTHIKUMAR, S.R., MALACHI, S.A. & MAJIYAGBE, K.A., 1985. Rinderpest outbreak in free-living wildlife in Nigeria. The Veterinary Record, 117, 469–470.
- SHARMA, B., BANDYOPADHAY, S.K., JOSHI, P.C. & BANSAL, R.P., 1984. Antibody response of cattle to rinderpest vaccine. Veterinary Microbiology, 10, 189–192.
- SHARMA, B., JOSHI, R.C., LAHIRI, D.K. & BANSAL, R.P., 1983. Evaluation of enzyme-linked immunosorbent assay in rinderpest. Indian Journal of Animal Science, 53, 1292–1295.
- SHARMA, B., LAHIRI, D.K., JOSHI, R.C. & BANSAL, R.P., 1986. A comparison of immunofluorescent and immunoperoxidase test for the detection of rinderpest antigen in acetone fixed smears. Indian Journal of Animal Science, 55, 1292–1295.
- SHILSTON, A.W., 1917. The vitality of the rinderpest virus outside the body under natural conditions. Memoirs of the Department of Agriculture in India, Veterinary Series, 3, 1032.
- SIMON, N., 1962. Between the Sunlight and the Thunder. The Wildlife of Kenya. London: Collins.
- SINGH, S.N., 1972. A passive haemagglutination test to detect rinderpest virus in spleen and blood. Indian Veterinary Journal, 49, 888–889.
- SINGH, K.V. & ATA, F., 1967. Experimental rinderpest in camels. A preliminary report. Bulletin of Epizootic Diseases of Africa, 15, 19–23.
- SINGH, V.P. & MURTY, D.K., 1979. An outbreak of rinderpest in wild spotted deer (Axis axis). Indian Veterinary Journal, 56, 988–990.
- SRIPAD, K., RAMESH BABU, N.G., BABU, M.M., GOPAL, T. & KESHAVAMURTHY, B.S., 1995. Experimental transmission with ovine rinderpest virus. Small Ruminant Research, 16, 277–282.
- STEVENSON-HAMILTON, J., 1911. The relation between game and tsetse flies. Bulletin of Entomological Research, 2, 113.
- STEVENSON, P., ROSSITER, P.B., MUNGA, L., NDUNG’U, E.K. & DOLAN, R.B., 1999. Rinderpest vaccination and the incidence and development of trypanosomiasis in cattle. Tropical Animal Health and Production, 31, 65–73.
- STEWART, D.R.M., 1968. Rinderpest among wild animals in Kenya, 1963–66. Bulletin of Epizootic Diseases of Africa, 16, 139–140.
- STIRLING, R.F., 1932. Some experiments in rinderpest vaccination: Active immunization of Indian plains cattle with goat-adapted virus alone in field conditions. Veterinary Journal, 88, 192–204.
- STONE, S.S. & DE LAY, P.D., 1961. The inactivation of rinderpest virus by b-propriolactone and its effect on homologous complement-fixing and neutralising antibodies. Journal of Immunology, 87, 464–467.
- SUGIMOTO, M. & YAMANOUCHI, K., 1994. Characteristics of an attenuated vaccinia virus strain, LC16m0, and its recombinant virus vaccines. Vaccine, 12, 675–681.
- TAJIMA, M. & USHIJIMA, T., 1970. The pathogenesis of rinderpest in the lymph nodes of cattle. American Journal of Pathology, 62, 221–235.
- TAJIMA, M., USHIJIMA, T., KISHI, S. & NAKAMURA, J., 1967. Electron microscopy of cytoplasmic inclusion bodies in cells infected with rinderpest virus. Virology, 31, 92–100.
- TAKEMATSU, M. & MORIMOTO, T., 1954. Studies on tissue culture with rinderpest virus. Japanese Journal of Veterinary Service, 16, 185–193.
- TAYLOR, W.P., 1968. The susceptibility of the one-humped camel (Camelus dromedarius) to infection with rinderpest virus. Bulletin of Epizootic Diseases of Africa, 16, 405–410.
- TAYLOR, W.P., 1986. Epidemiology and control of rinderpest. Revue Scientifique et Technique de l’Office International des Épizooties, 5, 407–410.
- TAYLOR, W.P., BHAT, P.N. & NANDA, Y.P., 1995. The principles and practise of rinderpest eradication. Veterinary Microbiology, 44, 359–367.
- TAYLOR, W.P. & PERRY, C.T., 1970. A plaque assay system for rinderpest virus and its use in characterising virus adsorption. Archives of Virology, 32, 269–282./li>
- TAYLOR, W.P. & PLOWRIGHT, W., 1965. Studies on the pathogenesis of rinderpest in experimental cattle. III. Proliferation of an attenuated strain in various tissues following sub-cutaneous inoculation. Journal of Hygiene, Cambridge, 63, 263–275.
- TAYLOR, W.P., PLOWRIGHT, W., PILLINGER, R., RAMPTON, C.S. & STAPLE, R.F., 1965. Studies on the pathogenesis of rinderpest in experimental cattle. IV. Proliferation of the virus following contact infection. Journal of Hygiene, Cambridge, 63, 497–506.
- TAYLOR, W.P. & WATSON, R.M., 1967. Studies of the epizootiology of rinderpest in blue wildebeest and other game species of Northern Tanzania and Southern Kenya, 1965–1967. Journal of Hygiene, Cambridge, 65, 537–545.
- TEUSHER, E., RAMACHANDRAN, S. & HARDING, H.P., 1981. Observations on the pathology of Jembrana disease in Bali cattle. Zentralblatt für Veterinärmedizin, 28A, 608–622.
- THEILER, A., 1909. Notes on stock disease of German and British East Africa and Uganda, and the resolutions of the International Veterinary Congress at the Hague, Holland, 1909. Transvaal Agricultural Journal, 8, 183–197.
- THIÉRY, G., 1956. Influence du type de virus et de l’espèce affectée sur les lésions de la peste bovine. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 9, 109–115.
- THIÉRY, G., 1956. Hématologie, histopathologie et histochimie de la peste bovine. Intérêt de l’étude histochimique des inclusions cellulaires de la peste bovine pour la signification générale des inclusions dans les maladies à virus. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 9, 117–140.
- THOMAS, A.D. & REID, N.R., 1944. Rinderpest in game: A description of an outbreak and an attempt at limiting its spread by means of a bush fence. Onderstepoort Journal of Veterinary Science and Animal Industry, 20, 7–23.
- TODD, C. & WHITE, R.G., 1914. Experiments on Cattle Plague. Cairo: Government Press.
- TSUKIYAMA, K., SUGIYAMA, M., YOSHIKAWA, Y. & YAMANOUCHI, K., 1987. Molecular cloning and sequence analysis of the rinderpest mRNA encoding the haemagglutinin protein. Virology, 163, 48–54.
- TSUKIYAMA, K., YOSHIKAWA, Y. & YAMANOUCHI, K., 1988. Fusion glycoprotein (F) of rinderpest virus: Entire nucleotide sequence of the F mRNA, and several features of the F protein. Virology, 164, 523–530.
- TURNER, G., 1906. Rinderpest: Its prevention and cure. Report of the British Association, 1905, 552.
- VARNELL, G. & PRITCHARD, W., 1866. On the treatment of the cattle plague. Third report of the commissioners into the origin and nature etc. of the cattle plague. London: Her Majesty’s Stationery Office.
- VITTOZ, R., 1963. Report of the Director of the Scientific and Technical Activities of the Office International des Épizooties, May 1962–May 1963. Paris: O.I.E.
- VIZOZO, A.D. & THOMAS, W.E., 1981. Paramyxovirus of the morbilli group in the wild hedgehog Erinaceus europeus. British Journal of Experimental Pathology, 62, 79–86.
- VOGEL, S.W. & HEYNE, H., 1996. Rinderpest in South Africa—100 years ago. Journal of the South African Veterinary Association, 67, 164–170.
- VON OSTERTAG, R., 1916. Ueber Rinderpest. Ein Beitrag zum Stande und der Bekamfung der Tierseuchen in Deutsch-Ostafrika. Zeitschrift für Infektionskrankheiten, Parasitenkund Krankheiten und Hygiene der Haustiere, 18, 1–48.
- WAFULA, J.S., MIRANGI, P.K., IRERI, R.G. & MBUGUA, N., 1986. Development and stability of rinderpest virus antigens in cattle tears and lymph nodes. Tropical Animal Health and Production, 18, 26–30.
- WAFULA, J.S., ROSSITER, P.B., WAMWAYI, H.M. & SCOTT, G.R., 1989. Preliminary observations on rinderpest in pregnant cattle. The Veterinary Record, 124, 485–486.
- WAFULA, J.S. & WAMWAYI, H.M., 1989. Some factors which could cause rinderpest vaccination failure in cattle. Bulletin of Animal Health and Production in Africa, 37, 251–254.
- WAGNER, G.G., JESSETT, D.M., BROWN, C.G.D. & RADLEY, D.E., 1975. Diminished antibody response to rinderpest vaccination in cattle undergoing experimental East Coast fever. Research in Veterinary Science, 19, 209–211.
- WALKER, J., 1922. In: Bulletin Number 4. Department of Agriculture, Division of Veterinary Research, Kenya.
- WALKER, R.V.L., BAKER, J.A. & JENKINS, D.L., 1946. Rinderpest. II. Certain immunity reactions. American Journal of Veterinary Research, 7,
- WALSH, J., 1987. War on cattle disease divides the troops. Science, 237, 1289–1291
- WALSH, E.P., BARON, M.D., ANDERSON, J. & BARRETT, T., 2000. Development of a genetically marked recombinant rinderpest vaccine expressing green fluorescent protein. Journal of General Virology, 81, 709–718.
- WALSH, E.P., BARON, M.D., RENNIE, L.F., MONAGHAN, P., ANDERSON, J. & BARRETT, T., 2000. Recombinant rinderpest vaccines expressing membrane-anchored proteins as genetic markers: Evidence of exclusion of marker protein from the virus envelope. Journal of Virology, 74, 10165–10175.
- WAMBURA, P.N., MOSHY, D.W., MBISE, A.N., MOLLEL, G.O., TAYLOR, W.P., ANDERSON, J. & BRUNING, A., 2000. Diagnosis of rinderpest in Tanzania by a rapid chromatographic strip-test. Tropical Animal Health and Production, 32, 141–145.
- WAMWAYI, H.M., 1989. Efficacy of some chemicals and disinfectants against rinderpest virus. Bulletin of Animal Health and Production in Africa, 37, 255–260.
- WAMWAYI, H.M., FLEMING, M. & BARRETT, T., 1995. Characterisation of African isolates of rinderpest virus. Veterinary Microbiology, 44, 151–163.
- WAMWAYI, H.M., KARIUKI, D.P., WAFULA, J.S., ROSSITER, P.B., MBUTHIA, P.G. & MACHARIA, S.R., 1992. Observations on rinderpest in Kenya, 1986–1989. Veterinary Pathology, 30, 544–554. Revue Scientifique et Technique de l’Office International des Epizooties, 11, 769–784.
- WAMWAYI, H.M., WAFULA, J.S. & ROSSITER, P.B., 1995. Comparison between virus neutralisation tests and an indirect enzyme linked immunosorbent assay for detecting antibodies to rinderpest virus in cattle sera. Bulletin of Animal Health and Production in Africa, 43, 45–52.
- WAMWAYI, H.M. & WAFULA, J.S., 1987. Microtitre technique for detection and titration of rinderpest virus in infected bovine tissue and secretions. Bulletin of Animal Health and Production in Africa, 35, 318–320.
- WARREN, J.L., 1960. The relationships of the viruses of measles, canine distemper and rinderpest. Advances in Virus Research, 7, 27–60.
- WATANABE, M., 1970. Rinderpest: Diagnosis and prophylaxis. National Institute of Animal Health Quarterly, 10, 29–43.
- WATERSON, A.P., ROTT, R. & RUCKLE-ENDERS, G., 1963. The components of measles virus and their relation to rinderpest and distemper. Zentralblatt für Naturforschung, 18b, 377–384.
- WESTON, E.A., 1924. Rinderpest in Australia. Journal of the American Veterinary Medical Association, 19, 337–350.
- WHITE, R.G., 1958. A specific diffusible antigen of rinderpest virus demonstrated by the agar double-diffusion precipitation reactions. Nature, 181, 1409.
- WHITE, R.G., SIMPSON, R.M. & SCOTT, G.R., 1961. An antigenic relationship between the viruses of bovine rinderpest and canine distemper. Immunology, 4, 203–205.
- WILDE, J.K.H., 1948. Rinderpest in some African wild mammals. Journal of Comparative Pathology, 58, 64–72.
- WILDE, J.K.H. & SCOTT, G.R., 1961. Rinderpest interference with caprinized rinderpest virus. Journal of Comparative Pathology, 71, 222–227.
- WOHLSEIN, P., TRAUTWEIN, G., HARDER, T.C., LIESS, B. & BARRETT, T., 1993. Viral antigen distribution in organs of cattle experimentally infected with rinderpest virus. Veterinary Pathology, 30, 544–554.
- WOHLSEIN, P. WAMWAYI, H.M., TRAUTWEIN, G., POHLENZ,J. LIESS, B. & BARRETT, T., 1995. Pathomorphological and immunohistological findings in cattle experimentally infected with rinderpest virus isolates of different pathogenicity. Veterinary Microbiology, 44, 141–147.
- YAMANAKA, M., DALE, B., CRISP, T., CORDELL, B., GRUBMAN, M. & YILMA, T., 1992. Sequence analysis and editing of the phophoprotein (P) gene of rinderpest virus. Virology, 190, 1255–1261.
- YAMANAKA, M., HSU, D., CRISP, T., DALE, B., GRUBMAN, M. & YILMA, T., 1988. Cloning and sequence analysis of the hemagglutinin gene of the virulent strain of rinderpest virus. Virology, 166, 251–253.
- YAMANOUCHI, K., 1980. Comparative aspects of pathogenicity of measles, canine distemper and rinderpest virus. Japanese Journal of Medical Science and Biology, 33, 41–66.
- YAMANOUCHI, K. & BARRETT, T., 1994. Progress in the development of a heat-stable recombinant rinderpest vaccine using an attenuated vaccinia virus vector. Revue Scientifique et Technique de l’Office International des Epizooties, 13, 721–735.
- YAMANOUCHI, K., CHINO, F., KOBUNE, F., FUKUDA, A. & YOSHIKAWA, Y., 1974. Pathogenesis of rinderpest infection in rabbits. I. Clinical signs, immune response, histological changes and virus growth patterns. Infection and Immunity, 9, 199–205.
- YAMANOUCHI, K., FUKUDA, A., KOBUNE, F., YOSHIKAWA, Y. & CHINO, F., 1974. Pathogenesis of rinderpest virus infection in rabbits. II. Effect of rinderpest virus on the immune function of rabbits. Infection and Immunity, 9, 201–211.
- YAMANOUCHI, K., INUI, K., SUGIMOTO, M., ASANO, K., NISHIMAKI, F., KITCHING, R.P., TAKAMATSU, H. & BARRETT, T., 1993. Immunisation of cattle with a recombinant vaccinia vector expressing the haemagglutinin gene of rinderpest virus. The Veterinary Record, 132, 152–156.
- YILMA, T., HSU, D., JONES, L., OWENS, S., GRUBMAN, M., MEBUS, C., YAMANAKA, M. & DALE, B., 1988. Protection of cattle against rinderpest with vaccinia virus recombinants expressing the HA or F gene. Science, 242, 1058–1060.
- YOUSSEF, H.M., SAMI, A.M., EL-SANOUSI, A.A., SABER, M.S. & REDA, I.M., 1991. A localised outbreak of rinderpest in Egyptian buffaloes in 1990. Veterinary Medical Journal, Giza, 39, 57–69.
- ZEIN, A. & SROUR, E., 1985. Production of interferon and of plaque enhancing factor by rinderpest-virus. Zentralblatt für Veterinärmedizin, B, 32, 466–471.
- ZONCHELLO, A., 1917. II gultian nel Sahel. Clinica Veterinaria, 30, 113–132.
- ZWART, D. & MACADAM, I., 1966. Resistance of two bulls against rinderpest without detectable neutralizing antibodies in their sera. Bulletin of Epizootic Diseases of Africa, 14, 53–54.
- ZWART, D. & MACADAM, I., 1967. Transmission of rinderpest by contact from cattle to sheep and goats. Research in Veterinary Science, 8, 37–47.
- ZWART, D. & MACADAM, I., 1967. Observations on rinderpest in sheep and goats and transmission to cattle. Research in Veterinary Science, 8, 53–57.
- ZWART, D. & ROWE, L.W., 1966. The occurrence of rinderpest antibodies in the sera of sheep and goats in northern Nigeria. Research in Veterinary Science, 7, 504–511.