- Infectious Diseases of Livestock
- Part 2
- Equine coital exanthema
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Equine coital exanthema
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Equine coital exanthema
Synonyms: Genital horse pox, eruptive venereal disease, equine venereal vulvitis or balanitis, and coital vesicular exanthema
Previous authors: G P ALLEN and N W UMPHENOUR
Current authors:
M, CAROSSINO, DVM, PhD, Dipl. ACVM (Virology), Dipl. ACVP, Assistant Professor of Veterinary Pathology, Department of Pathobiological Sciences & Louisiana Animal Disease Diagnostic Laboratory (LADDL), School of Veterinary Medicine, Louisiana State University, Louisiana State University, United States
M A VISSANI, DVM, PhD, Director, Laboratory of Equine Virology, National Institute of Agricultural Technology (INTA), Buenos Aires, Argentina; Principal Investigator, National Council for Scientific and Technical Research (CONICET), Buenos Aires, Argentina; Assistant Professor of Infectious Diseases, School of Veterinary Medicine, Universidad del Salvador, Buenos Aires, Argentina.
M E BARRANDEGUY, DVM, PhD, Director, Research Institute, School of Veterinary Medicine, Universidad del Salvador, Buenos Aires, Argentina; Professor of Infectious Diseases, School of Veterinary Medicine, Universidad del Salvador, Buenos Aires, Argentina; Associate Principal Investigator, National Institute of Agricultural Technology (INTA), Buenos Aires, Argentina
U B R BALASURIYA, - BVSc, MS, PhD, FSLCVS, Director and Professor of Virology, Louisiana Animal Disease Diagnostic Laboratory (LADDL), and Department of Pathobiological Sciences, School of Veterinary Medicine, Louisiana State University, River Road, Room 1043, Baton Rouge, Louisiana, 70803, United States of America
Introduction
Equine coital exanthema (ECE) is an infectious and highly contagious, venereally-transmitted mucocutaneous disease, characterized by the formation of papules, vesicles, pustules and ulcers on the skin of external genital organs of mares and stallions, caused by Equid alphaherpesvirus 3 (EHV-3). 4, 11, 55
First reported in 1920 during an outbreak of the disease in one Thoroughbred stallion and six mares in Ireland,21 EHV-3 was then concurrently isolated during the 1960s in the United States, Canada and Australia.18, 30, 40 Since then, reports of ECE have been recorded almost worldwide under a variety of names such as genital horse pox, eruptive venereal disease, equine venereal vulvitis or balanitis, or coital vesicular exanthema.10, 12, 20, 27-29, 31, 41, 43, 51, 52, 58, 61, 62 The latest report was the confirmation, for the first time, of ECE in the native Icelandic horse population.50
The infection is relatively benign and does not result in systemic illness, infertility or abortion.39, 52 However, the negative impact on the equine industry, mainly in the Thoroughbred breed, relies on the forced, temporary withdrawal of affected animals with the consequent disruption of mating activities in breeding facilities. For affected stallions, such disruptions may translate into significant end-of-season decreases in the mare-book size. Similarly, affected mares will miss breeding opportunities. In artificial insemination and embryo transfer centres, affected mares may be reluctant to be inspected, inseminated or transferred with the consequent loss of opportunity to become pregnant. In both scenarios (natural mating or artificial insemination), delayed foaling dates or reduced pregnancy rates may occur in those mares that miss breeding opportunities because of the disease.4, 10, 55, 58
Aetiology
The aetiological agent of ECE, Equid alphaherpesvirus 3 (EHV-3), is a member of the Herpesviridae family (see Herpesviridae: General Introduction) with a typical alphaherpesviral architecture, size, and genome structure.4, 24, 32 Its biological features place EHV-3 in the Alphaherpesvirinae subfamily, within the genus Varicellovirus, which also includes Equid alphaherpesvirus 1 (EHV-1), Equid alphaherpesvirus 4 (EHV-4), Equid alphaherpesvirus 8 (EHV-8) and Equid alphaherpesvirus 9 (EHV-9). EHV-3 is antigenically, genetically, and pathogenically distinct from EHV-1 and EHV-4, and even more distantly related to equid gammaherpesviruses (EHV-2 and EHV-5).4, 24 It shares no protective or neutralization epitopes and only minor genetic homology with these other herpesviruses of the domestic horse (Equus caballus).4, 13, 15 The restriction endonuclease cleavage patterns of the DNA of EHV-3 are unique when compared with those reported for the genomic DNA from EHV-1, EHV-2, EHV-4, and EHV-5.4 Although causing disease of clinical similarity, the virus of ECE is unrelated, by serum neutralization tests, to Bovine alphaherpesvirus 1 (BHV-1) that causes infectious pustular vulvovaginitis and balanoposthitis in cattle.17 (see Chapter Infectious bovine rhinotracheitis/Infectious pustular vulvovaginitis and infectious pustular balanoposthitis)
As for physicochemical properties, the buoyant density of EHV-3 has been determined as 1.727 g/cm3, with a sedimentation coefficient of approximately 55.4 S, which corresponds to a molecular weight value of 90 to 100 megadaltons (Md).7 Regarding the structure of the EHV-3 genome, it has a double-stranded DNA class D genome consisting of a long and a short unique region (UL and US), both flanked by inverted repeats (TRL/IRL and IRS/TRS).7, 44 Its full-length genome sequence consists of 151,601 bp (G + C content of 68.1 per cent) encoding for 76 open reading frames (ORF), four of which are duplicated (ORFs 64, 65, 66, and 76), resulting in a total of 80 protein products. 7, 44 Based on the nucleotide sequence, EHV-3 is the most divergent of the equine alphaherpesviruses, with overall nucleotide identities with the other equine alphaherpesviruses ranging from 62.1 to 64.9 per cent, whereas EHV-1, EHV-4, EHV-8, and EHV-9 have identities of at least 78.2 per cent. 7, 44 As for EHV-6, its counterpart in donkeys (also referred to as asinine herpesvirus 1; AsHV-1), the overall nucleotide sequence identity with EHV-3 glycoproteins (g) B, C, and D is 87.02 per cent.16, 36
Purified virions of EHV-3 possess, in addition to the DNA-containing nucleocapsid, a proteinaceous tegument and glycoprotein-laden envelope.3 As for other herpesviruses, EHV-3, is environmentally labile, and infectivity is quickly destroyed by lipid solvents, detergents, heat, and drying as well as by the common disinfectants available for veterinary use.26, 42 Storage at temperatures below −60 °C is required for maintaining its long-term viability.4
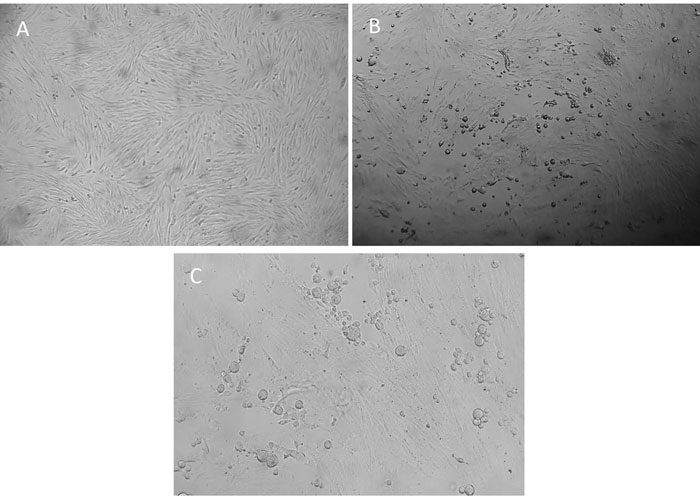
Equid alphaherpesvirus 3is highly host specific. It replicates only in cell lines derived from equids and, to date, experimental in vivo studies have solely been done in the natural host.4, 14, 38, 55, 60 In vitro, EHV-3 progeny DNA and virions are detected by two and six hours post-infection (PI), respectively. While it has been previously noted that cytopathic effect (CPE) is detectable as early as six hours PI after a high multiplicity of infection,4 in the authors’ experience, CPE from clinical isolates is normally visible >24 hours PI, with a few instances in which the clinical specimen may contain a high viral titre and CPE is visible as early as 12-24 hours PI. The CPE of EHV-3 is typically herpetic in nature with rapidly enlarging foci of rounded, refractile cells (Figure 1). When infected cell cultures are stained with haematoxylin and eosin, eosinophilic, intranuclear inclusion bodies are visible.4 Although it has been previously reported that 34 °C is the optimal temperature for in vitro replication of EHV-3,4 it has been extensively described that 37°C is a permissive temperature to isolate EHV-3 in E. Derm (NBL-6) cells (ATCC CCL-57) and equine explants.11, 38, 56, 60 Although restriction endonuclease fingerprint analysis of viral DNA has been used to demonstrate the genetic individuality of EHV-3 isolates, there is no reported evidence of antigenic diversity among clinical isolates recovered from different disease outbreaks.4 In Argentinean field isolates, restriction endonuclease fingerprint analysis showed that Argentinean strains differ from the reference strain by HindIII and EcoRI patterns.9
Regarding antigenic variants, there is not detailed analysis of the antigenic and immunogenic proteins/glycoproteins of EHV-3, but a genetic analysis of the partial sequence of the gG gene from 25 field isolates demonstrated that there are at least four genetically distinguishable strains in circulation. These strains, named CAT, AAT, CAG and ACT are a consequence of three base substitutions at positions 904, 1103 and 1264 of the gG gene. The mutation at 904 position is a silent mutation, while those at 1103 and 1264 produce changes in the amino acid sequence: aspartic acid to alanine in the amino acid position 368, and serine to alanine in the 422 of the gG protein, respectively.10 Neither the restriction endonuclease pattern nor the mutations of the partial gG gene could be associated with any particular clinical manifestation and their biological implications are still undetermined.
The cloning of EHV-3 in a bacterial artificial chromosome (BAC) has been reported and the generation of mutant EHV-3 viruses devoid of gE or gG has demonstrated that these glycoproteins are dispensable for virus growth in vitro.2, 54 Small-plaque variants of EHV-3 arise during passage in cell culture and are characterized by alterations in their DNA restriction endonuclease patterns caused by the presence of a 5,7 kbp nucleic acid insert in the unique (U) sequence of the small (S) component of their DNA genomes.4
Epidemiology
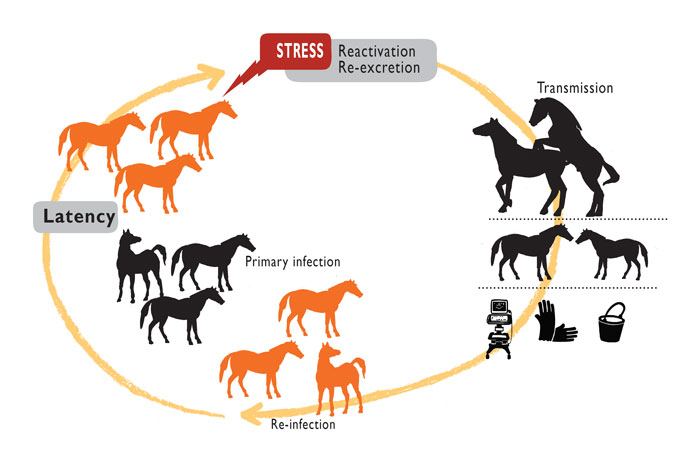
Equid alphaherpesvirus 3 is endemic in most equine breeding populations in the world.4, 6, 13, 27, 32, 33, 41, 43, 50, 52, 61 The only known biological reservoir of EHV-3 is the latently infected horse and it is well-known that, as for other herpesviral infections, periods of reactivation from latency, with production and shedding of infectious virus are the source of infection for other comingling susceptible animals.4, 11, 55 (Figure 2) Reactivation from latency was demonstrated following experimental administration of glucocorticoids in two seropositive mares;15 and subsequently natural reactivation in both mares, during an isolation period of 11 months, has been reported.13 Episodes of reactivation and shedding, either with or without clinical manifestation, and together with variable levels of serum antibodies, have been observed. Epidemiological data suggest that the original viral source of an outbreak of ECE may be either a visiting mare brought onto the stud farm for breeding or virus reactivated from a member of the resident stallion or mare population.4, 13, 58 A study carried out in a breeding farm in Argentina showed that 48 per cent of the mares were seropositive, thus potentially latently infected, and 6 per cent were shedding the virus without clinical signs.13 Although the anatomical site that harbours the latent herpesvirus is still unknown, it has been inferred, based on comparison with Human herpesvirus 2 and Caprine alphaherpesvirus 1 (CpHV-1), to be in the sciatic and/or sacral ganglion cells.11 (Figure 3).
Surveys to estimate the seroprevalence of EHV-3 infection in several horse populations have demonstrated the existence of EHV-3 specific antibodies in 10 to 53 per cent of horses at breeding age.4, 6, 13 In contrast to these expected results for breeding stocks, a study conducted in Japan reported a lower prevalence of EHV-3 antibodies in sexually mature horses (4.1 per cent).33 Maternally-derived antibodies to EHV-3 are present in new-born foals but they decay to undetectable levels by four months of age. The seroprevalence is low in young as well as in unmated horses, but increases with age in breeding animals.4 As the infection is benign and the disease is self-limiting, ECE is sporadically reported and thus leads to underestimation of its true incidence. Once recrudescence of EHV-3 infection has occurred, ECE is highly contagious; post-coital infection rates as high as 100 per cent have been reported.55
The virus is transmitted mainly through direct contact during sexual intercourse4, 32; nevertheless, non-coital transmission has been described in several instances, associated with the genito–nasal contact by behavioural nuzzling/sniffing8, 21, 22, 34 (Figure 2). The incubation period is five to nine days.4 However, during experimental infection, the incubation period could be as short as one to three days.14, 56 The potential for virus spread during artificial insemination has been suggested.25, 37 However, there is no information regarding the susceptibility of the cervical and endometrial epithelium to EHV-3 infection. Iatrogenic transmission of infection during reproductive manoeuvres can occur by virus-contaminated objects such as buckets and elements used for hygiene of the genital area of mares and stallions, ultrasound scanners and examination sleeves used for rectal palpation or gynaecological examination and/or surgical instruments in the post-partum management.4, 11, 58 (Figure 2). The use of a contaminated endoscope was involved in an outbreak of an atypical presentation of EHV-3 infection resulting in unilateral rhinitis in training Thoroughbred horses.12 The likelihood of mechanical transmission by stable flies and urine from infected animals has also been suggested.25, 29

Figure 3 Pathogenesis of Equid alphaherpesvirus 3 (EHV-3):(1) EHV-3 infects the stratified epithelium of epidermal surfaces at muco–cutaneous margins and skin of external genital organs;(2) the virus replicates and laterally spreads;(3) lytic replication occurs and the virus is shed; (4) destruction of epithelial cells elicits a vigorous, localized inflammatory response; (5) the virus does not breach the basement membrane, and thus systemic dissemination is limited; (6) after active infection the virus induces latency; the anatomic site has not been demonstrated yet but it has been inferred to be in the sciatic and/or sacral ganglion cells3 from which reactivation can occur (dashed arrow).
Pathogenesis
The virus is easily transmitted by simple skin contact; the epidermal surface does not need to be damaged for infection to be established.4 After initial infection, EHV-3 replicates in the stratified epithelium of the epidermis present at muco–cutaneous margins and skin of external genital organs of mares and stallions (Figure 3). Viral invasion of the basement membrane or infection of immune cells to infect the lamina propria does not occur.4, 38 Thus, systemic dissemination of EHV-3 is limited, but it is still unknown which factors are involved in this localized replication site. This non-invasive behaviour of EHV-3 is comparable with that of EHV-4, but is in stark contrast to the pathogenesis of EHV-1.38, 53
Destruction of epithelium by the lytic virus infection elicits a vigorous, localized inflammatory response that gives rise to the formation of the characteristic cutaneous lesions of ECE.1–3 Anorectal lymphadenopathy has been suggested as an additional complication associated with EHV-3 infection.10 However, it could not yet be elucidated if the enlargement of the anorectal lymph nodes is due to EHV-3 infection itself, to the outcome of the inflammatory process in the region, or to a secondary bacterial infection.
Secondary bacterial infection with Streptococcus equi subsp. zooepidemicus is common and influences the character, severity, and duration of the epithelial lesions. Recovery from ECE is complete in a matter of two to three weeks, while depigmentation can persist longer.4 The immunity induced by EHV-3 infection has not been studied in detail to date. Serological responses of the horse to EHV-3 infection comprise the production of serum complement-fixing and virus-neutralizing antibodies that reach maximal levels 14 to 21 days after infection.4 Similarly to other equine alphaherpesviruses, antibody responses against EHV-3 tend to reach similar titres to those against EHV-1.11, 56 Complement fixation titres decline rapidly and are usually not detectable 60 days after infection, whereas serum-neutralizing antibodies decline more slowly, with detectable levels persisting for a year or more.4, 14, 15 There is no available information regarding mucosal immunity, but as reactivation and shedding occur even in the presence of higher serum-neutralizing antibody titres,13, 56 it would be reasonable to hypothesize that mucosal immunity plays an important role in the control of infection and shedding after reactivation, and also in the severity of clinical lesions.
Reactivation from latency can be associated with low (or decreasing) neutralizing antibody titres, as seen for CpHV-1, another genital alphaherpesvirus.47 Nevertheless, EHV-3 spontaneous reactivation and shedding in the presence of varying levels of neutralizing antibodies was detected in mares kept in isolation13, 56 and also observed in two natural outbreaks of ECE.54
Clinical signs and Pathology
After the incubation period of approximately 1-3 days, small (1–2 mm), raised and reddened papules, which can often go unnoticed, appear on the penis and prepuce of stallions, and on the vagina, perineum and, occasionally, on the skin of the ventral surface of the tail, buttocks, medial aspect of the thighs, or on teats of mares. Lesions commonly extend beyond the mucodermal junction of the vulva and may be present on its inner, non-pigmented mucosal surface as well as on the clitoris, fossa clitoridis, or the walls of the vaginal vestibule in mares and on the urethral orifice of stallions.4 (Figures 4 and 5). Sequentially, lesions progress to vesicles and pustules, and after epidermal sloughing of the necrotic dome of the pustule, a shallow, raw or encrusted erosion or ulcer appears.4, 32, 55 Small “cracks” in the rectal sphincter were also reported as the most common clinical sign in Thoroughbred mares during an extensive outbreak of ECE.58 Localized inflammation, erythema, congestion and oedema of the genital area are commonly observed, along with vulvar discharge, tail swithing, frequent urination or arching of the back.4, 11, 32 Together with the description of lesions around the anus, lymphadenopathy, constipation, tenesmus and evacuation of firm, dry, mucus-covered faeces have been also reported during a severe outbreak of ECE at an artificial insemination centre.10 In the stallion, oedema of the prepuce and sheath may occur and extend laterally onto the ventral abdominal wall and scrotum4 It is noteworthy that stallions with severe lesions can exhibit discomfort, loss of libido and refusal to copulate.4, 11, 46, 51, 55 General signs of infection, such as fever, anorexia or dullness, are rare, but if present they are more severe in stallions than in mares.4, 11, 55, 56
Both severity and duration of the disease varys considerably among individual horses and the disease progression can be also be influenced by secondary bacterial infections (Streptococcus equi subsp. zooepidemicus being the most common) and/or external myiasis.4, 10, 32, 56 (Figure 4). Lesions infected secondarily with bacteria may ooze a white discharge. The lesions may be few or many in number and may be at different clinical stages.4 Uncomplicated cases typically resolve within 10 to 14 days, while cutaneous scars and depigmentation can persist longer.1, 11, 55
The in vivo replication of EHV-3 in the horse is restricted to anatomical sites covered by true cutaneous (stratified squamous) epithelium or by the transitional epithelium at the mucocutaneous junctions of genital and respiratory tissues.4 However, when ex vivo replication in nasal and vaginal mucosa is performed, EHV-3-induced plaques are significantly higher in the vaginal mucosa than in the nasal mucosae. This suggests a replicative advantage in genital mucosa that may explain viral tropism.38 Because of this anatomical tropism, the lesions of ECE are most commonly located on the external (cutaneous) surface of the vulva and surrounding perineal skin in the mare, and, in the stallion, on the glans and free body of the penis as well as on both the parietal and visceral folds of the prepuce. Occasionally, extragenital epidermal sites on the muzzle, lips, or nostrils can be observed.4, 56
Unlike coital exanthema of cattle (infectious pustular vulvovaginitis/balanoposthitis) caused by BHV-1, lesions within the cranial vaginal canal of the mare are not a prominent feature of EHV-3 infection.4 The disease is not known to cause any long-term impairment of fertility in either mares or stallions.4, 39, 52 Abortions caused by natural EHV-3 infection of the foetus have not been reported.4, 55 Experimental inoculation of EHV-3 into young horses by the intranasal route resulted in ulcerations of the nasal mucosa with accompanying pyrexia and nasal discharge.
Horses with chronically recurrent ECE in which typical lesions occur annually during the terminal period of pregnancy or after foaling have been observed, most commonly in aged broodmares.4 In two outbreaks in Argentina, mares in which lesions were first detected were those that had recently delivered and/or been subjected to postpartum management procedures (rectal palpation and suturing).58
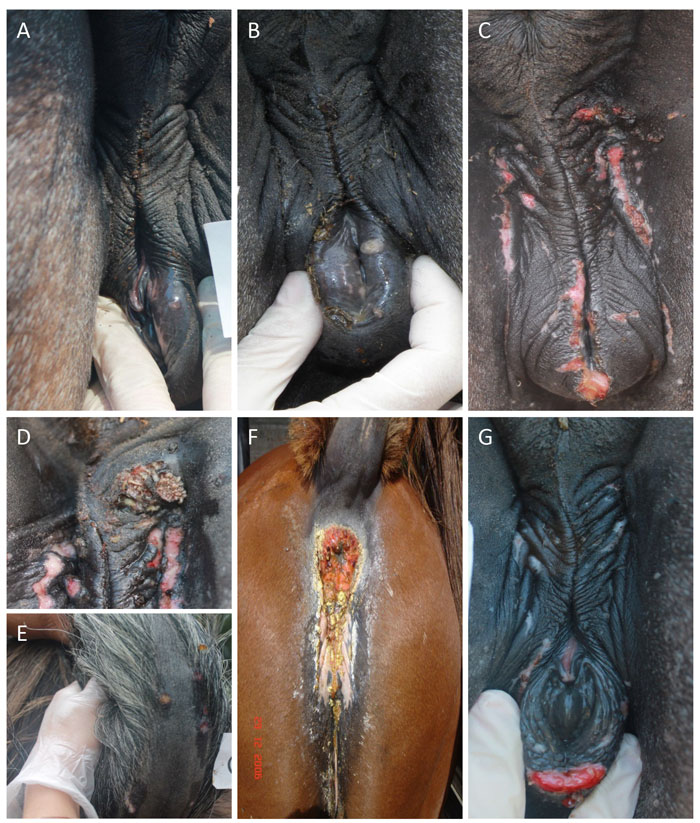
Figure 4 Lesions of equine coital exanthema (ECE) observed on the vulva of experimentally infected mares; (a): vesicle; (b): pustule; (c, f, g): erosions and ulcers; (f): secondary bacterial infection (d): myiasis; (e): lesions out of the inoculation area. , M. A. Vissani, C. O. Perglione, O. Zabal, G. Alvarez, E. Thiry, M. Barrandeguy, V. Parreno, 2020. Reduces viral excretion in mares with equine coital exanthema, Topical Ganciclovir, 94, 1-9.56 Copyright Elsevier (2020). Reproduced with permission.
So far, no studies with detailed histopathological examination of ECE lesions through the course of their development have been conducted. Necrotic epithelium covering the erosions or ulcers is sloughed and replaced by a fibrin exudate in which leukocytes are trapped. The underlying dermis is oedematous and heavily infiltrated with inflammatory cells. The transition from necrotic tissue to normal epithelium at the edges of the erosions/ulcers is sharply defined. Typical herpesvirus intranuclear inclusion bodies are usually present in some epithelial cells at the periphery of the erosions.4
Diagnosis
The genital lesions of ECE in both mares and stallions are characteristic, and a clinical diagnosis can therefore be made during the clinical inspection. Laboratory diagnosis is needed for confirmation and it is essential for subclinical infections.4, 32, 55 Perineal and genital swabs are the appropriate clinical samples for diagnosis and should be collected by firm swabbing or scraping of the edges of fresh, active lesions and maintained in 5–6 ml of viral transport medium (containing antibiotics and antifungal drugs).4, 55 Serum blood samples for serological testing are useful for epidemiological studies.
Primary equine-derived cells (eg. foetal equine kidney, equine embryonic lung, equine thyroid or equine testis) for isolation and propagation of EHV-34, 11 are the best but this can also be easily performed on equine-derived cell lines named, E. Derm (NBL-6) cells (ATCC; CCL-57) (Figure 1) or FHK-Tcl3.1.5, 11, 35 Increased success of virus isolation is achieved if material collected from cutaneous erosions is inoculated directly (i.e. without prior homogenization, centrifugation, or filtration) onto monolayers of susceptible cells,4 and also a 1:2 dilution in foetal calf serum improves isolation as it diminishes the non-specific toxicity induced by the inoculum in cell culture.11 Characteristic CPE by EHV-3 is generally evident by one to two days after inoculation of cell monolayers.
Regardless, detection of EHV-3 DNA by conventional or real-time PCR, which is carried out in many laboratories,20, 33, 41, 50, 52, 58 contributes to rapid confirmation of EHV-3 infection, allowing the immediate implementation of control measures. Although less useful, a serological diagnosis of infection with EHV-3 can be made retrospectively by the demonstration of a significant rise (four-fold) in the level of neutralizing antibody in paired serum samples obtained during the acute and convalescent disease period.4, 55 A very promising new platform based on the fluorescent probe hydrolysis-based insulated isothermal PCR (iiPCR) technology was described in 2018.57 This on-site diagnostic tool serves for the rapid and accurate detection of EHV-3 in breeding farms, facilitating the identification of subclinically infected mares prior to mating, hence avoiding contagion of the stallion and contributing to prevention of huge outbreaks of ECE. While this technology has been validated, it is yet to be implemented in the field.
Differential diagnosis
Clinically, typical cases of ECE can usually be diagnosed without difficulty. In ECE-like outbreaks accompanied by exanthematous lesions on the lips or nostrils of horses, vesicular stomatitis must be considered. In geographical areas where true horse pox still occurs, this orthopoxvirus should be included in the differential diagnosis. Also, in geographical areas in which contagious equine metritis (caused by Taylorella equigenitalis) has been reported, this agent should be excluded by careful laboratory examination in mares in which a mucopurulent vulvar discharge is the prominent feature of the clinical picture. It should be borne in mind that exanthematous lesions on the external genitalia of mares as a result of infection with equid herpesvirus 1 have, on occasion, been reported.4
Treatment, Prevention and Control
The therapeutic objective of the treatment of breeding stallions and mares exhibiting ECE is shortening of the required period of suspended mating by promoting the rapid and uncomplicated healing of genital lesions. The treatment of stallions showing clinical ECE is mainly based on sexual rest until lesions have healed, daily cleansing of the genitalia, and administration of anti-inflammatories and broad-spectrum antimicrobials to avoid secondary bacterial infections.4, 32, 55 Antiseptics, antimicrobials and anti-inflammatories are applied topically, once or twice daily, as creame- or ointment-based emollients. During cleansing or topical application of these, care should be taken to avoid accidental, traumatic removal of the crusts of healing sores.
The use of topical antiviral compounds has been explored recently. Nucleoside analogues, such us acyclovir (ACV) and ganciclovir (GCV), have demonstrated to be effective against EHV-3 in vitro23, 45 and subsequent studies confirmed that among both, GCV has a higher efficiency to inhibit EHV-3 replication.49, 60 Further studies performed in vivo showed that 1 per cent topical GCV, once a day, contributes to the clearance of EHV-3 shedding,thus reducing the time mares and stallions should be segregated from reproduction in the face of an ECE outbreak.56, 59 The implementation of a preventive treatment in subclinically infected mares in order to diminish virus replication and shedding has been studied.56 It has been observed in a Thoroughbred breeding farm with history of ECE outbreaks that the use of 1 per cent topical GCV in mares, during three days after foaling was helpful in diminishing the occurrence of clinical ECE during two consecutives breeding seasons.54
Taking into account that the most important negative consequence of ECE is the occurrence of the disease in stallions during the breeding season, the preventive measures rely mainly on clinical examination of mares before mating and segregating those with clinical evidence of ECE. This procedure does not identify subclinically infected animals, leading to a high risk of contagion for stallions. Thus, in breeding farms with heavily-scheduled calendars, it is highly recommended to perform strict clinical examination before breeding, and to implement an accurate biosecurity management protocol during the pre-mating hygienic procedures in mares and post-mating in stallions.4, 55 Moreover, to overcome the possibility that subclinically infected mares will transmit the virus to stallions, the adoption of additional preventive measures, such as identification of these mares by, for example, on-site real-time PCR and segregation from mating, is strongly recommended.55
A commercial vaccine against ECE is not available, and the alternative for an EHV-3 vaccine has not been explored yet. However, a vaginal immunization against CpHV-1, EHV-3 counterpart in goats, has been developed containing the inactivated virus and a mutant enterotoxin of Escherichia coli as adjuvant. Vaccinated goats displayed high levels of secretory IgA and were significantly protected after challenge with the virulent CpHV-1 strain, with pronounced decrease in virus shedding.19, 48 Because of the existence of EHV-3 latently infected carrier animals in most horse populations, occasional reactivations of latent virus with recrudescence of clinical or subclinical infections are unavoidable. As reactivation of latent virus is not preventable, the basis for controlling the negative impact of outbreaks of ECE in breeding establishments is containment of the spread of infection.
A stringent code of practice should be implemented within breeding sheds following observation of a case of ECE. The three priorities necessary for successful ECE control are:
- Cessation of breeding of clinically affected animals;
- Heightened vigilance for early recognition of new clinical cases;
- Strict adherence to breeding shed hygiene procedures designed to eliminate mechanical transmission of the virus.
The most effective method for restricting the transmission of EHV-3 infection remains the immediate cessation of mating activities of clinically affected stallions and mares. The decision to return an EHV-3 infected stallion or mare to breeding service should be based on a clinical evaluation together with a negative result of EHV-3 PCR on perineal-genital swab.4, 55 The quarantine time interval, which may vary from horse to horse, will be influenced by the extent and severity of the lesions and by the rapidity of the healing process. As previously mentioned, the recovery period may be prolonged by secondary bacterial infection.4, 55
Personnel assisting with the preparation of mares or stallions for breeding should be informed of the importance of early detection of new cases of ECE and trained in recognition of the lesions of ECE. Assistants in the mare chute area responsible for preparing mares for cover (e.g. washing the genitalia and wrapping the tail) can be instrumental in identifying animals with suspect lesions. Similarly, stallion crew members working close to the animals during mating often have the best vantage point for recognition of a new case of ECE.
Strict adherence to hygienic measures (or biosecurity measures) designed to prevent the mechanical spread of EHV-3 during an ECE outbreak is a critical component of the disease control plan. Care must be taken to avoid iatrogenic transmission of the virus through contaminated equipment such as rectal sleeves, gloves, vaginal specula orinsemination utensils.
Breeding shed personnel who have direct contact with horses should wear long, disposable examination sleeves and short latex gloves which are changed between each horse handled. Mare preparation chutes should be washed down with water and then disinfected between the preparation of each mare to prevent cross-infection. A thorough rinsing of each stallion’s penis and prepuce with plain warm water after each mating has been used with the aim of reducing the viral load of any potential inoculum of EHV-3 acquired from the covered mare. Finally, all instruments, buckets, and supporting devices used during the breeding procedure should be washed and sterilized between use or fitted with clean disposable covers or liners.
Although there is still no commercial product available for use, the use of antivirals as a therapeutic treatment in clinically affected animals contributes to the clearance of virus shedding, thus reducing the time mares and stallions are segregated, and also reducing the risk of contagion and environmental contamination. As the administration of the treatment does not require specific expertise, owners or breeders can easily do it, which would be helpful for treatments during the breeding season.
Finally, the combined implementation of an on-site diagnostic PCR (i.e. iiPCR) during pre-mating inspection in mares with the consequent segregation of EHV-3 positives, together with the application of a topical treatment in quarantined mares, would minimize EHV-3 transmission, avoiding the infection of stallions, the possible progression to an outbreak, and, hence, the temporal disruption of matings.
References
- The effect of chemical tick control on cattle on free-living ixodid ticks and on ticks parasitic on sympatric impala in the Central Province, Zambia.
- AKHMEDZHANOV, M., SCROCHI, M., BARRANDEGUY, M., VISSANI, A., OSTERRIEDER, N. &DAMIANI, A. M., 2017. Construction and Manipulation of a Full-Length Infectious Bacterial Artificial Chromosome Clone of Equine Herpesvirus Type 3 (EHV-3). Virus Research, 228. https://doi.org/10.1016/j.virusres.2016.11.012.
- ALLEN, G. &RANDALL, C., 1979. Proteins of Equine Herpesvirus Type 3. I. Polypeptides of the Purified Virion. Virology, 92, 252-257.
- ALLEN, G. P. &UMPHENOUR, N. W., 2004. Equine Coital Exanthema. In: Infectious Diseases of Livestocks; COETZER, J. & TUSTIN, R., (eds.). Oxford Press: Cape Town, 860-867.
- ANDOH, K., KAI, K., MATSUMURA, T. &MAEDA, K., 2009. Further Development of an Equine Cell Line That Can Be Propagated over 100 Times. Journal of Equine Veterinary Science, 20(2), 11–14. https://doi.org/10.1294/jes.20.11.
- ATASEVEN, V. S., DOGAN, F., BILGE DAGALP, S., TAN, M. T., ALTINBAY, K. &ATES, C. T., 2014. Serological Survey of Equid Herpesvirus 3 Infection in Turkish Horses. Turkish Journal of Veterinary and Animal Sciences, 38, 295-298. https://doi.org/10.3906/vet-1311-72.
- ATHERTON, S. S., SULLIVAN, D. C., DAUENHAUER, S. A., RUYECHAN, W. T. &O’CALLAGHAN, D. J., 1982. Properties of the Genome of Equine Herpesvirus Type 3. Virology, 120(1), 18-32. https://doi.org/10.1016/0042-6822(82)90003-4.
- BARBA, M., STEWART, A. J., SHRADER, S. M., KOEHLER, J. W., JOHNSON, A. K. &CALDWELL, F. J., 2015. Keratitis and Periocular Lesions Associated with Equine Herpesvirus-3 in a 3-Month-Old Filly. Equine Veterinary Education, 27(12), 618–622. https://doi.org/10.1111/eve.12365.
- BARRANDEGUY, M., 2010. Virological Aspects and Pathogenesis of Natural and Experimental Equid Herpesvirus 3 Infection in Horses, PhD Degree, Université of Liége, Belgium.
- BARRANDEGUY, M., PERKINS, J., DONOUGH, J., MAC., VISSANI, A., OLGUIN, C. &THIRY, E., 2010. Occurrence of Equine Coital Exanthema in Mares from an Embryo Transfer Center. Journal of Equine Veterinary Science, 30(3), 145-149. https://doi.org/10.1016/j.jevs.2010.01.059.
- BARRANDEGUY, M. &THIRY, E., 2012. Equine Coital Exanthema and Its Potential Economic Implications for the Equine Industry. Veterinary Journal, 191(1), 35-40 https://doi.org/10.1016/j.tvjl.2011.01.016.
- BARRANDEGUY, M., ULLOA, N., BOK, K. &FERNÁNDEZ, F., 2010. Outbreak of Rhinitis Caused by Equine Herpesvirus Type 3. Veterinary Record, 166(6), 178. https://doi.org/10.1136/vr.c444.
- BARRANDEGUY, M., VISSANI, A., LEZICA, F. P., SALAMONE, J., HEGUY, A., BECERRA, L., OLGUIN PERGLIONE, C. &THIRY, E., 2010. Subclinical Infection and Periodic Shedding of Equid Herpesvirus 3. Theriogenology, 74(4), 576–580. https://doi.org/10.1016/j.theriogenology.2010.03.014.
- BARRANDEGUY, M., VISSANI, A., OLGUIN, C., BARBARA, G., VALENZUELA, H., BECERRA, L., TORDOYA, M., MIÑO, S. &THIRY, E., 2012. Experimental Infection with Equid Herpesvirus 3 in Seronegative and Seropositive Mares. Veterinary Microbiology, 160(3-4), 319-326. https://doi.org/http://dx.doi.org/10.1016/j.vetmic.2012.06.024.
- BARRANDEGUY, M., VISSANI, A., OLGUIN, C., BECERRA, L., MIÑO, S., PEREDA, A., ORIOL, J. &THIRY, E., 2008. Experimental Reactivation of Equine Herpesvirus-3 Following Corticosteroid Treatment. Equine Veterinary Journal, 40(6), 593-595. https://doi.org/10.2746/042516408X333399.
- BROWNING, G. F., FICORILLI, N. &STUDDERT, M. J., 1988. Asinine Herpesvirus Genomes: Comparison with Those of the Equine Herpesviruses. Archives of Virology, 101(3-4), 183-190. https://doi.org/10.1007/BF01310999.
- BRYANS, J. T. &ALLEN, G. P., 1972. In Vitro and in Vivo Studies of Equine Coital Exanthema. In: Third International Conference on Equine Infectious Diseases; BRYANS, J. & GERBER, H., (eds.). Paris, 322-342.
- BRYANS, J. T. &ALLEN, G. P., 1974. No Equine Infectious Diseases. Paris 1972. Basel, Karger. In: Proceedings 3rd International Conference, 322–336. https://doi.org/10.1159/000393546.
- CAMERO, M., BELLACICCO, A. L., TARSITANO, E., DECARO, N., MARTELLA, V., TEMPESTA, M. &BUONAVOGLIA, C., 2007. Intravaginal Administration of an Inactivated Vaccine Prevents Lesions Induced by Caprine Herpesvirus-1 in Goats, 25, 1658-1661. https://doi.org/10.1016/j.vaccine.2006.11.002.
- COCHARD, T., LÉON, A., HUET, C., FORTIER, G. &PRONOST, S., 2002. Exanthème Coïtal Dans Un Élevage de Poneys Fjord. Intérêt Du Diagnostic Par PCR. In: Proceedings of the Association Vétérinaire Equines Français. Le Touquet, France, 423.
- CRAIG, J. F. &KEHOE, D., 1921. Horse Pox and Coital Exanthema. Journal of Comparative Pathology, 34, 126-129. doi: 10.1016/S0368-1742(21)80014-4.
- CRANDEL, R. &DAVIS, E., 1985. Isolation of Equine Coital Exanthema Virus (Equine Herpesvirus 3) from the Nostril of a Foal. Journal of the American Veterinary Medical Association, 187, 503-504.
- CULLINANE, A., MCGING, B. &NAUGHTON, C., 1994. The Use of Acyclovir in the Treatment of Coital Exanthema and Ocular Disease Caused by Equine Herpesvirus 3. In: Equine Infectious Diseases VII; PLOWRIGHT, W. & NAKAJIMA, H., (eds.). R & W Publications: Newmarket (Suffolk), 55.
- DAVISON, A. J., 2010. Herpesvirus Systematics. Veterinary Microbiology, 143(1), 52-69. https://doi.org/10.1016/j.vetmic.2010.02.014.
- DAYARAM, A., SEEBER, P. A. &GREENWOOD, A. D., 2021. Environmental Detection and Potential Transmission of Equine Herpesviruses. Pathogens, 10(4), 423. https://doi.org/10.3390/pathogens10040423.
- DWYER, R. M., 2004. Environmental Disinfection to Control Equine Infectious Diseases. Veterinary Clinics of North America: Equine Practice, 20(3), 531-542. https://doi.org/10.1016/j.cveq.2004.07.001.
- FERREIRA, C., COSTA, E. A., FRANÇA, S. A., MELO, U. P. D., DRUMOND, B. P., BOMFIM, M. R. Q., COELHO, F. M., RESENDE, M., PALHARES, M. S. &SANTOS, R. L., 2010. Equine Coital Exanthema in a Stallion. Arquivo Brasileiro de Medicina Veterinaria e Zootecnia, 62(6), 1517-1520.
- FILHO, G. B. S., BOM, H. A. S. C., FONSECA, S. M. C., COSTA, É. A., SANTOS, B. S. A. S., SANTOS, R. L., SOUZA, F. A. L., NETO, J. E. &MENDONÇA, F. S., 2021. Equine Coital Exanthema Caused by Equid Alphaherpesvirus 3: A Report of an Outbreak in Northeastern Brazil. Pesquisa Veterinária Brasileira, 41, 1-7. https://doi.org/10.1590/1678-5150-PVB-6877.
- GIBBS, E. P. J., ROBERTS, M. C. &MORRIS, J. M., 1972. Equine Coital Exanthema in the United Kingdom. Equine Veterinary Journal, 4(2), 74-80. https://doi.org/10.1111/j.2042-3306.1972.tb03883.x.
- GIRARD, A., GREIG, A. S. &MITCHELL, D. A., 1968. Virus Associated with Vulvitis and Balanitis in the Horse. Preliminary Report. Canadian Journal of Comparative Medicine and Veterinary Science, 32(4), 603-604.
- HUE, E., FORTIER, C., NORMAND, C., SUTTON, G., THIEULENT, C., MARCILLAUD-PITEL, C., LEGRAND, L. &PRONOST, S., 2021. Circulation and Characterisation of Equid Herpesvirus-3 Strains in France between 2010 and 2021. In: Equine Infectious Diseases Conferences, 62.
- HUSSEY, G. S. &LANDOLT, G. A., 2015. Equine Alphaherpesviruses. In: Robinson’s Current Therapy in Equine Medicine, Elsevier, 158-161. https://doi.org/10.1016/B978-1-4557-4555-5.00037-6.
- KIRISAWA, R., TOISHI, Y., AKAMATSU, A. &SOEJIMA, K., 2017. Isolation of Equine Herpesvirus 3 ( EHV- 3 ) from Equine Coital Exanthema of Two Stallions and Sero-Epidemiology of EHV-3 Infection in Japan. Journal of Veterinary Medical Science, 79(3), 636-643. https://doi.org/10.1292/jvms.16-0518.
- KROGSRUD, J. &ONSTAD, O., 1971. Equine Coital Exanthema. Isolation of a Virus and Transmission Experiments. Acta Veterinaria Scandinavica, 12(1), 1-14.
- MAEDA, K., YASUMOTO, S., TSURUDA, A., ANDOH, K., KAI, K., OTOI, T. &MATSUMARA, T., 2007. Establishment of a Novel Equine Cell Line for Isolation and Propagation of Equine Herpesviruses. Journal of Veterinary Medical Science, 69(9), 989–991. https://doi.org/10.1292/jvms.69.989.
- MARTELLA, V., LANAVE, G., CAMERO, M., LAROCCA, V., LORUSSO, E., CATELLA, C., CAPOZZA, P., TEMPESTA, M. &BUONAVOGLIA, C., 2020. Identification of a Novel α-Herpesvirus Associated with Ulcerative Stomatitis in Donkeys. Emerging Infectious Diseases, 26(12), 3044-3047. https://doi.org/10.3201/EID2612.200201.
- METCALF, C., 2001. The Role of International Transport of Equine Semen on Disease Transmission. Animal Reproduction Science, 68, 229-237. https://doi.org/10.1046/j.1440-172x.2001.00279.x.
- NEGUSSIE, H., LI, Y., TESSEMA, T. S. &NAUWYNCK, H. J., 2016. Replication Characteristics of Equine Herpesvirus 1 and Equine Herpesvirus 3 : Comparative Analysis Using Ex Vivo Tissue Cultures. Veterinary Research, 1-11. https://doi.org/10.1186/s13567-016-0305-5.
- PASCOE, R., 1981. The Effect of Equine Coital Exanthema on the Fertility of Mares Covered by Stallions Exhibiting the Clinical Disease. Australian Veterinary Journal, 57, 111-114.
- PASCOE, R. R. &BAGUST, T. J., 1975. Coital Exanthema in Stallions. Journal of Reproduction and Fertility. Supplement, 147-150.
- PRONOST, S., BRAU, S., LEGRAND, L. &CADORÉ, J. L., 2012. Equine Coïtal Exanthema in France: Evidence of EHV-3 Infection by Real Time PCR. Journal of Equine Veterinary Science, 32(10), S77-S78. https://doi.org/10.1016/j.jevs.2012.08.165.
- REED, S. M. &TORIBIO, R. E., 2004. Equine Herpesvirus 1 and 4. Veterinary Clinics of North America: Equine Practice, 20(3), 631-642. https://doi.org/10.1016/j.cveq.2004.09.001.
- SEKI, Y., SEIMIYA, Y. M., YAEGASHI, G., KUMAGAI, S.-I., SENTSUI, H., NISHIMORI, T. &ISHIHARA, R., 2004. Occurrence of Equine Coital Exanthema in Pastured Draft Horses and Isolation of Equine Herpesvirus 3 from Progenital Lesions. Journal of Veterinary Medical Science, 66(12), 1503-1508. https://doi.org/10.1292/jvms.66.1503.
- SIJMONS, S., VISSANI, A., TORDOYA, M. S., MUYLKENS, B., THIRY, E., MAES, P., MATTHIJNSSENS, J., BARRANDEGUY, M. &VAN RANSTA, M., 2014. Complete Genome Sequence of Equid Herpesvirus 3. Genome Announcemants, 2(5). https://doi.org/10.1128/genomeA.00797-14.
- SMITH, K. O., GALLOWAY, K. S., KENNELL, W. L., OGILVIE, K. K. &RADATUS, B. K. A., 1982. New Nucleoside Analog , 9- [[ 2-Hydroxy-1- ( Hydroxymethyl ) Ethoxy ] Methyl ] Guanine, Highly Active In Vitro Against Herpes Simplex Virus Types 1 and 2. Antimicrobial Agents and Chemotherapy, 22(1), 55-61.
- STUDDERT, M., 1996. Equine Coital Exanthema (Equine Herpesvirus 3). In: Virus Infections of Equines; STUDDERT, M., (ed.). Elsevier,: Amsterdam, 39-46.
- TEMPESTA, M., BUONAVOGLIA, D., SAGAZIO, P., PRATELLI, A. &BUONAVOGLIA, C., 1998. Natural Reactivation of Caprine Herpesvirus 1 in Latently Infected Goats. Veterinary Record, 143, 200.
- TEMPESTA, M., CAMERO, M., BELLACICCO, A. L., TARSITANO, E. &CRESCENZO, G., 2007. Potent Inhibition of Genital Herpesvirus Infection in Goats by Cidofovir. Antiviral Therapy, 12, 977-979.
- THIEULENT, C. J., HUE, E. S., FORTIER, C. I., DALLEMAGNE, P., ZIENTARA, S., MUNIER-LEHMANN, H., HANS, A., FORTIER, G. D., PITEL, P.-H., VIDALAIN, P.-O. &PRONOST, S. L., 2019. Screening and Evaluation of Antiviral Compounds against Equid Alpha-Herpesviruses Using an Impedance-Based Cellular Assay. Virology, 526, 105-116. https://doi.org/10.1016/J.VIROL.2018.10.013.
- THORSTEINSDÓTTIR, L., GUÐMUNDSSON, G. Ö. &JENSSON, H., 2021. Torsteinsdóttir, S. Isolation of Equid Alphaherpesvirus 3 from a Horse in Iceland with Equine Coital Exanthema. Acta Veterinaria Scandinavica, 4-7. https://doi.org/10.1186/s13028-021-00572-4.
- TOISHI, Y., TSUNODA, N. &KIRISAWA, R., 2017. Occurrence of Equine Coital Exanthema (ECE) in Stallions in Japan and Effectiveness of Treatment with Valacyclovir for ECE. Journal of Veterinary Medical Science, 79(3), 632-635. https://doi.org/10.1292/jvms.16-0511.
- VAN DER MEULEN, K., CAIJ, A., SMETS, K. &NAUWYNCK, H., 2006. Equine Coital Exanthema in a Mare in Belgium. Vlaams Diergeneeskundig Tijdschrift, 75, 286-289.
- VANDEKERCKHOVE, A. P., GLORIEUX, S., GRYSPEERDT, A. C., STEUKERS, L., VAN DOORSSELAERE, J., OSTERRIEDER, N., VAN DE WALLE, G. R. &NAUWYNCK, H. J., 2011. Equine Alphaherpesviruses ( EHV-1 and EHV-4 ) Differ in Their Efficiency to Infect Mononuclear Cells during Early Steps of Infection in Nasal Mucosal Explants. Veterinary Microbiology, 152(1-2), 21-28 https://doi.org/10.1016/j.vetmic.2011.03.038.
- VISSANI, M. A., 2022. Personal Comunication; p Instituto de Virología CICVyA, Instituto Nacional.
- VISSANI, M. A., DAMIANI, A. M. &BARRANDEGUY, M. E., 2021. Equine Coital Exanthema: New Insights on the Knowledge and Leading Perspectives for Treatment and Prevention. Pathogens, 10(8), 1-10. https://doi.org/10.3390/pathogens10081055.
- VISSANI, M. A., PERGLIONE, C. O., ZABAL, O., ALVAREZ, G., THIRY, E., BARRANDEGUY, M. &PARREÑO, V., 2020. Topical Ganciclovir Reduces Viral Excretion in Mares With Equine Coital Exanthema. Journal of Equine Veterinary Science, 94, 103199. https://doi.org/10.1016/j.jevs.2020.103199.
- VISSANI, M. A., TORDOYA, M. S., TSAI, Y. L., LEE, P. Y. A., SHEN, Y. H., LEE, F. C., WANG, H. T. T., PARREÑO, V. &BARRANDEGUY, M., 2018. On-Site Detection of Equid Alphaherpesvirus 3 in Perineal and Genital Swabs of Mares and Stallions. Journal of Virological Methods, https://doi.org/10.1016/j.jviromet.2018.04.002.
- VISSANI, M. A., VENTIMIGLIA, A., ALONSO, J. C., OLGUIN-PERGLIONE, C., ALAMOS, F. &BARRANDEGUY, M., 2021. Diagnosis of Equid Alphaherpesvirus 3 before Mating: Its Contribution to Control Outbreak. 11th International Equine Infectious Diseases Conference, under review.
- VISSANI, M. A., VENTIMIGLIA, A., ALONSO, J. C., TORDOYA, M. S., PARREÑO, V. &BARRANDEGUY, M., 2021. Mares Naturally Infected with Equid Alphaherpesvirus 3: Evaluation of Treatment Effectiveness. 11th Equine Infectious Diseases Conference, under review.
- VISSANI, M. A., ZABAL, O., TORDOYA, M. S., PARREÑO, V., THIRY, E. &BARRANDEGUY, M., 2018. In Vitro Comparison of Acyclovir, Ganciclovir and Cidofovir against Equid Alphaherpesvirus 3 and Evaluation of Their Efficacy against Six Field Isolates. Revista Argentina de Microbiología, 50(4), 380-390. https://doi.org/10.1016/j.ram.2018.01.003.
- YANG, J. &LIM, Y. K., 2012. Epizootiologic Evaluation on Equine Coital Exanthema in Korea. Korean Journal of Veterinary Research, 52(1), 61-64 https://doi.org/10.14405/kjvr.2012.52.1.061.
- YUROV, K. P. &ALEKSEENKOVA, S. V., 2019. Coital Exanthema of Horses: Diagnosis and Identification of the Pathogen. Russian Veterinary Journal. Productive Animals, 6, 5-8.