- Infectious Diseases of Livestock
- Part 2
- Foot-and-mouth disease
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Foot-and-mouth disease
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
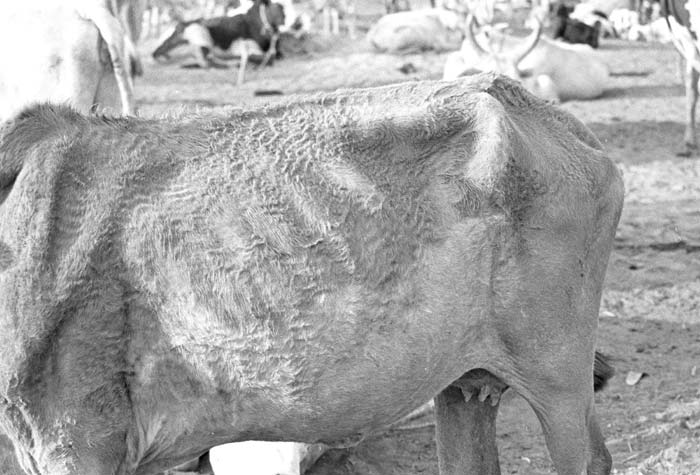
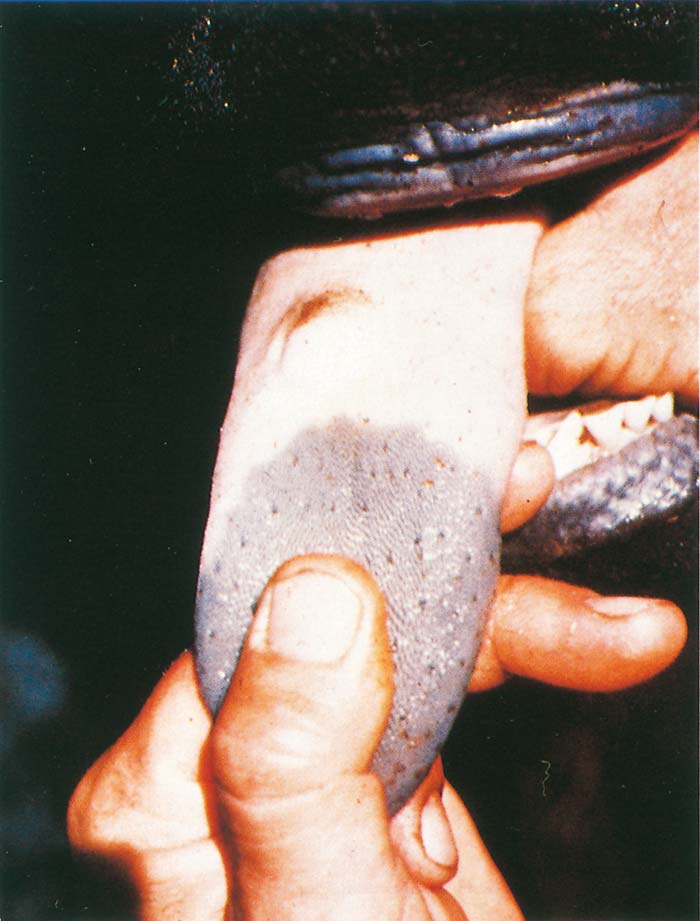
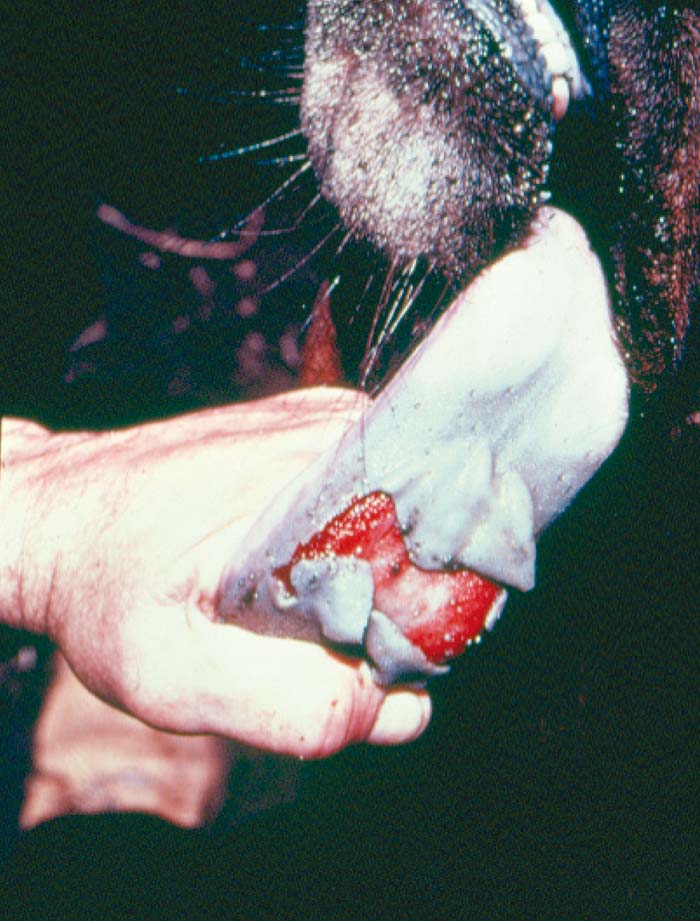
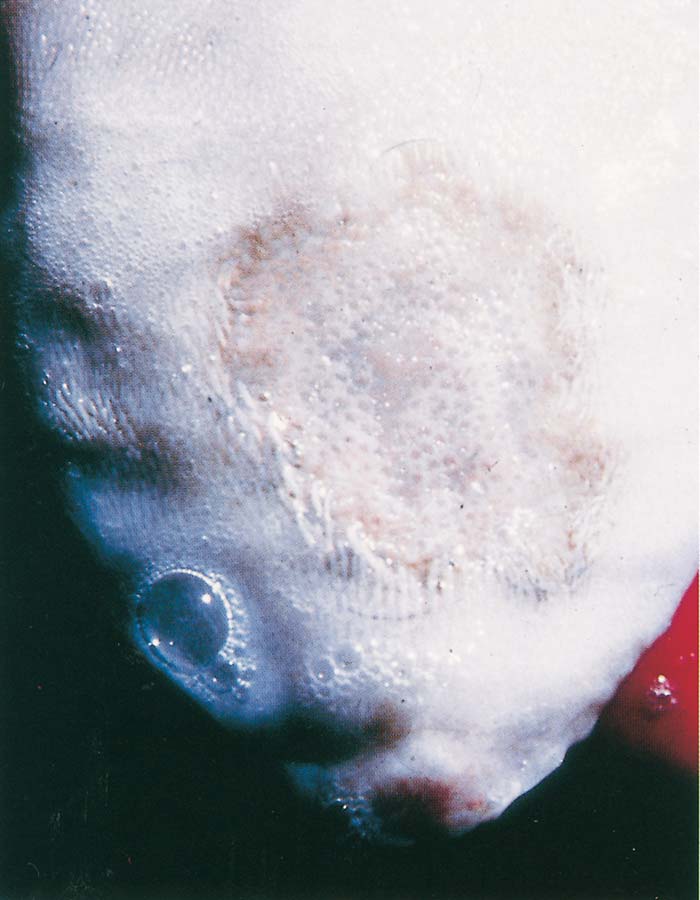
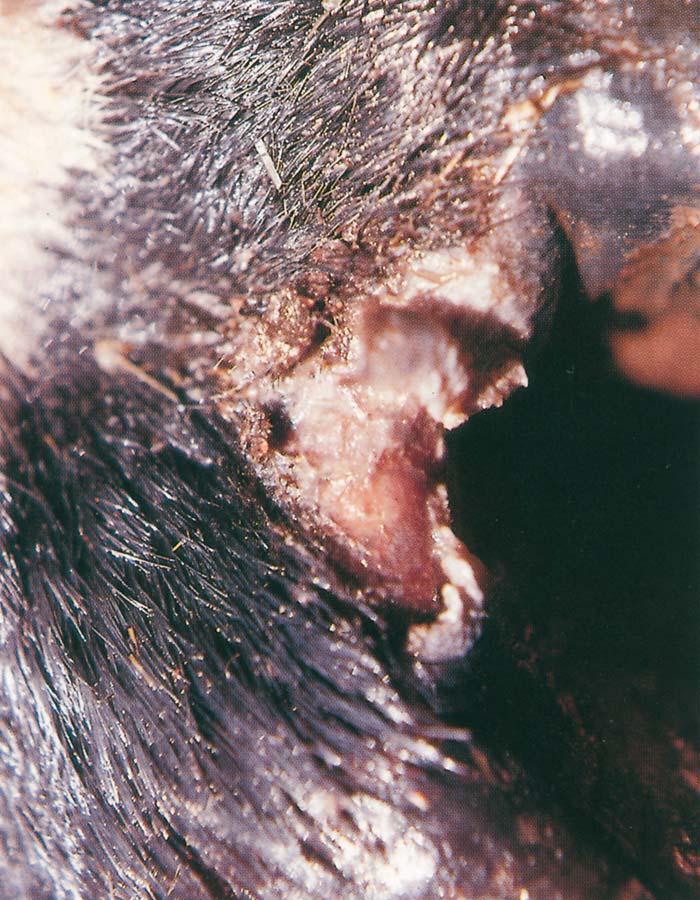
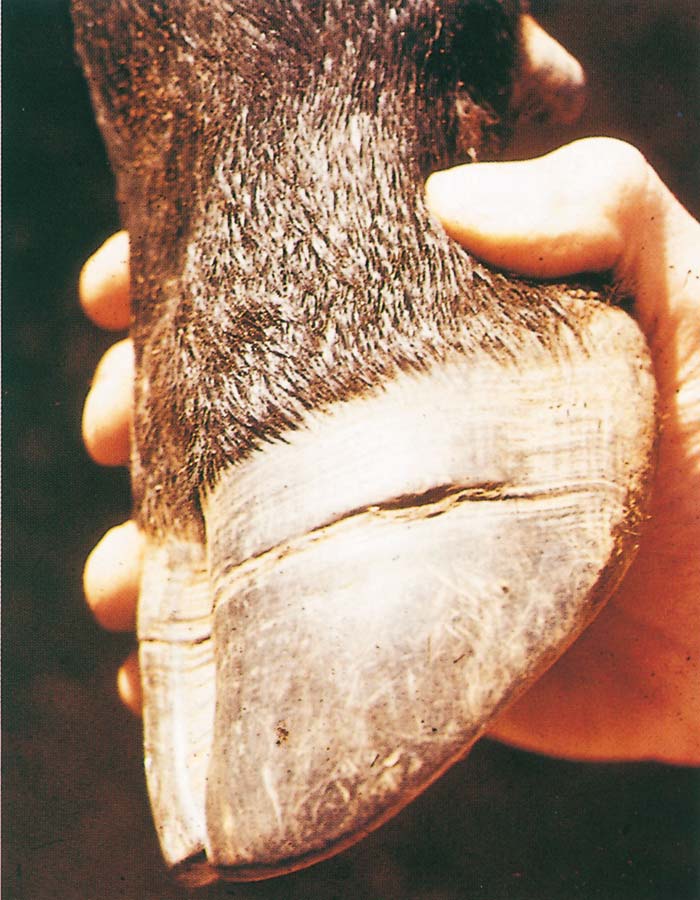
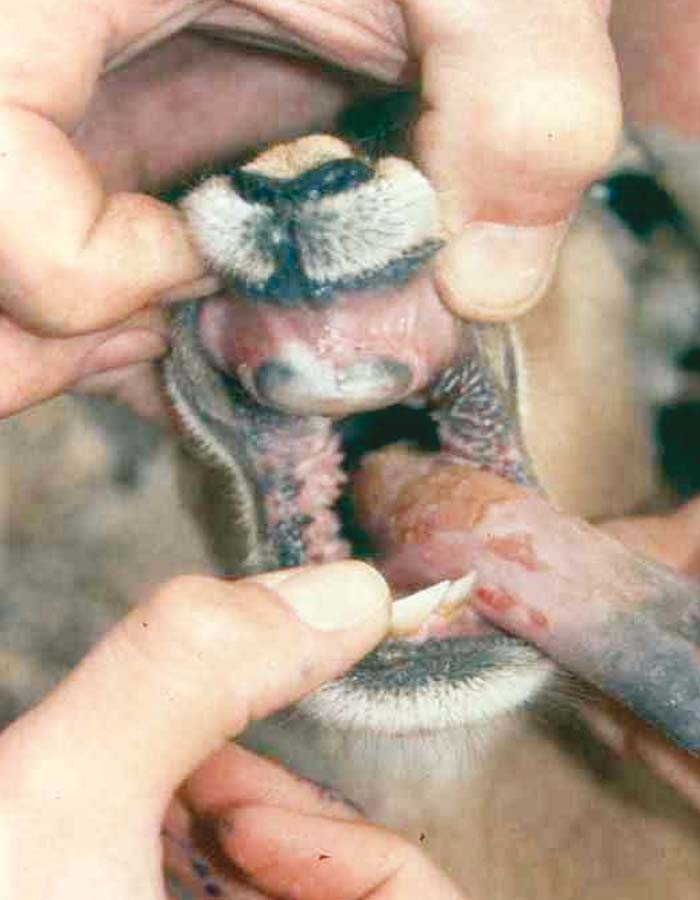
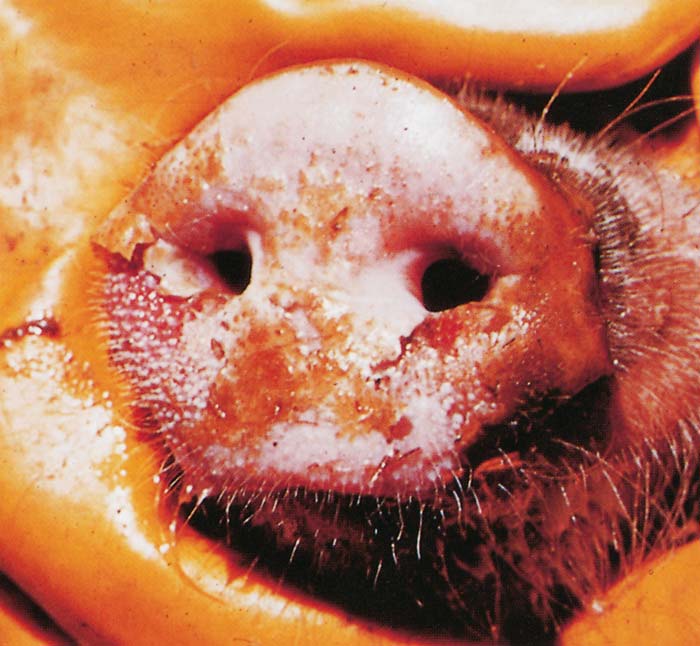
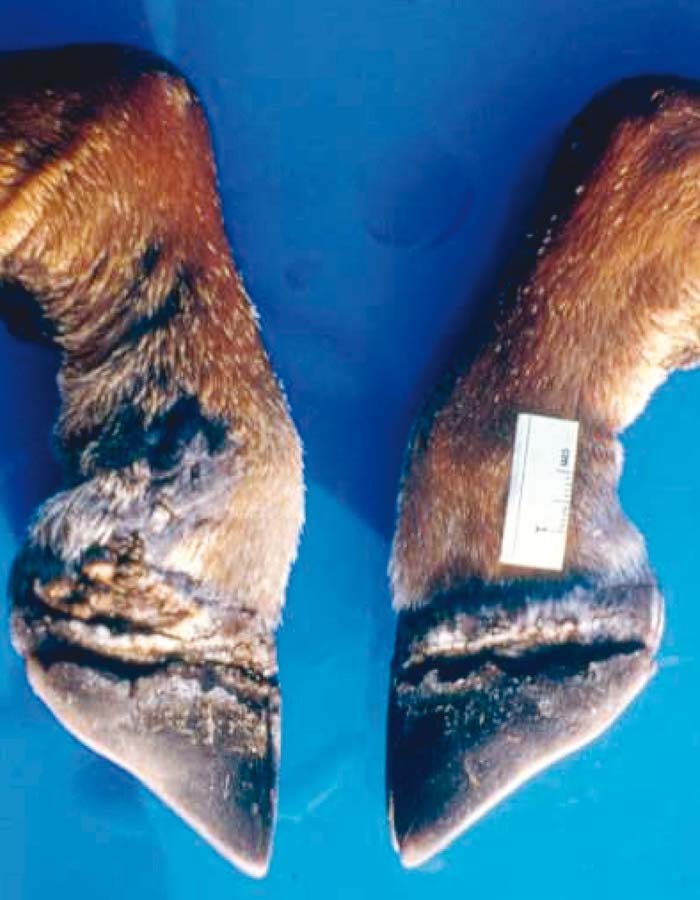
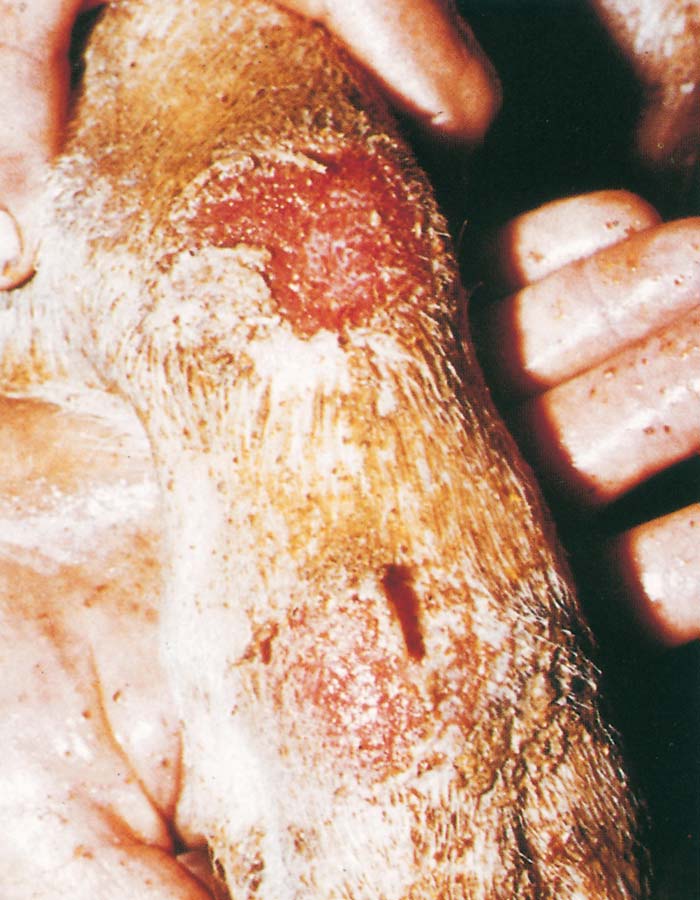
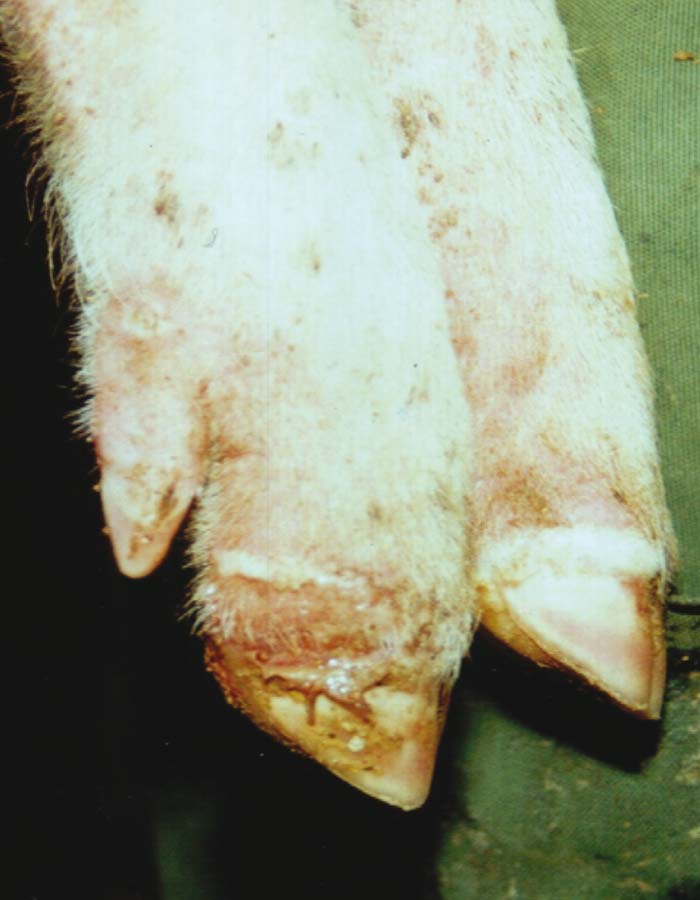
Foot-and-mouth disease (FMD) is a highly contagious and usually acute affliction of cloven-hoofed animals and camelids caused by a virus of the family Picornaviridae. The susceptibility of different species to infection and their ability to transmit it, however, are highly variable. In cloven- hoofed livestock the disease is usually characterized by high morbidity, low mortality and the development of vesicles and erosions in the mucosa of the mouth and skin of the interdigital spaces and coronary bands.
During the second half of the nineteenth and first half of the twentieth centuries in western Europe and North America repeated epidemics of rapidly spreading FMD resulted in serious losses, predominantly among high-producing livestock raised under increasingly intensive systems. As the economic effects of the disease and the difficulties of controlling it became apparent, concerted efforts towards eradicating it were undertaken. This was eventually achieved in North America in 1929 and in the European Union in 1992. Among other major livestock producers, Australia only experienced FMD prior to the turn of the nineteenth century and New Zealand has never been required to respond to an outbreak. Japan achieved eradication in 1908.
The logistically difficult and costly efforts required to eradicate the disease resulted in countries which had achieved eradication becoming wary of re-importing it, particularly from regions where exotic types of FMD virus (FMDV) were prevalent. They consequently instituted measures to prevent this, including embargoes on agricultural imports from countries where efficient control is not practised or where the epidemiological situation with respect to FMD has not been accurately established. Embargoes were also sometimes used as barriers to exclude imports of livestock and livestock products from regions that were able to produce them more cheaply than the importer. Curbing the use of non-tariff barriers was one of the objectives of the General Agreement on Tariffs and Trade (GATT) and remains an ideal of its successor, the World Trade Organization (WTO).
In addition, the Office International des Epizooties (OIE), the international animal health body primarily concerned with facilitating international trade in animals and animal products, is increasingly involved in instituting rational guidelines to foster international trade on the one hand without endangering the FMD-free status of major importers on the other. This is proving increasingly contentious because of the conflicting desires of exporters for decreasing restrictions as opposed to the increasing fear of importers for the inadvertent arrival of FMDV in legally as well as illegally imported animals or animal products.
The recent epidemics of FMD that have occurred in countries long free from FMD such as Taiwan, Japan, South Korea, the UK, Ireland, France and the Netherlands,133 have re-emphasized the devastating repercussions that FMD can have. These effects have primarily been economic: the direct and indirect costs of the 2001 outbreak in the UK, for example, may have been as high as US$ 12 billion.317, 321 Similarly, the outbreak in Taiwan had far-reaching consequences particularly in the loss of the valuable export market to Japan for pork. However, other issues relating to animal welfare, ecological effects occasioned by large-scale carcass disposal and sociological problems experienced by farming and other rural industries (e.g. tourism) have also been profound.78, 205, 315 This upsurge in the prevalence of FMD has emphasized the difficulties of protecting countries and regions of the world free of FMD from reincursion of the infection. It also highlighted the extraordinary measures necessary to protect the livelihoods of farmers whose livestock and products are subject to movement restriction/ market access as a result of FMD outbreaks.65
Perhaps the most profound effect of these events on the conventional approach to animal health has been the questioning of accepted control practices on both technical and moral grounds. It is clear that in both developed and developing countries large-scale ‘stamping out’ (slaughter of infected and in-contact animals, and burial or incineration of the carcasses) is no longer socially or politically acceptable. This presents a new and acute challenge in the field of animal health control.
It is an unfortunate fact that FMD is still widespread in Africa, although in North and southern Africa considerable success in reducing the prevalence of the disease and in developing FMD-free zones has been achieved. In many countries outside these two regions there is little attempt to control the infection while in others the policies and practices applied sometimes ignore important epidemiological principles and are therefore largely futile. If this situation is not changed it will continue to retard the development of many parts of Africa, particularly in the arid zones of the continent. The reason is simply that livestock are one of the few tradable commodities that those in arid parts of Africa possess and FMD, together with some other important infections of livestock, precludes access of livestock and livestock products from countries in these regions to international markets where good prices are achievable. If, on the other hand, such countries were to gain access to markets where the best prices prevail — such as has been the case for southern African countries under the terms of the Lomé Convention funded by the European Union — assistance with escape from the poverty trap may be provided. In terms of competition between developing countries for access to markets in the developed world, there is an additional danger on the horizon for African countries. South America and parts of south-east Asia, which have historically suffered severely from the effects of FMD, are making rapid strides towards improved control or eradication of the disease, despite recent set-backs. This will enable them to access markets which African countries could target and thus limit the potential for growth in livestock- based exports from Africa in future. Basically, the danger for many African countries is that FMD will increasingly preclude access to international trade and render escape from the poverty trap on the part of rural communities more difficult than it would otherwise be.
Countries in sub-Saharan Africa face another problem that is unique in the context of FMD: the African buffalo (Syncerus caffer) is a maintenance host for the South African Territories (SAT) types of FMDV158 and large herds of infected buffalo are present in many of the larger countries of the subcontinent, particularly in eastern and southern Africa. This complicates control or eradication of the infection. For this reason there have, in at least one country, been attempts to eliminate buffaloes from areas other than wildlife reserves. However, the wholesale eradication of buffalo populations is aesthetically, ecologically and economically untenable. Therefore, the control ofFMDin wildlife populations in Africa needs to be addressed more actively than has been the case hitherto.
Foot-and-mouth disease is probably not an ancient disease; the first identifiable description was that of Hieronymus Francastorius in 1546, who described an outbreak in cattle in what is now Italy.164 However, probably because of the prevalence of other major animal plagues such as rinderpest in Europe during the nineteenth century, relatively little mention of FMD was made prior to 1886.50
After that FMD became increasingly important, and a number of FMD institutes to counter the ravages of the disease were established in Europe and South America in the first quarter of the twentieth century.
The virus of FMD was one of the first filterable agents to be identified.337 Prior to this discovery by Loeffler and Frosch in 1898, no known micro-organisms were capable of passing through Berkfeld filters. This finding led Loeffler and Frosch to deduce that the agent causing FMD was beyond the resolution of the light microscope.271
It is probable that FMD viruses have been present in Africa for a long time although, so far, there is no direct proof of this. Supporting evidence for this contention lies in the fact that the SAT types of FMDV are uniquely adapted to long-term survival in free-living African buffalo populations in southern, central and eastern Africa (see below). Furthermore, these viruses, as far as is known, have existed nowhere else (and therefore could not have been imported) and comprise a group of types clearly distinguishable, both genetically and immunologically, from the other four types of FMDV, viz types A, O, C and Asia-1, which presumably evolved in Asia and Europe.26, 246, 262
Because there is no written record of early historical events in sub-Saharan Africa, the earliest records of animal diseases south of the Sahara were provided by explorers, missionaries and settlers. Perhaps the best synopsis of these accounts was provided by Henning in 1956 164 and the following passage summarizing early accounts of FMD in South Africa is quoted in full:
In 1780 le Vailant 210 described a disorder in cattle, ‘klowsiekte’, which ‘attacked the feet of oxen causing them to swell prodigiously, and often producing suppuration; sometimes the hoofs dropped off. It was not attended with any danger and generally terminated in a fortnight’. Gordon Cumming in 1850 also reported serious losses among his oxen from the ‘tongue or hoof sickness’, along the Vaal River.79 He was detained for several days and was forced to purchase or barter fresh oxen from Mahura and his tribe. From enquiries made by him it appeared that the disease was well known to his Hottentot servants. In 1858 General S.J.P. Kruger reported on the campaign against Mahura along the Harts River, stating: ‘By this time our bullocks suffered much from the tongue and claw sickness.’200 According to information obtained from some of the oldest inhabitants of the Transvaal and Free State, a disease resembling foot-andmouth disease had been prevalent for a considerable time before it was recognized officially. On the other hand, there seems to be very little doubt that some forms of blue-tongue have been mistaken for foot-and-mouth disease in the past.37
The first official record of foot-and-mouth disease in South Africa was made by Hutcheon in 1892173 when an outbreak occurred in Griqualand West.
Although the source of the infection could not be traced, Hutcheon173 stated that the disease was first reported in parts of Mashonaland (Southern Rhodesia), from where it was considered to have spread along the traffic routes through Bechuanaland into Griqualand West, the Transvaal, the Orange Free State and Natal. Otto Henning,163 who investigated the outbreak found the disease to be identical with the European disorder. The scourge was well known to several farmers and, according to information obtained from some of the older inhabitants, it had been prevalent in South Africa for many years before this outbreak was reported. Although over 75 per cent of the incontact cattle contracted the disease, the mortality in adult animals did not exceed 3 per cent; but the losses in calves were reported to be considerably greater. At first attempts were made to confine the disease to the north of the Orange River by preventing all movements of cattle from the north to the south of the river; but this was not possible, and as soon as Hutcheon174 realized that outbreaks were occurring in several districts south of the river, he withdrew the restrictions. During the two following years mild epizootics occurred in different parts of the country,135 but the disease never assumed serious proportions, and no further outbreaks were reported after 1895. The advent of rinderpest in 1896 completely eclipsed foot-and-mouth disease in importance, and it may be that the devastation of the cattle population by that plague was responsible for its disappearance.
In April 1903 foot-and-mouth disease reappeared in South Africa.261 The infection was introduced into the Cape Peninsula by means of shipments of cattle from the Argentine. Fortunately the disease was confined to two places only, a farm where the imported cattle were kept and a local dairy that harboured a runaway heifer from the Argentine shipment. Both areas were immediately placed under strict quarantine; the affected animals were dressed with antiseptic lotions and kept under as hygienic conditions as circumstances permitted; and the premises were thoroughly disinfected. Although several of the Argentine animals succumbed to the secondary infection that resulted from their exposure to dirty, muddy surroundings, no deaths occurred in the local dairy herd. At about the end of July of the same year there was no further evidence of the disease, and the quarantine restrictions were raised.
Apart from the 1903 incident, FMD disappeared from the southern African subcontinent with the advent of the Great Rinderpest Pandemic that reached southern Africa in 1896, and did not reappear until April, 1931, when it occurred on what was then known as Nuanetsi Ranch in Southern Rhodesia (now Zimbabwe). The reappearance of the disease was a source of great consternation at the time as the ability of buffalo to provide a reservoir of infection was unknown and many believed that the infection had been reintroduced by imported animals or animal products, although no evidence for this could be found.106
Foot-and-mouth disease has occurred regularly in most southern African countries since 1931 and the cost of control has undoubtedly eclipsed that of any other viral disease from that time.
In other parts of sub-Saharan Africa, the Great Rinderpest Pandemic (see Rinderpest), which began in north-east Africa in 1889 and spread throughout the continent, presumably had the same effect on the prevalence of FMD as it did in southern Africa. There is unfortunately no information on this matter. In general, however, the effects of FMD in central, western and eastern Africa have been less pronounced than in southern Africa because other epidemic diseases that caused high mortality, particularly rinderpest, have coexisted with FMD for more than the last 100 years. This, together with the fact that intensive cattle and pig farming in these parts of Africa are poorly developed, has resulted in FMD having a lower profile than in southern Africa.
Other than in southern Africa, the records for the occurrence of FMD in Africa are incomplete, mainly because most cases and even outbreaks are unreported because the effects of the disease in extensive husbandry systems are often unremarkable. Officially recorded outbreaks by virus type, country and year have recently been reviewed for the last approximately 60 years.346 These, however, probably reflect only a small fraction of outbreaks that have occurred over this time period.
Furthermore, the knowledge of the distribution of serotypes in sub-Saharan Africa is imprecise; and even more so in the case of intratypic variants, i.e. lineages and topotypes which in the past were referred to as subtypes. Asia-1 is the only type that has so far not been recorded on the African continent.
Aetiology
Foot-and-mouth disease virus belongs to the genus Aphthovirus within the positive strand RNA virus family Picornaviridae. Previously, the sole member of this genus, FMDV is now the prototype for this taxon which takes its name from the Greek word aptha meaning ‘vesicles in the mouth’. At present, the Aphthovirus genus incorporates equine rhinitis A virus (previously equine rhinovirus type 1), on the basis of genomic organization and sequence similarity with FMDV.211, 338
In common with other picornaviruses, FMDV is non enveloped and has a roughly spherical capsid, exhibiting icosahedral symmetry.257, 266 The dry capsid diameter is 27 to 28 nm, and the virion consists of approximately 70 per cent protein and 30 per cent RNA, as well as a small quantity of lipid.22, 68 It has a molecular mass of about 8,5 × 106D with a sedimentation constant of 146S.52, 266
This characteristic sedimentation rate in sucrose gradients is widely used in vaccine manufacture to determine the mass of intact virions present in culture harvests because disintegration of virus particles results in loss of immunogenicity.
Seven immunologically distinct FMDV types occur, namely types A, O, C (the so-called European types), Asia-1 and the three South African Territories (SAT) types 1, 2 and 3. This serological classification is based on the inability of viruses from different types to induce cross-protection in animals.253 Under conditions of severe challenge, such as when a virus is inoculated into the epithelium of the tongue of cattle (intradermalingual inoculation), there is no cross protection between virus types.50 Likewise, a single dose of a monotypic vaccine fails to protect against heterotypic challenge.64 The serological classification of FMDV types is supported by molecular data. Graphical representation of the genetic relationships between the seven types, as given by phylogenetic trees, reveal that they form two genetically distinct groups, one comprising the three SAT types and the other consisting of types A, O, C and Asia-1 (Figure 125.1).
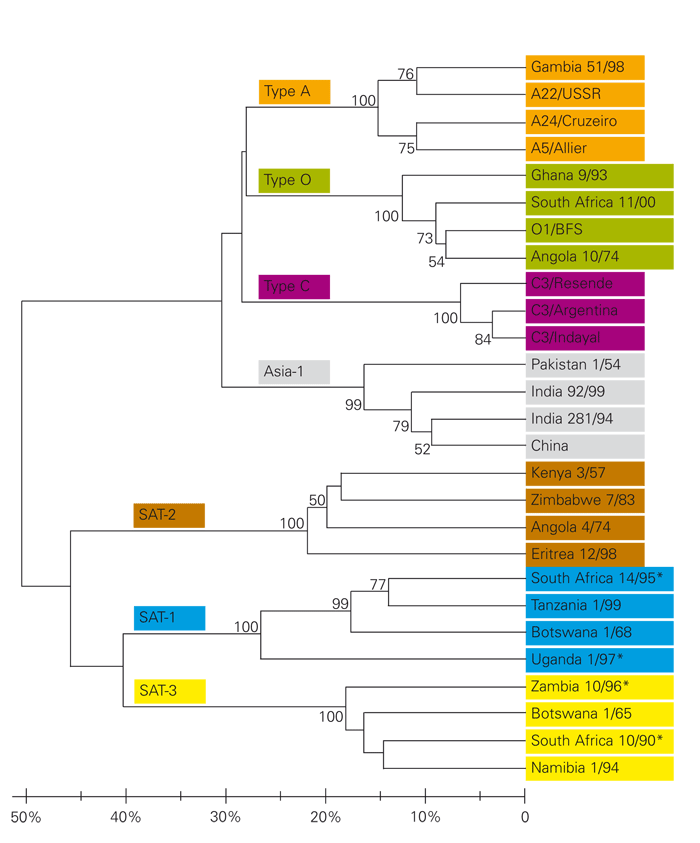
Figure 125.1 Phylogenetic tree depicting the VP1 gene relationships of the seven FMDV types based on amino acid sequences corresponding to the C-terminal half of the ID protein (positions 90–221 in SAT1 type viruses). The scale indicates the percentage divergence (calculated using p-distances) and support for internal nodes is indicated by bootstrap values > 50 per cent (based on 10 000 replications)
* Denotes viruses recovered from African buffalo. All other isolates were obtained from livestock
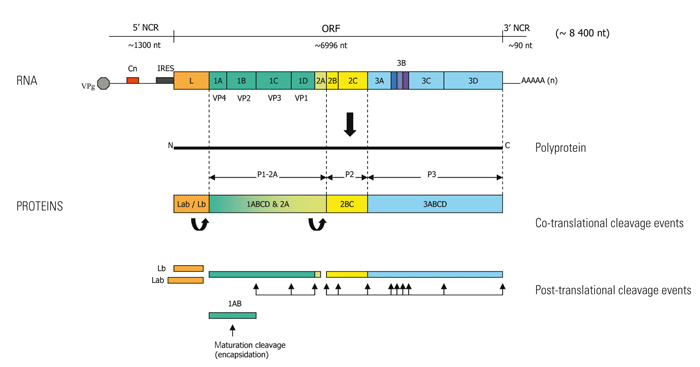
The viral RNA comprises a single plus (messengersense) strand, approximately 8 400 nucleotides (nt) long, consisting of a 5’ non-coding region (NCR), a single large open reading frame (ORF) approximately 6 996 nt long and a short 3’ NCR of about 90 nt (Figure 125.2). There is a small virus-encoded protein, VPg that is covalently attached to the 5’ terminus and a genetically encoded poly (A) tail at the 3’ end. The 5’ NCR is approximately 1300 nt long and consists of an S fragment (about 400 nt long), followed by a homopolymeric tract consisting of cytidyl (C) residues, 150 to 250 nt in length, referred to as the polycytodylic or poly C tract. Downstream of the poly C is a 720 nt region containing inverted repeats which are predicted to form pseudo-knots.69The internal ribosomal entry site (IRES) which spans the 435 nt region immediately upstream of the first functional AUG initiation codon and which contains several non-initiator AUG codons, also occurs within this region.38, 201 Both the IRES and the 3’ NCR have highly ordered structures and antisense RNA corresponding to these NCRs is known to inhibit FMDV translation and formation of infectious virus particles.146 Furthermore, virus attenuation has been associated with a shortening of the poly (C) tract,153 whilst deletion of the 3’ NCR abrogates virus replication presumably because this region contains major cis-acting signals that are essential for negative strand RNA synthesis.282
The large ORF of the virus genome encodes a single polyprotein, 2332 amino acids long in type O viruses,118 which yields 12 different products following proteolytic processing by viral proteases (Figure 125.2). The aphthovirus polyprotein is composed of four distinct regions, the Leader (L), P1-2A, P2 and P3 and differs slightly from the L, P1 (4 proteins), P2 (3 proteins) and P3 (4 proteins) or L434 convention which is strictly applicable for other picorna -viruses.272 This is because the FMD capsid protein precursor P1-2A consists of five rather than four proteins and arises from 2A/2B cleavage instead of P1/2A cleavage as occurs in most other picornaviruses. The five proteins constituting the FMD protein precursor P1-2A are 1A (VP4), 1B (VP2), 1C (VP3) and 1D (VP1), and the short 2A oligopeptide (Figure 125.2). VP1-4 encode structural proteins, whilst the remaining three coding regions, L, P2 and P3 are precursors for non-structural proteins that play a role in protein processing and RNA replication. L and 3C are proteases, with 3C being the major viral protease and also playing a role in shutting down host cell transcription through cleavage of host cell histone H3.109 3D encodes the RNA-dependent RNA polymerase (replicase) and 3B encodes three non-identical, functional copies of VPg, a feature unique to FMDV.110, 211 The exact functions of 2B, 2C and 3A are unknown; but length and sequence variation in the 3A region of serotypes O and C FMDV has been linked to differences in pathogenesis.34, 198, 239
Serological differentiation between animals that have been vaccinated and those that have been infected, which is important for trade in animals and animal products (see Control), uses the fact that inactivated vaccines that have been at least partially purified induce antibodies only against the viral structural proteins exposed on the surface of the virion (VP1-3) and to the polymerase (because each virion, on average, contains one polymerase molecule). The identification of antibodies against non-structural proteins therefore provides good evidence that the animal concerned has been infected by FMDV in the past (see Diagnosis).
On entering the cell, replication and translation of the FMD viral genome occurs in the cytoplasm. Delivery of an intact genome is essential as the viral RNA acts as mRNA for the host cell translation machinery and as a template for replication of RNA by the virally encoded RNA polymerase. Viral RNA is translated into a large polyprotein that is sequentially cleaved by proteases into the structural and non-structural proteins.334 Proteolytic processing occurs rapidly and probably whilst translation is in progress as the complete polyprotein has never been observed.
Initiation of translation of the FMD viral genome directed by the IRES occurs by a cap-independent mechanism.201 Following ribosome recognition of the IRES, initiation of FMD VRNA translation starts at one of two functional in-frame AUG codons that are separated from each other by 84 nt.35 Two forms of the L proteinase occur, namely Lab and Lb, with the latter truncated version of the L proteinase arising following initiation of translation at the second AUG start site.
The first polyprotein processing event is the co-translational cleavage of the L/P1 by the L proteinase. Both forms of the L protein can cleave the L/P1 junction and both ensure the proteolytic degration of eIF4G, the cellular cap-binding protein complex.229 Following this primary processing event, co-translational cleavage by 2A occurs at the 2A/2B junction to deliver a P1-2A product.279 Post-translational processing of P1 is mediated by the major viral protease 3C to produce 1AB (VP0), 1C (VP3) and 1D (VP1). The 3C also cleaves translation initiation factors eIF4AI and eIF4G in mammalian cells, with the former being a feature apparently unique to FMDV infected cells and occurring from about three hours post-infection.39 The final cleavage event is directed at VP0 (VP4 + VP2) and only occurs on encapsidation of the RNA. Prior to this maturation cleavage of VP0, a myristate moiety is attached to the N-terminus of 1AB that appears to be critical for capsid stability.68
Shortly after infection, host cell transcription and translation is inhibited mainly through the effects of the virally encoded proteases on the cellular protein and RNA synthesis machinery. In contrast, viral RNA synthesis in the cytoplasm proceeds rapidly to give rise to a pool of structural and non-structural proteins. Progeny RNA, produced through a complementary minus (negative sense) strand, and the structural proteins are then assembled into new virions that are released by infection-mediated disintegration of the host cell.112, 271 In cell cultures FMDV may produce 100 000 virus particles and the time between infection and the liberation of new virus can be as short as six hours.
Both the single-stranded, positive-sense RNA and the double-stranded, replicative form (RF) were shown to be infectious for baby hamster kidney (BHK) cells, following microinjection of the RNA into the cytoplasm.47 The infectious nature of theRNAhas been used in the laboratory to develop and test genetically modified genomes where infectious RNA transcripts are derived from bioengineered cDNA.209, 260, 281, 356
These artificial transcripts are also infectious, but to a lesser extent than the viral RNA that has cellular replication complex proteins associated with it that are believed to form part of the RNA packaging signal.144, 236
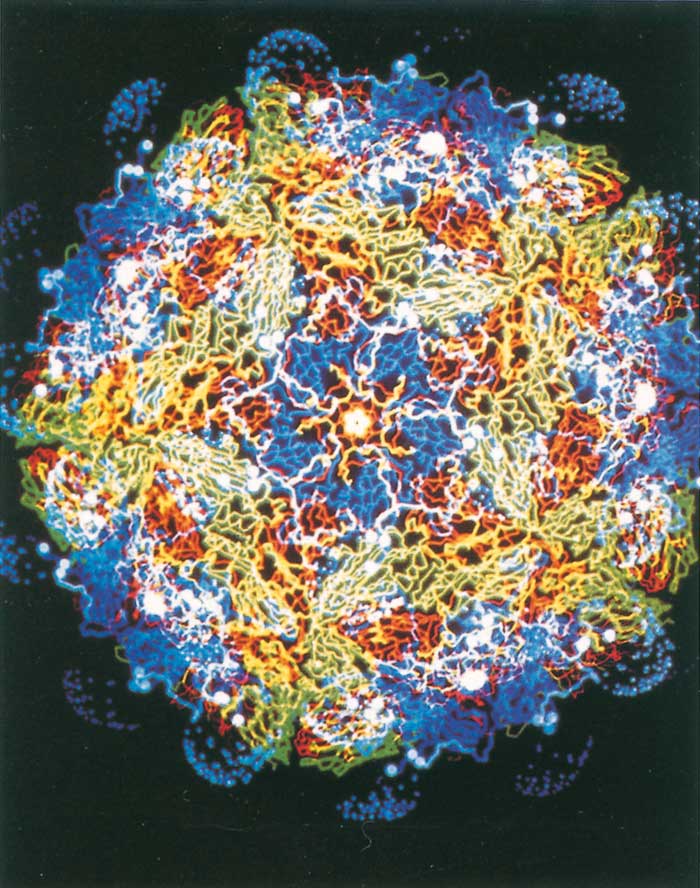
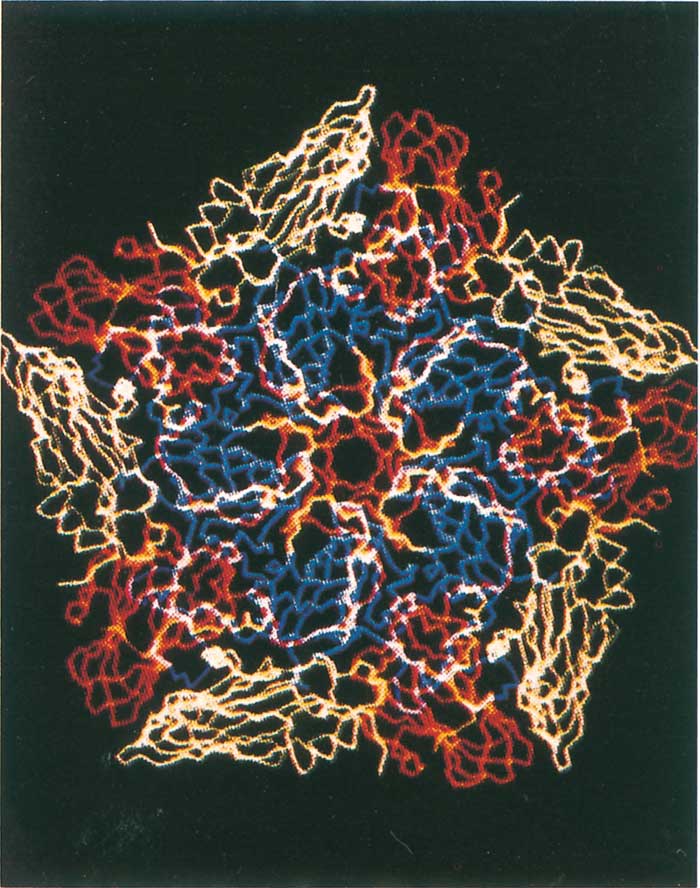
The virion capsid is made up of 60 asymmetrical protomers (poorly defined capsomers), containing one molecule of each of the three surface-exposed proteins (VP1–3), and an internal VP4-containing N-terminal myristic acid.68 A few protomers within each capsid are immature and contain VP0 instead of VP2 and VP4.52, 271 Pentamers are formed when five protomers associate and ultimately give rise to a capsid consisting of 12 pentamers and incorporating a single copy of genomic RNA. Each virion on average also contains one molecule of RNA polymerase, sometimes referred to as virion infection- associated antigen (VIA or VIAA).52 Some cell culture harvests contain virus particles that lack RNA, and their protomers do not contain VP2 and VP4, as the VP0 is uncleaved. These particles have a sedimentation rate of 75S instead of 146S and are referred to as ‘empty particles’.
When the virion is disassembled by mild denaturation (such as heat or acid treatment), pentameric subviral particles with a sedimentation coefficient of 12S (Figure 125.3b) are obtained.52, 271 Unlike the 146S and 75S fractions, 12S pentamers do not elicit an effective immune response, i.e. neutralizing antibodies. For this reason vaccines containing 146S or 75S particles are only effective while the particles are intact. Disaggregation into 12S pentamers results in loss of effective immunogenicity. Therefore, determination of post-inactivation 146S content of vaccine-virus harvests is vital in predicting the potency during vaccine manufacture (see Control).
Generally, picornaviruses are stable across the pH 3 to pH 9 range; but FMDV is distinguished from other picornaviruses by its narrow pH stability range (pH 7 to pH 9). The acid-lability of FMDV is essential for efficient uncoating and endocytic entry as agents preventing acidification of endosomes block infection.63 Another characteristic feature of FMDV is the relatively high CsCl density (1,41 to 1,45 g/ml), which can be ascribed to the penetration of caesium ions via the highly hydrophobic hole at the 5-fold axis.1
The surface-exposed VP1, VP2 and VP3 share an eightstranded antiparallel beta-barrel configuration and differ from each other primarily in the loops connecting the beta-barrel segments.246 By convention, the eight betabarrels are labelled B-I with the variable loop regions being denoted by the letters of the two beta-barrels they connect. Amino acid alignment of FMDV with other picornaviruses indicates that the loop regions connecting the beta-barrels in VP1 and VP2 are truncated, particularly in the regions contributing to the pentameric apex of the virion where poliovirus and rhinovirus have major antigenic sites.231 In contrast to other picornaviruses, FMDV has a relatively smooth capsid and contains none of the ‘canyons’ or ‘pits’ that are believed to render the residues involved in cell-attachment inaccessible to the host immune system.
Instead, the cell attachment site of FMDV is located on an exposed, disordered loop that protrudes from the surface of the capsid (Figure 125.3a and b)1, 2 This multifunctional domain not only contains the highly conserved Arg-Gly- Asp (RGD) cell-attachment site, but is also highly immunogenic as monoclonal antibody (Mab) studies have shown that the G-H loop of VP1 elicits neutralizing antibodies in six of the seven FMDV types studied thus far.77, 196, 207, 218, 283 Thus the conserved cell-attachment site of the virus which is known to bind many extracellular ligands to integrin cell surface receptors273 is flanked by hypervariable sites delivering a domain that is capable of adopting different conformations. Presumably this situation ensures access to cells by the virus but, at the same time, enables it to elude the immune response of the host.267, 268 Unfortunately, the flexibility of the loop region has also meant that resolution of the loop structure eluded researchers until recently. Using different approaches including synthetic peptides and chemical reduction of disulphide bonds, the loop structure of types O, A and C viruses was resolved.120, 212, 341 Results from these studies indicate that the loop structure is similar across types with the RGD adopting an open turn conformation and being flanked by a beta-sheet and alpha helix on the amino and carboxy sides, respectively. Thus despite the low G-H loop sequence identity observed both within and between serotypes, structural similarities appear to remain intact.
The importance of the RGD site for interaction of FMDV with cellular receptors is well-established.33, 119, 223 However, it was also shown that viruses of different subtypes are able to use different cellular receptors32, 294 and that cell-culture adapted variants that do not contain RGD are able to replicate normally.221 Thus FMDV is able to gain entry to cells by using different receptors. The use of multiple receptors by the virus has been confirmed by independent studies and all receptors thus far identified can be categorized into two main types, namely the RGD-dependent integrins and heparan sulphate proteoglycans (HSPG).179, 181, 235 These distinct cell-surface receptors are associated with virulence and loss of virulence following cell culture adaptation, respectively.281
The amino acids flanking the RGD, particularly at positions –1, +1 and +2 are known to affect integrin binding affinity and specificity although the relative importance of each of these flanking sites may vary between types.223 Complete conservation at sites flanking the RDG was observed at positions +2 and +4, –1 and +4, and –1 and +4, for diverse field strains of SAT1, SAT2 and SAT3 types of the virus respectively.28
Although the residues present at the –1 position differed between types (SAT2 subtypes had a positively charged arginine and those of SAT3 had a non-polar isoleucine), the +4 residue for different isolates of the three SAT types was exclusively leucine.
Despite the considerable levels of antigenic and genetic variation found within each of the types, arising from the highly variable and adaptable nature of FMDV, all FMDV isolates can be grouped into one of the seven serological types. The high levels of variation observed for FMDV types arise from the error-prone replication of viral RNA due to the absence of proofreading by the viral replicase and from selection imposed by functional and structural constraints that are continuously acting on the genome.91 The inherent variability coupled with short generation times, results in RNA evolution rates that can be a million-fold greater than that observed in the DNA of their hosts.166, 302 Rates of mutation have been quantified both in vivo and in vitro for different FMDV types, with the rate of accumulation of mutation being between 0,9 × 10−2 and 7,4 × 10−2 substitutions per nucleotide per year (s/n/y) for European type viruses recovered from persistently infected cattle,131 and between 1,54 × 10−2 and 1,64 × 10−2 s/n/y in SAT-type viruses in persistently infected African buffalo.342 This implies at least one substitution error per genome replication cycle and gives rise to a population of phylogenetically related variants in accordance with the quasispecies concept.89, 90 Thus, RNA virus populations are a dynamic equilibrium with mutants arising at a high rate on the one hand, but being selected against on the other.91 Selective forces acting on specific capsid protein sites have been identified in sequences generated for both field and laboratory-adapted strains.111, 152 However, antigenic variants can also arise in the absence of immune pressure.88
Another important variation-generating mechanism is homologous recombination which occurs at a relatively high frequency in picornaviruses and coronaviruses. Intratypic and intertypic FMDV recombinants have been identified both in vivo and in vitro.136, 179, 199, 224 However, the limited evidence of intertypic recombination in the field may be due to the lower probability of co-infection of animals with unrelated strains. In Africa where six types and numerous subtypes occur, and where buffalo are known to be co-infected with different types,155 recombination is more likely and indications for the occurrence thereof have recently been forwarded.339
One of the consequences of generation of genetic variation through mutation, selection and recombination is that new FMDV antigenic variants are constantly being generated, a factor which has significant implications for selection of appropriate vaccine strains.16, 172, 222, 276 Historically, the occurrence and range of intratypic variation was described on the basis of subtypes. Conventionally, subtyping was based on cross-neutralization or complement fixation (CF) tests which generally show good correlation with cross-protection in vivo.253 The latter technique, however, waned in popularity as the CF reaction is not exclusively dependent on antigens that are relevant to immunological protection.249, 250, 251More recently, development of techniques for the analysis of RNA genomes has enabled more accurate determination of antigenic variation of FMDV. However, variation based on genome sequencing and variants established by antigenic analysis differ and are not directly comparable (see below).
Initially over 60 different subtypes were identified by the World Reference Laboratory (WRL) for FMD, with the majority of discernible subtypes being identified for types A and O (such as A5, A24, O1, O5 and others).253, 262 It was, however, difficult to identify subtypes within the SAT types107 and the concept of a continuous spectrum of intratypic antigenic variation (without there necessarily being a clear distinction between variants) has become established instead,253 leading to the abandonment of subtype classification. Instead the practical significance of the r-value was recognized.277 and is used to provide a measure of antigenic similarity between two strains. The r-value is defined as the ratio of the serum titre against a heterologous strain (virus A) to the serum titre against the homologous strain (Virus B).275
In practice, the heterologous strain is usually an outbreak strain and the homologous strain a vaccine strain, with the r-value providing a measure of similarity between the two and therefore a measure of vaccine strain suitability for a given outbreak situation. The ‘r-value’ may be either r1 or r2 or R depending on the serological tests performed using antisera derived from animals that were either infected or immunized with the viruses in question.
Thus,

R is therefore the composite value expressed as a percentage but, because asymmetric relationships are common, R is often misleading.251 Since the ability of a vaccine strain to induce an immune response which protects against the ‘new’ outbreak strain is what is important in practice, r1 values provide the most valuable information. Generally, an r1 value greater than 0,4 is indicative of a satisfactory antigenic relationship and confirms suitability of a vaccine strain, whilst r1 values below 0,4 reflect a weak to poor antigenic relationship between the outbreak and vaccine strain. These cut-offs are based on the correlation between the serum neutralizing antibody titre and protection from challenge in cattle277 and should be taken as guidelines of vaccine strain suitability rather than as absolute values.
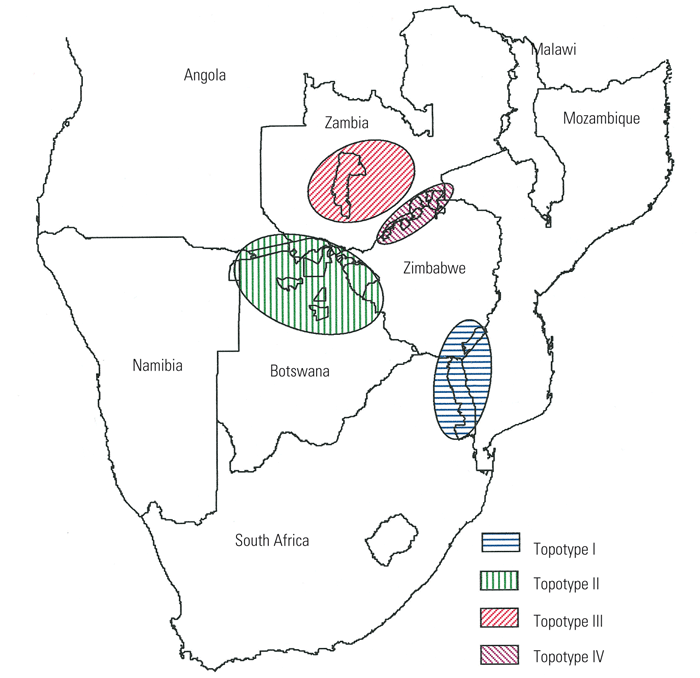
Although it does not provide a direct measure of immunological relationships, sequencing part of the VP1 gene of FMDV is useful for determining intratypic variation of FMDV, for identifying epidemiological links between viruses and for studying evolutionary processes operating on the FMD viral genome. Genetic distance between two FMDVs, as reflected by VP1 gene nucleotide sequences, is used as a measure of genetic relatedness with those differing by less than 5 per cent from each other being considered part of the same epidemic.259 Although this measure appears to be applicable across all FMDV types, the demarcation of genotypes differs between types, with SAT types having higher genetic divergence values than those observed for the European types.16, 26, 27, 29, 30, 220, 289, 344 The higher levels of variability within the SAT types of virus most likely reflect their apparently longer period of existence as the 1D phylogeny incorporating viruses representative of the seven types (Figure 125.1) indicates that the three SAT types arose earlier than the European and Asian types and therefore have had more time to diversify. In addition to supplying insight into type ancestry, VP1-based phylogenies reveal that different genotypes within the O and SAT types evolve in discrete geographical regions, with these variants being referred to as topotypes. 27, 30, 289 Grouping of viruses according to geographical origin provides a powerful tool for determining the origin of outbreaks and for tracing the course of an infection, as exemplified by the emergence and spread of the pan-Asian virus (type O) that caused the outbreaks of FMD in the UK in 2000.288 VP1 gene sequencing has also been instrumental in identifying transboundary and transcontinental virus transmission, for confirming or refuting the involvement of vaccine administration in causing FMD outbreaks and for identifying prolonged persistence of particular virus variants in the field.29, 36, 127, 199, 217, 248, 290
Since the advent of polymerase chain reaction (PCR)- sequencing of FMD viral genomes, a comprehensive database of virus variants distributed throughout the world is becoming established, with many sequences readily available in the Genbank database for comparative purposes. As more and more sequences become available, the ability to determine the exact origin of the virus causing an outbreak is achieved with greater accuracy. This vast database also provides a valuable resource for designing and refining PCR-based methods used for the rapid detection and characterization of the virus recovered from an outbreak.
Epidemiology
Distribution and occurrence
At present, most of the major livestock-producing regions of the world are free of FMD: North America, Western Europe, Australia, New Zealand and some parts of South America. Up-to-date situation maps with respect to countries or zones (i.e. portions of countries) that are free from FMD are available on the website of the OIE (www.oie.int).
In South America rapid progress was made during the 1990s towards eradicating FMD from major livestock producing areas of the so-called southern cone covering Chile, Uruguay, Argentina and southern Brazil. However, resurgence of the disease in 2000/1 in these areas, with the exception of Chile, at least temporarily reversed the progress. This experience, together with re-incursion of infection into countries long free of the disease such as Taiwan (1997), Japan (2000) and South Korea (2000/2) in southeastern Asia, and the UK, Ireland, France and the Netherlands (2001) in western Europe, has shown that initial optimism over the success of eradication programmes was exaggerated. These events demonstrated the difficulty of keeping FMD permanently out of countries that import large quantities of livestock and their products because the infection is still widespread in developing regions of the world. This includes much of northern South America, most of Africa, the Middle East, south and south-eastern Asia, and parts of central and eastern Europe. Much speculation on the prevalence of FMD on mainland China has been prevalent for some years because, although there are no official reports of the disease in that large and populous country, unofficial rumours abound including the assertion that attenuated vaccines against FMD are or have recently been used there. The fact that China has now joined the WTO is likely to result in the situation improving and becoming clearer. The Indian subcontinent is another region that harbours a major reservoir of infection because of the very large livestock population and wide distribution of types O, A and Asia-1. There is, however, a clear intention by countries that comprise that region to address the problem more effectively than in the past.
The geographic distribution of the seven virus types is heterogeneous. Types A and O are prevalent in South America, south-eastern Europe, southern and south-eastern Asia, Africa and the Middle East. Type C at one time had a similar distribution but now occurs infrequently and in recent years has only been recorded in Kenya. Currently, type O has the widest distribution and prevalence of all types.188, 189 A particular phenomenon has been the recent dramatic spread of what has become known as the pan-Asian lineage of type O, first detected in Northern India in the early 1990s.289 It was next detected in the Middle East and then spread to the Far East where Taiwan, South Korea and Japan suffered serious outbreaks. It reached the Balkans in 1996 and in September 2000 arrived in South Africa via a ship’s galley waste fed to pigs on a farm in the Camperdown district in KwaZulu-Natal.290 Most dramatically, an almost identical virus arrived in the UK in February 2001 and subsequently spread to Ireland, France and the Netherlands.89 In Figure 125.4 the spread of this topotype over a 10-year period is shown. Asia-1 was confined to the Far and Middle East and the Balkans but in 2000 occurred in south-eastern Europe (Greece) for the first time.199
The three SAT types are restricted to sub-Saharan Africa, although there have been incursions of SAT1 into North Africa and the Middle East in 1962 and 1970.115, 254 In 1990 SAT2 was recorded in the Republic of Yemen115 and in 2000 SAT2 spread to Saudi Arabia and Kuwait.3
In Africa, as indicated above, six of the seven FMDV types occur and the reported distribution of outbreaks by country and type since 1948 has been reviewed.346 There is no doubt, however, that FMD is severely under-reported in the continent and therefore the available information is incomplete. Nevertheless, it is clear that types O, SAT1 and SAT2 are widely distributed while types A, C and SAT3 are less so.346 In fact, disease in livestock caused by SAT3 has not been reported outside southern Africa despite having been identified in buffalo in East Africa in the 1970s.161 Of the three SAT types, SAT2 has the highest prevalence in domestic animals: between 1900 and 1987 it was the cause of 48 per cent of out breaks in livestock in southern Africa that were typed. SAT1 and SAT3 accounted for 36 per cent and 16 per cent respectively. 322 In the last ten years (1992 to 2001) the causative virus in outbreaks reported to the OIE from southern Africa — much reduced in comparison with former 10-year periods — were almost equally divided between the three SAT types.57, 346 The reason for this remains to be determined.
Conversely, types other than SAT1, 2 and 3 have not occurred in the southern-most part of the African continent (South Africa, Namibia, Botswana and Swaziland) except when types A and O were imported from their northern neighbours into respectively Namibia (1958, 1962, 1968) and Zambia (1976, 1982 to 1984);322 or by intercontinental spread such as occurred most recently when the pan-AsianOlineage caused an outbreak in KwaZulu-Natal in September 2000 (Figure 125.5).290 Circumstantial evidence indicates that type O viruses causing outbreaks in Mozambique in the 1970s and 1980s were related to cattle imports from South America.322 Retrospectively, the role of cattle importation in outbreaks in southern African countries has been confirmed on the basis of a molecular epidemiological study where it was shown that the type O virus recovered from outbreaks in Angola in 1974 and 1975 most likely originated from South America.290 Similarly, type A viruses of European–South American origin were introduced into two different southern African regions, namely, Angola (1973) and Malawi (1974 to 1975).197
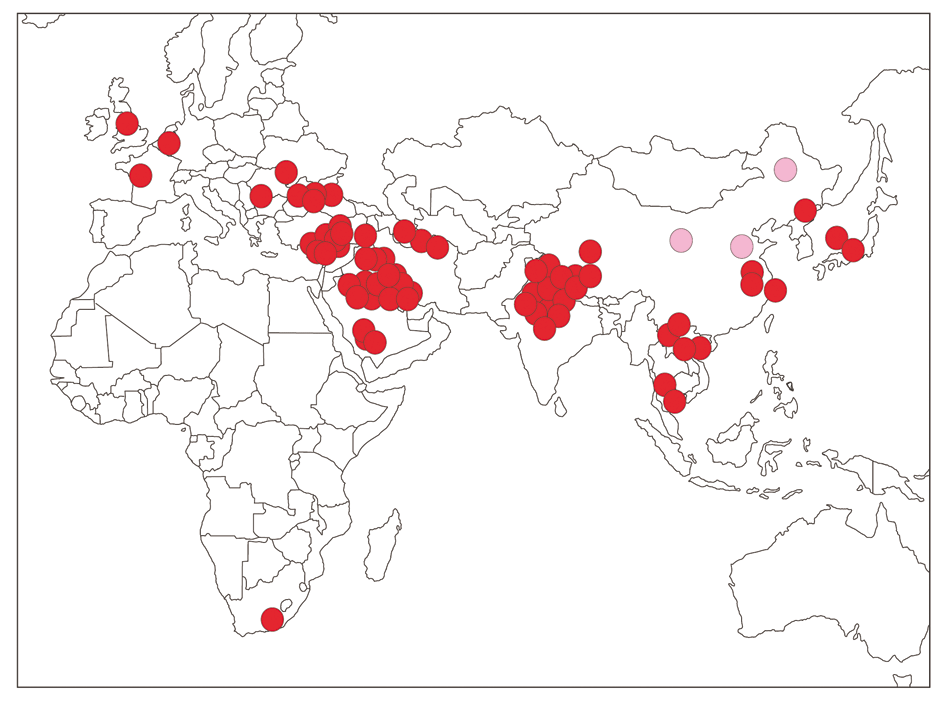
Figure 125.4 Spread of the South Asian (now commonly referred to as the Pan-Asian) topotype or lineage: 1990–2001. Light-coloured circles denote instances where the virus concerned was not characterized but where there is epidemiological evidence for the virus concerned being a member of this lineage. (By courtesy of the Food and Agriculture Organization, Rome)
The origin of the other A, O and C viruses that caused many outbreaks of FMD in livestock in Angola between 1958 and 1975115, 322, 346 is unknown but may have been at least partly derived from Europe bearing in mind the then colonial relationship between Angola and Portugal and the high prevalence of FMD in western Europe at that time.
Molecular epidemiology
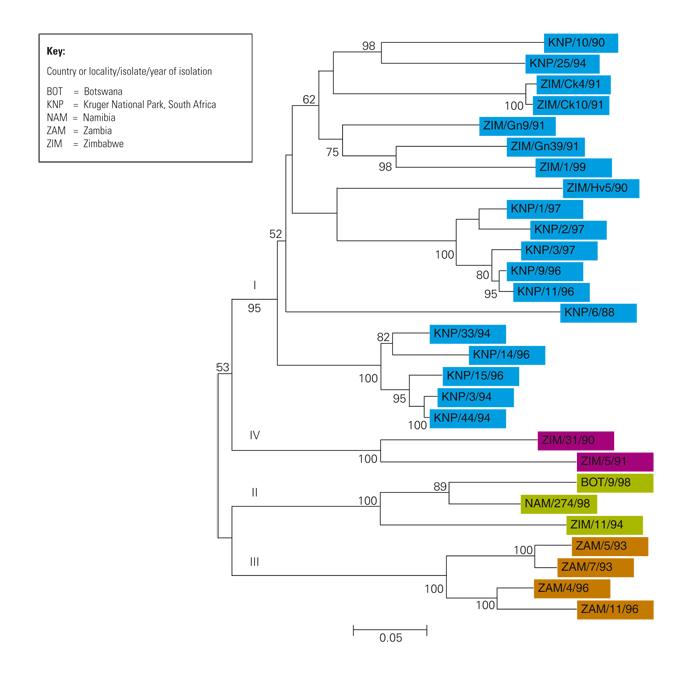
It is increasingly apparent that to understand the epidemiology of FMD it is essential to be able to differentiate between intratypic variants because outbreaks of FMD caused by the same type may constitute epidemiologically unrelated events. An example occurred in Zimbabwe in 1989 when two outbreaks caused by SAT2 viruses were detected within a few days of each other — one at Mutorashanga in Mashonaland and the other around Gweru in the Midlands. Sequence analysis of portions of the 1D genes of the two viruses concerned showed that, although they were of the same lineage, they differed by more than 5 per cent over the region sequenced, indicating that the two viruses had different origins.48, 322
Statistical comparison of nucleotide sequences over a 200 to 500 base region covering the 3’ end of the 1D and a portion of 2A genes of FMDV, has now become a standard method for deriving relationships between FMD viruses within the same type.240 Using this approach on SAT viruses derived from African buffalo in wildlife reserves in southern Africa has revealed that all three SAT types have evolved, and continue to evolve, independently in different geographic areas.29, 30, 342, 344, 345 These lineages or topotypes (see Aetiology) are clearly distinguishable. This basic information has permitted the geographic origin of buffalo to be traced using the viruses isolated from them343 and also enabled the source of infection to be identified in FMD outbreaks in cattle and impala (Aepyceros melampus) populations in contact with African buffalo. 30, 330
The same approach has identified lineages and topotypes in other regions of the world and enabled their differentiation, the most famous of which—the pan-Asian O lineage288, 289 — has been mentioned above.
Impact of foot-and-mouth disease
A remarkable feature of FMD is that, although the clinicopathology of the disease varies little from one geographic region to another, the impact of the disease differs considerably in different parts of the world. In the developed world it is the most feared of all animal diseases with the possible exception of those that have zoonotic potential such as bovine spongiform encephalopathy. The reason is the devastating economic consequences it can have. Most recently this was illustrated by the 2001 UK outbreak where, within three months, it resulted in the destruction of over three million animals, cost the tourist industry £5,2 billion and caused considerable human misery.319
Ultimately, more than six million animals were culled to ‘stamp out’ the disease.293, 318 Conversely, in many pastoral communities in sub-Saharan Africa where the disease is prevalent and well recognized by livestock owners they often — although not always — ascribe little importance to it. The simple reason for this difference is that in extensive production systems dependent on ‘unimproved’ livestock, the disease is usually relatively trivial and the animals recover uneventfully in a week or two. The loss of production in such systems is usually unremarkable although the disease may result in a lack of draught power for ploughing at critical times and therefore results in agricultural and social disruption. It may also affect production by the growing peri-urban, small-scale dairies that are developing in many parts of Africa.
The lower impact of the disease in sub-Saharan Africa may also, to some extent, be due to indigenous cattle having greater inherent resistance to the disease than breeds selected in developed countries for high yield in milk or beef production. In high performance animals, although few adult animals die (pigs have been an exception in some outbreaks) as a result of the infection, production losses may be dramatic and, especially in dairy cattle, long lasting.190, 191 Other reasons for the high impact of FMD and difficulty in controlling it in intensive livestock systems are:
- the extreme rapidity with which the virus can spread over long distances and so infect large numbers of animals in a short period of time;
- immunological variants (types and subtypes) which make it possible for animals to suffer repeated attacks of FMD;
- a proportion (sometimes more than 50 per cent) of ruminants recovered from acute disease retain the infection in the pharyngeal region and subsequently may serve as a source of infection for susceptible animals in contact with them. The latter event occurs only rarely at best and some have argued that it is so infrequent as to be epidemiologically insignificant. On the other hand, the repeated outbreaks caused by SAT2 viruses that occurred in Zimbabwe in the 1980s and which were traced back to carrier cattle, provide strong circumstantial evidence for carriers being important in propagating the infection in some circumstances;324 and
- in sub-Saharan Africa, especially in southern Africa, unlike elsewhere in the world, wildlife is important in the propagation and persistence of FMDV (particularly the SAT types).
Each of these factors is covered in more detail below.
Transmission
Foot-and-mouth disease virus may spread by either direct or indirect contact.177, 278, 296 Most commonly, it occurs by direct contact between animals excreting virus and susceptible individuals.349 Infrequently, indirect transmission is effected by objects or materials contaminated with virus-containing secretions, excretions or tissues or by animal products, such as milk, or by air currents in which virus-containing aerosols are suspended. People, animals, vehicles, and birds may serve as mechanical transmitters of the infection,296 although doubt has been expressed about birds.264 Analysis of the mechanisms of farm to farm spread during the UK outbreaks that occurred in 1967 and 2001 has revealed that what has been termed ‘local spread’ (within a radius of 3 km) accounted for the majority (about 80 per cent) of secondary outbreaks. However, although infected aerosols are thought to have accounted for most of this type of spread there is no certainty on the issue.270, 292
Strong circumstantial and theoretical evidence for long distance (greater than 1 km) air-borne transmission of FMD outbreaks in northern Europe has been reported.93, 116, 137, 176
The most impressive contemporary example occurred in 1981 where cattle on the Isle of Wight (UK) were infected by windborne aerosols produced by infected pigs in Brittany (France) and carried across the English Channel over a distance of more than 250 km.193 Models that predict the direction and distance of spread from a primary source are now available.304 However, no convincing evidence for the involvement of air-borne transmission in other parts of the world, including sub-Saharan Africa and South America, has so far been advanced.
As is the case with other directly transmitted infectious agents, there are three factors that determine the characteristics of FMD transmission. These are:
- the quantity, duration and means by which virus is liberated into the environment;
- the ability of the virus to survive outside the animal body; and
- the quantities of virus required to initiate infection at the primary infection sites of animals that are exposed.
Virus excretion
With the exception of oesophago-pharyngeal secretions, measurable quantities of FMDV are present in animal secretions and excretions for less than two weeks after infection.130 In viraemic animals, FMDV is present in most physiological fluids and hence potentially in all secretions and excretions.177 There are, however, differences in this respect between virus strains, host species and different secretions and excretions.94, 322 The fact that FMD in southern Africa has often spread more slowly than would be expected in Europe106, 108, 204 is not as a result of the SAT virus types being excreted at lower rates than is the case for the ‘European’ types.322
Significant excretion may occur up to four days, or perhaps longer, before the appearance of lesions, so that apparently healthy animals can be an important source of infection.61, 177, 308 In this regard milk is of particular concern because of the rapid dispersal of the product and the movement of bulk tankers between farms, in addition to the fact that milk may contain up to 105,5 infectious doses/ml.322
Domestic pigs are the most efficient excretors of FMDV into the environment. Individual pigs excrete 105,8 to 108,6 TCID50 infectious doses of virus into the atmosphere per 24- hour period.98 There is obviously considerable variation between viruses in this respect because these values differed by a 600-fold factor in the studies conducted.98 Since pigs are often intensively farmed, the occurrence of disease in piggeries may result in enormous multiplication of infectivity but this is not always so. This is exempified by the differences between the 1967 and 2001 FMD outbreaks in the UK: in the former outbreak, air-borne infection derived from pigs was an important factor in transmission while in the 2001 outbreak this was not so.270 Long-distance air-borne spread of infection from primary outbreaks in Europe has occurred only rarely other than where pigs were the predominant source of infection.95
Peak excretion of air-borne virus by cattle and pigs coincides with development of early clinical disease whereas in sheep peak levels generally precede development of clinical disease.98 Aerosol excretion in pigs generally lasts for three to five days.
Virus-containing aerosols are probably derived from the upper respiratory tract102 as they are present in the breath of pigs. The particle size of such aerosols range in diameter from about 3 to 10 μm, the majority being greater than 5 μm.273 Most infectivity is associated with droplets larger than 6 μm.104 Because aerosols are subject to evaporation and may coalesce, the particle size structure is not static,273 and it may be that aerosols smaller than 3 μm are rapidly destroyed by atmospheric conditions.298 Should meteorological factors be suitable for virus survival in aerosols (see below), their fall-rate is such that they remain air-borne long enough to be conveyed for more than 10 km from the source by air currents273 and, rarely, for up to hundreds of kilometres.93
Of all the secretions and excretions in acutely infected animals (certainly in cattle and pigs), saliva contains the highest concentrations of virus: up to 108,5 infectious doses/ml in cattle.322 It has been pointed out that when infectivity levels in saliva exceed 107/ml they could contribute to virus-containing aerosols as well as cause gross contamination of the immediate environment.176, 177 There is a non-specific viral inhibitor (dialysable inactivating factor) in the saliva of cattle, which limits virus survival outside the body94, 101 but this aspect has not been quantified.
Other secretions which contain appreciable quantities of virus are those of the nasal passages and the oesophago-pharyngeal region as well as the urogenital tract and milk.322 Semen, which in bulls may attain virus levels of up to 104,7 MLD50 prior to the development of visible lesions, is also a potential source of infection in both cattle and pigs.297 Artificial insemination is thus a potential avenue of transmission. In respect of embryo transfer, FMDV does not appear to penetrate the zona pellucida although it may be capable of infecting hatched bovine embryos.301 It has been shown by risk assessment that as long as the recommendations of the Import/Export Committee of the International Embryo Transfer Society are observed,15 the likelihood of such transmission is negligible.314
Both urine and faeces have been found to contain high levels of virus;187 in the case of urine this is for prolonged periods as a result of supposed persistent kidney infection. 165, 247, 348 These early reports of persistent kidney infection have not subsequently been confirmed. Levels in urine have reached 104,9 cell culture infective doses.187 However, the fact that urogenital secretions, particularly those of the preputium, may contain high virus levels,322 presents the possibility that virus in urine may be derived largely from such secretions. A number of investigators have found little or no virus in faeces;74, 129, 177 one hypothesis for this being that environmental contamination after defaecation is responsible for most of the virus detected in faeces.247
Nevertheless, faeces may contain up to 105 cell culture infective doses per gram.
Carrier animals
An aspect which is still unresolved after nearly a century of speculation177 is the ability of carriers to transmit the infection.59 Carriers are defined as animals in which FMDV persists in the mucosa of the soft palate, pharynx and cranial oesophagus for more than 28 days after acute infection.287 In effect therefore, carriers are apparently healthy animals in which the virus is shed in small quantities from basal epithelial cells of the pharynx and dorsal soft palate.355 Persistent infection occurs in cattle, sheep, goats, African buffalo, and other wild ruminants but not in pigs.50, 96, 131, 158, 177, 336 Like pigs, camelids apparently do not become carriers.81 More than 50 per cent of cattle that have recovered from infection, or that were vaccinated and subsequently exposed to infection — whether they develop clinical disease or not—become persistently infected.190 In cattle virus levels in the oesophago-pharyngeal region begin to decline soon after initial infection59, 104 and they may fluctuate.131, 336 By six months after infection, the presence of persistent virus becomes erratic131, 336 but virus may persist in up to 20 per cent of animals for a year155 and in some animals for as long as two years.155 A small proportion of cattle examined during a field investigation in Zimbabwe were found to be persistently infected three years after initial infection.287, 324 Sheep may remain carriers for up to nine months.287
Viral persistence occurs despite the occurrence of antibody in oesophago-pharyngeal secretions122 and superinfection with more than one type clearly occurs.228, 336
Circumstantial evidence indicates that persistently infected cattle are able to transmit FMDV occasionally,177, 336 but this has not been proven conclusively59, 310, 324, 336, 349 and is currently a point of major contention.24, 308 In many of the FMD outbreaks that occurred in cattle in southern Africa since 1931, aphthization or ‘firing’ (the deliberate infection of all animals present within the outbreak focus) was practised. Aphthized cattle were often allowed to move to disease-free areas within a few months of the end of the outbreak, i.e. quarantine restrictions were lifted after three to four months. Despite this, FMD control in South Africa, Botswana and Zimbabwe was historically successful, indicating that persistently infected cattle are not a regular source of infection.108
In vitro studies have shown that viral persistence possibly involves co-evolution of both the infecting virus and the host cells.84 A similar situation may obtain in the pharyngeal mucosa. Because every virus isolate is a fleeting representative of a continuously evolving population,131 and such transmission only rarely occurs, it is possible that infection derived from persistently infected cattle (or other species) provides a mechanism whereby dominant virus populations are periodically replaced.131
A theory that transmission by carrier animals is precipitated by stress developed following indication that such a situation was responsible for FMDV transmission from a carrier African buffalo to cattle in Zimbabwe.159 However, experiments based on the effect of dexamethasone administration to carrier cattle did not support the theory.178 In fact, dexamethasone administration had the unexpected effect of causing the persistent virus to become undetectable in probang specimens obtained from treated carriers. The virus re-appeared as soon as dexamethasone administration ceased. In another experiment, carriers were infected with rinderpest virus which destroys T cells but this did not result in increased levels of viral recovery from the FMDV carriers. Similarly, experimental infection of cattle persistently infected with FMDV with bovine herpesvirus 1 (the cause of infectious bovine rhinotracheitis) also failed to precipitate transmission of FMDV to susceptible cohorts.227
Transmission of FMDV by livestock carriers therefore appears, at most, to be an extremely rare event. Conversely, where African buffalo are concerned, such transmission both to in-contact cohorts and cattle has been clearly demonstrated (see The role of wildlife, below).
Virus survival outside the living animal
Foot-and-mouth disease virus is very labile in even mildly acid solutions. At pH 6,0 the rate of inactivation is 90 per cent per minute while at pH 5,0 it is 90 per cent per second.18 In mildly acidic conditions FMD virions disintegrate into their constituent 12S subunits (pentamers) liberating RNA. Although the latter is infectious, environmental ribonucleases will generally rapidly inactivate naked RNA.19 The virus is also labile in alkaline solutions; at pH 10 its loss of infectivity is 90 per cent every 14 hours.18 For this reason, 2 per cent NaOH or KOH and 4 per cent Na2CO3 are used as cheap and effective disinfectants. They are generally more effective than acid solutions because they are not as easily neutralized by organic material such as faeces and blood.
Lactic acid formation in skeletal muscle after slaughter, where the pH usually drops to pH 5,5 to 6,0 within 48 hours at 4°C, renders meat free of infectious virus.75 However, in lymph nodes, which are not subject to acid formation, the virus can persist for at least 120 days at 4°C.74 The same applies to bone marrow and blood in large blood vessels. Details on the occurrence and persistence of FMDV in meat and meat products has been published.14, 43, 44
There is variation between virus strains so far as their lability at low or high pH is concerned. Although inactivation is rapid at first, a residuum of infectivity may remain after exposure to extreme pH as is the case with exposure to high temperature.19, 20 Foot-and-mouth disease virus may also be protected from the effects of pH by chemical agents,18 and in dairy products by the association of the virus with a variety of cell structures and casein micelles.44
In milk from FMD- infected cows, the virus survives in small quantities in casein after iso-electric precipitation from skim milk at pH 4,6, in butter ripened with lactic acid cultures at pH 4,6, in cheese at pH 5,1, and in sweet whey at pH 5,2.44 However, the levels of persisting virus are low and of doubtful epidemiological significance (see below).
In contrast to the effects of pH, FMDV is relatively resistant to heat inactivation although there is considerable variation between virus stains.19, 86 In the case of primarily infected milk this resistance is extreme.350 The reason for this is at least partly the association of the virus with cell and fat structures.44 The virus can only be inactivated with certainty by exposure of milk to low-temperature pasteurization for periods in excess of 20 minutes, while 2,5 seconds is sufficient at 148°C.350 At 37°C, 146S particles lose 90 per cent of their infectivity within eight hours.55 Foot-and-mouth disease virus in infected ground beef products survived cooking to an internal temperature of up to 72°C but not 79,4 °C.44, 226
Foot-and-mouth disease virus in various dairy products subjected to heat and chemical treatments has generally involved the survival of only a small fraction of the original infectivity that has required highly artificial means to demonstrate. There is, therefore, some doubt — because the amounts of residual virus are likely to be below the threshold of infection required to infect pigs that may be fed such material—as to the necessity for the disruption of international trade which these results occasioned in the past.95, 97 In general, denaturation of protomer proteins is relatively rapid at temperatures above 43°C,18 while virus inactivated by prolonged incubation at 37°C apparently loses infectivity through disruption of the RNA without damage to the protein coat.19, 55
At temperatures of 55 and 61 °C, type A virus in cell culture fluid showed initial first-order inactivation followed by ‘tailing’, indicating the presence of a small thermal-resistant fraction.20 Hence, the common practice of heating sera and other products to 56°C for 30 minutes in order to render them free of FMDV has the disadvantage that a small fraction of infectivity may remain after such treatment. Whether the surviving fraction is biologically significant is doubtful.
Prevailing ambient temperatures have relatively little effect on the survival of FMDV in aerosols; at 27 °C high recoveries were achieved between 30 and 60 minutes after the formation of the aerosols.94
As is the case for most non-enveloped viruses, FMDV survives best in aerosols and microdroplets under conditions where relative humidity exceeds 60 per cent. There is a critical relative humidity range of 55 to 60 per cent, below which virus survival is poor,23, 92, 273 although the reason for this is not clearly understood.100 The fact that many regions of the developing world where FMD is prevalent are relatively dry, is probably an explanation as to why longdistance air-borne transmission of FMDV has not been identified outside western Europe.
The nature of the suspending fluid is important as far as FMDV survival in aerosols is concerned. Survival of virus in aerosols generated from saliva was much poorer than in those generated from milk, nasal fluid or faeces.95 This was ascribed to an organic antiviral molecule in saliva that is inactivated within three hours at 70°C, but not at 60°C.94
Detailed studies conducted earlier this century58 indicated that the long-term survival of FMDV on the surface of hay and bran, but not on other materials tested, was possible under conditions of relative dryness and absence of light. An unidentified rehydrating agent extractable from hay and bran was held to be responsible for this effect.
There is an ingrained belief in southern Africa that, because of the susceptibility of FMDV to ultraviolet light, FMD is less easily transmitted in that region than, for example, in northern Europe. Available evidence, however, suggests that this is not the case because FMDV has been found to be photoresistant to the ultraviolet levels in sunlight — albeit northern European sunlight.99
More detail on the interaction of FMDV with chemicals and disinfectants is available from review articles18, 295 and Simm’s300 bibliography.
Quantities of virus required to initiate infection
In cattle and sheep, infection with FMDV by the respiratory route can be achieved with as little as 10 cell culture infective doses.98, 105, 134 Conversely, from comparative titrations involving intradermalingual inoculation of sheep and cattle with five different FMDVs (four of them SAT types), sheep appeared to be less susceptible than cattle.132
Since infection by the oral route in cattle requires at least 10 000 times as much virus as by the respiratory route,60, 296 the respiratory tract appears to be the virus’s usual portal of entry in cattle (and probably also in sheep and goats). As indicated above, most infectivity in aerosols is associated with larger droplets104 which are likely to be predominantly trapped in the upper respiratory tract.
Cattle are efficient samplers of infected air because of their relatively large inspiratory volume (86 to 167 m3 per day for adults),296 and they therefore serve as the most efficient indicators of the presence of infection among domestic host species. Presumably for this reason the attack rate in the 1967 FMD epidemic in Britain increased with the density of the cattle population.170 Sheep, although having a similar minimum infectious dose to cattle so far as FMDV is concerned, are in practice less susceptible than cattle because of their considerably smaller inspiratory volume. The relatively low stocking rates for both cattle and small stock in sub-Saharan Africa are likely to be another explanation as to why long-distance air-borne transmission has so far not been reported in the subcontinent.
For pigs the situation is probably different in that the minimal infectious doses by both the oral and respiratory routes are relatively high.98 Recent studies 4–7 using several virus strains have shown that a pig may require up to 6 000 TCID50, i.e. as much as 600 times more than required by cattle105 or sheep,134 to cause infection. Furthermore, circumstantial evidence suggests that, in Western Europe at least, infection in pigs usually results from the feeding of contaminated and untreated swill.306 Pigs, therefore, present an interesting dichotomy: they excrete more infectivity than other domestic animal species while being themselves relatively resistant to infection.
Relatively little is known about the role that sheep and goats play in the epidemiology of FMD in sub-Saharan Africa. As mentioned above, sheep are highly susceptible to infection and both species excrete significant quantities of virus when acutely infected.322 Although infection has occurred in these species during epidemics in the subcontinent, 13, 195 cattle are far more important than sheep and goats in the spread of infection. This is probably due to their generally lower rates of virus excretion and lower susceptibility to air-borne virus because of their smaller inspiratory volumes.322 In East Africa, sheep and goats were found to have little importance in the epidemiology of FMD.11 In southern Africa none of the many outbreaks recorded since 1931 have been reported as having significant involvement of sheep and goats. For this reason these species are not included in routine prophylactic vaccination programmes aimed at the control of FMD. Conversely, in the Middle East there are FMD strains, particularly within type O, that appear to circulate preferentially in sheep and goats.195 There are, furthermore, many examples of FMD being carried into previously free countries by sheep and goat imports.195 The tendency of sheep and goats to develop mild or even clinically silent infection, makes them a particularly dangerous source of infection.252 It has been pointed out195 that although direct contact between infected and susceptible animals is the usual mechanism of transmission within sheep and goat populations, the rate of transmission within flocks is lower than that which generally occurs in pig or cattle herds. In an outbreak in Greece in 1994, for example, towards the end of the epidemic only about 20 per cent of individuals within herds became infected.214 A similar phenomenon was encountered during the 2001 epidemic in the UK in which sheep clearly played a major part.195 However, a recent laboratory study using the Greece 1994 virus has shown that there was a significant reduction in the levels of infection and estimated transmission rates over time during serial passage through groups of sheep.168 This supports the hypothesis that some, if not most, FMDV strains will die out in sheep populations if the levels of infection are not ‘boosted’ periodically by infection of cattle or pigs.195 On the other hand, it is clear from the pattern of the outbreak that occurred in the UK in 2001 that the pan-Asian O lineage involved was spread widely by sheep.133 Therefore, on the basis of observations in other parts of the world it behoves countries in sub-Saharan Africa to be aware of the potential of sheep and goats to spread the infection surreptitiously.
Coitus and artificial insemination as methods of transmission by infected bulls are possible,297 but the minimal infectious dose for cattle by this route is high, viz. 103,4 MLD50. It has been stated, but without corroborating evidence, that this route does not appear possible for sows and nanny goats.297
Other routes of viral entry are also possible. For example, infection of cattle was achieved with as little as 100 infective doses inoculated onto the conjunctiva.311 This suggested that face flies (Musca spp.) could possibly transmit the virus mechanically, but a preliminary investigation into this possibility failed to substantiate it.329 Arthropods, in general, probably play no part in FMDV transmission.146, 177, 328 Small rodents and hedgehogs (family Erinaceidae) in Europe177 and capybaras (Hydrochoerus hydrochoeris) in South America138 have been implicated in spreading FMDV, but as yet there is no evidence that similar animals are important in this respect in sub-Saharan Africa.
The role of wildlife
A general observation has been that wherever in the world FMD has been eradicated from livestock, it has also generally disappeared from wildlife in those regions.326, 330 In all parts of the world with the exception of sub-Saharan Africa, FMD in free-ranging or captive wildlife appears to have been an extension of the disease in livestock. This has been documented for free-ranging moose (Alces alces),215 as well as in fallow (Cervus dama),25 roe (Capreolus capreolus) and red (Cervus elaphus) deer in Europe.70 In the former Soviet Union, FMD was described in free-ranging reindeer (Rangifer tarandus)202, 242 and saiga (Saiga tatarica),185 while in India severe clinical signs and mortality were reported in blackbuck (Antilope cervicapra). 182 High morbidity and mortality also occurred in free-ranging mountain gazelles (Gazella gazella) in Israel.299 All these episodes in wildlife occurred during epidemics in cattle. Similarly, outbreaks of FMD in zoological gardens in Paris,333 Zurich8 and Buenos Aires145 coincided with outbreaks of FMD in domestic animals. Even in sub-Saharan Africa, where wildlife are clearly involved in the maintenance of FMD, livestock sometimes transmit the infection to wildlife rather than vice versa.12, 157, 327
The only locality in which overt FMD has been reported regularly in wildlife over the last 60 years is the Kruger National Park (KNP) in South Africa, where there have been 31 recorded outbreaks in impala since 1938, 23 of which were detected after routine surveillance was introduced in the KNP in the mid-1960s. Eight (26 per cent) were caused by SAT1, 15 (48 per cent) by SAT2, three (10 per cent) by SAT3, and five (16 per cent) were untyped. However, since 1983, nine of the 10 outbreaks in impala were caused by SAT2. Sequence analysis of the recent SAT2 isolates involved has shown that these outbreaks were causally distinct.29, 345
In the KNP, FMD in impala appears to occur generally in localities where high densities of this species occur. In addition, because impala depend on water, infection frequently has spread along water courses in the park, i.e. it is assumed that the virus is not transmitted through the water itself but by contact between animals congregated along rivers and streams.
With few exceptions, obvious clinical disease has not occurred in other species in the vicinity of outbreaks in impala.183 Direct contact between impala inside the park and domestic animals outside is largely prevented by a perimeter fence, and cattle immediately outside the fence on the South African side are vaccinated every six months. Perimeter fencing and vaccination of cattle close to the perimeter of the KNP are presumed to have prevented many FMD outbreaks in cattle raised in that vicinity (see below).
There is some uncertainty about the dimensions of fences required to prevent the spread of FMD in southern Africa, particularly in view of the potential for air-borne spread of FMDV. In south-eastern Zimbabwe, double fence lines with a defoliated zone about 10 m wide between the two lines have been used to form the perimeters of commercial wildlife conservancies. The idea is that direct transmission across the fence lines would be precluded by prevention of direct contact between animals on either side of the fence. In the initial design, one of the fence lines was at least 1,8 m in height to prevent antelope from jumping over the fence.151 An analysis of the risks posed by such a system to the livestock industry of Zimbabwe was conducted soon after the establishment of three such conservancies. 312 This showed that there was a risk that, despite the double fence line and the height of the fence, impala and kudu (Tragelaphus strepsiceros) antelope would be able to get out of the conservancies in significant numbers by jumping the perimeter fences. Conversely, the risk of airborne spread across the perimeter fence lines was found to be insignificant. A few months after the risk analysis was completed an outbreak of FMD caused by a SAT2 virus occurred in cattle immediately adjacent to one of the conservancies. Subsequent investigation, which included genome sequencing of isolates recovered from cattle involved in the outbreak, showed that the causative virus had been introduced into the conservancy two years previously by African buffalo translocated from the Hwange National Park in western Zimbabwe.151 There was strong circumstantial evidence that the herd of buffalo introduced from Hwange had infected antelope (impala and kudu) in its vicinity and that the antelope had subsequently transmitted the infection to cattle outside the conservancy.151
Although African buffalo in the KNP in South Africa have been shown to be the usual source of infection for impala on the basis of sequencing studies,27, 29 persistent infection in impala has not been demonstrated.10, 340 However, FMD epidemics caused by apparently identical viruses have recurred in impala 6 to 18 months after the original outbreak183, 345 indicating that the virus may have been maintained within the impala population. Were that so, the mechanism whereby the virus survived in inter-epidemic periods remains to be explained. The alternative explanation is that the same virus has been transmitted on more than one occasion from buffalo to impala in the same vicinity.
Infection and attack rates have varied in the outbreaks of FMD in impala that have been studied, with the latter sometimes being much lower than the former, indicating that subclinical infection is common, as has been noted in impala exposed experimentally.40
Paradoxically, clinical FMD has not been diagnosed in impala other than in the KNP (this species is widely prevalent in sub-Saharan Africa), although there is serological evidence of infection in other parts of the subcontinent.12 It is assumed that the reason for this is that in the KNP, unlike most other parts of sub-Saharan Africa where impala occur, active surveillance for clinical FMD is conducted routinely.
African buffalo were recognized as major reservoirs of SAT types in the 1970s160 although it seems that these animals rarely develop clinical FMD in natural circumstances. In this respect, SAT types affect buffalo and impala in the KNP differently. Persistence of the virus in some individual buffalo for at least five years probably explains why it has been maintained for a period of over 20 years in a small isolated group of buffalo.73 However, persistence of infection in individual buffalo is probably not life-long.158
Infection of individual animals within breeding herds of buffalo usually occurs when colostrum-derived immunity starts to wane at two to four months of age.72 Calves are not necessarily infected by their dams,71 and it is presumed that SAT types of virus spread mainly during minor epidemics among young animals in breeding herds, with carriers ensuring that the virus survives interepidemic periods.331 Since most buffalo in southern Africa are born in midsummer, they become susceptible to infection more or less synchronously during the dry winter months when passively acquired antibody wanes. Other susceptible species, principally impala, probably become exposed while infection is circulating among buffalo calves, possibly around permanent water points, where animals congregate. It is assumed, therefore, that there is a time during each year when large breeding herds of buffalo are a potent source of infection for other species of animals that come into contact with them, even though there is no obvious clinical disease within such herds.
Transmission of SAT types of virus between individual buffalo appears to occur by two processes:
- contact transmission between acutely infected and susceptible individuals which is likely to account for most infections; and
- occasional transmission between carrier buffalo and susceptible individuals.
However, the mechanism whereby carrier transmission occurs between buffalo is obscure. A possibility, for which the evidence is still tenuous, is sexual transmission.28 In a study conducted in the KNP, samples were collected from the urogenital tracts of 20 buffalo bulls. From the semen and sheathwash of a 3,5-year-old animal with measurable circulating antibody to all three SAT types, SAT3 type virus was recovered.
This suggests that carrier bulls may shed SAT viruses in their semen. Buffalo bulls in the field have been observed by farmers to mount domestic cows on occasion150 and it is possible that sexual activity may be a way in which SAT types of FMDV are transmitted from African buffaloes to cattle.
It appears that the rate at which the three SAT types circulate within buffalo populations in the KNP differs. SAT1 viruses were consistently recovered at a higher rate than those of the other two SAT types over a 10-year period from probang specimens collected from buffalo.330 Studies into circulating antibody levels in the same population showed that antibody to SAT1 rose consistently sooner in young animals than for the other two types.322, 331 This indicates that SAT1 viruses circulate more rapidly in the KNP than do SAT2 and 3.
Genome sequencing studies conducted over the last ten years in southern Africa have shown that buffalo populations in different geographic locations maintain distinct lineages of SAT viruses, i.e. so-called topotypes. For SAT2 and SAT3, four topotypes have so far been identified within each type while for SAT1 only three were distinguishable.26, 30, 31, 344 Figures 125.6 and 125.7, as examples, show the geographic distribution of SAT3 topotypes in southern Africa. Topotype I comprises viruses from north-eastern South Africa, southern Zimbabwe and Mozambique; topotype II is represented by viruses from western Zimbabwe, Namibia and Botswana; topotype III contains viruses originating from Zambia; whilst topotype IV is represented by virus from northern Zimbabwe. The geographic distribution of the SAT1 and 2 topotypes is similar.27 This demonstrates clearly that SAT topotypes are in a process of evolving independently in buffalo in different wildlife locations. The natural geographic distribution of intratypic variants within each SAT type are therefore now clearly recognized in southern Africa. Similar work in other regions of Africa is needed.
At the applied level this has enabled the source of outbreaks of FMD in cattle in southern Africa to be traced to their origin in individual buffalo populations346, 347 and has been used to trace the origin of captive buffalo moved illegally.343 It also enables selection of appropriate virus strains for inclusion into vaccines used to protect cattle in the vicinity of infected buffalo populations.171
There is, however, a paradox in understanding the transmission of FMDV from buffalo to other species. While SAT1 viruses appear to circulate more rapidly within buffalo herds in theKNP(see above), most FMD outbreaks in impala within the KNP as well as in cattle adjacent to the KNP have been caused by SAT2 types.30, 322 A possibility is that impala as well as cattle are, in general, more susceptible to infection by SAT2 viruses than SAT1 but there is no direct evidence for this.
Epidemiological investigation into two recent outbreaks of FMD in cattle in the Mpumalanga and Limpopo provinces of South Africa show, with a high degree of probability, that the infections were derived from contact with buffalo that escaped across the perimeter fence of the KNP.347
The first of these outbreaks occurred initially in communal cattle in the Nkomazi area immediately south of the KNP in November 2000.56 Unfortunately, this SAT1 infection spread to a feedlot near Middelburg (Mpumalanga), about 200 km to the west, before it was detected and thence to an abattoir at Manzini in Swaziland by cattle exported from the feedlot for slaughter. The outbreak also spread across the border between South Africa and north-eastern Swaziland by illegal transborder movement of cattle from Nkomazi. These events were deduced by comparison of partial 1D nucleotide sequences of viruses obtained from the various localities and investigation on the ground.56 The sequencing results clearly showed that the virus from the Swaziland outbreak and those from the South African outbreak shared more than 99 per cent sequence homology and that they are most closely related to a southern KNP buffalo virus genotype, represented by KNP/22/96.56 The FMD outbreaks were preceded by a devastating one in 100-year flood in eastern Mpumalanga that destroyed the perimeter fence of the KNP in many places and allowed buffalo to escape into adjacent farming areas. The significance of the flood is demonstrated by the fact that, after the flood of 2000, a total of 468 buffalo were chased back into the KNP by helicopter or destroyed during the third quarter of the year in comparison with significantly smaller numbers in the four previous years.56, 347 Some of the escaped buffalo were observed to have mingled with cattle in the surveillance zone adjacent to the KNP, where vaccination is not practised. As mentioned above, the SAT1 virus causing the outbreak was shown to be closely related to SAT1 virus isolates from buffalo in the southern part of the KNP.
The other FMD outbreak, caused by a SAT2 virus, occurred at Bushbuck Ridge — in Limpopo Province but also adjacent to the KNP — in February 2001.347 This outbreak was similarly attributed to the escape of buffalo from the KNP following the unusual flood of the previous year. The virus involved was shown by partial 1D sequencing to be closely related to SAT2 viruses previously isolated from buffalo in the nearby Orpen Gate area of the KNP (data not shown).
Pathogenesis
The pathogenesis of FMD has been studied mainly in cattle and pigs.53, 54, 59 Acquisition of infection, other than in pigs in which it is generally oral, usually occurs by inhalation and the initial site of virus replication may be the respiratory bronchioles of the lung.54However, an earlier study showed initial replication occurred in the mucosa and possibly the lymphoid tissues of the pharynx, particularly in the tonsillar region of the soft palate.60 Furthermore, recent work indicates that, in pigs at least, replication of virus does not occur in the respiratory epithelium.6
The fact that high levels of virus could be recovered from retropharyngeal, mandibular and parotid lymph nodes in more than 50 per cent of cattle in the pre-viraemic stage of infection suggests that spread from the initial replication site usually occurs via the lymphoid system60 and that replication occurs in lymphoid tissue. The liberation of virus into the efferent lymphatic system results in an initial viraemia whereby virus reaches a wide variety of organs and tissues.
In domestic ruminants and African buffalo the pharynx and dorsal soft palate are predilection sites for FMDV replication. Not only does early viral replication occur at these sites but virus persists there for prolonged periods. The initially high virus levels (>106,0MLD50/ml) which occur in the secretions of the pharyngeal region in cattle, fall sharply during the following eight weeks,104 so that in persistently infected cattle the quantities of virus usually recoverable from pharyngeal secretions are low.60 Thevirus persists particularly in basal epithelial cells of the pharynx and dorsal soft palate where, unusually, the infection does not result in lysis of cells. Basal epithelial cells lie adjacent to the basement membrane of the specialized non-cornified, stratified, squamous epithelium that occurs at these sites.355 The virus is not present in the more superficial layers of this epithelium and it is not clear how it is excreted into the pharynx. The mechanism of viral persistence in basal epithelial cells also remains to be identified but a number of mechanisms have been proposed for persistent infection with FMDV.286, 352 Viral persistence in the pharyngeal region of pigs has so far not been shown to occur, which accords with the lack of other evidence for the existence of carriers in this species.
In persistently infected ruminants, virus has, as yet, only been recovered from cells and secretions collected from the pharynx and cranial oesophagus using ‘probang cups’.309 However, FMDV-specific genome sequences in multiple sites have been found, although not in pharyngeal specimens in cattle, up to two years after infection.42 This finding contradicts previous observations based on detection of live virus and its significance is therefore uncertain. Similarly, finding evidence for the presence of FMD viral genome, but not live virus, in the sera of pigs recovered from FMDV infection needs corroboration because it questions the widely held view that persistent infection does not occur in pigs.230
High virus levels may occur in organs and tissues which do not generally develop gross changes referable to FMDV replication, such as lymph nodes, myocardium, pancreas, and adrenals, thyroid and mammary glands,45, 60, 74 as well as skin of unaffected areas.128 In mammary tissue and myocardium, however, viral replication in secretory epithelial cells of the alveoli and myocytes respectively results in clear microscopic lesions45, 46, 103 and it is therefore possible that this situation also occurs in other organs and tissues. Replication of the virus in mammary tissues is the probable cause of the dramatic fall in milk production characteristic of FMD in cows. They often do not recover during the lactation concerned and permanent loss of quarters as a result of secondary mastitis may occur.
Infecting virus is thought to spread via the bloodstream to Langerhans cells (macrophage-like dendritic cells) in epithelia at sites throughout the body.85 All epithelial cells in contact with an infected Langerhans cell become infected.53 An interesting demonstration of this phenomenon (i.e. infection of dendritic macrophages of the Langerhans type) is the effect that prior African swine fever virus infection has on subsequent exposure of pigs and dendritic cell cultures to FMDV. Both in vivo and in vitro, African swine fever virus is able to prevent, or at least reduce, the capacity of FMDV to cause infection.142, 143
Epithelial lesions in FMD are initiated by infection of single cells in the stratum spinosum.352 Following infection of these cells, bullae develop either by lysis of cells swollen as a result of ballooning degeneration and the release of intracellular fluid, or by the formation of areas of focal intercellular oedema. The bullae then coalesce and rupture or, more rarely, the fluid seeps away resulting in desiccation of the lesion.352
Development of characteristic vesicular lesions in FMD is dependent on two factors — infection of squamous epithelium and persistent local irritation or friction.256 In transplantation studies in guinea pigs it was shown that epithelium from predilection sites grafted to other body areas lost that predilection and vice versa. This explains why the mouth, feet and teats are predilection sites for the development of lesions, and why pigs often develop lesions on the dorsum of the snout, as a result of ‘snuffling’. Similarly, warthog (Phacochoerus aethiopicus) which often ‘kneel’ on their carpal joints while feeding tend to develop lesions on their ‘knees’.
Detectable viraemias resulting from SAT type infections in cattle and pigs do not last longer than three days129 but in sheep infected with other virus types viraemias were shown to last as long as five days.195
It has been observed that yearling cattle may fail to recover their productive potential fully as a result of damage to ‘glandular tissue’, such as the thyroid. These have been referred to as ‘hairy panters’ because their coat becomes long and rough (Figure 125.8) and their tolerance of high ambient temperatures is apparently impaired190 (see Clinical signs and pathology).
Immunity
The immune response of domestic animals to infection with FMDV is characteristically ephemeral and this, together with the wide immunological diversity present within this group of viruses, often results in poor herd immunity.
Although the responses of very young piglets and calves differ from those of more mature animals, they are immunologically competent to FMDV from an early age.124, 238, 280 The poor responses of calves and piglets to immunization is probably due more to immunological interference by colostrumderived antibody124, 237 than to immunological immaturity. Colostrum-derived antibody disappears before pigs and cattle attain the age of six months,123, 237 the half-life being 21,5 days in cattle.280 In pigs the half-life varies with the class of immunoglobulin which predominates in the colostrum: the half-life of IgM is four to eight days while that of IgG is 7 to 21 days.123 For this reason it has been recommended that sows not be vaccinated or revaccinated within 30 days of farrowing since this will elevate the IgM level with its short half-life.121
Cattle are immune to reinfection with homologous virus for one to three years, and occasionally for up to 4,5 years.18, 50 Circumstantial evidence suggests that the duration of immunity after infection with SAT types of virus may be shorter. The duration of immunity in other domestic species is largely unknown, but it is probably shorter than in cattle.18 That there is considerable species variation, has been shown by serological studies in wildlife.160
Since recovery from infection with FMDV is the rule, little attention has been paid to the immunological mechanisms involved in recovery, although interferon levels and the ‘opsonophagocytic’ activity of peripheral blood leukocytes have been examined in acutely infected animals.9 Conversely, since it is clear that the level of neutralizing antibody in the circulation correlates with resistance to infection or reinfection in immunized and recovered animals respectively,219, 249, 313 the humoral immune response and the antigens which are able to induce it have been extensively studied. The relationship between neutralizing antibody level and the degree of resistance to infection is, however, not simple and depends on the virus type as well as, in immunized animals, the period between immunization and exposure to infection.335
Neutralization of the FMD virion can occur at several sites.225, 320, 353 The major immunogenic site is composed of a complex arrangement of two regions of VP1 (amino acid residues 140–160 and 200–213) in addition to contributions from at least one of the other surface proteins, VP2 or VP3.52, 234, 353 The 140–160 region of VP1 seems to be the major determinant involved in neutralization,269 and amino acid residues 133–158 of VP1 form disordered protrusions on the virion surface.1 VP1 is the only structural protein of FMDV which, on its own, is at all immunogenic.21, 203
The antibody response to FMDV is T-cell dependent.125Since different species recognize different determinants as T-cell recognition sites, not all FMDV antigens are equally immunogenic in different species.52 This has presented problems in the development of synthetic vaccines.
Because the usual route of infection in FMD is the respiratory tract, local immunity may be important as far as resistance to infection is concerned, but the extent to which this is so with respect to overall immunity remains to be established. Neutralizing activity occurs in the secretions of the upper respiratory tract in cattle and pigs recovered from FMD. In cattle it reaches a peak within one week of infection achieved by inoculation of virus into the epithelium of the tongue.122, 126 The major constituent of the local response in cattle is IgA, although IgM is also present in appreciable amounts for approximately the first 10 days.126
Clinical signs and pathology
All cloven-hoofed species and camelids are susceptible to infection with FMDV, although the resulting disease varies with the level of immunity, the virus strain and quantity of infectivity to which the animal is exposed, host species, breed and even between individuals of a given breed. There are virus strains with a predilection for individual species.49, 177 Some strains of the pan-Asian type O lineage that have occurred in Taiwan and the Philippines appear to be adapted to pigs and do not readily infect cattle.193 Goats and sheep generally suffer mild or, sometimes, completely inapparent infection.11, 129 This appears to be particularly so for goats in southern Africa although no definitive studies have so far been conducted. In the 2001 UK outbreak, sheep were the species responsible for most spread of infection and in many cases this was to some extent because infection was not accompanied by obvious disease, i.e. in many cases the disease that occurred was inapparent to the owner or dealer. In specific studies vesicular disease was not found in about 25 per cent of sheep that became infected (it was claimed by some involved in the UK outbreak that in some flocks this was as high as 95 per cent) while a further 20 per cent may develop only a single lesion.135, 195 A further important point is that lesions in sheep may be visible for only a few days.195 In southern African outbreaks involving indigenous or Bos indicustype cattle are often mild and difficult to recognize.108, 184
In adult animals mortality rates are low, although instances of rates as high as 30 per cent have been recorded in cattle.174 Some pan-Asian O lineage that have occurred recently have caused high mortality in pigs.193One of the reasons why the diagnosis of FMD was delayed in the FMD outbreak that occurred at Camperdown in South Africa in 2000 was due to the mortality rate in pigs on the index farm being higher than expected for FMD. This lead to an initial suspicion of toxicosis.
Foot-and-mouth disease is characteristically a rapidly spreading infection.139 The incubation period is generally short, ranging from two to eight days but may last up to 11 to 14 days. The incubation period following between-farm spread may be even longer as was observed in France in 2001.67 In most outbreaks the morbidity rate approaches 100 per cent. An important observation in southern Africa, dating from the earliest investigations, however, is that spread between cattle may be surprisingly slow.106 Morbidity rates as low as 0,2 per cent have been described in the so-called occult form of FMD,108 but this has not been described from anywhere other than Botswana.
As its name implies, FMD is characterized by the development of lesions in the mouth and on the feet. However, the earliest clinical signs are those of a non-specific infectious illness and include fever, dullness, inappetence, a fall in milk yield and the cessation of rumination. Because the disease is usually rapid in onset and development, these signs are superseded within a few hours by signs more obviously referable to the development of lesions. These include lameness, a disinclination to stand, lagging behind the herd/flock, salivation and other signs of discomfort in the mouth such as smacking of the lips and grinding of the teeth. There is often a concomitant serous nasal discharge, which usually becomes mucopurulent during the following two to three days.
The young of domestic species susceptible to FMD may die suddenly without premonitory signs as a result of myocarditis (sometimes referred to as ‘tiger heart disease’ because of the macroscopic appearance of the myocardium). In a number of instances in western Europe prior to the eradication of FMD in 1992, as well as in the Middle East, the first indications of the occurrence of FMD were sudden deaths among piglets and lambs.
Lesions in cattle are usually present in the mouth, particularly at the tip and/or on the dorsal surface of the tongue, and on all four feet (Figures 125.9 to 125.13). This is not invariable and lesions may be absent from the mouth or one or more feet.108, 139 Lesions in the mouth may also occur inside the lips, on the gums, dental pad, hard palate, and even the muzzle. They do not occur on the ventral surface of the tongue which is one of the features that distinguishes lesions caused by FMD from those of rinderpest. Lesions due to FMD have also been described in the mucous membrane of the nostrils and even in the conjunctiva of cattle.164
Mouth lesions are first visible as small blanched foci. These foci rapidly increase in size and develop into vesicles containing serous fluid (Figure 125.9) which rupture easily. For this reason unruptured vesicles are not often seen during the examination of field cases. In acute cases approximately circular erosions with shreds of epithelium adhering to the edges of the lesion are the common finding. Other than on the dorsal surface of the tongue, these lesions are usually less than 20 mm in diameter, but on the tongue they may be more than 60 mm in diameter.
Rarely, the epithelium of a large part of the dorsal surface of the tongue desquamates during examination leaving a raw and bleeding surface (Figure 125.10). Healing of mouth lesions is far more rapid than is the case with foot lesions. By 11 days after infection healing lesions have a regenerated epithelium although without papillae which take longer to regenerate (Figure 125.11).190 Lesions that are more than two weeks old are often only discernible as circumscribed areas with poorly developed papillae and can be easily overlooked.
Mouth lesions are less common and less pronounced in other domestic animal species such as goats, sheep and pigs18, 132, 164, 252 and may proceed directly from epithelial necrosis to erosion without vesicle formation.94, 132 In sheep and other small ruminants, lesions commonly occur on the dental pad where they may be difficult to detect (Figure 125.14). In pigs, lesions often occur on the snout (Figures 125.15 and 125.16) but in other species lesions on the muzzle are rare. Lesions on the tongues of pigs tend to occur further back than is the case in other species.193
Foot lesions in all species develop in the skin of the interdigital space (Figure 125.12), the bulbs of the heel and the coronet.139 In cattle, lesions predominate at the two former sites while coronary band lesions are more common in smaller species, including antelope such as impala (Figure 125.17).
In severe cases lesions are usually present at all three sites and may be so severe that the horn of the hoof is lost completely. Severe lesions of the feet may result in the formation of a fault in the hooves (Figure 125.13). Pigs often develop lesions at the base of the supernumary digits and in the skin covering the carpal and metacarpal joints (Figure 125.18) as well as that covering joints of the hind legs when housed on rough surfaces.193
The skin covering intact or recently ruptured vesicles is pale (Figure 125.19) and in the course of the one or two days following vesicle development becomes necrotic and friable. In less severe cases fluid liberated from small vesicles becomes mixed with soil particles to form brown scabs or crusts. Secondary bacterial infection of foot lesions occurs frequently, particularly where animals are kept in muddy unhygienic conditions. The severity of this varies, but it may result in suppuration extending into the joints, tendon sheaths and even the bones of the foot with crippling consequences.
In cows and sows, lesions often develop on the teats and the resulting pain means that such cows are difficult if not impossible to milk properly. Secondary mastitis is a frequent complication in cows.
So-called chronic FMD, was encountered historically in cattle in both Europe and South America307 and is apparently also recognized by pastoralists in East Africa who have specific names for it.66, 216, 255 The condition characteristically occurs three to six months after acute FMD in adult cattle and is manifested by heat intolerance resulting in an increased rate of respiration, a ‘woolly’ or ‘hairy ‘ coat (Figure 125.8) and general unthriftiness. These animals are sometimes referred to as ‘hairy panters’. Other signs that may be present are reduced milk production, mastitis and infertility. Typically, this condition may affect up to 5 per cent of cattle in a herd.307 Although there is no unequivocal evidence that the condition is a sequel to FMD, it is widely thought to be due to some form of hormonal imbalance resulting from infection with FMDV.
Diagnosis
The ability to diagnose FMD rapidly and accurately is a prerequisite for efficient animal health control as well as for the support of export-oriented agricultural economies in large parts of the world.
Because of the renowned ability of FMDV to spread rapidly, the handling of this agent for diagnostic purposes should be restricted to high-security laboratories.241
In domestic animals the diagnosis of FMD is dependent upon clinical and epidemiological assessment of the situation where the disease is suspected followed by identification of FMDV, viral RNA or viral antigens in the early stages of infection, and serology if the condition has been in progress for longer than two weeks. The diagnosis of persistently infected or carrier animals is effected by the isolation of virus or identification of viral RNA in oesophagopharyngeal secretions.265
Since both the fluid contained in, and the epithelium covering, vesicles usually contain high concentrations of FMD virus (>106 infectious doses/millilitre or gram), these are the specimens of choice in acute cases. The fluid is easily aspirated from unruptured vesicles using a needle and syringe while epithelial fragments may simply be cut free from the edges of lesions with scissors. Epithelial specimens from mouth and foot lesions contain detectable quantities of live virus for four and seven days respectively after the appearance of the lesions.244
It is essential to add a transport medium buffered between pH 7 and 8 to specimens collected for laboratory examination to prevent desiccation and changes in pH during transport. Phosphate-buffered saline (PBS) at pH 7,4 is ideal and is the usually recommended medium. The molarity of such solutions is not as important as the pH, but should be close to normal. If suspect specimens can be kept cool (4 to 10 °C) and reach a suitable laboratory within about 12 hours, no further preservative is needed. If this cannot be achieved, 50 per cent glycerol and antibiotics may be added to the PBS in which epithelial fragments are suspended, to prevent putrefaction.194 Glycerol should not be added to specimens of vesicular fluid because it is toxic to cell cultures. There is no practical way to separate vesicular fluid from the glycerol except by dilution, which could hamper the isolation of virus present in small quantities. Glycerol can be removed from epithelial specimens by washing them in PBS. Detailed instructions for the collection, packaging, and dispatch of FMD specimens have been published.194 The collection of probang specimens (see below) from animals persistently infected with FMDV has similarly been described.162
Viral identification, typing and subtyping
Initial identification of FMDV contained in material collected from acute cases of the disease is usually done using a type-specific antigen-capture enzyme-linked immunosorbent assay (ELISA).114, 263, 351 Some laboratories still use short incubation-time complement fixation tests for this purpose as this was widely employed prior to the introduction of the antigen-capture ELISA. If the material under test contains sufficient virus (approximately 105 TCID50/millilitre or gram), results can be available within a few hours.
In a well-equipped laboratory susceptible cell cultures— ideally primary bovine thyroid but pig, lamb or calf kidney cells are also apt—should be set up at the same time in case the initial viral content of the material available is too low for detection by ELISA.303 In addition, multiplication of the virus in cell cultures will provide virus for more detailed viral characterization. If the viral content of the initial sample is low or the virus concerned requires adaptation to the cell culture system used, it may take a further two blind passages (i.e. several days) in cell cultures for recovery of the virus. Thus it may take four to five days before a field sample can be conclusively declared free of FMDV. In general, the virus grows rapidly in susceptible cell cultures and cytopathic effects are usually visible within 24 hours.
Increasingly, highly sensitive polymerase chain reaction (RT-PCR)-based systems are used on initial field material not only to determine whether or not FMDV is present but also to type and assign the virus present in the material submitted to viral lineages.26, 154, 243, 258, 288 The partial genomic sequences of the FMDVs obtained from field material using this approach can rapidly be compared with those stored in the laboratory data-base or available on the Internet (e.g. from Genbank). This enables the virus type to be determined in less than a day and the viral lineage to be established within a further 24 to 48 hours (see Aetiology). This information can be vital in establishing the origin of the outbreak and for following its subsequent spread.
It is sometimes argued that PCR-based systems make virus isolation unnecessary and that therefore the isolation of field virus using cell culture is obsolete. It should be remembered, however, that it is essential to isolate the viruses involved in outbreaks to determine antigenic relationships with potential vaccine strains and for biological characterization of the virus. Furthermore, because PCR systems are so sensitive, they are prone to contamination and may provide false positive results. The notion that virus isolation is unnecessary is therefore erroneous.
Probang specimens
When vesicular epithelium/fluid is not available, usually because the suspect lesions are healing, the most useful specimen is throat scrapings obtained with the use of a probang cup that is inserted into the pharyngeal region of ruminants (Figure 125.20). From such material the virus may be isolated, albeit in small amounts, or identified by PCR-based tests.
Virus in the pharyngeal area of ruminants is likely to persist in a proportion (commonly about 50 per cent) of animals for several months after the acute phase of infection (see Epidemiology). However, because the virus does not persist in all animals and also because when it does it is erratically present in material collected in this way,87, 169 at least four to five animals should be sampled.
It is important that probang material is mixed immediately after collection with an equal volume (usually 4 to 6 mm) of a buffer solution that will maintain the pH at about 7,4. For reliable results the buffered material should immediately be placed in cryo-tubes and frozen using liquid nitrogen or dry ice for transport to the laboratory. Keeping such material for several hours in a ‘cool box’ during transport to the laboratory will significantly reduce the likelihood of virus recovery. For reasons that are not clear, some virus-containing probang specimens are identified by virus isolation but not PCR and vice versa. For that reason both PCR and virus isolation should be attempted on probang material. If virus identification is indicated in cases where the animals were infected more than seven to ten days previously, probang material is the only potential source.
It is important that whenever probang material is collected from animals, blood for serum preparation is also collected from the same animals. This will enable the laboratory to correlate the results of virus detection and serology to provide a more reliable interpretation of the likelihood of infection in the animals in question. This is particularly so when vaccine may have been administered to the animals involved.
Serology
For approximately the last 15 years serological testing as a means of diagnosing FMD has relied on tests — virus neutralization and ELISA — in which antibodies directed against the surface proteins of the virus are detected (see Aetiology).240 These type-specific tests detect antibodies resulting from infection or vaccination with equal facility and therefore can be used for monitoring vaccine responses as well as retrospective detection of evidence of infection. However, they are unable to differentiate between responses to infection and vaccination. More particularly, there is the problem of differentiating between animals that have simply been infected and those that become infected subsequent to vaccination and which may then become carriers. Work conducted over some years directed at developing tests that enable such differentiation213 have now resulted in several tests (e.g. ELISAs and immuno[Western]-blot’s for a variety of non-structural viral proteins) that, if properly applied, can do that.41, 213, 305 In essence, these tests rely on the fact that inactivated vaccines prepared using at least partially purified virus antigens (i.e. free of non-structural viral proteins [NSPs]) do not stimulate antibody responses to NSPs of the virus. Therefore, if antibodies to NSPs are found, their development must have been induced by viral replication following natural infection (or have been derived from colostrum of such animals). Unfortunately, so far, this differentiation is only possible on a herd basis because in some animals that become infected subsequent to vaccination, viral replication may be suppressed to such an extent that antibody responses to NSPs remain below the threshold of detection.191 The development of tests for detection of antibodies to NSPs has, nevertheless, added significant benefit to the laboratory diagnosis of FMD.
In situations that are well controlled it should be possible to differentiate between animals that are carriers and those that are not by judicial use of antibody tests for both structural and non-structural viral proteins combined with probang testing. However, this requires a detailed understanding of the tests and their limitations and a laboratory in which the various tests can be competently performed. This would only be practical for small numbers of animals.
The newly developed tests for NSPs should not be confused with the so-called VIA (virus infection-associated) test—using either agar-gel diffusion or ELISA detection systems — that were long used in South America to measure antibody to the viral polymerase (protein coded by the 3D gene). Initially it was thought that these tests differentiated infected animals from those that had been vaccinated but, because FMD virions each contain one molecule of polymerase, the VIA is inefficient in this respect.
Large-scale serology for FMD is currently usually conducted using the ‘liquid-phase blocking ELISA’147–149 which has the benefit of being adaptable to the sera of all FMD-susceptible species. This test, although highly sensitive, is less specific and therefore has the tendency to produce false positive results, with potentially serious repercussions.56Recently, a solid-phase ELISA has been developed which has higher specificity.113 This test was used extensively during the 2001 UK FMD outbreak and the subsequent testing required for the UK to regain its FMD-free status from the OIE.
Differential diagnosis
Vesicular stomatitis, swine vesicular disease and vesicular exanthema of pigs all produce lesions in cattle or pigs which are clinically indistinguishable from FMD.62, 112 The definitive hosts of swine vesicular disease virus are either fish or sea lions (family Otariidae), so that pigs only become infected by contact with these animals or their tissues. For this reason the more appropriate name for this virus is San Miguel sea lion virus.112 These viruses, as far as is known, are confined to the Pacific coast of the Americas.
Conditions which produce erosions or ulceration in the mouth or on the muzzle of cattle, goats, sheep or pigs and which need to be differentiated from FMD include bovine virus diarrhoea/mucosal disease, rinderpest, bluetongue, malignant catarrhal fever, lumpy skin disease, papular stomatitis, infectious bovine rhinotracheitis and orf (contagious pustular dermatitis). None of these diseases, except bluetongue and occasionally contagious pustular dermatitis, usually produce foot lesions.
Foot lesions caused by bacteria and fungi in animals housed in unhygienic, wet and crowded conditions, such as foot-rot and foul-in-the-foot, may produce lesions that are difficult to differentiate from those of FMD which have progressed beyond the vesicle stage. The absence of mouth lesions in such cases usually obviates confusion with FMD.
During the 2001 FMD epidemic in the UK oral lesions in sheep and cattle unassociated with FMD were regularly encountered in Scotland which complicated the diagnosis of FMD.17 A similar condition in sheep was described in England and Wales and referred to as idiopathic mouth ulcer.83 In most cases ulcers occurred in the gingival mucosa below the incisor arcade. The aetiology of the condition was not established although it was thought to be traumatic in origin. The disease was humorously referred to as ‘ovine mouth and gum obscure disease’ (OMAGOD).17
Furocoumarins contained in celery and parsnips, particularly if infected with the fungus Sclerotinia sclerotiorum and which interact with ultraviolet rays in sunlight, are capable of producing both snout and foot lesions in whiteskinned pigs.232, 233
Control
In general, control of FMD in sub-Saharan Africa has two distinct but overlapping situations to deal with. The first is cattle to cattle transmission involving all the types of FMDV prevalent in Africa. The second is disease associated with wildlife, especially African buffalo, caused by the three SAT virus types. Although small ruminants and pigs are frequently involved in the persistence and spread of FMD elsewhere in the world, this is not often the case in sub-Saharan Africa and clinical disease in domestic livestock is seldom encountered other than in cattle. Wildlife-associated FMD and the measures taken to control it have only been studied to any extent in southern Africa. It can safely be surmised, however, that it occurs in other parts of Africa where buffalo and cattle occur in the same vicinity. This is perhaps a reflection of the fact that there have been few systematic efforts to control FMD other than in southern Africa. For that reason, control strategies appropriate to Africa discussed here are largely based on the southern African example.
The developed world has now largely eradicated FMD. In western Europe, which serves as a good example, FMD was controlled after the Second World War by annual mass vaccination of cattle and occasionally pigs within the various countries of the region. By the 1970/80s this brought the number of outbreaks down to low figures. A few of these outbreaks were, however, caused by improperly inactivated vaccine or virus escapes from laboratories.24, 208, 306 In 1992 vaccination as a preventive measure was discontinued in the European Union (EU) and efforts were then concentrated on keeping the infection beyond the borders of the EU and developing contingency plans to deal with the infection if and when it were inadvertently reintroduced. This was based on comparative cost/benefit analyses of continued mass vaccination and the new strategy.208 The latter approach has been largely successful despite the now infamous incursion of type O virus via the UK in 2001.133 The sources of the few outbreaks that have occurred in Europe since the cessation of mass vaccination have been analysed.208 They were ascribed to (1) smuggling of live animals (Italy 1993/Greece 1994); (2) both legal and illegal importation of meat and animal products (Russia 1995/Balkans 1996/UK 2001); (3) escape from laboratories (Russia 1993); (4) indirect contacts: immigrants (Greece 1996); and (5) unknown (Bulgaria 1993 and 1996/Greece 2000).
However, the public and political fall-out resulting from the slaughter of millions of animals to eliminate the type O outbreak in Europe in 2001 — estimates for the UK alone vary between 6,5 to 11 million animals76, 293 — have resulted in FMD control becoming a matter of public concern in Europe and North America. In South America the unfortunate reoccurrence of FMD in Argentina, Uruguay and southern Brazil (the so-called southern cone of South America) in 2000/1 after cessation of annual mass vaccination has likewise caused widespread consternation. The consequence has been, and will continue for some time to be, widespread re-appraisal of approaches to FMD control in various parts of the world. This, in turn, is likely to result in fundamental changes in the next few years, the nature and extent of which are currently impossible to predict other than that there will be a move away from mass ‘stamping out’. Interestingly, ‘stamping out’ is one of the few subjects on which there is general agreement between developed and developing countries. In both it is increasingly considered that wholesale slaughter of healthy animals to halt or prevent FMD outbreaks (as well as those of other epidemic diseases) is morally and financially unacceptable.
On the other hand, particularly in the most developed countries, strategies are still currently based on ‘stamping out’, i.e. slaughter and disposal by burning or burial of infected and in-contact animals of susceptible species.293 ‘Stamping out’ presents other practical difficulties. In small but highly developed countries with large human populations there are undesirable ecological consequences related to mass carcass disposal. Incineration on a large scale results in atmospheric pollution while burial holds the possibility of contamination of underground water supplies. In 2001 this was a major problem in countries such as Ireland, the Netherlands and the UK.82, 315 The logistics of the rapid disposal of large numbers of carcasses in a short period of time is another factor making ‘stamping out’ sometimes impractical in practice. In the UK in 2001 the objective was to slaughter animals on infected and in-contact farms within 48 hours. At the height of the outbreak this resulted in an enormous back-log of carcasses awaiting disposal by incineration or burial293, 315 which was accompanied by public revulsion.
The perception of the public at large is that, because vaccines against FMD exist and have been successfully used on a large scale in the past, vaccination is the obvious way to control or to eradicate FMD. While that is basically true, interacting technical, economic and logistical realities make the issue complex.
Vaccination
Currently almost all vaccines used throughout the world against FMD are inactivated. In other words, the virus incorporated into the vaccine is first treated with an inactivant to destroy viral infectivity but its antigenicity is retained. Almost invariably aziride compounds (usually binary ethylene immine [BEI]) are currently employed but in the past formaldehyde was commonly used.24 Aziridines induce a first order chemical reaction — unlike formaldehyde — that results in RNA fragmentation and consequent inability of the virus to replicate. The inactivated vaccine may contain one or more virus strains of any of the seven types (generally incorporating about 1 μg per dose of semi-purified antigen). The latter are generally produced using cultures of BHK21 (baby hamster kidney) cells. These may be grown in monolayer or suspension cultures but, because suspension cultures are amenable to large-volume production (1 000 to 10 000 litres), the latter are increasingly being used. It is essential that cell-culture derived virus is concentrated either by ultrafiltration or precipitation with polyethylene glycol (PEG)24 because there is insufficient virus in one or two millilitres of cell-culture fluid to induce effective immune responses. To further potentiate the immune response it is essential that an adjuvant be added to the inactivated virus. Traditionally, the adjuvant has consisted of a mixture of aluminium hydroxide (to which the virus adsorbs) and saponin, a surface-active agent extracted from tree-bark. Purified saponin is known as Quil A.80 This type of formulation works well in ruminants but in pigs it induces poor antibody responses.140 For pigs therefore, and now increasingly for ruminants as well, oil-based adjuvants — single or double oil emulsions — are used.24 In cattle these adjuvants are held to stimulate longer lasting antibody responses, thereby lengthening the duration of effective immunity. This has not always been substantiated by experimental work but nevertheless the much improved performance of FMD vaccines in South America is claimed to be at least partly due to the widespread adoption of vaccines incorporating oil adjuvants. International agreement stipulates that each dose of vaccine should deliver at least three protective doses (3 PD50) although it is generally accepted that 6 PD50 provides more effective and rapid responses. 24, 47, 240 Thus the latter is generally recommended in emergency situations and it has been shown that effective immune responses occur within three to five days in cattle, sheep and pigs.24
There are a number of vaccine banks around the world117 that are capable of supplying emergency vaccines to their subscribers at short notice. The concentrated antigens are stored in the gas-phase of liquid nitrogen, which keeps the antigen stable for many years. There are, however, no banks for FMD vaccines in Africa and, as far as is known, no African country has automatic drawing rights on any of the international banks.
Foot-and-mouth disease vaccines, whether adjuvanted with aluminium hydroxide/saponin or oil-based preparations should be stored at normal refrigeration temperatures and not be frozen because freezing/thawing will disrupt the activity of the adjuvant. The maintenance of an effective cold chain therefore presents a difficulty during mass vaccination campaigns.
Because FMD vaccines contain non-living antigen they should ideally be administered as a primary and a secondary dose two to eight weeks apart to establish basic immunity followed by booster doses at four to six monthly intervals. Once animals have reached an age of two years only annual booster doses are necessary to maintain the level of immunity. However, due to the practical difficulties of applying such a regimen, mass vaccination against FMD generally involves biannual or annual vaccination of all animals and this has mostly proved to be satisfactory where field challenge is not severe. Vaccination of calves under six months of age should be avoided because colostrum-derived antibodies interfere with the response to the vaccine.
It needs to be remembered, however, that where turnover in the animal population is high — for example on pig farms — more frequent administration of vaccine is necessary to maintain a high level of herd immunity. Minimal levels of herd immunity required for effective prevention of FMD remain to be determined.
An important but frequently overlooked issue when it comes to the application of FMD vaccines is that the vaccines used in a particular locality need to contain virus strains that will invoke immune responses that are effective against the challenge which the animals are likely to meet in the field. As described in the section on Aetiology, not only are there seven different types but within types there is intratypic variation. Within specific types, especially SAT2 and A, antigenic variation is considerable. For that reason it should as far as possible be ensured that the so-called r-value of field viruses in the locality are ≥0,4 against viruses contained in the vaccine (see Aetiology). In southern Africa, where the major challenge is presented by viruses circulating in wildlife, it is important to establish which viruses are circulating in buffalo populations in the vicinity because there may be considerable geographic variation between localities (see Epidemiology). Elsewhere in Africa information on the viruses in circulation in particular countries or regions is generally sketchy other than for some historic information on the prevalent types.346 This means that vaccination against FMD in Africa is currently often a hit and miss affair.
Tradability of vaccinated animals
There are basically two reasons why current acceptance of a country’s (or a zone’s, i.e. a demarcated region within a country) freedom from FMD is incompatible with the presence of vaccinated animals. The first is that should vaccinated animals be present in a country or a zone in significant numbers, it could mask the occurrence of clinical disease were FMDV to be circulating. Secondly, it is possible for cattle—in theory sheep and goats too—that have been vaccinated against FMD to be protected against clinical disease following exposure to infection while nevertheless becoming infected and retaining the infection for months or years in the pharyngeal region (see Epidemiology). It is at least theoretically possible for such persistently infected animals to transmit the virus to susceptible cohorts (i.e. behave as carriers). This is a contentious issue and currently there is considerable debate on the practical significance of carriers. Some maintain that, in practice, vaccinated carriers have never been shown to transmit the infection.308
Fortunately technical developments are assisting in the resolution of this problem. As indicated above (see Diagnosis) there are now serological tests that are able to differentiate between, on the one hand, antibody responses of animals that have merely been vaccinated with inactivated vaccine and, on the other, animals that have either been infected or both vaccinated and infected. These tests, which aim to detect antibodies to NSPs (e.g. 3ABC ELISA and a Western-blot test commonly referred to as the EITB test) can be used to differentiate between potential carriers and animals that are not. However, at present, these tests have so far only been recognized as herd tests because in some individual animals the level of replication that occurs after vaccination is too limited to induce antibodies to NSPs.192
Essentially, vaccines against foot-and-mouth disease can be used in three ways:
- for routine prophylaxis;
- in emergencies to eliminate incursion of an exotic virus where, once the spread of infection has been halted, the animals are killed (so-called vaccination to die); and
- also in emergencies, but in this instance the animals are not subsequently slaughtered (so-called vaccination to live).
The reason why the second category exists is that current OIE recommendations241 place limitations on the time periods required before export of livestock and livestock products from countries where vaccination has taken place (and the animals not subsequently slaughtered) to FMD-free countries can be resumed. Thus for countries that are generally FMD-free and are dependent on trade in livestock and their products and which use vaccination to control an outbreak, there are likely to be restrictions on resumption of normal trade unless animals that have been vaccinated are slaughtered. This is a contentious issue and the OIE is in the process of reviewing its recommendations.
Response to outbreaks in areas free of FMD
Appropriate reaction to FMD outbreaks depends on the livestock husbandry systems practised in the area concerned and the extent to which livestock and livestock products are exported to parts of the world engaged in intensive livestock production. In extensive systems where livestock are used primarily for local consumption and provide a means of longterm investment, the monetary return they provide is low. In such cases there is usually simply not the finance or infrastructure to control FMD effectively. This is why, in most of Africa, FMD is currently an uncontrolled disease. Outbreaks that occur periodically have limited impact and do not affect trade and for that reason are simply disregarded. Conversely, in intensive production systems where FMD can impact severely on both production and trade in livestock, immediate and effective response is imperative. The type of response is usually predetermined in considerable detail by emergency planning and by legislation or regulations of the country or trade block (e.g. the EU) concerned. This not only determines the initial strategy but also aspects such as sources of emergency finance; details of personnel of various categories that can be called upon at short notice; and the location and quantities of emergency equipment (such as vehicles, earthmoving equipment, maps and vaccine) that can be readily accessed. Prior institution of appropriate legislation enabling quick and effective responses is also vital.323
In most developed countries the immediate response to an outbreak, especially if it is localized, is ‘stamping out’ which is carried out as quickly as possible. 167, 293 In the UK epidemic of 2001 it was attempted to kill all animals on infected properties within 24 hours and those on adjacent farms or that had contact with the infected premises within 48 hours.293 Unfortunately, the sheer extent of the outbreak quickly overwhelmed the available capacity and so this was not always achieved.
This illustrates one of the problems of emergency/contingency planning, viz. the impossibility of predetermining the initial size of an outbreak. Secondly, as has been emphasized,293 no two outbreaks are alike and therefore the ability to adapt responses early to prevailing circumstances is important. An illustration of this flexibility is reflected in an account on the responses to the FMD outbreaks that occurred in South Africa in 2000/01.56
It is increasingly recognized that where large numbers of animals are involved in an outbreak, ring vaccination (i.e. vaccination around the perimeter of the outbreak focus) may be necessary to provide an immune belt between infected and susceptible populations. However, where multifocal outbreaks occur, as was the case in the UK in 2001, by the time infection is recognized, it becomes very difficult to differentiate infected areas from those that are not. In such situations, ring vaccination becomes impractical and in that case extensive vaccination over a wide area may become necessary.
The first step in the event of an outbreak is to confirm the clinical diagnosis by laboratory tests, which include typing and determination of the lineage of virus involved and testing the outbreak virus against antisera to available vaccine strains to establish which would be most appropriate. A quarantine area should immediately be declared and enforced around the affected area. This is initially usually applied to an administratively defined area such as a magisterial district or department but, if the situation so dictates, this area may be reduced or increased in size. Within the EU and some individual countries the sizes of various control zones round an infected focus is predetermined by statute (e.g. 10 km). Whether this is a sensible approach or not is a moot point because it limits the latitude for those with the responsibility of managing the outbreak for making decisions that are based on the epidemiological merits of the case. On the other hand, it is helpful in avoiding initial uncertainty and confusion in the absence of clear-cut data.
It is important to impose an immediate and binding ‘standstill’ on the movement of susceptible animals over a wide geographical area (how wide will depend on circumstances) as soon as the outbreak is diagnosed. This will help ensure that the disease does not spread before the control/eradication measures are instituted. It is claimed by some that the failure of the UK authorities to proclaim a country-wide standstill immediately the outbreak of 2001 was diagnosed contributed to the duration and spread of the outbreak.
Thereafter, it is important to establish the exact extent of spread by inspecting all herds within the quarantine area. It is essential to ensure that inspectors do not themselves spread the infection. Thus the policy should be to ensure strict separation of personnel working inside the affected area and those inspecting areas considered ‘clean’. For this reason, it is advisable that inspection should start at the periphery of the quarantined area and progress inwards towards the infection focus. Once the size of the outbreak has been ascertained the extent of the quarantine area can be re-assessed.
Continuing inspection both inside and outside the focus is, however, essential in order to establish the clinical end-point and any subsequent transmission to previously uninfected localities.
In southern Africa it is traditional to erect around the infected area a cordon that makes use of natural physical barriers such as rivers, as well as fences which may have to be erected specifically for this purpose. It is necessary that the cordon be staffed along its entire length (not only at road entrances) to prevent unauthorized movement of people, animals and animal products in and out of the control area. In particularly dangerous localities a double cordon may be advisable.
Ideally, an animal-free zone outside the cordon, between infected and ‘clean’ areas, should be established to prevent contact between infected and non-infected animals. However, this is not always possible under the conditions that prevail in southern Africa.
Early in the eradication campaign a study of permits or other records relating to animal movements into and out of the affected area before the imposition of control measures should be made. This may reveal the source of infection as well as possible movement of infected animals out of the affected area and enable them to be traced.
As soon as the extent of the infected focus has been established, the strategy for eradication of the infection needs to be decided upon. This could involve ‘stamping out’ of the infection focus or ring vaccination, which will depend on many factors including the epidemiological circumstances, predetermined policy and financial and logistical resources. This usually involves a political decision, which is likely to be influenced by a whole range of socio-economic factors.
If ring vaccination is decided upon, immunization with vaccine containing the appropriate virus strain should commence immediately, both within the infection focus to immunize animals not already infected, and in the rest of the quarantine area. Different teams for the infected and ‘clean’ areas should be used for this purpose to ensure that vaccinators do not spread the infection. If conventionally adjuvanted vaccine (alhydrogel/saponin) is used a second inoculation should be administered to all ruminants two or three weeks later, but pigs should only be immunized if oil-adjuvanted vaccines are available. Booster immunizations should be given to all animals every three months for the duration of the outbreak.
If no spread has occurred in the quarantine area within 6 to 12 weeks of the clinical end-point it can be accepted that the outbreak has been contained. At that point or earlier, if vaccination has been applied, a decision as to what to do with the vaccinated animals will have to be made. This will be determined to a large extent by the importance of international trade to the country or region concerned. The OIE, as mentioned above, currently recommends (although this is under review) that trading partners favour countries or zones that slaughter vaccinated livestock and so can lay claim again to ‘freedom from FMD without vaccination’.241
If the vaccinated animals are not slaughtered once the outbreak has been shown to have come to an end, precisely what should be allowed as far as the subsequent movement of potential carriers is concerned, is a matter over which disagreement exists. Strictly, carriers are a potential source of infection for up to three years after vaccination.323 Serological screening, including the use of tests for antibodies to NSPs, may be sufficient to show that the danger of carriers being present within the area vaccinated is minimal. On the other hand, the experience in southern Africa, as in other parts of the world, is that carrier cattle have rarely, if ever, been responsible for initiating outbreaks.308
It is vital that all those involved in, and affected by, the control measures applied to control/eradication of FMD outbreaks be fully informed (and consulted if time allows) as to what the rules are and why they are necessary. Unless their co-operation can be gained, effective control/eradication is unlikely to be easily achieved.
Control of disease in endemic areas
WhereFMDis endemic or occurs sporadically in the livestock population of a large country or region, routine vaccination of the cattle population on an annual or biannual basis combined with the control of livestock movement and marketing, as well as effective responses to outbreaks when they occur (vaccination within and around the outbreak focus accompanied by quarantine of infected and in-contact animals), is generally applied. The experience of western Europe and, more recently, that of the southern cone of South America shows that this can be highly effective in reducing the prevalence of infection to low levels and even eliminating the infection altogether. However, without the co-operation and active participation of the livestock owners and associated industries such policies are usually not effective because the country or region concerned usually does not have a veterinary service large enough to do all that is necessary in addition to police control activities. It is also vital that infection is not reintroduced from neighbouring countries or regions where FMD is less effectively controlled. To aid countries in such situations the OIE provides for recognition of ‘FMD freedom with vaccination’ as an intermediate stage in progress towards full ‘freedom from FMD infection without vaccination’.241 The problem in such situations is to be sure that routine vaccination has effectively arrested circulation of FMDV and is not just masking the presence of infection by suppression of its clinical manifestation. This, together with the low probability that carrier animals may transmit the infection were they to come in contact with susceptible animals, complicates the use of vaccination as a control strategy for countries that wish to export livestock and livestock products to FMD-free countries. However, as mentioned above (see Epidemiology and Diagnosis), the development of vaccines that do not contain non-structural viral proteins and of tests capable of differentiating between antibody responses to structural and non-structural viral proteins, has provided a significant step forward in certification with respect to virus circulation in vaccinated populations.
In regions of sub-Saharan Africa where wildlife, particularly African buffalo, provide a reservoir of infection for the SAT types of virus, there is the constant threat that livestock (in effect cattle) will be infected from this source. Southern African countries, over several decades, have developed complicated but effective ways to deal with this problem. Some (e.g. South Africa, Botswana and Namibia) have obtained recognition from the OIE for large parts of the country concerned (zones) as being ‘free from FMD without vaccination’. This has involved:
- biannual (in the case of Namibia it is only annual) vaccination of cattle against SAT1 and 2 (sometimes also SAT3) types in areas where infected buffalo and cattle interface;
- maintenance of surveillance zones between the vaccination (buffer) zones and FMD-free zones of the country concerned. Within surveillance zones routine clinical inspection of all cattle is conducted on a weekly, fortnightly or monthly basis. This generally requires each owner within the area concerned to produce all his/her cattle for inspection and the numbers of cattle inspected and their owners’ details are recorded. These activities generally centre on dip tanks to which cattle are brought for routine dipping on a regular basis to counter tick-borne diseases; and
- movement of cattle — sheep and goats are variably included— is controlled within the infected (defined by the presence of infected buffalo), buffer and surveillance zones by permit systems. Movement of livestock out of these zones into the free zone is precluded (other than transport to abattoirs for direct slaughter).
A vital but contentious adjunct to livestock movement control is the use of fencing not only to regulate contact between domestic animals but also that between wildlife and domestic livestock. In southern Africa the historic approach has been to separate domestic livestock from wildlife by means of fences (double fence lines that preclude direct contact between animals on either side of the fence are increasingly being used).312 In addition, cattle in localities adjacent to wildlife areas are usually vaccinated biannually against FMD. This generally has been successful in preventing transmission of FMD from wildlife to livestock.330 However, the use of fencing has been severely criticized by conservationists because the fences sometimes have blocked migration routes and access of wildlife to water, resulting in ecological disturbance and wildlife mortality.245 The necessity for the use of fencing is increasingly being questioned; the argument being that vaccination alone should be sufficient. Conversely, the contribution of fencing to effective FMD control is increasingly apparent.346
A study on the environmental, social and economic impact of fences aimed at animal disease control in Ngamiland (north-western Botswana) has highlighted the complexities of the issues involved. Essentially, the economic, social and environmental costs and benefits are viewed differently by the various affected parties, e.g. local communities with a livestock tradition, commercial farming interests, the tourist industry and the environmental lobby.292 Wildlife management may be further complicated by restrictions placed on areas where particular species, such as buffalo, may be farmed or ranched, on wildlife translocation, and on the distribution of products derived from wildlife, such as meat, hides and trophies.
There is no doubt that the combination of measures applied to FMD control in southern Africa has been highly successful over the last 30 to 40 years in enabling maintenance of FMD-free areas and therefore permitting international trade in livestock products. This success seems to be threatened, however, by recent developments in some of these countries.
Decentralization of veterinary authority has meant that the national veterinary service is unable to exert the same degree of enforcement of animal health regulations as was formerly the case. A contributing factor is political developments that have resulted in a decrease of discipline in rural areas and among livestock owners that has resulted in long-established disease control regulations being increasingly ignored without penalty.
Whatever the FMD status of a country, it is essential to preclude the importation of FMDV—if only to make certain that ‘foreign’ virus types and subtypes are not introduced. It is therefore necessary to control and monitor the import of susceptible animals and their products and to ensure that all access routes into a country are covered—road, rail, air and sea. The proper testing and quarantining of all imported cloven-hoofed animals from potentially infected regions is absolutely essential. It is also important that wastes from ships, aeroplanes and trains be decontaminated immediately and not used as swill for pigs (Figure 125.5).
References
- ACHARYA, R., FRY, E., STUART, D., FOX, D., ROWLANDS, D. & BROWN, F., 1989. The three-dimensional structure of foot-and-mouth disease virus at 2.9A revolution. Nature, 337, 709–716.
- ACHARYA, A., FRY, E., STUART, D., FOX, G., ROWLANDS, D. & BROWN, F., 1990. The structure of foot-and-mouth disease virus: Implications for its physical and biological properties. Veterinary Microbiology, 23, 21–34.
- AIDEROS, H.A., 2002. Regional status and approaches to control/ eradication of foot-and-mouth disease in the Middle East and North Africa. Scientific and Technical Review, 21, 451–458.
- ALEXANDERSEN, S., BROTHERHOOD, I. & DONALDSON, A.I., 2002. Natural aerosol transmission of foot-and-mouth disease aerosol to pigs: Minimal dose for strain O1 Lausanne. Epidemiology and Infection, 128, 301–312.
- ALEXANDERSEN, S. & DONALDSON, A.I., 2002. Further studies to quantify the dose of natural aerosols of foot-and-mouth disease virus for pigs. Epidemiology and Infection, 128, 313–323.
- ALEXANDERSEN, S., OLEKSIEWICZ, M.B. & DONALDSON, A.I., 2001. Early pathogenesis of foot-and-mouth disease in pigs infected by contact: A quantitative time course study using TaqMan RT-PCR. Journal of General Virology, 82, 747–755.
- ALEXANDERSEN, S. ZHANG, Z. & DONALDSON, A.I., 2001. Further studies to quantify the dose of natural aerosols of foot-and-mouth disease virus for pigs. Epidemiology and Infection, 128, 313–323.
- ALLENSPACH, V., 1950. Foot-and-mouth disease in the zoological gardens of Zu¨rich [in German]. Schweizer Archiv fu¨r Tierheilkunde, 92, 42–47.
- AMADORI, M. & MELEGARI, M., 1987. Humoral and cellular parameters of experimental foot-and-mouth disease in heifers. Journal of Veterinary Medicine, B34, 480–488.
- ANDERSON, E.C., ANDERSON, J., DOUGHTY, W.J. & DREVMO, S., 1975. The pathogenicity of bovine strains of foot-and-mouth disease virus for impala and wildebeest. Journal of Wildlife Diseases 11, 248–255.
- ANDERSON, E.C., DOUGHTY, W.J. & ANDERSON, J., 1976. The role of sheep and goats in the epizootiology of foot-and-mouth disease in Kenya. Journal of Hygiene, 76, 395–402.
- ANDERSON, E.C., FOGGIN, C., ATKINSON, M., SORENSON, K.J., MADEKUROZVA & NQINDI, J., 1993. The role of wild animals other than buffalo, in the current epidemiology of foot-and-mouth disease in Zimbabwe. Epidemiology and Infection, 111, 559–563.
- ANON., 1972-86, FAO-WHO-OIE Animal Health Year Books. bellver-gallent, m., (ed.). International Office of Epizootics, Food and Agriculture Organisation of the United Nations, World Health Organisation.
- ANON., 1986. Investigation on the possible effect of electrical stimulation on pH and survival of foot-and-mouth disease virus in meat and offals from experimentally infected animals. Agriculture, Report EUR 10048 en. Luxembourg: Commission of the European Communities.
- ANON., 1992. Conclusions of the Research Subcommittee of the International Embryo Transfer Society Import/Export Committee. Scientific and Technical Review, 11, 938–939.
- ARAÚJO, J.P., MONTASSIER, H.J. & PINTO, A.A., 2002. Extensive antigenic and genetic variation among foot-and-mouth disease type A viruses isolated from the 1994 and 1995 foci in São Paulo, Brazil. Veterinary Microbiology, 84, 15–27.
- AYRES, E., CAMERON, E., KEMP, R., LEITCH, H., MOLLISON, A., MUIR, I., REID, H., SMITH, D. & SPROAT, J., 2001. Oral lesions of foot-and-mouth disease in sheep and cattle in Dumfries and Galloway. The Veterinary Record, 148, 720–723.
- BACHRACH, H.L., 1968. Foot-and-mouth disease. Annual Review of Microbiology, 22, 201–244.
- BACHRACH, H.L., 1977. Foot-and-mouth disease virus: Properties, molecular biology and immunogenicity. In: ROMBERGER, J.A., (ed.). Beltsville Symposia in Agricultural Research. I. Virology in Agriculture. Montclair: Allanheld, Osmun & Co.
- BACHRACH, H.L., BREESE, S.S., CALLIS, J.J., HESS, W.R. & PATTY, R.E., 1957. Inactivation of foot-and-mouth disease virus by pH and temperature changes and by formaldehyde. Proceedings of the Society of Experimental Biology and Medicine, 95, 147–152.
- BACHRACH, H.L. MOORE, D.M., MCKERCHER, P.D. & POLATNICK, J., 1975. Immune and antibody responses to an isolated capsid protein of foot and mouth disease virus. Journal of Immunology, 115, 1635–1641.
- BACHRACH, H.L., TRAUTMAN, R. & BREESE, S.S., 1964. Chemical and physical properties of virtually pure foot-and-mouth disease virus. American Journal of Veterinary Research, 25, 333–342.
- BARLOW, D.F., 1972. The aerosol stability of a strain of foot-and-mouth disease virus and the effects on its stability of precipitation with ammonium sulphate or other chemicals. General Virology, 15, 17–24.
- BARTELING, S.J., 2002. Development and performance of inactivated vaccines against foot-and-mouth disease. Scientific and Technical Review, 21, 577–588.
- BARTELS, S. & CLAASSEN, P., 1936. Wild game in the etiology of foot-and-mouth disease [in German]. Berliner Tierärztliche Wochenschrift, 52, 230–233.
- BASTOS, A.D.S., 1998. Detection and characterization of foot-and-mouth disease virus in sub-Saharan Africa. Onderstepoort Journal of Veterinary Research, 65, 37–47.
- BASTOS, A.D.S., 2001. Molecular epidemiology and diagnosis of SAT-type foot-and-mouth diseases in southern Africa. PhD thesis, University of Pretoria, Pretoria, South Africa. pp. 148.
- BASTOS, A.D.S., BERTSCHINGER, H.J., CORDEL, C., VAN VUUREN, C. DE W.J., KEET, D., BENGIS, R.G., GROBLER, D.G. & THOMSON, G.R., 1999. Possibility of sexual transmission of foot-and-mouth disease from African buffalo to cattle. The Veterinary Record, 145, 77–79.
- BASTOS, A.D.S, BOSHOFF, C.I., KEET, D.F., BENGIS, R.G. & THOMSON, G.R., 2000. Natural transmission of foot-and-mouth disease virus between African buffalo (Syncerus caffer) and impala (Aepyceros melampus) in the Kruger National Park, South Africa. Epidemiology and Infection, 124, 591–598.
- BASTOS, A.D.S., HAYDON, D.T., FORSBERG, R., KNOWLES, N.J., ANDERSON, E.C., BENGIS, R.G., NEL, L.H. & THOMSON, G.R., 2001. Genetic heterogeneity of SAT-1 type foot-and-mouth disease viruses in southern Africa. Archives of Virology, 146, 1537–1551.
- BASTOS, A.D.S., HAYDON, D.T., SANGARE, O., BOSHOFF, C.I., EDRICH, J.L. & THOMSON, G.R., 2003. The implications of viral diversity in the SAT2 serotype for control of foot-and-mouth disease in sub-Saharan Africa. Journal of General Virology, 84, 1959–1606.
- BAXT, B. & BARACH, H.L., 1982. The adsorption and degradation of foot-and-mouth disease virus by isolated BHK 21 cell plasma membranes. Virology, 116, 391–405.
- BAXT, B. & BECKER, Y., 1990. The effect of peptides containing the arginine-glycine-aspartic acid sequence on the adsorption of foot-and-mouth disease virus to tissue culture cells. Virus Genes, 4, 73–83.
- BEARD, C.W. & MASON, P.W., 2000. Genetic determinants of altered virulence of Taiwanese foot-and-mouth disease virus. Journal of Virology, 74, 987–991.
- BECK, E., FORSS, S., STREBEL, K., CATTANEO, R. & FEIL, G., 1983. Structure of the FMDV translation initiation site and of the structural proteins. Nucleic Acids Research, 11, 7873–7885.
- BECK, E. & STROHMAIER, K., 1986. Subtyping of European FMDV outbreak strains by nucleotide sequence determination. Paper presented at the 17th Conference of the O.I.E. Foot-and-Mouth Disease Commission, Paris, 1–3 October, 1986.
- BEKKER, J.G., DE KOCK, G. & QUINLAN, J.B., 1934. The occurrence and identification of blue tongue in cattle and the so-called pseudofoot- and-mouth disease in South Africa. Onderstepoort Journal of Veterinary Research, 2, 393–507.
- BELSHAM, G.J. & BRANGWYN, J.K., 1990. A region of the 5’ non-coding region of foot-and-mouth disease virus RNA direct efficient internal initiation of protein synthesis within cells: Involvement with the role of L protease in translational control. Journal of Virology, 64, 5389–5395.
- BELSHAM, G.J., MCINERNEY, G.M. & ROSS-SMITH, N., 2000. Foot-and-mouth virus 3C protease induces cleavage of translation initiation factors eIF4A and iEF4G within infected cells. Journal of Virology, 74, 272–280.
- BENGIS, R.G. & THOMSON G.R., 1992. P.O. Box 12, Skukuza, South Africa, and P.O. Box 30786, Nairobi, Kenya. Unpublished data.
- BERGMANN, I.E., 2002. Validation of the I-ELISA 3ABC/EITB system for use in foot-and-mouth disease surveillance. In: Proceedings of the International Symposium on Foot-and-Mouth Control Strategies. 2–5 June 2002. Foundation Merieux, Lyon, France.
- BERGMANN, I.E., MALIRAT, V., AUGE DE MELLO, P. & GOMES, I., 1996. Detection of foot-and-mouth disease viral sequences in various fluids and tissues during persistence of the virus in cattle. American Journal of Veterinary Research, 57, 134–137.
- BLACKWELL, J.H., 1980. Internationalism and the survival of foot-and-mouth disease in cattle and food products. Journal of Dairy Science, 63, 1019–1030.
- BLACKWELL, J.H., 1984. Foreign animal disease agent survival in animal products: Present developments. Journal of the American Veterinary Medical Association, 184, 674–679.
- BLACKWELL, J.H., MCKERCHER, P.D., KOSIKOWSKI, F.V., CARMICHAEL, L.E. & GOREWIT, R.C., 1983. Histological and histochemical characterisation of mammary gland tissue of cows infected with foot-and-mouth disease by contact exposure. Research in Veterinary Science, 35, 106–113.
- BLACKWELL, J.H., WOOL, S. & KOSIKOWSKI, F.V., 1981. Vesicular exocytosis of foot-and-mouth disease virus from mammary gland secretory epithelium of infected cows. Journal of General Virology, 56, 207–212.
- BOLDRINI, G.M., 1978. Vaccination and the control of foot-and-mouth disease. The Veterinary Record, 102, 194–198.
- BOSCH (VOSLOO), w., 1989. Unpublished data. Exotic Disease Division, ARC-Onderstepoort Veterinary Institute, Onderstepoort 0110, South Africa.
- BROOKSBY, J.B., 1950. Strains of the virus of foot-and-mouth disease showing natural adaption to swine. Journal of Hygiene, 47, 184–195.
- BROOKSBY, J.B., 1982. Portraits of viruses: Foot-and-mouth disease virus. Intervirology, 18, 1–23.
- BROWN, F., 1972. Comments on immunology of foot-and-mouth disease in swine. Journal of the American Veterinary Medical Association, 160, 526–527.
- BROWN, F., 1985. Antigenic structure of foot-and-mouth disease virus. In: van regenmortel, m.h.v. & neurath, a.r., (eds). Immunochemistry of Viruses. The Basis for Serodiagnosis and Vaccines. Elsevier Scientific Publishers B.V. (Biomedical Division).
- BROWN, C.C., OLANDER, H.J. & MEYER, R.F., 1995. Pathogenesis of foot-and-mouth disease in swine, studied by in-situ hybridization. Journal of Comparative Pathology, 113, 51–58.
- BROWN, C.C., PICCONE, M.E., MASON, P.W., MCKENNA, T.S. & GRUBMAN, M.J., 1996. Pathogenesis of wild-type and leaderless foot-and-mouth disease virus in cattle. Journal of Virology, 70, 5638–5641.
- BROWN, F. & WILD, T.F., 1966. The effect of heat on the structure of foot-and-mouth disease virus and the viral ribonucleic acid. Biochemica et Biophysica Acta, 119, 301–308.
- BRU¨CKNER, G.K., VOSLOO, W., DU PLESSIS, B.J.A., KLOECK, P.E.L.G., CONNOWAY, L., EKRON, M.D., WEAVER, D.B., DICKASON, C.J., SCHREUDER, F.J., MARAIS, T. & MOGAJANE, M.E., 2002. Foot-and-mouth disease: The experience of South Africa. Scientific and Technical Review, 21, 751–764.
- BUNJEBARA, K., 2002. OIE, 12 Rue de Prony, 75017, Paris. Personal communication.
- BURBURY, Y.M., 1928. VII. Survival of foot-and-mouth disease virus when dried upon various materials. Third Progress Report of the Foot-and-Mouth Disease Research Committee. London: His Majesty’s Stationery Office.
- BURROWS, R., 1966. Studies on the carrier state of cattle exposed to foot-and-mouth disease virus. Journal of Hygiene, 64, 81–90.
- BURROWS, R., MANN, J.A., GARLAND, A.J.M., GREIG, A. & GOODRIDGE, D., 1981. The pathogenesis of natural and simulated natural foot-and-mouth disease infection in cattle. Journal of Comparative Pathology, 91, 599–609.
- BURROWS, R., MANN, J.A., GRIEG, A., CHAPMAN, W.G. & GOODRIDGE, D., 1971. The growth and persistence of foot-and-mouth disease virus in the bovine mammary gland. Journal of Hygiene, 69, 307–321.
- CALLIS, J.J., 1984. Foot-and-mouth disease. In: Foreign Animal Diseases: Their Prevention, Diagnosis and Control. Richmond, Virginia: Committee on Foreign Animal Diseases of the United States Animal Health Association.
- CARILLO, E.C., GIACHETTI, C. & CAMPOS, R.H., 1984. Effect of lysosomotropic agents on the foot-and-mouth disease virus replication. Virology, 135, 542–545.
- CARTWRIGHT, B., CHAPMAN, W.G. & SHARPE, R.T., 1982. Stimulation of heterotypic antigens of foot-and-mouth disease virus antibodies in vaccinated cattle. Research in Veterinary Science, 32, 338–342.
- CASSAGNE, M.-H., 2002. Managing compensation for economic losses in areas surrounding foot-and-mouth disease outbreaks: The response of France. Scientific and Technical Review, 21, 823–829.
- CATLEY, A., OKOTH, S., OSMAN, J., FISON, T., NJIRU, Z., MWANGI, J., JONES, B.A. & LEYLAND, T.J., 2001. Participatory diagnosis of a chronic wasting disease in cattle in southern Sudan. Preventive Veterinary Medicine, 45, 1–21.
- CHMITLIN, I. & MOUTOU, F., 2002. Foot-and-mouth disease: Lessons to be learned from the experience of France. Scientific and Technical Review, 21, 731–737.
- CHOW, M., NEWMAN, J.F.E., FILMAN, D., HOGLE, J.M., ROWLANDS, D.J. & BROWN, F., 1987. Myristylation of picornavirus capsid protein VP4 and its structural significance. Nature, 327, 482–486.
- CLARKE, B.E., BROWN, A.L., CURREY, K.M., NEWTON, S.E., ROWLANDS, D.J. & CARROLL, A.R., 1987. Potential secondary and tertiary structure in the genomic RNA of foot-and-mouth disease virus. Nucleic Acids Research, 15, 7067–7079.
- COHRS, P. & WEBER-SPRINGE, W., 1939. Maul- und Klauenseuche beim Reh und Hirsch (Foot-and-mouth disease in roe deer and red deer). Deutsche Tierärztliche Wochenschrift, 47, 97–103.
- CONDY, J.B. & HEDGER, R.S., 1974. The survival of foot-and-mouth disease virus in African buffalo with non-transferance of infection to domestic cattle. Research in Veterinary Science, 16, 182–185.
- CONDY, J.B. & HEDGER, R.S., 1978. Experiences in the establishment of a herd of foot-and-mouth disease free African buffalo (Syncerus caffer). South African Journal of Wildlife Research, 8, 87–89.
- CONDY, J.B., HEDGER, R.S., HAMBLIN, C. & BARNETT, I.T.R., 1985. The duration of the foot-and-mouth disease virus carrier state in African buffalo (i) in the individual animal and (ii) in a free-living herd. Comparative Immunology, Microbiology and Infectious Diseases, 8, 257–265.
- COTTRAL, G.E., 1969. Persistence of foot-and-mouth disease virus in animals, their products and the environment. Bulletin de L’Office International des Epizooties, 71, 549–568.
- COTTRAL, G.E., COX, B.F. & BALDWIN, D.E., 1960. The survival of foot-and-mouth disease virus in cured and uncured meat. American Journal of Veterinary Research, 21, 288–297.
- CRISPIN, S.M., ROGER, P.A., O’HARE, H. & BINNS, S.H., 2002. The 2001 foot-and-mouth disease epidemic in the United Kingdom: animal welfare perspectives. Scientific and Technical Review, 21, 877–883.
- CROWTHER, J.R., ROWE, C.A. & BUTCHER, R., 1993. Characterization of monoclonal antibodies against a type SAT-2 foot-and-mouth disease virus. Epidemiology and Infection, 111, 391–406.
- CUIJPERS, M.P. & OSINGA, K.J., 2002. The position of the Dutch Farmers’ Union on lessons learned and future prevention and control of foot-and-mouth disease. Scientific and Technical Review, 21, 839–850.
- CUMMING, R.G., 1850. Five years of a Humbert’s life in South Africa. Vol. II. London: Murray.
- DALSGARD, K., 1977. The adjuvant activity of ‘Quil A’ in trial vaccination of cattle and guinea pigs against foot-and-mouth disease. Acta Veterinaria, Scandanavia, 18, 367.
- DAVID, M., TORRES, A., MEBUS, C., CARRILLO, B.J., SCHUDEL, A., FONDEVILLA, N., BLANCO VIERA, J. & MARCOVECCHIO, F.E., 1993. Further studies on foot-and-mouth disease in the llama. Proceedings of the U.S. Animal Health Association, 97, 280–285.
- DE KLERK, P.F., 2002. Carcass disposal: Lessons from the Netherlands after the foot-and-mouth disease outbreak of 2001. Scientific and Technical Review, 21, 789–796.
- DE LA RUA, R., WATKINS, G.H. & WATSON, P.J., 2001. Idiopathic mouth ulcers in sheep. The Veterinary Record, 149, 30–31.
- DE LA TORRE, J.C., MARTINEZ-SALAS, E., DIEZ, J., VILLAVERDE, F., GEBAUER, F., ROCHA, E., DAVILA, M. & DOMINGO, E., 1980. Co-evolution of cells and viruses in a persistent infection of foot-and-mouth disease virus in cell culture. Journal of Virology, 62, 2050–2058.
- DI GIROLAMO, W., SALAS, M. & LAGUENS, R.P., 1995. Role of Langerhans cells in the infection of the guinea-pig epidermis with foot-and-mouth disease virus. Archives of Virology, 83, 331–336.
- DOEL, T.R. & BACCARINI, P.J., 1981. Thermal stability of foot-and-mouth disease virus. Archives of Virology, 70, 21–32.
- DOEL, T.R., WILLIAMS, L. & BARNETT, P.V., 1994. Emergency vaccination against foot-and-mouth disease: Rate of development of immunity and its implication for the carrier state. Vaccine, 12, 592–600.
- DOMINGO, E., DIEZ, J., MARTINEZ, M.A., HERNANDEZ, J., HOLGUIN, A., BORREGO, B. & MATEU, M.G., 1993. New observations on the antigenic diversification of RNA viruses. Antigenic variation is not dependent on immune selection. Journal of General Virology, 74, 2039–2045.
- DOMINGO, E., ESCARMIS, C., MARTINEZ, M.A., MARTINEZ-SALAS, E. & MATEU, M.G., 1992. Foot-and-mouth disease virus populations are quasispecies. Current Topics in Microbiology and Immunology, 176, 33–47.
- DOMINGO, E., HOLLAND, J.J., BEIBRICHER, C. & EIGEN, M., 1995. Quasispecies: The concept and the word. In: gibbs, a., calisher, c. & garcia- arenal, f., (eds). Molecular Basis of Virus Evolution. Cambridge: Cambridge University Press. pp. 171–180.
- DOMINGO, E., MATEU, M.G., ESCARMIS, C., MARTINEZ-SALAS, E., ANDREU, D., GIRALT, E., VERAGUER, N. & FITA, I., 1996. Molecular evolution of aphthovirus. Virus Genes, 11, 197–207.
- DONALDSON, A.I., 1972. Influence of relative humidity on the aerosol stability of different strains of foot-and-mouth disease virus suspended in saliva. Journal of General Virology, 15, 25–33.
- DONALDSON, A.I., 1979. Air-borne foot-and-mouth disease. The Veterinary Bulletin, 49, 653–659.
- DONALDSON, A.I., 1983. Quantitative data on airborne foot-and-mouth disease virus: Its production, carriage and deposition. Philosophical Transactions of the Royal Society, London, B302, 529–534.
- DONALDSON, A.I., 1986–8. Institute for Animal Health, Pirbright, Surrey. Personal communications.
- DONALDSON, A.I., 1987. Foot-and-mouth disease: The principle features. Irish Veterinary Journal, 41, 325–327.
- DONALDSON, A.I., 1997. Risks of FMD through milk and dairy products. Scientific and Technical Review, 16, 117–124.
- DONALDSON, A.I. & ALEXANDERSON, S., 2002. Predicting the spread of foot-and-mouth disease by airborne virus. Scientific and Technical Review, 21, 569–575.
- DONALDSON, A.I. & FERRIS, N.P., 1975. The survival of foot-and-mouth disease virus in open air conditions. Journal of Hygiene, 74, 409–416.
- DONALDSON, A.I. & FERRIS, N.P., 1976. The survival of some air-borne animal viruses in relation to relative humidity. Veterinary Microbiology, 1, 413–420.
- DONALDSON, A.I. & FERRIS, N.P., 1978. The influence of inhibitors and antibody on the airborne survival of foot-and-mouth disease virus. Proceedings of the First International Conference on Aerobiology. Berlin: Schmidt Verlag.
- DONALDSON, A.I. & FERRIS, N.P., 1980. Sites of release of airborne foot-and-mouth disease from infected pigs. Research in Veterinary Science, 29, 315–319.
- DONALDSON, A.I., FERRIS, N.P. & WELLS, G.A.H., 1984. Experimental foot-and-mouth disease in fattening pigs, sows and piglets in relation to outbreaks in the field. The Veterinary Record, 115, 509–512.
- DONALDSON, A.I., GARLAND, A.J.M., FERRIS, N.P. & COLLEN, T., 1981. The 1975 foot-and-mouth disease epidemic in Malta. I. Experimental studies with the causal virus strain 01 Malta. British Veterinary Journal, 137, 300–307.
- DONALDSON, A.I., GIBSON, C.F., OLIVER, R., HAMBLIN, C. & KITCHING, R.P., 1987. Infection of cattle by airborne foot-and-mouth disease virus: Minimal doses with 01 and SAT 2 strains. Research in Veterinary Science, 43, 339–346.
- DU TOIT, P.J., 1932. Foot-and-mouth disease in Southern Rhodesia. Journal of the South African Veterinary Medical Association, 3, 26–37.
- ESTERHUYSEN, J.J., THOMSON, G.R., ASHFORD, W.A., LENTZ, D.W., GAINARU, M.D., SAYER, A.J., MEREDITH, C.D., VAN RENSBURG, D.J. & PINI, A., 1988. The suitability of a rolled BHK21 monolayer system for the production of vaccines against the SAT types of foot-and-mouth disease virus. 1. Adaption of virus isolates to the system, immunogen yields achieved and assessment of serotype cross reactivity. Onderstepoort Journal of Veterinary Research, 55, 77–84.
- FALCONER, J., 1972. The epizootiology and control of foot-and-mouth disease in Botswana. The Veterinary Record, 91, 354–359.
- FALK, M.M., GRIGERA, P.R., BERGMAN, I.E., ZIBERT, A., MULTHAUP, G. & BECK, E., 1990. Foot-and-mouth disease virus protease 3C induces specific proteolytic cleavage of host cell histone H3. Journal of Virology, 64, 748–756.
- FALK, M.M., SOBRINO, F. & BECK, E., 1992. VPg gene amplification correlates with infective particle formation in foot-and-mouth disease virus. Journal of Virology, 66, 2251–2260.
- FARES, M.A., MOYA, A., ESCARMIS, C., BARANOWSKI, E., DOMINGO, E. & BARRIO, E., 2001. Evidence for positive selection in the capsid protein-coding region of the foot-and-mouth disease virus (FMDV) subjected to experimental passage regimens. Molecular Biology and Evolution, 18, 10–21.
- FENNER, F., BACHMANN, P.A., GIBBS, E.P.J., MURPHY, F.A., STUDDART, M.J. & WHITE, D.O., 1987. Veterinary Virology. Orlando, New York, London, Sydney, Tokyo, Toronto: Academic Press Inc.
- FERRIS, N.P., BULUT, A.N., RENDLE, T., DAVIDSON, F. & MACKAY, D.K.J., 2000. A solid phase competition ELISA for measuring antibody to foot-and-mouth disease virus. Session of the Research Group of the Standing Technical Committee: European Commission for the Control of Foot-and-Mouth Disease. September 2000. Borovets, Bulgaria.
- FERRIS, N.P. & DAWSON, M., 1988. Routine application of enzyme-linked immunosorbent assay in comparison with complement fixation for the diagnosis of foot-and-mouth and swine vesicular diseases. Veterinary Microbiology, 16, 201–209.
- FERRIS, N.P. & DONALDSON, A.I., 1992. The World Reference Laboratory for Foot-and-Mouth Disease: A review of 33 years of activity (1958–1991). Scientific and Technical Review, 11, 657–684.
- FOGEDBY, E.G., MALINQUIST, W.A., OSTEEN, O.L. & JOHNSON, M.L., 1960. Air-borne transmission of foot-and-mouth disease virus. Nordisk Veterinaermedicin, 12, 490–498.
- FORMAN, A.J. & GARLAND, A.J.M., 2002. Foot-and-mouth disease: the future of vaccine banks. Scientific and Technical Review, 21, 601–612.
- FORSS, S., STREBEL, K., BECK, E. & SCHALLER, H., 1984. Nucleotide sequence and genome organisation of foot-and-mouth disease virus. Nucleic Acids Research, 12, 6587–6601.
- FOX, G., PARRY, N., BARNETT, P.V., MCGINN, B., ROWLANDS, D.J. & BROWN, F., 1989. The cell attachment site on foot-and-mouth disease virus includes the amino acid sequence RGD (arginine-glycine-aspartic acid). Journal of General Virology, 70, 625–637.
- FRANCE, L.L., PIATTI, P.G., NEWMAN, J.F.E., TOTH, I., GIBBONS, W.A. & BROWN, F., 1994. Circular dichroism, molecular modelling and serology indicate that the structural basis of antigenic variation in foot-and-mouth disease virus is helix formation. Proceedings of the National Academy of Science of the USA, 91, 8442–8446.
- FRANCIS, M.J., 1986. The humoral immune response of young animals to foot-and-mouth disease vaccination. Foot and Mouth Disease Bulletin, 23, 1–44. Pirbright: Coopers Animal Health Ltd.
- FRANCIS, M.J. & BLACK, L., 1983. Antibody response in nasal fluid and serum following foot-and-mouth disease infection or vaccination. Journal of Hygiene, 91, 329–334.
- FRANCIS, M.J. & BLACK, L., 1984. The effect of vaccination regimen on the transfer of foot and mouth disease antibodies from the sow to her piglets. Journal of Hygiene, 93, 123–131.
- FRANCIS, M.J. & BLACK, L., 1986. Response of young pigs to foot-and-mouth disease oil emulsion vaccine in the presence and absence of maternally derived neutralizing antibodies. Research in Veterinary Science, 41, 33–39.
- FRANCIS, M.J., HASTINGS, G.Z., SYRED, A.D., MCGINN, B., BROWN, F. & ROWLANDS, D.J., 1987. Non-responsiveness to a foot-and-mouth disease virus peptide overcome by addition of foreign helper T-cell determinants. Nature, 300, 168–170.
- FRANCIS, M.J., OULDRIDGE, E.J. & BLACK, L., 1983. Antibody response in bovine pharyngeal fluid following foot-and-mouth disease vaccination and/or exposure to live virus. Research in Veterinary Science, 35, 206–210.
- FREIBERG, B., RAHMAN, M.M. & MARQUARDT, O., 1999. Genetical and immunological analysis of recent Asian type A and O foot-and-mouth disease virus isolates. Virus Genes, 19, 167–182.
- GAILIUNAS, P. & COTTRAL, G.E., 1966. Presence and persistence of foot-and-mouth disease virus in bovine skin. Journal of Bacteriology, 91, 2333–2338.
- GAINARU, M.D., 1988. Veterinary Research Institute, Onderstepoort, South Africa. Unpublished data.
- GAINARU, M.D., THOMSON, G.R., BENGIS, R.G., ESTERHUYSEN, J.J., BRUCE, W. & PINI, A., 1986. Foot-and-mouth disease and the African buffalo (Syncerus caffer). II. Virus transmission and excretion during acute infection. Onderstepoort Journal of Veterinary Research, 53, 75–85.
- GEBAUER, F., DE LA TORRE, J.C., GOMES, I., MATEN, M.G., BARAHONA, H., TIRABOSCHI, B., BERGMANN, I., DE MELLO, P.A. & DOMINGO, E., 1988. Rapid selection of genetic and antigenic variants of foot-and-mouth disease during persistence in cattle. Virology, 62, 2041–2049.
- GEERING, W.A., 1967. Foot-and-mouth disease in sheep. Australian Veterinary Journal, 43, 485–489.
- GIBBENS, J.C., SHARPE, C.E., WILESMITH, J.W., MANSLEY, L.M., MICHALOPOULOU, E., RYAN, J.B.M. & HUDSON, M., 2001. Descriptive epidemiology of the 2001 foot-and-mouth disease epidemic in Great Britain. The Veterinary Record, 149, 729–743.
- GIBSON, C.F. & DONALDSON, A.I., 1986. Exposure of sheep to natural aerosols of foot-and-mouth disease virus. Research in Veterinary Science, 41, 45–49.
- GIBSON, C.F., DONALDSON, A.I. & FERRIS, N.P., 1984. Response of sheep vaccinated with large doses of vaccine to challenge by airborne foot-and-mouth disease virus. Vaccine, 2, 157–161.
- GIRAUDO, A.T., GOMES, I., AUGÉ DE MELLO, P., BECK, E., LA TORRE, J.L., SCODELLER, E.A. & BERGMANN, I.E., 1988. Behaviour of intertypic recombinants between virulent and attenuated aphthovirus strains in tissue culture and cattle. Journal of Virology, 62, 3789–3794.
- GLOSTER, J., SELLERS, R.F. & DONALDSON, A.I., 1982. Long distance transport of foot-and-mouth disease virus over the sea. The Veterinary Record, 110, 47–52.
- GOMES, I. & ROSENBERG, F.J., 1984/85. A possible role of capybaras (Hydrochoerus hydrochoeris hydrochoeris) in foot-and-mouth disease (FMD) endemicity. Preventive Veterinary Medicine, 3, 197–205.
- GRAHAM, A.M., 1967. Foot-and-mouth disease. The Veterinary Record, (Special Issue—Information Supplement), 84, 25–30.
- GRAVES, J.H., MCKERCHER, P.D., FARRIS, H.E. & COWAN, K.M., 1968. Early response of cattle and swine to inactivated foot-and-mouth disease vaccine. Research in Veterinary Science, 9, 35.
- GREENBERG, B., 1973. In: Flies and disease. Biology and disease transmission. Vol. II. New Jersey, USA: Princeton University Press.
- GREGG, D.A., MEBUS, C.A. & SCHLAFER, D.H., 1995. African swine fever interference with foot-and-mouth disease infection and seroconversion in pigs. Journal of Veterinary Diagnostics and Investigation, 7, 31–43.
- GREGG, D.A., SCHLAFER, D.H. & MEBUS, C.A., 1995. African swine fever virus infection of skin-derived dendritic cells in vitro causes interference with subsequent foot-and-mouth disease virus infection. Journal of Veterinary Diagnostics and Investigation, 7, 44–51.
- GRIGERA, P.R., TISMINETZSKY, S.G., LEBENDIKER, M.B., PERIOLO, O.H. & LA TORRE, J.L., 1988. Presence of a 43-kDa host-cell polypeptide in purified aphthovirions. Virology, 165, 584–588.
- GROSSO, A.M., 1957. Foot-and-mouth disease in the Buenos Aires Zoo in the past five years [in Spanish]. Gaceta Veterinaria (Buenos Aires), 19, 54–55.
- GUTIÉRREZ, A., MARTINEZ-SALAS, E., PINTADO, B. & SOBRINO, F., 1994. Specific inhibition of aphthovirus infection by RNAs transcribed from both 5’ and 3’ noncoding regions. Journal of Virology, 68, 7426–7432.
- HAMBLIN, C., BARNETT, I.T.R. & CROWTHER, J.R., 1986. A new enzyme-linked immunosorbent assay (ELISA) for the detection of antibodies against foot-and-mouth disease virus. II. Application. Journal of Immunological Methods, 93, 123–129.
- HAMBLIN, C., BARNETT, I.T.R. & HEDGER, R.S., 1986. A new enzyme-linked immunosorbent assay (ELISA) for the detection of antibodies against foot-and-mouth disease virus. I. Development and method of ELISA. Journal of Immunological Methods, 93, 115–121.
- HAMBLIN, C., KITCHING, R.P., DONALDSON, A.I., CROWTHER, J.R. & BARNETT, I.T.R., 1987. Enzyme-linked immunosorbent assay (ELISA) for the detection of antibodies against foot-and-mouth disease virus. III. Evaluation of antibodies after infection and vaccination. Epidemiology and Infection, 99, 733–744.
- HARGREAVES, S., 1992. Director of Veterinary Services, P.O. Box 8012, Causeway, Harare, Zimbabwe. Personal communications.
- HARGREAVES, S.K., FOGGIN, C.M., ANDERSON, E.C., BASTOS, A.D.S., THOMSON, G.R., FERRIS, N. & KNOWLES, N. An investigation into the source and spread of foot-and-mouth disease virus from a wildlife conservancy in Zimbabwe. Scientific and Technical Review. In press.
- HAYDON, D.T., BASTOS, A.D., KNOWLES, N.J. & SAMUEL, A.R., 2001. Evidence for positive selection in foot-and-mouth disease virus capsid genes from field isolates. Genetics, 137, 7–15.
- HARRIS, T.R. & BROWN, F., 1977. Biochemical analysis of a virulent and an avirulent strain of foot-and-mouth disease virus. Journal of General Virology, 34, 87–105.
- HEARPS, A., ZHANG, Z. & ALEXANDERSON, S., 2001. Evaluation of the portable Cepheid SmartCycler real-time PCR machine for rapid diagnosis of foot-and-mouth disease. The Veterinary Record, 150, 612–628.
- HEDGER, R.S., 1968. The isolation and characterization of footand- mouth disease virus from clinically normal herds of cattle in Botswana. Journal of Hygiene, 66, 27–36.
- HEDGER, R.S., 1972. Foot-and-mouth disease and the African buffalo (Syncerus caffer). Journal of Comparative Pathology, 82, 19–28.
- HEDGER, R.S., 1976. Foot-and-mouth disease in wildlife with particular reference to the African buffalo (Syncerus caffer). In: PAGE, A.L., (ed.). Wildlife Diseases. New York: Plenum Publishing Corporation.
- HEDGER, R.S., 1976. The maintenance of foot-and-mouth disease in Africa. Ph.D. thesis, London School of Hygiene and Tropical Medicine, University of London.
- HEDGER, R.S. & CONDY, J.B., 1985. Transmission of foot-and-mouth disease from African buffalo virus carriers to bovines. The Veterinary Record, 117, 205.
- HEDGER, R.S., CONDY, J.B. & GOLDING, S.M., 1972. Infection of some species of African wild life with foot-and-mouth disease virus. Journal of Comparative Pathology, 82, 455–461.
- HEDGER, R.S., FORMAN, A.J. & WOODFORD, M.H., 1973. Foot-and-mouth disease in East African buffalo. Bulletin of Epizootic Diseases of Africa, 21, 99–101.
- HEDGER, R.S. & STUBBINS, A.G.J., 1971. The carrier state in foot-and-mouth disease, and the probang test. State Veterinary Journal (U.K.), 26, 45–50.
- HENNING, O., 1892. Annual Report of the Colonial Veterinary Surgeon for 1892, 15.
- HENNING, M.W., 1956. Foot-and-mouth disease, Mond-en-Klouseer. In: Diseases in South Africa. South Africa: Central News Agency.
- HESS, W.R., BACHRACH, H.L. & CALLIS, J.J., 1960. Persistence of foot-and-mouth disease virus in bovine kidneys and blood as related to the occurrence of antibodies. American Journal of Veterinary Research, 21, 1104–1108.
- HOLLAND, J., SPINDLER, K., HORODYSKI, F., GRABOU, E., NICOL, S. & VAN DE POL, S., 1982. Rapid evolution of RNA genomes. Science, 215, 1577–1585.
- HOWARD, S.C. & DONNELLY, C.A., 2000. The importance of immediate destruction in epidemics of foot-and-mouth disease. Research in Veterinary Science, 69, 189–196.
- HUGHES, G.J., 2001. Modelling the maintenance and transmission of foot-and-mouth disease virus in sheep. PhD thesis, University of Edinburgh.
- HUGHES, G.J., MIOULET, V., KITCHING, P.R., WOOLHOUSE, M.E.J., ALEXANDERSEN, S. & DONALDSON, A.I., 2002. Foot-and-mouth disease infection of sheep: implications for diagnosis and control. The Veterinary Record, 150, 724–727.
- HUGH-JONES, M.E., 1972. Epidemiological studies on the 1967–68 foot-and-mouth epidemic: Attack rates and cattle density. Research in Veterinary Science, 13, 411–417.
- HUNTER, P., 1996. The performance of South African Territories serotypes of foot-and-mouth disease antigen in oil-adjuvented vaccines. Scientific and Technical Review, 15, 913–922.
- HUNTER, P., 1998. Vaccination as a means of control of foot-and-mouth disease in sub-Saharan Africa. Vaccine, 16, 261–264.
- HUTCHEON, D., 1892. Annual Report of the Colonial Veterinary Surgeon, 1892, 9.
- HUTCHEON, D., 1893. Annual Report of the Colonial Veterinary Surgeon, 1893, 10.
- HUTCHEON, D., 1894. Annual Report of the Colonial Veterinary Surgeon, 1894, 8.
- HYSLOP, N. ST.G., 1965. Airborne infection with the virus of foot-and-mouth disease. Journal of Comparative Pathology, 75, 119– 126.
- HYSLOP, N. ST.G., 1970. The epizootiology and epidemiology of foot and mouth disease. Advances in Veterinary Science, 14, 261–307.
- ILLOTT, M.C., SALT, J.S., GASKELL, R.M. & KITCHING, R.P., 1997. Dexamethasone inhibits virus production and the secretory IgA response in oesophageal-pharyngeal fluid in cattle persistently infected with foot-and-mouth disease virus. Epidemiology and Infection, 118, 181–187.
- JACKSON, T., BLAKEMORE, W., NEWMAN, J.W., KNOWLES, N.J., MOULD, A.P., HUMPHRIES, M.J. & KING, A.M., 2000. Foot-and-mouth disease virus is a ligand for the high affinity binding conformation of integrin alpha 5 beta 1: influence of the leucine residue within the RGDL motif on selectivity of integrin binding. Journal of General Virology, 81, 1383– 1391.
- JACKSON, T., ELLARD, F.M., ABU GHAZALEH, R., BROOKS, S.M., BLAKEMORE, W.E., CORTEYN, A.H., STUART, D.I., NEWMAN, J.W.I. & KING, A.M.Q., 1996. Efficient infection of cells in culture by type O foot-and-mouth disease virus requires binding to cell surface heparin sulphate. Journal of Virology, 70, 5282–5287.
- JACKSON, T., SHEPPARD, D., DENYER, M., BLAKEMORE, W. & KING, A.M.Q., 2000. The epithelial integrin alpha 5 beta 6 is a receptor for foot-and-mouth disease virus. Journal of Virology, 74, 4949–4956.
- KAR, B.C., HOTA, N. & ACHARJYO, L.N. , 1983. Occurrence of foot-and-mouth disease among some wild ungulates in captivity. Indian Veterinary Journal, 60, 237–239.
- KEET, D.F., HUNTER, P., BENGIS, R.G., BASTOS, A.D. & THOMSON, G.R., 1996. The 1992 foot-and-mouth disease epizootic in the Kruger National Park. Journal of the South African Veterinary Association, 67, 83–87.
- KENNEDY, D.J., JACKSON, R.B. & RAMSAY, G.C., 1984. Difficulties experienced in recognizing foot-and-mouth disease in an outbreak in Zimbabwe. Australian Veterinary Journal, 61, 163–164.
- KHUKOROV, V.M., PRONINA, N.A., KORSUN, L.N., KARPEMKO, I.G. & KRUGLIKOVA, B.A., 1974. Foot-and-mouth disease in saiga antelopes (in Russian). Veterinariya (Moscow), 5, 60–61.
- KING, A.M.Q., MCCAHON, D., SAUNDERS, K., NEWMAN, J.W.I. & SLADE, W.R., 1985. Multiple sites of recombination within the RNA genome of foot-and-mouth disease virus. Virus Research, 3, 373–384.
- KITCHING, R.P., 1992. Foot-and-mouth disease. In: ANDREWS, A.H., BLOWEY, R.W., BOYD, H. & EDDY, R.G., (eds.). Diseases and Husbandry of Cattle. Oxford: Blackwell Scientific. pp. 537–543.
- KITCHING, R.P., 1998. A recent history of foot-and-mouth disease. Journal of Comparative Pathology 118, 89–108.
- KITCHING, R.P., 1999. Foot-and-mouth disease: Current world situation. Vaccine, 17, 1772–1774.
- KITCHING, R.P., 2002. Clinical variation in foot-and-mouth disease: cattle. Scientific and Technical Review, 21, 499–504.
- KITCHING, R.P., 2002. Identification of foot-and-mouth disease virus carrier and subclinically infected animals and their differentiation from vaccinated animals. Scientific and Technical Review, 21, 531–538.
- KITCHING, R.P., 2002. National Center for Foreign Animal Diseases, 1015 Arlington St., Winnipeg, Manitoba, R3E 3M4. Canada. Unpublished data.
- KITCHING, R.P. & ALEXANDERSON, S., 2002. Clinical variation in foot-and-mouth disease: pigs. Scientific and Technical review, 21, 513–518.
- kitching, r.p. & donaldson, a.i., 1987. Collection and transportation of specimens for vesicular virus investigation. Revue Scientifique et Technique, 6, 263–272.
- KITCHING, R.P. & HUGHES, G.J., 2002. Clinical variation in foot-and-mouth disease: sheep and goats. Scientific and Technical Review, 21, 505–512.
- KITSON, J.D.A., MCCAHON, D. & BELSHAM, G.J., 1990. Sequence analysis of monoclonal antibody resistant mutants of type O foot-and-mouth disease virus: Evidence for the involvement of the three surface exposed capsid proteins in four antigenic sites. Virology, 179, 26–34.
- KNOWLES, N.J., ANSELL, D.M. & SAMUEL, A.R., 1998. Molecular comparison of recent foot-and-mouth disease type A viruses from west Africa with historical and reference virus strains. Session of the Research Group of the Standing Technical Committee of the European Commission for the Control of Foot-and-Mouth Disease. 14–18 September 1998. Pirbright, UK.
- KNOWLES, N.J., DAVIES, P.R., HENRY, T., O’DONNELL, V., PACHECO, J.M. & MASON, P.W., 2001. Emergence in Asia of foot-and-mouth disease viruses with an altered host range: Characterization of alternations in the 3A protein. Journal of Virology, 75, 1551–1556.
- KREBS, O. & MARQUARDT, O., 1992. Identification and characterization of foot-and-mouth disease virus O1 Burgwedel/1987 as an intertypic recombinant. Journal of General Virology, 73, 613–619.
- KRUGER, S.J.P., 1858. Quoted from ‘Staats Courant’ No. 43, November 19th, 1858. Bloemhof Blue Book. Evidence taken in connection with the diamond fields arbitration, 1871, 313.
- KUHN, R., LUZ, N. & BECK, E., 1990. Functional analysis of the internal initiation site of foot-and-mouth disease virus. Journal of Virology, 64, 4625–4631.
- KVITKIN, V.P., 1959. Physiopathology of experimental foot-and-mouth disease in reindeer. Veterninariya (Moscow), 36, 25–28.
- LAPORTE, J., GROSCLAUDE, J., WONTYGEM, J., BERNARD, S. & RONZE, P., 1973. Neutralisation en culture cellulaire du ponvoir infectieux du virus de la fièvre aphteuse des sérums provenant de porcs immunisés à l’aide d’une protéin viral purifiée. Compte Rendu Hebdomadaire des Séances de l’Academie des Sciences, 276D, 3399–3402.
- LAWRENCE, D.A., 1935. Foot-and-mouth disease in Southern Rhodesia. Journal of the South African Veterinary Medical Association, 6, 253–262.
- LAWRENCE, C.J., 2002. Animal welfare consequences in England and Wales of the 2001 epidemic of foot-and-mouth disease. Scientific and Technical Review, 21, 863–868.
- LAWRENCE, J.A., FOGGIN, C.M. & NORVAL, R.A.I., 1980. The effects of war on the control of diseases of livestock in Rhodesia (Zimbabwe). The Veterinary Record, 107, 82–85.
- LEA, S., HERNANDEZ, J., BLAKEMORE, W., BROCCHI, E., CURRY, S., DOMINGO, E., FRY, E., ABU-GHAZALEH, R., KING, A., NEWMAN, J., et al., 1994. The structure and antigenicity of a type C foot-and-mouth disease virus. Structure, 15, 123–139.
- LEFORBAN, Y., 2002. How predictable were the outbreaks of foot and mouth disease in Europe in 2001 and is vaccination the answer? Scientific and Technical Review, 21, 549–556.
- LEIPPERT, M., BECK, E., WEILAND, F. & PFAFF, E., 1997. Point mutations within the G- H loop of foot-and-mouth disease virus O1K affect virus attachment to target cells. Journal of Virology, 71, 1046–1051.
- LE VAILLANT, M., 1795. Travels into the Interior Parts of Africa, Vol. II. 1780–1785.
- LI, F., BROWNING, G.F., STUDDERT, M.J. & CRABB, B.S., 1996. Equine rhinovirus 1 is more closely related to foot-and-mouth disease virus than to other picornaviruses. Proceedings of the National Academy of Sciences of the USA, 93, 990–995.
- LOGAN, D., ABU-GHALZALEH, R., BLAKEMORE, W., CURRY, S., JACKSON, T., KING, A., LEA, S., LEWIS, R., NEWMAN, J., PARRY, N., ROWLANDS, D., STUART, D. & FRY, E., 1993. Structure of a major immunogenic site on foot-and-mouth disease virus. Nature, 362, 566–568.
- MACKAY, D.K., FORSYTH, M.A., DAVIES, P.R., BERLINZANI, A., BELSHAM, G.J., FLINT, M. & RYAN, M.D., 1998. Differentiating infection from vaccination in foot-and-mouth disease using a panel of recombinant, non-structural proteins in ELISA. Vaccine, 16, 446.
- MACKAY, D.K.W., NEWMAN, B. & SACHPATZIDIS, A., 1995. Epidemiological analysis of the serological survey for antibody to FMD virus, Greece 1994. Report of the Institute for Animal Health, Pirbright, UK and the Animal Health Office, Xanthi, Greece.
- MAGNUSSON, H., 1939. A case of foot-and-mouth disease in an elk [in German]. Deutsche Tierärtzliche Wochenschrift, 47, 509–511.
- MAGOMA, G., SAMUEL, S., KONDELA, A., MADEGE, M. & MAKUNGU, S., 2000. Case study: A preliminary clinical investigation of a condition known as luzwiga in cattle in the lake zone of Tanzania. Paper presented at the Annual Scientific Conference of the Tanzania Veterinary Association, Arusha, December 2000.
- MARQUARDT, O. & HAAS, B., 1998. VP1-coding sequences of recent isolates of foot-and-mouth disease virus types A, O and Asia1. Virus Genes, 16, 185–193.
- MARQUARDT, O., RAHMAN, M.M. & FREIBERG, B., 2000. Genetic and antigenic variance of foot-and-mouth disease virus type Asia 1. Archives of Virology, 145, 149–157.
- MARTIN, W.B. & CHAPMAN, W.G., 1961. The tissue culture colour test for assaying the virus and neutralizing antibody of foot-and-mouth disease and its application to the measurement of immunity in cattle. Research in Veterinary Science, 2, 53–61.
- MARTINEZ, M.A., DOPAZO, J., HERNANDEZ, J., MATEU, M.G., SOBRINO, F., DOMINGO, E. & KNOWLES, N.J., 1992. Evolution of the capsid protein genes of foot-and-mouth disease virus: antigenic variation without accumulation of amino acid substitutions over six decades. Journal of Virology, 66, 3557–3565.
- MARTINEZ, M.A., VERDAGUER, N., MATEU, M.G. & DOMINGO, E., 1997. Evolution subverting essentiality: Dispensability of the cell attachment Arg-Gly-Asp motif in multiple passaged foot-and-mouth disease virus. Proceedings of the National Academy of Sciences, USA, 94, 6798–6802.
- MATEU, M.G., DA SILVA, J.L., ROCHA, E., DE BRUM, D.L., ALONSO, A., ENJUANES, L., DOMINGO, E. & BARAHONA, H., 1988. Extensive antigenic heterogeneity of foot-and-mouth disease virus of serotype C. Virology, 167, 113–124.
- MATEU, M.G., VALERO, M.L., ANDREU, D. & DOMINGO, E., 1996. Systematic replacement of amino acid residues within an Arg-Gly-Asp containing loop of foot-and-mouth disease virus and effect on cell recognition. Journal of Biological Chemistry, 271, 12814–12819.
- MCCAHON, D., KING, A.M., ROE, D.S., SLADE, W.R., NEWMAN, J.W. & CLEARY, A.M., 1985. Isolation and biochemical characterization of intertypic recombinants of foot-and-mouth disease virus. Virus Research, 3, 87–100.
- MCCULLOUGH, K.C., CROWTHER, J.W., CARPENTER, W.C., BROCCHI, E., CAPUCCI, L., DEÁSIMONE, F., XIE, Q. & MCCAHON, D., 1987. Epitopes on foot-and-mouth disease particles. I. Topology. Virology, 157, 516–525.
- MCKERCHER, P.D., MORGAN, D.O., MCVICAR, J.W. & SHNOT, N.J., 1980. Thermal processing to inactivate viruses in meat products. Proceedings of the 84th Annual Meeting of the United States Health Association, San Diego, California, 320–328.
- MCVICAR, J.W., 1977. The pathobiology of foot-and-mouth disease in cattle: A review. Bulletin Centro Panamericano Fiebre Aftosa, 26, 9–14.
- MCVICAR, J.W. & SUTMU¨LLER, P., 1976. Growth of foot-and-mouth disease virus in the upper respiratory tract of non-immunized, vaccinated, and recovered cattle after intranasal inoculation. Journal of Hygiene, 76, 467–481.
- MEDINA, M., DOMINGO, E., BRANGWYN, J.K. & BELSHAM, G.J., 1993. The two species of foot-and-mouth disease virus leader protein, expressed individually, exhibit the same activities. Virology, 194, 355–359.
- MEZENCIO, J.M., BABCOCK, G.D., KRAMER, E. & BROWN, F., 1999. Evidence for the persistence of foot-and-mouth disease virus in pigs. Veterinary Journal, 157, 379–389.
- MINOR, P.D., 1990. Antigenic structure of picornaviruses. Current Topics in Microbiology and Immunology, 161, 121–154.
- MONTGOMERY, J.F., OLIVER, R.E. & POOLE, W.S.H., 1987. A vesiculo-bullous disease in pigs resembling foot-and-mouth disease. I. Field codes. New Zealand Veterinary Journal, 35, 21–26.
- MONTGOMERY, J.F., OLIVER, R.E., POOLE, W.S.H. & JULIAN, A.F., 1987. A vesiculo-bullous disease in pigs resembling foot-and-mouth disease. II. Experimental reproduction of the lesions. New Zealand Veterinary Journal, 35, 27–30.
- MORRELL, D.J., MELLOR, E.J.C., ROWLANDS, D.J. & BROWN, F., 1987. Surface structure and RNA-protein interactions of foot-and-mouth disease virus. Journal of General Virology, 68, 1649–1658.
- NEFF, S., SA-CARVALHO, D., RIEDER, E. MASON, P.W., BLYSTONE, S.D., BROWN, E.J. & BAXT, B., 1998. Foot-and-mouth disease virus virulent for cattle utilizes the integrin v3 as its receptor. Journal of Virology, 72, 3587–3594.
- NEWMAN, J.F.E. & BROWN, F., 1997. Foot-and-mouth disease virus and poliovirus particles contain proteins of the replication complex. Journal of Virology, 71, 7657–7662.
- NICHOLLS, M.J., BLACK, L. & RWEYEMAMU, M.M., 1984. The effect of maternally derived antibodies on the response of calves to vaccination against foot-and-mouth disease. Journal of Hygiene, 92, 105–116.
- NICHOLLS, M.J., BLACK, L., RWEYEMAMU, M.M. & GRADWELL, D.V., 1985. Effect of age on response of cattle to vaccination against foot-and-mouth disease. British Veterinary Journal, 141, 17–26.
- NUNEZ, J.I., BARANOWSKI, E., MOLINA, N., RUIZ-JARABO, C.M., SANCHEZ, C., DOMINGO, E. & SOBRINO, F., 2001. A single amino acid substitution in nonstructural protein 3A can mediate adaptation of foot-and-mouth disease virus to the guinea pig. Journal of Virology, 75, 3977–3983.
- OFFICE INTERNATIONAL DES EPIZOOTIES, 2000. Manual of Standards for Diagnostic Tests and Vaccines. OIE, Paris.
- OFFICE INTERNATIONAL DES EPIZOOTIES, 2001. International Animal Health Code. OIE, Paris.
- OGRYZKOV, S.E., 1963. The pathology of foot-and-mouth disease in reindeer [in Russian]. Trudy 2 Vsesojuznoi Konfernzii Patologii i Anatomii Zhivotnych (Izdanie Moskovskoi Veterinarnoi Akademii, Tome 46). pp. 420–425. (English abstract, Veterinary Bulletin, 36, 2571, 1966).
- OLEKSIEWICZ, M.B., DONALDSON, A.I. & ALEXANDERSON, S., 2001. Development of a novel real-time RT-PCR assay for quantitation of foot-and-mouth disease virus in diverse porcine tissues. Journal of Virological Methods, 92, 23–35.
- OLIVER, R.E., DONALDSON, A.I., GIBSON, C.F., ROEDER, P.L., LE BLANC SMITH, P.M. & HAMBLIN, C., 1988. Detection of foot-and-mouth disease antigen in bovine epithelial samples: Comparison of sites of sample collection by an enzyme linked immunosorbent assay (ELISA) and complement fixation test. Research in Veterinary Science, 44, 315–319.
- OWEN, M. & OWEN, D., 1980. The fences of death. African Wildllife, 31, 25–27.
- PALMENBERG, A.C., 1989. Sequence alignments of picornaviral capsid proteins. In: SEMLER, B.L. & EHRNFELD, E., (eds). Molecular Aspects of Picornavirus Infection and Detection. Washington D.C.: American Society for Microbiology. pp. 211–241.
- PARKER, J., 1971. Presence and inactivation of foot-and-mouth disease virus in animal faeces. The Veterinary Record, 88, 659–662.
- PATTNAIK, P., VENKATARAMANAN, R., TOSH, C., SNYAL, A., HEMADRI, D., SAMUEL, A.R., KNOWLES, N.J. & KITCHING, R.P., 1998. Genetic heterogeneiety of Indian field isolates of foot-and-mouth disease virus serotype O as revealed by partial sequencing of 1D gene. Virus Research, 55, 115–127.
- PAY, T.W.F., 1983. Variation in foot-and-mouth disease: Application to vaccination. Revue Scientifique et Technique, 2, 701–723.
- PAY, T.W.F., 1985. Factors influencing the performance of footand- mouth disease vaccines under field conditions. In: DELLA-PORTA, A.J., (ed.). Veterinary Viral Diseases: Their Significance in South-East Asia and the Western Pacific. Sydney, New York, London: Academic Press Inc.
- PAY, T.W.F., 1985. The comparison of the antigenic relationship of a vaccine strain with a new field isolate of foot-and-mouth disease virus. Foot-and-mouth Disease Bulletin, 23, 1–6.
- PAY, T.W.F., 1988. Foot-and-mouth disease in sheep and goats: A review. Foot-and-mouth Disease Bulletin, 26, 2–13.
- PEREIRA, H.G., 1977. Subtyping of foot-and-mouth disease virus. Developments in Biological Standardization, 35, 167–174.
- PEREIRA, H.G., 1981. Foot-and-mouth disease. In: GIBBS, E.P.J., (ed.). Virus Diseases of Food Animals. Vol. II. Disease Monographs. London, New York: Academic Press Inc.
- PEREKA, A.E., MAKENE, V.W., CHIBUNDA, R.T., MASELLE, R.M., PHIRI, E.C.J.H. & KAMBARAGE, D.M., 2001. Heat intolerance associated with foot-and-mouth disease in cattle: haematological parameters and plasma metabolites. Tanzania Veterinary Journal, 21, 63–68.
- PLATT, H., 1960. The localization of lesions in experimental foot-and-mouth disease. British Journal of Experimental Pathology, 41, 150–159.
- PUTNAK, J.R. & PHILLIPS, B.A., 1981. Picornaviral structure and assembly. Microbiological Reviews, 45, 287–315.
- REID, S.M., FERRIS, N.P., HUTCHINGS, G.H., ZHANG, Z., BELSHAN, G.J. & ALEXANDERSON, S., 2001. Diagnosis of foot-and-mouth disease by real-time fluorogenic PCR assay. The Veterinary Record, 149, 621–623.
- RICO-HESSE, R., PALLANSCH, M.A., NOTTAY, B.K. & KEW, O.M., 1987. Geographic distribution of wild poliovirus type 1 genotypes. Virology, 160, 211–322.
- RIEDER, E., BAXT, B., LUBROTH, J. & MASON, P.W., 1994. Vaccines prepared from chimeras of foot-and-mouth disease virus (FMDV) induce neutralizing antibodies and protective immunity to multiple serotypes of FMDV. Journal of Virology, 68, 7092–7098.
- ROBERTSON, W., 1903. Annual Report of the Colonial Veterinary Surgeon, 42.
- ROBSON, K.J.H., CROWTHER, J.R., KING, A.M.Q. & BROWN, F., 1979. Comparative biochemical and serological analysis of five isolates of a single serotype of foot-and-mouth disease virus. Journal of General Virology, 45, 579–590.
- ROEDER, P.L. & LE BLANC SMITH, P.M., 1987. Detection and typing of foot-and-mouth disease virus by enzyme-linked immunosorbent assay: A sensitive, rapid and reliable technique for primary diagnosis. Research in Veterinary Science, 43, 225–232.
- ROHRER, H., 1983. Differential significance of epizootiological factors in the control of foot-and-mouth disease. A brief review. Acta Virologica, 27, 371–375.
- ROSSI, M.S., SADIR, A.M., SCHUDEL, A.A. & PALMA, E.L., 1988. Detection of foot-and-mouth disease virus with DNA probes in bovine esophageal-pharyngeal fluids. Archives of Virology, 99, 67–74.
- ROSSMAN, M.G., ARNOLD, E., ERICSON, J.W., FRANKENBERGER, E.A., GRIFFITH, J.P., HECHT, H-J., JOHNSON, J.E., KAMER, G., LUO, M., MOSSER, A.G., RUECKERT, R.R., SHERRY, B. & VRIEND, G., 1985. Structure of human common cold virus and functional relationship to other picornaviruses. Nature, 317, 145–153.
- ROSSMAN, M.G. & PALMENBERG, A.A., 1988. Conservation of the putative receptor attachment site in picornaviruses. Virology, 164, 373–382.
- ROSSMAN, M.G. & RUECKERT, R.R., 1987. What does the molecular structure of viruses tell us about viral functions? Microbiological Sciences, 4, 206–214.
- ROWLANDS, D.J., 1985. Novel approaches to foot-and-mouth disease vaccination. Vaccine, 3, 37–38.
- ROYAL SOCIETY, 2002. Infectious diseases in livestock: Scientific questions relating to the transmission, prevention and control of epidemic outbreaks of infectious diseases in livestock in Great Britain. London: The Royal Society.
- RUECKERT, R.R., 1985. Picornaviruses and their replication. In: FIELDS, B.N., KNIPE, D.M., MELNICK, J.L., CHANOCK, R.M., ROIZMAN, R. & SHOPE, R.E., (eds). Virology. New York: Raven Press.
- RUECKERT, R.R. & WIMMER, E., 1984. Systematic nomenclature of picornavirus proteins. Journal of Virology, 50, 957–959.
- RUMNEY, R.P., 1986. Meteorological influences on the spread of foot-and-mouth disease. Journal of Applied Bacteriology, Symposium Supplement 1986, 105S–114S.
- RUOSLAHTI, E. & PIERSCHBACHER, M.D., 1987. New perspectives on cell adhesion: RGD and integrins. Science, 238, 4911–497.
- RWEYEMAMU, M.M., BOOTH, J.C., HEAD, M. & PAY, T.W.F., 1978. Microneutralization tests for serological typing and subtyping of foot-and-mouth disease virus strains. Journal of Hygiene, 81, 107–123.
- RWEYEMAMU, M.M. & HINGLEY, P.J., 1984. Foot-and-mouth disease virus strain differentiation: Analysis of the serological data. Journal of Biological Standards, 12, 323–337.
- RWEYEMAMU, M.M., PAY, T.W.F. & PARKER, M.J., 1977. Serological differentiation of foot-and-mouth disease virus strains in relation to selection of suitable vaccine strains. Developments in Biological Standards, 35, 205–214.
- RWEYEMAMU, M.M., PAY, T.W.F. & SIMMS, M.J., 1982. The control of foot-and-mouth disease by vaccination. Veterinary Annual, 22, 63–80.
- RYAN, M.D., KING, A.M.Q. & THOMAS, G.P., 1991. Cleavage of foot-and-mouth disease virus polyprotein is mediated by residues located within a 19 amino acid sequence. Journal of General Virology, 72, 2727–2732.
- SADIR, A.M., SCHUDEL, A.A. & LAPORTE, O., 1988. Response of foot-and-mouth disease vaccines in newborn calves. Influence of age, colostral antibodies and adjuvants. Epidemiology and Infection, 100, 135–144.
- SA-CARVALHO, D., RIEDER, E., BAXT, B., RODARTE, R., TANURI, A. & MASON, P.W., 1997. Tissue culture adaptation of foot-and-mouth disease selects viruses that bind to heparin and are attenuated in cattle. Journal of Virology, 71, 5115–5123
- SAIZ, M., GOMEZ, S., MARTINEZ-SALAS, E. & SOBRINO, F., 2001. Deletion or substitution of the aphthovirus 3’ NCR abrogates infectivity and virus replication. Journal of General Virology, 82, 93–101.
- AIZ, J.C., GONZALEZ, M.J., BORCA, M.V., SOBRINO, F. & MOORE, D.M., 1991. Identification of neutralizing antigenic sites on VP1 and VP2 of type A5 foot-and-mouth disease virus, defined by neutralization-resistant variants. Journal of Virology, 65, 2518–2524.
- SAIZ, J.C., SOBRINO, F. & DOPAZO, J., 1993. Molecular epidemiology of foot-and-mouth disease virus type O. Journal of General Virology, 74, 2281–2285.
- SAKAMOTO, K. & YOSHIDA, K., 2002. Recent outbreaks of foot-and-mouth disease in countries of east Asia. Scientific and Technical Review, 21, 459–463.
- SALT, J.S., 1993. The carrier state of foot-and-mouth disease—an immunological review. British Veterinary Journal, 149, 207–223.
- SALT, J.S., SAMUEL, A.R. & KITCHING, R., 1996. Antigenic analysis of type O foot-and-mouth disease virus in persistently infected bovines. Archives of Virology, 141, 1407–1421.
- SAMUEL, A.R. & KNOWLES, N.J., 2001. Foot-and-mouth disease virus: Cause of the recent crisis for the UK livestock industry. Trends in Genetics, 17, 421–424.
- SAMUEL, A.R. & KNOWLES, N.J., 2001. Foot-and-mouth disease type O viruses exhibit genetically and geographically distinct evolutionary lineages (topotypes). Journal of General Virology, 82, 609–621.
- SANGARE, O., BASTOS, A.D., MARQUARDT, O., VENTER, E.H., VOSLOO, W. & THOMSON, G.R., 2001. Molecular epidemiology of serotype O foot-and-mouth disease virus with emphasis on West and South Africa. Virus Genes, 22, 345–351.
- SCOTT, F.W., COTTRAL, G.E. & GAILIUNAS, P., 1966. Resistance of foot-and-mouth disease virus in external lesions and saliva of experimentally infected cattle. American Journal of Veterinary Research, 27, 1531–1536.
- SCOTT WILSON RESOURCE CONSULTANTS, 2000. Environmental assessment of veterinary fences in Ngamiland. 23 Chester Street, Edinburgh EH3 7ET, UK.
- SCUDAMORE, J.M. & HARRIS, D.M., 2002. Control of foot-and-mouth disease: Lessons from the experience of the outbreak in Great Britain in 2001. Scientific and Technical Review, 21, 699–710.
- SEKIGUCHI, K., FRANKE, A.J. & BAXT, B., 1982. Competition for cellular receptor sites among selected aphthoviruses. Archives of Virology, 74, 53–64.
- SELLERS, R.F., 1968. The inactivation of foot-and-mouth disease virus by chemicals and inactivants. The Veterinary Record, 83, 504–506.
- SELLERS, R.F., 1971. Quantitative aspects of the spread of foot-and-mouth disease. The Veterinary Bulletin, 41, 431–439.
- SELLERS, R.F., 1983. Transmission of viruses by artificial breeding techniques: A review. Journal of the Royal Society of Medicine, 76, 772–775.
- SELLERS, R.F. & PARKER, J., 1969. Airborne excretion of foot-and-mouth disease virus. Journal of Hygiene, 67, 671–677.
- SHIMSHONY, A., ORGAD, U., BAHARAV, D., PRUDOVSKY, S., YAKOBSON, B., BAR MOSHE, B. & DAGAN, D., 1986. Malignant foot-and-mouth disease in mountain gazelles. The Veterinary Record, 119, 175–176.
- SIMMS, M.J., 1969. Inactivation of foot-and-mouth disease virus. Foot-and-mouth Disease Bulletin: Supplement No. 2. The Wellcome Research Laboratories, Beckenham, Kent. 12.
- SINGH, E., 1984. Proceedings of the 10th International Congress on Animal Reproduction and Artificial Insemination, 10–14 June 1984, Urbana IL (USA).
- SMITH, D.B. & INGLIS, S.C., 1987. The mutation rate and variability of eukaryotic viruses: An analytical review. Journal of General Virology, 68, 2729–2740.
- SNOWDON, W.A., 1966. Growth of foot-and-mouth disease virus in monolayer cultures of calf thyroid cells. Nature, 210, 1079–1080.
- SORENSON, J.H., MACKAY, D.K., JENSON, C.C. & DONALDSON, A.I., 2000. An integrated model to predict the atmospheric spread of foot-and-mouth disease virus. Epidemiology and Infection, 124, 577–590.
- SORENSON, K.J., MADSEN, K.G., MADSEN, E.S., SALT, J.S., NQINDI, J. & MACKAY, D.K., 1998. Differentiation of infection from vaccination in foot-and-mouth disease by the detection of antibodies to the non-structural proteins 3D, 3AB and 3ABC in ELISA using antigens expressed in baculovirus. Archives of Virology, 143, 1461–1476.
- STROHMAIER, K., 1987. Die Maul- und Klauenseuche. III. Erfahrungen und Folgerungen aus 20 Jahren Bekämpfung der Maul- und Klauenseuche. Tierärztliche Umschau, 5, 417–422.
- SUTMÖLLER, P., 2001. 1502 Largo Road, # 101, Richmond, Virginia 23233, USA. Personal communication.
- SUTMÖLLER, P. & CASAS OLASCOAGA, R., 2002. Unapparent foot-and-mouth disease infection (sub-clinical infections and carriers): Implications for control. Scientific and Technical Review, 21, 519–529.
- SUTMÖLLER, P. & GAGERO, A., 1965. Foot-and-mouth disease carriers. The Veterinary Record, 77, 968–969.
- SUTMÖLLER, P. & MCVICAR, J.W., 1972. The epizootiological importance of foot-and-mouth disease carriers. III. Exposure of pigs to bovine carriers. Archiv fu¨r die Gesamte Virusforschung, 37, 78–84.
- SUTMÖLLER, P. & MCVICAR, J.W., 1973. Foot-and-mouth disease: Growth of virus after conjunctival inoculation of cattle. Archiv fu¨r die Gesamte Virusforschung, 43, 284–287.
- SUTMÖLLER, P., THOMSON, G.R., HARGREAVES, S.K., FOGGIN, C.M. & ANDERSON, E.C., 2000. The foot-and-mouth disease risk posed by African buffalo within wildlife conservancies to the cattle industry of Zimbabwe. Preventive Veterinary Medicine, 44, 43–60.
- SUTMÖLLER, P. & VIERIRA, A., 1980. The relationship of neutralizing antibody titres for foot-and-mouth disease virus and the protection of cattle. Bulletin Centro Panamericano Fiebre Aftosa, 39–40, 57–62.
- SUTMOLLER, P. & WRATHALL, A.E., 1997. A quantitative assessment of the risk of transmission of foot-and-mouth disease, bluetongue and vesicular stomatitis by embryo transfer in cattle. Preventive Veterinary Medicine, 32, 111–132.
- TAYLOR, K.C., 2002. Environmental impacts of the foot-and-mouth disease outbreak in Great Britain in 2001: The use of risk analysis to manage the risks in the countryside. Scientific and Technical Review, 21, 797–813.
- TAYLOR, R.D. & MARTIN, R.B., 1987. Effects of veterinary fences on wildlife conservation in Zimbabwe. Environmental Management, 11, 327–334.
- THE ECONOMIST, 27 JULY – 2 AUGUST 2001. London.
- THE SUNDAY TELEGRAPH, 13 january 2002. United Kingdom.
- THE TIMES, 24 MAY 2001. United Kingdom.
- THOMAS, A.A.M., WOORTMEIJER, R.J., BARTELING, S.J. & HELOEN, R.H., 1988. Evidence for more than one important neutralizing site on foot-and-mouth disease virus. Archives of Virology, 99, 237–242.
- THOMPSON, D., MUIEL, P., RUSSEL, D., OSBORNE, P., BROMLEY, A., ROWLANDS, M., CREIGH-TYTE, S. & BROWN, C., 2002. Economic costs of the foot-and-mouth disease in the United Kingdom in 2001. Scientific and Technical Review, 21, 675–687.
- THOMSON, G.R., 1994. Foot-and-mouth disease. In: COETZER, J.A.W., THOMSON, G.R. & TUSTIN, R.C., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town, London, New York: Oxford University Press. pp. 825–952.
- THOMSON, G.R., 1995. Overview of foot-and-mouth disease in southern Africa. Scientific and Technical Review, 14, 503–520.
- THOMSON, G.R., 1996. The role of carrier animals in the transmission of foot-and-mouth disease. O.I.E. Comprehensive Reports on Technical Items Presented to the International Committee or to Regional Commissions. pp. 87–103.
- THOMSON, G.R., 1999. Alternatives for controlling animal diseases resulting from interaction between livestock and wildlife in southern Africa. South African Journal of Science, 95, 71–76.
- THOMSON, G.R., BENGIS, R.G. & BROWN, C.C., 2001. Picornavirus infections. In: WILLIAMS, E.S. & BARKER, I.K. (eds). Infectious Diseases of Wild Mammals. 3rd edn. Ames: Iowa State University Press. pp. 119–130.
- THOMSON, G.R., BENGIS, R.G., ESTERHUYSEN, J.J. & PINI, A., 1984. Maintenance mechanisms for foot-and-mouth disease virus in the Kruger National Park and potential avenues for its escape into domestic animal populations. Proceedings of the XIIIth World Congress on Diseases of Cattle. Vol. I, September 1984. Durban, South Africa.
- THOMSON, G.R., DOUBE, B.M., BRAAK, L.F.O., GAINARU, M.D. & BENGIS, R.G., 1988. Failure of Haematobia thirouxi potans (Bezzi) to transmit foot-and-mouth disease virus mechanically between viraemic and susceptible cattle. Onderstepoort Journal of Veterinary Research, 55, 121–122.
- THOMSON, G.R. & NEVILL, E.M., 1988. Onderstepoort Veterinary Institute, South Africa. Unpublished data.
- THOMSON, G.R., VOSLOO, W. & BASTOS, A.D.S., in press. Foot-and-mouth disease in wildlife. In: ROWLANDS, D. (ed.). Foot-and-Mouth Disease. Amsterdam: Elsevier.
- THOMSON, G.R., VOSLOO, W., ESTERHUYSEN, J.J. & BENGIS, R.G., 1992. Maintenance of foot-and-mouth disease viruses in buffalo (Syncerus caffer Sparrman, 1779) in southern Africa. Scientific and Technical Review, 11, 1097–1107.
- TORRES, A., DAVID, M.J. & BOWMAN, Q.P., 2002. Risk management of international trade: Emergency preparedness. Scientific and Technical Review, 21, 493–498.
- URBAIN, A., BULLIER, P. & NOUVEL, J., 1938. A small outbreak of aphthous fever in wild animals in captivity [in French]. Bulletin Academie Vétérinaire de France 11, 59–73.
- VAKHARIA, V.N., DAVANEY, M.A., MOORE, D.M., DUNNAND, J.J. & GRUBMAN, M.J., 1987. Proteolytic processing of foot-and-mouth disease virus polyproteins expressed in a cell-free system from clone-derived transcripts. Journal of Virology, 61, 3199–3207.
- VAN BEKKUM, J.G., FISH, R.C. & NATHAUS, I., 1969. Immunologic responses in Dutch cattle vaccinated with foot-and-mouth disease vaccines under field conditions: Neutralizing antibody responses and immunity to O, A and C types. American Journal of Veterinary Research, 30, 2125–2129.
- VAN BEKKUM, J.G., FRENKEL, H.S., FREDERIKS, H.H.J. & FRENKEL, S., 1959. Observations on the carrier state of cattle exposed to foot-and-mouth disease virus. Tijdschrift voor Dieregeneeskunde, 84, 1159–1164.
- VAN KAMMEN, A., 1999. Beijerinck’s contribution to the virus concept— an introduction. In: CALISHER, C.H. & HORZINEK, M.C., (eds). 100 Years of Virology: The Birth and Growth of a Discipline. Wien, New York: Springer-Verlag. pp. 1–8.
- VAN REGENMORTEL, M.H.V., FAUQUET, C.M., BISHOP, D.H.L., CARSTENS, E.B., ESTES, M.K., LEMON, S.M., MANILOFF, J., MAYO, M.A., MCGEOCH, D.J., PRINGLE, C.R. & WICKNER, R.B, 2000. Virus Taxonomy: Classification and nomenclature of viruses. Sixth Report of the International Committee on Taxonomy of Viruses. Wien: Springer-Verlag.
- VAN RENSBURG, H., HAYDON, D., JOUBERT, F., BASTOS, A., HEATH, L. & NEL, L., 2002. Genetic heterogeneity in the foot-and-mouth disease virus Leader and 3C proteinases. Gene, 289, 19–29.
- VAN VUUREN, C. DE W., 1997. ARC-Institute for Exotic Diseases, Onderstepoort 0110, South Africa. Personal communication.
- VERDAGUER, N, MATEU, M.G., ANDREU, D., GIRALT, E., DOMINGO, E. & FITA, I., 1995. Structure of the major antigenic loop of foot-and-mouth disease virus complexed with a neutralizing antibody: Direct involvement of the Arg-Gly-Asp motif in the interaction. EMBO Journal, 14, 739–748.
- VOSLOO, W., BASTOS, A.D, KIRKBRIDE, E., ESTERHUYSEN, J.J., JANSE VAN RENSBURG, D., BENGIS, R.G., KEET, D.F. & THOMSON, G.R., 1996. Persistent infection of African buffalo (Syncerus caffer) with SAT-type foot-and-mouth disease viruses: Rate of fixation of mutations, antigenic change and interspecies transmission. Journal of General Virology, 77, 1457–1467.
- VOSLOO, W., BASTOS, A.D., MICHEL, A. & THOMSON, G.R., 2001. Tracing movement of African buffalo in southern Africa. Scientific and Technical Review, 20, 630–639.
- VOSLOO, W., KIRKBRIDE, E., BENGIS, R.G., KEET, D.F. & THOMSON, G.R., 1995. Genome variation in the SAT types of foot-and-mouth disease viruses prevalent in buffalo (Syncerus caffer) in the Kruger National Park and other regions of southern Africa, 1986–1993. Epidemiology and Infection, 114, 203–218.
- VOSLOO, W., KNOWLES, N.J. & THOMSON, G.R., 1992. Genetic relationships between southern African SAT-2 isolates of foot-and-mouth disease virus. Epidemiology and Infection, 109, 547–558.
- VOSLOO, W, BASTOS, A.D.S., SANGARE, O., HARGREAVES, S.K. & THOMSON, G.R., 2002. Review of the status and control of foot-and-mouth disease in sub-Saharan Africa. Scientific and Technical Review, 21, 437–449.
- VOSLOO, W., BOSHOFF, K., DWARKA, R. & BASTOS, A.D.S., 2002. The possible role that buffalo played in the recent outbreaks of foot-and-mouth disease in South Africa. Annals of the New York Academy of Sciences, 969, 187–190.
- WALDMAN, O., TRAUTWEIN, K. & PYL, F., 1931. Die Persistenz des Maul und Klauenseuche virus im korper durchgeseuchter Fiere und Seine Auscheidung. Zentralblatt fu¨r Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene, Abteiling I Originale, 121, 1932.
- WALKER, J., 1934. Some observations on foot-and-mouth disease in the Bechuanaland Protectorate. Journal of the South African Veterinary Medical Association, 5, 73–91.
- WALKER, J.S., DE LEEUW, P.W., CALLIS, J.J. & VAN BEKKUM, J.G., 1984. The thermal death time curve of foot-and-mouth disease virus contained in primarily infected milk. Journal of Biological Standardization, 12, 185–189.
- WESTBURY, H.A., DOUGHTY, W.J., FORMAN, A.J., TANGCHAITRONG, S. & KONGTHON, A., 1988. A comparison of enzyme-linked immunosorbent assay, complement fixation and virus isolation for foot-and-mouth disease diagnosis. Veterinary Microbiology, 17, 21–28.
- WOODBURY, E.L., 1995. A review of the possible mechanisms for the persistence of foot-and-mouth disease virus. Epidemiology and Infection, 114, 1–13.
- XIE, Q-C., MCCAHON, D., CROWTHER, J.R., BELSHAM, G.J. & MCCULLOUGH, K.C., 1987. Neutralization of foot-and-mouth disease virus can be mediated through any of at least three separate antigenic sites. Journal of General Virology, 68, 1637–1647.
- YILMA, T., 1980. Morphogenesis of vesiculation in foot-and-mouth disease. American Journal of Veterinary Research, 41, 1537–1542.
- ZHANG, Z.D. & KITCHING, R.P., 2001. The localisation of persistent foot-and-mouth disease virus in the epithelial cells of the soft palate and pharynx. Journal of Comparative Pathology, 124, 89–94.
- ZIBERT, A., MAAS, G., STREBEL, K., FALK, M.M. & BECK, E., 1990. Infectious foot-and-mouth disease virus derived from a cloned full-length cDNA. Journal of Virology, 64: 2467–2473.