- Infectious Diseases of Livestock
- Part 2
- Bovine ephemeral fever
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Bovine ephemeral fever
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). PD kirkland and TD St George, Bovine ephemeral fever, 2018.

Bovine ephemeral fever
Previous Authors: T D ST GEORGE
Current Authors:
P D KIRKLAND - Senior Principal Researcher Scientist, Manager Virology Laboratory, BVSc, PhD, FAMS, Elizabeth Macarthur Agriculture Institute, Woodbridge Road, Menangle, New South Wales, 2568, Australia
T D ST GEORGE - Retired from Commonwealth Scientific & Industrial Research Organisation, DVM, MVSc, BVSc, Virus Consultants International, 44/260 Cliveden Avenue, Corinda, Queensland, 4075, Australia
Introduction
Bovine ephemeral fever (BEF) is an infectious disease of cattle and water buffalo (Bubalus bubalis) caused by an arthropod-borne Ephemerovirus and is characterized by short duration, fever, stiffness, and disinclination to move, but which in most cases results in complete recovery.
The first recognition of ephemeral fever as a major disease in southern Africa was during an epidemic in Zimbabwe in 1906, although elderly inhabitants there considered it to be the re-emergence of a disease for which they already had a name in their tribal language. Brief reference in Schweinfurth’s4 account of his exploration of central Africa takes its history back a further 40 years to 1868–71. The first scientific account from Africa, giving an excellent description of an epidemic in Egypt in 1895, was in a prize-winning essay by Piot, a French veterinarian.41 It was not until 1936 that BEF was recognized in Australia and, much later again, in Japan.
The name of the disease has varied: ‘Dengue of cattle’;41 ‘drie-dae-stywesiekte’ (Afrikaans); ‘three days’ or ‘three-day stiffsickness’ in Africa and Australia; and ‘bovine epizootic fever’ in Japan.29 The term ‘ephemeral fever’, coined sometime between 1908 and 1910 in southern Africa, is the most widely used name.4 The obvious losses caused by BEF are loss of milk production, loss of draught power, abortion, and infertility in bulls and occasionally deaths of valuable animals as well as the expense of care or treatment and vaccines.48 The insidious losses result from a decrease in muscle mass during the time of the year when weight gain should be most rapid.
Bovine ephemeral fever is regarded as an important exotic disease in Europe and the Americas and the virus may be handled only in high security laboratories there. This seems paradoxical in view of the dependence on mosquitoes for its transmission.
Aetiology
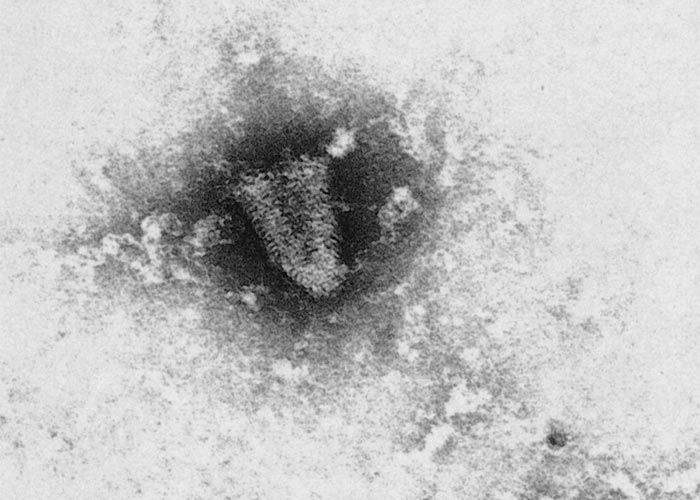
Bovine ephemeral fever virus (BEFV) belongs to the family Rhabdoviridae, genus Ephemerovirus. It is a single-stranded negative sense RNA virus with five structural and one non-structural protein.69 Van der Westhuizen66 first illustrated the bullet-shaped morphology (Figure 1) by electron microscopy after his pioneering isolation of BEFV. Other than full bullet shapes, there are shortened forms and cones, which are probably defective interfering particles with uncertain antigenic characteristics.
Bovine ephemeral fever virus is susceptible to both high and low pH27 and does not survive long outside its vertebrate or invertebrate host. It is rapidly inactivated by the high concentrations of lactic acid which develop in the muscles of cattle after death. Fomites, body discharges or tissues therefore play no part in the transmission of BEF.
There is no evidence of immunogenic diversity within the BEFV population.45 Epitope mapping has demonstrated antigenic variation.12
For many years BEFV was considered to be unrelated to other. However, antigenic and genetic similarities to BEFV have been found in Kimberley, Berrimah and Adelaide River rhabdoviruses,14, 23, 24 which are now classified in the genus Ephemerovirus. Therefore, any virus isolated from a suspect case of BEF must be identified by tests using monoclonal antibodies, virus-specific RT-PCR or nucleic acid sequencing. A neutralization test using BEFV-specific antiserum is an alternative. Group reactive fluorescence tests have limited value. Furthermore, infection of cattle with related viruses can induce low titres of neutralizing antibodies to BEFV. Consequently, serological surveys of cattle and wildlife that have been conducted exclusively with BEFV do not provide definitive evidence of infection with this virus. Since Africa has a rich store of rhabdoviruses, it is likely that further antigenically related viruses will be found.
It is a curious feature of BEFV that it replicates poorly, or not at all, in tissue cultures of bovine origin and induces high titres of interferon. Furthermore, the virus also appears to lose antigenicity for cattle with repeated passage in cultures of baby hamster kidney, hamster lung or monkey kidney tissue cultures.31
Epidemiology
The seasonal occurrence of BEF suggested insect transmission to the early investigators in Africa.4, 5, 22, 56 Epidemiological and pathogenesis studies implicate mosquitoes as the important vectors of BEFV. The range of species involved is likely to be wide as BEFV has been isolated from culicine (mixed pool) and anophilines (Anopheles bancroftii)46 and detected by real-time reverse transcription PCR (qRT-PCR) in Culex annulirostris20 in Australia. It has not yet been isolated from mosquitoes elsewhere in the world.
Experimental transmission is only achieved when BEFV infected blood is injected into the bloodstream. Mosquitoes feed by inserting their mouthparts and saliva directly into a venule. In contrast, Culicoides spp. are pool feeders and take up virus in the blood that oozes into the tiny surface wound they make on cattle. There is evidence that BEF virus may replicate in Culicoides spp. as the virus has been isolated from wild-caught mixed species in Kenya,16 C. imicola and C. coarctatus in Zimbabwe7 and C. brevitarsis in Australia.13 The presence of virus in these Culicoides spp. does not prove that they can transmit it. By the use of microtechniques, BEFV has been shown to be excreted in the saliva of infected mosquitoes (Culex annulirostris) but not in the saliva of midges (C. brevitarsis).37 If efficient BEFV isolation techniques34, 40 or qRT-PCR20 are applied to mosquitoes collected in an epidemic, the vectors can be further defined. The whole of Africa, Asia south of the former USSR, and Australasia (but excluding Papua New Guinea and New Zealand) may be regarded as falling within the infected zone even if clinical disease has not been formally reported there. Europe and the Americas are free of the infection. The limits of occurrence in countries where only part of the country is infected, such as Japan, China, and Australia, are mediated by climate through its effects on insect vectors.
In Australia, ephemeral fever has spread rapidly in cattle along watercourses even during times of drought, and well beyond the distribution of the Culicoides spp that are recognised arbovirus vectors,21 consistent with a mosquito vector. Bovine ephemeral fever is essentially a summer and autumn disease in subtropical and temperate areas of Africa, Asia, and Australia, disappearing with the first frosts. In tropical areas, it is linked to the rainy season. This is illustrated, in particular, by sporadic outbreaks several weeks after unseasonal heavy rainfalls in Kenya and tropical Australia.15, 48 In Egypt, BEF occurs at a time when rain is virtually unknown,42 but its presence is probably linked to the effect of the Nile River’s water level on vector breeding. In Israel, epidemics have occurred in arid areas where vector-breeding sites are associated with waste-water from dairy farms.
The movement of the 1906 epidemic in southern Africa was clearly from north to south; first Matabeleland in Zimbabwe, then the Limpopo, North West, Mpumalanga and Gauteng provinces, KwaZulu-Natal Province, and finally the Eastern and Western Cape provinces of South Africa.22 In Australia, similar north to south movement has been repeated many times.39, 44, 53, 63 In the northern hemisphere, in contrast, the movement is south to north, as in Egypt, China and Taiwan. In Japan, only the southern part of the main island has so far been affected.
None of the information that has become available since the 1906 epidemic has given any indication of a north-south or otherwise directional movement in southern Africa. Serious outbreaks may be widespread in some years while in other years they are sporadic and restricted to certain parts of the country. Bovine ephemeral fever appears to have had a high prevalence and been more widespread than usual in 1955 and the periods 1966–68, 1974–78, 1981–84 and 1999–2000.3, 68 However, the causes of this periodicity (including cyclical weather patterns) remain unknown.
In South Africa, cases of BEF are most common in the second half of summer, but tend to occur a few months earlier in the winter rainfall region of the Western Cape Province.68
In Australia, the apparent speed of movement of epidemics has varied from six weeks to two years to spread among essentially the same cattle population over a distance of 3 000 km. The dominating influences are probably the proportion of susceptible cattle in the population, the effects of weather on breeding, and down-wind drift of vectors. The duration of disease within a herd is influenced essentially by these factors as well as by the size of the herd. In an explosive, fast-moving epidemic, individual cases often occur five to seven days ahead of the main wave. Concurrent infection with other arthropod-borne viruses may interfere with the infection of the cattle with BEFV.47
The over-wintering mechanisms of the infection are unknown. Serological monitoring of sentinel cattle in an endemic area in Australia indicated that the virus was unlikely to over-winter in cattle. The presence of antibodies in the bloodstream would preclude any hypothetical resurgent virus becoming available to an insect vector.
Viraemia in experimentally infected cattle usually lasts about four to five days, commencing the day before fever and terminating one to two days after clinical recovery.36, 64 Very occasionally viraemia may persist for as long as 13 days after infection.
In general, one attack of BEF confers life-long immunity. However, within a single epidemic, double or triple bouts of clinical disease have been observed in Australia. MacFarlane and Haig35 reported two sequential attacks in individual South African cattle and attributed the cause to different strains of BEFV, but did not publish any supporting data. The statement common to sequential disease episodes is that subsequent bouts are always more severe than the preceding ones. An alternative explanation for this phenomenon is that the signs of other diseases, such as infection with another Ephemerovirus, Ibaraki disease or epizootic haemorrhagic disease of deer closely resemble ephemeral fever.
In Africa, a number of species of free-living ruminants have neutralizing antibodies to BEFV; Davies et al.15 found antibody in Kenyan African buffalo (Syncercus caffer), waterbuck (Kobus ellipsiprymnus), wildebeest (Connochaetes taurinus) and hartebeest (Acelaphus baselaphus). Young and Van den Heever70 found neutralizing antibody in S. caffer in South Africa. However, the specificity of the antibodies detected remains to be confirmed.
In Australia, marsupials, which form the bulk of the indigenous wildlife, are free of antibodies, though introduced species of deer and water buffaloes develop antibodies without exhibiting disease. Sheep appear to be unaffected even when pastured with cattle during an epidemic, and there is no serological evidence that subclinical infection occurs.49 However, BEFV has been passaged through sheep and back to cattle under experimental conditions.25 There is a single report of neutralizing antibody in goats.6
Pathogenesis
Bovine ephemeral fever primarily affects the endothelium of small blood vessels.36, 50 The inflammatory nature of the disease is associated with arterioles, venules and capillaries of the synovial membranes, tendon sheaths, muscles, fasciae and skin.3 A two- to fourfold increase in plasma fibrinogen levels confirms the major systemic inflammatory response. The result is the effusion of fibrin-rich fluid into joint, peritoneal, pleural and pericardial cavities and discharges from nasal and ocular mucosal surfaces.
The secondary effect is on the plasma mineral components 51 (Table 1; Figures 2); there being a fall in the total calcium, iron and zinc levels and a rise in copper level. The fall in plasma calcium occurs in both the ionized and bound fractions.38 Absorption of calcium from the food which has been ingested ceases, creating a cycle that will only be broken when the effect of the virus is removed. Reduction in the ionized fraction of the plasma calcium is the result of an alkalosis during clinical disease. More severe clinical disease and a higher mortality rate than usual occurred in Australia in well-conditioned cattle grazing virtually pure buffel grass (Cenchrus ciliaris) pastures which have a high oxalate content. It is presumed that the high oxalate levels lower plasma calcium further than would generally be the case.49
The paresis that is usually encountered in acute disease is accompanied by the other signs of hypocalcaemia, namely depression, muscular fibrillation, ruminal stasis, inability to swallow, constipation and recumbency. These are accompanied by the same biochemical changes that occur in parturient hypocalcaemia (milk fever).52
The supposition that neural lesions are not usually responsible for the temporary paralysis is proved by the response of specific hypocalcaemic signs to calcium infusion.52 The rapid breathing that occurs at intervals during illness reduces the carbon dioxide level in the blood affecting its pH (Figure 2e). There is, furthermore, a similarity between the biochemical and haematological changes which occur in BEF and those surrounding normal parturition, suggesting that there is a common basis for the hypocalcaemia which occurs in both conditions.52 The pathogenesis of the Wallerian degeneration observed in the cervical spinal cord in those few cases which remain permanently paralysed28 is obscure. However, detailed studies of peripheral nerves and the central nervous system of naturally infected cattle have shown the presence and persistence of viral antigen in lesions in affected tissues.2
The paralysis induced by BEFV infection appears to have at least 2 origins – biochemical and neural-as treatment does not always restore limb function. Recent research into Guillain-Barre syndrome of humans suggests that cytokines produced by various viruses causes temporary demyelinization of nerves and an ascending paralysis. While studies have shown the presence of viral antigens in peripheral nerves and brain of some affected animals, it has been suggested that some of the lesions in permanently paralysed animals may be due to pressure effects induced by prolonged recumbency.2
Interferon circulates in high titre during the clinical course of BEF.74 After fever has subsided and the affected animal is fully alert, recovery from paresis can still occur.
It is probable that BEF virus does not cause tissue damage directly by destroying infected cells, but rather by inducing massive cytokine production. In addition to the beneficial effects of limiting virus infections, interferon produces a toxic effect, mediated through other substances, which produces clinical signs similar to those of early BEF. Thus, the principal clinical signs are accounted for by the generalized inflammation, which may be initiated by the very high interferon levels in natural and experimental cases,55 and a consequent biochemical dyscrasia (especially the fall in ionized calcium levels).38
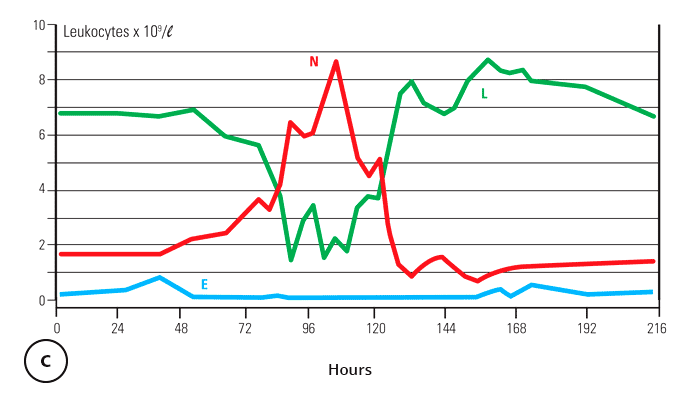
The published evidence indicates that BEFV is contained in the leukocyte fraction of the blood during fever36, 57 and, more particularly, in neutrophils.68, 71, 72 The site of initial replication of BEFV in cattle is unknown, but the virus is circulating in the blood in high titre approximately one day prior to the onset of fever and associated neutrophilia (Figure 2c).
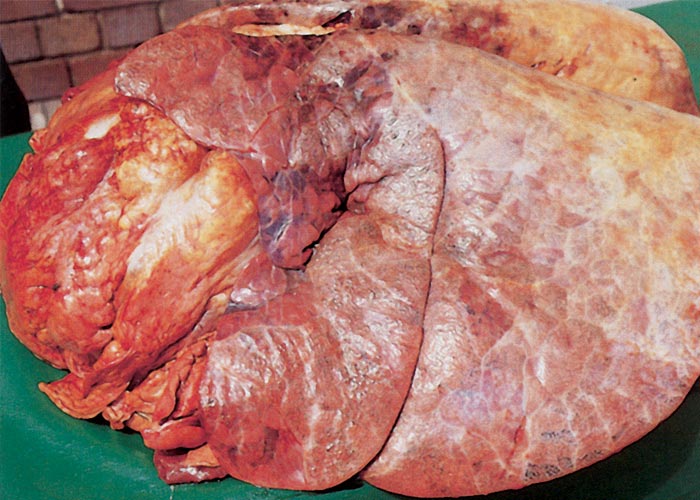
The pathogenesis of the pulmonary and subcutaneous emphysema seen in a small proportion of cases is not known but may be due to partial blockage of the air passages with exudate as well as to necrosis of the bronchiolar walls resulting in rupture of bronchioles and alveoli.59 Air is then able to reach the connective tissue septa and lymphatics of the lungs and extends subpleurally to reach the mediastinum. From here air can spread through the thoracic inlet to the subcutaneous tissues.
Table 1 Changes in clinical, haematological and biochemical parameters in cattle during ephemeral fever1, 4
| UNIT | BEFORE ILLNESS (RANGE) | DURING ILLNESS (RANGE) | INCREASE (+) OR DECREASE (−) | |
| BLOOD | ||||
| Lymphocytes | % | 65–80 | 20–50 | − |
| ×109/l | 2,5–6,5 | 1,5–5,0 | − | |
| Neutrophils | ×109/l | 1,0–3,5 | 5,0–17,0 | + |
| Monocytes | % | 1–2 | 4–10 | + |
| Eosinophils2 | % | 1–10 | 0–1 | − |
| pH (venous) | 7,3–7,4 | 7,5–7,6 | + | |
| PLASMA | ||||
| Fibrinogen | g/l | 3,7–10,0 | 12,3–21,3 | + |
| Ammonia | µmol/l | 30–50 | 70 | + |
| Glucose | µmol/l | 1,7–4,3 | 4,1–5,5 | + |
| Phosphate | µmol/l | 2,16–2,96 | 0,97–1,30 | − |
| SERUM | ||||
| Interferon | Units | < 8 | 512–2048 | + |
| Albumin | g/l | 32–40 | 24–30 | − |
| Calcium | mmol/l | 2,2–2,8 | 1,9–2,1 | − |
| Magnesium | mmol/l | 0,8–1,3 | 0,7–0,8 | − |
| Iron | µg/l | 1500–2100 | 340–780 | − |
| Copper | µg/l | 360–600 | 730–1300 | + |
| Zinc | µg/l | 660–1400 | 100–500 | − |
| Non esterified fatty acids | µmol/l | < 50 | 400 | + |
| Neut. antibody titre3 | 0 | 16–512 | + | |
| Serum carboxy peptidase B | Units of activity | 0,035–0,044 | 0,016–0,028 | − |
| Rectal temperature °C | 38–38,8 | 39,5–42 | + | |
| Heart rate/min. | 60–70 | 80–120+ | + | |
| Respiratory rate/min | 10–30 | 60–100 | + |
- Except where specified, the data in the table was provided by T.D. St. George and M.F. Uren, CSIRO Division of Tropical Animal Production, Australia, and G.M. Murphy, Queensland Department of Primary Industries, Australia. Unpublished data 1988
- Cattle with an eosinophilia prior to illness experience a marked depletion of eosinophils
- Cattle previously exposed to an antigenically related rhabdovirus may have cross-reacting neutralizing antibody to BEFV before clinical disease and may experience a secondary rise to titres in excess of 512
- Uren and Murphy59
Clinical signs
Bovine ephemeral fever is in most cases a self-limiting, disabling disease of a few days’ duration.36 Convalescence is rapid, even in those animals that are severely affected. Individual cattle show a wide variation in the range, severity and duration of clinical signs but the onset is always sudden and accompanied by a sharp rise in body temperature.
Clinical signs are more severe in lactating than in nonlactating cows, in heavily conditioned animals and in mature cattle than in calves. Mortality is low, seldom exceeding 2 or 3 per cent.26
The incubation period in the field is unknown but three to five days is common in experimental infections with extremes of between 24 hours and 7 days.
The fever is phasic, with two or more peaks 12 to 24 hours apart. Clinical signs in the first febrile phase (40 to 42 °C), which lasts 12 to 24 hours, are mild or even imperceptible unless the animals are closely observed, and are always milder than in the later phase or phases. During the first febrile episode cattle may exhibit nothing more than depression, disinclination to move, and stiffness, but nevertheless maintain their appetite. Many young cattle recover completely after this febrile phase but if the disease progresses they may become separated, anorexic, and stiff, developing a shifting lameness and moving their weight from limb to limb when standing. There is usually a serous nasal discharge and, less frequently, an ocular discharge and swelling of the joints (Figure 3). The pulse rate accelerates, the respiratory rate is increased in cycles, and subcutaneous muscles may fasciculate or exhibit tremors. In some cases patchy, and particularly periorbital, subcutaneous oedema of the head occurs. Clinical recovery is usually complete within one or two days of the subsidence of fever.
Recumbency is the obvious feature of the next order of severity (Figure 4). The affected animal is at first in sternal recumbency and able to rise in response to a strong stimulus, but is later completely unable to do so. Loss of the swallowing reflex, bloat, ruminal stasis, constipation, and excessive salivation may be evident and the head may be turned on the flank. From sternal recumbency, the animal lapses into lateral recumbency with further reflex loss, progressing to coma and death. In these cases there is no voluntary or reflex response to the flies that settle on the facial discharges. Resolution of clinical signs may occur spontaneously up to, and including, the early paralytic stage.
An early sign of the disease in lactating cows is an abrupt decline in milk production. The quality of the milk is severely affected and there is a high cell count. Except in those cows which are at a very early stage in lactation, the milk yield does not return to pre-illness levels.17, 60 Cows in the second half of lactation may not return to production.
Moist râles can be heard in the lungs as the second febrile stage commences. Pneumonia may be a secondary complication, and the clinical signs associated with severe pulmonary emphysema, which may eventually result in subcutaneous emphysema of the backline rarely extending over most of the body, have been observed in a small proportion of cases.19, 35, 59
Abortion occurs in about five per cent of pregnant cows, especially those in late pregnancy. This is economically important not only because the foetus, but also the whole of the impending lactation, is lost. Fertility subsequent to abortion does not seem to be affected.62 The cause of the temporary infertility and of abnormal spermatozoa in bulls is unknown but is likely to be related to the high temperature fever.10 However, early treatment of bulls with anti-inflammatory drugs will prevent effects on fertility.18
Pathology
There are few descriptions of the pathological changes in field cases of BEF. However there is one detailed study of a group of naturally infected cattle2 and the lesions in experimental cases have been described.3
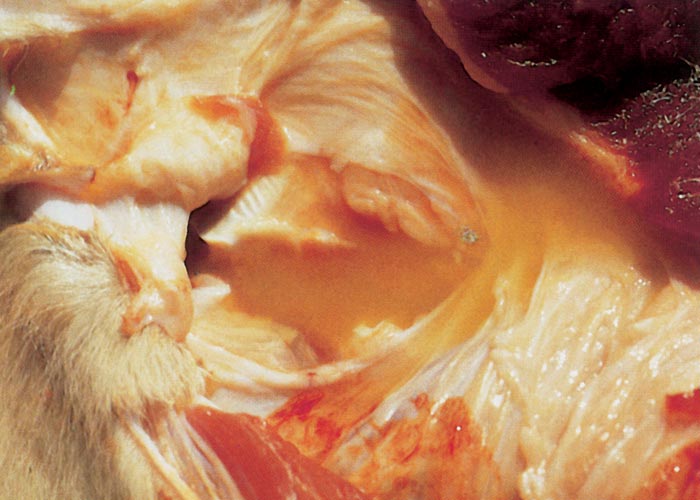
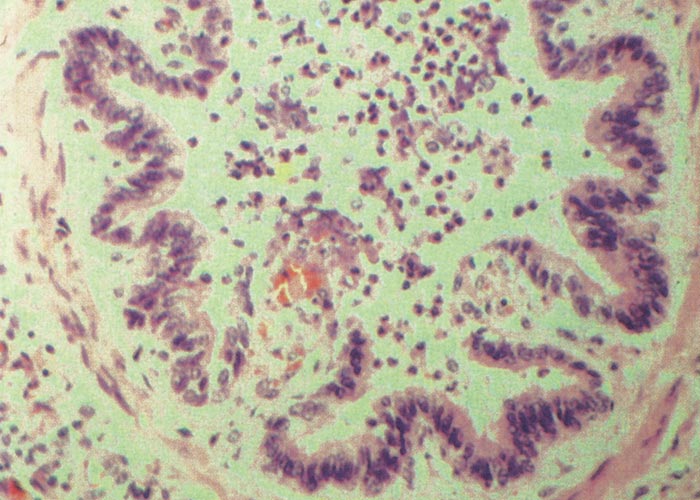
The most conspicuous macroscopic changes are serofibrinous polysynovitis (Figure 5), tendovaginitis, cellulitis and focal necrosis of skeletal muscles, with the quadriceps group and the larger muscles of the shoulder and back being particularly affected. These lesions are variable in severity, being barely discernible in some animals and very marked in others. The lesions in both joints and muscles are invariably more severe in the limbs on which the animal was limping. Muscle lesions are predominantly located near the origins and insertions. The amount of fibrin-flecked fluid in joint capsules, and peritoneal, pericardial and pleural cavities varies. A generalized oedema of lymph nodes is invariably present. The lungs show patchy oedema and, in a small number of cases, severe alveolar and interstitial emphysema (Figure 6).59 The latter may result in the formation of bullae 20 to 100 mm in diameter in the lungs, as well as subpleurally, in the mediastinum, subperitoneally and in the subcutis.
Inflammatory changes in the small blood vessels are considered to be the fundamental histopathological lesion3, 5 but these tend to have an irregular distribution. Initially, swelling and hyperplasia of the endothelium is accompanied by perivascular neutrophil infiltration and oedema. Subsequent changes comprise focal or extensive necrosis of vessel walls and thrombosis. Focal areas of hyaline necrosis in skeletal muscles are almost invariably associated with thrombosis. The lymph nodes show oedema and neutrophils in the sinuses. The lungs have areas of patchy, lobular congestion, oedema and atelectasis. Exudate, which consists of desquamating epithelial cells, inflammatory cells and fibrin,59 may be evident in bronchioles and bronchi (Figure 7) and is often accompanied by emphysema and bullae formation.59 Although the major organs appear to be essentially normal macroscopically, microscopic changes do occur in some blood vessels. Cattle killed in the acute stages of infection infrequently have lesions in the central nervous system, but Wallerian degeneration may occur in the upper cervical spinal cord in cattle with long-standing ataxia or paralysis after recovery from the acute febrile stage of the disease.27 Detailed studies of the central and peripheral nerves of naturally infected cattle killed at various times after infection have demonstrated a distinct neurotropism for BEFV, with viral RNA detected in nervous tissue during the acute viraemic stage of infection and viral antigen detected in both peripheral and central nervous tissue at later times after infection. There was evidence of degenerative changes in both central and peripheral nerves with tropism for specific cell types and it has been postulated that a peripheral neuropathy may play an important role in the development of recumbency in BEFV infected animals.2
Diagnosis
A reliable presumptive diagnosis of BEF is possible. This is based on the sudden onset of disease in late summer, affecting sizeable numbers of cattle in areas where BEF is known to occur. However, when a single animal is presented early in an epidemic a diagnosis may be difficult to achieve unless blood samples are collected from the animal in the very early stages of disease.
The introduction of a qRT-PCR assay has revolutionised confirmation of BEFV infection, provided blood samples are collected within the first few days of disease. This assay detects viral RNA in the blood sample. EDTA treated blood samples are preferred. Once samples reach a laboratory, a diagnosis can be confirmed by qRT-PCR within a few hours, supporting ‘same day’ confirmation of BEF.20
Another direct confirmatory test is a differential leukocyte count, which may be performed crudely on a blood smear. If there is no neutrophilia with at least 30 per cent of band forms (Table 1), the animal does not have BEF but may have another viral disease. However, a high percentage of neutrophils is not pathognomonic of ephemeral fever, as generalized bacterial infections can have the same effect. The clots formed by blood taken during the latter stages of illness contract poorly, even if left for several days.
Bovine ephemeral fever virus is isolated most efficiently in cultures of Aedes albopictus cells70 and then adapts to mammalian cells such as hamster kidney or lung cell cultures. The intracerebral inoculation of mice less than two days old 66 is very inefficient by comparison.
Serological confirmation of ephemeral fever may be obtained by the detection of a rise in neutralizing antibody titre between sera collected from individual animals during acute illness and again two to three weeks later. There is, however, no clear guideline as to what constitutes a diagnostic rise, because animals, which have been exposed to an antigenically related virus, such as Kimberley virus, may have low titres (<20) of neutralizing antibody to BEFV in acute phase serum. Instead of a primary antibody response commencing two or three days after clinical recovery, an anamnestic response begins on the second day of illness and the antibody titre reaches a plateau approximately five days later.11, 51 The blocking ELISA test73 gives similar results to the neutralization test.
Differential diagnosis
Stiffness, lameness and recumbency are characteristic of a variety of conditions prevalent in cattle in southern Africa. Conditions such as aphosphorosis which is characterized by osteomalacia; the toxicosis caused by Crotalaria burkeana and certain other Crotalaria spp., which produces laminitis and overgrowth of the hooves;32 botulism; blackquarter (Clostridium chauvoei infection); the neurotoxicosis resulting from the ingestion of maize infected with the fungus, Stenocarpella maydis ( Diplodia maydis);32, 33 and physical injury may all initially be confused with BEF. The colloquial nomenclature commonly used for the first two of these conditions, ‘stywesiekte’ (literal translation — stiff sickness) may also be a source of confusion.
The pulmonary and subcutaneous emphysema that occurs in a small proportion of BEF cases should be differentiated from fog fever or acute pulmonary oedema and emphysema associated with the production of the pneumotoxin 3-methylindole from dietary L- tryptophan in the rumen.9, 32
In Japan, the clinical signs of Ibaraki disease were initially confused with those of BEF.30 The main distinguishing feature of Ibaraki disease is permanent paralysis of the oesophagus due to the degeneration of striated muscle, and erosions of the muzzle and mouth.
Control
Bovine ephemeral fever is one of the rare viral diseases for which rationally based treatment is possible.54, 64 The treatment has six elements:
- Rest, with protection from the elements where possible, as well as the provision of water and feed when recovery begins.
- Valuable breeding stock may merit parenteral rehydration in hot weather.
- Anti-inflammatory (either steroidal or non-steroidal) treatment designed to be effective for several days should be given as early as possible.64 The fever and clinical appearance of cows are modified or restored to apparent normality by treatment with anti-inflammatory drugs, but neither the fall in plasma zinc and iron nor the rise in plasma copper38 is affected.
- If signs of hypocalcaemia are observed (such as ruminal stasis, paresis, or loss of reflexes), then the treatment for parturient hypocalcaemia should be instituted. Calcium borogluconate solution, as used for the treatment of milk fever, is injected subcutaneously (if treated by a stockman) or intravenously by a veterinarian who can monitor the response of the heart to avoid an overdose. Follow-up administrations given subcutaneously every six hours for one day are necessary in severe cases. The first response is a slowing of the breathing and a return of the eye, rumen, oesophageal and tail reflexes. The animal becomes more alert and makes an effort to resume sternal recumbency. It will then try to move to water, or will drink copiously if water is offered. Further improvement is sometimes slow.
- Nothing should be given by mouth unless the swallowing reflex is observed to be functional or until after the administration of calcium borogluconate.
- Convalescent animals should not be stressed or worked for several days, since clinical recovery occurs before all biochemical functions have returned to normal and before tissue repair is complete.
- In endemic areas, it is important to immunize cattle, particularly dairy and feedlot herds and valuable breeding stock, to preclude production losses caused by BEF. Immunization should ideally be carried out in spring (e.g. September in the southern hemisphere) to ensure a high level of immunity during the ensuing summer and autumn. In calves younger than six months old, maternally derived antibody may interfere with the response to vaccine.
- All vaccines currently in use are produced in tissue culture but, unusually for vaccines containing live virus, most also incorporate an adjuvant.8, 58, 67 Revaccination each season is necessary to maintain immunity. A killed vaccine that uses QuilA as an adjuvant is commercially available in Australia. A subunit vaccine has produced sterile immunity under experimental conditions.65
- The vectors involved in transmitting BEFV have not all been identified. At present there is no effective means of interrupting the cycle of the virus within the vector stage of its life cycle.
References
- ANON., 1956–1986. Annual reports of the Directorate of Animal Health. Department of Agriculture and Water Supply, South Africa.
- BARIGYE, R., DAVIS, S., HUNT,R., HUNT,N., WALSH,S., ELLIOTT,N., BURNUP, C., AUMANN, S., DAY, C., DYRTING, K., WEIR, R. & MELVILLE, L.F. 2016. Viral neurotropism, peripheral neuropathy and other morphological abnormalities in bovine ephemeral fever virus-infected downer cattle. Australian Veterinary Journal 94, 362–370
- BASSON, P.A., PIENAAR, J.G. & VAN DER WESTHUIZEN, B., 1969. The pathology of ephemeral fever: A study of the experimental disease in cattle. Journal of the South African Veterinary Medical Association, 40, 385–397.
- BEVAN, L.E.W., 1907. Preliminary report on the so called ‘stiff-sickness’ or ‘three-day-sickness’ of cattle. Journal of Comparative Pathology and Therapeutics, 20, 104–113.
- BEVAN, L.E.W., 1912. Ephemeral fever, or three days’ sickness of cattle. Veterinary Journal, 68, 458–461.
- BIGGS, H., 1988. Central Veterinary Laboratory, Windhoek, Namibia. Unpublished data.
- BLACKBURN, N.K., SEARLE, L. & PHELPS, R.J., 1985. Viruses isolated from Culicoides (Diptera: Ceratopogonidae) caught at the veterinary research farm, Mazowe, Zimbabwe. Journal of the Entomological Society of Southern Africa, 48, 331
- CAMERON, C.M., BARNARD, B.J.H., ERASMUS, E. & BOTHA, W.J.S., 1987. Antibody response in cattle to oil emulsion rabies and ephemeral fever vaccines. Onderstepoort Journal of Veterinary Research, 54, 157–158.
- CARLSON, R.J. & BREEZE, R.G., 1984. Ruminal metabolism of plant toxins with emphasis on indolic compounds. Journal of Animal Science, 58, 1040–1049.
- CHENOWETH, P.J. & BURGESS, G.W., 1972. Mid-piece abnormalities in bovine semen following ephemeral fever. Australian Veterinary Journal, 48, 37–38.
- CYBINSKI, D.H., 1987. Homologous and heterologous antibody reactions in sera from cattle naturally infected with bovine ephemeral fever group viruses. Veterinary Microbiology, 13, 1–9.
- CYBINSKI, D.H., DAVIS, S.S. & ZAKRZEWSKI, H., 1999. Antigenic variation of the bovine ephemeral fever virus glycoprotein. Archives Virology, 124, 211–224.
- CYBINSKI, D.H. & MULLER, M.J., 1990. Isolation of arboviruses from cattle and insects at two sentinel sites in Queensland, Australia, 1979–1985. Australian Journal of Zoology, 38, 25–32.
- CYBINSKI, D.H. & ZAKRZEWSKI, H., 1983. The isolation and preliminary characterization of a rhabdovirus in Australia related to bovine ephemeral fever virus. Veterinary Microbiology, 8, 221–235.
- DAVIES, F.G., SHAW, T. & OCHIENG, P., 1975. Observations on the epidemiology of ephemeral fever in Kenya. Journal of Hygiene, Cambridge, 75, 231–235.
- DAVIES, F.G. & WALKER A.R., 1974. The isolation of ephemeral fever virus from cattle and Culicoides midges in Kenya. The Veterinary Record, 95, 63–64.
- DAVIS, S.S., GIBSON, D.S. & CLARK, R., 1984. The effect of bovine ephemeral fever on milk production. Australian Veterinary Journal, 61, 128–129.
- DOWSETT, K. & ST. GEORGE, T.D., 1971. Commonwealth Scientific and Industrial Research Organisation, Canberra, Australia. Unpublished data.
- ERASMUS, B.J., COETZER, J.A.W., THEODORIDIS, A. & PIENAAR, J.G., 1974. Extensive interstitial emphysema as a complication of ephemeral fever. Journal of the South African Veterinary Medical Association, 45, 240.
- FINLAISON, D.S., READ, A.J. & KIRKLAND, P.D., 2010. An epizootic of bovine ephemeral fever in New South Wales in 2008 due to long distance dispersal of vectors. Australian Veterinary Journal, 88, 301-306.
- FINLAISON, D.S., READ, A.J., ZHANG, J., PASKIN, R. & KIRKLAND, P.D. 2014. Application of a real time polymerase chain reaction assay to the diagnosis of bovine ephemeral fever during an outbreak in New South Wales and northern Victoria in 2010. Australian Veterinary Journal, 92, 24-27.
- FREER, G.W., 1910. Ephemeral fever or three-day sickness of cattle. Veterinary Journal, 66, 19–22.
- GARD, G.P., CYBINSKI, D.H. & ST. GEORGE, T.D., 1983. The isolation in Australia of a new virus related to bovine ephemeral fever virus. Australian Veterinary Journal, 60, 89–90.
- GARD, G.P., CYBINSKI, D.H. & ZAKRZEWSKI, H., 1984. The isolation of a fourth bovine ephemeral fever group virus. Australian Veterinary Journal, 61, 332.
- HALL, W.T., DADDOW, K.N., DIMMOCK, C.K., ST. GEORGE, T.D. & STANDFAST, H.A., 1975. The infection of merino sheep with bovine ephemeral fever virus. Australian Veterinary Journal, 51, 344–346.
- HENNING, W.H., 1956. Ephemeral fever, three-day-sickness, drie-daesiekte. Animal Diseases in South Africa. 3rd edn. Pretoria, South Africa: Central News Agency.
- HEUSCHELE, W.P., 1970. Bovine ephemeral fever. I. Characteristics of the causative virus. Archiv für die Gesamte Virusforschung, 30, 195–202.
- HILL, M.W.M. & SCHULTZ, L., 1977. Ataxia and paralysis associated with bovine ephemeral fever infection. Australian Veterinary Journal, 53, 217–221
- INABA, Y., 1968. Bovine epizootic fever. Japan Agricultural Research Quarterly, 3, 36–42.
- INABA, Y., 1975. Ibaraki disease and its relationship to bluetongue. Australian Veterinary Journal, 51, 178–185.
- INABA, Y., TANAKA, Y., SATO, K., ITO, H., OMORI, T. & MATUMOTO, M., 1969. Bovine epizootic fever. III. Loss of virus pathogenicity and immunogenicity for the calf during serial passage in various host systems. Japanese Journal of Microbiology, 13, 181–186.
- KELLERMAN, T.S., COETZER, J.A.W. & NAUDÉ, T.W., 1988. Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa. Cape Town: Oxford University Press Southern Africa.
- KELLERMAN, T.S., RABIE, C.J., VAN DER WESTHUIZEN, G.C.A., KRIEK, N.P.J. & PROZESKY, L., 1985. Induction of the neuromycotoxicosis, diplodiosis, in domestic ruminants with cultures of indigenous and exotic isolates of Diplodia maydis. Onderstepoort Journal of Veterinary Research, 43, 147–154.
- KIRKLAND, P.D., 1993. The epidemiology of bovine ephemeral fever in south-eastern Australia: Evidence for a mosquito vector. Bovine Ephemeral Fever and Related Rhabdoviruses, Australian Centre for International Agricultural Research Proceedings No. 44, Canberra, Australia.
- MACFARLANE, I.S. & HAIG, D.A., 1955. Some observations on three-day stiff-sickness in the Transvaal in 1954. Journal of the South African Veterinary Medical Association, 36, 1–7.
- MACKERRAS, I.M., MACKERRAS, M.J. & BURNET, F.M., 1940. Experimental studies of ephemeral fever in Australian cattle. Council for Scientific and Industrial Research, Melbourne, Australia, Bulletin, 20a, No. 136.
- MULLER, M.J., 1989. Commonwealth Scientific and Industrial Research Organisation, Canberra, Australia. Unpublished data.
- MURPHY, G.M., ST. GEORGE, T.D., UREN, M.F. & COLLINS, R.G., 1986. The biochemistry of ephemeral fever in cattle. In: ST. GEORGE, T.D., AKY, B.H. & BLOK, J., (eds). Proceedings of the Fourth Symposium on Arbovirus Research in Australia Brisbane, Australia, 6–9 May 1986. Commonwealth Scientific and Industrial Research Organization/ Queensland Institute for Medical Research, Brisbane, 307–313.
- MURRAY, M.D., 1970. The spread of ephemeral fever of cattle during the 1967–68 epizootic in Australia. Australian Veterinary Journal, 46, 77–82.
- MURRAY, M.D., 1977. Possible vectors of bovine ephemeral fever in the 1967–68 epizootic in northern Victoria. Australian Veterinary Journal, 75, 220.
- PIOT, J.B., 1896. Epizootic of dengue fever of cattle in Egypt. Prix Monbinne. Paris, France: National Academy of Medicine.
- RABAGLIATI, D.S., 1924. ‘Three days’ fever or stiff sickness in cattle. The Veterinary Record, 4, 23, 503–505.
- SCHWEINFURTH, G., 1878. The Heart of Africa. Tr. E.E. Frewer. London: Sampson Low, Marston, Low & Searle.
- SEDDON, H.R., 1938. The spread of ephemeral fever (three-day sickness) in Australia in 1936–37. Australian Veterinary Journal, 14, 90–101.
- SNOWDON, W.A., 1970. Bovine ephemeral fever: The reaction of cattle to different strains of ephemeral fever virus and the antigenic comparison of two strains of virus. Australian Veterinary Journal, 46, 258–266.
- STANDFAST, H.A., ST. GEORGE, T.D. & DYCE, A.C., 1976. The isolation of ephemeral fever from mosquitoes in Australia. Australian Veterinary Journal, 52, 242
- ST. GEORGE, T.D., 1985. Studies on the pathogenesis of bovine ephemeral fever in sentinel cattle. I. Virology and serology. Veterinary Microbiology, 10, 493–504.
- ST. GEORGE, T.D., 1986. The epidemiology of bovine ephemeral fever in Australia and its economic effect. In: ST. GEORGE, T.D., KAY, B.H. & BLOK, J., (eds). Proceedings of the Fourth Symposium on Arbovirus Research in Australia, Brisbane, Australia, 6–9 May 1986. Canberra: Commonwealth Scientific and Industrial Research Organization.
- ST. GEORGE, T.D., 1990. Commonwealth Scientific and Industrial Research Organization, Brisbane, Australia. Unpublished data.
- ST. GEORGE, T.D., 1993. The natural history of ephemeral fever of cattle. In: Bovine Ephemeral Fever and Related Rhabdoviruses. Australian Centre for International Agricultural Research Proceedings No. 44, Canberra, Australia.
- ST. GEORGE, T.D., CYBINSKI, D.H., MURPHY, G.M. & DIMMOCK, C.K., 1984. Serological and biochemical factors in bovine ephemeral fever. Australian Journal of Biological Sciences, 37, 341–349.
- ST. GEORGE, T.D., MURPHY, G.M., BURREN, B. & UREN, M.F., 1995. Studies on the pathogenesis of bovine ephemeral fever. IV. A comparison with the inflammatory events in milk fever. Veterinary Microbiology, 46, 131–142.
- ST. GEORGE, T.D., STANDFAST, H.A., CHRISTIE, D.G., KNOTT, S.G. & MORGAN, I.R., 1977. The epizootiology of bovine ephemeral fever in Australia and Papua-New Guinea. Australian Veterinary Journal, 53, 17–28.
- ST. GEORGE, T.D., UREN, M.F. & ZAKRZEWSKI, H., 1986. The pathogenesis and treatment of bovine ephemeral fever. In: ST. GEORGE, T.D., KAY, B.H. & BLOK, J., (eds). Proceedings of the Fourth Symposium on Arbovirus Research in Australia, Brisbane, Australia 6–9 May 1986. Canberra: Commonwealth Scientific and Industrial Research Organization.
- ST. GEORGE, T.D., UREN, M.F. & ZAKRZEWSKI, H., 1988. Commonwealth Scientific and Industrial Research Organization, Canberra, Australia. Unpublished data.
- THEILER, A., 1908. Stiff sickness or three-day sickness of cattle. Reports of the Government Veterinary Bacteriologist, 1906–07, 22–23.
- THEODORIDIS, A., 1969. Fluorescent antibody studies on ephemeral fever virus. Onderstepoort Journal of Veterinary Research, 36, 187–190.
- THEODORIDIS, A., BOSHOFF, S.E.T. & BOTHA, M.J., 1973. Studies on the development of a vaccine against bovine ephemeral fever. Onderstepoort Journal of Veterinary Research, 40, 77–82.
- THEODORIDIS, A. & COETZER, J.A.W., 1979. Subcutaneous and pulmonary emphysema as complications of bovine ephemeral fever. Onderstepoort Journal of Veterinary Research, 46, 125–127.
- THEODORIDIS, A., GIESECKE, W.H. & DU TOIT, I.J., 1973. Effect of ephemeral fever on milk production and reproduction of dairy cattle. Onderstepoort Journal of Veterinary Research, 40, 83–91.
- UREN, M.F. & MURPHY, G.M., 1985. Studies on the pathogenesis of bovine ephemeral fever. 2. Changes in serum calcium and enzyme levels. Veterinary Microbiology, 10, 505–515.
- UREN, M.F., ST. GEORGE, T.D., KIRKLAND, P.D., STRANGER, R.S. & MURRAY, M.D., 1987. Epidemiology of bovine ephemeral fever in Australia 1981– 1985. Australian Journal of Biological Science, 40, 125–136.
- UREN, M.F., ST. GEORGE, T.D. & STRANGER, R.S., 1983. Epidemiology of ephemeral fever of cattle in Australia 1975–1981. Australian Journal of Biological Science, 36, 91–100.
- UREN, M.F., ST. GEORGE, T.D. & ZAKRZEWSKI, H., 1989. The effect of anti-inflammatory drugs on bovine ephemeral fever. Veterinary Microbiology, 19, 99–111.
- UREN, M.F., WALKER, P.J., ZAKRZEWSKI, H., ST. GEORGE, T.D. & BYRNE, K.A., 1994. Effective vaccination of cattle using the virion G protein of bovine ephemeral fever virus as an antigen. Vaccine, 12, 845–850.
- VAN DER WESTHUIZEN, B., 1967. Studies on bovine ephemeral fever. I. Isolation and preliminary characterization of a virus from natural and experimentally produced cases of bovine ephemeral fever. Onderstepoort Journal of Veterinary Research, 34, 29–40.
- VANSELOW, B.S., ABETZ, I. & TRENFIELD, K., 1985. Bovine ephemeral fever vaccine incorporating adjuvant Quil A: A comparative study using adjuvant Quil A, aluminium hydroxide gel and dextran suphate. The Veterinary Record, 117, 37–43.
- VENTER, G.J., 1985. Veterinary Research Institute, Onderstepoort, South Africa. Unpublished data.
- WALKER, P.J., BYRNE, K.A., CYBINSKI, D.H., DOOLAN, D.L. & WONGHONG YANG., 1991. Proteins of bovine ephemeral fever virus. Journal of General Virology, 72, 67–74.
- YOUNG, E. & VAN DEN HEEVER, L.W., 1969. The African buffalo as a source of food and by-products. Journal of the South African Veterinary Medical Association, 40, 83–88.
- YOUNG, P.L. & SPRADBROW, P.B., 1980. The role of neutrophils in bovine ephemeral fever virus infection of cattle. Journal of Infectious Diseases, 142, 50–55.
- YOUNG, P.L. & SPRADBROW, P.B., 1985. Transmission of virus from serosal fluids and demonstration of antigen in neutrophils and mesothelial cells of cattle infected with bovine ephemeral fever. Veterinary Microbiology, 10, 199–207.
- ZAKRZEWSKI, H. & CYBINSKI, D.H., 1988. Commonwealth Scientific and Industrial Research Organization, Canberra, Australia. Unpublished data.
- ZAKRZEWSKI, H. & ST. GEORGE, T.D., 1987. Commonwealth Scientific and Industrial Research Organization, Canberra, Australia. Unpublished data.