- Infectious Diseases of Livestock
- Part 2
- Bovine respiratory syncytial virus infection
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Bovine respiratory syncytial virus infection
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Bovine respiratory syncytial virus infection
Previous authors: M VAN VUUREN
Current authors:
J L MCGILL - Assistant Professor, MS, PhD, Department of Veterinary Microbiology and Preventive Medicine, Iowa State University, 1907 Christensen Drive, VMRI Building 5, Iowa, 50011, United States of America
RE SACCO - Research Microbiologist/Immunologist /Lead Scientist, PhD, Ruminant Diseases and Immunology Research Unit, National Animal Disease Center/USDA/ARS, 1920 Dayton Avenue, Ames, Iowa, IA 50010, United States of America
Introduction
Infection with bovine respiratory syncytial virus (BRSV) is inapparent in the majority of animals, but in some it does cause mild to severe23 respiratory tract disease characterized by fever, coughing, serous nasal and ocular discharges, dyspnoea and, in some animals, subcutaneous emphysema. It is one of several viruses that are primary pathogens in the bovine respiratory disease complex (see Pneumonic pasteurellosis in cattle). Respiratory syncytial virus also infects sheep and goats and may cause rhinitis in sheep.25 Evidence for the existence of a respiratory syncytial virus (RSV) affecting horses has been reported.24
The virus was first isolated from cattle in Switzerland in 197040 and has subsequently been found worldwide. Its prevalence and importance in Africa is largely unknown, although antibody to it is widespread in feedlot cattle in South Africa.61
Several reviews on BRSV have been published.3, 7, 28, 36, 44, 46, 59
Aetiology
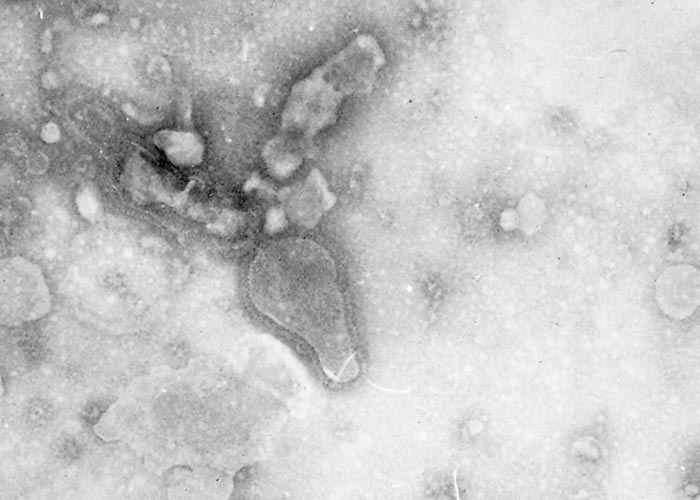
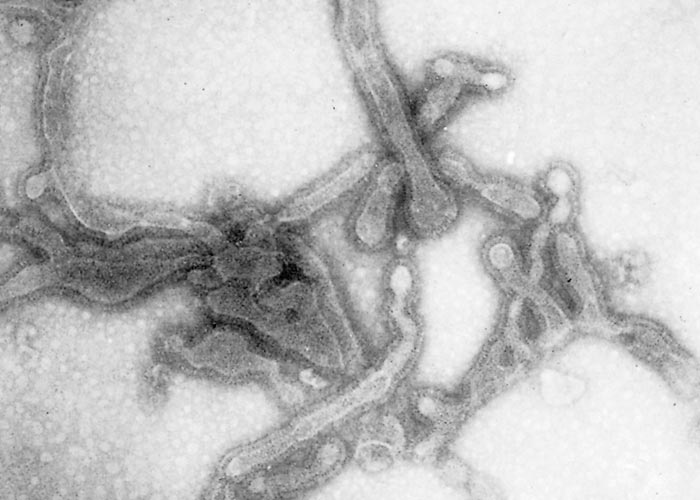
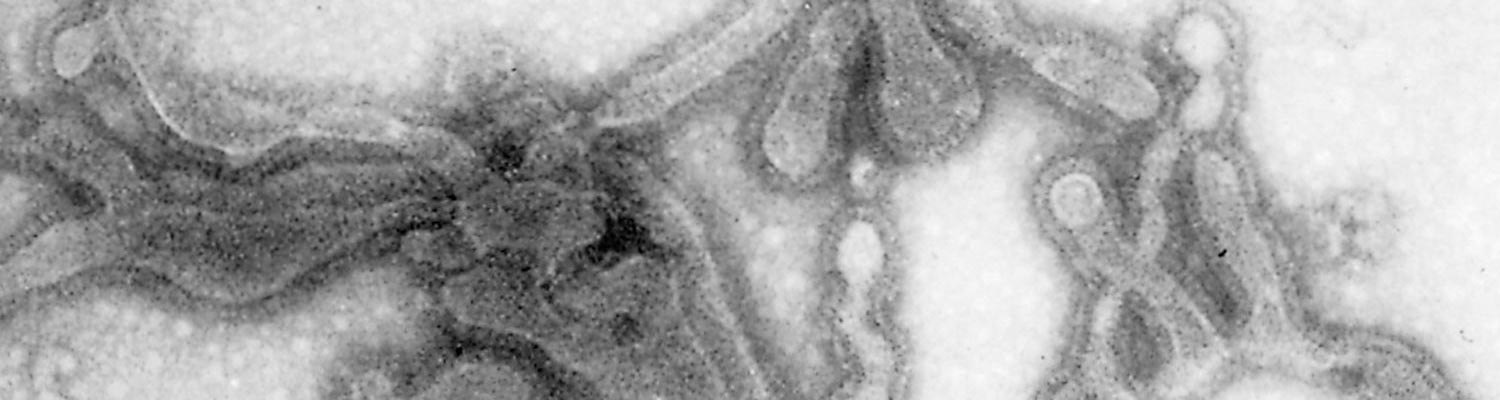
The virus is classified as a member of the genus Pneumovirus in the family Paramyxoviridae. Virions are pleomorphic, enveloped and measure 80 to 500 nm in diameter for the round and pleomorphic forms (Figure 1), and up to 5 microns in length for filamentous forms (Figure 2). The elongated nucleocapsid is helically coiled and tortuous and contains single-stranded RNA.
The envelope contains transmembrane surface glyoproteins, but, in contrast to ortho- and paramyxoviruses, pneumoviruses lack neuraminidase and haemagglutinin. Virions mature by budding from the cytoplasmic membrane.
A wide range of cell cultures are susceptible to infection with BRSV, including those prepared from most bovine tissue types, as well as kidney cells of pigs, hamsters, monkeys and humans.38 Cytopathic effects are characterized by multinucleate syncytial cell formation, intracytoplasmic inclusions and necrosis.
Bovine respiratory syncytial virus is antigenically related to human respiratory syncytial virus (HRSV),40 but the two viruses are distinct.48 It has been proposed that ruminant RSVs can be divided into 2 subgroups, BRSV and ovine RSV, with caprine RSV more closely related to BRSV.20
The genome of respiratory syncytial viruses is approximately 15.2 kb and is transcribed into 10 major subgenomic mRNAs encoding 11 proteins, because M2 mRNA contains two overlapping reading frames, which encode 2 proteins, M2-1 and M2-2.16
Epidemiology
Cattle are probably the principal reservoirs of the virus. Sheep and goats may also be infected but their role in the epidemiology is not clear, and inapparent infections are common in these species.2, 15, 19, 20, 32, 37, 43
Since the initial isolation of BRSV in Switzerland in 1970, the virus has been detected worldwide in cattle herds, with rates of infection impacted by multiple factors. In some countries, it has been estimated that frequency of BRSV exposure exceeds 50 per cent in some dairy and beef herds.28 Respiratory disease due to BRSV is most severe in calves less than 6 months of age, and infection can occur in the presence of maternal antibody. In some geographic areas, by 12 months of age, exposure of calves to BRSV may exceed 70 per cent.58 Reinfection in calves is common, likely as a result of reintroduction of BRSV into herds.36 Spread of the infection may result from direct contact with or aerosol droplets from infected animals, or indirectly from contaminated surfaces.29 Respiratory disease in cattle in the northern hemisphere caused by BRSV occurs mainly in late autumn, winter and early spring, although outbreaks have also been reported in the summer. In addition to a role for fluctuations in temperature, risk of BRSV infections can be impacted by stresses associated with weaning, transport, handling, crowding, and mixing of cattle from different sources.
Pathogenesis
The severity of clinical disease caused by BRSV is dependent on the age and immune status of the calf, the route of infection, and the strain of the virus.8 Some strains are significant primary pathogens of young cattle because, acting alone, they are capable of causing severe damage to the lower respiratory tract.12, 39, 45 However, the virus more frequently predisposes the respiratory tract of cattle to the pathogenic effects of secondary bacterial infections.6
The virus replicates in epithelial cells of the nasal cavity, pharynx, trachea, bronchi and bronchioli, and in type II pneumocytes and alveolar macrophages. It induces cytopathic changes characterized by loss of cilia and/or necrosis of bronchial and bronchiolar epithelial cells.11 The virus also alters opsonization and phagocytosis by alveolar macrophages.38, 56 Reduced mucociliary clearance resulting from a loss of cilia is responsible for the accumulation of fluid and tissue debris in the airways and alveoli, which provides an ideal environment for the growth of bacterial opportunists.
Studies conducted over the last two decades have enhanced our knowledge of the mechanisms whereby BSRV is able to establish respiratory tract infection and induce inflammation and subsequent respiratory disease. In particular, roles have been shown for non-structural proteins NS1 and NS2, as well as surface-expressed fusion and attachment glycoproteins.59 For example, NS1 and NS2 are known to regulate host type I interferons and calves infected with BRSV NS deletion mutants had no microscopic and macroscopic lung lesions.59 Likewise, it was shown that calves challenged with BRSV fusion protein deletion mutants exhibited significantly reduced pulmonary inflammation.
Clinical signs
Although most infections are subclinical, some infected animals show mild to severe disease13, 23 after an incubation period of five to seven days. In naturally infected dairy calves, early clinical signs are characterized by fever and coughing with nasal, oral and sometimes ocular discharges followed within two to three days by rapid abdominal breathing.26 Severely affected calves may show signs of respiratory distress, such as breathing through an open mouth and forced grunting expiration.60 The clinical signs that result from experimental BRSV infection mirror those observed during natural outbreaks. Upper respiratory tract signs such as nasal discharge and coughing develop on days three to five after infection, progressing to severe lower respiratory tract signs on days seven to nine after infection, including fever, anorexia, dyspnoea and hypoxia.5, 22, 55, 64 Animals with subclinical infections may have some reduction in feed and water intake, with subtle reductions in weight gain; however, severe disease can lead to rapid weight loss and dehydration. Animals that survive severe disease lag in weight gain and growth, and are susceptible to secondary bacterial infections.44
Pathology
In calves that die from disease, irregular areas of atelectasis and pneumonia, particularly of the cranioventral parts of the lungs, together with lung oedema and interstitial emphysema are usually evident. A mucopurulent exudate may be present in the bronchi of atelectatic and pneumonic areas. The interstitial emphysema may affect all the lobes of the lungs but is often more pronounced in the caudal lobes. In severe cases, emphysematous lesions may develop subpleurally, resulting in pneumothorax, pneumomediastinum or pneumopericardium.9, 33 Subcutaneous emphysematous bulla formation may be prominent. Bronchial and mediastinal lymph nodes are enlarged and oedematous.6, 41
Microscopic changes associated with BRSV infection are characterized by an acute necrotizing bronchiolitis with the development in many bronchioli of syncytial cells formed by the proliferation of epithelial cells, some of which may contain eosinophilic intracytoplasmic inclusion bodies. The lumens of the bronchioli contain neutrophils, plasma cells, macrophages, mucus and cellular debris.
Evidence of atelectasis and pneumonia is present, the exudate in alveoli containing neutrophils, macrophages and cellular debris. Epithelialization may be evident in some alveoli, even with a tendency to form syncytia, some of which may contain intracytoplasmic inclusion bodies.12, 39, 60
Microscopically, tracheobronchial lymph nodes have enlarged cortical areas with prominent follicles, expanded parafollicular areas, and enlarged medullary cords due to lymphocytic hyperplasia. The medullary sinuses commonly contain macrophages, variable numbers of lymphocytes and plasma cells, and sporadic neutrophils and eosinophils.44
As viral infection progresses, an attempt to repair necrotic airway epithelium may result in epithelial hyperplasia and bronchiolitis obliterans.14 Persistent fibrinous and exudative inflammation is followed by fibroblast infiltration and neovascularization in an attempt at healing. Fibrous polyps covered by respiratory epithelium may extend into the airway lumen, often resulting in permanent decreases in airflow and alveolar ventilation.14, 44
Diagnosis
Since respiratory infections in cattle cannot be differentiated clinically, a diagnosis of disease caused by BRSV infection can only be made with laboratory support including post mortem examination, histopathology, immunohistochemistry, real-time reverse transcription PCR (RT-PCR) and occasionally virus isolation.44
The gold standard for BRSV diagnosis is RT-PCR. It is rapid, specific and highly sensitive compared to other techniques. A commercial RT-PCR kit detects all BRSV strains assessed with 99.3 per cent efficiency and a detection limit of 0.1 TCID50.54 Several multiplex RT-PCR assays have also been developed that can detect multiple viral and bacterial pathogens that are commonly associated with bovine respiratory disease, including BRSV.35, 50, 52 For ante mortem testing, tracheal washes and bronchoalveolar lavage samples offer the highest sensitivity, although guarded nasopharyngeal swabs are also acceptable for detecting BRSV.18 Nasal swabs are the least sensitive sample for BRSV diagnosis. Intranasal vaccination with modified live viruses can result in BRSV detection by RT-PCR, underlining the importance of case history when interpreting diagnostic test results.27, 53
Antigen detection tests, such as fluorescent antibody and immunoperoxidase-staining techniques applied to cells recovered either from the nasal passages or tissue sections, are commonly used for the detection of the virus.30, 34, 47, 51, 62, 63 Due to challenges with endogenous (background) fluorescence, immunofluorescence stains are less commonly used and often more difficult to interpret.7, 44 Immunohistochemistry on formalin-fixed tissues is generally more successful, particularly when assessing tissues that have undergone some degree of autolysis during transport to the laboratory. 1-step enzyme-linked immunosorbent assays are generally more sensitive than traditional antigen-capture enzyme immunoassays.42, 44 Because of the close antigenic similarity between bovine and human respiratory syncytial viruses, a rapid, patient-side strip test designed for detection of human RSV has been shown to be effective for detecting BRSV in nasal and tracheal swabs taken from cattle.49, 57
Bovine respiratory syncytial virus is labile and difficult to recover by the use of cell cultures.12, 31, 48 For this reason, virus isolation is less commonly used. When attempting virus isolation, it is imperative to use a sucrose-containing transport medium to preserve the virus if any delay during transport is anticipated, and to inoculate cell cultures as soon as possible after the specimens have been collected.1 Tracheal washes and bronchoalveolar lavage yield cells from deeper parts of the respiratory tract than do nasopharyngeal swabs,4 and therefore offer the possibility of more efficient detection of BRSV.
Retrospective detection of BRSV infections can be accomplished by antibody detection using indirect immunofluorescence, virus neutralization and complement fixation tests. However, since most infections are inapparent, and vaccination programmes are widely used, care should be exercised when attributing respiratory disease in cattle to BRSV based on serology.
Control
Prevention of BRSV infection is the optimal approach for controlling the disease in the individual and at the herd level. Multiple modified-live and killed vaccines are commercially available generally marketed as multivalent products that also target other common pathogens of the respiratory disease complex. Given the susceptibility of young calves to BRSV infection, use of a maternal vaccination programme and attention to colostrum intake are highly encouraged. A modified-live, multivalent vaccine containing BRSV, BHV-1 and PI3 has been approved for intranasal administration and has shown some efficacy against BRSV infection in calves as young as three days of age.17, 21 However, protection induced by intranasal vaccination appears to wane within a few months22 and should be followed by a second dose around 60 days later.17
Treatment of sick animals should be based on early detection of disease and focused on supportive care, including administration of NSAIDS and correction of dehydration.10 Antibiotics are commonly administered to limit secondary bacterial infections. There are few data available to support the use of antihistamines or corticosteroids, although this approach has been suggested for animals demonstrating severe respiratory signs associated with BRSV infection.6 Some clinicians believe that a change of feed or a 48-hour fast is an important part of the early treatment of acute respiratory distress in calves.26 Most animals will recover in several days without treatment.
References
- BAKER, J. C., 1986. What to do about bovine RSV. Norden News. 4–14.
- BAKER, J. C., AMES, T. R. & MARKHAM, R. J. F., 1985. Serologic studies of bovine respiratory syncytial virus in Minnesota cattle. American Journal of Veterinary Research, 46, 891–892.
- BAKER, J. C., ELLIS, J. A. & CLARK, E. G., 1997. Bovine respiratory syncytial virus. Veterinary Clinics of North America: Food Animal Practice, 13, 425-454.
- BATEMAN, E. D. & AINSLIE, G. M., 1990. Broncho-alveolar lavage—a window to the lung. South African Medical Journal, 77, 550–552.
- BLODÖRN, K., HÄGGLUND, S., GAVIER-WIDEN, D., ELEOUET, J. F., RIFFAULT, S., PRINGLE, J., TAYLOR, G. & VALARCHER, J. F., 2015. A bovine respiratory syncytial virus model with high clinical expression in calves with specific passive immunity. BMC Veterinary Research, 11, 76.
- BOHLENDER, R. E., MCCUNE, M. W. & FREY, M. L., 1982. Bovine respiratory syncytial virus infection. Modern Veterinary Practice, 8, 613-618.
- BRODERSEN, B. W., 2010. Bovine respiratory syncytial virus. Veterinary Clinics of North America: Food Animal Practice, 26, 323-333.
- BRYSON, D. E., MCNULTY, M. S., ALLAN, G. M., CUSH, P. F. & LOGAN, E. F., 1982. Experimental respiratory syncytial virus pneumonia in calves. Proceedings of the Twelfth World Congress on Diseases of Cattle. Amsterdam.
- BRYSON, D. G., 1993. Necropsy findings associated with BRSV pneumonia. Veterinary Medicine, 88, 894–899.
- CAMPBELL, J., 2016. Viral Respiratory Tract Infections in Cattle. In: AIELLO SE, MOSES, M. A. & ALLEN, D. G. (eds.), The Merck Veterinary Manual, 11th edition, Kenilworth, New Jersey, USA: Merck & Co., Inc.
- CASTLEMAN, W. L., CHANDLER, S. K. & SLAUSON, D. O., 1985. Experimental bovine respiratory syncytial virus infection in conventional calves: Ultrastructural respiratory lesions. American Journal of Veterinary Research, 46, 554–560.
- CASTLEMAN, W. L., LAY, J. C., DUBOVI, E. J. & SLAUSON, D. O., 1985. Experimental bovine respiratory syncytial virus infection in conventional calves: Light microscopic lesions, microbiology, and studies on lavaged lung cells. Journal of Veterinary Research, 46, 547–553.
- CASTLEMAN, W. L., TORRES-MEDINA, A., HAWKINS, K. L., DUBOVI, E. J. & ATZ, J. M., 1985. Severe respiratory disease in dairy cattle in New York state associated with bovine respiratory syncytial virus infection. Cornell Veterinarian, 75, 473–483.
- CASWELL, J. L. & WILLIAMS, K. J., 2008. Respiratory System. In: MAXIE, M.G., (eds.). Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals, 5th edition, Edinburgh, Scotland: Saunders, Ltd.
- COLLINS, J. K., TEEGARDEN, R. M., MACVEAN, D. W., SMITH, G. H. & FRANK, G. R., 1988. Prevalence and specificity of antibodies to bovine respiratory syncytial virus in sera from feedlot and range cattle. American Journal of Veterinary Research, 49, 1316–1319.
- COLLINS, P. L., DICKENS, L. E., BUCKLER-WHITE, A., OLMSTED, R. A., SPRIGGS, M. K., CAMARGO, E. & COELINGH, K. V., 1986. Nucleotide sequences for the gene junctions of human respiratory syncytial virus reveal distinctive features of intergenic structure and gene order. Proceedings of the National Academy of Sciences of the United States of America. 83, 4594-4598.
- CORTESE, V. S., SEEGER, J. T., TREJO, C., SHORT, T. H. & ELLIS, J. A., 2018. The Impact of Neonatal Intranasal Vaccination for Bovine Respiratory Syncytial Virus on Subsequent Vaccination and Responses to Natural Exposure. JSM Allergy Asthma,, 3, 1021.
- DOYLE, D., CREDILLE, B., LEHENBAUER, T. W., BERGHAUS, R., ALY, S. S., CHAMPAGNE, J., BLANCHARD, P., CORSSLEY, B., BERGHAUS, L., COCHRAN, S. & WOOLUMS, A. R., 2017. Agreement Among 4 Sampling Methods to Identify Respiratory Pathogens in Dairy Calves with Acute Bovine Respiratory Disease. Journal of Veterinary Internal Medicine, 31, 954-959.
- DUNBAR, M. R., JESSUP, D. A., EVERMANN, J. F. & FOREYT, W. J., 1985. Seroprevalence of respiratory syncytial virus in free-ranging Bighorn sheep. Journal of the American Veterinary Medical Association, 187, 1173–1174.
- ELERAKY, N. Z., KANIA, S. A., EVERMANN, F. F. & POTGIETER, L. N. D., 2003. Comparison of targeting F and G protein genes to detect bovine and ovine respiratory syncytial viruses. Journal of Veterinary Diagnostic Investigation, 15, 277-280.
- ELLIS, J. A., GOW, S. P. & GOJI, N., 2010. Response to experimentally induced infection with bovine respiratory syncytial virus following intranasal vaccination of seropositive and seronegative calves. Journal of the Veterinary Medical Association, 236, 991-999.
- ELLIS, J. A., GOW, S. P., MAHAN, S. & LEYH, R., 2013. Duration of immunity to experimental infection with bovine respiratory syncytial virus following intranasal vaccination of young passively immune calves. Journal of the American Veterinary Medical Association, 243, 1602-1608.
- ELVANDER, M., ALENIUS, S. & JACOBSSON, S. O., 1991. Severe outbreaks of respiratory disease in dairy herds caused by bovine respiratory syncytial virus. Bovine Practitioner, 26, 166-168.
- EUGESTER, A. K., 1996. Foal pneumonia: The possible involvement of a respiratory syncytial virus. Texas Veterinarian, August 1996. 19-21.
- EVERMANN, J. F., LIGGIT, H. D. & PARISH, S. M., 1985. Properties of a respiratory syncytial virus isolated from a sheep with rhinitis. American Journal of Veterinary Research, 46, 947–951.
- FREY, M. L., 1983. Bovine respiratory syncytial virus and acute respiratory distress syndrome in cattle. Proceedings of the Eighteenth Conference of the American Association of Bovine Practitioners, Oklahoma City.
- FULTON, R. W., D’OFFAY, J. M., LANDIS, C., MILES, D. G., SMITH, R. A., SALIKI, J. T., RIDPATH, J. F., CONFER, A. W., NEILL, J. D., EBERLE, R., CLEMENT, T. F., CHASE, C. C. L., BURGE, L. J. & PAYTON, M. E., 2016. Detection and characterization of viruses as field and vaccine strains in feedlot cattle with bovine respiratory disease. Vaccine, 34, 3478-3492.
- GERSHWIN, L. J., 2007. Bovine respiratory syncytial virus infection: immunopathogenic mechanisms. Animal Health Research Reviews, 8, 207-213.
- HAGGLAND, S., SVENSSON, C., EMANUELSON, U., VALARCHER, J. F. & ALENIUS, S., 2006. Dynamics of virus infections involved in the bovine respiratory disease complex in Swedish dairy herds. Veterinary Journal, 172, 320-328.
- HAINES, D. M., CLARK, E. G. & CHELACK, B. J., 1989. The detection of bovine respiratory syncytial virus in formalin fixed bovine lung with commercially available monoclonal antibodies and avidin biotin complex immunohistochemistry. Canadian Journal of Veterinary Research, 53, 366–368.
- JACOBS, J. W. & EDINGTON, N., 1971. Isolation of respiratory syncytial virus from cattle in Britain. Veterinary Record, 88, 694.
- KEY, D. W. & DERBYSHIRE, J. B., 1984. Serological studies of parainfluenza type 3 virus, bovine adenovirus type 3 and bovine respiratory syncytial virus infection in beef calves. Veterinary Microbiology, 9, 587–592.
- KIMMAN, T. G., STRAVER, P. J. & ZIMMER, G. M., 1989. Pathogenesis of naturally acquired bovine respiratory syncytial virus infection in calves: morphologic and serologic findings. American Journal of Veterinary Research, 50, 684–693.
- KIMMAN, T. G., ZIMMER, G. M., STRAVER, P. J. & DE LEEUW, P. W., 1986. Diagnosis of bovine respiratory syncytial virus infections inproved by virus detection in lung lavage samples. American Journal of Veterinary Research, 47, 143-147.
- KISHIMOTO, M., TSUCHIAKA, S., RAHPAYA, S. S., HASEBE, A., OTSU, K., SUGIMURA, S., KOBAYASHI, S., KOMATSU, N., NAGAI, M., OMATSU, T., NAOI, Y., SANO, K., OKAZAKI-TERASHIMA, S., OBA, M., KATAYAMA, Y. & SATO, R., 2017. Development of a one-run real-time PCR detection system for pathogens associated with bovine respiratory disease complex. Journal of Veterinary Science, 79(3), 517-523.
- LARSEN, L. E., TJORNEHOJ, K. & VIUFF, B., 2000. Extensive sequence divergence among bovine respiratory syncytial viruses during recurrent outbreaks in closed herds. Journal of Clinical Microbiology, 38, 4222-4227.
- LEHMKUHL, H. D. & GOUGH, P. M., 1977. Investigation of causative agents of bovine respiratory tract disease in a beef cow-calf herd with an early weaning program. American Journal of Veterinary Research, 38, 1717– 1720.
- MATUMOTO, M., INABA, Y., KUROGI, H., SATO, K., OMORI, T., GOTO, Y. & HIROSE, O., 1974. Bovine respiratory syncytial virus: Host range in laboratory animals and cell cultures. Archiv für die Gesamte Virusforschung, 44, 280-290.
- MOHANTY, S. B., INGLING, A. L. & LILLIE, M. G., 1975. Experimentally induced respiratory syncytial virus infection in calves. American Journal of Veterinary Research, 4, 417-419.
- PACCAUD, M. F. & JACQUIER, C., 1970. A respiratory syncytial virus of bovine origin. Archiv für die Gesamte Virusforschung, 30, 327–342.
- PIRIE, H. M., PETRIE, L., PRINGLE, C. R., ALLAN, E. M. & KENNEDY, G. J., 1981. Acute fatal pneumonia in calves due to respiratory syncytial virus. Veterinary Record, 108, 411-416.
- QUINTING, B., ROBERT, B., LETELLIER, C., BOXUS, M., KERKHOFS, P., SCHYNTS, F. & COLLARD, A., 2007. Development of a 1-Step Enzyme-Linked Immunosorbent Assay for the Rapid Diagnosis of Bovine Respiratory Syncytial Virus in Postmortem Specimens. Journal of Veterinary Diagnostic Investigation, 19, 238–243.
- ROSSI, C. R. & KIESEL, G. K., 1974. Serological evidence for the association of bovine respiratory syncytial virus with respiratory tract disease in Alabama cattle. Infection and Immunity, 10, 293–298.
- SACCO, R. E., MCGILL, J. L., PILLATZKI, A. E., PALMER, M. V. & ACKERMANN, M. R., 2014. Respiratory Syncytial Virus Infection in Cattle. Veterinary Pathology, 51, 427–436.
- SHARMA, R. & WOLDEHIWET, Z., 1990. Pathogenesis of bovine respiratory syncytial virus in experimentally infected lambs. Veterinary Microbiology, 23, 267–272.
- SHARMA, R. & WOLDEHIWET, Z., 1991. Bovine respiratory syncytial virus: A review. Veterinary Bulletin, 61, 1117–1131.
- SMITH, G. H., COLLINS, J. K., SANDERS, J. & ANDERSON, G., 1987. Increased sensitivity of diagnosis of infectious bovine rhinotracheitis virus and bovine respiratory syncytial virus infection using immunoperoxidase on fixed tissue. Agri-practice, April 1987. 27-30.
- SMITH, M. H., FREY, M. L. & DIERKS, R. E., 1975. Isolation, characterization and pathogenicity studies of a bovine respiratory syncytial virus. Archives of Virology, 47, 237–247.
- SOCHA, W. & ROLA, J., 2012. Use of rapid human respiratory syncytial virus strip tests for detection of bovine respiratory syncytial virus in experimentally vaccinated calves. Polish Journal of Veterinary Sciences, 15, 629-634.
- THANTHRIGE-DON, N., LUNG, O. O., FURUKAWA-STOFFER, T., BUCHANAN, C., JOSEPH, T., GODSON, D. L., GILLEARD, J., ALEXANDER, T. & AMBAGALA, A. P., 2018. A novel multiplex PCR-electronic microarray assay for rapid and simultaneous detection of bovine respiratory and enteric pathogens. Journal of Virological Methods, 261, 51-62.
- THOMAS, L. H. & STOTT, E. J., 1981. Diagnosis of respiratory syncytial virus infection in the bovine respiratory tract by immunofluorescence. The Veterinary Record, 108, 432–435.
- THONUR, L., MALEY, M., GILRAY, J., CROOK, T., LAMING, E., TURNBULL, D., NATH, M. & WILLOUGHBY, K., 2012. One-step multiplex real time RT-PCR for the detection of bovine respiratory syncytial virus, bovine herpesvirus 1 and bovine parainfluenza virus 3. BMC Veterinary Research, 8, 37.
- TIMSIT, E., LE DREAN, E., MAINGOURD, C., BELLOC, C., GUATTEO, R., BAREILLE, N., SEEGERS, H., DOUART, A., SELLAL, E. & ASSIE, S., 2009. Detection by real-time RT-PCR of a bovine respiratory syncytial virus vaccine in calves vaccinated intranasally. The Veterinary Record, 165, 230-233.
- TIMSIT, E., MAINGOURD, C., LE DREAN, E., BELLOC, C., SEEGERS, H., DOUART, A. & ASSIE, S., 2010. Evaluation of a commercial real-time reverse transcription polymerase chain reaction kit for the diagnosis of Bovine respiratory syncytial virus infection. Journal of Veterinary Diagnostic Investigation, 22, 238-241.
- TJORNEHOJ, K., UTTENTHAL, A., VIUFF, B., LARSEN, L. E., RONTVED, C. & RONSHOLT, L., 2003. An experimental infection model for reproduction of calf pneumonia with bovine respiratory syncytial virus (BRSV) based on one combined exposure of calves. Research in Veterinary Science, 74, 55-65.
- TOTH, T. & HESSE, R. A., 1983. Replication of five bovine respiratory viruses in cultured bovine alveolar macrophages. Archives of Virology, 75, 219–224.
- URBAN‐SCHMIEL, R., WERNICKI, A., GROOMS, D. L., BARBU, N. I., ROLA, J. & SOCHA, W., 2015. Rapid Detection of Bovine Respiratory Syncytial Virus in Poland Using a Human Patient‐Side Diagnostic Assay. Transboundary and Emerging Diseases, 62, 407-410.
- VALARCHER, J. F., SCHELCHER, F. & BOURHY, H., 2000. Evolution of bovine respiratory syncytial virus. Journal of Virology, 74, 10714-10728.
- VALARCHER, J. F. & TAYLOR, G., 2007. Bovine respiratory syncytial virus infection. Veterinary Research, 38, 153-180.
- VAN DEN INGH, T. S. G. A. M., VERHOEFF, J. & VAN NIEUWSTADT, A. P. K. M. I., 1982. Clinical and pathological observations on spontaneous bovine respiratory syncytial virus infections in calves. Research in Veterinary Science, 33, 152–158.
- VAN VUUREN, M., 1990. Serological studies of bovine respiratory syncytial virus in feedlot cattle in South Africa. Journal of the South African Veterinary Association, 61, 168–169.
- WATT, N. J., PIRIE, H. M., ALLAN, E. M., SELMAN, I. E., PETRIE, L., GIBBS, H. A. & WISEMAN, A., 1982. Parainfluenza type III virus and respiratory syncytial virus antigens in paraffin-embedded sections of pneumonic lung. Proceedings of the Twelfth World Congress on Diseases of Cattle. Amsterdam.
- WELLEMANS, G., 1977. Laboratory diagnosis methods for bovine respiratory syncytial virus. Veterinary Science Communications, 1, 179–189.
- WOOLUMS, A. R., ANDERSON, M. L., GUNTHER, R. A., SCHELEGLE, E. S., LAROCHELLE, D. R., SINGER, R. S., BOYLE, G. A., FRIEBERTSHAUSER, K. E. & GERSHWIN, L. J., 1999. Evaluation of severe disease induced by aerosol inoculation of calves with bovine respiratory syncytial virus. American Journal of Veterinary Research, 60, 473–480.