- Infectious Diseases of Livestock
- Part 2
- Visna-maedi
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Visna-maedi
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). B Murphy, D Knowles and M Highland, Visna-maedi, 2018.
Visna-maedi
Previous authors: D W VERWOERD AND R C TUSTIN
Current authors:
D P KNOWLES - Research Leader, DVM, PhD, DACVP, Department of Veterinary Microbiology and Pathology, Washington State University, 405 Bustad Hall, Pullman, Washington, 99164, USA
B G MURPHY - Associate Professor, DVM, PhD, Dip ACVP, VetMed PMI, 4206 VetMed, 3A One Shields Ave, Davis, California, USA
M A HIGHLAND - Veterinary Medical Officer - Researcher/Scientist, DVM, PhD, Diplomate ACVP, USDA-ARS-ADRU, Washington State University, 405 Bustad Hall, Pullman, Washington, 99164, USA
Introduction
Visna-maedi, one of the so-called slow viral diseases of sheep, is caused by a non-oncogenic retrovirus of the Lentivirus genus. The Icelandic name denotes the two most common forms of the disease, maedi referring to the respiratory form of the disease (“breathlessness”, chronic interstitial pneumonia) and visna referring to the neurologic form of the disease (“wasting”, demyelinating leukoencephalomyelitis). Two other disease syndromes, arthritis and chronic mastitis, are more rarely seen. The disease is also known as ovine progressive pneumonia or Montana sheep disease in the USA, zwoegersiekte in the Netherlands, la bouhite in France, and Graaff-Reinet disease in South Africa.15, 17
It was first described in 1915 in South Africa by Mitchell,38 who regarded the condition as an aberrant form of jaagsiekte (see Jaagsiekte), followed by reports of disease in the state of Montana (USA).11, 36 The confusion with pulmonary adenomatosis (jaagsiekte) caused by Mitchell’s report was resolved by De Kock,20 who was the first to realize that they were two distinct diseases, often coexisting in the same animal. Chronic pneumonia, named Graaff-Reinet disease after the town in which the experimental station was located, and where the diseased animals were located.
In Iceland, the introduction of visna-maedi virus (VMV) was traced to the importation in 1933 of Karakul rams from Germany, one of which carried both visna-maedi and jaagsiekte viruses.44 Sigurdson and collaborators first isolated VMV in 1960. Most of the early work on visna-maedi was done in Iceland, where it was initially considered to be two separate diseases.49
Aetiology
Visna-maedi virus is closely related to the caprine arthritis encephalitis virus (CAEV) of goats (see Caprine arthritis-encephalitis). Both diseases have been reviewed elsewhere.4, 8, 37, 42, 45, 48, 49 Visna-maedi virus and CAEV are genetically and pathogenically closely related. These viruses share a predictable tropism for specific anatomic locations including joint synovium, tissues of the central nervous system (brain and spinal cord), lung, and mammary gland. Visna-maedi virus and CAEV also share a common tropism for histiocytic cells (monocytes, macrophages and dendritic cells) and do not infect CD4 T cells as is the case with the immunosuppressive lentiviruses such as the human immunodeficiency viruses. Well- documented cross-species transmission of CAEV and VMV between sheep and goats has resulted in a collective designation of these viruses as small ruminant lentiviruses (SRLV).37 Although capable of cross-infecting their mammalian hosts and genetically recombining, VMV and CAEV have a predisposition for either sheep or goats, respectively.
Mature virions are 80 to 100 nm in diameter. Each virus particle contains two copies of the 8.4 to 9.2 kb long single stranded RNA genome. As is the case in all members of the Retroviridae, VMV replicates via a proviral DNA intermediate generated from the viral RNA genome through the viral reverse transcriptase enzyme. The genome contains three well conserved genes: the env gene that encodes the envelope glycoprotein, the gag gene coding for the three core proteins surrounding the viral nucleic acid, the pol gene that codes for the viral RNA-dependent DNA polymerase (reverse transcriptase) and several other viral enzymes. The structural proteins of SRLV include the surface (SU) and transmembrane (TM) proteins (both encoded by env) and CA, MA and NC (encoded by gag). In addition to the gag-pol-env genes present in all retroviruses, VMV has three additional open reading frames (vif, vpr-like and rev) which play a role in regulating viral replication.8, 49 The vpr-like gene was formerly referred to as tat, but is now thought to be functionally analogous to the vpr gene of the immunodeficiency viruses.
Epidemiology
With the exception of Australasia, ovine lentiviruses have been isolated from all the major sheep-producing countries, and are often found with jaagsiekte (sheep pulmonary adenomatosis). They also occur in areas with a very low prevalence of jaagsiekte, such as the USA,12 and were responsible for large-scale mortalities in Iceland after the eradication of jaagsiekte.44 Studies of concurrent infections with the viruses causing visna-maedi and jaagsiekte reveal an increase in the seroprevalence of VMV antibodies in flocks affected by jaagsiekte. This effect was independent of other factors such as breed of sheep or size of flock. It was concluded that jaagsiekte plays a role in the replication and/or spread of VMV, although a synergistic effect of the simultaneous infections on the expression of concurrent lesions does not seem to occur.18, 29, 50
Differences in breed susceptibility to SRLV have not been found in South Africa, but have been reported elsewhere.17, 60
Horizontal transmission of the disease occurs under conditions of close contact. Droplet infection via the respiratory route has therefore been implicated, especially between adult animals.44 As is true of CAEV, transmission from ewe to lamb has been shown to occur mainly through the colostrum,19 presumably by means of infected monocytes or macrophages.41 Although not shown, transmission by bloodsucking insects could also play a role. Transplacental transmission is possible but rare.7 Semen has not been implicated as a source of infection nor has any evidence been published of indirect transmission by excretions or fomites.
The virus is able to persist latently within the host cell genome in the presence of high antibody titres, and infected animals are therefore carriers for life. Transcriptionally silent latent virus, in the form of integrated provirus (DNA form), may be activated at any time, providing stimulation for the immune system and an explanation for the slow development of the disease. Antigenic drift has been proposed as a mechanism for the evasion of the immune response.30, 40 Most infected sheep in fact show little sign of disease, but remain carriers and can transmit the infection to others. No sex or age predilection has been found in the susceptibility of animals to infection.
Pathogenesis
Both natural and experimental infections of sheep with VMV give rise to systemic multiorgan disease. Visna-maedi virus has an anatomic tropism and predisposition for infecting the tissues of the lung and central nervous system. The primary target cells for VMV are monocytes and macrophages,41 but a variety of cells are permissive for infection. These additional targets include epithelial cells in the mammary gland and endothelial cells and microglial cells of the CNS. However, it is difficult to establish whether detection of viral antigen or nucleic acid indicates infection of or uptake of viral products by these cells.4 Unlike the immunodeficiency-causing lentiviruses [the human immunodeficiency virus (HIV), simian immunodeficiency virus (SIV), feline immunodeficiency virus (FIV)], VMV does not replicate in CD4 T cells. Antigenic variation in viral strains and differences in tissue tropism in sheep breeds may play a role in determining cell tropism. An in vitro study demonstrated that sequence variability in the noncoding long terminal repeat (LTR) affected the cell tropism of the virus.1 The cellular receptor of VMV has yet to be identified.
Due to low fidelity of viral reverse transcriptase, mutations are predictably introduced during each replication cycle. In addition, recombination between VMV and CAEV can occur in dually infected cells, resulting in dramatic shifts in the viral genome. Random SRLV mutability can and does lead to the ability of the virus to evade the host immune response and ensure persistence of the infection.37 Neutralizing antibodies are usually detected two to three months after infection and consist of a different subpopulation of IgG molecules than the complement-fixation (CF) antibodies. Commonly used agar gel immunodiffusion (AGID) and ELISA tests detect antibodies to the gp135 glycoprotein and the p30 core protein of the virus. Although neutralizing antibodies serve to eliminate cell-free virus, these antibodies are unable to destroy infected cells, especially when the viral genome is latent (not expressing viral antigen). In an infected animal, many of the antiviral antibodies, perhaps the majority, are non-neutralizing.
There is strong evidence that the pathogenesis of the lesions of SRLV is immunologically mediated.4 Non-neutralizing antibodies may serve to augment viral pathogenesis and function as enhancing antibodies. Macrophages and dendritic cells have Fc receptors on their plasma membranes; virus-antibody complex internalization and cellular infection may be facilitated through the binding of these surface receptors. As a result, SRLVs take advantage of the immune system to aid their replication.4 Immunosuppression of sheep prior to experimental infection with VMV severely limits the development of lesions in the CNS, and conversely, more severe lesions are found in immunostimulated animals. Though less clear in the case of maedi, pulmonary lesions also seem to be caused by a cell-mediated immune response (CMI) against virus-specific antigens produced by persistently infected cells.
Both the humoral and cellular immune responses to VMV infection, as well as the role played by the various cell components in the immunopathology, have been studied in great detail. After infection, there is a restriction of viral replication in circulating monocytes, at least partially due to a transcriptional block.8, 48, 49 Upon maturation of monocytes into macrophages in vitro and in selected populations of tissue macrophages, the restriction is lifted, and a low level of expression of viral antigens is detectable.8, 41, 48, 49 Explants from many different organs will produce cell cultures in which the virus can replicate to high titres, indicating that the latent virus is not defective.8, 49 Viral DNA levels in alveolar macrophages are significantly higher in sheep with maedi lesions in the lung, suggesting an important role for infected macrophages in the pathogenesis of the disease.33, 62 It has been shown that cultured monocyte-derived macrophages from VMV-infected sheep express low levels of major histocompatibility complex (MHC) class II antigens and efficiently present VMV antigen to antigen-specific T cell lines.32 Increased alveolar macrophage related neutrophil chemotactic activity and fibronectin by macrophages from infected lungs may also play a role in the recruitment of inflammatory cells to the lesion.10
Virus-specific cytotoxic T lymphocytes are present in efferent lymph and peripheral blood of sheep persistently infected with VMV. CD8+ mediated cytotoxicity is specific for particular strains of VMV.5 In bronchoalveolar lavage samples from naturally infected sheep, the percentage of CD8+ cells is significantly increased, while percentages of CD4+, CD5+, gamma delta+ T cells, and the ratio of CD4+/CD8+ cells are decreased.34 Within these lymphocyte shifts, only the increase in CD8+ cells showed a significant correlation with the severity of lung lesions, suggesting that CD8+ lymphocytes are key cells in the development of the interstitial lymphocytic alveolitis characteristic of maedi.34 A similar preponderance of CD8+ cells has also been demonstrated in the carpal synovium of VMV-infected sheep with clinical arthritis.3
In studying the immune response in lymph nodes after experimental infection, it was shown that there is a balanced increase of all cell types, suggesting a selective migration of CD8+ cells from the node as the cause of CD8+ preponderance in affected tissues.6 It was also found that CD8+ depletion of sheep does not affect VMV replication in the lymph nodes, suggesting that these cells are not required for the control of the primary infection.25 Conversely, CD4+ depletion reduces the levels of infected macrophages and it was shown that CD4+ T cells are required for VMV infection in macrophages but not in dendritic cells.24
Clinical signs
The onset of clinical disease is insidious, and loss of condition is usually the first clinical sign recognized in both the maedi and visna syndromes. Respiratory signs of maedi are rarely seen in animals before the age of three or four years. Dyspnoea is common, initially following exercise, but later also at rest. Static lung volumes and the transfer factor for carbon monoxide are significantly reduced.9 A dry cough may develop but nasal discharges are typically absent in uncomplicated cases. In the absence of secondary infections there is no fever, and the animal eventually dies six to twelve months after the appearance of clinical signs. Secondary bacterial pneumonia, often associated with members of the Pasteurellaceae family, is commonly the cause of death in field cases.
Clinical evidence of visna, characterized by neurological signs, is much less common, even though the incubation period is shorter than in maedi. Unsteady gait, particularly affecting the pelvic limbs, stumbling and falling, trembling of facial muscles, and inability to extend the fetlock may be early signs of the disease. Gradual progression leads to paresis and eventually to total paralysis, accompanied by loss of condition (wasting), despite the appetite being usually unaffected. Fever is absent and the animal remains alert throughout the course of the disease. The clinical course may last up to a year, but there is no recovery once clinical signs are manifested.17, 30 Other clinical signs reported include swollen joints and mastitis resulting in induration of the mammary gland.15, 59 Conflicting results were obtained in studies measuring the effect of VMV-associated mastitis on milk production and lamb growth. Statistically significant reductions in the growth rate of lambs from infected ewes were found in some cases51, 56 whereas no such differences were found in others.22, 23, 57 These discrepancies can probably be ascribed to differences in the virulence of the viral strains used and/or variations in the susceptibility of various sheep breeds.
Pathology
Although clinical signs are typically referable to disease of the CNS or lung, the lungs, regional lymph nodes, brain, spinal cord, synovial joints, and mammary glands can be affected to a greater or lesser extent, and any of these tissues may be involved in a single sheep.15 In advanced cases of maedi, the lungs do not collapse fully when the thoracic cavity is opened, and the impressions of the ribs may be retained. The lungs are enlarged, perhaps two to four times their normal weight, have a firm rubbery consistency, lack normal crepitations, and are greyish in colour. The cut surface is dry, and the pulmonary exudate typical of jaagsiekte is absent. Secondary bacterial pneumonia may be present. The bronchial and mediastinal lymph nodes are usually grossly enlarged, may be three to five times their normal size, and have a whitish homogenous appearance on cut section.15, 17, 27, 28, 30
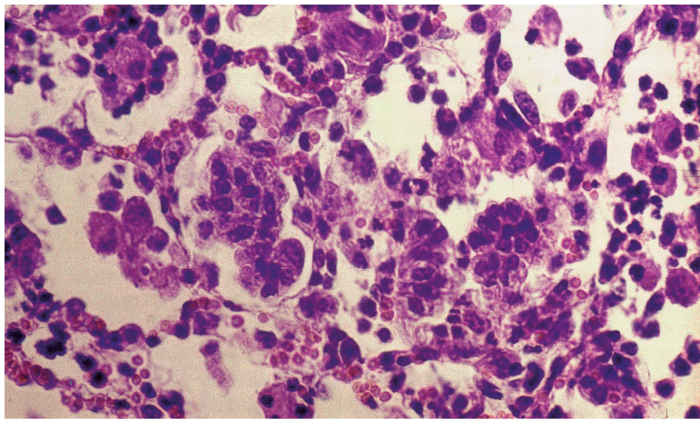
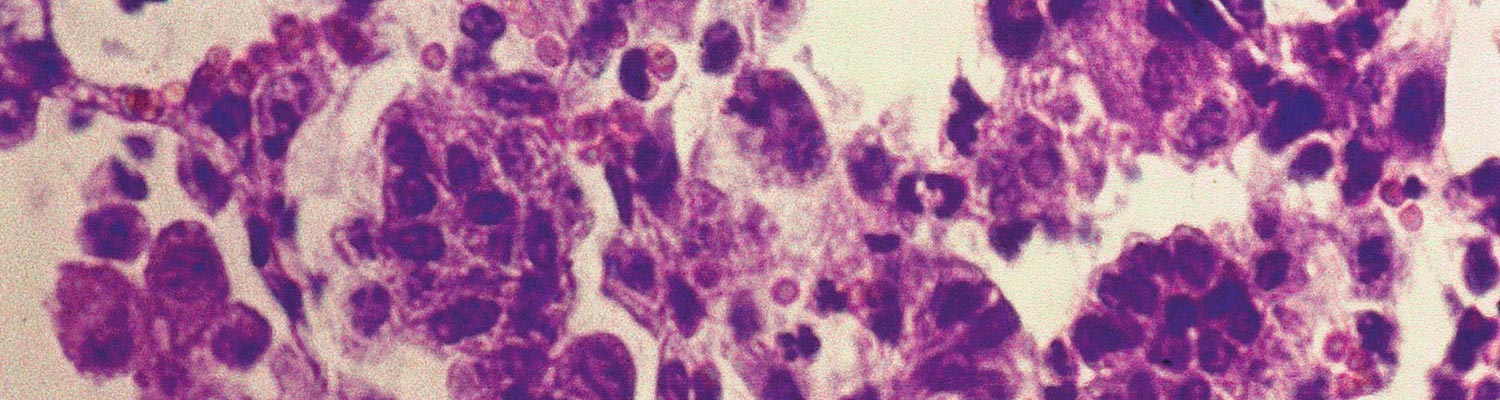
The most characteristic histopathological feature in the lungs is diffuse, often pronounced, thickening of the alveolar septa caused by infiltration of mononuclear cells, mainly lymphocytes and monocytes, and hyperplasia of smooth muscle cells (interstitial pneumonia, Figure 1). These changes may lead to complete or partial obliteration of alveoli. Fibrosis is not a prominent feature, but perivascular, peribronchial and peribronchiolar lymphoid hyperplasia, often with development of lymphoid follicles, is a consistent and conspicuous change. Hyperplasia of lining type II pneumocytes (palisading cuboidal cells), which could be confused with early jaagsiekte lesions, may be present. Intra-alveolar exudate is generally inconspicuous and consists mainly of alveolar macrophages and some cellular debris, unless secondary bacterial pneumonia is also present.27, 28
Visna has been characterized as a chronic demyelinating nonsuppurative encephalomyelitis.54 It commences in the ependyma of the ventricles of the brain and central canal of the spinal cord, and spreads to involve particularly the white matter of the brain and grey matter of the spinal cord. The lesions may be widespread but patchy in distribution. In brains taken from advanced cases, lesions are characterized by lymphocytic choroiditis with, in some places, necrosis and fragmentation of the ependyma. A lymphocytic infiltration, which may be extensive and show evidence of lymphoid follicle formation, and prominent perivascular infiltrations in the nervous tissues, composed of lymphocytes, macrophages and plasma cells, are characteristic lesions. The number of cells, chiefly lymphocytes, in the cerebrospinal fluid may increase in some animals to 1000/mm3 or more.31 Demyelination, foci of necrosis and gliosis in the white matter (that of the cerebrum and cerebellum often being most severely affected), and mild to severe lymphocytic meningitis are usually present.27, 43 In the spinal cord, lesions occur multifocally along almost its entire length but vary in intensity. There is a nonsuppurative poliomyelitis, patchy demyelination and gliosis of white matter, and a lymphocytic meningitis and choroiditis. Cellular exudate may be present in the central canal.27
When arthritis is present, it is usually the carpal, metacarpal and/or tarsal joints which are affected, but more commonly carpal.16, 63 The histological lesion is similar to that of caprine arthritis caused by CAEV, and is characterized by hyperplasia and necrosis of the synovial membrane, necrosis and erosions of articular cartilage, necrosis and fibrosis of subchondral bone, and extensive periarticular fibrosis. Some affected joints may contain a small amount of fibrin, but generally lack neutrophils. The inflammatory reaction, which is prominent, is chiefly lymphocytic.
The mastitis which some infected sheep develop is characterized by interstitial accumulations of lymphocytes, the formation of periductal lymphoid nodules, and necrosis at the site of these nodules.13, 21, 35 Vascular lesions have also been reported in some cases of VMV infection. These are most frequently associated with lesions in other organs, and are associated with intramural oedema and haemorrhage, mononuclear cell infiltration, and fibrinoid necrosis of the media of arteries. Thrombosis may be present.14
Diagnosis
A tentative diagnosis of visna-maedi can be based on clinical signs and serological results. Confirmation by means of histopathology may be necessary however, as the clinical signs are not specific and clinical disease can occur in the absence of antibodies. Due to the slowly progressive nature of the infection and disease pathogenesis, VMV can become widespread in a flock or region before clinical cases may be observed37 Viral isolation provides supportive evidence but in view of the persistent nature of the infection, a positive result may only be indicative of subclinical infection.48, 49
Detection of proviral DNA by means of the polymerase chain reaction (nested or real- time PCR) can be a sensitive diagnostic technique. In cell cultures, a positive result can be obtained by PCR 24 hours after infection, four days before viral protein can be detected.61 Proviral DNA, has also been detected in peripheral blood monocytes. Detection of viral RNA in cultured cells is possible by means of reverse-transcriptase PCR or in situ hybridization with labelled viral DNA and can be combined with immunohistochemistry to demonstrate viral antigens.53 However, viral genetic heterogeneity is a common cause of decreased PCR sensitivity.47 The sensitivity and specificity of immunohistochemical diagnosis of VMV is considerably enhanced by using a panel of monoclonal antibodies prepared against the envelope as well as core proteins of the virus.26, 52
Various serological methods have been used for the detection of SRLV antibodies, including neutralization, complement fixation, immunofluorescence, AGID, and ELISA tests.48, 49, 55 In general, serology-based assays such as ELISA have been found to be the more sensitive than PCR-based tests, except for the detection of recent infections.2, 39 Neutralizing antibodies develop late in the infection cycle and are strain-specific, and therefore not suitable for general diagnostic purposes. The AGID test using antigen containing gp135 is not strain-specific and has proven to be a simple and reliable test. Complement-fixing (CF) antibodies can be detected three to four weeks after infection, and titres tend to remain constant for years.8, 48 However, it has largely been replaced by the more sensitive ELISA. Many different variations of the ELISA have been developed for the diagnosis of VMV
Virus isolation from live animals can be achieved by in vitro cultivation of macrophages from peripheral blood or milk, with or without the addition of indicator cells. Most ovine cell cultures are suitable indicator cells, but sheep choroid plexus cells are most often used. After the death of an animal, virus is most readily isolated by explanting affected tissues such as lung, choroid plexus, synovial membrane, or mammary gland. Cytopathic effects develop slowly (over several weeks) and include the appearance of refractile stellate cells and syncytia. The presence of virus can be detected by reverse transcriptase assay, electron microscopy, or immunolabelling techniques.17, 49
Differential diagnosis
The respiratory signs of visna-maedi are basically the same as those associated with chronic pneumonia. Pneumonia caused by bacteria and certain other viral infections is usually more acute and accompanied by fever, although such secondary infections often occur in maedi and complicate the diagnosis. Proliferative interstitial pneumonia,58 shown to be associated with Mycoplasma ovipneumonia, resembles maedi, as does the pneumonia associated with Dictyocaulus filaria infestation. Typical cases of jaagsiekte and maedi are easy to distinguish by means of the presence or absence of lung exudate, but it is not always so straightforward when secondary infection is present. Wasting and nervous signs may resemble those caused by scrapie, internal parasites, meningeal worm and/or secondary to severe anaemia, and vitamin E/selenium deficiency (white muscle disease/nutritional myopathy).
Control
Except for symptomatic medication, there is no specific treatment for visna-maedi and the outcome of the disease is invariably fatal. Successful vaccination has not yet been achieved, leaving eradication of infection as the only current viable option. This has been achieved on a national basis in Iceland by the implementation of a drastic test and slaughter policy. Most control programs focus on elimination of infected animals and their progeny46 Such programs have been in operation for many years in several European countries, resulting in successful control. A nucleus of disease-free animals can be established in a short time by isolating lambs at birth and rearing them artificially.15, 16, 49
References
- AGNARSDOTTIR, G., THORSTEINSDOTTIR, H., OSKARSSON, T., MATTHIASDOTTIR, S., HAFLIDADOTTIR, B. S., ANDRESSON, O. S. & ANDRESDOTTIR, V., 2000. The long terminal repeat is a determinant of cell tropism of visna-maedi virus. Journal of General Virology, 81, 1901–1905.
- ALVAREZ, V., DALABUIT-TEST, M., ARRANZ, J., I., L., JUSTE, R. A., AMORENA, B., DE ANDRES, D., LUJAN, L., BADIOLA, J. J. & BERRIATUA, E., 2006. PCR detection of colostrum-associated maedi-visna virus (MVV) infection and relationship with ELISA-antibody status in lambs. Research in Veterinary Science, 80, 226-234.
- ANDERSON, A. A., HARKISS, G. D. & WATT, N. J., 1994. Quantitative analysis of immunohistological changes in the synovial membrane of sheep infected with visna-maedi virus. Clinical Immunology and Immunopathology, 72, 21-29.
- BLACKLAWS, B. A., 2012. Small ruminant lentiviruses: Immunopathogenesis of visna-maedi and caprine arthritis encephalitis. Comparative immunology, microbiology and infectious diseases, 35, 259-269.
- BLACKLAWS, B. A., BIRD, P., ALLEN, D. & MC CONNELL, I., 1994. Circulating cytotoxic T lymphocyte precursors in visna-maedi virus-infected sheep. Journal of General Virology, 75, 1589–1596.
- BLACKLAWS, B. A., BIRD, P., ALLEN, D., ROY, D. J., MAC LENNAN, I. C. M., HOPKINS, J., SARGAN, D. R. & MCCONNELL, I., 1995. Initial lentivirus-host interactions within lymph nodes: A study of visna-maedi virus infection in sheep. Journal of Virology, 69, 1400–1407.
- BROUGHTON-NEISWANGER, L. E., WHITE, S. N., KNOWLES JR, D. P., MOUSEL, M. R., WHITE, S. N., MOUSEL, M. R., LEWIS, G. S., MARSHALL, K. L. & KNOWLES, J., D. P., 2010. Non-maternal transmission is the major mode of ovine lentivirus transmission in a ewe flock; A molecular epidemiology study. Infection, Genetics and Evolution, 10, 998-1007.
- CAREY, N. & DALZIEL, R. G., 1993. The biology of visna-maedi virus—an overview. British Veterinary Journal, 149, 437–454.
- COLLIE, D. D. S., WATT, N. J., WARREN, P. M., BEGARA, I. & LUJAN, L., 1993. Effects on lung compliance, lung volume, and single-breath transfer factor for carbon monoxide in sheep with lentivirus-induced lymphoid interstitial pneumonia. Journal of Veterinary Research, 54, 454–462.
- CORDIER, G., COZON, G., GREENLAND, T., ROCHER, F., GUIGEN, F., GUERRAT, S., BRUNE, J. & MORNEX, J. F., 1990. In vivo activation of alveolar macrophages in ovine lentivirus infection. Clinical Immunology and Immunopathology, 55, 355–367.
- COWDRY, E. V. & MARSH, H., 1927. Comparative pathology of South African jaagziekte and Montana progressive pneumonia of sheep. Journal of Experimental Medicine, 45, 571–585.
- CUTLIP, R. C., JACKSON, T. A. & LAIRD, G. A., 1977. Prevalence of ovine progressive pneumonia in a sampling of cull sheep from western and midwestern United States. American Journal of Veterinary Research, 38, 2091–2093.
- CUTLIP, R. C., LEHMKUHL, H. D., BROGDEN, K. A. & BOLIN, S. R., 1985. Mastitis associated with ovine progressive pneumonia virus infection in sheep. American Journal of Veterinary Research, 46, 326–328.
- CUTLIP, R. C., LEHMKUHL, H. D., BROGDEN, K. A. & MC CLURKIN, A. W., 1985. Vasculitis associated with ovine progressive pneumonia infection in sheep. American Journal of Veterinary Research, 46, 61–64.
- CUTLIP, R. C., LEHMKUHL, H. D., SCHMERR, M. F. & BROGDEN, K. A., 1988. Ovine progressive pneumonia (Visna-maedi) in sheep. Veterinary Microbiology, 17, 237–250.
- CUTLIP, R. C., LEHMKUHL, H. D., WOOD, R. L. & BROGDEN, K. A., 1985. Arthritis associated with ovine progressive pneumonia. American Journal of Veterinary Research, 46, 65–68.
- DAWSON, M., 1980. Visna-maedi: A review. The Veterinary Record, 106, 212–216.
- DAWSON, M., DONE, S. H., VENABLES, C. & JENKINS, C. E., 1990. Visna-maedi and sheep pulmonary adenomatosis: A study of concurrent infection. British Veterinary Journal, 146, 531–538.
- DE BOER, G. F., 1970. Zwoegerziekte, een persisterende virus infection bij schapen. PhD Thesis, University of Utrecht, The Netherlands.
- DE KOCK, G., 1929. Are the lesions of jaagsiekte in sheep of the nature of a neoplasm? Fifteenth Annual Report of the Director of Veterinary Services, Union of South Africa, 611–641.
- DENG, P., CUTLIP, R. C., LEHMKUHL, H. D. & BROGDEN, K. A., 1986. Ultrastructure and frequency of mastitis caused by ovine progressive pneumonia virus infection in sheep. Veterinary Pathology, 23, 184–189.
- DOHOO, J. R., HAENY, D. P., STEVENSON, R. G., SAMAGH, B. S. & RHODES, C. S., 1987. The effects of Visna-maedi infection on productivity in ewes. Preventive Veterinary Medicine, 4, 471–484.
- DUNGU, B., VORSTER, J., BATH, G. F. & VERWOERD, D. W., 2000. The effect of a natural visna-maedi virus infection on the productivity of South African sheep Onderstepoort Journal of Veterinary Research, 67, 87–96.
- ERIKSSON, K., MC INNES, E., RYAN, S., TONKS, P., MC CONNELL, I. & BLACKLAWS, B., 1999. CD4+ T-cells are required for the establishment of visna-maedi virus infection in macrophages but not dendritic cells in vivo. Virology, 258, 355–364.
- ERIKSSON, K., MC INNES, E., RYAN, S., TONKS, P., MC CONNELL, I. & BLACKLAWS, B., 1999. In vivo depletion of CD8+ cells does not affect primary maedi visna virus infection in sheep. Veterinary Immunology and Immunopathology, 70, 173–187.
- GELMETTI, D., GIBELLI, L., BROCCHI, E. & CAMMARATA, G., 2000. Using a panel of monoclonal antibodies to detect Maedi virus (MV) in chronic pulmonary distress of sheep. Journal of Virological Methods, 88, 9-14.
- GEORGSSON, G., NATHANSON, N., PALSSON, P. A. & PÉTURSSON, G., 1976. The pathology of visna and maedi in sheep. In: KIMBERLIN, R.H., (ed.). Slow Virus Diseases of Animals and Man. Amsterdam: North Holland Publishing Company.
- GEORGSSON, G. & PALSSON, P. A., 1971. The histopathology of maedi. A slow, viral pneumonia of sheep. Veterinary Pathology, 8, 63-80.
- GONZALEZ, L., JUSTE, R. A., CUERVO, L. A., IDIGORAS, I. & SAEZ DE OCARIZ, C., 1993. Pathological and epidemiological aspects of the coexistence of visna-maedi and sheep pulmonary adenomatosis. Research in Veterinary Science, 54, 140–146.
- GUDNADOTTIR, M., 1974. Visna-maedi in sheep. Progress in Medical Virology, 18, 336–349.
- JUBB, K. V. F., KENNEDY, P. C. & PALMER, N., 1985. Pathology of Domestic Animals. 3rd Edition. Orlando, Florida, USA: Academic Press, 1, 2, 3, 574, 582 & 527.
- LEE, W. C., MCCONNEL, I., BLACKLAWS, B. A. & LEE, W. C., 1997. Antigen presenting function of visna-maedi virus infected macrophages. Journal of the Chinese Society of Veterinary Science, 23, 256–264.
- LUJAN, L., BEGARA, I., COLLIE, D. D. S. & WATT, N. J., 1994. Ovine lentivirus (visna-maedi virus) protein expression in sheep alveolar macrophages. Veterinary Pathology, 31, 695–703.
- LUJAN, L., BEGARA, I., COLLIE, D. D. S. & WATT, N. J., 1995. CD8+ lymphocytes in bronchoalveolar lavage and blood: In vivo indicators of lung pathology caused by visna-maedi virus. Veterinary Immunology and Immunopathology, 49, 89–100.
- LUJAN, L., GARCIA-MARIN, J. F., FERNANDEZ DE LUCO, D., VARGAS, A. & BADIOLA, J. J., 1991. Pathological changes in the lungs and mammary glands of sheep and their relationship with visna-maedi infection. The Veterinary Record, 129, 51–54.
- MARSH, D. T., 1923. Progressive pneumonia in sheep. Journal of the American Veterinary Association, 62, 458–473.
- MINGUIJÓN, E., REINA, R., PÉREZ, M., POLLEDO, L., VILLORIA, M., RAMÍREZ, H., LEGINAGOIKOA, I., BADIOLA, J. J., GARCÍA-MARÍN, J. F., DE ANDRÉS, D., LUJÁN, L., AMORENA, B. & JUSTE, R. A., 2015. Small ruminant lentivirus infections and diseases. Veterinary Microbiology, 181, 75-89.
- MITCHELL, D. T., 1915. Investigations into Jaagsiekte. Third and Fourth Reports of the Director of Veterinary Research, Union of South Africa, 585–614.
- MUZ, D., OGUZOGLU, T. C., ROSATI, S., REINA, R., BERTOLOTTI, L. & BURGU, I., 2012. First molecular characterization of visna/maedi viruses from naturally infected sheep in Turkey. Archives of Virology, 158, 559-570.
- NARAYAN, O., GRIFFIN, D. E. & CHASE, J., 1977. Antigenic shift of visna virus in persistently infected sheep. Science, 197, 376–378.
- NARAYAN, O., WOLINSKY, J. S., CLEMENTS, J. E., STRANDBERG, J. D., GRIFFIN, D. E. & CORK, L. C., 1982. Slow virus replication: The role of macrophages in the persistence and expression of visna viruses of sheep and goats. Journal of General Virology, 345–356.
- NARAYAN, O., ZINK, M. C., GORRELL, M., CRANE, S., HUSO, D., JOLLY, P., SALTARELLI, M., ADAMS, R. J., CLEMENTS, J. E. & LEVY, J. A., 1993. The lentiviruses of sheep and goats. The Retroviridae, 2, 229–255.
- OLIVER, R. E., GORHAM, J. R., PARISH, S. F., HADLOW, W. J. & NARAYAN, O., 1981. Ovine progressive pneumonia: Pathologic and virologic studies on the naturally occurring disease. American Journal of Veterinary Research, 42, 1554–1559.
- PALSSON, P. A., 1976. Maedi and visna in sheep. In: KIMBERLIN, R.H., (ed.). Slow Virus Diseases of Animals and Man. Amsterdam: North-Holland Publishing Company.
- PEPPIN, M., VITY, C., RUSSO, P., MORNEX, J. F. & PETERHANS, E., 1998. Visna-maedi virus infection in sheep: A review. Veterinary Research, 29, 341–367.
- PEREZ., 2010.
- PETERHANS, E., GREENLAND, T., BADIOLA, J., HARKISS, G., BERTONI, G., AMORENA, B., ELIASZEWICZ, M., JUSTE, R. A., KRASSNIG, R., LAFONT, J. P., LENIHAN, P., PÉTURSSON, G., PRITCHARD, G., THORLEY, J., VITU, C., MORNEX, J. F. & PÉPIN, M., 2004. Routes of transmission and consequences of small ruminant lentiviruses (SRLVs) infection and eradication schemes. Veterinary Research, 35, 257-274.
- PETURSSON, G., GEORGSSON, G., PALSSON, P. A. & DINTER, Z., 1990. Visna-maedi virus. In: MOREIN, B., (ed.). Virus infections of ruminants. Amsterdam: Elsevier, 431–440.
- PETURSSON, G. & HOFF-JORGENSEN, R., 1990. Visna-maedi and Related Diseases. Dordrecht: Kluwer Academic Publishers.
- PRITCHARD, G. C. & DONE, S. H., 1990. Concurrent visna-maedi virus infection and pulmonary adenomatosis in a commercial breeding flock in East Angola. Veterinary Record, 127, 197–200.
- QUERAT, G., AUDOLY, G., SONIGO, P. & VIGNE, R., 1990. Nucleotide sequence analysis of SA-OVMV, a visna-related ovine lentivirus: Phylogenetic history of lentiviruses. Virology, 175, 434–447.
- REYBURN, H. T., ROY, D. J., BLACKLAWS, B. A., SARGAN, D. R. & MC CONNELL, I., 1992. Expression of visna-maedi virus major core protein, p25: Development of a sensitive p25 antigen detection assay. Journal of Virological Methods, 37, 305–320.
- ROY, D. J., WATT, N. J., INGMAN, T., HOUWERS, D. J., SARGAN, D. R. & MC CONNELL, I., 1992. A simplified method for the detection of visna-maedi virus RNA by in situ hybridization. Journal of Virological Methods, 36, 1-11.
- SIGURDSSON, B. & PALSSON, P. A., 1958. Visna of sheep. A slow, demyelinating infection. British Journal of Experimental Pathology, 39, 519–528.
- SIMARD, C. L. & BRISCOE, M. R., 1990. An enzyme-linked immunosorbent assay for detection of antibodies to visna-maedi virus in sheep. I. A simple technique for production of antigen using sodium dodecyl sulfate treatment. II. Comparison to conventional agar gel immunodiffusion test. Canadian Journal of Veterinary Research, 54, 446–450, 451–456.
- SMITH, C., 1992. Ovine Lentivirus: A Real or Imaginary threat? Journal of the American Veterinary Medicine Association, 200, 139–143.
- SNOWDER, G. D., GATES, N. L., A., G. H. & GORHAM, J. R., 1990. Prevalence and effect of subclinical ovine progressive pneumonia virus infection on ewe wool and lamb production. Journal of the American Veterinary Medicine Association, 197, 475–479.
- SULLIVAN, N. D., ST GEORGE, T. D. & HORSFALL, N., 1973. A proliferative interstitial pneumonia of sheep associated with mycoplasma infection. Australian Veterinary Journal, 49, 57–62.
- WATT, N. J., KING, T. J., COLLIE, D., MC INTYRE, N., SARGAN, D. & MC CONNELL, I., 1992. Clinicopathological investigation of primary, uncomplicated visna-maedi virus infection. The Veterinary Record, 131, 455–461.
- WHITE, S. N. & KNOWLES, D. P., 2013. Expanding possibilities for intervention against small ruminant lentiviruses through genetic marker-assisted selective breeding. Viruses, 5(6), 1466-1499.
- ZANONI, R., PAULI, U. & PETERHANS, E., 1990. Detection of caprine arthritis-encephalitis and visna-maedi viruses using the polymerase chain reaction. Experientia, 46, 316–319.
- ZHANG, Z., WATT, N. J., HOPKINS, J., HARKISS, G. & WOODALL, C. J., 2000. Quantitative analysis of visna-maedi virus DNA load in peripheral blood monocytes and alveolar macrophages. Journal of Virological Methods, 86, 13-20.
- ZINK, M. C., NARAYAN, O., KENNEDY, P. G. E. & CLEMENTS, T. E., 1987. Pathogenesis of visna-maedi and caprine arthritis-encephalitis: New leads in the mechanism of restricted virus replication and persistent inflammation. Immunology and Immunopathology, 15, 167–180.