- Infectious Diseases of Livestock
- Part 2
- African horse sickness
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
African horse sickness
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
African horse sickness
Previous authors: JAW COETZER AND AJ GUTHRIE
Current authors:
JAW COETZER - BVSc, BVSc(Hons), M.Med.Vet(Path), DVSc(Honoris Causa), Emeritus Professor, Faculty of Veterinary Science, University of Pretoria, South Africa.
M QUAN - Associate Professor, BVSc, MSc, PhD, Department of Veterinary Tropical Diseases, University of Pretoria, South Africa.
AJ GUTHRIE - Professor, Director of Equine Research Centre, BVSc, PhD, Faculty of Veterinary Science, University of Pretoria, South Africa.
CT WEYER - BVSc, MSc, PhD, State Veterinarian Boland Authorised Veterinarian and Research Officer, Equine Health Fund, WITS Health Consortium, Cape Town, South Africa.
Introduction
African horse sickness (AHS) is a peracute, acute, subacute or subclinical non-contagious disease of equids caused by an orbivirus, of which there are nine serotypes, all transmitted biologically by Culicoides midges. The disease is manifested by fever, inappetence and clinical signs and lesions compatible with impaired respiratory and circulatory functions characterized by oedema of subcutaneous and intermuscular tissues and the lungs, transudation into the body cavities, and haemorrhages, particularly of the serosal surfaces. The mortality rate in naive horses, the most susceptible species, may be as high as 95 per cent while donkeys and mules are considerably less susceptible and generally develop only a febrile disease. The disease occurs regularly in most countries in sub-Saharan Africa and is regarded as one of the major scourges of the continent.
The first known historical reference to a disease resembling AHS is in an Arabian document ‘Le Kitâb El-Akouâ El- KâfiahWaEl Chafiâh’, in which an outbreak of the disease in Yemen in 1327 was reported. Another early reference to what was probably AHS in horses imported into East Africa from India is by Father Monclaro in his account of the journey of Francisco Baro to East Africa in 1569.98, 201
Horses and donkeys were introduced into South Africa shortly after the arrival of the first settlers of the Dutch East India Company in the Cape of Good Hope in 1652.98 According to Theiler,201 frequent references were made to the disease in the records of the Dutch East India Company. In 1719, for instance, nearly 1 700 horses succumbed to the dreaded ‘perreziekte’ or ‘pardeziekte’ in the Cape of Good Hope. The ability of frost to arrest outbreaks was recognized at this time. In 1830, Thomas Perry, the District Surgeon at Graaff-Reinet wrote “If the animal is rode during the Sickness, or urged by driving or otherwise to any degree of speed, he falls at once, literally suffocated by the quantity of frothy matter which fills his trachea and issues in abundance from his nostrils”. In 1850 Gordon Cumming and other hunters reported AHS among their horses while Livingstone was unable to use horses on some of his journeys in southern, central and East Africa and was forced to travel on foot or on the back of an ox.201 The Voortrekkers also suffered severe losses amongst their horses.98One of the worst recorded epidemics occurred in 1854/1855 when mortalities were close to 64 850.32 At that time this represented a staggering 40 per cent of the horse population of the Cape and the cost at that time was estimated to be around £525 000. The result was a decline in the growth rate and expansion of the Cape Colony. Horses that survived AHS were considered “salted” and were worth six to ten times more in value.
At first, AHS was confused with anthrax and biliary fever. Dew, grass and cobwebs were suspected as possible causes, where “The distemper was everywhere most destructive in the low grounds and along the watershed of each district; the fatal miasma seeming to be held in suspension during the day, and to descend again at night with the heavy dews”.32 Edington considered the causal agent to be a mushroom.69 In 1900, M’Fadyean118 succeeded in transmitting the disease with a bacteria-free filtrate of blood from an infected horse. According to Henning,98this finding was confirmed independently by Theiler and by Nocard a year later, and by Sieber in 1911. From these experiments, it was concluded that the disease was caused by a virus.
Winged nocturnal insects, such as mosquitoes, were suggested by Watkins-Pitchford to be responsible for the transmission of AHS.161He showed that horses could be protected against infection when housed in mosquito-proof enclosures. However, it was only in 1944 when Du Toit66 reported that Culicoides midges were probably vectors of both AHS and bluetongue viruses (see Vectors: Culicoides.)
One of the reasons why Sir Arnold Theiler sited the now Onderstepoort Veterinary Institute at Onderstepoort in 1908, was the high prevalence of AHS in the immediate area. The earliest reasonably successful AHS vaccine was developed by Theiler in 1905 and its use continued until 1933. The ‘vaccine’ consisted of simultaneous inoculation of the virus together with serum obtained from horses that had recovered from the disease. This method provided unpredictable results and was not widely used. Theiler’s pioneering research194, 195, 196, 197, 201suggested that there was a plurality of ‘immunologically distinct strains’ of AHS virus (AHSV) since immunity acquired against one ‘strain’ did not always afford protection against infection by ‘heterologous strains’; 26 to 81 per cent of horses reacted when challenged with heterologous strains. This work was expanded on by Alexander and co-workers,9, 11, 13, 14 who employed antisera from horses recovered from AHS in cross-neutralization tests in mice.
A major advance in research on AHSV came in 1933 and 1935 when Alexander8, 9showed that viscerotropic isolates of AHSV became neurotropic but did not lose their immunogenicity after serial intracerebral passage in mice. Alexander in 193812was the first to succeed in propagating AHSV in chicken embryos by inoculation of the chorio-allantoic membrane, and he showed that the virus became attenuated during passage in embryonated hens’ eggs.
Although cases of AHS occur every year in horses in South Africa, major outbreaks before 1953 seem to have occurred at intervals of roughly 20 to 30 years. Severe losses were reported in 1780, 1801, 1839, 1855, 1862, 1891, 1914, 1918, 1923, 1940, 1946 and 1953. The outbreak of 1855 is still considered to have been the most severe.32
Before the advent of mechanized transport, the horse played a very important role in transportation, was vital in military operations, and was used for draught power in agriculture and mining. Heavy losses due to AHS were therefore very disruptive and deeply felt. Although horses are nowadays seldom used for these purposes, many have considerable monetary value as performance horses or animals used for other forms of recreation. Furthermore, the exportation of horses from areas where AHS occurs can only be accomplished following strict quarantine and testing procedures.19, 21
Since a polyvalent vaccine against AHS has been available in southern Africa, severe losses due to the disease have largely ceased although it continues to occur in individual or small groups of horses. Recently, outbreaks of AHS occurred due to the transmission of polyvalent AHSV modified live vaccine strains that had reverted to virulence and reassorted.213 Epidemics of the disease in recent years in countries outside the endemic regions in Africa such as Thailand and Malaysia17, 93, 108, 115, 116, 143, 193serve as a warning that the disease may spread to continents hitherto free of the disease.
Aetiology
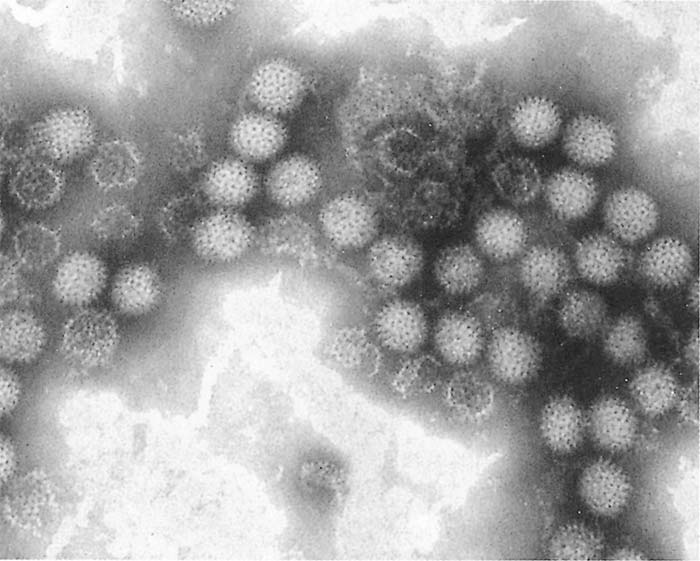
African horse sickness virus is classified in the genus Orbivirus in the family Reoviridae. It is similar in morphology (Figure 1), and shares many properties with other orbiviruses such as bluetongue and equine encephalosis viruses47, 188, 189, 191, 208(see Bluetongue).
Nine types of AHSV are known, of which the last was isolated in 1960.102, 133Antisera that neutralize homologous virus may cross-neutralize heterologous serotypes (e.g. there is cross-neutralization between serotypes 1 and 2, 3 and 7, 5 and 8, and 6 and 9). Field evidence indicates, however, that no significant intratypic variation occurs.
The morphology as well as the physio-chemical, biological and immunological properties of AHSV have been reviewed.9, 136, 208Virions are c. 70 nm in diameter and have icosahedral symmetry.111, 162, 163They contain ten double-stranded RNA genome segments encapsulated within a double-layered capsid made up of 32 capsomeres, each comprising seven structural proteins.43, 44, 147
The virus is relatively heat-stable as the infectivity of citrated plasma containing AHSV is not inactivated by heating at 55 to 75 °C for 10 minutes. Virus derived from cell cultures in a medium containing calf serum is stable for three months at 4 °C but at –25 °C there is a marked reduction in virus titre, which occurs within 48 hours unless the virus is diluted in a stabilizer. The virus can be stored for at least six months at 4 °C in saline containing 10 per cent serum. African horse sickness virus is best stored in a lyophilized state; minimal loss of titre occurs in lyophilized vaccine strains kept at 4 °C. Putrefaction does not destroy the virus; putrid blood may retain its infectivity for more than two years.194 Virus can be recovered for 12 months from washed infected erythrocytes stored at 4 °C.100 The optimal pH for virus survival is 7,0 to 8,5; the virus is sensitive to acid pH values but is relatively resistant to pH changes on the alkaline side of neutrality.9It is resistant to ether and other lipid solvents.
African horse sickness virus may be attenuated by serial passage in mice, embryonated hens’ eggs and cell cultures. Attenuation by passage of the virus in suckling mice results in less neurotropism than does passage in adult mice.74Strains of AHSV that have been attenuated by approximately 100 intracerebral passages in adult mice brains have been used in the preparation of ‘horse sickness neurotropic mouse brain vaccine’ and ‘horse sickness neurotropic tissue culture vaccine’.133, 153These vaccines were used extensively until 1974 when a start was made on replacing some of these strains with large plaque variants of AHSV.61
Virus attenuation occurs faster during passage in cell culture than in mouse brains. The virulent virus loses its viscerotropic properties after 8 to 13 passages in monkey kidney stable (MS) cell or baby hamster kidney (BHK) cell lines.135Within 23 passages in these cell cultures, the infectivity of the virus for mice decreases considerably and it is avirulent for horses. However, different strains of the virus become attenuated at different rates.13, 74
The size of plaques in cell culture is a marker of the virulence of AHS viruses; large plaque variants are much less virulent than small plaque variants. Therefore, large plaque variants are selected for the preparation of attenuated vaccine strains.20, 75
Isolation of the virus can be performed using BHK21, African green monkey (Vero) or MS cells.71, 73, 136, 181Cytopathic effects, characterized by focal or dispersed increased refractivity and detachment of cells, may appear three to seven days after inoculation of cultures, and may occasionally only become conspicuous in the second passage. After three passages, advanced cytopathic effects develop within two to four days.20The virus has also been adapted to calf, horse and lamb kidney cell cultures73 and has been propagated in a mosquito (Aedes albopictus) cell line.136Ultrastructural changes in mammalian-derived cell cultures associated with replication of AHSV include the formation of intracytoplasmic tubular structures (of undetermined length and 18 nm wide), often in close association with virus particles and ‘virus factories’, composed of a virus-specific non-capsid polypeptide of about 54 000 daltons.42, 105, 111, 147
Two to four-day-old suckling mice, cell cultures or intravascular inoculation of embryonated hens’ eggs are sensitive methods of isolating AHSV. Although adult mice can also be used for this purpose they are not as susceptible as suckling mice.20, 208Other laboratory animals such as ferrets, guinea pigs, mice, and various strains of rats have been used to cultivate AHSV.8, 9, 71, 130, 131, 136Rabbits, however, are not susceptible to infection.9, 133, 136In the first passage the incubation period in suckling mice inoculated intracerebrally with infected horse blood varies between 4 and 20 days and mortality may reach 100 per cent. In subsequent passages, the incubation period is reduced to two to four days.20, 104, 130, 136Mice show nervous signs before death; high concentrations of the virus occur in the brains.
Epidemiology
African horse sickness occurs regularly throughout sub-Saharan Africa. It is endemic in eastern, central and most parts of southern Africa.201 The disease is also reported from time to time in countries in North Africa from where it has occasionally extended into countries of the Middle East and Spain.12, 48 However, its intrusion into North Africa and countries around the Mediterranean Sea and Asia is impeded by the Sahara Desert.40 African horse sickness virus can be distributed over great distances if equids incubating the disease are translocated by land, sea or air.116, 172 Spread of AHS can also occur as a result of the wind-borne spread of infected Culicoides midges.67, 184Apparently AHS does not occur in Madagascar or Mauritius.
In Egypt the disease occurred in 1928, 1943, 1953, 1958 and 1971 (all outbreaks originated in the areas of Aswan and Qena provinces, and the international boundaries between Egypt and Sudan),181 in Yemen in 1930 and in the then Palestine, Syria, Lebanon and Jordan in 1944.12, 103, 104, 136During the summer of 1959, AHS caused by serotype 9 occurred in the south-eastern regions of Iran. This was followed by outbreaks during the spring of 1960 in most of the Persian Gulf area (Cyprus, Iraq, Syria, Lebanon, Jordan) as well as in Afghanistan, Pakistan, India and Turkey. Between 1959 and 1961 this region lost more than 300 000 equine animals.136, 169In 1965 the disease occurred in Libya, Tunisia, Algeria and Morocco and also spread across the Strait of Gibraltar into Spain in 1966.97Fortunately it occurred at the end of the summer and the outbreak in Spain was quickly terminated by the application of strict control measures. Between 1987 and 1990 outbreaks of AHS occurred again in Spain; the source of infection was suspected to be ten zebras (Equus burchelli) imported from Namibia.93, 116African horse sickness was also diagnosed in southern Portugal in 1989 and northern Morocco between 1989 and 1991; these outbreaks are thought to be extensions of outbreaks that occurred in August of the same year in southern Spain.93, 222 In 1989 a minor outbreak of AHS, caused by serotype 9, was reported in Saudi Arabia after an absence of 30 years.17 In 1997 outbreaks of AHS occurred in Saudi Arabia and Yemen and in 1999 on the Cape Verde Islands. Serotypes 6 and 9 of AHSV were isolated from samples collected from equids in Ethiopia in 20032, 219 and serotype 2 resulted in the death of approximately 2 000 horses in Ethiopia in 2008. Serotypes 2 and 7 of AHSV were isolated in Senegal in 2007 and serotype 2 was isolated in Nigeria in 2007. African horse sickness was reported in The Gambia in 2007. A possible outbreak of AHS occurred in Saudi Arabia in 2014.4 Outbreaks of AHS were reported in Chad in 2019 which resulted in the death of more than 85 per cent of the 376 reported cases.138 In 2020, an outbreak of AHS occurred in Thailand that resulted in the death of more than 93 per cent of the 604 reported cases.18, 108, 115, 193 The source of infection was suspected to be zebras (Equus burchelli) imported from southern Africa.112 In September 2020, Malaysia also reported an outbreak of AHS involving five horses none of which showed severe clinical signs or died.143
In South Africa, the disease usually first occurs in the early summer months (October or November) in the eastern parts of the country. The extent of the spread is influenced by the time of the year in which the disease first makes its appearance and by the extent of favourable climatic conditions for the breeding of Culicoides midges.40 Although the three Cape provinces are usually free of the disease, the infection sometimes extends into these areas and caused serious losses. According to Du Toit,62 heavy losses were reported even in Belfast, a town situated at almost the highest altitude (about 2 100m above sea level) in Mpumalanga, during the severe epidemic of 1923. Outbreaks of AHS in the Cape Peninsula in the Western Cape Province have been reported in 1967,40 1990,601999,33 2004,186, 213 2011,213 2014,213 201686 and most recently in 2021. The outbreaks in 2004, 2011, 2014 and 2016 were all associated with reversion to virulence and reassortment of live attenuated vaccine viruses.86, 213
In the summer rainfall areas of southern Africa, AHS is generally most prevalent in warm coastal regions or low lying, moist, inland areas, such as valleys, marshes, and riverine vegetation during the second half of summer. Early and heavy rains followed by warm, dry spells favour the occurrence of epidemics. In South Africa, the most serious outbreaks are commonly encountered in March and April. Following the first frosts, which are usually experienced towards the end of April or in May, the disease disappears abruptly. However, in the Lowveld, where frost is less common, cases of the disease may continue into May and June.98
During outbreaks of AHS in endemic areas, different virus serotypes may be active simultaneously within an area but usually, one serotype dominates during a particular season, followed the next year by the dominance of another serotype. All serotypes are commonly responsible for outbreaks of AHS in South Africa, while serotypes 1, 3, 4 and 9 have been recorded outside Africa. Outside southern Africa serotype 9 appears to be widespread and occurs in North and West Africa158 and was responsible for the epidemics in the Middle East.
African horse sickness affects primarily equine species. Horses are most susceptible to the disease (mortality rate between 70 and 95 per cent), but mules are less so (mortality rate between 50 and 70 per cent). Donkeys and zebras are very resistant, most infections being subclinical.12, 201Generally horses of all breeds are equally susceptible to AHS but variation in susceptibility to the same virus of individual horses has been reported.201 However, there are horses in North and West Africa which descend from animals that have been present there since at least 2000 BC, and which have apparently acquired natural resistance to AHS.41 Theiler201 was unable to produce fatal disease in donkeys inoculated with large quantities of virulent blood; at most a mild fever reaction was produced. Donkeys in the Middle East, however, appear to be far more susceptible to disease (the mortality rate may reach 10 per cent) than are South African donkeys.12 Zebras are highly resistant to the disease and only show a mild fever following artificial infection.77
Foals born to immune mares acquire passive immunity by the ingestion of colostrum soon after birth.14 This immunity is variable in individual foals and progressively declines as a result of catabolism of the immunoglobulins until it is completely lost after about four to six months.52, 212 Foals born to fully susceptible mares are just as susceptible as are previously unexposed adult horses.
In addition to equids, dogs are the only other species that contract a highly fatal form of the disease after infection with AHSV.A possible reason for this may be that both equine and canine blood contains proteases that cleave the AHSV VP2 outer protein.123, 124African horse sickness virus serotype 9 has been isolated from the blood of stray dogs in Egypt,181 and antibodies to AHSV have been detected in the sera of dogs in India,185 Egypt,23, 24Botswana,6, 7 Kenya,6, 7 Nigeria,26, 27 and South Africa.131, 206The mean seroprevalence reported is 7 per cent but is variable with up to 50 per cent of dogs testing positive for AHSV antibodies. Antibodies to serotypes 1,913,131 4,6, 7 6,144, 145 76, 7 and 927 have been reported.
The disease has been transmitted to dogs by the ingestion of infected horse meat35, 53, 160and the inoculation of AHSV infected blood. It iswas believed that dogs play only a minor role if any in the epidemiology of AHS as all reports of naturally occurring canine AHS clinical cases before 2013 occurred after dogs ingested AHSV infected horse meat35, 91, 160, 202 and the low affinity of Culicoides midges for canine blood meals. This assumption has been brought into question as recent cases of AHS have been reported in dogs without apparent ingestion of infected horse meat.146, 206, 216
Culicoides midges are more opportunist feeders than previously thought and canine blood meals have been found in multiple species including in the primary vector of AHSV, C. imicola.127, 170, 187 Midge transmission of the closely related bluetongue virus has been reported in dogs.150, 151The role of dogs in the epidemiology of AHSV should be investigated further.149
Clinical and pathological findings in dogs are consistent with the pulmonary form of AHS seen in equids.146, 199 Most cases become subclinically infected and may show a brief febrile response.119, 199, 206 Survival is uncommon once clinical signs occur.146
Besides early reports,69, 195, 204little evidence of antibody to AHSV has been detected in domestic or wild ruminants30, 36, 81, 92with the possible exception of camels.25, 181Pigs, cats and monkeys are refractory to infection with AHSV.136 Among wildlife, antibodies have been found only in zebras,30, 36, 55, 77African elephants (Loxondonta africana)36, 55, 77and black and white rhinoceroses (Diceros bicornis and Ceratotherium simium respectively).81However, the possibility that elephants are reservoir hosts of AHSV is questioned as 100 elephants tested in an endemic AHS area in South Africa had high complement fixing titres to both bluetongue and AHS viruses, but no neutralizing antibodies to these viruses, indicating that the sera of elephants react non-specifically in the complement fixation test.77 In contrast, in a similar study in Kenya, elephants were shown to have neutralizing antibodies to AHSV.36
It has not been shown that any mammalian species can act as a carrier of AHSV . There is a continuous transmission cycle of AHSV between Culicoides midges and zebras in the Kruger National Park in South Africa.29Under such circumstances a sufficiently large zebra population can act as a source for the virus.31, 113, 114It has been suggested that donkeys may play a similar role in parts of Africa where there are large donkey populations.95 Given the high mortality rate in horses, this species is regarded as an accidental or indicator host. Equids that have recovered from infection with AHSV mount a strong humoral antibody response and clear the virus, which explains the failure of the disease to become established outside tropical and subtropical Africa, despite the occurrence of many outbreaks outside endemic areas.98
African horse sickness is not contagious. The disease spreads by the movement of either infected vertebrate hosts, which then infect vectors in the ‘new’ region, or vectors. It can be distributed over great distances if equids incubating the disease are translocated by land, sea or air. Because the disease is transmitted by Culicoides midges, it has a seasonal occurrence and its prevalence is therefore influenced by climatic and other conditions which favour the breeding of Culicoides spp (see Vectors: Culicoides spp.) The virus is transmitted biologically by Culicoides spp., of which C. imicola and C. bolitinos have been shown to play an important role in Africa.134, 136, 207Culicoides variipennis, a midge prevalent in the USA but not present in southern Africa, can become infected by feeding on the chorio-allantoic membranes of chicken embryos infected with AHSV and midges of this species can transmit the virus seven days after becoming infected to develop chicken embryos by feeding on their chorio-allantoic membranes.39
On the open veld, most animals become infected during the period between sunset and sunrise when Culicoides midges are most active. During their lifetimes, the midges generally remain within a radius of a few kilometres of the site where they breed, but it has been postulated that they can be borne for many hundreds of kilometres in air currents.184Spread of AHSV in infected midges in this manner has been postulated as being responsible for several outbreaks. For example, it has been suggested that infected Culicoides midges transported by wind were responsible for the spread of the disease over the sea from Senegal to the Cape Verde Islands in 1943, from Turkey to Cyprus in 1960, and from Morocco to Spain in 1966.184
Although other insects have also been incriminated as possible vectors of AHSV, none has been conclusively shown to play a role under natural conditions. As long ago as 1934 it was shown that mosquitoes of the genus Aedes could harbour AHSV for one week but could not transmit the disease.140, 141While some workers have shown experimentally that Anopheles stephensi, Culex pipiens and Aedes aegypti can transmit the virus,154, 155, 156others failed to infect Ae. aegypti and Culex pipiens fatigans by allowing them to feed on virus suspensions or infected horses.211 Biting flies may play a minor role in the mechanical transmission of AHS but, as the virus is susceptible to desiccation and high temperature, and viraemia in infected horses is relatively low (approximately 105 TCID50/ml), this method of transmission is probably inefficient.
Ticks do not play a significant role in the transmission or maintenance of the virus under natural conditions. However, the dog tick (Rhipicephalus sanguineus sanguineus) may harbour and, under experimental conditions, transmit AHSV.99, 181 The virus can also replicate in Hyalomma dromedarii,25 which usually parasitizes camels.
Experimentally, AHS can be transmitted to horses by the parenteral inoculation of infective equine blood or organ suspensions; it is more readily transmitted by the intravenous than by the subcutaneous route.98
Pathogenesis
The outcome of infection in horses, including the incubation period and severity of disease, depends largely on the virulence of the virus and susceptibility of the animal. After infection, the initial multiplication of the virus occurs in the regional lymph nodes and is followed by a primary viraemia with subsequent infection of target organs, namely the lungs and lymphoid tissues throughout the body. Virus multiplication at these sites gives rise to a secondary viraemia of variable duration; in horses it is generally not higher than 105 TCID50/ml and lasts four to eight days but does not exceed 21 days, whereas in donkeys and zebras the levels of viraemia are lower but they may last as long as four weeks. In zebras, viraemia has been reported to occur in the presence of circulating antibodies.77 African horse sickness virus is closely associated with the erythrocytes in the blood.201
In experimentally infected horses high concentrations of the virus are found in the spleen, lungs, caecum, pharynx, choroid plexus and most lymph nodes by the second day after inoculation. This precedes the onset of fever or detectable viraemia. By the third day after virus inoculation, it is present in most organs.75 Even in the ‘cardiac’ (‘dikkop’, ‘thick’ or ‘swollen head’) form of AHS, virus levels in the myocardium are no higher than in the blood, indicating that the myocardium is not a primary site of virus replication.75 High concentrations of virus in lymphoid tissues may be responsible for the depletion of lymphocytes which occurs during the disease.75 However, transmission electron microscopic studies have not revealed the presence of virus in lymphocytes.139
Effusions into body cavities and oedematous changes of various tissues (particularly of the lungs), as well as serosal and visceral haemorrhages, are often evident in fatal cases of AHS and indicate endothelial cell damage. Although no significant ultrastructural changes or evidence of viral replication could be detected in endothelial cells in the lungs in one study,139 in another the presence of virus and ultrastructural changes in, and separation of, endothelial cells in the lungs were found.85
The factors determining the course and severity of the infection are not fully understood. Analysis of the transcriptome in peripheral blood mononuclear cells after virulent AHSV stimulation showed that AHSV may impair the innate immune response through the inhibition of type I and III interferons and as a result natural killer cell responses, with a prolonged expression of pro-inflammatory cytokines and chemokines that may cause immunopathology that may play a major role in the pathogenesis of AHSV infection.78, 79
Theiler201 described four forms of AHS in horses, and these are still useful in categorizing the different effects AHSV may have on equids. These are the horse sickness fever form; the peracute, ‘pulmonary’ or ‘dunkop’ (‘thin head’) form, i.e. cases in which subcutaneous swelling of the head is absent); the oedematous, ‘cardiac’ or ‘dikkop’ (‘thick’ or ‘swollen head’) form; and the ‘mixed’ form. It has been shown that small plaque variants of AHSV produce severe clinical reactions, while large plaque variants produce no or mild reactions. 75 There is some evidence that serial passage of AHSV in horses using a virus that has originated from the lung tissue consistently produced the peracute disease with marked pulmonary involvement, whereas passage using spleen homogenates from the same animal resulted in milder disease.75
Fully susceptible horses, such as foals that have lost their colostral immunity or horses that have never been exposed to the AHSV, usually develop the ‘dunkop’ form of AHS. Exercise during the febrile stage of the disease may also precipitate the ‘dunkop’ form.75, 104 Field observations indicated that the rigid distinction between different forms of AHS is not fully justifiable and that most cases of AHS are of the ‘mixed’ form.48, 75, 129 Although more than one serotype may be active during an outbreak, isolation of more than one serotype from a naturally infected animal has never been recorded.104
There are usually no lesions in the myocardium of horses that die of AHS, but mild or severe changes may occasionally be present.129 During the 1959 Middle East epidemic of AHS caused by serotype 9, severe myocardial lesions characterized by extensive areas of degeneration and necrosis of myocytes accompanied by a marked inflammatory response were described in fully susceptible horses, particularly those suffering from the ‘dikkop’ form of the disease.129 In AHS cases much of the oedema of the subcutaneous and other loose tissue is distributed in the dorsal parts of the body, which is inconsistent with cardiac failure.75
Pulmonary alveolar flooding by oedematous fluid would seem to occur terminally in most fatal cases139 although once it is initiated it may develop very rapidly, particularly in ‘dunkop’ cases. The histological demonstration of an abundance of fibrin in the pulmonary alveoli of horses that have died of the ‘dunkop’ form indicates that the oedema fluid has a high protein content, compatible with that of permeability oedema.139 This oedema results in respiratory distress, and ultimately, asphyxia.
Clinical signs
The clinical signs of naturally and experimentally infected horses have been described by various workers.34, 116, 129, 139, 201In experimentally induced cases, the incubation period usually varies between five and seven days, but it may be as short as two days and is rarely longer than ten days. Its duration depends on the virulence of the virus and the dose of virus received; the more virulent the virus and the larger the dose of the virus, the shorter the incubation period.
'Dunkop' or 'pulmonary' form
This form of the disease occurs most commonly when AHSV infects fully susceptible horses, notably foals that have lost their passively acquired immunity. It is also the usual form in dogs.
Following the incubation period, a fever may be the only sign for one or two days, with the rectal temperature reaching a maximum of 41 °C or sometimes higher. The characteristic clinical signs in this form of AHS are severe dyspnoea, paroxysms of coughing, and, in some, discharge of large quantities of frothy, serofibrinous fluid from the nostrils. In most cases, the discharge from the nose only appears after death. The onset of dyspnoea is generally very sudden, and death often occurs within a few hours of its appearance. Sometimes an apparently healthy horse, at work, becomes listless, suddenly severely dyspnoeic, and dies shortly thereafter. Dyspnoea and hyperpnoea are rapidly progressive; the respiratory rate may reach 75 per minute. Terminally, the nostrils are flared, the mouth is open, the tongue protrudes, the head and neck are extended, and the animal sweats profusely. At first, the appetite of affected animals remains good despite the high fever and respiratory distress. Sometimes the animal may take a mouthful of hay without chewing it.
The prognosis for horses suffering from the ‘dunkop’ form is extremely grave; less than 5 per cent recover. In recovering horses, the fever gradually subsides but the breathing remains laboured for some days.
'Dikkop' or 'cardiac' form
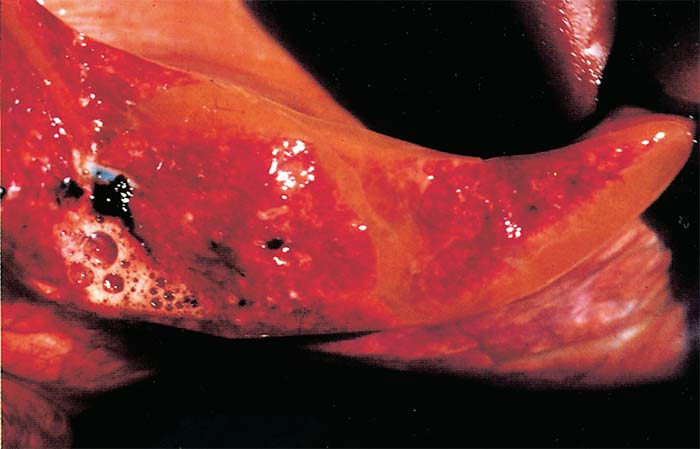
Subcutaneous oedematous swelling chiefly of the head and neck (Figure 2), and particularly the supraorbital fossae (Figure 3), characterize this form of AHS. The febrile reaction reaches its maximum at a later stage in the course of the disease than in the ‘dunkop form’ and may remain high for three to six days before declining. However, some animals develop only a very mild fever.
The swelling of the head and neck usually appears late in the course of the disease but if it should commence early, the course is always more acute, the condition more serious, and death frequently ensues. As the swellings increase, dyspnoea and cyanosis may supervene.
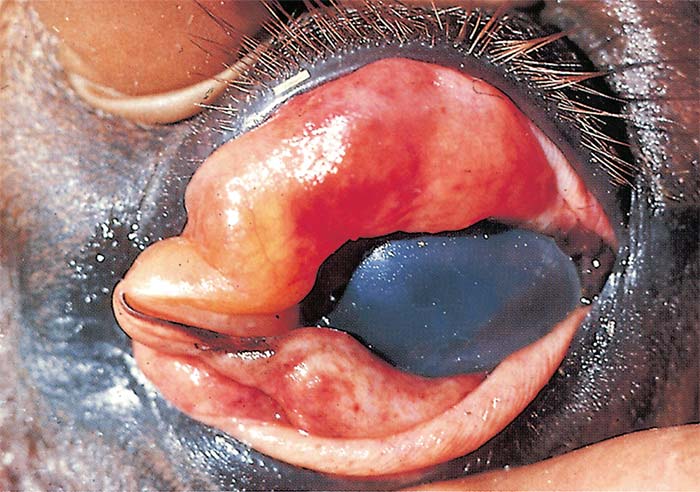
Varying degrees of swelling of the supraorbital fossae and other parts of the head are evident in affected horses, bulging of the supraorbital fossae being characteristic. In severe cases, the eyelids (Figure 4), lips, cheeks, tongue, intermandibular space, and sometimes also the neck, chest and shoulders, but generally not the lower parts of the legs (i.e. below the elbow or stifle joints) are involved. Animals have difficulty in moving the swollen eyelids, with the result that the eyes are kept partly closed. Unfavourable prognostic signs are petechiae in the mucosa of the conjunctivae and mouth, particularly that of the ventral aspect of the tongue. These, if they occur, become evident shortly before death.
Some animals may repeatedly lie down or are restless when standing, and frequently paw the ground with their front feet as a result of severe colic which is often refractory to standard treatment with analgesics. In the ‘dikkop’ form the course of the disease is always more protracted and milder than in the ‘dunkop’ form, with a mortality rate of about 50 per cent. Death usually occurs within four to eight days of the onset of the febrile reaction.
Interference or difficulty in swallowing as a result of paralysis of the oesophagus may be a complication, particularly in those cases in which severe oedematous swellings involve the head.200 When the animal drinks, water containing particles of food may run out of the nose. Partly masticated food found in the manger or on the stable floor is also evidence of oesophageal paralysis. In severely affected animals the oesophagus becomes stuffed with food and acquires a visibly cord-like appearance. Affected animals lose weight rapidly and may die from foreign-body pneumonia.
Equine babesiosis or biliary fever is a common complication of AHS during recovery,25, 98 presumably because AHS lowers the resistance of animals carrying the parasite. In such cases icterus, anaemia, and constipation are evident, and the immediate cause of death may be babesiosis.
'Mixed' form
Although this is the most common form of AHS it is very rarely diagnosed as such clinically; one of the other preceding forms predominates and this is reflected in the diagnosis. It is only during necropsy when lesions of both the ‘pulmonary’ and ‘cardiac’ forms are observed that the diagnosis is made.
Horses affected by this form may show signs either of respiratory distress followed by oedematous swellings or, initially, of the ‘dikkop’ form before suddenly developing respiratory distress from which they may die. The mortality rate in horses affected by this form of the disease is approximately 70 per cent. Death usually follows three to six days after the onset of the febrile reaction.
Horse sickness fever
This form usually occurs in horses immune to one or more serotypes of AHSV which become infected with a heterologous serotype against which there is some cross-protection. It also occurs in species such as donkeys and zebras, which are resistant to the development of clinical disease.
Horse sickness fever is a very mild form of the disease and is frequently not diagnosed clinically. The most characteristic finding is a rise of the rectal temperature (39 to 40 °C) lasting one to six days followed by a drop in temperature to normal, and recovery. Some horses may show partial loss of appetite, congestion of the conjunctivae, slightly laboured breathing, and increased heart rate, but these signs are transient. Mild subclinical cases of horse sickness fever have been documented in vaccinated horses in South Africa which were subsequently infected with field strains of AHSV215 and in outbreaks of AHS in the Western Cape Province of South Africa associated with genome reassortment and reversion to virulence of live, attenuated vaccine viruses.87, 213
Pathology
Macroscopic pathology
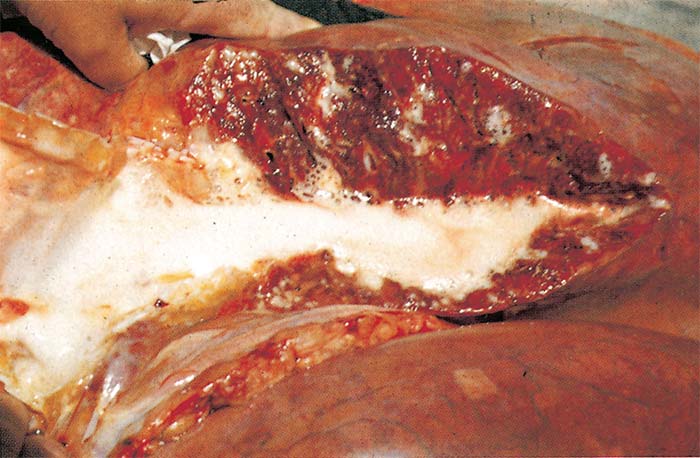
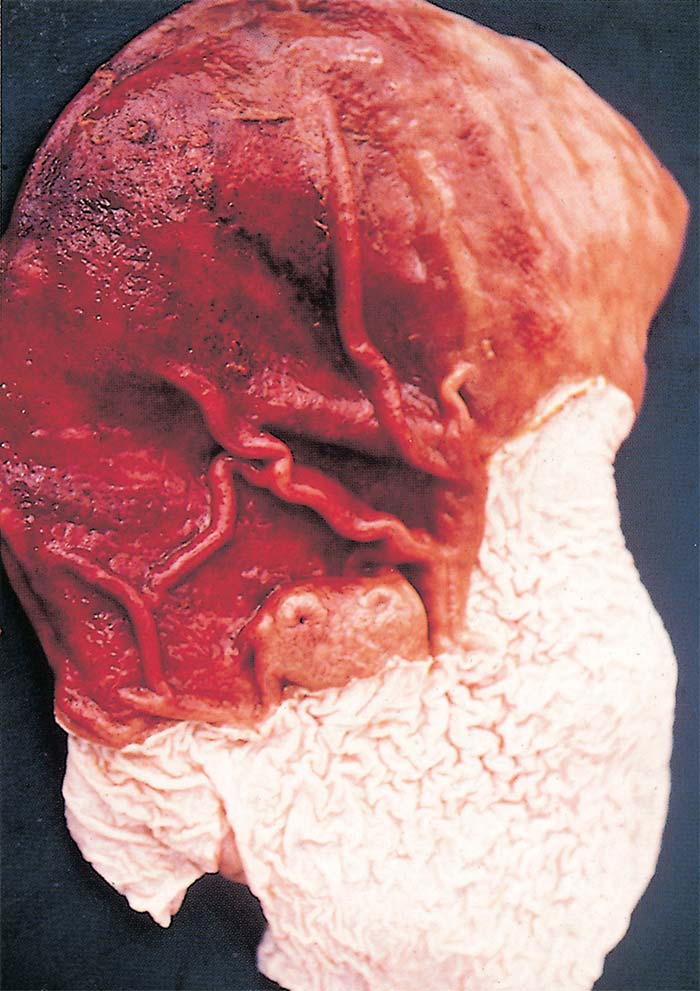
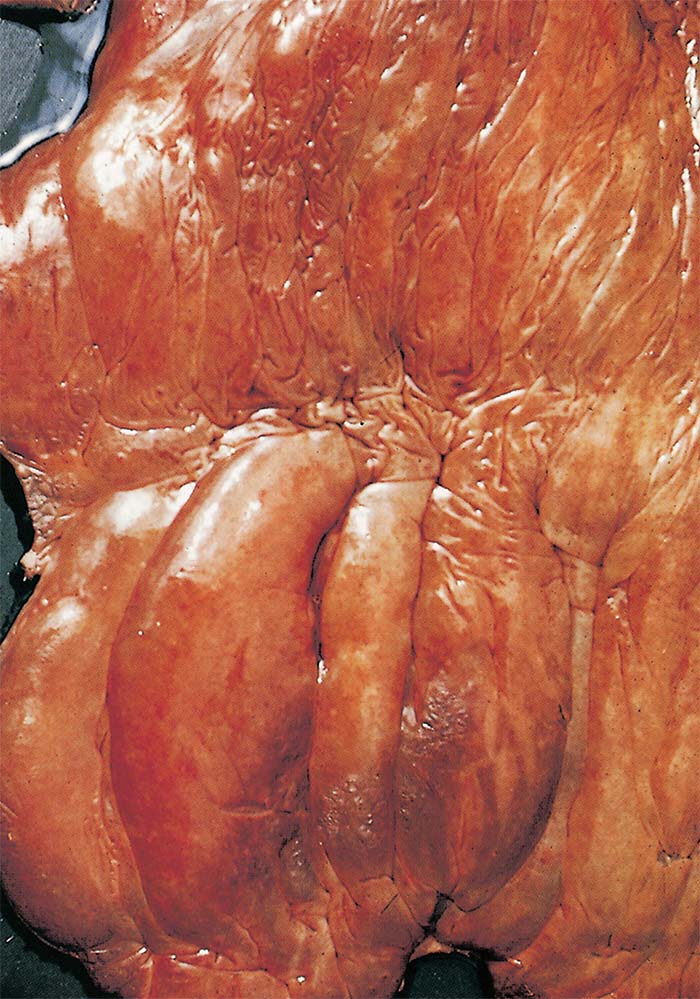
'Dunkop' or 'pulmonary' form: The most striking changes are severe oedema of the lungs (Figures 5-8) and hydrothorax. Several litres of pale yellow fluid, which may coagulate on exposure to air are found in the thoracic cavity. Oedema of the subcutaneous and intermuscular tissues is usually absent or mild.
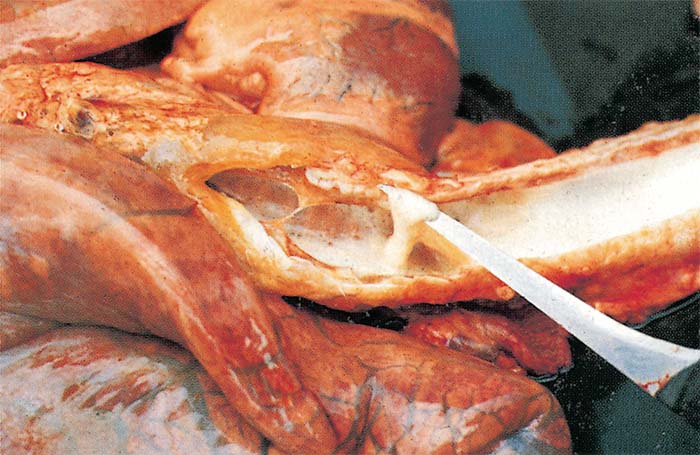
The mediastinum, and often the loose connective tissue around major blood vessels in the thorax, oesophagus, trachea and thymus, may also be oedematous. As a result of oedema, the lungs are much heavier than normal, do not collapse on opening the thoracic cavity, the interlobular septa often contain a gelatinous fluid (Figures 6 and 7), and, on the cut surface, copious amounts of serofibrinous fluid ooze from them. Often the impression is gained that the animal has drowned because of the copious amount of fluid in the lungs. The trachea and bronchi usually contain large amounts of froth and serofibrinous fluid which may be gelatinous (Figure 8). In some cases, the diameter of the trachea may be reduced as a result of severe gelatinous oedema of its mucosa. Petechiae and ecchymoses are sometimes present in the mucosa of the trachea and the parietal and visceral pleura.
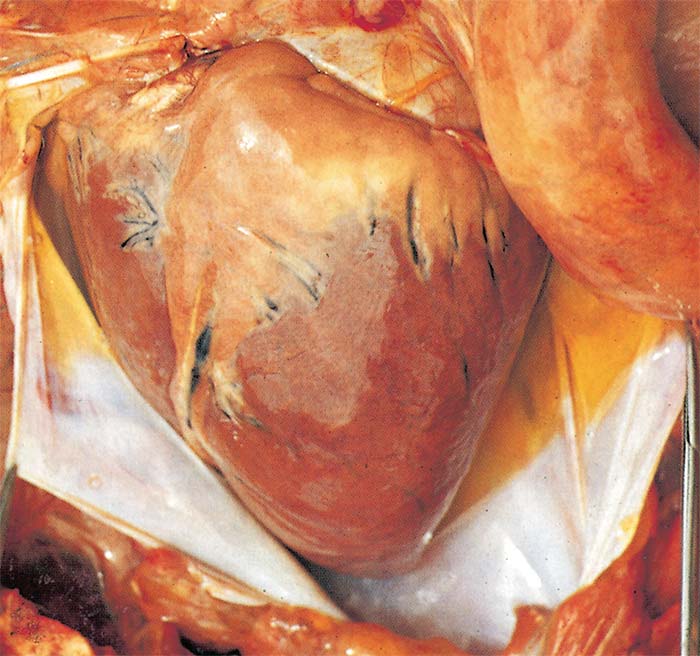
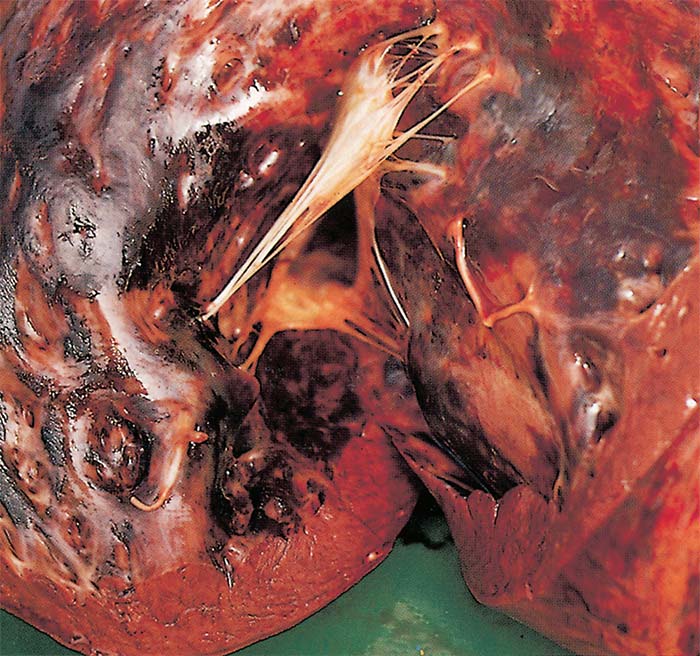
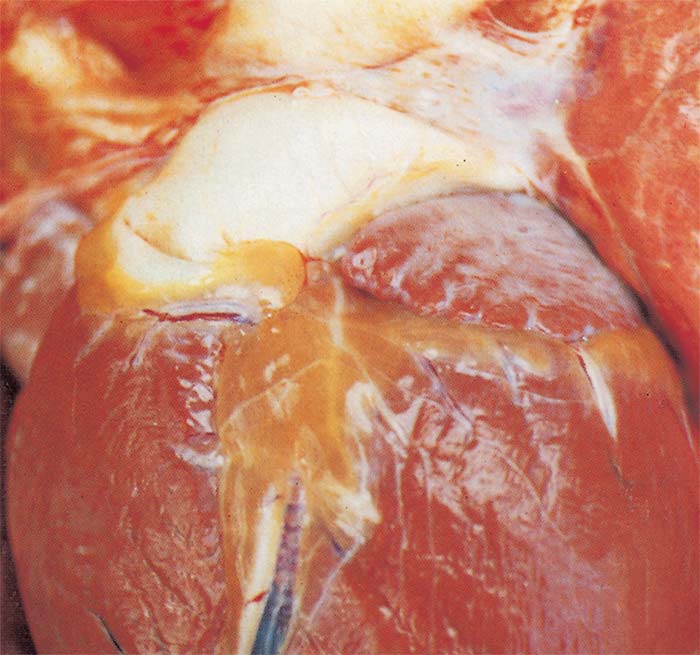
Hydropericardium is rare in this form of the disease (Figure 9). Epi- and endocardial haemorrhages of varying size (particularly severe in the left ventricle) (Figure 10), sometimes accompanied by slight oedema of the fat in the coronary grooves (Figure 11) and of the atrioventricular valves (Figure 12), may be evident.
All the lymph nodes are slightly to moderately enlarged, but the bronchial and mediastinal nodes are most severely swollen and oedematous. In most cases the spleen is normal in size or slightly enlarged, the white pulp is more prominent than normal, and the capsule may contain haemorrhages of varying sizes.
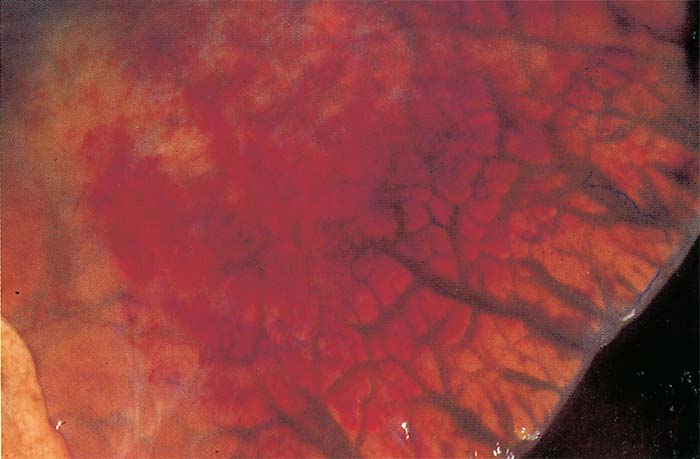
Marked diffuse congestion of the mucosa of the glandular part of the stomach is a consistent finding (Figure 13) This may be accompanied by patchy congestion and petechiation of the serosa and, sometimes, of the mucosa of the intestine. The liver may be slightly enlarged and congested, and its lobulation slightly more distinct than normal. There is usually some degree of ascites.
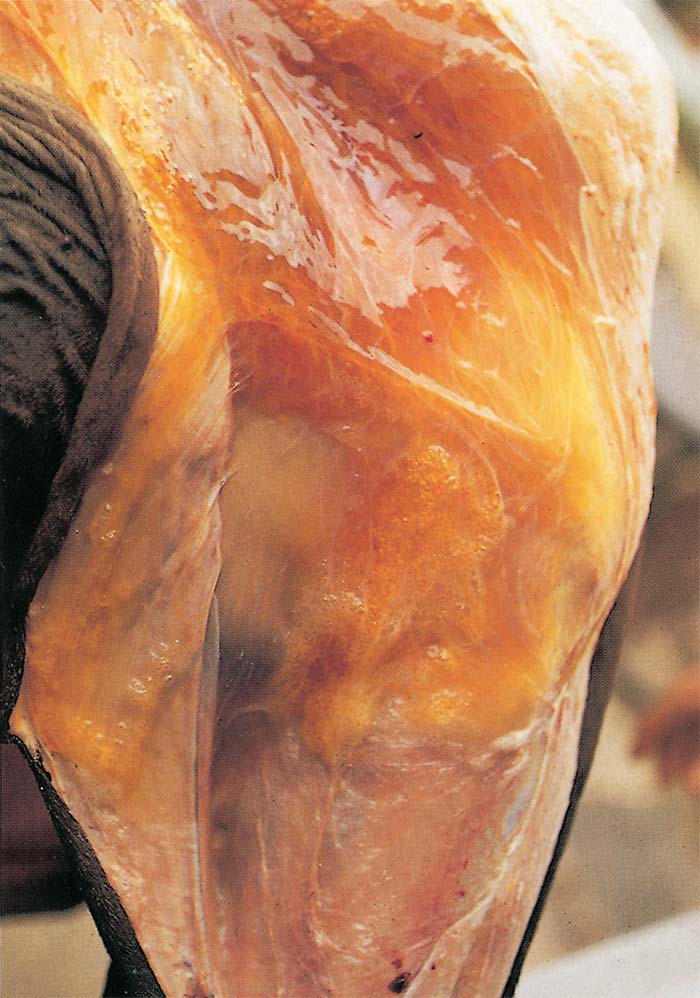
‘Dikkop’ or ‘cardiac’ form: The most characteristic change in this form of AHS is the presence of distinctly yellowish gelatinous oedema of the subcutaneous and intermuscular connective tissues (Figure 14) of the head and neck. In severe cases, this extends to the back, shoulders and chest. The oedema is particularly severe around the ligamentum nuchae. The eyelids, supraorbital fossae, lips, cheeks, tongue, and intermandibular space are commonly involved. The tongue may be severely swollen, cyanotic and have mucosal petechiae on its ventral surface.
Lesions similar to, but more severe than, those described for the ‘dunkop’ form are found in the heart. Very rarely pale greyish blanched areas of varying size may be noticeable in the myocardium. Severe hydropericardium is almost invariably present (Figure 9).
The lungs are usually normal or slightly congested, and only occasionally slightly oedematous. The thoracic cavity seldom contains an excess of fluid. Many lymph nodes are swollen and oedematous, but the spleen is only occasionally slightly enlarged. The liver is engorged and its lobulation is distinct. There is usually mild nephrosis.
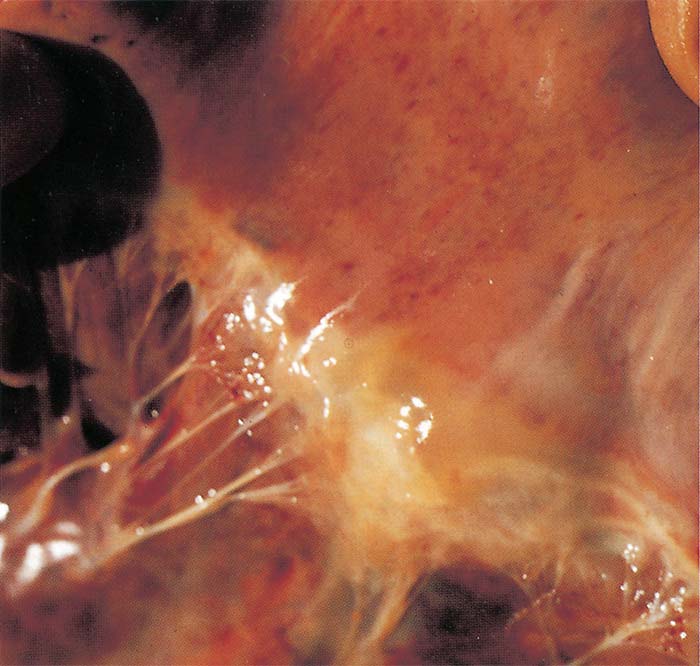
Lesions in the gastrointestinal tract are similar to but more severe than, those found in the ‘dunkop’ form. Moderate to severe oedema, congestion and petechiation of the mucosa of the caecum, colon and rectum are regular findings (Figure 15). In cases that manifest severe paralysis of the oesophagus, the organ is distended with a variable amount of compressed food.
'Mixed' form: Lesions made up of a mixture of both the ‘dunkop’ and ‘dikkop’ forms of AHS are present in animals that die of this form of the disease.
Microscopic pathology
There are few published reports on the histopathology of AHS.129, 139In horses with involvement of the lungs, copious protein-rich oedematous fluid is found in the alveoli and interstitial tissues. The amount of fibrin in the fluid ranges from a few strands to dense masses filling the alveolar spaces. A few macrophages with abundant eosinophilic cytoplasm occur in close association with the fibrin in some alveoli. Variable numbers of neutrophils are present in some areas of the lungs. Based on the presence of fibrin and inflammatory cells in the lungs it has been suggested that the pulmonary lesion should be classified as exudative pneumonia. 129, 139Alveolar capillaries and many arterioles and venules are congested and contain increased numbers of neutrophils, lymphocytes and monocytes. Perivasculitis, characterized by infiltration principally of lymphocytes but also of monocytes, macrophages and occasional neutrophils and plasma cells, in the adventitia are evident in the lungs.139 Separation of Type I pneumocytes from the alveolar wall is occasionally apparent.139
There are usually no microscopic lesions in the myocardium, but if present they vary considerably in distribution and extent of involvement.129 They may consist of small foci or very extensive areas which involve the entire thickness of the wall of the ventricles and atria and consist of degenerative and necrotic myocytes, haemorrhages, occasional fibrin strands between myocyte fibres, and increased numbers of neutrophils in the capillaries.116, 139Fully susceptible horses that have succumbed to disease caused by serotype 9 have been reported to show, in addition to areas of degeneration and necrosis of myocytes, extensive oedema and haemorrhages of the heart muscle accompanied by infiltration of some mononuclear cells, plasma cells, siderocytes and polymorphonuclear leukocytes.129 Lysis of necrotic myocytes and evidence of early fibroplasia may be evident.
The bronchiolar and intestinal lymphoid nodules, germinal centres of most lymph nodes, and white pulp of the spleen show varying degrees of lymphocyte depletion. Pyknosis and karyorrhexis occur in cells of the lymphoid series. The hepatocytes and the epithelium of convoluted tubules in the kidneys reveal cloudy swelling and hydropic degeneration.
Ultrastructural pathology
Separation of Type I pneumocytes from the alveolar walls is common.139 Newsholme139 found no injury to cells, including endothelial cells, in oedematous lungs, nor was there any evidence of virus or virus replication in the lungs, the bronchiole-associated lymphoid nodules, bronchial, mediastinal and peripheral lymph nodes or the spleen. 139 However, in another study, virus, as well as ultrastructural changes, were evident in endothelial cells in the lungs.85
Diagnosis
Clinical signs, history and macroscopical lesions are usually sufficient to suggest a diagnosis of AHS.167 A confirmatory diagnosis can be obtained by identification of the virus or in naïve populations, specific antibodies.20A very important aspect of the diagnosis is the selection of samples and their safe transportation to the laboratory.
Traditionally, viral isolation was considered the gold standard for diagnosis. The virus can be isolated from blood collected, preferably in heparin, during the febrile stage of the disease or from specimens of the lungs, spleen and lymph nodes collected at necropsy and kept at 4 °C.20, 75 Virus isolation can be performed by the inoculation of a variety of cell cultures e.g. baby hamster kidney-21, Vero, monkey stable (see Aetiology), by the intracerebral inoculation of suckling mice or by the intravenous inoculation of embryonated hens’ eggs.20, 167 Blood collected in chelating agents should be diluted five- to ten-fold to prevent detachment of the monolayer of cell cultures.
Alternative group-specific tests to identify AHSV are the complement fixation (CF) test,132 agar gel immunodiffusion (AGID), direct and indirect immunofluorescence (IFA),54 microsphere-based immunoassay,182, 183 lateral flow assay,51 rapid capture (indirect sandwich) enzyme-linked immunosorbent assay (ELISA),176 using either polyclonal antibodies (PAbs) or monoclonal antibodies (MAbs), and sandwich ELISAs that detect viral antigen in mammalian and insect tissue homogenates as well as in cell culture supernatant fluid.59, 60, 94 Genomic probes have been developed for in situ hybridization in tissues.46 Immunohistochemical staining methods have also been used successfully to determine the localization of AHSV antigen within various tissues.217, 218
The reverse transcription-polymerase chain reaction (RT-PCR) assay is a highly sensitive technique that provides rapid identification of AHSV viral nucleic acid in the blood and other tissues of infected animals. The latter has greatly improved the laboratory diagnosis of AHS by increasing the sensitivity of detection and shortening the time required for the diagnosis. Several agarose gel-based RT-PCR assays for the specific detection of AHSV RNA have been described targeted at viral segments 3, 5, 7, 8 or 10.22, 45, 110, 137, 180, 190, 220, 221 A widely used method employs primers that target the terminal ends of RNA segment 7 (coding for VP7) amplifying the complete viral segment.220 Real-time RT-PCR methods for the highly sensitive and specific detection of AHSV RNA have been developed based on the use of a pair of primers and a labelled probe from conserved sequences of viral segments 3, 5 or 7.1, 28, 80, 171 A loop-mediated isothermal amplification assay has been developed that is suitable for use in the field.83 A duplex real-time RT-PCR has also been described that targets segments 7 and 8 of the genome (coding for VP7 and NS2 respectively).166In 2015 the OIE Reference Laboratories for AHS carried out an international ring trial to gather information on the performance of the different methods used in the main AHSV diagnostic laboratories. Ten different RT-PCR protocols were evaluated. The study identified that the real-time RT-PCR methods of Agüero et al. (2008)1 and Guthrie et al. (2013)89 correctly detected all the representative strains included in the international ring trial with high sensitivity in the analysis of field samples.
Virus typing is achieved with a plaque inhibition neutralization test using type-specific antisera.20, 102, 133, 208 Electrical insulating fish-spine beads filled with type-specific antiserum (produced in sheep) are used to indicate virus-antibody neutralization on Vero cell monolayers inoculated with the test sample.167 The development of type-specific gel-based RT-PCR120, 178, 205 and real-time RT-PCR28, 109, 214 assays targeting the VP2 gene encoding the neutralizing epitopes of AHSV has significantly increased the speed of the identification of the nine serotypes of AHSV.
Techniques have been developed which allow full-length sequences of viral dsRNA genomes to be obtained.84, 88, 121, 164, 165, 213 This technology can be used to investigate and reveal viral segment reassortments. The information gained from these sequences can be used to investigate viral attributes such as viral tropism and virulence. It can also be used to study viral evolution of outbreaks in the field, assessing attenuation and adaptation to cell culture.165
Classical virology techniques such as viral isolation remain the only tests able to provide a measure of the amount of infectious virus in a sample. On the other hand, these techniques are time-consuming: virus neutralization tests for virus typing usually takes a minimum of two weeks to obtain a result and this time constraint can be costly in the face of an outbreak situation, where a rapid diagnosis is needed. Molecular assays can be performed rapidly and address this concern, but these assays need to target conserved regions of the genome and the genetic drift of the AHSV genome, in particular the VP2 coding region, can make the reliable detection of all genetic variants within each serotype difficult. Although molecular methods can rapidly type AHSV, classical virology techniques still play a vital role and provide a vital backup to serotyping AHSV isolates.
The advantages of molecular assays are that the procedures can be used on specimens that do not contain live viruses,1 that contain attenuated strains or strains of low virulence179 and can be used for earlier detection of viraemia than viral isolation.190, 191 The RT-PCR procedure will detect virus-specific nucleic acid after the virus is no longer viable and capable of establishing a new infection in either insects or mammalian cells. Therefore, positive results do not necessarily indicate the presence of an infectious virus.
Real-time RT-PCR provides advantages over agarose gel-based RT-PCR methods, with faster analysis times, higher sensitivity and suitability for high-throughput automation. Nevertheless, gel-based RT-PCR methods, particularly those amplifying long RNA fragments,110, 220 can be very useful in the further genetic characterization of the virus by sequencing of the amplicons. In addition, it may be beneficial in laboratories without the capacity to perform real-time RT-PCR.
Antibody to AHSV can be detected by utilizing CF,20, 37, 100 AGID,20, 100 IFA,54, 100 virus neutralization (VN) 20, 37, 100 and ELISA tests.20 In horses that have recovered from the disease, high CF antibody titres indicate infection with the virus within the previous few months.37, 104Antibodies detectable by VN tests and ELISA persist for several years, making these tests unsuitable for diagnostic investigations in endemic areas where there is circulating field virus and routine vaccination, as previous exposure is likely. Furthermore, serological assays have a limited value for the diagnosis of acute infection, as many horses may die of AHS before a significant antibody response is noted.172, 179
If possible, more than one test should be performed to diagnose an outbreak of AHS, especially the index case. The initial test can be a quick test such as ELISA or PCR, followed by virus isolation in tissue culture. Virus neutralization for serotype identification, type-specific RT-PCR or sequencing should be performed as early in the outbreak as possible so that the serotype can be identified, and the correct vaccine selected.
At present, there are no international standards for viruses or diagnostic reagents, and there is no standard methodology for the identification of AHSV. However, a viral and antibody panel has been evaluated, and comparative studies between different ELISAs for AHSV antigen and antibody determination have been carried out in different laboratories, including in the European Union (EU) Reference Laboratory for AHS. The results have demonstrated a high level of correlation for both antigen and antibody determination with an in-house test and commercial kits. Similar studies have been conducted with several RT-PCR assays also providing a high level of correlation. Further information on comparative studies of different test methods and kits is available from the OIE Reference Laboratories for AHS.
Differential diagnosis
It is not possible to differentiate the horse sickness fever form of the disease from the early febrile stages of other forms of AHS or the early febrile stages of many other equine infectious diseases.
The clinical signs and lesions of AHS may be confused with those of equine encephalosis (see Equine encephalosis). As many of the epidemiological features of the two diseases are similar, they may occur simultaneously in South Africa and possibly also in other regions in southern Africa. Horses manifesting swelling of the eyelids, supraorbital fossae or of the entire head as a result of equine encephalosis cannot be differentiated clinically from the ‘dikkop’ form of AHS, and can occasionally be severe enough to cause death. However, the mortality rate of AHS is much higher than that of equine encephalosis.
Although the lesions of AHS at necropsy are fairly characteristic, the disseminated small haemorrhages and oedema found in some parts of the body that are associated with the ‘dikkop’ form of AHS may be very similar to those found in cases of purpura haemorrhagica and equine viral arteritis. In horses affected by these conditions, subcutaneous oedema often occurs ventral to the elbow and stifle joints, as well as in other locations while in those suffering from AHS it does not occur below these joints. In purpura haemorrhagica, oedema and haemorrhages tend to be more numerous, widespread and severe than in AHS.
The early stages of biliary fever, when Babesia parasites may be difficult to demonstrate in blood smears, are occasionally confused with AHS. African horse sickness may also be complicated by babesiosis, and in such cases, ventral oedema may be severe. Any cause of sudden death can be confused with AHS peracute disease, and even other causes of vasculitis induced respiratory disease such as equid herpesvirus infections can be a differential diagnosis..
Control
Preventative/control strategies, should be aimed at breaking the transmission cycle. This can be done by reducing contact of horses with Culicoides vectors, preventing or controlling the movement of viraemic horses, and immunization of horses.
In endemic countries immunization remains the most effective preventative measure available. All AHSV serotypes are present in South Africa and most parts of sub-Saharan Africa, although their temporal distribution may differ. The use of a polyvalent vaccine is, therefore, necessary to protect horses in these areas. Currently, the control of AHSV in endemic African countries relies on a polyvalent live attenuated vaccine (LAV) administering seven serotypes in two doses; AHSV-5 and AHSV-9 are not included in the vaccine since cross-protection with serotypes 8 and 6 respectively has been documented.209, 213
Several methods of immunization were attempted before 1930 with varying success.62, 63, 64, 65, 68, 82, 196, 197, 198, 201 Since the demonstration in the early 1930s that AHSV can be attenuated by the serial intracerebral passage in mice, the immunization of horses against the disease has been greatly simplified and improved.9, 11, 12, 16By exploiting these findings Alexander, Neitz, and Du Toit15 showed in 1936 that a highly effective attenuated vaccine could be produced. Nowadays large plaque variants of AHSV are selected as candidate vaccine strains.20, 61
In endemic areas, and in regions where AHS occurs almost every year, viz. most parts of Africa south of the Sahara, annual vaccination of horses is the most practical means of control. Although prophylactic immunization against AHS is a very efficient method of preventing serious losses, it cannot be relied upon fully to protect horses against infection or disease. However, horses that have received three or more courses of immunization are usually well protected against the disease. In South Africa, annual immunization with a live polyvalent attenuated vaccine in the low vector activity season of June to October (winter to spring), which is some time before the peak AHS season, is advocated and allows immunized animals to respond adequately to the vaccine before possibly being challenged by natural exposure. Onderstepoort Biological Products currently produces a polyvalent vaccine containing attenuated strains prepared in two components: trivalent (serotypes 1, 3 and 4) and quadrivalent (serotypes 2, 6, 7 and 8).61 A course of immunization consists of the administration of these component vaccines a minimum of three weeks apart. Serotypes 5 and 9 are not included in the vaccines as serotypes 8 and 6, respectively, afford adequate cross-protection.
Generally, immunization has only limited side effects. A temperature response may ensue between 5 and 13 days after inoculation because of a low-level virus replication in immunized animals, which may progress to mild swelling of the supraorbital fossae. Horses should not be excessively exerted for about three weeks after the first course of immunization. These animals could also be a potential source of vaccine virus that could be transmitted by Culicoides midges and therefore vector control should be applied to these cases routinely. Cases of fatal encephalitis, characterized by blindness and neurological disorders, in fully susceptible horses and donkeys six to eight weeks after initial vaccination with neurotropic mouse strains of the virus, have occasionally been reported.72, 106, 142, 148, 152, 159, 168 Serological evidence has been found of accidental aerosol infection of four humans who were working with vaccine strains attenuated by the passage in mice brains (particularly serotypes 1 and 6) in the vaccine packing section of Onderstepoort Biological Products.192 They suffered from nonfatal encephalitis and chorioretinitis, which resulted in permanent partial loss of vision or blindness.
Immunity to live attenuated AHSV strains is solid against the homologous virus and probably lasts indefinitely.131 Cross-immunity between serotypes may be enhanced by repeated inoculation of the same virus.9, 10 There is evidence that animals that recover from infection with a field virus acquire a broader cross-immunity to the other serotypes than that which is obtained from immunization.38 The simultaneous inoculation of several serotypes of attenuated AHSVs in horses usually results in the production of antibodies against each serotype, although the response of individual horses may vary, and in some animals, antibodies against one or more of the serotypes inoculated may not be detectable by VN tests.14, 38This is possibly because of interference between viruses in the polyvalent vaccine or over attenuation of vaccine strains.76, 133For these reasons annual vaccination of horses with the polyvalent vaccine is advocated in high-risk areas in Africa to ensure the production of the widest possible polyvalent immunity to the different serotypes. During outbreaks of AHS in Spain, about 10 per cent of animals (horses, mules and donkeys) immunized for the first time with an attenuated monovalent AHSV serotype 4 vaccine failed to seroconvert. However, at least some animals that fail to respond serologically are resistant to challenge infection.93
In most instances, the levels of antibody acquired by foals from colostrum correlate well with the levels of antibody in the sera of their dams and determine the duration of their passive immunity.14, 38, 52Because of the passive immunity acquired by foals born to immune mares, it is generally recommended that foals should not be immunized before they are six months of age. However, in foals that acquire low levels of antibody to one or more AHSV serotypes via the colostrum, neutralizing antibodies to individual serotypes may decline to undetectable levels two to four months after birth, suggesting that foals could become susceptible to infection well before the age of six months, which is the age commonly recommended for initial vaccination.38 As a result of the restricted breeding season of Thoroughbreds, most foals in southern Africa are three to five months of age by late February when possible challenge with AHSV can be expected to be highest. It is therefore recommended in Zimbabwe that foals should be immunized in January and again in September.38 Animals should be vaccinated twice in their first and second years of life and thereafter only once annually to avoid unresponsiveness.38
Recombinant DNA technology has recently been applied to AHSV in an attempt to produce subunit vaccines.61, 175 Preliminary results from these studies suggest that it may be necessary to include multiple viral proteins in such vaccines to stimulate protective immunity in horses.61
The monovalent, attenuated AHSV-9 vaccine (National Laboratory, Senegal) is used extensively in West Africa, because, before the appearance of serotypes 2 and 7 in 2007, AHSV-9 was the only serotype known to be circulating in this region. Monovalent vaccination (against serotype 9 or 4) has also been used successfully in combination with other control measures to eradicate the virus during outbreaks in the incursive areas, e.g. the 1959 to1961 outbreaks in the Middle East, the 1955 and 1956 outbreaks in North Africa and Spain, and the 1987 to 1991 outbreaks in Spain, Portugal and Morocco.116
Despite the evident success of these live attenuated vaccines in controlling endemic transmissions, there are still concerns about their use in epidemic situations in non-endemic countries, namely:
- the seed virus strains used in vaccines may be introduced into non-endemic regions,
- live virus vaccines may cause teratogenic effects and are therefore not recommended for use in pregnant mares,
- attenuated virus vaccines are likely to result in low-level viraemia in some vaccinated equids and may lead to infection of Culicoides midges,
- gene reassortment may occur between live attenuated vaccine viruses and wild-type viruses, in either the vertebrate or invertebrate hosts that leads to the establishment of new genetic variants209, 210, 213, 215 that have different or even enhanced virulence characteristics.
Consequently, efforts should be made to develop safer vaccines, such as inactivated, sub-unit or recombinant vaccines. An inactivated vaccine was used to successfully combat the 1987 to1991 AHS epidemics in Spain and Portugal.101 Clinical trials in France and field trials in Spain and Morocco showed that the inactivated vaccine was effective against AHSV transmission. However, once the epidemic had ended, the vaccine was withdrawn, and there are currently no inactivated vaccines on the market. Although inactivated vaccines may be effective, they are expensive to produce and sometimes multiple inoculations are needed to elicit high levels of protective immunity. Consequently, efforts are made to develop improved and less expensive inactivated vaccines. To address the need for safe and more effective vaccines, several candidates have been evaluated including subunit vaccines, virus-like particles (VLPs), avian reovirus muNS protein microspheres (MS), recombinant poxviruses and reverse genetic approaches.3, 5, 49, 50, 56, 57, 58, 70, 90, 107, 117, 122, 125, 126, 128, 173, 174, 177, 203 Promising vaccine candidates are under development that are based on the administration of a poxvirus vector expressing VP2 protein, alone or in combination with VP5,5, 90 and recombinant capsid proteins, e.g. VP2 alone, or in association with VP5 and VP7, that self-assemble into immunogenic virus-like particles.107, 175 These vaccine candidates have yet to be tested in horses, but their immunogenicity and safety are satisfactory in mammal models (e.g. guinea pigs).
As Culicoides midges are mostly crepuscular, infection of susceptible horses can be prevented to a large degree by stabling them during the hours that are high risk for vector activity, namely, some hours before sunset and letting them out a few hours after sunrise, (see Vectors: Culicoides spp.). Although Culicoides imicola, the most common vector responsible for AHSV transmission, does not readily enter buildings, it should be noted that there are other species such as C.bolitinos, which may, and therefore stable design can be optimised with the use of shade cloth to close all openings for vector protection. This can be further protected by impregnation of the mesh with pyrethroid insecticides and the use of insecticides on the walls of the structure. Vector protected stabling can also be utilized in outbreak areas for the isolation of infected horses to reduce the transmission of the virus to the vector population, as well as isolation of vaccinated animals if they are vaccinated with live attenuated vaccines in the face of high vector activity.
The application of insect repellents, especially those containing diethyltoluamide (DEET) and the use of pyrethroid containing insecticides on animals’ coats will also discourage Culicoides from feeding on them.96, 157
Following an outbreak of AHS in a country that has previously been free of the disease, attempts should be made to limit further transmission of the virus and to achieve eradication as soon as possible. Control measures must be instituted as soon as a suspected diagnosis of AHS has been made without waiting for the diagnosis to be confirmed. In epidemic situations, the following measures should be taken:21
- delineate the area of infection, taking into consideration geographical borders such as mountains and rivers;
- movement control should be implemented, and the use of antigen detection methods of diagnosis can be used for risk mitigation as part of movement control protocols;
- equids should be stabled, preferably with vector protection of the stables as far as possible, at least from dusk to dawn. Insect control methods should also be implemented;
- the rectal temperatures of all horses, mules and donkeys should be recorded twice a day to detect infected animals as early as possible as overt disease is generally preceded by viraemia for about three days. Vector protection methods as described above should be implemented for animals with a fever to prevent spread of the disease;
- in the event of an epidemic, emergency vaccination protocols should be considered carefully, taking into account the source of the virus, the serotype circulating, and the susceptibility of the equid population involved. All vaccination animals should be positively identified.
- the virus type should be identified as soon as possible as this will be pivotal in the decisions needed around vaccination protocols. The decision on whether to use a monovalent or polyvalent vaccine will be dependent on whether one or more serotypes are circulating, as well as what is commercially available on a large scale at short notice.
- the World Health Organization for Animal Health should be notified immediately of the outbreak of disease.
Treatment revolves around mainly supportive therapy including nonsteroidal drugs, cardiac inotropes, ACE inhibitors and diuretics. Affected animals should be carefully nursed, well-fed and given rest as the slightest exertion may result in death. After recovery, they should be rested for at least four weeks before being returned to light work. As babesiosis may be a complication of AHS, blood smears, as well as the rectal temperature (to detect a secondary febrile reaction), should be taken regularly and, if the smears are found to be positive, animals should be appropriately treated. Careful massaging of the oesophagus in those cases in which it is paralysed may result in a gradual improvement of the condition.
References
- AGUERO, M., GOMEZ-TEJEDOR, C., ANGELES CUBILLO, M. & RUBIO, C., 2008. Real-time fluorogenic reverse transcription polymerase chain reaction assay for detection of African horse sickness virus. Journal of Veterinary Diagnostic Investigation, 20, 325-328.
- AKLILU, N., BATTEN, C., GELAYE, E. & JENBERIE, S., 2012. African horse sickness outbreaks caused by multiple virus types in Ethiopia. Transboundary and Emerging Diseases, 61, 2.
- AKSULAR, M., CALVO-PINILLA, E., MARIN-LOPEZ, A., ORTEGO, J., CHAMBERS, A. C., KING, L. A. & CASTILLO-OLIVARES, J., 2018. A single dose of African horse sickness virus (AHSV) VP2 based vaccines provides complete clinical protection in a mouse model. Vaccine, 36, 7003-7010.
- AL-GHAMDI, G. M., 2017. African horse sickness: a possible cause of horse fatalities in Al-Baha, southwestern of the Kingdom of Saudi Arabia. International Journal of Current Advanced Research, 6, 4713-4714.
- ALBERCA, B., BACHANEK-BANKOWSKA, K., CABANA, M., CALVO-PINILLA, E., VIAPLANA, E., FROST, L., GUBBINS, S., URNIZA, A., MERTENS, P. & CASTILLO-OLIVARES, J., 2014. Vaccination of horses with a recombinant modified vaccinia Ankara virus (MVA) expressing African horse sickness (AHS) virus major capsid protein VP2 provides complete clinical protection against challenge. Vaccine, 32, 3670-3674.
- ALEXANDER, K. A., KAT, P. W., HOUSE, J., HOUSE, C., O'BRIEN, S. J., LAURENSON, M. K., MCNUTT, J. W. & OSBURN, B. I., 1995. African horse sickness and African carnivores. Vet Microbiol, 47, 133-140.
- ALEXANDER, K. A., KAT, P. W., HOUSE, J., HOUSE, C., O'BRIEN, S. J., LAURENSON, M. K., MCNUTT, J. W. & OSBURN, B. I., 1995. African horse sickness and African carnivores. Veterinary Microbiology, 47, 133-140.
- ALEXANDER, R. A., 1933. Preliminary note on the infection of white mice and guinea pigs with the virus of horsesickness. Journal of the South African Veterinary Association, 4, 1-9.
- ALEXANDER, R. A., 1935. Studies on the neurotropic virus of horsesickness. II. Some physical and chemical properties. Onderstepoort Journal of Veterinary Science and Animal Industry, 4, 323-348.
- ALEXANDER, R. A., 1935. Studies on the neurotropic virus of horsesickness. III. The intracerebral protection test and its application to the study of immunity. Onderstepoort Journal of Veterinary Science and Animal Industry, 4, 349-377.
- ALEXANDER, R. A., 1938. Studies on the neurotropic virus of horsesickness. VI. Propagation in the developing chick embryo. Onderstepoort Journal of Veterinary Science and Animal Industry, 11, 9-19.
- ALEXANDER, R. A., 1948. The 1944 epizootic of horsesickness in the Middle East. Onderstepoort Journal of Veterinary Science and Animal Industry, 23, 77-92.
- ALEXANDER, R. A. & DU TOIT, P. J., 1934. The immunization of horses and mules against horsesickness by means of the neurotropic virus of mice and guinea pigs. Onderstepoort Journal of Veterinary Science and Animal Industry, 2, 375-391.
- ALEXANDER, R. A. & MASON, J. H., 1941. Studies on the neurotropic virus of horsesickness. VII. Transmitted immunity. Onderstepoort Journal of Veterinary Science and Animal Industry, 16, 19-32.
- ALEXANDER, R. A., NEITZ, W. O. & DU TOIT, P. J., 1936. Horse sickness: Immunization of horses and mules in the field during the season 1934-1935 with a description of the technique of preparation of polyvalent mouse neurotropic vaccine. Onderstepoort Journal of Veterinary Science and Animal Industry, 7, 17-30.
- ALEXANDER, R. A. & VAN DER VYFER, B., 1935. Horse sickness field experiments with neurotropic vaccine during the season 1933-1934. Journal of the South African Veterinary Association, 6, 33-38.
- ANDERSON, E. C., MELLOR, P. & HAMBLIN, C., 1989. African horse sickness in Saudi Arabia. Veterinary Record, 125, 489-489.
- ANONYMOUS., African horse sickness situation update: Equine Veterinary Association of Thailand.
- ANONYMOUS., 1997. 97/10/EC: Commission Decision of 12 December 1996 amending Council Decision 79/542/EEC and Commission Decisions 92/160/EEC, 92/260/EEC and 93/197/EEC in relation to the temporary admission and imports into the Community of registered horses from South Africa. Official Journal, L003, 9-24.
- ANONYMOUS., 2019. African horse sickness (Infection with African horse sickness virus). Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. World Organisation for Animal Health. Paris.
- ANONYMOUS., 2020. Infection with African horse sickness virus. Terrestrial Animal Health Code. World Organisation for Animal Health. Paris.
- ARADAIB, I. E., MOHEMMED, M. E., SARR, J. A., IDRIS, S. H., ALI, N. O. M., MAJID, A. A. & KARRAR, A. E., 2006. Short communication: a simple and rapid method for detection of African horse sickness virus serogroup in cell cultures using rt-PCR. Veterinary Research Communications, 30, 319-324.
- AWAD, F. I., AMIN, M. M., SALAMA, S. A. & ALY, M. M., 1981. The incidence of African horse sickness antibodies in animals of various species in Egypt. Bull Anim Health Prod Afr, 29, 285-287.
- AWAD, F. I., AMIN, M. M., SALAMA, S. A. & ALY, M. M., 1981. The incidence of African horse sickness antibodies in animals of various species in Egypt. Bulletin of Animal Health and Production in Africa, 29, 285-287.
- AWAD, F. I., AMIN, M. M., SALAMA, S. A. & KHIDE, S., 1981. The role played by Hyalomma dromedarii in the transmission of African horsesickness virus in Egypt. Bulletin of Animal Health and Production in Africa, 29E, 337-340.
- BABA, S. S., AKINYELE, H. A. & OLALEYE, O. D., 1992. Prevalence of complement-fixing antibody to the African horse sickness virus in domestic animals in Nigeria. Beitrage zur tropischen Landwirtschaft und Veterinarmedizin, 30, 471-477.
- BABA, S. S., OLALEYE, O. D. & AYANBADEJO, O. A., 1993. Haemagglutination-inhibiting antibodies against African horse sickness virus in domestic animals in Nigeria. Veterinary Research, 24, 483-487.
- BACHANEK-BANKOWSKA, K., MAAN, S., CASTILLO-OLIVARES, J., MANNING, N. M., MAAN, N. S., POTGIETER, A. C., DI NARDO, A., SUTTON, G., BATTEN, C. & MERTENS, P. P. C., 2014. Real time RT-PCR assays for detection and typing of African horse sickness virus. PLoS One, 9, e93758.
- BARNARD, B. J. H., 1993. Circulation of African horsesickness virus in zebra (Equus burchelli) in the Kruger National Park, South Africa, as measured by the prevalence of type specific antibodies. Onderstepoort Journal of Veterinary Research, 60, 111-117.
- BARNARD, B. J. H., 1997. Antibodies against some viruses of domestic animals in southern African wild animals. Onderstepoort Journal of Veterinary Research, 64, 95-110.
- BARNARD, B. J. H., 1998. Epidemiology of African horse sickness and the role of the zebra in South Africa. Archives of Virology: Supplementa, 14, 13-19.
- BAYLEY, T. B., 1856. Notes on the Horse-sickness at the Cape of Good Hope, in 1854-1855. Cape Town: Saul Solomon & Co.
- BELL, R. A., 1999. Outbreak of African horse sickness in the Cape Province of South Africa. Veterinary Record, 144, 483.
- BEST, J. R., ABEGUNDE, A. & TAYLOR, W. P., 1975. An outbreak of African horse sickness in Nigeria. Veterinary Record, 97, 394.
- BEVAN, L. E. W., 1911. The transmission of African horsesickness to the dog by feeding. Veterinary Journal, 67, 402-408.
- BINEPAL, V. S., WARIRU, B. N., DAVIES, F. G., SOI, R. & OLUBAYO, R., 1992. An attempt to define the host range for African horse sickness virus (Orbivirus, Reoviridae) in East Africa, by a serological survey in some Equidae, Camelidae, Loxodontidae and Carnivore. Veterinary Microbiology, 31, 19-23.
- BLACKBURN, N. K. & SWANEPOEL, R., 1988. African horse sickness in Zimbabwe: 1972 to 1981. Tropical Animal Health and Production, 20, 169-176.
- BLACKBURN, N. K. & SWANEPOEL, R., 1988. Observations on antibody levels associated with active and passive immunity to African horse sickness. Tropical Animal Health and Production, 20, 203-210.
- BOORMAN, J., MELLOR, P. S., PENN, M. & JENNINGS, M., 1975. The growth of African horse-sickness virus in embryonated hen eggs and the transmission of virus by Culicoides variipennis Coquillet (Diptera, Ceratopogonidae). Archives of Virology, 47, 343-349.
- BOSMAN, P., BRUCKNER, G. K. & FAUL, A., 1995. African horse sickness surveillance systems and regionalization/zoning: the case of South Africa. Revue scientifique et technique (International Office of Epizootics), 14, 645-653.
- BOURDIN, P., 1973. Ecology of African horse sickness. In: KARGER S., (ed.). Proceedings of the Third International Conference of Equine Infectious Diseases. Basel.
- BREESE, S. S., OZAWA, Y. & DARDIRI, A. H., 1969. Electron microscopic characterization of African horse-sickness virus. Journal of the American Veterinary Medical Association, 155, 391-400.
- BREMER, C. W., 1976. A gel electrophoretic study of the protein and nucleic acid components of African horse sickness virus. Onderstepoort Journal of Veterinary Research, 43, 193-200.
- BREMER, C. W., HUISMANS, H. & VAN DIJK, A. A., 1990. Characterization and cloning of the African horse sickness virus genome. Journal of General Virology, 71, 793-799.
- BREMER, C. W. & VILJOEN, G. J., 1998. Detection of African horse sickness virus and discrimination between two equine orbivirus serogroups by reverse transcription polymerase chain reaction. Onderstepoort Journal of Veterinary Research, 65, 1-8.
- BROWN, C. C., MEYER, R. F. & GRUBMAN, M. J., 1994. Presence of African horse sickness virus in equine tissues, as determined by in situ hybridization. Veterinary Pathology, 31, 689-694.
- CALISHER, C. H. & MERTENS, P. P., 1998. Taxonomy of African horse sickness viruses. Archives of Virology Supplement, 14, 3-11.
- CARPANO, M., 1931. African horse sickness as observed particularly in Egypt and in Eritrea. Cairo: Government Press.
- CASTILLO-OLIVARES, J., CALVO-PINILLA, E., CASANOVA, I., BACHANEK-BANKOWSKA, K., CHIAM, R., MAAN, S., NIETO, J. M., ORTEGO, J. & MERTENS, P. P. C., 2011. A modified vaccinia Ankara virus (MVA) vaccine expressing African horse sickness virus (AHSV) VP2 protects against AHSV challenge in an IFNAR -/- mouse model. PLoS One, 6, e16503.
- CHIAM, R., SHARP, E., MAAN, S., RAO, S., MERTENS, P., BLACKLAWS, B., DAVIS-POYNTER, N., WOOD, J. & CASTILLO-OLIVARES, J., 2009. Induction of antibody responses to African horse sickness virus (AHSV) in ponies after vaccination with recombinant modified vaccinia Ankara (MVA). PLoS One, 4, e5997.
- COSTA, S., SASTRE, P., PEREZ, T., TAPIA, I., BARRANDEGUY, M., SANCHEZ-VIZCAINO, J. M., SANCHEZ-MATAMOROS, A., WIGDOROVITZ, A., SANZ, A. & RUEDA, P., 2016. Development and evaluation of a new lateral flow assay for simultaneous detection of antibodies against African Horse Sickness and Equine Infectious Anemia viruses. Journal of Virological Methods, 237, 127-131.
- CRAFFORD, J. E., LOURENS, C. W., GARDNER, I. A., MACLACHLAN, N. J. & GUTHRIE, A. J., 2013. Passive transfer and rate of decay of maternal antibody against African horse sickness virus in South African Thoroughbred foals. Journal of Equine Veterinary, 45, 604-607.
- DARDIRI, A. H. & OZAWA, Y., 1969. Immune and serologic response of dogs to neurotropic and viscerotropic African horse-sickness viruses. Journal of the American Veterinary Medical Association, 155, 400-407.
- DAVIES, F. G. & LUND, L. J., 1974. The application of fluorescent antibody techniques to the virus of African horse sickness. Research in Veterinary Science, 17, 128-130.
- DAVIES, F. G. & OTEINO, S., 1977. Elephants and zebras as possible reservoir hosts for African horse sickness virus. Veterinary Record, 100, 291-292.
- DE LA POZA, F., CALVO-PINILLA, E., LOPEZ-GIL, E., MARIN-LOPEZ, A., MATEOS, F., CASTILLO-OLIVARES, J., LORENZO, G. & ORTEGO, J., 2013. Ns1 is a key protein in the vaccine composition to protect Ifnar(-/-) mice against infection with multiple serotypes of African horse sickness virus. PLoS One, 8, e70197.
- DENNIS, S. J., MEYERS, A. E., GUTHRIE, A. J., HITZEROTH, I. I. & RYBICKI, E. P., 2018. Immunogenicity of plant-produced African horse sickness virus-like particles: implications for a novel vaccine. Plant Biotechnology Journal, 16, 442-450.
- DENNIS, S. J., O’KENNEDY, M. M., RUTKOWSKA, D., TSEKOA, T., LOURENS, C. W., HITZEROTH, I. I., MEYERS, A. E. & RYBICKI, E. P., 2018. Safety and immunogenicity of plant-produced African horse sickness virus-like particles in horses. Veterinary Research, 49, 105.
- DU PLESSIS, D. H., VAN WYNGAARDT, W. & BREMER, C. W., 1990. An indirect sandwich ELISA utilising F(ab')2 fragments for the detection of African horsesickness virus. Journal of Virological Methods, 29, 279-290.
- DU PLESSIS, D. H., VAN WYNGAARDT, W., GERDES, G. H. & OPPERMAN, E., 1991. Laboratory confirmation of African horse sickness in the Western Cape: application of a F(ab)2-based indirect ELISA. Onderstepoort Journal of Veterinary Research, 58, 1-3.
- DU PLESSIS, M., CLOETE, M., AITCHISON, H. & VAN DIJK, A. A., 1998. Protein aggregation complicates the development of baculovirus-expressed African horse sickness virus serotype 5 VP2 subunit vaccines. Onderstepoort Journal of Veterinary Research, 65, 321-329.
- DU TOIT, P. J., 1924. Horse sickness in 1923. Journal of the Department of Agriculture, Union of South Africa (1924), 8, 370-382.
- DU TOIT, P. J. & ALEXANDER, R. A., 1930. The immunization of horses against horse sickness by the use of formalized virus. Onderstepoort Journal of Veterinary Science and Animal Industry, 85-104.
- DU TOIT, P. J., ALEXANDER, R. A. & NEITZ, W. O., 1933. The immunization of horses against horsesickness by the use of formalized virus. Onderstepoort Journal of Veterinary Science and Animal Industry, 1, 25-48.
- DU TOIT, P. J. & NEITZ, W. O., 1932. The immunization of mules, with formalized horsesickness virus. Onderstepoort Journal of Veterinary Science and Animal Industry, 1, 35-47.
- DU TOIT, R. M., 1944. The transmission of blue tongue and horsesickness by Culicoides. Onderstepoort Journal of Veterinary Science and Animal Industry, 19, 7-16.
- DURR, P. A., GRAHAM, K. & VAN KLINKEN, R. D., 2017. Sellers’ Revisited: A Big Data Reassessment of Historical Outbreaks of Bluetongue and African Horse Sickness due to the Long-Distance Wind Dispersion of Culicoides Midges. Frontiers in Veterinary Science, 4.
- EDINGTON, A., 1900. South African horsesickness. Its pathology and methods of protective inoculation. Journal of Comparative Pathology and Therapeutics, 13, 200-231/281-300.
- EDINGTON, A., 1903. Note on the co-relation of several diseases occurring among animals in South Africa. Journal of Hygiene, 3, 137-154.
- EL GARCH, H., CRAFFORD, J. E., AMOUYAL, P., DURAND, P. Y., TOULEMONDE, C. E., LEMAITRE, L., COZETTE, V., GUTHRIE, A. & MINKE, J. M., 2012. An African horse sickness virus serotype 4 recombinant canarypox virus vaccine elicits specific cell-mediated immune responses in horses. Veterinary Immunology and Immunopathology, 149, 76-85.
- ERASMUS, B. J., 1963. Cultivation of horse sickness virus in tissue culture. Nature, 200, 716-719.
- ERASMUS, B. J., 1963. Preliminary observations on the value of the guinea-pig in determining the innocuity and antigenicity of neurotropic attenuated horse sickness strains. Onderstepoort Journal of Veterinary Research, 30, 11-22.
- ERASMUS, B. J., 1964. Some observations on the propagation of horse sickness virus in tissue culture. Bulletin de l'Office International des Epizooties, 62, 923-928.
- ERASMUS, B. J., 1966. The attenuation of Horse sickness virus: problems and advantages associated with the use of different host systems In: BRYANS JT., (ed.). Equine Infectious Diseases I. Princeton, New Jersey: Veterinary Publications Incorporated, 208-213.
- ERASMUS, B. J., 1973. Pathogenesis of African horse sickness In: GERBER H., (ed.). Equine Infectious Diseases III. Basel: Karger SA, 1-11.
- ERASMUS, B. J., 1978. A new approach to polyvalent immunization against African horse sickness In: BRYANS, J. T. & GERBER, H., (eds.). Equine Infectious Diseases IV. Princeton, New Jersey: Veterinary Publications Incorporated, 401-403.
- ERASMUS, B. J., YOUNG, E., PIETERSE, L. M. & T., B. S., 1976/1978. The Susceptibility of zebra and elephants to African horse sickness virus. Proceedings of the Fourth International Conference on Equine Infectious Diseases, Lyon. Princeton, NJ: Veterinary Publications Inc, 409-413.
- FABER, E., TSHILWANE, S. I., KLEEF, M. V. & PRETORIUS, A., 2021. Virulent African horse sickness virus serotype 4 interferes with the innate immune response in horse peripheral blood mononuclear cells in vitro. Infection, Genetics and Evolution, 91, 104836.
- FABER, E., TSHILWANE, S. I., VAN KLEEF, M. & PRETORIUS, A., 2022. Apoptosis versus survival of African horse sickness virus serotype 4-infected horse peripheral blood mononuclear cells. Virus Research, 307, 198609.
- FERNANDEZ-PINERO, J., FERNANDEZ-PACHECO, P., RODRIGUEZ, B., SOTELO, E., ROBLES, A., ARIAS, M. & SANCHEZ-VIZCAINO, J. M., 2009. Rapid and sensitive detection of African horse sickness virus by real-time PCR. Research in Veterinary Science, 86, 353-358.
- FISCHER-TENHAGEN, C., HAMBLIN, C., QUANDT, S. & FROLICH, K., 2000. Serosurvey for selected infectious disease agents in free-ranging black and white rhinoceros in Africa. Journal of wildlife diseases, 36, 316-323.
- FOURIE, P. J. J., 1935. Blood groups of the horse with special reference to their significance in blood transfusion and in horse sickness immunisation. Onderstepoort Journal of Veterinary Science and Animal Industry, 4, 7-47.
- FOWLER, V. L., HOWSON, E. L. A., FLANNERY, J., ROMITO, M., LUBISI, A., AGUERO, M., MERTENS, P., BATTEN, C. A., WARREN, H. R. & CASTILLO-OLIVARES, J., 2017. Development of a Novel Reverse Transcription Loop-Mediated Isothermal Amplification Assay for the Rapid Detection of African Horse Sickness Virus. Transboundary Emerging Diseases, 64, 1579-1588.
- GAUDREAULT, N. N., MAYO, C. E., JASPERSON, D. C., CROSSLEY, B. M., BREITMEYER, R. E., JOHNSON, D. J., OSTLUND, E. N., MACLACHLAN, N. J. & WILSON, W. C., 2014. Whole genome sequencing and phylogenetic analysis of Bluetongue virus serotype 2 strains isolated in the Americas including a novel strain from the western United States. Journal of Veterinary Diagnostic Investigation, 26, 553-557.
- GOMEZ-VILLAMANDOS, J. C., SANCHEZ, C., CARRASCO, L., LAVIADA, M. M., BAUTISTA, M. J., MARTINEZ-TORRECUADRADA, J., SANCHEZ-VIZCAINO, J. M. & SIERRA, M. A., 1999. Pathogenesis of African horse sickness: ultrastructural study of the capillaries in experimental infection. Journal of Comparative Pathology, 121, 101-116.
- GREWAR, J. D., WEYER, C. T., VENTER, G. J., VAN HELDEN, L. S., BURGER, P., GUTHRIE, A. J., COETZEE, P., LABUSCHAGNE, K., BUHRMANN, G., PARKER, B. J. & THOMPSON, P. N., 2019. A field investigation of an African horse sickness outbreak in the controlled area of South Africa in 2016. Transboundary Emerging Diseases, 66, 743-751.
- GREWAR, J. D., WEYER, C. T., VENTER, G. J., VAN HELDEN, L. S., BURGER, P., GUTHRIE, A. J., COETZEE, P., LABUSHAGNE, K., BUHRMANN, G., PARKER, B. J. & THOMPSON, P. N., 2013. The 2011 outbreak of African horse sickness in the African horse sickness-controlled area in South Africa. Journal of the South African Veterinary Association, 84.
- GUTHRIE, A. J., COETZEE, P., MARTIN, D. P. & LOURENS, C., 2015. Complete Genome Sequences of Four African Horse Sickness Virus Strains from a Commercial Tetravalent Live Attenuated Vaccine. Genome Announcements, 3.
- GUTHRIE, A. J., MACLACHLAN, N. J., JOONE, C., LOURENS, C. W., WEYER, C. T., QUAN, M., MONYAI, M. S. & GARDNER, I. A., 2013. Diagnostic accuracy of a duplex real-time reverse transcription quantitative PCR assay for detection of African horse sickness virus. Journal of Virological Methods, 189, 30-35.
- GUTHRIE, A. J., QUAN, M., LOURENS, C. W., AUDONNET, J., MINKE, J. M., YAO, J., HE, L., NORDGREN, R., GARDNER, I. A. & MACLACHLAN, N. J., 2009. Protective immunization of horses with a recombinant canarypox virus vectored vaccine co-expressing genes encoding the outer capsid proteins of African horse sickness virus. Vaccine, 27, 4434-4438.
- HAIG, D. A., MCINTOSH, B. M. & CUMMING, R. B., 1956. An outbreak of horse sickness, complicated by distemper in a pack of foxhounds. Journal of the South African Veterinary Association, 27, 245-249.
- HAMBLIN, C., ANDERSON, E. C., JAGO, M., MLENGEYA, K. & HIPJI, K., 1990. Antibodies to some pathogenic agents in free-living wild species in Tanzania. Epidemiology and Infection, 105, 585-594.
- HAMBLIN, C., MELLOR, P. S., GRAHAM, S. D., HOOGHUIS, H., MONTEJANO, R. C., CUBILLO, M. A. & BONED, J., 1991. Antibodies in horses, mules and donkeys following monovalent vaccination against African horse sickness. Epidemiology and Infection, 106, 365-371.
- HAMBLIN, C., MERTENS, P. P. C., MELLOR, P. S., BURROUGHS, N. J. & CROWTHER, J. R., 1991. A serogroup specific enzyme-linked immunosorbent assay for the detection and identification of African horse sickness viruses. Journal of Virological Methods, 31, 285-292.
- HAMBLIN, C., SALT, J. S., MELLOR, P. S., GRAHAM, S. D., SMITH, P. R. & WOHLSEIN, P., 1998. Donkeys as reservoirs of African horse sickness virus. Archives of Virology, 14, 37-47.
- HARRUP, L., MIRANDA, M. A. & CARPENTER, S., 2016. Advances in control techniques for Culicoides and future prospects. Veterinaria Italiana.
- HAZRATI, A., 1967. Identification and typing of horse-sickness virus strains isolated in the recent epizootic of the disease in Morocco, Tunisia and Algeria. Archives of Razi Institute, 19, 131-143.
- HENNING, M. W., 1956. African horse sickness, perdesiekte, pestis equorum. Animal Diseases of South Africa. 3rd edition. Pretoria: Central News Agency Ltd, 785-808.
- HESS, W. R., 1988. African horse sickness In: MONATH, T. P., (ed.). The arboviruses: Epidemiology and Ecology. Florida: Boca Raton, CRC Press Inc, (3), 1-18.
- HOUSE, C., MIKICUIK, P. E. & BERNINGER, M. L., 1990. Laboratory diagnosis of African horse sickness: comparison of serological techniques and evaluation of storage methods of samples for virus isolation. Journal of Veterinary Diagnostic Investigation, 2, 44-50.
- HOUSE, J. A., LOMBARD, M., DUBOURGET, P., HOUSE, C. & MEBUS, C. A., 1994. Further studies on the efficacy of an inactivated African horse sickness serotype 4 vaccine. Vaccine. 12, 142-144.
- HOWELL, P. G., 1962. The isolation and identification of further antigenic types of African horse sickness virus. Onderstepoort Journal of Veterinary Research, 29, 139-149.
- HOWELL, P. G., 1963. African horsesickness. In Emerging Diseases of Animals Rome Food and Agriculture Organization of the United Nations, 71-108.
- HOWELL, P. G., 1979. African horse sickness. In: ROHRER, H., (ed.). Handbuch Der Virusinfektionen Bei Tieren: Jena: Fischer, 593-625.
- HUISMANS, H. & ELS, H. J., 1979. Characterization of the tubules associated with the replication of three different orbiviruses. Virology, 92, 397-406.
- HUQ, M. M., 1961. African horse sickness in Pakistan. Bulletin de l'Office International Epizooties, 55, 278-284.
- KANAI, Y., VAN RIJN, P. A., MARIS-VELDHUIS, M., KANAME, Y., ATHMARAM, T. N. & ROY, P., 2014. Immunogenicity of recombinant VP2 proteins of all nine serotypes of African horse sickness virus. Vaccine, 32, 4932-4937.
- KING, S., RAJKO-NENOW, P., ASHBY, M., FROST, L., S., C. & BATTEN, C., 2020. Outbreak of African horse sickness in Thailand, 2020. Transboundary and Emerging Diseases, 67, 1764-1767.
- KOEKEMOER, J. J., 2008. Serotype-specific detection of African horse sickness virus by real-time PCR and the influence of genetic variations. Journal of Virological Methods, 154, 104-110. doi: 10.1016/j.jviromet.2008.08.010.
- LAVIADA, M. D., SANCHEZ-VIZCAINO, J. M., SOBRINO, F. & ROY, P., 1997. Detection of African horsesickness virus by the polymerase chain reaction: a note. Investigacion en Sanidad Animal, 12, 97-102.
- LECATSAS, G. & ERASMUS, B. J., 1967. Electron microscopic study of the formation of African horse-sickness virus. Archiv fur die gesamte Virusforschung, 22, 442-450.
- LESTÉ-LASSERRE, C., 2020. Thailand scrambles to contain major outbreak of horse-killing virus. ScienceMag: Science.
- LORD, C. C., WOOLHOUSE, M. E. & BARNARD, B. J., 1997. Transmission and distribution of virus serotypes: African horse sickness in zebra. Epidemiology and Infection, 118, 43-50.
- LORD, C. C., WOOLHOUSE, M. E. & BARNARD, B. J., 1998. Transmission and distribution of African horse sickness virus serotypes in South African zebra. Archives of Virology. Supplementa, 14, 21-28.
- LU, G., PAN, J., OU, J., SHAI, R., HU, X., WANG, C. & LI, S., 2020. African horse sickness: Its emergence in Thailand and potential threat to other Asian countries. Transboundary and Emerging Diseases, 67, 1751-1753.
- LUBROTH, J., 1988. African horse sickness and the epizootic in Spain 1987. Equine Practice, 10, 26-33.
- LULLA, V., LOSADA, A., LECOLLINET, S., KERVIEL, A., LILIN, T., SAILLEAU, C., BECK, C., ZIENTARA, S. & ROY, P., 2017. Protective efficacy of multivalent replication-abortive vaccine strains in horses against African horse sickness virus challenge. Vaccine, 35, 4262-4269.
- M'FADYEAN, J., 1900. African horse sickness. Journal of Comparative Pathology and Therapeutics, 13, 1-20.
- M'FADYEAN, J., 1910. The Susceptibility of the Dog to African Horse-Sickness. Journal of Comparative Pathology and Therapeutics, 23, 27-33.
- MAAN, N. S., MAAN, S., NOMIKOU, K., BELAGANAHALLI, M. N., BACHANEK-BANKOWSKA, K. & MERTENS, P. P. C., 2011. Serotype specific primers and gel-based RT-PCR assays for 'typing' African horse sickness virus: identification of strains from Africa. PLoS One, 6, e25686.
- MAAN, S., RAO, S., MAAN, N. S., ANTHONY, S. J., ATTOUI, H., SAMUEL, A. R. & MERTENS, P. P. C., 2007. Rapid cDNA synthesis and sequencing techniques for the genetic study of bluetongue and other dsRNA viruses. Journal of Virological Methods, 143, 132-139.
- MANNING, N. M., BACHANEK-BANKOWSKA, K., MERTENS, P. P. C. & CASTILLO-OLIVARES, J., 2017. Vaccination with recombinant Modified Vaccinia Ankara (MVA) viruses expressing single African horse sickness virus VP2 antigens induced cross-reactive virus neutralising antibodies (VNAb) in horses when administered in combination. Vaccine, 35, 6024-6029.
- MARCHI, P. R., RAWLINGS, P., BURROUGHS, J. N., WELLBY, M., MERTENS, P. P., MELLOR, P. S. & WADE-EVANS, A. M., 1995. Proteolytic cleavage of VP2, an outer capsid protein of African horse sickness virus, by species-specific serum proteases enhances infectivity in Culicoides. J Gen Virol, 76 ( Pt 10), 2607-2611.
- MARCHI, P. R., RAWLINGS, P., BURROUGHS, J. N., WELLBY, M., MERTENS, P. P., MELLOR, P. S. & WADE-EVANS, A. M., 1995. Proteolytic cleavage of VP2, an outer capsid protein of African horse sickness virus, by species-specific serum proteases enhances infectivity in Culicoides. Journal of General Virology, 76(Pt 10), 2607-2611.
- MAREE, S., MAREE, F. F., PUTTERILL, J. F., DE BEER, T. A. P., HUISMANS, H. & THERON, J., 2016. Synthesis of empty African horse sickness virus particles. Virus Research, 213, 184-194.
- MARÍN-LÓPEZ, A., BARREIRO-PIÑEIRO, N., UTRILLA-TRIGO, S., BARRIALES, D., BENAVENTE, J., NOGALES, A., MARTINEZ-COSTAS, J., ORTEGO, J. & CALVO-PINILLA, E., 2020. Cross-protective immune responses against African horse sickness virus after vaccination with protein NS1 delivered by avian reovirus muNS microspheres and modified vaccinia virus Ankara. Vaccine, 38, 882-889.
- MARTINEZ-DE LA PUENTE, J., MARTINEZ, J., FERRAGUTI, M., MORALES-DE LA NUEZ, A., CASTRO, N. & FIGUEROLA, J., 2012. Genetic characterization and molecular identification of the bloodmeal sources of the potential bluetongue vector Culicoides obsoletus in the Canary Islands, Spain. Parasit Vectors, 5, 147.
- MARTÍNEZ-TORRECUADRADA, J. L., DÍAZ-LAVIADA, M., ROY, P., SANCHEZ, C., VELA, C., SANCHEZ-VIZCAINO, J. M. & CASAL, J. I., 1996. Full protection against African horse sickness (AHS) in horses induced by baculovirus-derived AHS virus serotype 4 VP2, VP5 and VP7. Journal of General Virology, 77, 1211-1221.
- MAURER, F. D. & MCCULLY, R. M., 1963. African horse sickness - with emphasis on pathology. American Journal of Veterinary Research, 24, 235-266.
- MCINTOSH, B. M., 1953. The isolation of virus in mice from cases of horse sickness in immunized horses. Onderstepoort Journal of Veterinary Research, 26, 183-195.
- MCINTOSH, B. M., 1955. Horse sickness antibodies in the sera of dogs in enzootic areas. Journal of the South African Veterinary Association, 26, 269-272.
- MCINTOSH, B. M., 1956. Complement fixation with horse sickness viruses. Onderstepoort Journal of Veterinary Research, 27, 165-169.
- MCINTOSH, B. M., 1958. Immunological types of horse sickness virus and their significance in immunization. Onderstepoort Journal of Veterinary Research, 27, 465-539.
- MEISWINKEL, R., BAYLIS, M. & LABUSCHAGNE, K., 2000. Stabling and the protection of horses from Culicoides bolitino (Diptera: Ceratopogonidae), a recently identified vector of African horse sickness. Bulletin of entomological research, 509-515.
- MIRCHAMSY, H. & H., T., 1964. Attempts to vaccinate foals with living tissue culture adapted horse sickness virus. Bulletin de l'Office International des Epizooties, 62, 911-921.
- MIRCHAMSY, H. & HAZRATI, A., 1973. A review on aetiology and pathogeny of African horse sickness. Archives of Razi Institute, 25, 23-46.
- MIZUKOSHI, N., SAKAMOTO, K., IWATA, A., UEDA, S., KAMADA, M. & FUKUSHO, A., 1994. Detection of African horse sickness virus by reverse transcriptase polymerase chain reaction (RT-PCR) using primers for segment 5 (NS1 gene). Journal of Veterinary Medical Science, 56, 347-352.
- N., R., 2019. Immediate notification: African horse sickness, first occurrence of a listed disease in the country, Chad. Paris: World Organisation for Animal Health.
- NEWSHOLME, S. J., 1983. A morphological study of the lesions of African horse sickness. Onderstepoort Journal of Veterinary Research, 50, 7-24.
- NIESCHULZ, O., BEDFORD, G. A. H. & DU TOIT, R. M., 1934. Results of a mosquito survey at Onderstepoort during the summer 1931-32 in connection with the transmission of horse sickness. Onderstepoort Journal of Veterinary Science and Animal Industry, 3, 43-77.
- NIESCHULZ, O. & DU TOIT, R. M., 1934. Investigation into transmission of horse sickness at Onderstepoort during the season 1932-1933. Onderstepoort Journal of Veterinary Science and Animal Industry, 8, 213-269.
- NOBEL, T. R., 1961. Vaccination against African horse sickness and post-vaccination reaction in Israel. Refuah Veterinarith, 18, 168-173.
- NOOR, N. M., 2020. Immediate notification: African horse sickness, First occurrence of a listed disease in the country, Malaysia. Paris: World organization for Animal Health.
- O'DELL, N., 2018. Pathology of fatal natural African horsesickness virus infection in dogs. Journal, Master of Veterinary Medicine (Pathology)
- O'DELL, N., 2018. Pathology of fatal natural African horsesickness virus infection in dogs, in Pathology. University of Pretoria: Pretoria.
- O'DELL, N., ARNOT, L., JANISCH, C. E. & STEYL, J. C., 2018. Clinical presentation and pathology of suspected vector transmitted African horse sickness in South African domestic dogs from 2006 to 2017. Veterinary Record, 182, 715.
- OELLERMANN, R. A., ELS, H. J. & ERASMUS, B. J., 1970. Characterization of African horse sickness virus. Arch Gesamte Virusforsch, 29, 163-174.
- ORHAN, A., 1961. Notes on African horse sickness in Cyprus. Bulletin de l'Office International Epizooties, 55, 250-257.
- OURA, C., 2018. A possible role for domestic dogs in the spread of African horse sickness virus. Veterinary Record, 182, 713-714.
- OURA, C. A. & EL HARRAK, M., 2011. Midge-transmitted bluetongue in domestic dogs. Epidemiol Infect, 139, 1396-1400.
- OURA, C. A. & EL HARRAK, M., 2011. Midge-transmitted bluetongue in domestic dogs. Epidemiology and Infection, 139, 1396-1400.
- OZAWA, Y. & HAZRATI, A., 1967. Post-vaccination reactions in donkeys vaccinated with African horse-sickness polyvalent vaccine. Bulletin de l'Office International Epizooties, 67, 949-959.
- OZAWA, Y., HAZRATI, A. & EROL, N., 1956. African horse-sickness live-virus tissue culture vaccine. American Journal of Veterinary Research, 26, 154-168.
- OZAWA, Y. & NAKATA, G., 1965. Experimental transmission of African horse-sickness by means of mosquitoes. American Journal of Veterinary Research, 26, 744-748.
- OZAWA, Y., NAKATA, G., SHAD-DEL, F. & NAVAI, S., 1966. Transmission of African horse-sickness by a species of mosquito, Aedes aegypti Linnaeus. American Journal of Veterinary Research, 27, 695-697.
- OZAWA, Y., SHAD-DEL, F. & NAKATA, G., 1966. Transmission of African horse sickness by means of mosquito bites and replication of the virus in Aedes aegypti. Proceedings of the 1st International Equine Infectious Diseases Conference, 196-207.
- PAGE, A. E., SLOVIS, N. M. & HOROHOV, D. W., 2014. Lawsonia intracellularis and equine proliferative enteropathy. Veterinary Clinics: Equine Practice, 30, 641-658.
- PARKER, J., ARMSTRONG, R. M., ABEGUNDE, A. & TAYLOR, W. P., 1977. African horse sickness virus antibodies in northern Nigeria 1974-1975. Research in Veterinary Science, 22, 274-280.
- PAVRI, K. M. & ANDERSON, C. R., 1963. Isolation of vaccine strain of African horse sickness virus from brains of two horses given polyvalent vaccine. Indian Journal of Veterinary Sciences and Biotechnology, 33, 215-219.
- PIERCY, S. E., 1951. Some observations on African horse-sickness including an account of an outbreak among dogs. East African Agricultural and Forestry Journal, 17, 62-64.
- PITCHFORD, N. & THEILER, A., 1903. Investigations into the nature and causes of horse sickness. Agricultural Journal of the Cape of Good Hope, 23, 153-156.
- POLSON, A., 1941. The particle size of African horse sickness virus as determined by ultrafiltration and ultracentrifugation. Onderstepoort Journal of Veterinary Research, 16, 33-50.
- POLSON, A. & DEEKS, D., 1963. Electron microscopy of neurotropic African horse-sickness virus. Journal of Hygiene (London), 61, 149-153.
- POTGIETER, A. C., PAGE, N. A., LIEBENBERG, J., WRIGHT, I. M., LANDT, O. & VAN DIJK, A., 2009. Improved strategies for sequence-independent amplification and sequencing of viral double-stranded RNA genomes. Journal of General Virology, 90, 1423-1432.
- POTGIETER, A. C., WRIGHT, I. M. & VAN DIJK, A. A., 2015. Consensus Sequence of 27 African Horse Sickness Virus Genomes from Viruses Collected over a 76-Year Period (1933 to 2009). Genome Announcements, 3.
- QUAN, M., LOURENS, C. W., MACLACHLAN, N. J., GARDNER, I. A. & GUTHRIE, A. J., 2010. Development and optimisation of a duplex real-time reverse transcription quantitative PCR assay targeting the VP7 and NS2 genes of African horse sickness virus. Journal of Virological Method, 167, 45-52.
- QUAN, M., VAN VUUREN, M., HOWELL, P. G., GROENEWALD, D. & GUTHRIE, A. J., 2008. Molecular epidemiology of the African horse sickness virus S10 gene. Journal of General Virology, 89, 1159-1168.
- RAFYI, A., 1961. Horse sickness. Bulletin de l'Office International Epizooties, 56, 216-250.
- REID, N. R., 1961. African horse sickness. British Veterinary Journal, 118, 137-142.
- RIDDIN, M. A., VENTER, G. J., LABUSCHAGNE, K. & VILLET, M. H., 2019. Culicoides species as potential vectors of African horse sickness virus in the southern regions of South Africa. Medical and Veterinary Entomology, 33, 498-511.
- RODRIGUEZ-SANCHEZ, B., FERNANDEZ-PINERO, J., SAILLEAU, C. & ZIENTARA, S., 2008. Novel gel-based and real-time PCR assays for the improved detection of African horse sickness virus. Journal of Virological Methods, 151, 87-94.
- RODRIGUEZ, M., HOOGHUIS, H. & CASTANO, M., 1992. African horse sickness in Spain. Veterinary Microbiology, 33, 129-142.
- ROY, P., 1996. Multiple Gene Expression in Baculovirus System: Third Generation Vaccines for Bluetongue Disease and African horse sickness Disease. Annals of the New York Academy of Sciences, 791, 318-332.
- ROY, P., BISHOP, D. H. L., HOWARD, S., AITCHISON, H. & ERASMUS, B., 1996. Recombinant Baculovirus-Synthesized African horse sickness Virus (AHSV) Outer-capsid Protein VP2 Provides Protection Against Virulent AHSV Challenge. Journal of General Virology, 77, 2053-2057.
- ROY, P. & SUTTON, G., 1998. New generation of African horse sickness virus vaccines based on structural and molecular studies of the virus particles. Archives of Virology: Supplementa, 14, 177-202.
- RUBIO, C., CUBILLO, M. A., HOOGHUIS, H., SANCHEZ-VIZCAINO, J. M., DIAZ-LAVIADA, M., PLATEAU, E., ZIENTARA, S., CRUCIERE, C. & HAMBLIN, C., 1998. Validation of ELISA for the detection of African horse sickness virus antigens and antibodies. Archives of Virology: Supplementa, 14, 311-315.
- RUTKOWSKA, D. A., MOKOENA, N. B., TSEKOA, T. L., DIBAKWANE, V. S. & O’KENNEDY, M. M., 2019. Plant-produced chimeric virus-like particles - A new generation vaccine against African horse sickness. BMC Veterinary Research, 15.
- SAILLEAU, C., HAMBLIN, C., PAWESKA, J. T. & ZIENTARA, S., 2000. Identification and differentiation of the nine African horse sickness virus serotypes by RT-PCT amplification of the serotype-specific genome segment 2. Journal of General Virology, 81, 1-7.
- SAILLEAU, C., MOULAY, S., CRUCIERE, C., LAEGREID, W. W. & ZIENTARA, S., 1997. Detection of African horse sickness virus in the blood of experimentally infected horses: comparison of virus isolation and a PCR assay. Research in Veterinary Science, 62, 229-232.
- SAKAMOTO, K., PUNYAHOTRA, R., MIZUKOSHI, N., UEDA, S., IMAGAWA, H., SUGIURA, T., KAMADA, M. & FUKUSHO, A., 1994. Rapid detection of African horse sickness virus by the reverse transcriptase polymerase chain reaction (RT-PCR) using the amplimer for segment 3 (VP3 gene). Archives of Virology, 136, 87-97.
- SALAMA, S. A., DARDIRI, A. H., AWAD, F. I., SOLIMAN, A. M. & AMIN, M. M., 1981. Isolation and identification of African horse sickness virus from naturally infected dogs in Upper Egypt. Canadian Journal of Comparative Medicine, 45, 392-396.
- SANCHEZ-MATAMOROS, A., BECK, C., KUKIELKA, D., LECOLLINET, S., BLAISE-BOISSEAU, S., GARNIER, A., RUEDA, P., ZIENTARA, S. & SANCHEZ-VIZCAINO, J. M., 2016. Development of a Microsphere-based Immunoassay for Serological Detection of African Horse Sickness Virus and Comparison with Other Diagnostic Techniques. Transboundary Emerging Diseases, 63, e270-e277.
- SANCHEZ-MATAMOROS, A., NIETO-PELEGRIN, E., BECK, C., RIVERA-ARROYO, B., LECOLLINET, S., SAILLEAU, C., ZIENTARA, S. & SANCHEZ-VIZCAINO, J. M., 2016. Development of a Luminex-Based DIVA Assay for Serological Detection of African Horse Sickness Virus in Horses. Transboundary Emerging Diseases, 63, 353-359.
- SELLERS, R. F., PEDGLEY, D. E. & TUCKER, M. R., 1977. Possible spread of African horse sickness on the wind. Journal of Hygiene, 79, 279-298.
- SHAH, K. V., 1964. Investigation of African horse sickness in India. I. Study of the natural disease and the virus. Indian Journal of Veterinary Science and Biotechnology, 34, 1-14.
- SINCLAIR, M., BUHRMANN, G. & GUMMOW, B., 2006. An epidemiological investigation of the African horse sickness outbreak in the Western Cape Province of South Africa in 2004 and its relevance to the current equine export protocol. Journal of the South African Veterinary Association, 77, 191-196.
- SLAMA, D., HAOUAS, N., MEZHOUD, H., BABBA, H. & CHAKER, E., 2015. Blood meal analysis of culicoides (Diptera: ceratopogonidae) in central Tunisia. PLoS One, 10, e0120528.
- SPENCE, R. P., MOORE, N. F. & NUTTALL, P. A., 1984. The biochemistry of orbiviruses. Brief review. Archives of Virology, 82, 1-18.
- STANLEY, N. F., 1981. Reoviridae: orbivirus and reovirus infections of mammals and birds. Comparative Diagnosis of Viral Diseases, 67-104.
- STONE-MARSCHAT, M., CARVILLE A, S., A. & LAERGREID, W. W., 1994. Detection of African horse sickness virus by reverse transcription-PCR. Journal of Clinical Microbiology, 32, 679-700.
- STUART, D. I., GOUET, P., GRIMES, J., MALBY, R., DIPROSE, J., ZIENTARA, S., BURROUGHS, J. N. & MERTENS, P. P., 1998. Structural studies of orbivirus particles. Archives of Virology: Supplementa, 14, 235-250.
- SWANEPOEL, R., ERASMUS, B. J., WILLIAMS, R. & TAYLOR, M. B., 1992. Encephalitis and chorioretinitis associated with neurotropic African horse sickness virus infection in laboratory workers: Part III. Virological and serological investigations. South African Medical Journal, 81, 458-461.
- THANETO, S., 2020. Immediate notification: African horse sickness, First occurrence of a listed disease in the country, Thailand. Paris: World Organization for Animal Health.
- THEILER, A., 1905. Horse sickness - Introduction. Report of Government Veterinary Bacteriologist, 131-162.
- THEILER, A., 1907. On the co-relation of various disease of stock in South Africa. Report of Government Veterinary Bacteriologist, 67-80.
- THEILER, A., 1908. Further notes on immunity in horse sickness. Report of Government Veterinary Bacteriologist, 89-161.
- THEILER, A., 1908. The immunization of mules with polyvalent serum and virus. Report of Govern Veterinary bacteriology, 192-213.
- THEILER, A., 1909. Investigation into South African diseases (f). On the variability of the virulency of a particular strain of horse sickness virus. Report of the Government Veterinary Bacteriologist, 57-144.
- THEILER, A., 1910. The susceptibility of the dog to African horse sickness. Journal of Comparative Pathology, 23, 315-325.
- THEILER, A., 1920. Paralysis of the oesophagus in the horse as a sequal to horse sickness. Report of the Director of Veterinary Research, 7-8, 339-357.
- THEILER, A., 1921. African horse sickness (pestis equorum). Science Bulletin, 19, 1-29.
- VAN RENSBERG, I. B., DE CLERK, J., GROENEWALD, H. B. & BOTHA, W. S., 1981. An outbreak of African horsesickness in dogs. Journal of the South African Veterinary Association, 52, 323-325.
- VAN RIJN, P. A., MARIS-VELDHUIS, M. A., POTGIETER, C. A. & VAN GENNIP, R. G. P., 2018. African horse sickness virus (AHSV) with a deletion of 77 amino acids in NS3/NS3a protein is not virulent and a safe promising AHS Disabled Infectious Single Animal (DISA) vaccine platform. Vaccine, 36, 1925-1933.
- VAN SACEGHAM, R., 1918. La peste du cheval ou horse sickness au Congo Belge. Bulletin de la Societe de Pathologie Exotique, 11, 423-432.
- VAN SCHALKWYK, A., FERREIRA, M. L. & ROMITO, M., 2019. Using a new serotype-specific Polymerase Chain Reaction (PCR) and sequencing to differentiate between field and vaccine-derived African Horse Sickness viruses submitted in 2016/2017. Journal of Virological Methods, 266, 89-94.
- VAN SITTERT, S. J., DREW, T. M., KOTZE, J. L., STRYDOM, T., WEYER, C. T. & GUTHRIE, A. J., 2013. Occurrence of African horse sickness in a domestic dog without apparent ingestion of horse meat. Journal of the South African Veterinary Association, 84, 1-5.
- VENTER, G. J., GRAHAM, S. D. & HAMBLIN, C., 2000. African horse sickness epidemiology: vector competence of south african Culicoides species for virus serotypes 3, 5 and 8. Medical and Veterinary Entomology, 14, 245-250.
- VERWOERD, D. W., HUISMANS, H. & ERASMUS, B. J., 1979. Orbiviruses In: FRAENKEL-CONRAT, H. & WAGNER, R. R., (eds.). Comprehensive Virology. London: Plenum Press, 285-345.
- VON TEICHMAN, B. F., DUNGU, B. & SMIT, T. K., 2010. In vivo cross-protection to African horse sickness Serotypes 5 and 9 after vaccination with Serotypes 8 and 6. Vaccine, 28, 6505-6517.
- VON TEICHMAN, B. F. & SMIT, T. K., 2008. Evaluation of the pathogenicity of African Horse sickness (AHS) isolates in vaccinated animals. Vaccine, 26, 5014-5021.
- WETZEL, H., NEVILL, E. M. & ERASMUS, B. J., 1970. Studies on the transmission of African horse sickness. Onderstepoort Journal of Veterinary Research, 37, 165-168.
- WEYER, C. T., GREWAR, J. D., BURGER, P., JOONE, C., LOURENS, C., MACLACHLAN, N. J. & GUTHRIE, A. J., 2017. Dynamics of African horse sickness virus nucleic acid and antibody in horses following immunization with a commercial polyvalent live attenuated vaccine. Vaccine, 35, 2504-2510.
- WEYER, C. T., GREWAR, J. D., BURGER, P., ROSSOUW, E., LOURENS, C., JOONE, C., LE GRANGE, M., COETZEE, P., VENTER, E., MARTIN, D. P., MACLACHLAN, N. J. & GUTHRIE, A. J., 2016. African Horse Sickness Caused by Genome Reassortment and Reversion to Virulence of Live, Attenuated Vaccine Viruses, South Africa, 2004-2014. Emerging Infectious Diseases, 22, 2087-2096.
- WEYER, C. T., JOONE, C., LOURENS, C. W., MONYAI, M. S., KOEKEMOER, O., GREWAR, J. D., VAN SCHALKWYK, A., MAJIWA, S. O. A., MACLACHLAN, N. J. & GUTHRIE, A. J., 2015. Development of three triplex real-time reverse transcription PCR assays for the qualitative molecular typing of the nine serotypes of African horse sickness virus. Journal of Virological Methods, 223, 69-74.
- WEYER, C. T., QUAN, M., JOONE, C., LOURENS, C. W., MACLACHLAN, N. J. & GUTHRIE, A. J., 2013. African horse sickness in naturally infected, immunised horses. Equine Veterinary Journal, 45, 117-119.
- WHITEHEAD, Z., LE ROUX, C., O'DELL, N. & HANEKOM, J., 2018. Clinical presentation and management of African horse sickness in two dogs. Veterinary Record Case Reports, 6, e000664.
- WOHLSEIN, P., POHLENZ, J. F., DAVIDSON, F. L., SALT, J. S. & HAMBLIN, C., 1997. Immunohistochemical demonstration of African Horse Sickness viral antigen in formalin-fixed equine tissues. Veterinary Pathology, 34, 568-574.
- WOHLSEIN, P., POHLENZ, J. F., SALT, J. S. & HAMBLIN, C., 1998. Immunohistochemical demonstration of African horse sickness viral antigen in tissues of experimentally infected equines. Archives of Virology: Supplementa, 14, 57-65.
- ZELEKE, A., SORI, T., POWEL, K., GEBRE-AB, B. F. & ENDEBU, B., 2005. Isolation and identification of circulating serotypes of African horse sickness virus in Ethiopia. The Journal of Applied Research in Veterinary Medicine, 3, 40-43.
- ZIENTARA, S., SAILLEAU, C., MOULAY, S. & CRUCIERE, C., 1994. Diagnosis of the African horse sickness virus serotype 4 by a one-tube, one manipulation RT-PCR reaction from infected organsJournal of Virological Methods, 46, 179-188.
- ZIENTARA, S., SAILLEAU, C., MOULAY, S., WADE-EVANS, A. & CRUCIERE, C., 1995. Application of the polymerase chain reaction to the detection of African horse sickness viruses. Journal of Virological Methods, 53, 47-54.
- ZIENTARA, S., SAILLEAU, C., PLATEAU, E., MOULAY, S., MERTENS, P. P. & CRUCIERE, C., 1998. Molecular epidemiology of African horse sickness virus based on analyses and comparisons of genome segments 7 and 10. Archives of Virology: Supplementa, 14, 221-234.