- Infectious Diseases of Livestock
- Part 2
- Scrapie
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Scrapie
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). T Konold and E Vallino-Costassa, Scrapie, 2018.

Scrapie
Previous authors: R BRADLEY AND D W VERWOERD
Current authors:
T KONOLD - Veterinary Surgeon, Information Officer & Veterinary Research Officer, DrMedVet, PhD, MRCVS, ASU, Pathology Department, APHA Weybridge, Woodham Lane, New Haw, Addlestone, Surrey, KT15 3NB, United Kingdom
E VALLINO COSTASSA - Doctor in Veterinary Medicine, DVM, PhD, Centre for Animal Encephalopathies (CEA), Istituto Zooprofilattico Sperimentale Piemonte Liguria e Valle d’Aosta, Turin, Italy
Introduction
Scrapie is the oldest known transmissible spongiform encephalopathy (TSE) and was first described in sheep in 1750 in a German book on agriculture although others implied that it has been known since Roman times.169 Many theories existed about the cause of the disease; hereditary, sexually transmitted and/ or infectious but only in the 20th century was it demonstrated that the scrapie agent was transmissible.
Major events leading to our current knowledge of scrapie occurred as follows:
- 1898: Description of the microscopic neuropathology and neuronal vacuolation notably of motor neurons in the spinal cord.17
- 1936: Scrapie shown to be an experimentally transmissible disease in sheep.34
- 1936 to 1938: Iatrogenic scrapie occurred in hundreds of sheep in Great Britain through the use of louping-ill vaccine contaminated accidentally with scrapie agent from brains and spleens of sheep used for its manufacture. This was first reported at the Third International Congress for Microbiology75 and subsequently published in 1946.74 The transmissible nature of the scrapie agent was thus confirmed beyond reasonable doubt.
- 1957 to 1959: Zigas and Gajdusek reported the occurrence of kuru (trembling or shivering of the body, also known as the laughing death), a rare and unusual neurological disease in the Fore-speaking people of Papua New Guinea.221 They believed this was propagated by endocannibalistic consumption of the whole bodies of dead relatives during funeral rites. The American veterinary pathologist W.J. Hadlow made the important pathological connection between scrapie of sheep and kuru of humans by observing that ‘the overall resemblance between kuru and scrapie is too impressive to be ignored’.80 He went on to suggest the experimental transmission of kuru to a laboratory primate. This was subsequently achieved and was followed by successful transmission of human Creutzfeldt-Jakob disease (CJD) to primates, thus establishing that these rare human disorders were, like scrapie, transmissible spongiform encephalopathies (TSEs).
- 1961: Experimental transmission of scrapie to mice thus establishing a model system in which the disease, and the agent that caused it, could be studied in the laboratory26
- 1966 to 1967: Report of the very small size of the scrapie agent, its almost complete resistance to destruction by massive germicidal doses of ionizing radiation supporting the view that the agent may have a mode of replication independent of the integrity of a nucleic acid moiety.4, 5
- 1979: Report of the scrapie replication site hypothesis45 and the results of studies on the genetics of scrapie in sheep and mice.44 The latter report showed that murine genes influenced the phenotype of scrapie, notably its incubation period and lesion profile, when a small number of important factors were controlled. This work was the basis for identifying and distinguishing strains of scrapie agent and led to further opportunities to study the pathogenesis of scrapie notably by Bruce, Dickinson, Fraser and Kimberlin. The scrapie strain typing methods were to become vitally important after the discovery of bovine spongiform encephalopathy (see Bovine spongiform encephalopathy).
- 1979, 1980 and 1982: Reports on the tissue distribution of scrapie infectivity in goats and Suffolk sheep and in some other breeds of sheep with natural scrapie, including during the incubation period of Suffolk sheep from high scrapie-risk families82-84
- 1982: Establishment of the prion definition (small, proteinaceous infectious particles that resist inactivation by procedures that modify nucleic acids) and prion hypothesis.157
- 1990: Description of prion protein polymorphisms at position 171 associated with incubation control of scrapie in sheep.62 Additional polymorphisms were later identified at positions 136 and 154; all associated with scrapie susceptibility in sheep. This formed the basis for scrapie eradication by genotype selection in various countries.
- 2003: Report of a new type of scrapie in Norway, Nor98,16 which was subsequently found in other countries, including those considered free from scrapie. This disease was later termed ‘atypical scrapie’ to distinguish it from ‘classical scrapie’ that has been known for centuries. Retrospective studies confirmed atypical scrapie in archived sheep material from the 1970s and 1980s,27, 213 which suggests that it is not a new disease.
Substantial epidemiological studies have failed to reveal a connection between CJD in humans and scrapie; these were summarized by the European Food Safety Authority in 2011.46 Subsequently, it has been shown that some classical scrapie isolates can be transmitted to transgenic mice carrying the human prion protein gene and reproduce characteristics of a disease on second passage with similarities to sporadic CJD.25 Whilst this may indicate that some isolates of the scrapie agent may be zoonotic, it is not known whether transmission is possible in natural circumstances.49
Classical scrapie isolates have produced disease in cattle following intracerebral inoculation,29, 36, 115, 160 which was different to bovine spongiform encephalopathy, but there has been no evidence of infection following oral challenge.35, 119 Studies in the UK failed to infect pigs with classical scrapie,135 which is contrary to more recent studies in the USA, that reported infection by intracerebral and oral routes.77 Whether atypical scrapie can be transmitted to other farm animal species is currently unknown.
Classical scrapie has been found in most of the sheep-raising countries of the world at some time or another, often following importation of infected animals, but in some of these countries, including Australia, New Zealand and South Africa, the disease was eradicated before it could spread to indigenous sheep. The first case in South Africa was in 1966 in a Hampshire Down sheep imported from England.205 The disease was subsequently found in 11 sheep, all first- or second-generation progeny of the English imports of Hampshire Downs. A strict slaughter policy was applied, and no case has been diagnosed in South Africa since 1972, and the disease is considered to have been eradicated.
In contrast, atypical scrapie has been found even in sheep born in countries free from classical scrapie, such as New Zealand106 and Australia.31
Aetiology
It is commonly accepted that scrapie is an infectious disease caused by prions, i.e. proteinaceous infectious particles that are devoid of nucleic acid.158 The hypothesis is that the prion is composed exclusively of a modified isoform of PrP, named PrPSc. The latter is generated from the normal cellular form (PrPC) by re-folding part of its α-helix coil structure into a β sheet. These physical changes to the tertiary structure of the protein profoundly alter its physical properties and, it is also claimed, confers strain-specific (biological) properties.2 Thus, whereas PrPC is soluble in non-denaturing detergents, the disease-specific form is not. PrPC is denatured by proteases such as proteinase K but PrPSc is partially protease resistant, a feature that is utilized to aid in the diagnosis of prion diseases. PrPSc provokes no immune response in the host, presumably because the modified host protein, of which it is mostly comprised, is recognized as ‘self’. To prove the prion hypothesis, experiments usually relied on extraction of PrPC from live animals, which may have also contained DNA from viruses or bacteria, but no nucleic acid has been found despite diligent searching. In fact, infectious prions have been generated de novo from bacterially-expressed recombinant PrP, which can cause disease in mice.220 Strains are characterized by their structural, biochemical and stability differences of the prion protein.219
Prions are extraordinarily resistant to heat, ultraviolet and ionizing radiation, and most chemicals that inactivate microorganisms, although sodium hypochlorite and autoclaving in 2M sodium hydroxide are effective in destroying it.190 The use of 20,000 ppm sodium hypochlorite for 1 hour is recommended for use in practice to decontaminate surfaces.105
There are still questions about the function of the cellular prion protein because it is also found in various non-neural tissues. It may be involved in T cell, synaptic function or copper metabolism.222 Animals devoid of prion protein, however, appear to be clinically healthy and viable as reported for Norwegian dairy goats that have a natural mutation in the prion protein gene.15 However, inflammatory responses may be aggravated in these goats, at least after experimentally induced pulmonary inflammation,166 and it has been hypothesized that PrP may contribute to protection of vulnerable tissues against inflammatory damage.165
Epidemiology
Sheep and goats are the natural hosts of scrapie although there are considerable differences in the epidemiology between scrapie in these two species. Whilst disease in goats has been often attributed to contact with scrapie-infected sheep, outbreaks also occur in the absence of sheep.9 Scrapie has also been described in moufflon, a species of wild mountain sheep occurring in Europe.216
Classical and atypical scrapie occurs world-wide but the exact regional distribution is difficult to determine accurately due to the absence of a reliable ante-mortem test, differing scrapie test sensitivities, reliance on reporting of clinical suspects and the stigma attached to a declaration of scrapie occurrence. Mandatory sampling of at risk animals, such as fallen stock, often leads to discovery of cases, particularly those of atypical scrapie.55 Stringently enforced import regulations have enabled island countries such as Australia and New Zealand to maintain what is generally regarded as disease-free status although atypical scrapie has been detected in these and other countries where classical scrapie has not been reported.
Susceptibility to scrapie, both classical and atypical, is determined by the prion protein gene of the animal. Researchers demonstrated that sheep experimentally infected with a pool of brains from classical scrapie-affected sheep (named sheep scrapie brain pool 1, SSBP/1) had different susceptibilities resulting in varying incubation periods. The incubation period was controlled by a single gene (Sip gene) with sA (short incubation) or pA (prolonged incubation) alleles.96 It was subsequently shown that the Sip gene is identical to the PrP gene, which may vary between individual sheep and goats: multiple mutations (polymorphisms) exist, some of which are associated with differential susceptibility to scrapie, including disease phenotypes. Three codons in the sheep prion protein gene are important with regard to susceptibility: codon 136 (encoding arginine A or valine V), codon 154 (encoding arginine R or histidine H) and codon 171 (encoding glutamine Q or arginine R), usually written as VRQ/VRQ, ARQ/ARQ etc. Experimental studies in sheep infected with bovine spongiform encephalopathy, SSBP/1 and another isolate, CH1641 from a Cheviot sheep,56 have shown that polymorphisms at codons 136 and 171 affected disease incidence and incubation time.63 PrP alleles encoding for valine at codon 136 and glutamine at codon 171 (VRQ) are associated with increased susceptibility to classical scrapie, whereas those encoding for arginine at codon 171 (ARR) are associated with increased resistance.11 These findings formed the basis for control programs to eradicate classical scrapie, such as the National Scrapie Plan in Great Britain (Table 1). Histidine at codon 154 also appears to have some protective effect against classical scrapie.51
Table 1 Classical scrapie risk categories used in the National Scrapie Plan in Great Britain
| Risk category | Genotype | Resistance/susceptibility to classical scrapie |
| 1 | ARR/ARR | Sheep that are genetically most resistant to classical scrapie. |
| 2 | ARR/AHQ | Sheep that are genetically resistant to classical scrapie, but will need careful selection when used for further breeding. |
| 3 | AHQ/AHQ | Sheep that genetically have little resistance to classical scrapie and will need careful selection when used for further breeding. |
| 4 | ARR/VRQ | Sheep that are genetically susceptible to classical scrapie and should not be used for breeding unless in the context of a controlled breeding programme. |
| 5 | AHQ/VRQ | Sheep that are highly susceptible to classical scrapie and should not be used for breeding. |
Although ARR/ARR sheep are the most resistant sheep, resistance is not absolute since there have been rare cases of classical scrapie in sheep of this genotype.79 Other polymorphisms linked with resistance to classical scrapie have been found subsequently, such as K176 (encoding lysine) or T137 (encoding threonine).203
Susceptibility to atypical scrapie is also dictated by polymorphisms at these three codons but, contrary to classical scrapie, cases occur particularly in AA136 sheep, even ARR/ARR sheep, and sheep carrying the AHQ allele, whilst being rare in sheep with the VRQ allele.14
Polymorphisms at codon 136, 154 and 171 are irrelevant for susceptibility to classical scrapie in goats, which are either ARQ/ARQ or AHQ/AHQ. However, other polymorphisms have been associated with increased resistance to classical scrapie in goats; the most important being at codons 222 (encoding lysine K), 146 (encoding serine S or aspartate D) and 211 (encoding glutamine Q)202 (Table 2). Similar to sheep, H154 is associated with increased susceptibility to atypical scrapie in goats30 although it may offer some protection against classical scrapie.18, 153
Table 2 Classical scrapie risk in goats
| Genotype | Resistance/susceptibility to classical scrapie |
| K222 | Goats that are genetically most resistant to scrapie. |
| H154 | Goats that have some genetic resistance to scrapie. |
| Q222 | Goats that are highly susceptible to scrapie. |
M…Methionine, P…Proline, N…Asparagine, I…Isoleucine
Many studies have been carried out in various breeds to determine the frequency of alleles associated with resistance. These have shown that certain breeds may have little resistance in general or certain polymorphisms are breed- or region-specific. For example, Icelandic short-tailed sheep were all homozygous Q171.196 Whilst a high frequency of resistant genotypes (Q211, S146 and K222) was detected in the goat population in Greece,103 it was considerably lower in surveys in the United Kingdom (less than 7 per cent) and Boar goats were the main carriers of the S146 allele,64, 65 which was also the case for goats in Cyprus.153
Increasing the prevalence of the ARR allele has been shown to reduce the overall risk of scrapie at the population level in sheep.150 Indeed, genetic selection for scrapie resistance has led to a great reduction in the number of classical scrapie cases particularly in Europe, and especially in sheep where the influence of the prion protein gene on susceptibility has been studied the longest. The failure to effectively control scrapie in some affected European countries in the period between 2002 and 2012 was linked to regional variations in the implementation of genetic and non-genetic control measures. By contrast, the prevalence of atypical scrapie in Europe has been relatively consistent across countries, with approximately 6 cases per 10,000 tests. Testing for scrapie included fallen stock and healthy slaughter animals and was not just limited to neurological cases. Atypical scrapie is much rarer in goats, with an approximate prevalence of 1 per 10,000 tests, and only reported in five European countries.48
Risk factors for the occurrence of classical scrapie other than genotype were found to be flock size (higher with larger flocks), soil drainage (higher with naturally wet soils), purchase of replacement sheep through a market, spread of sheep compost on the land and disposal of placentae in the compost.87, 188
Scrapie is a disease of adult sheep and goats, usually between two and five years of age in classical scrapie42 although there are some reports of cases in sheep less than a year old.102, 172 Age-associated differences in scrapie incidence may be due to genotype, the level and age at exposure. Atypical scrapie generally affects older animals, above 5 years old.14, 30 Within-flock incidence can vary and is dependent on genotype distribution in the flock but can range from 1 to 20 per cent.42 In outbreaks of classical scrapie in sheep and goats, which led to herd cull with examination of central nervous and peripheral tissue for scrapie, a prevalence of above 30 per cent has been reported.33, 60
Studies that determined presence of prion protein or infectivity in various organs, body tissues or fluids were vital to gaining a better understanding of the routes of transmission in scrapie. It is commonly accepted that the risk of horizontal transmission is greater in animals that have wide-spread accumulation of scrapie-associated PrP in the body, which is ultimately linked to prion protein genotype and scrapie strain.
In classical scrapie, transmission occurs both vertically and horizontally. Infectivity has been demonstrated in foetal tissues of a scrapie-infected sheep183 and prion protein detected in foetal tissues by in vitro assay protein misfolding cyclic amplification (PMCA).58 This is indicative of maternal, pre-natal transmission. Whether scrapie can be transmitted via semen or embryo transfer is still debated because studies have produced conflicting results, and interpretation is often difficult because of small sample sizes. Prions have been found in semen from scrapie-affected rams using PMCA and infectivity confirmed in one study161 whereas there was no evidence of infectivity in semen in another study.167 Embryos collected from naturally-infected donors and implanted in scrapie-free sheep did not develop scrapie131, 212 but scrapie was confirmed in lambs produced by embryo transfer in experimentally infected recipient ewes.57 It was concluded that the risk of transmitting scrapie via embryo transfer was negligible when using ARR homozygous or heterozygous embryos and adhering to OIE procedures relating to embryo transfer.47 An epidemiological study found increased incidence of scrapie in the offspring of scrapie-affected dams but not in the offspring of scrapie-affected sires. That is suggestive of transmission from mother to offspring.90 Such transmission may occur perinatally or postnatally in sheep and goats via contaminated placental tissues7, 168 or by ingesting colostrum and milk.114, 120 The latter may be more efficient in the presence of concomitant lentivirus infection of dams.122, 128
Transmission of classical scrapie can also occur in the absence of contact with placentae or milk,163 and many studies have now confirmed infectivity or presence of the responsible prion in a range of secretions and excretions, such as faeces,192 urine162 and saliva189 and even skin,195 which may all contaminate the environment.76 This, together with the considerable resistance of prions to disinfection and biological inactivation, is the reason why scrapie is very difficult to eradicate once it has been established in a flock. It has been shown that the classical scrapie agent can remain biologically active in the environment for at least 16 years.61 Sheep on contaminated pasture can be infected in the absence of contact with infected sheep.43 Furthermore, prions can be detected on objects such as fence posts and water troughs133 and contact with such objects can result in disease in sheep although weathering may reduce infectivity.113 Disinfection with sodium hypochlorite may also not prevent re-infection in contaminated environments.85
Iatrogenic transmission of classical scrapie has also been reported in sheep vaccinated against Mycoplasma agalactiae.24 There was no evidence that the use of pituitary-derived hormones used in sheep breeding were associated with an increased risk for development of atypical scrapie.134
Little is known about transmission of atypical scrapie if it occurs at all. Results from an epidemiological study suggested that atypical scrapie was either not contagious or had a very low transmissibility under natural conditions.53 No significant risk factors for transmission were found in atypical scrapie case control studies.40, 54, 92 Although multiple cases of atypical scrapie have been found in flocks and atypical and classical scrapie may co-exist in flocks, there is currently no epidemiological evidence that these two disease types are linked or caused by lateral transmission149 and it may be a spontaneous disease.14 Experimental studies confirmed that atypical scrapie can be transmitted via the oral route177 and that the atypical scrapie agent can ‘shift’ into the classical scrapie strain CH1641 upon passage within sheep178 but it is not known whether this can occur under natural conditions.
Pathogenesis
After natural or oral infection, the scrapie agent generally accumulates in tissues of the lymphoreticular system prior to neuroinvasion via ascending neural and hematogenous pathways.
The pathogenesis of classical scrapie has been studied in naturally infected, highly susceptible VRQ/VRQ sheep killed at sequential time-points:207, 208 PrPSc was detected in the gastrointestinal tract-associated lymphoid tissue of the palatine tonsil, the Peyer’s patches of the small intestine and retropharyngeal, caudal jejunal and ileocaecal lymph nodes at 2 months of age. Immuno-labelling in lymph nodes was restricted to tingible body macrophages. PrPSc immune-labelling was more widespread, involving other peripheral lymph nodes and spleen, at 3 months of age and also included follicular dendritic cells. PrPSc was found in the enteric nervous system from 5 months of age. Neuroinvasion was confirmed from 10 months of age: PrPSc was detected in coeliac and mesenteric ganglion complex, dorsal motor (parasympathetic) nucleus of the vagus nerve in the medulla oblongata and intermediolateral column of thoracic spinal cord segments T8-T10. At the end-stage (26 months of age), the entire enteric nervous system, brain and spinal cord was affected. The circumventricular organs that have a less effective blood-brain barrier, may also act as an entry point for prions to the central nervous system, i.e. via the haematogenous route since PrPSc has been found in these organs.179
It has been shown that classical scrapie can be transmitted by blood transfusion, and infectivity in blood increased gradually during the incubation phase in VRQ/VRQ sheep.94 Monocytes, lymphocytes and platelets were found to carry infectivity in blood, originating from lymphoid germinal centers and possibly bone marrow.123
The risk of scrapie infection is correlated with the development of the Peyer’s patches and therefore highest in the first year of life. Thereafter susceptibility declines, presumably due to a reduction in surface area of ileal Peyer’s patches and lymphoid follicle density.185
Differences in the time-course of neuroinvasion and peripheral involvement may occur depending on prion protein genotype or prion protein strain. In naturally infected ARQ/ARQ Suffolk sheep, PrPSc was first detected at 8 months in sequential tonsillar biopsies starting at 3 months of age.100 No PrPSc immune-labelling was detected in ARR/VRQ sheep in a flock with natural scrapie up to 9 months of age whilst it was present in VRQ/VRQ sheep.6 PrPSc was found in a 3 month-old ARR/VRQ lamb in a different study.52 In some sheep or genotypes (ARQ/ARQ and ARQ/AHQ Sarda sheep) no evidence of peripheral PrPSc deposition was found, even though it was present in central and enteric nervous systems.129
Oral infection of sheep with classical scrapie resulted in early and consistent PrPSc accumulation in peripheral lymphoid tissue in VRQ/VRQ, VRQ/ARQ and ARQ/ARQ sheep whereas it was minimal, inconsistent or occurred later in VRQ/ARR and ARQ/ARR sheep, and ARQ/ARR sheep had the longest survival time.72
The finding of regular peripheral PrPSc accumulations in sheep with classical scrapie enables diagnosis in the live animal by examination of lymphoid tissue that is accessible thorough biopsies, such as third eyelid,144 tonsil100 and rectal mucosa.67
Although the pathogenesis of classical scrapie in goats is fundamentally the same, less is known in relation to breed, prion protein genotype and age. This was studied in 200 selected goats from a classical scrapie outbreak which demonstrated that peripheral PrPSc accumulation was frequent (68 of 72 scrapie-affected goats) although it did not necessarily include lymphoid tissue of the rectal mucosa. Also goats homozygous for isoleucine at codon 142 (II142) were more frequently infected than goats with a methonine allele (IM142 or MM142).71 Furthermore, the absence of detectable PrPSc in the enteric nervous system and thoracic spinal column, despite presence in the brain, suggested that the hematogenous route contributed significantly to neuroinvasion.70 The consistent involvement of the coeliac and mesenteric ganglion complex in scrapie-affected goats in Cyprus suggested that the spread from the enteric to the nervous system is similar to sheep.143
The pathogenesis of atypical scrapie is largely unknown, which is complicated by the majority of cases being detected by active surveillance of fallen stock or healthy slaughter animals and tissues for further examination are limited (brain only) or unsuitable (autolysed). Unlike classical scrapie, PrPSc is not found in peripheral lymphoid tissue.175 This suggests that the agent is predominantly restricted to the central nervous system.
Transmission studies in transgenic mice carrying the ovine prion protein gene (tg338 mice), however, demonstrated infectivity – although not always consistently – in peripheral tissues from field cases, such as retropharyngeal, popliteal, pre-scapular and parotid lymph nodes, but it was impossible to determine whether this was due to oral exposure similar to classical scrapie or due to centripetal spread from the brain.8
Oral challenge of sheep within 24 hours after birth resulted in atypical scrapie with infectivity demonstrated in the distal ileum at 24 months of age,177 i.e. considerably younger than that seen in natural cases of atypical scrapie.
It is currently not known which experimental infection route best mimics natural disease but the intracerebral route may be more suitable for a disease that presumably originates in the brain. AHQ/AHQ sheep inoculated intra-cerebrally with atypical scrapie brain homogenate presented with a uniform disease phenotype and had survival times between 378 and 1057 days, the mean of which was approximately half of the average age at death in field cases.176
Clinical signs
Both classical and atypical scrapie are slowly progressive neurological diseases. Although PrPSc is found throughout the brain in clinically affected animals, the sometimes poor correlation between clinical disease or clinical signs in particular, and detectable PrPSc raises questions about the cause of clinical signs.111
A detailed description of the clinical presentation of classical scrapie in sheep is given by Parry,154 although that was written prior to the discovery of atypical scrapie and the use of more sensitive post-mortem tests.
Most cases of scrapie have an insidious onset and progress slowly over several weeks but progression over several months has also been observed.16, 154 Usually, only one or a few animals are affected simultaneously in a flock or herd. Early clinical signs may not be noticed, especially in flocks or herds that are not closely monitored. It is questionable whether acute or peracute scrapie cases occur that do not display premonitory signs of illness. In one survey, 16 per cent of confirmed classical scrapie cases were found dead without exhibiting signs before death.28 It is more likely that clinical signs are not recognised or clinical signs are too subtle to be associated with scrapie. That would explain why so many cases, particular atypical cases, are detected by active surveillance in supposedly clinically healthy animals or fallen stock.
Early clinical signs can be fairly non-specific and are thus most likely to be observed by someone familiar with the animal. Farmers may suspect early scrapie in sheep or goats before neurological signs are evident due to their experience with the disease. These early clinical signs include weight loss and subtle behavioural changes, such as separation from other animals or changed behaviour during milking.
The main difference in the clinical presentation between classical and atypical scrapie is pruritus, which is largely absent in atypical scrapie. However, not all classical scrapie cases present with pruritus. Nevertheless, if pruritus is present it is usually easily spotted, especially in more advanced cases due to the loss of wool or hair as a result of frequent rubbing, nibbling and scratching. Circling has been described in cases of atypical scrapie, particularly in experimentally-induced disease.173, 175-177 This sign is rarely observed in sheep with classical scrapie although it has been reported in goats.109 In general, detailed descriptions of clinical signs of atypical scrapie are rare because very few natural cases are identified as clinical suspects.
Clinical signs can be crudely grouped into three different categories, viz. changes in mental status, behaviour and activity; changes in sensory responses and changes in movement.
Changes in mental status, behaviour and activity
These are often the earliest clinical signs noticed. There may be separation from the rest of the flock or herd and the animal may become more prone to flight when approached and thus difficult to catch. Classical scrapie-affected animals, particularly goats, may also appear hyper-alert, may become more excitable and display exaggerated responses to external stimuli, such as sudden movements or noises. Repeated startle responses to hand clapping were seen in approximately 30 per cent of 129 sheep with classical scrapie in Ireland.88 When left undisturbed, scrapie cases may appear somnolent or dull with a vacant gaze and low head carriage but this is more obvious in more advanced scrapie cases.
Sheep with classical scrapie tend to spend more time lying down than healthy sheep, are usually more inactive during these periods and are less socially responsive.86 Teeth grinding, either spontaneous or during handling, is frequent in sheep with classical scrapie. Appetite is usually not affected until the very late stage of the disease. Abnormal drinking patterns have also been reported in the early stages of classical scrapie; affected animals may drink more frequently but only small amounts taken.154 Difficult milking is a common sign in sheep and goats with classical scrapie.23
Changes in sensory responses
Classical scrapie is usually associated with increased grooming behaviour; affected animals spend more time rubbing on objects (e.g. fences, troughs), scratching (e.g. with horns), and nibbling themselves. The latter response is particularly frequent in goats.111 In the later stages, rubbing may be accompanied by lip-licking and smacking (‘spontaneous nibble reflex’). These responses to pruritus are, however, often only displayed when the animal is undisturbed or accustomed to an observing person. The response to pruritus is utilized as clinical test, i.e. scratching of the back often results in lip-licking and nibbling (termed ‘nibble reflex’ even though it is not a reflex (Figure 1)) but some animals may merely respond with head or body movements, which is why the term ‘positive scratch test’ may be more appropriate. Pruritus eventually leads to wool and hair loss, usually on the poll, around the tail base, lateral thorax and lower lateral neck in sheep111, 154 (Figures 2 and 3). Further consequences of intense rubbing may be discolouration, hyperpigmentation and lesions of the skin. Skin lesions initially present as superficial abrasions, excoriation with crusts if allowed to heal and lichenification. Secondary infections with skin pathogens, such as Staphylococus aureus, Dermatophilus congolensis and parapox virus (orf) may also occur.108
Although pruritus is generally associated with classical scrapie, it may not be present in all cases; this non-pruritic form was termed ‘ataxia type’, ‘paralytic form’ or ‘nervous form’102, 111, 148, 156 of scrapie. This is obviously dependent on the level of observation.
In a study of 162 classical scrapie-affected sheep, five (3 per cent) did not appear to be pruritic based on the absence of wool loss, scratch response and pruritic behaviour during 2.5 hours of passive observations.108 Such differences may be due to prion protein genotype118 and strain, such as atypical scrapie or CH1641 which usually do not cause pruritus in sheep.14, 178
There are infrequent reports of scrapie cases running into objects, possibly due to visual impairment.199, 204 This has been also described in atypical scrapie in both sheep and goats.141, 173
Easier to evaluate is the menace response, which may be absent in one or both eyes. This was observed for classical scrapie in approximately 10 per cent of sheep in a study in Ireland88 and 2 per cent of goats in a study in England.111 It was also reported for sheep with atypical scrapie,112, 176 (Figure 4) and may be a feature of cerebellar disease.
Changes in movement
Head tremor was seen in over 40 per cent of sheep88, 108, 209 and in 7-10 per cent of goats with classical scrapie.111, 218 It has also been described in sheep with atypical scrapie.112, 176 Frequency of occurrence increases as the disease progresses.
Gait abnormalities found in both scrapie types include incoordination, hypermetria (high stepping gait), jumping of the hind limbs (bunny hopping), a stilted or hypometric gait, lack of balance, falling and difficulty in rising, eventually resulting in permanent recumbency and death.16, 88, 111, 112, 141, 200, 210, 218 Collapsing episodes, which may be a form of cataplexy-narcolepsy and may be triggered by handling stress or when chased, have been reported in sheep and goats with classical scrapie.114, 121, 155, 170 Postural changes may be present in combination with gait abnormalities, such as a wide-based stance or crouching.88, 111
Circling, either spontaneous or when blindfolded, has been observed in sheep with atypical scrapie in both natural147, 173 and experimental disease.175, 176 It may also be seen occasionally in goats with classical scrapie.81, 109, 125
Other clinical signs
Hypersalivation, or more likely excess saliva due to swallowing deficits, which may increase the risk of aspiration pneumonia, may be observed in sheep and goats with classical scrapie.9, 23, 89, 127
Weight loss or loss of body condition is a frequent but non-specific sign, which has been described for all scrapie types in sheep and goats. There are no obvious physical changes (temperature, heart rate, respiratory rate) in scrapie cases.
There are no significant alterations in blood or cerebrospinal fluid that would be useful for ante-mortem diagnosis.
In the absence of a reliable live animal test, the clinical examination is currently the only method for diagnosis in live animals. However, as none of the clinical signs are pathognomonic, not all scrapie cases present with the full range of clinical signs and the clinical picture is affected by scrapie strains, confirmative tests following post-mortem are always necessary. Few protocols have been published to aid in the clinical diagnosis of classical scrapie in sheep37, 210 or scrapie in general116 based on the most frequent signs associated with the disease.
Other electrodiagnostic tests have been studied for use in sheep with scrapie, such as electroretinograms159, 180 and auditory evoked potentials,117 and although abnormalities were detected, these tests are expensive, time-consuming and have limited practical value.
Classical scrapie in goats
Atypical scrapie in sheep
Classical scrapie in sheep
Pathology
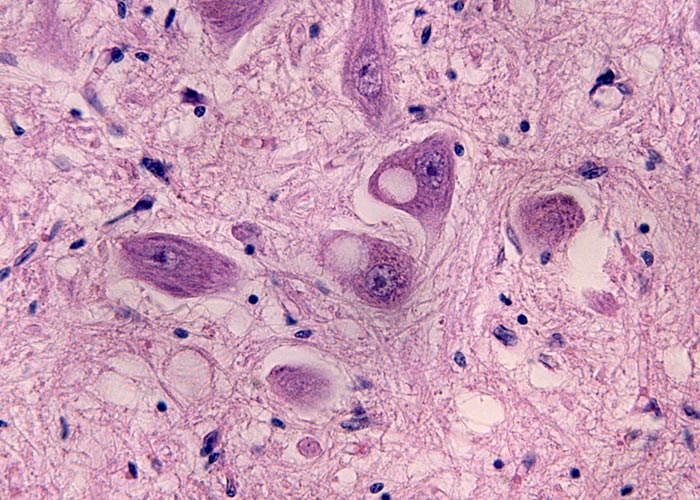
There are no pathognomonic gross lesions associated with scrapie. Morphological microscopic lesions are restricted to the central nervous system (CNS). The consistent microscopic lesions are vacuolar changes in the grey matter neuropil or neuronal perikarya, which are usually bilateral symmetrical, accompanied by astrocytosis.181, 217 Other forms of neuronal degeneration, such as chromatolysis or neuronophagia, may also be seen, and in some cases perivascular amyloidiosis may be present.215, 217 The latter appear to be more frequent in sheep with a V136 allele.130
Intraneuronal vacuolation is not a feature in atypical scrapie cases140 but descriptions of histopathological changes in atypical scrapie are limited because the brain material from confirmed cases is often autolysed.
Pathological changes have been observed in the retina, characterized by atrophy and loss of nuclei in nuclear layers and Müller glia hypertrophy in sheep with classical scrapie.93
Diagnosis
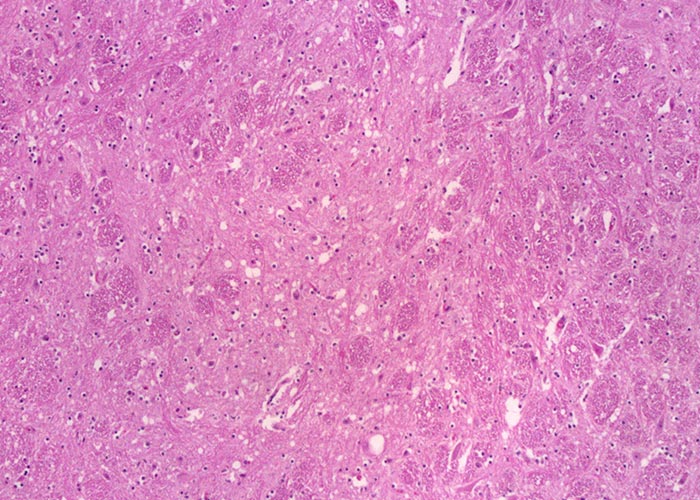
The standard method is the microscopic examination of the brain. Histological examination should be performed on formalin-fixed, paraffin embedded brain sections that are stained with haematoxylin and eosin (H&E). The histopathological changes in the CNS, predominantly vacuolation, are visible in the target areas at the level of the obex for classical scrapie, i.e. the dorsal motor (parasympathetic) nucleus of the vagus nerve (DMNV) (Figure 5), and the spinal tract nucleus of the trigeminal nerve (STNT) (Figure 6). In atypical scrapie, vacuolar changes are less prominent in the brainstem whereas neuropil vacuolation may be observed in the molecular layer of the cerebellum and the cerebral cortex.16
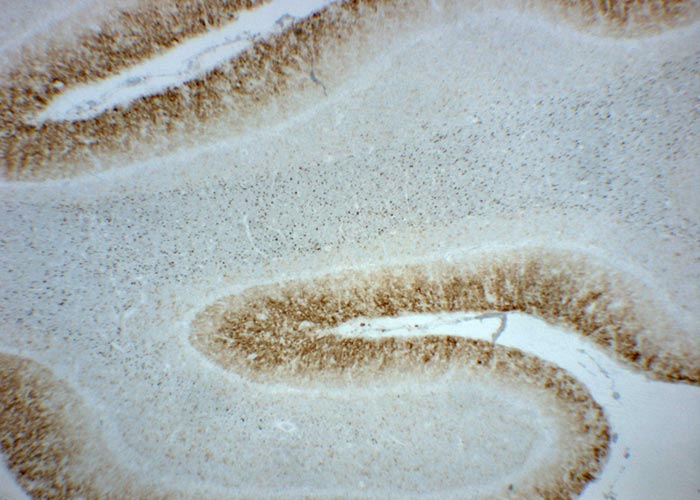
Immuno-histochemical analysis (IHC) can also be performed on paraffin embedded brain tissues in order to highlight the presence of PrPSc accumulation. The samples are deparaffinated, rehydrated, pretreated with 98 per cent formic acid and autoclaved at 121°C, then incubated at 4°C with monoclonal primary antibody anti PrPSc, incubated with avidin-biotin-peroxydase, reacted with chromogen 3-3’ diaminobenzidine (DAB), and counter-stained with Mayer’s hemalum. Different immuno-histochemical types of PrPSc deposition (see Bovine spongiform encephalopathy) can be observed in the brain of scrapie affected animals, which can be used to distinguish different scrapie strains. In sheep with classical scrapie, PrPSc is most frequently detected in the DMNV and less frequently and intensely in other neuroanatomical sites of the brainstem, such as solitray tract nucleus, STNT, midline raphe, olivary nuclei and accessory cuneate nucleus.164 PrPSc is frequently visible as granular or punctate deposit in the DMNV and STNT.182 Glial-type labelling with PrPSc is able to identify deposits branching out from the nucleus of glial cells on their processes and giving them a stellate appearance is predominantly found in the molecular layer of the cerebellum and granular pattern is frequently present in the granular layer of the cerebellum (Figure 7). A detailed description of the PrPSc types in sheep brains with scrapie has been published.69
The distribution of PrPSc is more restricted in atypical scrapie in sheep and the severity usually reduced when compared with classical scrapie. Immunolabelling is consistently greater in cerebellum than in the brainstem, and if detected at the level of the obex, restricted to the STNT.14, 140 That is why examination of the cerebellum, in addition to the brainstem, is advisable in small ruminants. Immunolabelling types of PrPSc are less variable in brains from atypical scrapie cases compared to classical scrapie, and intracellular and stellate PrPSc deposits, which feature in classical scrapie, are not evident in atypical scrapie brains. Fine granular immunolabelling in both molecular and granular layers of the cerebellum (Figure 8), but not stellate-type labelling, are most frequently seen in atypical scrapie brains.140 PrPSc was not detected in the cerebellum in a goat with atypical scrapie but the presence of immunolabelling in more rostral areas and only in the STNT in the brainstem171 is consistent with the findings in sheep with atypical scrapie. More data on goats with atypical scrapie is needed.
Another PrP-based confirmatory test is immunoblotting, which demands fresh or frozen tissue. It is also suitable for use on autolysed tissue95 that cannot be fixed for immunohistochemical examination. After digestion with proteinase K and labelling with anti-PrP-antibodies, three immunostained bands, di-, mono- and unglycosylated, of proteinase resistant PrPSc (PrPres) are distinguishable (Figure 9). The highest (diglycosylated) band of approximately 27 kDa and the lowest (unglycosylated) band of approximately 19 kDa occur in classical scrapie.187 Banding patterns are different in atypical scrapie (Figure 10) where an additional lower band of around 11 kDa is detectable.14, 201

Figure 9 Western Blotting analysis of positive and negative classical scrapie cases
C-: negative control; C+: classical scrapie control; Lanes S1, S3, and S4: classical scrapie cases; lane S2: negative case. Immunodetection was performed with monoclonal antibody P4. This image was kindly provided by Dr. Mazza Maria, IZS Turin.

Figure 10 Western Blotting analysis of classical and atypical scrapie cases
C-: negative control; C1+: classical scrapie control; C2+: atypical scrapie control (Nor 98); Lanes S1: negative case; Lanes S2: atypical scrapie case; Lane S3: classical scrapie case. Immunodetection was performed with monoclonal antibody P4. This image was kindly provided by Dr. Mazza Maria , IZS Turin.
In addition to comparing molecular mass and glycoform ratio using a single antibody, monoclonal antibodies targeted at different epitopes of the prion protein can also be used to visualize different PrPres fragments in Western immunoblots. This enables differentiation of classical scrapie from bovine spongiform encephalopathy (BSE) in sheep or goats and CH1641 scrapie.97, 124, 187 The latter is a distinct classical scrapie strain in both sheep and goats with molecular properties similar to BSE, which is thus diagnostically more challenging.91 Similarly, strain differentiation is possible by immunohistochemical examination of fixed tissue using a panel of different monoclonal and polyclonal antibodies. These recognize specific PrPSc sequences in specific cell types of the brain and lymphoreticular system (“epitope mapping”), which result in immunolabelling patterns that are characteristic for BSE, classical scrapie and CH1641 scrapie.98, 99, 198
In countries where animals are actively monitored for scrapie, for example by testing fallen stock or healthy slaughter animals, rapid (screening) tests are usually applied to brain samples and any positive or inconclusive sample is then subsequently examined by immunohistochemistry and/ or Western immunoblotting for disease confirmation as described in the OIE Manual of diagnostic tests and vaccines for terrestrial animals.145 A range of screening tests is available, e.g. enzyme-linked immunosorbent assays (reviewed by59), which either detect PrPres or do not use any proteinase digestion at all, although their diagnostic sensitivity may not be equal, which affects the efficacy of active surveillance systems.19 These tests may be inferior to immunohistochemistry in their sensitivity, particularly in goats, where cases may be missed if only screening tests are used.32, 71, 139, 151
As PrPSc is usually detectable in lymphoid tissue in most sheep and goats with classical scrapie and generally earlier than in brain tissue, this tissue type could also be examined for scrapie diagnosis although the rapid screening tests have not been formally evaluated for use on tissue other than brain. Accessible lymphoid tissues suitable for testing in live animals are the nictitating membrane, palatine tonsil and rectal tissue containing recto-anal mucosa-associated lymphoid tissue (RAMALT). Only the latter has been proposed as a live animal test66, 68 because it is less challenging, can be done under local anaesthesia of the rectum, usually yields adequate sample size, and is commercially available. Single-use specula exist to aid in the collection of these biopsies. Diagnostic sensitivity is obviously dependent on the peripheral distribution of PrPSc, which is influenced by strain (e.g. no detectable PrPSc in atypical scrapie), genotype (e.g. more limited peripheral distribution in sheep carrying an ARR allele) and species (less successful in goats with classical scrapie.71
The probability of detecting scrapie in small ruminant carcasses is highest if both brain and lymphoid tissue are tested because of PrPSc accumulation in peripheral tissue prior to neuroinvasion in most cases of classical scrapie. Limiting testing to brain by rapid tests may identify classical scrapie only in the last quarter of the incubation period.10 Testing retropharyngeal lymph node tissue using rapid tests has been considered for the national scrapie surveillance programme in New Zealand.107, 211 Other ultrasensitive PrP-based tests that exist make use of the ability for prions to convert PrPC into PrPSc or amyloid fibrils, such as PMCA and Real-Time Quaking Induced Conversion (RT-QuIC).104 These could potentially be used as a live animal test since PrPSc has been detected by PMCA in blood from classical scrapie-infected sheep.197 However, these tests are currently only used in specialist research facilities and have not been tested on a large number of animals infected with the various scrapie strains that exist.
For surveillance purposes, it is usually sufficient to detect scrapie and to further classify it as atypical scrapie, classical scrapie or BSE. Some scrapie isolates, particularly CH1641, can produce equivocal results where BSE cannot be excluded, and these are usually further investigated by transmission to a standardized panel of transgenic mice. The gold standard for strain characterization of the agent is still the mouse bioassay, which is however lengthy, expensive and impractical on a large scale although the creation of transgenic mice carrying the ovine prion protein gene has resulted in greatly reduced incubation periods. Strains can be differentiated in mice by clinical signs,41 incubation period,21 transmissibility,194, 214 histopathological lesion profile,22 imunolabelling profile,13 resistance to chemical or physical inactivation,191 resistance to proteinase K digestion126 and PrPres immunoblot profile.193 An overview of rodent models available for strain typing has been provided.78 Additional transgenic mice strains may become available as research tools for prion disease research.
Multiple classical scrapie strains exist, as demonstrated by mouse bioassays, such as ME7, 221C21 or strains that transmit poorly to wild-type mice.194 The current immunoblotting methods are unable to distinguish these strains, except for CH1641, but this may be possible by immunohistochemistry. Different immune-labelling patterns in the brain in classical scrapie-affected ARQ/ARQ sheep from different countries is suggestive of different scrapie strains73 although these patterns may also be affected by PrP gene polymorphisms at codons 136, 154 and 171.182 Vacuolar lesion profiles were found to be less useful to differentiate scrapie strains due to their high individual variability.12, 73
Differential diagnosis
Scrapie is a slowly progressive neurological disease, which should rule out diseases with an acute onset and short clinical course, such as hypomagnesaemia, botulism, rabies and pseudorabies (Aujezsky’s disease) although the level of prior observation is crucial in this respect. Infectious neurological diseases, such as Aujeszky’s disease or listeriosis, are usually associated with fever or other distinct clinical signs (facial paralysis in listeriosis) although the latter has been confused with classical scrapie in goats.109 Additional diagnostic tests, such as serology or blood biochemistry can be used to rule out other diseases (hypomagnesaemia, maedi-visna, caprine arthritis-encephalitis, ketosis) or there could be a response to treatment (polioencephalomalacia/ cerebrocortical necrosis/ thiamine deficiency). Abscesses, tumours or other space-occupying processes in the brain (e.g. cysts caused by larvae of the tapeworm Taenia multiceps) may cause clinical signs similar to atypical scrapie (circling) depending on the location of the lesion but are often also associated with extreme dullness, which is unusual for scrapie unless at the terminal stage. An overview of neurological diseases in sheep and goats and a list of diseases confused with scrapie is given by Brewer20 and Scott and Henshaw170 respectively. In a Swiss study of small ruminants with neurological signs, a goat with a brain tumour and ectoparasites had a clinical picture similar to scrapie.136
Pruritus can be persisting and caused by ectoparasites. Pruritus caused by Psoroptes ovis can be diagnosed by careful clinical examination coupled with parasitological examination of appropriate skin samples. Pruritic scrapie cases usually lack the skin lesions (crusts, serum-oozing papules) seen in sheep with mange. Emaciation may be caused by malnutrition (nutritional, bacterial or parasitic causes), which may also affect fleece quality, or dental disease but should not cause any neurological signs.
A range of plant poisonings can cause syndromes characterized by gait abnormalities and tremor142 but these affect usually groups of animals, have an acute onset and the clinical history may allow differentiation from scrapie.
Immunohistochemically, it has been shown that increased PrP immunolabelling in the brain may occur in a range of neurological conditions of sheep; such occurrence needs to be distinguished from immunolabelling in scrapie cases. Labelling of neurons has been associated with axonal swelling, neuronal necrosis and lysosomal storage disorders whilst labelling in glial cells was associated with bacterial infections and linked to possible oxidative stress.101 These studies support previous suggestions that altered PrP expression or transcription is associated with oxidative stress.
Control
As scrapie is a fatal disease and successful treatment does not exist, disease prevention is the only long-term control option once the diagnosis has been established.
Passive surveillance, which relies on reporting and examination of clinical suspects, is often the only method in many countries to monitor the disease but is less likely to detect disease particularly if present at low prevalence. Clinical suspects diagnosed as atypical scrapie are extremely rare, even in countries where the disease occurs, and targeted surveillance for scrapie by testing fallen stock and healthy slaughter animals is more likely to identify the disease.137
To determine the classical scrapie status of a country, zone, compartment or establishment certain criteria are necessary according to the World Organisation for Animal Health (OIE) Terrestrial Animal Health Code:146
- A risk assessment identifying all potential factors for scrapie occurrence (e.g. feeding practices, import of feedstuffs, sheep and goats, semen and embryos) and their historic perspective;
- an awareness programme to enable recognition of the clinical signs and
- a surveillance and monitoring system.
Several conditions have to be met before scrapie freedom can be considered, which includes testing of animals for scrapie and taking appropriate measures with regards to feeding practices and imports of animals to prevent introduction of disease, which are outlined in articles 14.8.2 – 14.8.5 of the chapter.
Scrapie prevention in countries with endemic classical scrapie is primarily achieved by genotype selection, i.e. removal of sheep with a susceptible prion protein genotype (VRQ) and increasing the number of sheep with a resistant genotype (ARR) will reduce susceptibility to classical scrapie in sheep. Such programmes are in place in many countries. A breeding program was set up within the European Union in 2003 to select for resistant genotypes138 although this may ultimately increase the number of animals in the population that are susceptible to atypical scrapie. However, detection of atypical scrapie does not usually result in the same strict legislative measures as for classical scrapie. In the European Union, any holding (establishment) where a confirmed case of atypical scrapie has been identified is currently subject to surveillance for two years. That involves testing of healthy slaughter animals and fallen stock over the age of 18 months.48
Studies in sheep have not shown any unfavorable association between the ARR allele and milk performance and certain production and performance traits.3, 38, 39
Reliance on tests following post-mortem alone to identify scrapie-infected animals without control involving genotype selection is unlikely to eradicate the disease.32 A similar genotype-based approach has not yet been tested in goats although it should be equally effective. The lack of resistant genotypes in certain goat and sheep breeds in some countries may present a problem, and herd cull has been used as an alternative option to eliminate the disease. However, re-introduction of the disease is likely if restocking is carried out using animals with susceptible genotypes given the persistence of the scrapie agent in the environment. In Iceland where a genotype-based approach is impossible due to the lack of sheep with resistant genotypes, current control measures include herd cull, destruction of hay, cleaning and disinfection of sheep barns and equipment by high pressure washing with soap and hypochlorite solution, replacing all worn materials in the sheep barn and/ or replacement with new buildings, restocking after a minimum of 2 years with lambs from scrapie-free areas remote from affected quarantine zones and continuous health control of new flocks to detect possible recurrence.61, 132 This has, however, not prevented re-occurrence. Mass restocking after whole herd cull increases the likelihood of re-occurrence of classical scrapie in goats.152
Scrapie eradication has become more important with the discovery of bovine spongiform encephalopathy (BSE) in cattle because the latter is zoonotic and its link to feeding of contaminated meat and bone meal to which small ruminants may have also be exposed. Whilst there has been no evidence of naturally occurring BSE in sheep so far,186 it was diagnosed in goats in the United Kingdom184 and France.50 The pathogenesis of BSE in goats is similar to classical scrapie in sheep206 and goats.1 The BSE agent can also be transmitted to sheep via blood.94 This is important in terms of risk for humans, should the BSE agent be confirmed in sheep under field conditions. Sheep were also used as a model to study transmission of variant CJD disease by blood products in humans because of similarities between the pathogenesis of TSEs in sheep and variant CJD in humans. Clinically, BSE is indistinguishable from classical scrapie in sheep110 and goats111 based on experimental studies, although BSE-affected sheep generally present with pruritus. BSE can be differentiated from scrapie by the diagnostic tests mentioned above although classical scrapie-BSE co-infection, if it occurs, may be more difficult to identify and may require mouse bioassay or other tests, like PMCA.174
The exclusion of known infectious materials (specified risk materials) from the food chain in the European Union is another measure to minimise exposure of humans to TSEs from any ruminant.
References
- AGUILAR-CALVO, P., FAST, C., TAUSCHER, K., ESPINOSA, J. C., GROSCHUP, M. H., NADEEM, M., GOLDMANN, W., LANGEVELD, J., BOSSERS, A., ANDREOLETTI, O. & TORRES, J. M., 2015. Effect of Q211 and K222 PRNP polymorphic variants in the susceptibility of goats to oral infection with goat bovine spongiform encephalopathy. Journal of Infectious Diseases, 212, 664-72.
- AGUZZI, A., 1998. Protein conformation dictates prion strain. Nature Medicine, 4, 1125.
- ALEXANDER, B. M., STOBART, R. H., RUSSELL, W. C., O'ROURKE, K. I., LEWIS, G. S., LOGAN, J. R., DUNCAN, J. V. & MOSS, G. E., 2005. The incidence of genotypes at codon 171 of the prion protein gene (PRNP) in five breeds of sheep and production traits of ewes associated with those genotypes1,2. Journal of Animal Science, 83, 455-459.
- ALPER, T., CRAMP, W. A., HAIG, D. A. & CLARKE, M. C., 1967. Does the agent of scrapie replicate without nucleic acid ? Nature, 214, 764.
- ALPER, T., HAIG, D. A. & CLARKE, M. C., 1966. The exceptionally small size of the scrapie agent. Biochemical and Biophysical Research Communications, 22, 278-284.
- ANDRÉOLETTI, O., BERTHON, P., MARC, D., SARRADIN, P., GROSCLAUDE, J., VAN KEULEN, L., SCHELCHER, F., ELSEN, J. M. & LANTIER, F., 2000. Early accumulation of PrP(Sc) in gut-associated lymphoid and nervous tissues of susceptible sheep from a Romanov flock with natural scrapie. Journal of General Virology, 81, 3115-3126.
- ANDRÉOLETTI, O., LACROUX, C., CHABERT, A., MONNEREAU, L., TABOURET, G., LANTIER, F., BERTHON, P., EYCHENNE, F., LAFOND-BENESTAD, S., ELSEN, J.-M. & SCHELCHER, F., 2002. PrPSc accumulation in placentas of ewes exposed to natural scrapie: influence of foetal PrP genotype and effect on ewe-to-lamb transmission. Journal of General Virology, 83, 2607-2616.
- ANDRÉOLETTI, O., ORGE, L., BENESTAD, S. L., BERINGUE, V., LITAISE, C., SIMON, S., LE, D. A., LAUDE, H., SIMMONS, H., LUGAN, S., CORBIÈRE, F., COSTES, P., MOREL, N., SCHELCHER, F. & LACROUX, C., 2011. Atypical/Nor98 scrapie infectivity in sheep peripheral tissues. PLoS Pathogens, 7, e1001285.
- ANDREWS, A. H., LAVEN, R. & MATTHEWS, J. G., 1992. Clinical observations on four cases of scrapie in goats. Veterinary Record, 130, 101.
- ARNOLD, M. & ORTIZ-PELAEZ, A., 2014. The evolution of the prevalence of classical scrapie in sheep in great Britain using surveillance data between 2005 and 2012. Preventive Veterinary Medicine, 117.
- BAYLIS, M., CHIHOTA, C., STEVENSON, E., GOLDMANN, W., SMITH, A., SIVAM, K., TONGUE, S. & GRAVENOR, M. B., 2004. Risk of scrapie in British sheep of different prion protein genotype. Journal of General Virology, 85, 2735-2740.
- BEGARA-MCGORUM, I., GONZÁLEZ, L., SIMMONS, M., HUNTER, N., HOUSTON, F. & JEFFREY, M., 2002. Vacuolar lesion profile in sheep scrapie: factors influencing its variation and relationship to disease-specific PrP accumulation. Journal of Comparative Pathology, 127, 59-68.
- BENCSIK, A. & BARON, T., 2011. Histopathological studies of "CH1641-like" scrapie sources versus classical scrapie and BSE transmitted to ovine transgenic mice (TgOvPrP4). PLoS One, 6, e22105.
- BENESTAD, S. L., ARSAC, J. N., GOLDMANN, W. & NÖREMARK, M., 2008. Atypical/Nor98 scrapie: properties of the agent, genetics, and epidemiology. Veterinary Research, 39, 19.
- BENESTAD, S. L., AUSTBØ, L., TRANULIS, M. A., ESPENES, A. & OLSAKER, I., 2012. Healthy goats naturally devoid of prion protein. Veterinary Research, 43, 87-87.
- BENESTAD, S. L., SARRADIN, P., THU, B., SCHÖNHEIT, J., TRANULIS, M. A. & BRATBERG, B., 2003. Cases of scrapie with unusual features in Norway and designation of a new type, Nor98. Veterinary Record, 153, 202-208.
- BESNOIT, C. & MOREL, C., 1898. Note sur les lésions nerveuses de la tremblante du mouton. Revue Vétérinaire, 23, 397-400.
- BILLINIS, C., PANAGIOTIDIS, C. H., PSYCHAS, V., ARGYROUDIS, S., NICOLAOU, A., LEONTIDES, S., PAPADOPOULOS, O. & SKLAVIADIS, T., 2002. Prion protein gene polymorphisms in natural goat scrapie. Journal of General Virology, 83, 713-721.
- BOZZETTA, E., NAPPI, R., CRUDELI, S., MELONI, D., VARELLO, K., LOPREVITE, D., MELIS, P. G., MAZZA, M., COLUSSI, S., INGRAVALLE, F., RU, G., NONNO, R. & LIGIOS, C., 2011. Comparative performance of three TSE rapid tests for surveillance in healthy sheep affected by scrapie. Journal of Virological Methods, 173, 161-168.
- BREWER, B. D., 1983. Neurologic disease of sheep and goats. Veterinary Clinics of North America. Large Animal Practice, 5, 677-700.
- BRUCE, M. E., BOYLE, A., COUSENS, S., MCCONNELL, I., FOSTER, J., GOLDMANN, W. & FRASER, H., 2002. Strain characterization of natural sheep scrapie and comparison with BSE. Journal of General Virology, 83, 695-704.
- BRUCE, M. E., BOYLE, A. & MCCONNELL, I., 2004. TSE strain typing in mice. Techniques in prion research. S. LEHMANN and J. GRASSI. Basel, Birkhäuser Verlag. 132-146.
- CAPUCCHIO, M. T., GUARDA, F., POZZATO, N., COPPOLINO, S., CARACAPPA, S. & DI MARCO, V., 2001. Clinical signs and diagnosis of scrapie in Italy: a comparative study in sheep and goats. Journal of Veterinary Medicine Series A - Physiology, Pathology, Clinical Medicine, 48, 23-31.
- CARAMELLI, M., RU, G., CASALONE, C., BOZZETTA, E., ACUTIS, P. L., CALELLA, A. & FORLONI, G., 2001. Evidence for the transmission of scrapie to sheep and goats from a vaccine against Mycoplasma agalactiae. Veterinary Record, 148, 531-6.
- CASSARD, H., TORRES, J.-M., LACROUX, C., DOUET, J.-Y., BENESTAD, S. L., LANTIER, F., LUGAN, S., LANTIER, I., COSTES, P., ARON, N., REINE, F., HERZOG, L., ESPINOSA, J.-C., BERINGUE, V. & ANDRÉOLETTI, O., 2014. Evidence for zoonotic potential of ovine scrapie prions. Nature Communications, 5, 5821.
- CHANDLER, R. L., 1961. Encephalopathy in mice produced by inoculation with scrapie brain material. The Lancet, 277, 1378-1379.
- CHONG, A., KENNEDY, I., GOLDMANN, W., GREEN, A., GONZÁLEZ, L., JEFFREY, M. & HUNTER, N., 2015. Archival search for historical atypical scrapie in sheep reveals evidence for mixed infections. Journal of General Virology, 96, 3165-3178.
- CLARK, A. M. & MOAR, J. A., 1992. Scrapie: a clinical assessment. Veterinary Record, 130, 377-378.
- CLARK, W. W., HOURRIGAN, J. L. & HADLOW, W. J., 1995. Encephalopathy in cattle experimentally infected with the scrapie agent. American Journal of Veterinary Research, 56, 606-612.
- COLUSSI, S., VACCARI, G., MAURELLA, C., BONA, C., LORENZETTI, R., TROIANO, P., CASALINUOVO, F., DI SARNO, A., MANIACI, M. G., ZUCCON, F., NONNO, R., CASALONE, C., MAZZA, M., RU, G., CARAMELLI, M., AGRIMI, U. & ACUTIS, P. L., 2008. Histidine at codon 154 of the prion protein gene is a risk factor for Nor98 scrapie in goats. Journal of General Virology, 89, 3173-3176.
- COOK, R., BINGHAM, J., BESIER, A., BAYLEY, C., HAWES, M., SHEARER, P., YAMADA, M., BERGFELD, J., WILLIAMS, D. & MIDDLETON, D., 2016. Atypical scrapie in Australia. Australian Veterinary Journal, 94, 452-455.
- CORBIÈRE, F., CHAUVINEAU-PERRIN, C., LACROUX, C., LUGAN, S., COSTES, P., THOMAS, M., BRÉMAUD, I., CHARTIER, C., BARILLET, F., SCHELCHER, F. & ANDRÉOLETTI, O., 2013. The limits of test-based scrapie eradication programs in goats. PLoS One, 8, e54911.
- CORBIÈRE, F., PERRIN-CHAUVINEAU, C., LACROUX, C., COSTES, P., THOMAS, M., BRÉMAUD, I., MARTIN, S., LUGAN, S., CHARTIER, C., SCHELCHER, F., BARILLET, F. & ANDREOLETTI, O., 2013. PrP-associated resistance to scrapie in five highly infected goat herds. Journal of General Virology, 94, 241-5.
- CUILLÉ, J. & CHELLE, P. L., 1936. Pathologie animal-la maladie dite tremblante du mouton est-elle inoculable? . Comptes rendus hebdomadaires des séances de l'Académie des Sciences, 203, 1552–1554.
- CUTLIP, R. C., MILLER, J. M., HAMIR, A. N., PETERS, J., ROBINSON, M. M., JENNY, A. L., LEHMKUHL, H. D., TAYLOR, W. D. & BISPLINGHOFF, F. D., 2001. Resistance of cattle to scrapie by the oral route. Canadian Journal of Veterinary Research, 65, 131-132.
- CUTLIP, R. C., MILLER, J. M., RACE, R. E., JENNY, A. L., KATZ, J. B., LEHMKUHL, H. D., DEBEY, B. M. & ROBINSON, M. M., 1994. Intracerebral transmission of scrapie to cattle. Journal of Infectious Diseases, 169, 814-820.
- D'ANGELO, A., MAURELLA, C., BONA, C., BORRELLI, A., CARAMELLI, M., ELENA, C. M., JAGGY, A. & RU, G., 2007. Assessment of clinical criteria to diagnose scrapie in Italy. Veterinary Journal, 174, 106-112.
- DE VRIES, F., DRÖGEMÜLLER, C., HAMANN, H., ANDRZEJEWSKI, M., GANTER, M. & DISTL, O., 2004. Influence of prion protein gene polymorphisms on performance traits in German meat sheep breeds. Deutsche Tierärztliche Wochenschrift, 111, 349-354.
- DE VRIES, F., HAMANN, H., DRÖGEMÜLLER, C., GANTER, M. & DISTL, O., 2005. Analysis of associations between the prion protein genotypes and production traits in East Friesian milk sheep. Journal of Dairy Science, 88, 392-398.
- DEL RIO VILAS, V. J., VINK, W. D. & HUBBARD, R., 2010. A case-control study of atypical scrapie in GB sheep flocks. Preventive Veterinary Medicine, 96, 241-251.
- DELL'OMO, G., VANNONI, E., VYSSOTSKI, A. L., DI BARI, M. A., NONNO, R., AGRIMI, U. & LIPP, H. P., 2002. Early behavioural changes in mice infected with BSE and scrapie: automated home cage monitoring reveals prion strain differences. European Journal of Neuroscience, 16, 735-742.
- DETWILER, L. A. & BAYLIS, M., 2003. The epidemiology of scrapie. Revue Scientifique et Technique - Office International des Epizooties, 22, 121-143.
- DEXTER, G., TONGUE, S. C., HEASMAN, L., BELLWORTHY, S. J., DAVIS, A., MOORE, S. J., SIMMONS, M. M., SAYERS, A. R., SIMMONS, H. A. & MATTHEWS, D., 2009. The evaluation of exposure risks for natural transmission of scrapie within an infected flock. BMC Veterinary Research, 5, 38.
- DICKINSON, A. G. & FRASER, H., 1979. An assessment of the genetics of scrapie in sheep and mice. Slow transmissible diseases of the nervous system. Vol 1. S. B. PRUSINER and W. J. HADLOW. New York, Academic Press. 1, 367–385.
- DICKINSON, A. G. & OUTRAM, G. W., 1979. The scrapie replication-site hypothesis and its implications for pathogenesis. Slow transmissible diseases of the nervous system. Vol 2. . S. B. PRUSINER and W. J. HADLOW. New York, Academic Press. 2, 13-31.
- EFSA, 2011. Joint scientific opinion on any possible epidemiological or molecular association between TSEs in animals and humans. EFSA Journal, 9, 1945.
- EFSA, 2013. Scientific opinion on the risk of transmission of classical scrapie via in vivo derived embryo transfer in ovine animals. EFSA Journal, 11, 3080.
- EFSA, 2014. Scientific opinion on the scrapie situation in the EU after 10 years of monitoring and control in sheep and goats. EFSA Journal, 12, 3781.
- EFSA, 2015. Scientific opinion on a request for a review of a scientific publication concerning the zoonotic potential of ovine scrapie prions. EFSA Journal, 13, 4197.
- ELOIT, M., ADJOU, K., COULPIER, M., FONTAINE, J. J., HAMEL, R., LILIN, T., MESSIAEN, S., ANDRÉOLETTI, O., BARON, T., BENCSIK, A., BIACABÉ, A. G., BERINGUE, V., LAUDE, H., LE DUR, A., VILOTTE, J. L., COMOY, E., DESLYS, J. P., GRASSI, J., SIMON, S., LANTIER, F. & SARRADIN, P., 2005. BSE agent signatures in a goat. Veterinary Record, 156, 523-524.
- ELSEN, J. M., AMIGUES, Y., SCHELCHER, F., DUCROCQ, V., ANDRÉOLETTI, O., EYCHENNE, F., KHANG, J. V., POIVEY, J. P., LANTIER, F. & LAPLANCHE, J. L., 1999. Genetic susceptibility and transmission factors in scrapie: detailed analysis of an epidemic in a closed flock of Romanov. Archives of Virology, 144, 431-445.
- ERSDAL, C., ULVUND, M. J., BENESTAD, S. L. & TRANULIS, M. A., 2003. Accumulation of pathogenic prion protein (PrPSc) in nervous and lymphoid tissues of sheep with subclinical scrapie. Veterinary Pathology, 40, 164-174.
- FEDIAEVSKY, A., MAURELLA, C., NOREMARK, M., INGRAVALLE, F., THORGEIRSDOTTIR, S., ORGE, L., POIZAT, R., HAUTANIEMI, M., LIAM, B., CALAVAS, D., RU, G. & HOPP, P., 2010. The prevalence of atypical scrapie in sheep from positive flocks is not higher than in the general sheep population in 11 European countries. BMC Veterinary Research, 6, 9.
- FEDIAEVSKY, A., MORIGNAT, E., DUCROT, C. & CALAVAS, D., 2009. A case-control study on the origin of atypical scrapie in sheep, France. Emerging Infectious Diseases, 15, 710-718.
- FEDIAEVSKY, A., TONGUE, S. C., NÖREMARK, M., CALAVAS, D., RU, G. & HOPP, P., 2008. A descriptive study of the prevalence of atypical and classical scrapie in sheep in 20 European countries. BMC Veterinary Research, 4, 19.
- FOSTER, J. D. & DICKINSON, A. G., 1988. The unusual properties of CH1641, a sheep-passaged isolate of scrapie. Veterinary Record, 123, 5-8.
- FOSTER, J. D., GOLDMANN, W. & HUNTER, N., 2013. Evidence in sheep for pre-natal transmission of scrapie to lambs from infected mothers. PLoS One, 8, e79433.
- GARZA, M. C., FERNÁNDEZ-BORGES, N., BOLEA, R., BADIOLA, J. J., CASTILLA, J. & MONLEÓN, E., 2011. Detection of PrPres in genetically susceptible fetuses from sheep with natural scrapie. PLoS One, 6, e27525.
- GAVIER-WIDÉN, D., STACK, M. J., BARON, T., BALACHANDRAN, A. & SIMMONS, M., 2005. Diagnosis of transmissible spongiform encephalopathies in animals: a review. Journal of Veterinary Diagnostic Investigation, 17, 509-527.
- GEORGSSON, G., ADOLFSDOTTIR, J. A., PALSDOTTIR, A., JORUNDSSON, E., SIGURDARSON, S. & THORGEIRSDOTTIR, S., 2008. High incidence of subclinical infection of lymphoid tissues in scrapie-affected sheep flocks. Archives of Virology, 153, 637-644.
- GEORGSSON, G., SIGURDARSON, S. & BROWN, P., 2006. Infectious agent of sheep scrapie may persist in the environment for at least 16 years. Journal of General Virology, 87, 3737-3740.
- GOLDMANN, W., HUNTER, N., FOSTER, J. D., SALBAUM, J. M., BEYREUTHER, K. & HOPE, J., 1990. Two alleles of a neural protein gene linked to scrapie in sheep. Proceedings of the National Academy of Sciences of the United States of America, 87.
- GOLDMANN, W., HUNTER, N., SMITH, G., FOSTER, J. & HOPE, J., 1994. PrP genotype and agent effects in scrapie: change in allelic interaction with different isolates of agent in sheep, a natural host of scrapie. Journal of General Virology, 75, 989-995.
- GOLDMANN, W., MARIER, E., STEWART, P., KONOLD, T., STREET, S., LANGEVELD, J., WINDL, O. & ORTIZ-PELAEZ, A., 2016. Prion protein genotype survey confirms low frequency of scrapie-resistant K222 allele in British goat herds. Veterinary Record, 178, 168.
- GOLDMANN, W., RYAN, K., STEWART, P., PARNHAM, D., XICOHTENCATL, R., FERNANDEZ, N., SAUNDERS, G., WINDL, O., GONZALEZ, L., BOSSERS, A. & FOSTER, J., 2011. Caprine prion gene polymorphisms are associated with decreased incidence of classical scrapie in goat herds in the United Kingdom. Veterinary Research, 42, 110.
- GONZÁLEZ, L., DAGLEISH, M. P., BELLWORTHY, S. J., SISÓ, S., STACK, M. J., CHAPLIN, M. J., DAVIS, L. A., HAWKINS, S. A., HUGHES, J. & JEFFREY, M., 2006. Postmortem diagnosis of preclinical and clinical scrapie in sheep by the detection of disease-associated PrP in their rectal mucosa. Veterinary Record, 158, 325-331.
- GONZÁLEZ, L., DAGLEISH, M. P., MARTIN, S., DEXTER, G., STEELE, P., FINLAYSON, J. & JEFFREY, M., 2008. Diagnosis of preclinical scrapie in live sheep by the immunohistochemical examination of rectal biopsies. Veterinary Record, 162, 397-403.
- GONZÁLEZ, L., HORTON, R., RAMSAY, D., TOOMIK, R., LEATHERS, V., TONELLI, Q., DAGLEISH, M. P., JEFFREY, M. & TERRY, L., 2008. Adaptation and evaluation of a rapid test for the diagnosis of sheep scrapie in samples of rectal mucosa. Journal of Veterinary Diagnostic Investigation, 20, 203-208.
- GONZÁLEZ, L., MARTIN, S., BEGARA-MCGORUM, I., HUNTER, N., HOUSTON, F., SIMMONS, M. & JEFFREY, M., 2002. Effects of agent strain and host genotype on PrP accumulation in the brain of sheep naturally and experimentally affected with scrapie. Journal of Comparative Pathology, 126, 17-29.
- GONZÁLEZ, L., MARTIN, S., HAWKINS, S. A., GOLDMANN, W., JEFFREY, M. & SISÓ, S., 2010. Pathogenesis of natural goat scrapie: modulation by host PRNP genotype and effect of co-existent conditions. Veterinary Research, 41, 48.
- GONZÁLEZ, L., MARTIN, S., SISÓ, S., KONOLD, T., ORTIZ-PELÁEZ, A., PHELAN, L., GOLDMANN, W., STEWART, P., SAUNDERS, G., WINDL, O., JEFFREY, M., HAWKINS, S. A. C., DAWSON, M. & HOPE, J., 2009. High prevalence of scrapie in a dairy goat herd: tissue distribution of disease-associated PrP and effect of PRNP genotype and age. Veterinary Research, 40, 65.
- GONZÁLEZ, L., PITARCH, J. L., MARTIN, S., THURSTON, L., SIMMONS, H., ACÍN, C. & JEFFREY, M., 2014. Influence of polymorphisms in the prion protein gene on the pathogenesis and neuropathological phenotype of sheep scrapie after oral infection. Journal of Comparative Pathology, 150, 57-70.
- GONZÁLEZ, L., SISÓ, S., MONLEÓN, E., CASALONE, C., VAN KEULEN, L. J. M., BALKEMA-BUSCHMANN, A., ORTIZ-PELÁEZ, A., IULINI, B., LANGEVELD, J. P. M., HOFFMANN, C., BADIOLA, J. J., JEFFREY, M. & ACÍN, C., 2010. Variability in disease phenotypes within a single PRNP genotype suggests the existence of multiple natural sheep scrapie strains within Europe. Journal of General Virology, 91, 2630-2641.
- GORDON, W. S., 1946. Advances in veterinary research: louping ill, tick-borne fever and scrapie. Veterinary Record, 58, 516-520.
- GORDON, W. S., BROWNLEE, A. & WILSON, D. R., 1939. Studies in louping-ill, tick-borne fever and scrapie. Journal, 362–363.
- GOUGH, K. C. & MADDISON, B. C., 2010. Prion transmission: Prion excretion and occurrence in the environment. Prion, 4, 275-282.
- GREENLEE, J. J., KUNKLE, R. A., SMITH, J. D. & GREENLEE, M. H. W., 2016. Scrapie in swine: a diagnostic challenge. Food Safety, 4, 110-114.
- GROSCHUP, M. H. & BUSCHMANN, A., 2008. Rodent models for prion diseases. Veterinary Research, 39, 32.
- GROSCHUP, M. H., LACROUX, C., BUSCHMANN, A., LUHKEN, G., MATHEY, J., EIDEN, M., LUGAN, S., HOFFMANN, C., ESPINOSA, J. C., BARON, T., TORRES, J. M., ERHARDT, G. & ANDREOLETTI, O., 2007. Classic scrapie in sheep with the ARR/ARR prion genotype in Germany and France. Emerging Infectious Diseases, 13, 1201-1207.
- HADLOW, W. J., 1959. Scrapie and Kuru. The Lancet, 274, 289-290.
- HADLOW, W. J., 1961. The pathology of experimental scrapie in the dairy goat. Research in Veterinary Science, 2, 289-314.
- HADLOW, W. J., KENNEDY, R. C. & RACE, R. E., 1982. Natural infection of Suffolk sheep with scrapie virus. Journal of Infectious Diseases, 146, 657-664.
- HADLOW, W. J., KENNEDY, R. C., RACE, R. E. & EKLUND, C. M., 1980. Virologic and neurohistologic findings in dairy goats affected with natural scrapie. Veterinary Pathology, 17, 187-199.
- HADLOW, W. J., RACE, R. E., KENNEDY, R. C. & EKLUND, C. M., 1979. Natural infection of the sheep with scrapie virus. Slow transmissible diseases of the nervous system. Vol. 2. S. B. PRUSINER and W. J. HADLOW. New York, Academic Press. 2, 3-12.
- HAWKINS, S. A., SIMMONS, H. A., GOUGH, K. C. & MADDISON, B. C., 2015. Persistence of ovine scrapie infectivity in a farm environment following cleaning and decontamination. Veterinary Record, 176, 99.
- HEALY, A. M., HANLON, A. J., WEAVERS, E., COLLINS, J. D. & DOHERTY, M. L., 2002. A behavioural study of scrapie-affected sheep. Applied Animal Behaviour Science, 79, 89-102.
- HEALY, A. M., HANNON, D., MORGAN, K. L., WEAVERS, E., COLLINS, J. D. & DOHERTY, M. L., 2004. A paired case-control study of risk factors for scrapie in Irish sheep flocks. Preventive Veterinary Medicine, 64, 73-83.
- HEALY, A. M., WEAVERS, E., MCELROY, M., GOMEZ-PARADA, M., COLLINS, J. D., O'DOHERTY, E., SWEENEY, T. & DOHERTY, M. L., 2003. The clinical neurology of scrapie in Irish sheep. Journal of Veterinary Internal Medicine, 17, 908-916.
- HIEPE, T., BERGMANN, V. & JUNGMANN, R., 1973. Beitrag zur klinischen und pathomorphologischen Diagnostik der Traberkrankheit des Schafes - Vorläufige Mitteilung [Clinical and pathomorphological diagnosis of scrapie - preliminary communication]. Monatshefte für Veterinärmedizin, 28, 905-907.
- HOINVILLE, L. J., TONGUE, S. C. & WILESMITH, J. W., 2010. Evidence for maternal transmission of scrapie in naturally affected flocks. Preventive Veterinary Medicine, 93, 121-128.
- HOPE, J., WOOD, S. C., BIRKETT, C. R., CHONG, A., BRUCE, M. E., CAIRNS, D., GOLDMANN, W., HUNTER, N. & BOSTOCK, C. J., 1999. Molecular analysis of ovine prion protein identifies similarities between BSE and an experimental isolate of natural scrapie, CH1641. Journal of General Virology, 80, 1-4.
- HOPP, P., OMER, M. K. & HEIER, B. T., 2006. A case-control study of scrapie Nor98 in Norwegian sheep flocks. Journal of General Virology, 87, 3729-3736.
- HORTELLS, P., MONZON, M., MONLEON, E., ACIN, C., VARGAS, A., BOLEA, R., LUJAN, L. & BADIOLA, J. J., 2006. Pathological findings in retina and visual pathways associated to natural scrapie in sheep. Brain Research, 1108, 188-194.
- HOUSTON, F., MCCUTCHEON, S., GOLDMANN, W., CHONG, A., FOSTER, J., SISÓ, S., GONZÁLEZ, L., JEFFREY, M. & HUNTER, N., 2008. Prion diseases are efficiently transmitted by blood transfusion in sheep. Blood, 112, 4739-4745.
- HUANG, H., SOUTYRINE, A., RENDULICH, J., O'ROURKE, K. & BALACHANDRAN, A., 2011. Investigation of the effects of experimental autolysis on the detection of abnormal prion protein in lymphoid and central nervous system tissues from elk and sheep using the Western blotting method. Canadian Journal of Veterinary Research, 75, 69-72.
- HUNTER, N., FOSTER, J. D., DICKINSON, A. G. & HOPE, J., 1989. Linkage of the gene for the scrapie-associated fibril protein (PrP) to the Sip gene in Cheviot sheep. Veterinary Record, 124, 364-6.
- JACOBS, J. G., SAUER, M., VAN KEULEN, L. J. M., TANG, Y., BOSSERS, A. & LANGEVELD, J. P. M., 2011. Differentiation of ruminant transmissible spongiform encephalopathy isolate types, including bovine spongiform encephalopathy and CH1641 scrapie. Journal of General Virology, 92, 222-232.
- JEFFREY, M., GONZÁLEZ, L., CHONG, A., FOSTER, J., GOLDMANN, W., HUNTER, N. & MARTIN, S., 2006. Ovine infection with the agents of scrapie (CH1641 isolate) and bovine spongiform encephalopathy: immunochemical similarities can be resolved by immunohistochemistry. Journal of Comparative Pathology, 134, 17-29.
- JEFFREY, M., MARTIN, S., GONZÁLEZ, L., FOSTER, J., LANGEVELD, J. P., VAN ZIJDERVELD, F. G., GRASSI, J. & HUNTER, N., 2006. Immunohistochemical features of PrP(d) accumulation in natural and experimental goat transmissible spongiform encephalopathies. Journal of Comparative Pathology, 134, 171-181.
- JEFFREY, M., MARTIN, S., THOMSON, J. R., DINGWALL, W. S., BEGARA-MCGORUM, I. & GONZÁLEZ, L., 2001. Onset and distribution of tissue PrP accumulation in scrapie-affected suffolk sheep as demonstrated by sequential necropsies and tonsillar biopsies. Journal of Comparative Pathology, 125, 48-57.
- JEFFREY, M., SCHOLES, S. F., MARTIN, S., MCGOVERN, G., SISO, S. & GONZALEZ, L., 2012. Increased immunohistochemical labelling for prion protein occurs in diverse neurological disorders of sheep: relevance for normal cellular PrP function. Journal of Comparative Pathology, 147, 46-54.
- JOUBERT, L., LAPRAS, M., GASTELLU, J., PRAVE, M. & LAURENT, D., 1972. Un foyer de tremblante du mouton en Provence. Bulletin de la Société des Sciences Vétérinaires et de Médecine Comparée (Lyon), 74, 165-184.
- KANATA, E., HUMPHREYS-PANAGIOTIDIS, C., GIADINIS, N. D., PAPAIOANNOU, N., ARSENAKIS, M. & SKLAVIADIS, T., 2014. Perspectives of a scrapie resistance breeding scheme targeting Q211, S146 and K222 caprine PRNP alleles in Greek goats. Veterinary Research, 45, 43.
- KANG, H.-E., MO, Y., RAHIM, R. A., LEE, H.-M. & RYOU, C., 2017. Prion diagnosis: application of real-time quaking-induced conversion. BioMed Research International, 2017:5413936.
- KIMBERLIN, R. H., WALKER, C. A., MILLSON, G. C., TAYLOR, D. M., ROBERTSON, P. A., TOMLINSON, A. H. & DICKINSON, A. G., 1983. Disinfection studies with two strains of mouse-passaged scrapie agent. Journal of the Neurological Sciences, 59, 355-369.
- KITTELBERGER, R., CHAPLIN, M. J., SIMMONS, M. M., RAMIREZ-VILLAESCUSA, A., MCINTYRE, L., MACDIARMID, S. C., HANNAH, M. J., JENNER, J., BUENO, R., BAYLISS, D., BLACK, H., PIGOTT, C. J. & O'KEEFE, J. S., 2010. Atypical scrapie/Nor98 in a sheep from New Zealand. Journal of Veterinary Diagnostic Investigation, 22, 863-875.
- KITTELBERGER, R., MCINTYRE, L., WATTS, J., MACDIARMID, S., HANNAH, M. J., JENNER, J., BUENO, R., SWAINSBURY, R., LANGEVELD, J. P., VAN KEULEN, L. J., VAN ZIJDERVELD, F. G., WEMHEUER, W. M., RICHT, J. A., SORENSEN, S. J., PIGOTT, C. J. & O'KEEFE, J. S., 2014. Evaluation of two commercial, rapid, ELISA kits testing for scrapie in retro-pharyngeal lymph nodes in sheep. New Zealand Veterinary Journal, 62, 343-50.
- KONOLD, T., BONE, G., ORTIZ-PELAEZ, A., TORTOSA, R., CLIFFORD, D., DEXTER, G., SIMMONS, M. M., SPIROPOULOS, J. & BERTHELIN-BAKER, C. F., 2009. Associations of clinical signs and prion protein genotypes in British sheep with scrapie. Deutsche Tierärztliche Wochenschrift, 116, 380-388.
- KONOLD, T., BONE, G., SIMMONS, M. M., DEXTER, G., MOORE, S. J. & PETTITT, R. G., 2007. Scrapie in goats. Veterinary Record, 161, 395-396.
- KONOLD, T., BONE, G., VIDAL-DIEZ, A., TORTOSA, R., DAVIS, A., DEXTER, G., HILL, P., JEFFREY, M., SIMMONS, M. M., CHAPLIN, M. J., BELLWORTHY, S. J. & BERTHELIN-BAKER, C., 2008. Pruritus is a common feature in sheep infected with the BSE agent. BMC Veterinary Research, 4, 16.
- KONOLD, T., BONE, G. E., PHELAN, L. J., SIMMONS, M. M., GONZÁLEZ, L., SISÓ, S., GOLDMANN, W., CAWTHRAW, S. & HAWKINS, S. A. C., 2010. Monitoring of clinical signs in goats with transmissible spongiform encephalopathies. BMC Veterinary Research, 6, 13.
- KONOLD, T., DAVIS, A., BONE, G., BRACEGIRDLE, J., EVERITT, S., CHAPLIN, M., SAUNDERS, G. C., CAWTHRAW, S. & SIMMONS, M. M., 2007. Clinical findings in two cases of atypical scrapie in sheep: a case report. BMC Veterinary Research, 3, 2.
- KONOLD, T., HAWKINS, S. A. C., THURSTON, L. C., MADDISON, B. C., GOUGH, K. C., DUARTE, A. & SIMMONS, H. A., 2015. Objects in contact with classical scrapie sheep act as a reservoir for scrapie transmission. Frontiers in Veterinary Science, 2.
- KONOLD, T., MOORE, S. J., BELLWORTHY, S. J., TERRY, L. A., THORNE, L., RAMSAY, A., SALGUERO, F. J., SIMMONS, M. M. & SIMMONS, H. A., 2013. Evidence of effective scrapie transmission via colostrum and milk in sheep. BMC Veterinary Research, 9, 99.
- KONOLD, T., NONNO, R., SPIROPOULOS, J., CHAPLIN, M. J., STACK, M. J., HAWKINS, S. A., CAWTHRAW, S., WILESMITH, J. W., WELLS, G. A., AGRIMI, U., DI BARI, M. A., ANDREOLETTI, O., ESPINOSA, J. C., AGUILAR-CALVO, P. & TORRES, J. M., 2015. Further characterisation of transmissible spongiform encephalopathy phenotypes after inoculation of cattle with two temporally separated sources of sheep scrapie from Great Britain. BMC Research Notes, 8, 312.
- KONOLD, T. & PHELAN, L., 2014. Clinical examination protocol to detect atypical and classical scrapie in sheep. Journal of Visualized Experiments, e51101.
- KONOLD, T., PHELAN, L. J., CAWTHRAW, S., SIMMONS, M. M., CHAPLIN, M. J. & GONZALEZ, L., 2016. Abnormalities in brainstem auditory evoked potentials in sheep with transmissible spongiform encephalopathies and lack of a clear pathological relationship. Frontiers in Veterinary Science, 3, 60.
- KONOLD, T., PHELAN, L. J., DONNACHIE, B. R., CHAPLIN, M. J., CAWTHRAW, S. & GONZALEZ, L., 2017. Codon 141 polymorphisms of the ovine prion protein gene affect the phenotype of classical scrapie transmitted from goats to sheep. BMC Veterinary Research, 13:, 122.
- KONOLD, T., SPIROPOULOS, J., CHAPLIN, M. J., STACK, M. J., HAWKINS, S. A., WILESMITH, J. W. & WELLS, G. A., 2013. Unsuccessful oral transmission of scrapie from British sheep to cattle. Veterinary Record, 173, 118.
- KONOLD, T., THORNE, L., SIMMONS, H. A., HAWKINS, S. A. C., SIMMONS, M. M. & GONZÁLEZ, L., 2016. Evidence of scrapie transmission to sheep via goat milk. BMC Veterinary Research, 12, 1-10.
- KÜMPER, H., 1991. Scrapie - Ein Fallbericht. Praktische Tierarzt, Collegium Veterinarium, 73, 32-34.
- LACROUX, C., SIMON, S., BENESTAD, S. L., MAILLET, S., MATHEY, J., LUGAN, S., CORBIERE, F., CASSARD, H., COSTES, P., BERGONIER, D., WEISBECKER, J. L., MOLDAL, T., SIMMONS, H., LANTIER, F., FERAUDET-TARISSE, C., MOREL, N., SCHELCHER, F., GRASSI, J. & ANDREOLETTI, O., 2008. Prions in milk from ewes incubating natural scrapie. PLoS Pathogens, 4, e1000238.
- LACROUX, C., VILETTE, D., FERNÁNDEZ-BORGES, N., LITAISE, C., LUGAN, S., MOREL, N., CORBIÈRE, F., SIMON, S., SIMMONS, H., COSTES, P., WEISBECKER, J. L., LANTIER, I., LANTIER, F., SCHELCHER, F., GRASSI, J., CASTILLA, J. & ANDRÉOLETTI, O., 2012. Prionemia and leukocyte-platelet-associated infectivity in sheep transmissible spongiform encephalopathy models. Journal of Virology, 86, 2056-66.
- LANGEVELD, J. P., JACOBS, J. G., ERKENS, J. H., BOSSERS, A., VAN ZIJDERVELD, F. G. & VAN KEULEN, L. J., 2006. Rapid and discriminatory diagnosis of scrapie and BSE in retro-pharyngeal lymph nodes of sheep. BMC Veterinary Research, 2, 19.
- LAVEN, R., 1990. Scrapie in a four year old goat. Goat Veterinary Society Journal, 11, 77-79.
- LEZMI, S., MARTIN, S., SIMON, S., COMOY, E., BENCSIK, A., DESLYS, J. P., GRASSI, J., JEFFREY, M. & BARON, T., 2004. Comparative molecular analysis of the abnormal prion protein in field scrapie cases and experimental bovine spongiform encephalopathy in sheep by use of western blotting and immunohistochemical methods. Journal of Virology, 78, 3654-3662.
- LIGIOS, C., BANDINO, E., DI GUARDO, G., FERRARI, G. C., PERFETTI, M. G., ELENI, C., RU, G., MUCELI, G., SPANU, G. P., AGRIMI, U. & POCCHIARI, M., 1999. Scrapie della pecora: osservazioni cliniche ed epidemiologiche in pecore di razza sarda [Clinical and epidemiological features in outbreaks of scrapie in Sarda sheep]. Obiettivi e Documenti Veterinari, 20, 65-70.
- LIGIOS, C., CANCEDDA, M. G., CARTA, A., SANTUCCIU, C., MAESTRALE, C., DEMONTIS, F., SABA, M., PATTA, C., DEMARTINI, J. C., AGUZZI, A. & SIGURDSON, C. J., 2011. Sheep with scrapie and mastitis transmit infectious prions through the milk. Journal of Virology, 85, 1136-1139.
- LIGIOS, C., CANCEDDA, M. G., MADAU, L., SANTUCCIU, C., MAESTRALE, C., AGRIMI, U., RU, G. & DI GUARDO, G., 2006. PrP(Sc) deposition in nervous tissues without lymphoid tissue involvement is frequently found in ARQ/ARQ Sarda breed sheep preclinically affected with natural scrapie. Archives of Virology, 151, 2007-2020.
- LIGIOS, C., DEXTER, G., SPIROPOULOS, J., MAESTRALE, C., CARTA, A. & SIMMONS, M. M., 2004. Distribution of vascular amyloid in scrapie-affected sheep with different genotypes. Journal of Comparative Pathology, 131, 271-276.
- LOW, J. C., CHAMBERS, J., MCKELVEY, W. A., MCKENDRICK, I. J. & JEFFREY, M., 2009. Failure to transmit scrapie infection by transferring preimplantation embryos from naturally infected donor sheep. Theriogenology, 72, 809-16.
- MACDIARMID, S. C., 1989. Aspects of scrapie and bovine spongiform encephalopathy. Surveillance, 16, 11-13.
- MADDISON, B. C., BAKER, C. A., TERRY, L. A., BELLWORTHY, S. J., THORNE, L., REES, H. C. & GOUGH, K. C., 2010. Environmental sources of scrapie prions. Journal of Virology, 84, 11560-11562.
- MARIER, E., DAWSON, M., SIMMONS, M., HOPE, J. & ORTIZ-PELAEZ, A., 2017. Case-control study on the use of pituitary-derived hormones from sheep as a potential risk factor for the occurrence of atypical scrapie in Great Britain. The Veterinary record, 180, 403.
- MATTHEWS, D. & COOKE, B. C., 2003. The potential for transmissible spongiform encephalopathies in non-ruminant livestock and fish. Revue Scientifique et Technique - Office International des Epizooties, 22, 283-296.
- MAURER, E., BOTTERON, C., EHRENSPERGER, F., FATZER, R., JAGGY, A., KOLLY, C., MEYLAN, M., ZURBRIGGEN, A. & DOHERR, M. G., 2005. Scrapie-Überwachung in der Schweiz. I. Klinische Aspekte von neurologischen Erkrankungen bei Schaf und Ziege [Swiss scrapie surveillance. I. Clinical aspects of neurological diseases in sheep and goats]. Schweizer Archiv für Tierheilkunde, 147, 425-33.
- MCINTYRE, K. M., DEL RIO VILAS, V. J. & GUBBINS, S., 2011. Demographic characteristics of scrapie-affected holdings identified by active and passive surveillance schemes in Great Britain: 2002-2005. Veterinary Journal, 187, 207-11.
- MELCHIOR, M. B., WINDIG, J. J., HAGENAARS, T. J., BOSSERS, A., DAVIDSE, A. & VAN ZIJDERVELD, F. G., 2010. Eradication of scrapie with selective breeding: are we nearly there? BMC Veterinary Research, 6, 24-24.
- MELONI, D., BOZZETTA, E., LANGEVELD, J. P. M., GROSCHUP, M. H., GOLDMANN, W., ANDREOLETTI, O., LANTIER, I., VAN KEULEN, L., BOSSERS, A., PITARDI, D., NONNO, R., SKLAVIADIS, T., INGRAVALLE, F., PELETTO, S., COLUSSI, S. & ACUTIS, P. L., 2017. EU-approved rapid tests might underestimate bovine spongiform encephalopathy infection in goats. Journal of Veterinary Diagnostic Investigation, 29, 232-236.
- MOORE, S. J., SIMMONS, M., CHAPLIN, M. & SPIROPOULOS, J., 2008. Neuroanatomical distribution of abnormal prion protein in naturally occurring atypical scrapie cases in Great Britain. Acta Neuropathologica, 116, 547-559.
- NENTWIG, A., OEVERMANN, A., HEIM, D., BOTTERON, C., ZELLWEGER, K., DRÖGEMÜLLER, C., ZURBRIGGEN, A. & SEUBERLICH, T., 2007. Diversity in neuroanatomical distribution of abnormal prion protein in atypical scrapie. PLoS Pathogens, 3, e82.
- NICHOLSON, S. S., 1989. Tremorgenic syndromes in livestock. Veterinary Clinics of North America. Food Animal Practice, 5, 291-300.
- NIEDERMEYER, S., EIDEN, M., TOUMAZOS, P., PAPASAVVA-STYLIANOU, P., IOANNOU, I., SKLAVIADIS, T., PANAGIOTIDIS, C., LANGEVELD, J., BOSSERS, A., KUCZIUS, T., KAATZ, M., GROSCHUP, M. H. & FAST, C., 2016. Genetic, histochemical and biochemical studies on goat TSE cases from Cyprus. Veterinary Research, 47, 99.
- O'ROURKE, K. I., BASZLER, T. V., BESSER, T. E., MILLER, J. M., CUTLIP, R. C., WELLS, G. A. H., RYDER, S. J., PARISH, S. M., HAMIR, A. N., COCKETT, N. E., JENNY, A. & KNOWLES, D. P., 2000. Preclinical diagnosis of scrapie by immunohistochemistry of third eyelid lymphoid tissue. Journal of Clinical Microbiology, 38, 3254-3259.
- OIE, 2016. Chapter 2.7.12. Scrapie. Manual of diagnostic tests and vaccines for terrestrial animals. Paris, World Organisation for Animal Health. 1-11.
- OIE, 2018. Chapter 14.8. Scrapie. Terrestrial Animal Health Code. Paris, World Organisation for Animal Health. 1-6.
- ONNASCH, H., GUNN, H. M., BRADSHAW, B. J., BENESTAD, S. L. & BASSETT, H. F., 2004. Two Irish cases of scrapie resembling Nor98. Veterinary Record, 155, 636-637.
- ONODERA, T. & SAEKI, K., 2000. Japanese scrapie cases. Japanese Journal of Infectious Diseases, 53, 56-61.
- ORTIZ-PELAEZ, A., ARNOLD, M. E. & VIDAL-DIEZ, A., 2016. Epidemiological investigations on the potential transmissibility of a rare disease: the case of atypical scrapie in Great Britain. Epidemiology and Infection, 144, 2107-16.
- ORTIZ-PELAEZ, A. & BIANCHINI, J., 2011. The impact of the genotype on the prevalence of classical scrapie at population level. Veterinary Research, 42, 31.
- ORTIZ-PELAEZ, A., GEORGIADOU, S., SIMMONS, M. M., WINDL, O., DAWSON, M., ARNOLD, M. E., NEOCLEOUS, P. & PAPASAVVA-STYLIANOU, P., 2015. Allelic variants at codon 146 in the PRNP gene show significant differences in the risk for natural scrapie in Cypriot goats. Epidemiology and Infection, 143, 1304-10.
- ORTIZ-PELAEZ, A., KELLY, L. & ADKIN, A., 2012. The risk of introducing scrapie from restocking goats in Great Britain. Preventive Veterinary Medicine, 107, 222-30.
- PAPASAVVA-STYLIANOU, P., WINDL, O., SAUNDERS, G., MAVRIKIOU, P., TOUMAZOS, P. & KAKOYIANNIS, C., 2011. PrP gene polymorphisms in Cyprus goats and their association with resistance or susceptibility to natural scrapie. Veterinary Journal, 187, 245-50.
- PARRY, H. B., 1983. Clinical features of natural scrapie. Scrapie disease in sheep. D. R. OPPENHEIMER. London, Academic Press. 60-72.
- PARRY, H. P., 1957. Scrapie and related myopathies in sheep. Veterinary Record, 69, 43-55.
- PATTISON, I. H. & MILLSON, G. C., 1961. Scrapie produced experimentally in goats with special reference to the clinical syndrome. Journal of Comparative Pathology, 71, 101-109.
- PRUSINER, S. B., 1982. Novel proteinaceous infectious particles cause scrapie. Science, 216, 136-144.
- PRUSINER, S. B., 1997. Prion diseases and the BSE crisis. Science, 278, 245-251.
- REGNIER, A., ANDREOLETTI, O., ALBARIC, O., GRUSON, D. C., SCHELCHER, F. & TOUTAIN, P. L., 2011. Clinical, electroretinographic and histomorphometric evaluation of the retina in sheep with natural scrapie. BMC Veterinary Research, 7, 25.
- ROBINSON, M. M., HADLOW, W. J., KNOWLES, D. P., HUFF, T. P., LACY, P. A., MARSH, R. F. & GORHAM, J. R., 1995. Experimental infection of cattle with the agents of transmissible mink encephalopathy and scrapie. Journal of Comparative Pathology, 113, 241-251.
- RUBENSTEIN, R., BULGIN, M. S., CHANG, B., SORENSEN-MELSON, S., PETERSEN, R. B. & LAFAUCI, G., 2012. PrPSc detection and infectivity in semen from scrapie-infected sheep. Journal of General Virology, 93, 1375-1383.
- RUBENSTEIN, R., CHANG, B., GRAY, P., PILTCH, M., BULGIN, M. S., SORENSEN-MELSON, S. & MILLER, M. W., 2011. Prion disease detection, PMCA kinetics, and IgG in urine from sheep naturally/experimentally infected with scrapie and deer with preclinical/clinical chronic wasting disease. Journal of Virology, 85, 9031-9038.
- RYDER, S., DEXTER, G., BELLWORTHY, S. & TONGUE, S., 2004. Demonstration of lateral transmission of scrapie between sheep kept under natural conditions using lymphoid tissue biopsy. Research in Veterinary Science, 76, 211-217.
- RYDER, S. J., SPENCER, Y. I., BELLERBY, P. J. & MARCH, S. A., 2001. Immunohistochemical detection of PrP in the medulla oblongata of sheep: the spectrum of staining in normal and scrapie-affected sheep. Veterinary Record, 148, 7-13.
- SALVESEN, Ø., REITEN, M. R., ESPENES, A., BAKKEBO, M. K., TRANULIS, M. A. & ERSDAL, C., 2017. LPS-induced systemic inflammation reveals an immunomodulatory role for the prion protein at the blood-brain interface. Journal of Neuroinflammation, 14, 106.
- SALVESEN, Ø., REITEN, M. R., KAMSTRA, J. H., BAKKEBØ, M. K., ESPENES, A., TRANULIS, M. A. & ERSDAL, C., 2017. Goats without prion protein display enhanced proinflammatory pulmonary signaling and extracellular matrix remodeling upon systemic lipopolysaccharide challenge. Frontiers in Immunology, 8, 1722.
- SARRADIN, P., MELO, S., BARC, C., LECOMTE, C., ANDREOLETTI, O., LANTIER, F., DACHEUX, J. L. & GATTI, J. L., 2008. Semen from scrapie-infected rams does not transmit prion infection to transgenic mice. Reproduction, 135, 415-8.
- SCHNEIDER, D. A., MADSEN-BOUTERSE, S. A., ZHUANG, D., TRUSCOTT, T. C., DASSANAYAKE, R. P. & O'ROURKE, K. I., 2015. The placenta shed from goats with classical scrapie is infectious to goat kids and lambs. Journal of General Virology, 96, 2464-2469.
- SCHNEIDER, K., FANGERAU, H., MICHAELSEN, B. & RAAB, W. H. M., 2008. The early history of the transmissible spongiform encephalopathies exemplified by scrapie. Brain Research Bulletin, 77, 343-355.
- SCOTT, P. R. & HENSHAW, C. J., 1995. Increasing the accuracy of the provisional antemortem diagnosis of scrapie. Agri-Practice, 16, 21-25.
- SEUBERLICH, T., BOTTERON, C., BENESTAD, S. L., BRUNISHOLZ, H., WYSS, R., KIHM, U., SCHWERMER, H., FRIESS, M., NICOLIER, A., HEIM, D. & ZURBRIGGEN, A., 2007. Atypical scrapie in a swiss goat and implications for transmissible spongiform encephalopathy surveillance. Journal of Veterinary Diagnostic Investigation, 19, 2-8.
- SIGURDARSON, S. & JARVEIKIVARNIR, S., 1991. Epidemiology of scrapie in Iceland and experience with control measures. Sub-acute spongiform encephalopathies. Proceedings of a seminar in the CEC agricultural resaerch programme, held in Brussels, 12-14 November 1990. R. BRADLEY, M. SAVEY, and B. MARCHANT. Dordrecht, Kluwer Academic Publishers. 233-242.
- SIMMONS, H. A., SIMMONS, M. M., SPENCER, Y. I., CHAPLIN, M. J., POVEY, G., DAVIS, A., ORTIZ-PELAEZ, A., HUNTER, N., MATTHEWS, D. & WRATHALL, A. E., 2009. Atypical scrapie in sheep from a UK research flock which is free from classical scrapie. BMC Veterinary Research, 5, 8.
- SIMMONS, M. M., CHAPLIN, M. J., VICKERY, C. M., SIMON, S., DAVIS, L., DENYER, M., LOCKEY, R., STACK, M. J., O'CONNOR, M. J., BISHOP, K., GOUGH, K. C., MADDISON, B. C., THORNE, L. & SPIROPOULOS, J., 2015. Does the presence of scrapie affect the ability of current statutory discriminatory tests To detect the presence of bovine spongiform encephalopathy? Journal of Clinical Microbiology, 53, 2593-2604.
- SIMMONS, M. M., KONOLD, T., SIMMONS, H. A., SPENCER, Y. I., LOCKEY, R., SPIROPOULOS, J., EVERITT, S. & CLIFFORD, D., 2007. Experimental transmission of atypical scrapie to sheep. BMC Veterinary Research, 3, 20.
- SIMMONS, M. M., KONOLD, T., THURSTON, L., BELLWORTHY, S. J., CHAPLIN, M. J. & MOORE, S. J., 2010. The natural atypical scrapie phenotype is preserved on experimental transmission and sub-passage in PRNP homologous sheep. BMC Veterinary Research, 6, 14.
- SIMMONS, M. M., MOORE, S. J., KONOLD, T., THURSTON, L., TERRY, L. A., THORNE, L., LOCKEY, R., VICKERY, C., HAWKINS, S. A. C., CHAPLIN, M. J. & SPIROPOULOS, J., 2011. Experimental oral transmission of atypical scrapie to sheep. Emerging Infectious Diseases, 17, 848-854.
- SIMMONS, M. M., MOORE, S. J., LOCKEY, R., CHAPLIN, M. J., KONOLD, T., VICKERY, C. & SPIROPOULOS, J., 2015. Phenotype shift from atypical scrapie to CH1641 following experimental transmission in sheep. PLoS One, 10, e0117063.
- SISÓ, S., JEFFREY, M. & GONZÁLEZ, L., 2009. Neuroinvasion in sheep transmissible spongiform encephalopathies: the role of the haematogenous route. Neuropathology and Applied Neurobiology, 35, 232-246.
- SMITH, J. D., GREENLEE, J. J., HAMIR, A. N. & GREENLEE, M. H. W., 2009. Altered electroretinogram b-wave in a Suffolk sheep experimentally infected with scrapie. Veterinary Record, 165, 179-181.
- SOFIANIDIS, G., PSYCHAS, V., BILLINIS, C., SPYROU, V., ARGYROUDIS, S., PAPAIOANNOU, N. & VLEMMAS, I., 2006. Histopathological and immunohistochemical features of natural goat scrapie. Journal of Comparative Pathology, 135, 116-129.
- SPIROPOULOS, J., CASALONE, C., CARAMELLI, M. & SIMMONS, M. M., 2007. Immunohistochemistry for PrPSc in natural scrapie reveals patterns which are associated with the PrP genotype. Neuropathology and Applied Neurobiology, 33, 398-409.
- SPIROPOULOS, J., HAWKINS, S. A., SIMMONS, M. M. & BELLWORTHY, S. J., 2014. Evidence of in utero transmission of classical scrapie in sheep. Journal of Virology, 88, 4591-4.
- SPIROPOULOS, J., LOCKEY, R., SALLIS, R. E., TERRY, L. A., THORNE, L., HOLDER, T. M., BECK, K. E. & SIMMONS, M. M., 2011. Isolation of prion with BSE properties from farmed goat. Emerging Infectious Diseases, 17, 2253-2261.
- ST ROSE, S., HUNTER, N., MATTHEWS, L., FOSTER, J., CHASE-TOPPING, M., KRUUK, L., SHAW, D., RHIND, S., WILL, R. & WOOLHOUSE, M., 2006. Comparative evidence for a link between Peyer's patch development and susceptibility to transmissible spongiform encephalopathies. BMC Infectious Diseases, 6, 5.
- STACK, M., JEFFREY, M., GUBBINS, S., GRIMMER, S., GONZÁLEZ, L., MARTIN, S., CHAPLIN, M., WEBB, P., SIMMONS, M., SPENCER, Y., BELLERBY, P., HOPE, J., WILESMITH, J. & MATTHEWS, D., 2006. Monitoring for bovine spongiform encephalopathy in sheep in Great Britain, 1998-2004. Journal of General Virology, 87, 2099-2107.
- STACK, M. J., CHAPLIN, M. J. & CLARK, J., 2002. Differentiation of prion protein glycoforms from naturally occurring sheep scrapie, sheep-passaged scrapie strains (CH1641 and SSBP1), bovine spongiform encephalopathy (BSE) cases and Romney and Cheviot breed sheep experimentally inoculated with BSE using two monoclonal antibodies. Acta Neuropathologica, 104, 279-286.
- STEVENS, K. B., DEL RIO VILAS, V. J. & GUITIAN, J., 2009. Classical sheep scrapie in Great Britain: spatial analysis and identification of environmental and farm-related risk factors. BMC Veterinary Research, 5, 33.
- TAMGÜNEY, G., RICHT, J. A., HAMIR, A. N., GREENLEE, J. J., MILLER, M. W., WOLFE, L. L., SIROCHMAN, T. M., YOUNG, A. J., GLIDDEN, D. V., JOHNSON, N. L., GILES, K., DEARMOND, S. J. & PRUSINER, S. B., 2012. Salivary prions in sheep and deer. Prion, 6, 52-61.
- TAYLOR, D. M., 2000. Inactivation of transmissible degenerative encephalopathy agents: a review. Veterinary Journal, 159, 10-17.
- TAYLOR, D. M., FRASER, H., MCCONNELL, I., BROWN, D. A., BROWN, K. L., LAMZA, K. A. & SMITH, G. R., 1994. Decontamination studies with the agents of bovine spongiform encephalopathy and scrapie. Archives of Virology, 139, 313-26.
- TERRY, L. A., HOWELLS, L., BISHOP, K., BAKER, C. A., EVEREST, S., THORNE, L., MADDISON, B. C. & GOUGH, K. C., 2011. Detection of prions in the faeces of sheep naturally infected with classical scrapie. Veterinary Research, 42, 65.
- THACKRAY, A. M., HOPKINS, L., LOCKEY, R., SPIROPOULOS, J. & BUJDOSO, R., 2011. Emergence of multiple prion strains from single isolates of ovine scrapie. Journal of General Virology, 92, 1482-1491.
- THACKRAY, A. M., HOPKINS, L., LOCKEY, R., SPIROPOULOS, J. & BUJDOSO, R., 2012. Propagation of ovine prions from "poor" transmitter scrapie isolates in ovine PrP transgenic mice. Experimental and Molecular Pathology, 92, 167-174.
- THOMZIG, A., SCHULZ-SCHAEFFER, W., WREDE, A., WEMHEUER, W., BRENIG, B., KRATZEL, C., LEMMER, K. & BEEKES, M., 2007. Accumulation of pathological prion protein PrPSc in the skin of animals with experimental and natural scrapie. PLoS Pathogens, 3, e66.
- THORGEIRSDOTTIR, S., SIGURDARSON, S., THORISSON, H. M., GEORGSSON, G. & PALSDOTTIR, A., 1999. PrP gene polymorphism and natural scrapie in Icelandic sheep. Journal of General Virology, 80, 2527-2534.
- THORNE, L. & TERRY, L. A., 2008. In vitro amplification of PrPSc derived from the brain and blood of sheep infected with scrapie. Journal of General Virology, 89, 3177-3184.
- THURING, C. M. A., VAN KEULEN, L. J. M., LANGEVELD, J. P. M., VROMANS, M. E. W., VAN ZIJDERVELD, F. G. & SWEENEY, T., 2005. Immunohistochemical distinction between preclinical bovine spongiform encephalopathy and scrapie infection in sheep. Journal of Comparative Pathology, 132, 59.
- TOUMAZOS, P., 1991. Scrapie in Cyprus. British Veterinary Journal, 147, 147-154.
- TOUMAZOS, P. & ALLEY, M. R., 1989. Scrapie in goats in Cyprus. New Zealand Veterinary Journal, 37, 160-162.
- TRANULIS, M. A., BENESTAD, S. L., BARON, T. & KRETZSCHMAR, H., 2011. Atypical prion diseases in humans and animals. Topics in Current Chemistry, 305, 23-50.
- VACCARI, G., PANAGIOTIDIS, C. H., ACIN, C., PELETTO, S., BARILLET, F., ACUTIS, P., BOSSERS, A., LANGEVELD, J., VAN KEULEN, L., SKLAVIADIS, T., BADIOLA, J. J., ANDRÉOLETTI, O., GROSCHUP, M. H., AGRIMI, U., FOSTER, J. & GOLDMANN, W., 2009. State-of-the-art review of goat TSE in the European Union, with special emphasis on PRNP genetics and epidemiology. Veterinary Research, 40, 48.
- VACCARI, G., SCAVIA, G., SALA, M., COSSEDDU, G., CHIAPPINI, B., CONTE, M., ESPOSITO, E., LORENZETTI, R., PERFETTI, G., MARCONI, P., SCHOLL, F., BARBARO, K., BELLA, A., NONNO, R. & AGRIMI, U., 2009. Protective effect of the AT(137)RQ and ARQK(176) PrP alleles against classical scrapie in Sarda breed sheep. Veterinary Research, 40, 19.
- VAN BOGAERT, L., DEWULF, A. & PALSSON, P. A., 1978. Rida in sheep. Pathological and clinical aspects. Acta Neuropathologica, 41, 201-206.
- VAN DER MERWE, G. F., 1966. The first occurrence of scrapie in the Republic of South Africa. Journal of the South African Veterinary Association, 37, 415-418.
- VAN KEULEN, L. J., VROMANS, M. E., DOLSTRA, C. H., BOSSERS, A. & VAN ZIJDERVELD, F. G., 2008. Pathogenesis of bovine spongiform encephalopathy in sheep. Archives of Virology, 153, 445-53.
- VAN KEULEN, L. J., VROMANS, M. E. & VAN ZIJDERVELD, F. G., 2002. Early and late pathogenesis of natural scrapie infection in sheep. APMIS, 110, 23-32.
- VAN KEULEN, L. J. M., SCHREUDER, B. E., VROMANS, M. E., LANGEVELD, J. P. M. & SMITS, M. A., 2000. Pathogenesis of natural scrapie in sheep. Archives of Virology. Supplementum, 57-71.
- VARGAS, F., BOLEA, R., MONLEON, E., ACIN, C., VARGAS, A., DE BLAS, I., LUJAN, L. & BADIOLA, J. J., 2005. Clinical characterisation of natural scrapie in a native Spanish breed of sheep. Veterinary Record, 156, 318-320.
- VARGAS, F., LUJAN, L., BOLEA, R., MONLEON, E., MARTIN-BURRIEL, I., FERNANDEZ, A., DE BLAS, I. & BADIOLA, J. J., 2006. Detection and clinical evolution of scrapie in sheep by 3rd eyelid biopsy. Journal of Veterinary Internal Medicine, 20, 187-193.
- VINK, W. D. & MCINTYRE, L. H., 2014. Active surveillance for scrapie in New Zealand: towards lymphoid tissue-based testing. New Zealand Veterinary Journal, 62, 361-362.
- WANG, S., FOOTE, W. C., SUTTON, D. L., MACIULIS, A., MILLER, J. M., EVANS, R. C., HOLYOAK, G. R., CALL, J. W., BUNCH, T. D., TAYLOR, W. D. & MARSHALL, M. R., 2001. Preventing experimental vertical transmission of scrapie by embryo transfer. Theriogenology, 56, 315-27.
- WEBB, P. R., POWELL, L., DENYER, M., MARSH, S., WEAVER, C., SIMMONS, M. M., JOHNS, E., SHEEHAN, J., HORSFIELD, P., LYTH, C., WILSON, C., LONG, A., CAWTHRAW, S., SAUNDERS, G. C. & SPENCER, Y. I., 2009. A retrospective immunohistochemical study reveals atypical scrapie has existed in the United Kingdom since at least 1987. Journal of Veterinary Diagnostic Investigation, 21, 826-829.
- WILSON, R., PLINSTON, C., HUNTER, N., CASALONE, C., CORONA, C., TAGLIAVINI, F., SUARDI, S., RUGGERONE, M., MODA, F., GRAZIANO, S., SBRICCOLI, M., CARDONE, F., POCCHIARI, M., INGROSSO, L., BARON, T., RICHT, J., ANDREOLETTI, O., SIMMONS, M., LOCKEY, R., MANSON, J. C. & BARRON, R. M., 2012. Chronic wasting disease and atypical forms of bovine spongiform encephalopathy and scrapie are not transmissible to mice expressing wild-type levels of human prion protein. Journal of General Virology, 93, 1624-1629.
- WOOD, J. & DONE, S., 1992. Natural scrapie in goats: neuropathology. Veterinary Record, 131, 93-96.
- WOOD, J. L., LUND, L. J. & DONE, S. H., 1992. The natural occurrence of scrapie in moufflon. Veterinary Record, 130, 25-27.
- WOOD, J. L., MCGILL, I. S., DONE, S. H. & BRADLEY, R., 1997. Neuropathology of scrapie: a study of the distribution patterns of brain lesions in 222 cases of natural scrapie in sheep, 1982-1991. Veterinary Record, 140, 167-174.
- WOOD, J. N., DONE, S. H., PRITCHARD, G. C. & WOOLDRIDGE, M. J., 1992. Natural scrapie in goats: case histories and clinical signs. Veterinary Record, 131, 66-68.
- ZABEL, M. D. & REID, C., 2015. A brief history of prions. Pathogens and Disease, 73, ftv087.
- ZHANG, Z., ZHANG, Y., WANG, F., WANG, X., XU, Y., YANG, H., YU, G., YUAN, C. & MA, J., 2013. De novo generation of infectious prions with bacterially expressed recombinant prion protein. The FASEB Journal, 27, 4768-4775.
- ZIGAS, V. & GAJDUSEK, D. C., 1957. Kuru: clinical study of a new syndrome resembling paralysis agitans in natives of the Eastern Highlands of Australian New Guinea. Medical Journal of Australia, 44, 745-54.
- ZOMOSA-SIGNORET, V., ARNAUD, J. D., FONTES, P., ALVAREZ-MARTINEZ, M. T. & LIAUTARD, J. P., 2008. Physiological role of the cellular prion protein. Veterinary Research, 39, 9.