- Infectious Diseases of Livestock
- Part 2
- Epizootic haemorrhagic disease
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Epizootic haemorrhagic disease
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). NJ Maclachlan , G Savini and BI Osburn, Epizootic haemorrhagic disease, 2018.
Epizootic haemorrhagic disease
Previous authors: N J MACLACHLAN AND B I OSBURN
Current authors:
N J MACLACHLAN - Distinguished Professor Emeritus, BVSc, PhD, Dip ACVP, Department of Pathology, Microbiology and Immunology, VetMed 3A, School of Veterinary Medicine / Veterinary Tropical Diseases, Faculty of Veterinary Science, University of California / University of Pretoria, One Shields Ave, Davis, California, 95616, United States
G SAVINI - Head of the Animal Health Department and of the National and OIE Reference Laboratory for BT, DVM, PhD, Istituto Zooprofilatico Sperimentale dell'Abruzzo e Molise "G. Caporale" Via Campo Boario, Teramo, 64101, Italy
B I OSBURN - Emmeritus Proffessor, Faculty of Veterinary Science, University of California / University of Pretoria, One Shields Ave, Davis, California, 95616, United States
Introduction
Epizootic haemorrhagic disease (EHD) is a Culicoides-transmitted haemorrhagic viral disease of wild and domestic ruminants, particularly white-tailed deer (Odocoileus virginianus).38, 58, 63, 73 The causative agent, EHD virus (EHDV), is an orbivirus that is very closely related to bluetongue virus (BTV) and both EHDV and BTV cause a similar disease in susceptible ruminants.24, 30, 38, 41, 57, 58, 62, 68, 73
Epizootic haemorrhagic disease was first described in white-tailed deer in North America in 1955 and substantial epidemics have since been described among populations of white-tailed deer and, to a lesser extent, other wild ruminant species including pronghorn antelope (Antilocapra americana), and bighorn sheep (Ovis canadensis).16, 19, 24, 38, 52 Infection with EHDV has also been documented in many other regions of the world.9, 20, 21, 29, 33, 35, 46-48, 54, 55, 59, 71, 73 Epizootic haemorrhagic disease virus is generally not regarded as a pathogen of domestic ruminants,1, 10, 17, 32, 33, 70 a notable exception is Ibaraki virus, a specific EHDV that caused an extensive outbreak of disease in cattle in Japan in the late summer and autumn of 1959 and 1960, and continues to cause disease amongst cattle in the Far East and perhaps elsewhere (see Ibaraki disease in cattle). Similarly, in recent years, EHDV has increasingly been identified as the cause of a disease syndrome of oral ulceration and coronitis in cattle in the Mediterranean Basin, Reunion Island, South Africa, South America and the USA.7, 13, 15, 18, 21, 28, 34-36, 39, 52, 63, 67, 72, 81 The increasing number of clinical cases reported in cattle has raised concerns amongst international authorities and in 2008, the disease was included in the OIE list of multispecies/ transboundary diseases.4
Aetiology
Epizootic haemorrhagic disease virus is a member of the family Reoviridae, subfamily Sedoreovirinae, genus Orbivirus.62 It is closely related to BTV, the prototype virus of the genus Orbivirus, and to African horse sickness virus.44 Viruses within the EHDV serogroup all share group-specific serologic cross-reactivity, and at least seven distinct serotypes are distinguished on the basis of neutralization tests with type-specific antisera and genetic and phylogenetic analyses.62 A novel additional serotype detected in an alpaca in South Africa has been proposed.80 The particles of EHDV are non-enveloped and include a genome of ten double-stranded RNA segments. The virion consists of two protein shells, with an inner ‘core’ that consists of viral proteins (VP) VP1, VP3, VP4, VP6 and VP7 and an outer shell of VP2 and VP5. Five nonstructural (NS) proteins (NS1, NS2, NS3/3A, NS4)14 also are produced in EHDV-infected cells. The coding assignments and functions of each protein are identical to those of BTV (see Bluetongue).
As for BTV, there is a strong correlation between EHDV genome-sequences and the geographic origin of the strains (topotypes)5-7 but no relationship exists between serotype and virulence. Like other orbiviruses, EHDV has a segmented genome and can undergo genetic reassortment. Individual genes also undergo genetic drift thus there is considerable genetic variation amongst the strains of EHDV that circulate within endemic regions. As an example, at least 3 different serotypes of EHDV currently circulate in North America, one of which (EHDV serotype 6) is a relatively recently introduced serotype that spread into the southeastern United States from the adjacent Caribbean ecosystem. Interestingly, however, only strains of EHDV serotype 6 that include reassorted genes from the historically endemic EHDV serotypes 1 and 2 were apparently able to expand their range beyond the southeastern United States.3, 75, 78
The original Ibaraki virus that was first described in Japan in 1959 is serologically related to the prototype strain of EHDV serotype 2 that was originally isolated in Alberta in 1964 (see Ibaraki disease in cattle). The serotype-specific L2 gene of this Ibaraki virus shares only partial homology (72 per cent) with isolates of EHDV serotype 2 from the USA,56 whereas the sequence of genes encoding core proteins (VP3, VP7) of Ibaraki virus are more conserved and very similar to those of Australian strains of EHDV.43 The similarity of the core protein genes of Australian strains of EHDV and Ibaraki virus suggests that they are included in an Asian ‘topotype’ of EHDV. Interestingly, recent outbreaks of Ibaraki-like disease among cattle in Japan were caused by EHDV serotypes 6 and 7, not EHDV 2.46, 64 These same EHDV serotypes have also been recently associated with outbreaks of EHD in cattle elsewhere in the world suggesting that the distinction between Ibaraki disease in Asia and EHD of cattle elsewhere in the world is increasingly less clear.
Epidemiology
Epizootic haemorrhagic disease virus infection occurs throughout extensive portions of North America, South America, Africa, the Middle East, Australia and Asia.52, 63 Its distribution is probably similar to that of BTV and includes tropical and temperate regions throughout the world between latitudes of approximately 50 °N and 35 °S.2, 9, 20, 25, 29, 33, 38, 55, 58, 73
The infection in ruminants is not contagious; the virus is transmitted between susceptible ruminants by haematophagous midges of the genus Culicoides that serve as biological vectors of the virus.38 The specific vector species differ throughout the world; Culicoides imicola is a vector of EHDV in Africa, Culicoides brevitarsis in Australia, and Culicoides sonorensis (previously designated as C. variipennis) in the USA.73
Serological evidence of EHDV infection has been reported in many ruminant species, both wild and domestic, including sheep, cattle, various species of Asian and North American deer, elk (Cervus canadensis), moose (Alces alces), pronghorn antelope, bighorn sheep, black (Diceros bicornis) and white (Ceratotherium simum) African rhinoceros, American black bears (Ursus americanus), yaks (Bos grunniens), camelids and Florida panthers (Felis concolor) among others.2, 24, 25, 28, 31, 36, 37, 44, 56, 58, 61, 73, 79 In temperate regions infection is most common in the late summer and autumn when vector populations peak, whereas infection occurs year-round in tropical regions of the world.
Female Culicoides midges become persistently infected with EHDV and can transmit the virus to susceptible ruminants after an external extrinsic period of approximately 10 to 14 days. As in BTV infection, viraemia can be prolonged beyond 50 days in EHDV-infected ruminants because of a novel association of the virus with ruminant erythrocytes.60, 65 Truly persistent infection of ruminants has not been documented in EHDV infected ruminants, although cattle probably serve as temporary reservoir hosts of the virus.1, 10, 17, 33, 70 The cycle of infection is completed when uninfected female Culicoides midges feed on viraemic ruminants.
Pathogenesis
The pathogenesis of EHDV infection of ruminants is probably very similar or identical to that caused by BTV (see Bluetongue), and has been most comprehensively described in white-tailed deer.30, 38, 40-42, 60, 65, 69 After infection has occurred by the feeding of infected Culicoides, the virus first replicates in lymph nodes draining the site of its inoculation. It is then disseminated to secondary sites of replication such as the lungs and spleen. Many of the clinical manifestations are a reflection of widespread vascular injury and subsequent disseminated intravascular coagulation that leads to haemorrhage, oedema, and tissue necrosis (infarction).38, 41, 69 Microvascular endothelial injury is characteristic of fulminant EHD of white-tailed deer, and both BTV and EHDV clearly replicate in the endothelium of infected ruminants.42, 62, 69 A prolonged cell-associated viraemia (50 days or longer) can occur in both BTV- and EHDV-infected ruminants that survive acute infection, the viraemia persisting despite the presence of neutralizing antibody.38, 60, 65 The intimate association of virus particles with erythrocytes is probably responsible for this prolonged viraemia, which provides a ready source of virus for the haematophagous vectors.65
Clinical signs
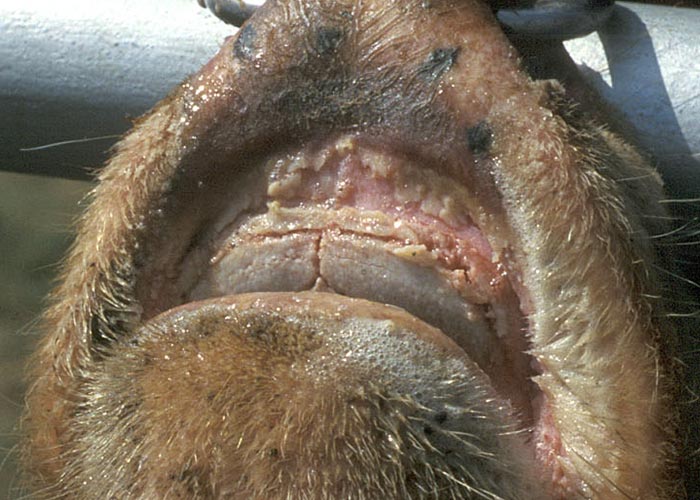
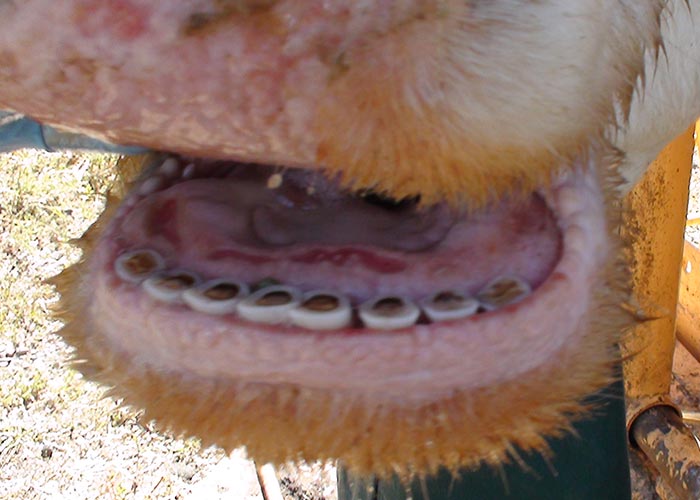
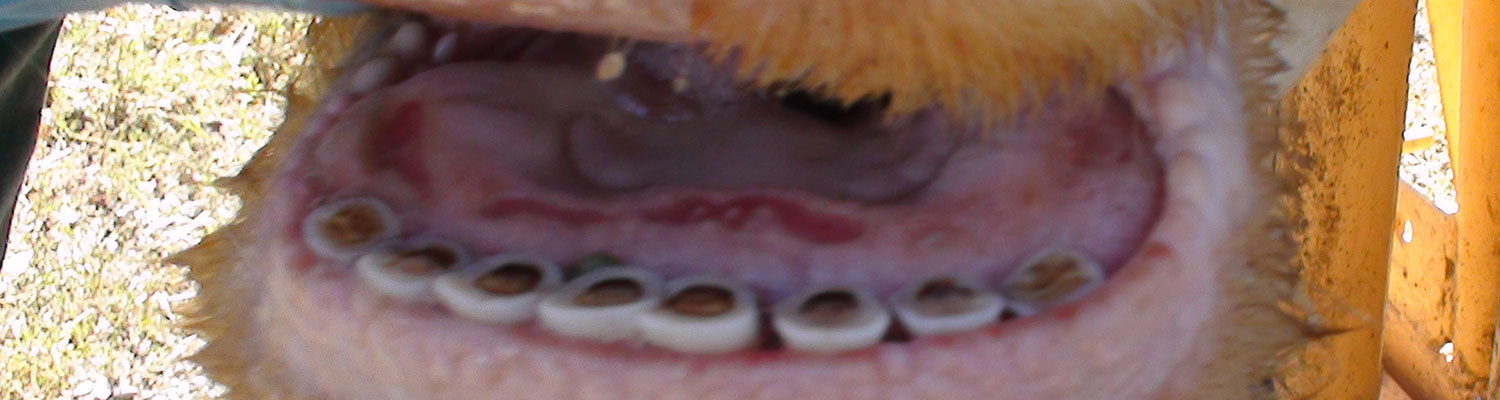
Epizootic haemorrhagic disease manifests as a haemorrhagic disease in deer, whereas EHDV infection typically is asymptomatic in domestic ruminants. The high seroprevalence of EHDV infection among domestic ruminants in endemic areas indicates that many animals survive infection or experience only subclinical infections.2, 25, 26, 29, 32, 33, 45, 57, 58 Although experimental infection of cattle with strains of EHDV isolated in the USA and Southeast Asia consistently has failed to induce clinical disease,1, 10, 17, 70 the virus increasingly has been incriminated as a cause of outbreaks of a distinctive disease syndrome among cattle in different areas of the world.13, 15, 18, 21, 34-36, 39, 52, 63, 67 Features of this disease include low morbidity and mortality, with signs in affected animals of excessive lachrymation and salivation, difficulty in swallowing, congestion and oedema of the conjunctivae, lips and tongue, linear to circular oral ulcers (Figures 1 and 2), and a stiff gait associated with visible hyperaemia at the coronary band and, occasionally, sloughing of the hoof.
Acute EHD in deer is manifested by fever, weakness, inappetance, excessive salivation, facial oedema, hyperaemia of the conjunctivae and the mucous membranes of the oral cavity, coronitis, and stomatitis with excessive salivation.30, 38 Oral ulcers may occur on the dental pad, hard palate and tongue in prolonged cases, and severe necrotizing glossitis occurs sporadically. The fulminant disease is characterized by excessive bleeding (haemorrhagic diathesis), which may manifest as bloody diarrhoea and/or haematuria, dehydration, diarrhoea, and death.
Pathology
Affected animals exhibit increasing perturbations in blood coagulation with progressive thrombocytopenia and prolongation of clotting assays such as the activated partial thromboplastin time and prothrombin time.
The lesions of EHD (see also clinical signs) have been described most comprehensively in white-tailed deer, and are characterized by extensive oral ulceration and widespread haemorrhage and oedema.30, 38 Haemorrhages vary considerably in extent (ranging from petechiae to ecchymoses) and distribution, and are often present in the skin and subcutis, mucosa and submucosa of the oral cavity, walls of the abomasum, forestomachs and urinary bladder, salivary glands, lymph nodes, serosal surfaces of the pleural and peritoneal cavities, endocardium, myocardium, and in the wall of the pulmonary artery and aorta. Hydropericardium and pulmonary oedema can be severe in acutely affected animals. Histologically, the lesions of EHD are characterized by haemorrhage and necrosis, with accompanying thrombosis of capillaries and small vessels in affected tissues. Tongue, salivary glands, walls of the forestomachs and aorta, and myocardium especially that of the papillary muscle of the left ventricle, are commonly affected.
Diagnosis
The diagnosis of EHD is accomplished by either virus detection or serology, or both. The optimal tissues for isolation of the virus include anticoagulated blood, spleen, lungs, and lymph nodes. It can be isolated by inoculation of embryonated hens’ eggs or insect (e.g. Kc and C6/36, cells derived from Culicoides sonorensis and Aedes albopictus, respectively) and/or mammal cells. The Kc cells are generally considered the most sensitive means of virus isolation,27 although Australian strains of EHDV are isolated in embryonated hens’ eggs and Aedes albopictus cell cultures just as well.33 Polymerase chain reaction (PCR)-based methods are increasingly used for detection of EHDV nucleic acid,9, 49, 51, 52, 72, 74, 76 however interpretation of these assays is complicated by the fact that EHDV nucleic acid may persist in the blood of infected ruminants far longer than does infectious virus. Both serogroup (that detect all serotypes of EHDV) targeting conserved genome segments 5, 6, 7, 9 and 10 and segment 2 directed serotype-specific RT-PCR assays are now available for rapid identification of field strains of the virus.8, 11, 12, 19, 22, 49, 50, 66, 72, 76, 77
Serological methods for detection of EHDV group-specific antibodies in the serum of ruminants include agar gel immunodiffusion test and competitive enzyme-linked immunosorbent assay (c-ELISA) that incorporates a monoclonal antibody to the EHDV group-specific antigen.31, 52, 82 While the former was widely used in the past, the more sensitive and specific c-ELISA is currently the recommended serological test for animal trade. Serotype-specific antibodies are detected by serum neutralization assay and is still the gold standard for their identification and quantification.62
Differential diagnosis
Epizootic haemorrhagic disease virus is the cause of a disease of deer and other wild ruminants that is indistinguishable from bluetongue.30, 38, 41, 42, 68 The lesions of EHD can also resemble some of those that occur in bovine virus diarrhoea/mucosal disease, vesicular stomatitis, and malignant catarrhal fever. Its diagnosis is complicated by the fact that both EHDV and BTV infections can be very common in endemic areas; thus animals in such areas are often seropositive to, or asymptomatically infected with the virus.32, 33, 57 The mere presence of either antibody or EHDV (or nucleic acid) in the blood of a ruminant is not proof of disease causality.
Serological cross-reactions with BTV further complicate the serological diagnosis of EHDV infection.17, 23 Thus, the presence of group-specific antibodies to either BTV or EHDV should always be confirmed to confirm the identity of the infecting virus. Furthermore, animals infected with more than one serotype of EHDV can produce cross-reacting antibodies that can lead to mis-identification when tested with virus-specific serum neutralization assays.
Control
With the notable exception of Ibaraki disease (see Ibaraki disease in cattle), methods to control EHDV infection are not widely available.52, 53 Vaccines are not commercially available, although they could be developed. Any efficacious vaccination strategy would involve the use of vaccines against all serotypes of EHDV endemic in a given area as humoral immunity is serotype-specific. Control of the Culicoides insect vectors theoretically might be achieved through use of insecticides and/or larvacides, the use of insect repellents on susceptible ruminants, and water management to minimize the breeding areas of those Culicoides spp. that breed in standing water.
References
- ABDY, M. J., HOWERTH, E. E. & STALLKNECHT, D. E., 1999. Experimental infection of calves with epizootic haemorrhagic disease virus. American Journal of Veterinary Research, 60, 621–626.
- AGUIRRE, A. A., MCLEAN, R. G., COOK, R. S. & QUAN, T. J., 1992. Serologic survey for selected arboviruses and other potential pathogens in wildlife in Mexico. Journal of Wildlife Diseases, 28, 435–442.
- ANBALAGAN, S., COOPER, E., KLUMPER, P., SIMONSON, R. R. & HAUSE, B. M., 2014. Whole genome analysis of epizootic hemorrhagic disease virus identified limited genome constellations and preferential reassortment. Journal of General Virology, 95, 434-441.
- ANON, 2018. Infection with Epizootic Haemorrhagic Disease Virus. OIE Code, available at http://www.oie.int/fileadmin/Home/eng/Health_standards/tahc/current/chapitre_ehd.pdf.
- ANTHONY, S. J., DARPEL, K.E., BELAGANAHALLI, M.N., MAAN, N., NOMIKOU, K., SUTTON, G., ATTOUI, H., MAAN, S. & MERTENS, P.P., 2011. RNA segment 9 exists as a duplex concatemer in an Australian strain of epizootic haemorrhagic disease virus (EHDV): Genetic analysis and evidence for the presence of concatemers as a normal feature of orbivirus replication. Virology, 420, 164-171.
- ANTHONY, S. J., DARPEL, K.E., MAAN, S., SUTTON, G., ATTOUI, H., & MERTENS, P.P., 2010. The evolution of two homologues of the core protein VP6 of epizootic haemorrhagic disease virus (EHDV), which correspond to the geographical origin of the virus. Virus Genes, 40, 67-75.
- ANTHONY, S. J., MAAN, S., MAAN, N., KGOSANA, L., BACHANEK-BANKOWSKA, K., BATTEN, C., DARPEL, K. E., SUTTON, G., ATTOUI, H. & MERTENS, P. P. C., 2009. Genetic and phylogenetic analysis of the outer-coat proteins VP2 and VP5 of epizootic haemorrhagic disease virus (EHDV): Comparison of genetic and serological data to characterise the EHDV serogroup. Virus Research, 145, 200–210.
- ARADAIB, I. E., MCBRIDE, J.W., WILSON, W.C. & OSBURN, B.I., 1995. Development of polymerase chain reaction for specific identification of epizootic hemorrhagic disease virus serotype 1. Archives of Virology, 140, 2273-2281.
- ARADAIB, I. E., MOHAMMED, M. E., MUKHTAR, M. M., GHALIB, H. W. & OSBURN, B. I., 1997. Serogrouping and topotyping of Sudanese and United States strains of epizootic haemorrhagic disease virus using PCR. Comparative Immunology and Microbiology of Infectious Diseases, 20, 211–218.
- ARADAIB, I. E., SAWYER, M. M. & OSBURN, B. I., 1994. Experimental epizootic haemorrhagic disease virus infection in calves: Virologic and serologic studies. Journal of Veterinary Diagnostic Investigation, 6, 489–492.
- ARADAIB, I. E., SMITH, W.L., OSBURN, B.I. & CULLOR, J.S., 2003. A multiplex PCR for simultaneous detection and differentiation of North American serotypes of bluetongue and epizootic hemorrhagic disease viruses. Comparative Immunology Microbiology of Infectious Diseases, 26, 77-87.
- ARADAIB, I. E., WILSON, W.C., CHENEY, I.W., PEARSON, J.E. & OSBURN, B.I., 1995. Application of PCR for specific identification of epizootic hemorrhagic disease virus serotype 2. Journal of Veterinary Diagnostic Investigation, 7, 388-392.
- BARNARD, B. J., GERDES, G. H. & MEISWINKEL, R., 1998. Some epidemiological and economic aspects of a bluetongue-like disease in cattle in South Africa. 1995/96 and 1997. Onderstepoort Journal of Veterinary Research, 65, 145–151.
- BELHOUCHET, M., MOHD, JAAFAR, F., FIRTH, A.E., GRIMES, J.M., MERTENS, P.P. & ATTOUI, H., 2011. Detection of a fourth orbivirus non-structural protein. PloS One, 6, e25697.
- BEN DHAOU, S., SAILLEAU, C., BABAY, B., VIAROUGE, C., SGHAIER, S., ZIENTARA, S., HAMMAMI, S. & BRÉARD, E., 2016. Molecular characterisation of epizootic haemorrhagic disease virus associated with a Tunisian outbreak among cattle in 2006. Acta Veterinaria Hungarica, 64, 250-262.
- BERINGER, J., HANSEN, L. P. & STALLKNECHT, D. E., 2000. An epizootic of haemorrhagic disease in white-tailed deer in Missouri. Journal of Wildlife Diseases, 36, 588–591.
- BOWEN, R. A., 1987. Serologic responses of calves to sequential infections with epizootic haemorrhagic disease virus serotypes. American Journal of Veterinary Research, 48, 1449–1452.
- BRÉARD, E., SAILLEAU, C., HAMBLIN, C., GRAHAM, S. D., GOURREAU, J. M. & ZIENTARA, S., 2004. Outbreak of epizootic haemorrhagic disease on the island of Réunion. The Veterinary Record, 155, 422-423.
- BRODIE, S. J., BARDSLEY, K. D., DIEM, K., MECHAM, J. O., NORELIUS, S. E. & WILSON, W. C., 1998. Epizootic haemorrhagic disease: Analysis of tissues by amplification and in situ hybridization reveals widespread orbivirus infection at low copy numbers. Journal of Virology, 72, 3863–3871.
- BROWN, S. E., GORMAN, B. M., TESH, R. B. & KNUDSON, D. L., 1992. Isolation of bluetongue and epizootic haemorrhagic disease viruses from mosquitoes collected in Indonesia. Veterinary Microbiology, 32, 241–252
- CÊTRE-SOSSAH, C., ROGER, M., SAILLEAU, C., RIEAU, L., ZIENTARA, S., BRÉARD, E., VIAROUGE, C., BERAL, M., ESNAULT, O. & CARDINALE, E., 2014. Epizootic haemorrhagic disease virus in Reunion Island: evidence for the circulation of a new serotype and associated risk factors. Veterinary Microbiology, 170, 383-390.
- CLAVIJO, A., SUN, F., LESTER, T., JASPERSON, D.C. & WILSON, W.C., 2010. An improved real-time polymerase chain reaction for the simultaneous detection of all serotypes of Epizootic hemorrhagic disease virus. Journal of Veterinary Diagnostic Investigation, 22, 588-593.
- DELLA-PORTA, A. J., PARSONSON, I. M. & MCPHEE, D. A., 1985. Problems in the interpretation of diagnostic tests due to cross-reactions between orbiviruses and broad serological responses in animals. Progress in Clinical and Biological Research. 178, 445–453.
- DULAC, G. C., DUBUC, C., AFSHAR, A., MYERS, D. J., BOUTTARD, A., SHAPIRO, J., SHETTIGARA, P. T. & WARD, D., 1988. Consecutive outbreaks of epizootic haemorrhagic disease of deer and bluetongue. The Veterinary Record, 122, 340.
- DUNBAR, M. R., CUNNINGHAM, M. W. & ROOF, J. C., 1998. Seroprevalence of selected disease agents from free-ranging black bears in Florida. Journal of Wildlife Diseases, 34, 612–619.
- DUNBAR, M. R., VELARDE, R., GREGG, M. A. & BRAY, M., 1999. Health evaluation of a pronghorn population in Oregon. Journal of Wildlife Diseases, 35, 496–510.
- ESCHBAUMER, M., WERNIKE, K., BATTEN, C. A., SAVINI, G., EDWARDS, L., DI GENNARO, A., TEODORI, L., OURA, C. A., BEER, M. & HOFFMANN, B., 2012. Epizootic hemorrhagic disease virus serotype 7 in European cattle and sheep: Diagnostic considerations and effect of previous BTV exposure. Veterinary Microbiology, 159(3-4), 298-306.
- FAVERO, C. M., MATOS, A.C., CAMPOS, F.S., CANDIDO, M.V., COSTA, E.A., HEINEMANN, M.B., BARBOSA-STANCIOLI, E.F. & LOABATO, Z.I., 2013. Epizootic hemorrhagic disease in brocket deer, Brazil. Emerging Infectious Diseases, 19, 346-348.
- FISCHER-TENHAGEN, C., HAMBLIN, C., QUANDT, S. & FROLICH, K., 2000. Serosurvey for selected infectious disease agents in free-ranging black and white rhinoceros in Africa. Journal of Wildlife Diseases, 36, 316–323.
- FLETCH, A. L. & KARSTAD, L. H., 1971. Studies on the pathogenesis of experimental epizootic haemorrhagic disease of white-tailed deer. Canadian Journal of Comparative Medicine, 35, 224–229.
- FORZAN, M., PIZZURRO, F., ZACCARIA, G., MAZZEI, M., SPEDICATO, M., CARMINE, I., SALINI, R., TOLARI, F., CERRI, D., SAVINI, G. & LORUSSO, A., 2017. Competitive enzyme-linked immunosorbent assay using baculovirus-expressed VP7 for detection of epizootic haemorrhagic disease virus (EHDV) antibodies. Journal of Virological Methods, 248, 212-216.
- FULTON, R. W., BURGE, L. J. & CUMMINS, J. M., 1989. Neutralizing antibody responses to bluetongue and epizootic haemorrhagic disease virus serotypes in beef cattle. American Journal of Veterinary Research, 50, 651–654.
- GARD, G. P., WEIR, R. P. & WALSH, S. J., 1988. Arboviruses recovered from sentinel cattle using several isolation methods. Veterinary Microbiology, 18, 119-125.
- GARRETT, E. F., PO, E., BICHI, E. R., HEXUM, S. K., MELCHER, R. & HUBNER, A. M., 2015. Clinical disease associated with epizootic hemorrhagic disease virus in cattle in Illinois. Journal of the American Veterinary Medical Association, 247, 190-195.
- GERDES, G. H., NESER, J. A., BARNARD, B. J. & LARSEN, J., 1996. Stomatitis and coronitis in cattle — an insect-borne viral disease. Journal of the South African Veterinary Association, 67, 103-104.
- GOLENDER, N., KHINICH, Y., GOROHOV, A., ABRAMOVITZ, I. & BUMBAROV, V., 2017. Epizootic hemorrhagic disease virus serotype 6 outbreak in Israeli cattle in 2015. Journal of Veterinary Diagnostic Investigation, 29, 885-888.
- HASSINE, T. B., AMDOUNI, J., MONACO, F., SAVINI, G., SGHAIER, S., SELIMEN, I. B., CHANDOUL, W., HAMIDA, K. B. & HAMMAMI, S., 2017. Emerging vector-borne diseases in dromedaries in Tunisia: West Nile, bluetongue, epizootic haemorrhagic disease and Rift Valley fever. Onderstepoort Journal of Veterinary Research, 84, e1-e3.
- HOFF, G. L. & TRAINER, D. O., 1978. Bluetongue and epizootic haemorrhagic disease viruses: Their relationship to wildlife species. Advances in Veterinary Science and Comparative Medicine, 22, 111–132.
- HOUSE, C., SHIPMAN, L. D. & WEYBRIGHT, G., 1998. Serological diagnosis of epizootic haemorrhagic disease in cattle in the USA with lesions suggestive of vesicular disease. Annals of the New York Academy of Sciences, 849, 497–500.
- HOWERTH, E. W., 2015. Cytokine release and endothelial dysfunction: a perfect storm in orbivirus pathogenesis. Veterinaria Italiana, 51, 275-281.
- HOWERTH, E. W., GREENE, C. E. & PRESTWOOD, A. K., 1988. Experimentally induced bluetongue virus infection in white-tailed deer: Coagulation, clinical pathologic, and gross pathologic changes. American Journal of Veterinary Research, 49, 1906–1913.
- HOWERTH, E. W. & TYLER, D. E., 1988. Experimentally induced bluetongue virus infection in white-tailed deer: Ultrastructural findings. American Journal of Veterinary Research, 49, 1914–1922.
- IWATA, H., MANABE, S., YOSHIDA, A., PEREIRA, E. M. & INOUE, T., 2001. The complete nucleotide sequences of L3 and S7 segments of Ibaraki virus encoding for the major inner capsid proteins, VP3 and VP7. Journal of Veterinary Medical Science, 63, 73-78.
- IWATA, H., YAMAGAWA, M. & ROY, P., 1992. Evolutionary relationships among gnat-transmitted orbiviruses that cause African horse sickness, bluetongue, and epizootic haemorrhagic disease as evidenced by their capsid protein sequences. Virology, 191, 251–261.
- JOHNSON, J. L., BARBER, T. L., FREY, M. L. & NASON, G., 1986. Serosurvey for selected pathogens in hunter-killed pronghorns in western Nebraska. Journal of Wildlife Diseases, 22, 87–90.
- KAMOMAE, Y., KAMOMAE, M., OHTA, Y., NABE, M., KAGAWA, Y., OGURA, Y., KATO, T., TANAKA, S., YANASE, T. & SHIRAFUJI, H., 2018. Epizootic hemorrhagic disease virus serotype 6 infection in cattle, Japan, 2015. Emerging Infectious Diseases, 24, 902-905.
- KATO, T., SHIRAFUJI, H., TANAKA, S., SATO, M., YAMAKAWA, M., TSUDA, T. & YANASE, T., 2016. Bovine Arboviruses in Culicoides Biting Midges and Sentinel Cattle in Southern Japan from 2003 to 2013. Transboundary and Emerging Diseases, 63, e160-e172.
- KEDMI, M., GALON, N., HERZIGER, Y., YADIN, H., BOMBAROV, V., BATTEN, C., SHPIGEL, N. Y. & KLEMENT, E., 2011. Comparison of the epidemiology of epizootic haemorrhagic disease and bluetongue viruses in dairy cattle in Israel. Veterinary Journal, 190, 77-83.
- MAAN, N. S., MAAN, S., NOMIKOU, K., JOHNSON, D. J., EL HARRAK, M., MADANI, H., YADIN, H., INCOGLU, S., YESILBAG, K., ALLISON, A. B., STALLKNECHT, D. E., BATTEN, C., ANTHONY, S. J. & MERTENS, P. P., 2010. RT-PCR assays for seven serotypes of epizootic haemorrhagic disease virus & their use to type strains from the Mediterranean region and North America. PloS One, 5, e12782.
- MAAN, N. S., MAAN, S., POTGIETER, A. C., WRIGHT, I. M., BELAGANAHALLI, M. & MERTENS, P. P. C., 2017. Development of Real-Time RT-PCR Assays for Detection and Typing of Epizootic Haemorrhagic Disease Virus. Transboundary and Emerging Diseases, 1120-1132.
- MAAN, N. S., MAAN, S., POTGIETER, A.C., WRIGHT, I. M., BELAGANAHALLI, M. & MERTENS, P.P.C., , 2016. Development of real-time RT-PCR assays for detection and typing of epizootic haemorrhagic disease virus. Transboundary and Emerging Diseases, 64, 1120–1132.
- MACLACHLAN, N. J., ZIENTARA, S., SAVINI, G. & DANIELS, P. W., 2015. Epizootic haemorrhagic disease. Revue Scientifique et Technique, 34, 341-351.
- MCVEY, D. S. & J., M. N., 2015. Vaccines for Prevention of Bluetongue and Epizootic Hemorrhagic Disease in Livestock: A North American Perspective. Vector-Borne and Zoonotic Diseases, 15, 385-396.
- MEJRI, S., DHAOU, S. B., JEMLI, M., BRÉARD, E., SAILLEAU, C., SGHAIER, S., ZOUARI, M., LORUSSO, A., SAVINI, G., ZIENTARA, S. & HAMMAMI, S., 2018. Epizootic haemorrhagic disease virus circulation in Tunisia. Veterinaria Italiana, 54, 87-90.
- MOORE, D. L., 1974. Bluetongue and related viruses in Ibadan, Nigeria: Serologic comparison of bluetongue, epizootic haemorrhagic disease of deer, and Abadina (Palyam) viral isolates. American Journal of Veterinary Research, 35, 1109–1113.
- NARA PEREIRA, E. M., IWATA, H. & INOUE, T., 2000. The complete nucleotide sequence of segment L2 of Ibaraki virus encoding for the antigen recognized by neutralizing antibodies. Journal of Veterinary Medical Science, 62, 317–321.
- ODIAWA, G., BLUE, J. L., TYLER, D. E. & SHOTTS, E. B., 1985. Bluetongue and epizootic haemorrhagic disease in ruminants in Georgia: Survey by serotest and virologic isolation. American Journal of Veterinary Research, 46, 2193–2196.
- PASICK, J., HANEL, K., ZHOU, E. M., CLAVIJO, A., COATES, J., ROBINSON, Y. & LINCOLN, B., 2001. Incursion of epizootic haemorrhagic disease into the Okanagan Valley, British Columbia in 1999. Canadian Veterinary Journal, 42, 207-209.
- PYBUS, M. J., RAVI, M. & POLLOCK, C., 2014. Epizootic hemorrhagic disease in Alberta, Canada. Journal of Wildlife Diseases, 50, 720-722.
- QUIST, C. F., HOWERTH, E. W., STALLKNECHT, D. E., BROWN, J., PISELL, T. & NETTLES, V. F., 1997. Host defence responses associated with experimental haemorrhagic disease in white-tailed deer. Journal of Wildlife Diseases, 33, 584–599.
- RAABIS, S. M., BYERS, S. R., HAN, S. & CALLAN, R. J., 2014. Epizootic hemorrhagic disease in a yak. Canadian Veterinary Journal, 55, 369-372.
- ROY, P., 2001. The Orbiviruses., In: KNIPE, D. M. & HOWLEY, P. M., (eds.). Fields Virology, 4th edn. Philadelphia: Lippincott, Williams & Wilkins,
- SAVINI, G., AFONSO, A., MELLOR, P., ARADAIB, I., YADIN, H., SANAA, M., WILSON, W., MONACO, F. & DOMINGO, M., 2011. Epizootic heamorragic disease. Research in Veterinary Science, 91, 1-17.
- SHIRAFUJI, H., KATO, T., YAMAKAWA, M., TANAKA, T., MINEMORI, Y. & YANASE, T., 2017. Characterization of genome segments 2, 3 and 6 of epizootic hemorrhagic disease virus strains isolated in Japan in 1985–2013: Identification of their serotypes and geographical genetic types. Infection, Genetics and Evolution, 53, 38-46.
- STALLKNECHT, D. E., HOWERTH, E. W., KELLOGG, M. L., QUIST, C. F. & PISELL, T., 1997. In vitro replication of epizootic haemorrhagic disease and bluetongue viruses in white-tailed deer peripheral blood mononuclear cells and virus-cell association during in vivo infections. Journal of Wildlife Diseases, 33, 574–583.
- SUN, F., COCHRAN, M., BECKHAM, T. & CLAVIJO, A., 2014. Molecular typing of epizootic hemorrhagic disease virus serotypes by one-step multiplex RT-PCR. Journal of Wildlife Diseases, 50, 639-644.
- TEMIZEL, E. M., YESILBAG, K., BATTEN, C., SENTURK, S., MAAN, N. S., MERTENS, P. P. C. & BATMAZ, H., 2009. Epizootic hemorrhagic disease in cattle, Western Turkey. Emerging Infectious Diseases, 15, 317-319.
- THORNE, E., WILLIAMS, E. S., SPRAKER, T. R., HELMS, W. & SEGERSTROM, T., 1988. Bluetongue in free-ranging pronghorn antelope (Antilocapra americana) in Wyoming. Journal of Wildlife Diseases, 24, 113–119.
- TSAI, K. & KARSTAD, L., 1973. The pathogenesis of epizootic haemorrhagic disease of deer: An electron microscopic study. American Journal of Pathology, 70, 379–400
- UREN, M. F., 1986. Clinical and pathological responses of sheep and cattle to experimental infection with five different viruses of the epizootic haemorrhagic disease of deer serogroup. Australian Veterinary Journal, 63, 199–201.
- VERDEZOTO, J., BREARD, E., VIAROUGE, C., QUENAULT, H., LUCAS, P., SAILLEAU, C., ZIENTARA, S., AUGOT, D. & ZAPATA, S., 2018. Novel serotype of bluetongue virus in South America and first report of epizootic haemorrhagic disease virus in Ecuador. Transboundary and Emerging Diseases, 65, 244-247.
- VIAROUGE, C., BREARD, E., ZIENTARA, S., VITOUR, D. & SAILLEAU, C., 2015. Duplex Real-Time RT-PCR Assays for the Detection and Typing of Epizootic Haemorrhagic Disease Virus. PloS One, 10, e0132540.
- WALTON, T. E. & OSBURN, B. I., 1992. Bluetongue and Related Orbiviruses. Boca Raton, Florida: CRC Press.
- WILSON, W. C., DANIELS, P., OSTLUND, E. N., JOHNSON, D. E., OBERST, R. D., HAIRGROVE, T. B., MEDIGER, J. & MCINTOSH, M. T., 2015. Diagnostic Tools for Bluetongue and Epizootic Hemorrhagic Disease Viruses Applicable to North American Veterinary Diagnosticians. Vector-Borne and Zoonotic Diseases, 15, 364-373.
- WILSON, W. C., GAUDREAULT, N. N., JASPERSON, D. C., JOHNSON, D. J., OSTLUND, E. N., CHASE, C. L. & RUDER, M. G., 2015. Stallknecht DE: Molecular evolution of American field strains of bluetongue and epizootic haemorrhagic disease viruses. Veterinaria Italiana, 51, 269-273.
- WILSON, W. C., HINDSON, B. J., O'HEARN, E. S., HALL, S., TELLGREN-ROTH, C., TORRES, C., NARAGHI-ARANI, P., MECHAM, J. O. & LENHOFF, R. J., 2009. A multiplex real-time reverse transcription polymerase chain reaction assay for detection and differentiation of Bluetongue virus and Epizootic hemorrhagic disease virus serogroups. Journal of Veterinary Diagnostic Investigation, 21, 760-770.
- WILSON, W. C., O’HEARN, E.S., TELLGREN-ROTH, C., STALLKNECHT, D.E., MEAD, D.G. & MECHAM, J.O., 2009b. Detection of all eight serotypes of Epizootic hemorrhagic disease virus by real-time reverse transcription polymerase chain reaction. Journal of Veterinary Diagnostic Investigation, 21, 220-225.
- WILSON, W. C., RUDER, M. G., JASPERSON, D., SMITH, T. P., NARAGHI-ARANI, P., LENHOFF, R., STALLKNECHT, D. E., VALDIVA-GRANDA, W. A. & SHEORAN, D., 2016. Molecular evolution of epizootic hemorrhagic disease viruses in North America based on historical isolates using motif fingerprints. Virus Genes, 52, 495-508.
- WORK, T. M., JESSUP, D. A. & SAWYER, M. M., 1992. Experimental bluetongue and epizootic haemorrhagic disease virus infection in California black-tailed deer. Journal of Wildlife Diseases, 28, 623–628.
- WRIGHT, I. M., 2013. Serological and Genetic Characterisation of Putative New Serotypes of Bluetongue Virus and Epizootic Haemorrhagic Disease Virus Isolated From an Alpaca. Dissertation submitted in partial fulfilment of the requirements for the degree Magister Scientiae in Biochemistry at the Potchefstroom Campus of the North-West University. North West University, South Africa, 102.
- YADIN, H., BRENNER, J., BUMBROV, V., OVED, Z., STRAM, Y., KLEMENT, E., PERL, S., ANTHONY, S., MAAN, S., BATTEN, C. & MERTENS, P.P., 2008. Epizootic haemorrhagic disease virus type 7 infection in cattle in Israel. The Veterinary Record, 162, 53-56.
- ZHOU, E. & AFSHAR, A., 1999. Characterization of monoclonal antibodies to epizootic haemorrhagic disease virus of deer (EHDV) and bluetongue virus by immunisation of mice with EHDV recombinant VP7 antigen. Research in Veterinary Science, 66, 247–252.