- Infectious Diseases of Livestock
- Part 1
- Vectors: Tsetse flies
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Vectors: Tsetse flies
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Vectors: Tsetse flies
Previous authors: R J PHELPS AND D F LOVEMORE
Current authors:
J BOUYER - Medical Entomologist, DVM, PhD, HDR, Insect Pest Control Laboratory, Joint FAO/IAEA Division of Nuclear Techniques in Food and Agriculture, Vienna, Austria & UMR CIRAD-Inra ASTRE « Animal, Santé, Territoires, Risques et Ecosystèmes », Montpellier, France
M J B VREYSEN - Laboratory Head, MSc, PhD, Insect Pest Control Laboratory, Wagramerstrasse 5, Vienna, Austria
Introduction
Blood-sucking muscoid Diptera of the family Glossinidae and the genus Glossina, known as tsetse flies, are restricted almost entirely to sub-Saharan Africa and some of its off-shore islands such as Mafia Island, although they are absent from others such as Madagascar and Pemba Island of Zanzibar. There are old records of tsetse flies from south-west Arabia,64 and near Gizar in Saudi Arabia,117 but they have not been reported there since then. It had long been known that domestic stock could not be kept where tsetse flies occurred and that disease was associated with the flies. However, it was not until Bruce, working in KwaZulu-Natal, South Africa in 1895, demonstrated the transmission of pathogenic trypanosomes of livestock by tsetse flies that the reason for livestock deaths became known. The role of tsetse flies as vectors of human trypanosomosis, and the importance of the Glossina palpalis group in this respect, was also demonstrated by Bruce and his co-workers in 1909.50 Nash (1969)270 appropriately referred to tsetse flies as ‘Africa’s bane’270 as they are the sole cyclical vectors of trypanosomes, the causative agents of ‘sleeping sickness’ or human African trypanosomosis (HAT) in humans and ‘nagana’ or African animal trypanosomosis (AAT) in livestock. During feeding on an infected host, the parasites are together with the blood, ingested by the fly, after which they undergo a cyclical development cycle in the fly.
Although there are 31 species and subspecies of tsetse flies257 all placed in the genus Glossina of the family Glossinidae,60 only eight to 10 species are considered of economic (agricultural-veterinary) or human sanitary importance. Tsetse flies occur in 38 African countries and infest an area of close to 10 million km2. These areas could not be explored easily before the advent of mechanized transport, as animal-drawn transport was excluded by AAT. The presence of tsetse flies was thus a major obstacle to the development of much of the continent, a situation that obtains in many places even today.8
There are those, even now, who look upon the tsetse fly as the guardian of the natural ecosystems of Africa, and who would like this fly to remain until humans have learnt to manage the land in a sustainable manner. At present, though, the human population of Africa is expanding rapidly, and the economic situation is such that the burgeoning population can, for the most part, be accommodated only as peasant farmers. The importance of livestock to such people is great, not only as a source of food, draught power, and money, but also for the important role that livestock, especially cattle, play in cultural affairs. Under these circumstances it is essential to try to manage the diseases of livestock, amongst which AAT plays a major role.
In view of the vast areas infested with tsetse flies, it is estimated that 60 million people are constantly exposed to the risk of becoming infected with HAT. Whereas in 2001, according to the World Health Organization, between 300 000-500 000 people were still suffering from HAT.65 The prevalence of HAT has declined drastically in the last decade due to increased surveillance, readily available drugs, more trained technicians, and a greater commitment of the international community.323, 324
Socio-economic impact
Tsetse flies, hunger, poverty
Hunger is the most extreme manifestation of poverty and with 34 per cent of the population being undernourished, it remains acute in rural sub-Saharan Africa. Alleviation of poverty can only start with the reduction of hunger and this can be achieved through the development of sustainable agricultural systems. Livestock is an essential part of this as it provide milk and meat for nourishment, transport, hides, manure for fuel or fertilizer, and it can support crop production through the provision of draught power. It is also an important means of savings for the farmers and is an essential source of income for the rural poor.124 Productive livestock is, however, largely absent in these vast fertile tsetse infested areas due to the omnipresence of an inconspicuous insect: the tsetse fly.396
The socio-economic impact of tsetse flies in sub-Saharan Africa is enormous. Direct annual cattle production losses due to the presence of the tsetse fly and AAT have been estimated at USD 600–1200 million,174 and the overall annual lost potential in livestock and crop production has been estimated at USD 4750 million.51 Importantly, the tsetse fly prevents the integration of crop farming and livestock keeping, which is considered crucial to the development of more efficient and sustainable livestock production systems.8 The presence of the tsetse fly and AAT is therefore considered to be one of the major root causes of hunger and poverty in sub-Sahara Africa; hence, the tsetse fly has been appropriately termed “the poverty fly”. This is evidenced by the remarkable overlap between the 38 tsetse-infested countries and the 34 heavily indebted poor countries in Africa.124
This link between tsetse flies, hunger and poverty is unfortunately being ignored by many decision and policy makers because it is perceived as a problem of the rural poor (85 per cent of the African poor are estimated to live in rural areas, of which 80 per cent rely on agriculture for their livelihood).247 Actions against this problem are only expected to provide results in the medium- or long- term, which is usually beyond the interest of many local politicians and/or international donors.124
Potential impact of tsetse control
In view of the above, it is therefore pertinent to solve the tsetse and trypanosomosis problem to alleviate hunger and poverty in Africa. African animal trypanosomosis significantly impairs agricultural and rural development, with an estimated 45-50 million cattle living under permanent risk of becoming infected with the disease; left untreated, the disease is often fatal.320, 341 An analysis of the direct (mortality, fertility, milk production, animal traction and weight) and indirect effects of AAT on the productivity of livestock indicated that annual calf mortality is 6-20 per cent higher, calving rate is 6-19 per cent lower, and milk yield is 20 per cent lower for livestock kept under AAT challenge compared with livestock outside the tsetse belt. An additional direct effect is weight loss reduced ploughing efficiency of oxen.178, 320 In addition, the introduction of more productive exotic cattle or cross-breeds is difficult, if not impossible, in AAT infested areas, where the disease impacts negatively on cattle population growth, their distribution, herd size and structure, prohibits the development of mixed farming systems and inhibits human settlements.25, 29, 124
A dynamic herd model210 indicated that the benefits of enhanced AAT control, alone in terms of increased meat and milk production would be USD 700 million per year. Although the number of cattle would increase under reduced AAT challenge, the creation of tsetse-free zones would stimulate the keeping of highly productive cross-breeds that can be kept at a lower average density than less productive breeds, which would help in preventing overgrazing, and hence, be beneficial for the environment, especially in fragile ecosystems.25
Case studies
Tsetse eradication on Unguja Island, Zanzibar: Two and five years after completion of the campaign that eradicated a population of Glossina austeni from Unguja Island, Zanzibar, two surveys were conducted to assess the socio -economic impact of this campaign. The data indicated that (1) the proportion of small farmer holdings with indigenous cattle increased from 31 to 94 per cent between 1985 and 2002, (2) the selling of milk from indigenous cattle increased from 11 to 62 per cent between 1985 and 1999, and (3) using oxen for ploughing increased to 5 per cent in 2002. The demand for improved livestock breeds was high and the percentage of holdings with improved cattle breeds increased from 2 to 24 per cent from 1985 to 2002. In addition, from 1985 to 1999, milk production nearly tripled. In general, the average monthly income of farming households increased by 30 per cent from 1999 to 2002, and the proportion of households with a monthly income of over 25 USD and over 50 USD increased from 69 to 86 per cent and from 22 to 36 per cent, respectively. This was associated with tsetse and AAT eradication as a strong correlation was observed between household income and milk yields, milk sales, and the use of manure and animal draught power for cultivation and transport.398
The eradication of Glossina austeni from Unguja Island in 1997 followed by the disappearance of AAT thereafter115 enabled farmers to integrate livestock keeping with cropping in areas where this had been impossible before. The increased livestock and crop productivity and the use of animals for transport and traction significantly contributed to an increase in the quality of people's lives.398
Tsetse eradication in the Niayes of Senegal: A socio-economic study assessed the potential benefits of the sustainable removal of Glossina pallidipes gambiensis from the Niayes, an area north-east of the capital Dakar in Senegal. Cattle were kept using three main farming systems, i.e. (1) a traditional system with trypanotolerant cattle, and (2) two “improved” systems with more productive cattle breeds for milk and meat production. Herd size was 45 per cent lower and annual cattle sales > 3 times higher in the improved farming systems compared with the traditional farming system (€ 250 (SD 513) per head versus € 74 (SD 38) per head, respectively). Thirty-four per cent (SD 4 per cent) of the farmers living in the tsetse-free areas owed improved breeds, whereas in the tsetse-infested areas only 6 per cent (SD 4 per cent) of the farmers did. After the sustainable removal of the tsetse fly population, the study considered two possible scenarios with respect to potential increases of cattle sales. The first, conservative scenario assumed a 2 per cent annual replacement rate of traditional by improved systems, and the second, more realistic scenario, assumed an increased replacement rate of 10 per cent five years after the eradication of the tsetse fly population. The final annual increase of cattle sales was estimated at ~€ 2800/km2 for a total cost of the eradication campaign of ~€ 6400/km2. Despite this relatively high cost, the benefit-cost analysis indicated that the project was highly cost-effective, with internal rates of return of 9.8 and 19.1 per cent and payback periods of 18 and 13 years for the two scenarios, respectively.27
Tsetse eradication in KwaZulu-Natal, South Africa: A socio-economic study in KwaZulu-Natal showed significant economic benefits should tsetse flies be removed from South Africa. The sustained removal of the two species present (Glossina austeni and Glossina brevipalpis) using an AW-IPM (Area Wide-Integrated Pest Management) approach that would include two cycles of SAT (Sterile or Surgical aseptic technique) followed by the release of sterile males, would reach a breakeven point after eight years. A cumulative total net benefit (net present value, taking into account a discount rate of eight per cent) of USD 51 million and an overall benefit to cost ratio of 3.4 would be obtained over a 15-year period, not taking into account additional benefits, such as improved agricultural productivity due to improved health of draught animals. As from year 9, the project would reach the maintenance phase and benefits would be fully established; the annual benefit cost ratio would fluctuate between USD 90 to 493 for each dollar invested (year 9 to year 15). Moreover, the project would have an internal rate of return of 23 per cent, meaning that the discount rate could be almost three times higher than the estimated value of eight per cent and the project would still break even in a 15-year time frame.199
Biology of tsetse flies
Anatomy and sensory systems
Detailed studies have been made of the anatomy69, 263 and the physiology218 of tsetse flies. Leak228 has recorded the advances in knowledge of the biology and ecology of tsetse flies over the previous 30 years.
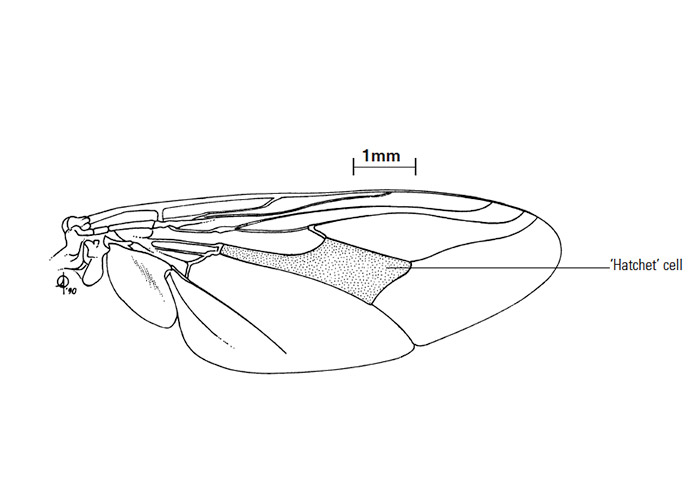
Tsetse flies (Figure 1) have one pair of membranous, mesothoracic, functional wings, while the metathoracic wings are modified to form halteres. They can fly at about 6,3 metres per second136 and the power of lift generated by their wings is unusually large.159 The age of tsetse flies may be estimated from the amount of wear on the hind margin of their wings, while the length of the vein forming the blade of the ‘hatchet’ cell (Figure 2) can be used as an index to the size of the fly.189 Geometric morphometrics further use the size and shape of the wing based on selected landmarks to infer parameters on the structure and dynamics of wild populations.36, 329 The energy substrate used in flight is proline,57 and the stored energy is in the form of abdominal fat. The amount of proline available to the flight muscles in the thorax is limited, so the fly cannot make sustained flights. When this fuel is exhausted, the fly must rest until further proline has been synthesized in the cells of the abdominal fat body58 and transported to the flight muscles of the thorax.
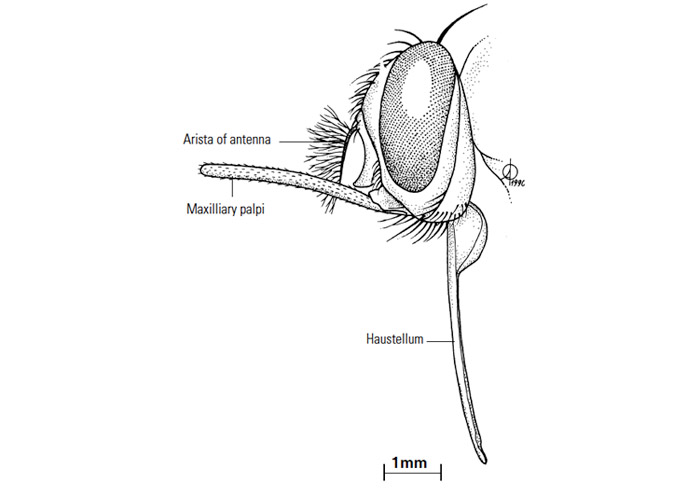
The functional mouthparts, or haustellum, of the tsetse fly consist of the labium, which is trough-like in cross section and terminates anteriorly with the denticulate labellum; the labrum, lying along the dorsal surface of the labial trough; and the hypopharynx, which lies within the tube formed between the labium and labrum and which has the salivary glands attached to its base. There are no mandibles, and the maxillae are represented by the stout maxillary palpi (Figure 3) between which the haustellum lies when at rest. The haustellum, or the haustellum plus the palpi, is often termed the proboscis. When the fly is feeding, the haustellum is lowered, the labellar teeth cut into the host’s skin and the haustellum positioned in the wound. Blood is then sucked up the central lumen of the haustellum, and saliva containing an anticoagulant is released into the wound. The labium is well supplied with sensilla which monitor the blood flow and quality.
Temperature of the host’s skin is a primary stimulus for the fly to start probing for a feeding site, and adenosine-5-triphosphate in the host’s blood stimulates the fly to engorge.134
Both sexes of the tsetse fly are obligate blood suckers and generally feed at three- to five-day intervals, although they may feed more frequently if the opportunity occurs. The blood meal is the source of both nutrition and water for the fly, although evidence from laboratory studies does exist that hungry tsetse flies can feed on water and water with sugar when no blood is available.334 Food reserves in the form of fat, and the amount of residual blood in a fly, give an indication of its nutritional status,307 to which its behaviour is related.46
Tsetse flies can detect odours by means of sensilla situated on the antennae; if their antennae (Figure 3) are removed, flies are less able to detect their hosts than are intact flies.375 Substances (kairomones) produced in the host’s breath, such as carbon dioxide and acetone, attract tsetse flies,373 while a further attractive component of cattle breath is 1-octen-3-ol.368 Kairomones in the urine of African buffaloes and cattle which attract tsetse flies have been isolated and identified as phenols.276, 381 However, some kairomones are repellent; the odour of humans repels some species of tsetse flies, notably Glossina pallidipes.372 One of the phenols present in ox urine, 2-methoxyphenol, is repellent to Glossina pallidipes and reduces trap catches by 60 per cent.359 Placing a source of 2-methoxyphenol next to a bait ox reduced the numbers of flies attracted to the ox, but did not affect the proportion of flies feeding on the ox after arrival. Only in areas of very light disease challenge would the repellent be of use in protecting animals in the field.359 Non-volatile substances may also be detected by sensilla, as is shown by the initiation of copulatory responses in the male fly by the contact of its tibial receptors with female sex pheromone, in the form of a wax on the surface of the female’s thorax.222
Well-developed compound eyes are present in the tsetse fly and are important in host location. Hungry flies and especially the morsitans group are particularly attracted to moving objects but at shorter ranges they can also recognize stationary hosts provided these are large enough to be resolved by the eyes, which have an absolute resolution of about three degrees.364 The eyes can also distinguish between light and dark, a useful attribute for seeking out shaded microclimates when ambient temperatures are at lethal levels.177 The spectral sensitivity of the compound eyes of tsetse flies has been investigated148 and it has been shown that it is the spectral reflectivity of the object which is detected by the fly. Blue-green and red covers on fly traps increase the capture rate of Glossina pallidipes and Glossina morsitans, while green, yellow and orange covers have the opposite effect.149 Comparison with a graded series of grey traps indicated that the flies employ some colour information and not contrast information alone. The same effects have been obtained with flat cloth screens of different colours.150
Eyes contain pteridine pigments that accumulate with age. This feature provides a cheap and convenient indicator of the mean age of a population221 (Lehane and Mail 1985) but the level of pteridine accumulation is highly dependent on temperature and fly size, and the method can therefore not be used to determine the precise age of individual insects.395
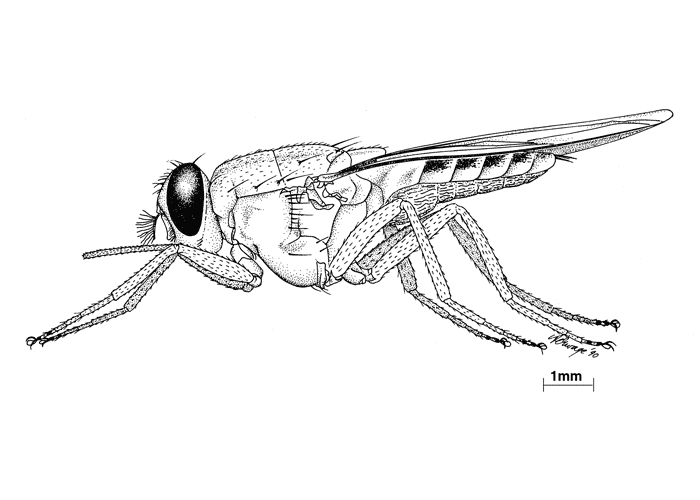
The sexes are easily recognized in tsetse flies as the male hypopygium is well developed (Figure 4). The male claspers are concealed within the hypopygium when not in use and dissection is necessary to display these structures, which are important for species identification. The internal reproductive system of females may be used to determine the physiological age of flies, as the shedding of eggs from the four ovarioles follows a regular sequence.69, 315
Many aspects of tsetse fly behaviour have been investigated,46, 47, 98, 352, 376, 406 several of which have been used to devise methods for their capture. When a full understanding of their behaviour has been attained, it may well lead to improvements in the present methods used for this purpose.
Life cycle and population dynamics
Female tsetse flies emerge from their puparia about two days before males191, 285 and, after expanding and hardening their wings, they seek a blood meal. In some species, females are receptive to males as soon as they start seeking food and often mate either when taking their first blood meal, or soon after. Females actively resist mating when they get older192 but polyandry is common in the field24 and can even reduce female survival in artificial rearing conditions through sexual harassment, if the male to female ratio is greater than 4 to 1.76 They are viviparous, ovulation taking place about four days after the first meal i.e. after eight to 10 days of imaginal life with the first larva being deposited about 10 days later. At deposition, the larva is at the end of its third instar, having been fed on a nutritious fluid during its development within the female. Each subsequent larva follows at an interval of about 10 days. The duration of larval development in the laboratory is temperature-dependent, and may be predicted by a formula.139 The female feeds at regular intervals throughout her life, and if not well nourished, her reproductive performance declines.135, 252 It has been shown in the field that high host density, resulting in very well-fed flies, may cause a reduction of the interlarval period by two days, relative to that predicted on the basis of mean ambient temperature.163 Females live longer than males in the field, and must live long enough to produce more than two mature larvae if the population is to remain static, and more than that to provide for mortality and population increase. The newly deposited larva moults to form the prepupa, but remains within the shed third instar cuticle, which then hardens to form the puparium within an hour of larviposition.424 Larvae are deposited in shady places with a soft substrate. Within the puparium, the prepupa moults to produce the pupa, and a final moult within the puparium produces the pharate adult, which emerges from the puparium in due course, the duration of the puparial stages being temperature dependent.
The insect does not feed from the time it leaves the female fly as a mature larva until the adult emerges from the pupa, a form of reproduction known as adenotrophic vivipary. At a constant temperature of 25 °C, male Glossina morsitans emerged after 30 days and females after 27 days, while the respective figures at a constant temperature of 16 °C were 101,8 and 100 days.284 Laboratory temperature can be monitored using electronic meteorological stations to study the relationship between temperature and life parameters, including mortality, and larval and pupal periods.277, 285 The life cycle of a tsetse fly is shown diagrammatically in Figure 5.
Male tsetse flies emerge in equal numbers to females and, after expanding and hardening, seek a blood meal. Although the males may copulate soon after emergence, they are not fully fertile until they are a few days old. In the field, males mate more than once but spermatogenesis occurs during the pupal period only.327 Males feed at similar intervals to females, but have greater energy reserves for flight as they do not have to nourish larvae.52 In general, the mean life span of males in the field is about four weeks, while that of females is at least eight weeks,191 but these durations may vary from place to place, depending on prevailing environmental factors.
The population dynamics of tsetse flies have been investigated for many years since the development of the mark– release–recapture method by Jackson.187 This method depends on marking flies with dots of artist’s oil paint186 or fluorescent dyes15, 318 and then recapturing them. Capture methods range from catching flies from a mobile or stationary bait with a hand net to the use of various types of traps, many patterns of which148, 160, 370, 374 have been developed since that used by Harris.165 However, no known capture method gives an unbiased sample with respect to population density, sex ratio, hunger stage, or age.228, 288, 316, 369
In view of the sampling biases, estimates of population density of tsetse flies vary widely. In addition, the assumptions underlying population estimates are often violated.391 Consequently, the population ecology of tsetse flies has been reviewed,308, 309 and a new analytical approach to population ecology has been developed.313
The rate at which tsetse fly populations grow is not known, although a mathematical modelling approach has been developed that indicates the limits within which the population growth rate must lie under given environmental conditions, based on current knowledge of tsetse fly biology.161 It has been shown that no population of tsetse flies can survive if the daily mortality of females is greater than 4 per cent, and in the field possibly 2 to 3 per cent.161 This is a very useful criterion for those concerned with tsetse fly eradication.
Ecology of tsetse flies
Tsetse flies are distributed over 10 million km2 in Africa between 14 °N and 29 °S. The northern limits of latitude are highest on the west coast, while the southern limits are greatest on the east coast, where the 17 °C effective temperature isotherm339 gives a fair representation of the historical limits of the tsetse fly. These limits are determined by climate, often through its effect on vegetation but also fragmentation of the habitat due to human influence.28, 33, 154 Although the tsetse fly and its puparium are well waterproofed,53, 54 regions situated below the 500mm isohyet do not support tsetse flies unless there are watercourses with some vegetation along them.
Temperature is of great importance in the ecology of the tsetse fly. Not only does it affect interlarval and puparial durations, but it also influences the fly’s activity. Below 15 °C tsetse flies are inactive and above 35 °C they seek out refuge sites, such as rot-holes in trees, animal burrows and deep fissures in bark, in which they are inactive. The eyes are important in this refuge-seeking behavior.177 Thus the temperatures must stay within the 16 to 35 °C range for long enough during the day for tsetse flies to remain active and able to seek food. They are inactive at night. For the smaller species of tsetse flies, the puparial duration at low temperatures is so long that the insects’ fat reserves become depleted prior to maturation; this is one of the ways in which tsetse fly distribution is limited.56 The larger fly species are not subject to this limitation to the same extent. Temperatures over 40 °C are rapidly lethal to both large and small species of tsetse fly184 and their puparia.283 Increasing altitude moderates temperature, and this is reflected in the distribution of tsetse flies. Female Glossina pallidipes were trapped up to an altitude of 1992 m in the southern Rift Valley of Ethiopia,397 but in Zimbabwe, which lies farther south, they do not occur at altitudes above 1 200 m.181 These limits are linked to the mean annual temperature and can of course vary with climatic changes.
The vegetation type at a given locality is greatly influenced by temperature and humidity. Apart from grasslands, which do not support tsetse flies, all forms of woodland, from savannah to rainforest, can provide a suitable habitat for some species of these flies, but no one vegetation type is suitable for all species. Artificial plantations can provide a suitable habitat for tsetse flies,365 and the thickets (sometimes comprising chiefly Lantana camara) which develop on abandoned agricultural land are often good habitats for them.
Host preferences and location
The distribution and abundance of some tsetse fly species, especially Glossina morsitans and Glossina pallidipes, which are often referred to as the game tsetse flies, are closely related to the numbers and habits of certain wild animals. The development of methods for determining the animals on which individual tsetse flies have fed, by analysis of their blood meals, has given an insight into their feeding preferences.75, 266, 303, 337, 410 Glossina morsitans and Glossina pallidipes showed some preference for the warthog (Phacochoerus aethiopicus) and bushpig (Potamochoerus porcus), as well as some Bovidae such as the kudu (Tragelaphus strepsiceros) and bushbuck (Tragelaphus scriptus). Other wild animals, including elephant (Loxodonta africana), black rhinoceros (Diceros bicornis) and African buffalo (Syncerus caffer), are also fed on, but less frequently. In the case of the elephant and the African buffalo, this may be due, in part, to their tendency to move over large distances. Primates are poor hosts for the morsitans group, and some animals such as impala (Aepyceros melampus) and zebra (Equus burchellii) are rarely fed on although they may be abundant in tsetse-infested country. In the case of zebra, tsetse selective pressure is even considered as one of the main hypothesis to explain their stripes.63 The blood of impala is adequate to nourish tsetse flies,217 and the reason for flies not feeding on this animal may lie in the behaviour of these animals.245 Behavioural reduction in the attack by tsetse flies has also been shown for juvenile warthog.355 It has been shown that, although the blood proteins of a range of wild animals differs, tsetse flies can effectively use all the different types of blood.258 In the absence of its preferred hosts, the tsetse fly can survive on the blood of other wild animal species.
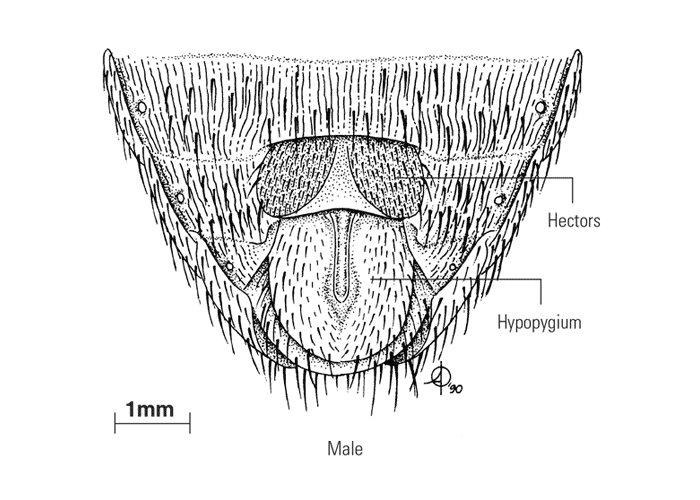
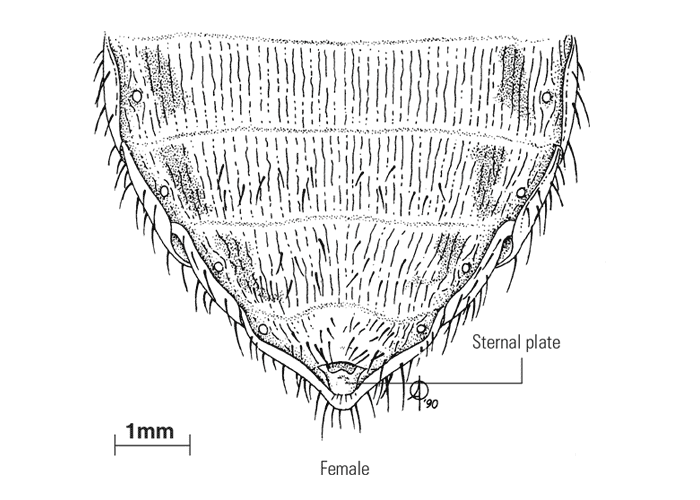
Figure 4 Ventral aspect of the terminal portion of the abdomen of both sexes of Glossina morsitans
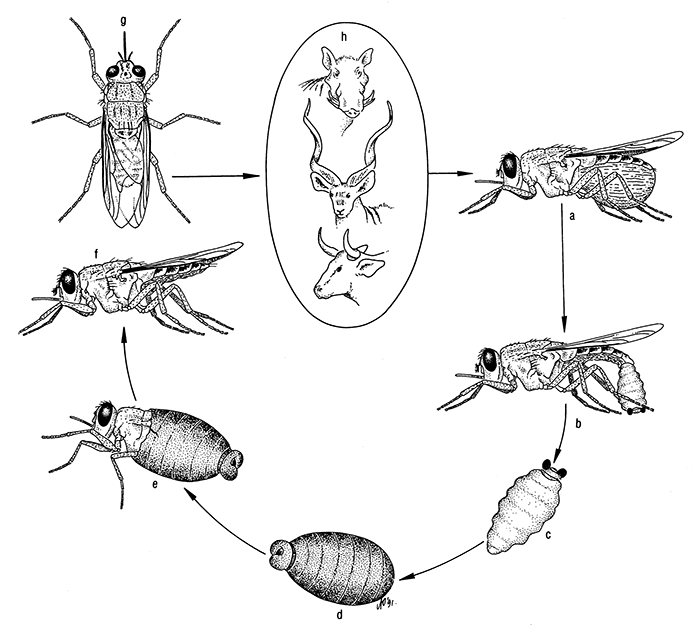
Figure 5 The life cycle of a tsetse fly
a = blood-engorged female
b = larvipositing female
c = third instar larva
d = puparium containing the pupa
e = fly emerging from the puparium
f = lateral aspect of a tsetse fly
g = dorsal aspect of a tsetse fly
h = some hosts of the morsitans group of tsetse flies
Domestic animals, particularly cattle41, 291 and donkeys,43 can be good hosts for tsetse flies, but sheep and goats are not. The analysis of tsetse fly blood meals from the northern region of the Nyanga district of Zimbabwe provides convincing evidence of Glossina morsitans’ ability to survive almost entirely on domestic cattle.303 It is also known from West Africa that Glossina palpalis, which feeds on a wide range of hosts, can adapt well to feeding on peri-domestic animals (animals such as dogs and pigs which occur around the houses of people) and humans.194 The palpalis group is very opportunistic in its choice of hosts and may even feed extensively on reptiles. They adapt to the availability of host very easily, even if pigs and humans are their preferred hosts. Glossina palpalis gambiensis has been shown to learn its trophic preferences, thus adapting to the most frequent hosts available during its lifespan.35 This is one of the main reasons why this group is more resilient to anthropic changes of ecosystems than the morsitans and the fusca groups.
In the laboratory, both in vivo feeding techniques using rabbits, guinea pigs, and goats as hosts or in vitro feeding methods where defibrinated blood is offered under a silicone membrane, are used to maintain colonies of tsetse flies.17, 254
Both vision and odour detection are used by the tsetse fly to locate its host.60, 81 In the case of vision, shape, movement, colour, and light and shade differences are important aspects.149, 166, 371 The contrast between light and shade may be important in determining alighting points on the host.45 As the range at which a moving ox can be seen by a tsetse fly is about 140 m,20 it seems that odours are responsible for attracting flies from greater distances.375 In riparian forests, the density of the vegetation is much more variable, and hence the range of attraction can vary from 10m to more than 300m.32
Studies have been made on the behaviour of tsetse flies in an odour plume,59, 136, 153, but full details of how the tsetse fly navigates in the plume have not been determined. At low wind speeds an odour plume meanders and, even 10m from the source, may give an incorrect indication of the direct line to the source of the odour.44, 152 Visual stimuli then become predominant when the fly gets close to its host.
At intermediate wind speeds the odour plume straightens and gives a better indication of the direction of the odour source, but at very high wind speeds the odour plume disintegrates rapidly and is not of use in indicating the source of the odour. Williams417 has modelled different tactics which may be used by the fly to locate an odour source, and it is likely that the fly is able to use a variety of methods, depending on the prevailing conditions. Glossina pallidipes has been reported to cross a clearing 923 m wide, where there was a conspicuous bush-covered cliff on the far side.344 Possibly senses other than vision are involved in this type of movement.
Vectors of trypanosomes
Tsetse flies are important mainly because they are vectors of certain species of protozoan parasites belonging to the genus Trypanosoma. This genus is split into four subgenera. Of these, one, Pycnomonas, contains only one species, T. suis, which is of minor importance in causing disease in Africa. The subgenera Duttonella, Nannomonas and Trypanozoon contain the species that cause most cases of AAT in livestock, while all the organisms causing HAT in humans belong to the subgenus Trypanozoon. The individual subgenera can be recognized on morphological grounds, but the species within the subgenus Trypanozoon are morphologically identical and their separation is based on epidemiological or molecular criteria. Trypanosomes belonging to the different subgenera tend to develop in different parts of the tsetse fly so, when the fly is dissected, one can often recognize the subgenus involved by its location (see African animal trypanosomoses, Figure 12.2). The development of DNA probes for the identification of trypanosomes in tsetse flies137 enables rapid and accurate identification of trypanosome species within the fly.328, 330, 333 Trypanosomes of major importance in Africa, and their usual development sites in the tsetse fly, are given in Table 1.
Transmission of the parasites after they have undergone a developmental cycle to produce the metacyclic infective stage, is known as cyclical transmission. Trypanosomes can also be transmitted mechanically by syringe passage and by contamination of the mouthparts of certain other species of biting flies including Stomoxinae and Tabanidae.412 Tabanidae can transmit T. vivax at high rates under field conditions and to a lesser degree even T. congolense.100, 101, 103
Table 1 Trypanosomes of major importance in Africa and their usual development sites in the tsetse fly
| SUBGENUS | SPECIES | SITE OF DEVELOPMENT |
| Duttonella | T. vivax | Haustellum |
| Nannomonas | T. congolense | Haustellum and midgut |
| T. simiae | Haustellum and midgut | |
| Trypanozoon | T. brucei brucei | Midgut and salivary glands |
| T. brucei gambiense | Midgut and salivary glands | |
| T. brucei rhodesiense | Midgut and salivary glands |
As trypanosomes survive for only a short time outside the mammalian or tsetse fly host, a fly with contaminated mouthparts has to feed on a susceptible host soon after the infective meal in order to transmit the parasite mechanically. Trypanosoma evansi, in North Africa, is transmitted entirely mechanically, like T. vivax in South America.102 In southern Africa, mechanical transmission of the trypanosomes normally transmitted by tsetse flies has not been unequivocally proven in the field.42, 412 All species of tsetse flies can transmit trypanosomes, although individual species may not have the same vectorial capacity for all the species of trypanosomes. The local removal of some populations of in particular the morsitans group, due to climatic or human influences, may thus favour species like T. vivax to the detriment of T. congolense.387 The infection level of the tsetse fly population varies from place to place, but is generally between 5 and 15 per cent. One of the main factors affecting infection rates in tsetse flies is temperature, with higher temperatures stressing the fly and making it more susceptible to trypanosome infection. Hunger due to starvation can also have the same effect.37
The conditions under which tsetse flies become infected when feeding have been reported.193, 260 Within the tsetse fly population some individuals are refractory to infection.251 This resistance is maternally inherited and is associated with the presence of rickettsia-like organisms in the midgut,228, 248 particularly in newly emerged flies.250 Lectins are involved in protecting the tsetse fly against infection by trypanosomes and these rickettsia-like organisms affect the level of the lectins.249 Sodalis glossinidius has been shown to be associated with infection of trypanosomes in tsetse flies in sleeping sickness foci122 and might represent a good target for transgenesis approaches aimed at blocking transmission.4
It has been suggested that the presence of trypanosomes in the haustellum of the tsetse fly interferes with the sensilla monitoring the rate of food intake and leads to infected flies probing with their mouthparts for a meal more often than uninfected flies.259 This would be an important factor in the frequency of disease transmission, but no increase in the probing activity in infected Glossina morsitans centralis has as yet been found in the field.255
Many wild animal species, particularly bushbuck, kudu, warthog and bushpig, are subclinically infected with the trypanosomes that affect humans and livestock and are important reservoirs of infection.194 Domestic animals such as cattle and pigs may be reservoir hosts of trypanosomes that affect humans.409, 415
Identification of Glossina
Tsetse flies were formerly treated as members of the family Muscidae, or a subfamily of it. They are now generally placed in a separate family Glossinidae, which has only one genus, Glossina and is part of the superfamily Hippoboscidea.
Flies in the genus Glossina can be recognized by the following features: they are long, robust, brown-black to brown coloured flies,327 and are between 6 and 16 mm long. Males are generally smaller than females; the proboscis points forward when at rest (Figure 6), and the antennae have feathered bristles on the arista. The wings, hyaline or slightly smoked, are at rest, crossed above the abdomen, their posterior end exceeding its extremity (Figure 7), a feature which separates them from other common blood-sucking Diptera such as horse flies (Tabanus subspecies) and stable flies (Stomoxys subspecies).
There are three groups of species within the genus Glossina, which may be distinguished largely by their habitat preferences. However, features of their genitalia are the definitive criteria. These groups have been given subgeneric names, but these are rarely used. Species and synonyms within the subgenera have been listed by Pont.294
The main criteria to identify species are the genital parts, the antenna and the tarsi. A key of determination allowing species identification is available in.229 (http://www.fao.org/docrep/011/i0535e/i0535e00.htm).
Distribution of tsetse flies in relation to various ecosystems
The fusca group (subgenus Austenina) inhabits dense forest belts (e.g. rain forest, evergreen rain forest) or dense riparian forest. Most species are usually found in wildlife areas with limited livestock development activities, and therefore their importance as vectors for AAT is limited. However, Glossina brevipalpis is an efficient vector of trypanosomes in southern Mozambique and KwaZulu-Natal in South-Africa.97 This group is very sensitive to changes in the vegetation caused by humans and the presence of most species is associated with the availability of specific wild host species, with the exception of Glossina brevipalpis.
The morsitans group (subgenus Glossina) is restricted mainly to savannah woodlands but is also found along water courses, in denser vegetation thickets or in forests. In the wet season they spread throughout the woodland, but in the hot, dry season they are often associated with vegetation along drainage lines. Their distribution is also strongly linked to the presence of wild fauna or livestock. This group is an important vector of AAT but they also transmit T. brucei rhodesiense between wild fauna, livestock and humans.
The palpalis group (subgenus Nemorhina) inhabits rainforests and vegetation bordering watercourses (e.g. riverine forest, protected forests, vegetation along lakes, mangroves), but they can disperse far out into savannah woodland, especially during the rainy season. Some species colonize tree plantations, like Glossina palpalis gambiensis in Senegal.38 The availability of specific hosts is generally not limiting for this group as they are very opportunistic feeders. Overall, the species of this group are the most resilient to anthropization and are important vectors of T. brucei gambiense. With the progressive regression of the morsitans group due to human encroachment, this group is gaining in importance as a vector of AAT.
The main factors affecting tsetse distribution are the suitability of vegetation and the availability of hosts. It is possible to model their distribution using satellite images and taking into account local climatic conditions, vegetation type and level of degradation. Suitable vegetation buffers macroclimatic variations, particularly for the fusca and palpalis groups. In eastern Africa, the tsetse belt largely follows north -south eco-climatic zones, with the fusca group being mostly limited to the Guinean area in the south whereas the morsitans and palpalis groups extend into the Sudanese area up to the edges of the Sahelian area in the north. In east and southern Africa, their distribution is patchier and mainly affected by altitude. Mapping suitable tsetse habitats is crucial to inform monitoring and control operations.31 Maps of tsetse distribution are available on the PAAT-IS website (http://www.fao.org/ag/againfo/programmes/en/paat/maps.html) but were developed using mostly outdated entomological data and their resolution is not suitable to guide control operations. They can, however, be used for prioritization of control operations on a regional scale. There is undoubtedly a great need to improve the knowledge of present tsetse distributions, which have changed drastically in the last decades due to local and global environmental changes.387
Recently, MaxEnt models have been developed to predict suitable tsetse habitats in western33, 110 and southern71, 121 Africa. These models are mostly based on MODIS (Moderate Resolution Imaging Spectroradiometer) data that have a spatial resolution of 250m and a temporal resolution of two images per day. In the model, the MODIS data are complemented with data of Landsat or Spot that have a high spatial resolution of 30m and 10m, respectively. MaxEnt models predict species distribution by contrasting the environmental conditions where the species is present to the global environment characterized by some generated pseudo-absence data,116 preferably in areas where the species has not been observed. The accuracy of the entomological data used to fit the model is critical and must account for trap efficiency. The output of the model is a quantitative indicator of habitat suitability for the species of interest in a given area. It is then possible to select a threshold of suitability to discriminate between suitable and unsuitable habitat, considering the desired level of sensitivity and specificity. These habitat types can then be used to maximize the cost-efficiency of entomological monitoring using traps, but also for control purpose, e.g. to calculate the density of sterile males to be released in function of the density of suitable habitat or to deploy insecticide-impregnated targets in the most suitable sites. In the tsetse project in Senegal, for example, the MaxEnt model provided accurate information on suitable habitat areas, which covered only 4 per cent of the total target area.109
Management of tsetse populations
The impact of past tsetse eradication campaigns is given in Table 2. The risk of false negative concerns only the cases when eradication was reported but flies were still present as the population had dropped below detectable level with the traps used; it was documented qualitatively considering the data reported by the authors. The risk of false positive concerns only the cases when eradication was achieved but not reported as rapid reinvasion occurred: it is based on the probability of not detecting surviving flies (Barclay and Hargrove, 2005). This probability is provided in brackets in the corresponding column. When eradication was not reported, the observed reduction rates are provided in brackets. Source: 37
Table 2 Impact of past tsetse eradication campaigns
| METHOD | COUNTRY | AREA | DURATION | TARGET SPECIES | ERADICATION OBTAINED | FALSE NEGATIVE | FALSE POSITIVE | REFERENCE |
| ITT 11/km river | Ivory Coast | 79 km river | 2 months | G. palpalis gamiensis G. tachinoides | No (98%) No (99.8%) | No (barrier) | 224 | |
| ITT 4/km2 | Zambia | 3 000 km2 | 4 years | G. morsitans centralis | Yes | Noa | 14 | |
| ITT 3-5/km2 | Zimbabwe | 600 km2 | 4 years | G. morsitans morsitans G. pallidipes | No (99.99%) | No (central area) | 384 | |
| ITT 7/km2 | Uganda | 32 km2 | 4 months | G. fuscipes fuscipes | No (97.3%) | No (barrier) | 274 | |
| ITT 5/km2 | Ethiopia | 150 km2 | 1 year | G. fuscipes fuscipes G. pallidipes | No (74%) No (92%) | No (barrier) | 230 | |
| ITT 4/km river | Nigeria | 1 500 km2 | >14 months | G. tachinoides | No (>90%) | No (barrier) | 345 | |
| ITT 10-20/km2 | Ethiopia | 200 km2 | 3 years | G. fuscipes fuscipes G. pallidipes G. morsitans submorsitans | No (no reduction) No (84%) No (83%) | No (insufficient reduction) | 227 | |
| SAT 5 cycles | Botswana | 16 000 km2 | 2 years | G. morsitans centralis | Yes | No (p<0.001) | 204 | |
| SAT 4 cycles | Ghana | 6 745 km2 | 1 month | G. tachinoides G. palpalis gambiensis | No (98%) | No (central area) | 3 | |
| SAT 9 cycles | Kenya | 300 km2 | 5 months | G. pallidipes | No (90-99.9%)c | No (central area) | 363 | |
| ITT 60/km2 + Netting of pig pens 20/km2 + ground spraying | Guinea | <20 km2 | 17 months | G. palpalis gambiensis | No (100%)4 | No (isolated island) | 196 | |
| ITC 6-18/km2 + ITT 4/km2 | Burkina Faso | 400 km2 | 3 years | G. tachninoides G. morsitans submorsitans | No (91.8%) No (98.4%) | Yes (no barrier) | 19 | |
| ITC 9-28/km2 + ITT 45-70/km2 | Unguja | 1 650 km2 | 5 years + 18 months | G. austeni | No (80% fem, 98% males) | No (Island) | 171, 405 | |
| ITC 2.5/km2 + ITT 1/km2 | Ghana | 18 000 km2 | 1 year | G. tachninoides G. palpalis gambiensis | No (≈96%) | No (barriers) | 3 | |
| ITC 9-28/km2 + ITT 45-70/km2 | Unguja | 1 650 km2 | (6.5) + 3.5 years | G. austeni | Yes | Nod | 399 | |
| ITT 2.4/km2 + SIT | Burkina Faso | 3 000 km2 | 3 years | G. tachinoides G. palpalis gambiensis | Yes | Yes (p=0.43)e Yes (p=0.56)e | 89, 293 | |
| ITT 4/km river + SIT | Nigeria | 1 500 km2 | +3 years | G. tachinoides | Yes | No (p<0.001)f | 345 |
a monitoring using stationary traps, screen fly rounds, electric net fitted on a motorcycle during one year in one block, two years in the second;
b 22 per cent of the adult females dissected after each cycle were survivors so that the number of cycles was limited to 4;
c over 99.9 per cent in main habitats and about 90 per cent in conifer plantation;
d no fly was captured in July 2010 (low efficiency of the traps used) but then tsetse were captured with sticky traps in 2011-2012;
d 399 leg-panel traps deployed in 55 fixed monitoring sites during 4 years ;
e considering the number of trapping events with 41 biconical traps set every month during 48h until July 1985 (trap efficiency of 1.2 per cent (±1.3 per cent) and 0.9 per cent(± 0.9 per cent) per km² per day for G. tachinoides and G. p. gambiensis, respectively) ;
f 384 monitoring traps were deployed permanently and collected every 24 to 48h.
ITT: insecticide-impregnated traps/targets
ITC: insecticide-impregnated cattle
SAT: sequential aerosol technique
SIT: sterile insect technique
Two critical concepts: Area-Wide Integrated Pest Management and the Phased Conditional Approach
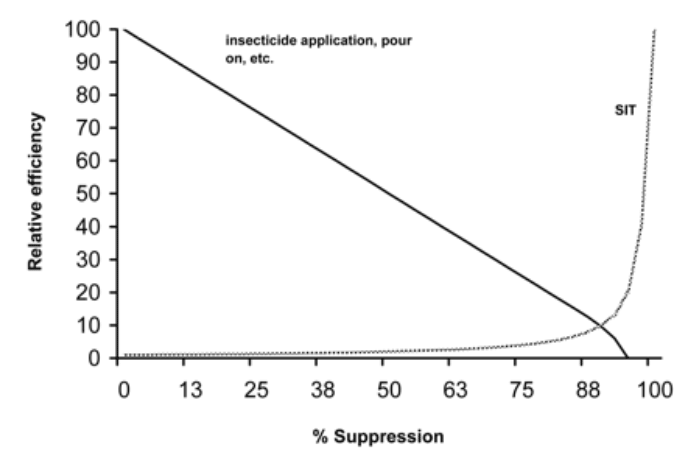
There are basically two broad strategies to manage tsetse populations,396 i.e. in a first approach, the populations are suppressed on a field-by-field (localised) basis, whereas in the second approach, the entire population is targeted on an area-wide basis (area-wide integrated pest management (AW-IPM)). Both strategies can be successful, but they have completely different goals (suppression versus eradication) and consequently will have different outcomes and associated costs.396
The localized, field by field approach has merit in ecological settings where the target tsetse population is not isolated and surrounded by other tsetse populations. The aim is to locally suppress (or reduce) the tsetse population below the transmission threshold to temporarily alleviate the burden of the disease in the area where the livestock is being kept. This strategy can only be sustained by the beneficiaries when the cost of the effort is low. In this “reactive approach” the farmer makes the decisions independently of the actions or intentions of his neighbours, and often after the pest population has reached damaging proportions. The aim of this bottom-up approach is to protect individual animals or herds and has the advantages that it (1) is independent of government funding, (2) does not require community coordination or a special organization that is responsible for programme implementation, (3) uses often ‘low tech’ control tactics, and (4) requires minimal planning. The biggest limitation of the approach is its vulnerability to reinvasion of the pest insects from untreated, neighbouring areas and hence, it proves difficult to sustain the results.205, 396 Moreover, any positive impact of vector control on disease prevalence or transmission tends to reduce the farmers’ involvement. Community efforts based on this approach are difficult to sustain as the farmers protect individual goods (their animals) instead of securing the entire production system (collective protection).26
The area-wide approach aims at the sustainable removal of an entire tsetse fly population within a delimited geographical area205, 207 used simple mathematical models to show the importance of targeting an entire pest population and emphasised the negative consequences of leaving small relic populations from which individuals can be recruited into the cleared areas.207 elaborated the basic concept of area-wide (or total population) pest management as:
“Uniform suppressive pressure applied against the total population of the pest over a period of generations will achieve greater suppression than a higher level of control on most, but not all of the population, each generation.”
AW-IPM is an approach that consists of a coordinated effort against all members of the pest population in an ecosystem before the pest population has reached damaging proportions. Therefore, the emphasis is on preventive pest management aimed at the protection of agriculture. The biggest advantage of the approach is that it minimizes the risk of reinvasion as also areas that are of no interest to the farmer are targeted. This top-down strategy usually requires, in view of its complexity, several years of planning and a specialised organisation with dedicated staff to implement the control activities. The approach can benefit from advanced technologies such as geographic information systems (GIS), population genetics, remote sensing and aerial release techniques.109, 205, 396 The limitations of this approach are that it is technology intensive and requires important and sustainable funding to reach its objectives. In the past, most control programmes against tsetse flies did not target an entire tsetse population and were therefore mostly not sustainable.402 In addition, very often the control was exercised using one control tactic rather than an integration of control tactics suited for the ecological situation. Ideally, an AW-IPM approach should initially deploy control tactics that are effective at high pest population densities (e.g. the aerial spraying of insecticides, traps, targets) followed by control tactics that are effective at low population densities (e.g. the sterile insect technique) (Figure 8).
Tsetse control efforts have often been plagued by political or stakeholders’ pressure to quickly start killing the insect, without detailed knowledge of the biology or dynamics of the targeted pest population. To avoid embarking on an operational programme without an appropriate strategy, a phased conditional approach (PCA) should be followed. The PCA is a strategy whereby programme implementation follows distinct phases and in which support to the next phase is conditional upon completion of all (or at least the majority of) activities in the previous phase.123 Whereas the diverse phases of the PCA might differ with the target pest species, or if a suppression rather than an eradication strategy is selected,170 the PCA for the tsetse programme in Senegal consisted of 4 phases, i.e. (1) training and commitment of all stakeholders, (2) baseline data collection and feasibility studies, (3) pre-operational activities, and (4) operational activities.402
A generalization of this approach was recently proposed for all situations of AAT control.104 Even when suppression is the selected strategy, it should still be implemented following AW-IPM principles to be sustainable. For example, it will often be more efficient to strengthen veterinary services and secure appropriate drugs and insecticides than to deliver insecticide-impregnated targets to a beneficiary community: whereas tsetse suppression is generally perceived as easy, sustaining the results will probably require empowerment of the beneficiaries rather than technology transfer.
Population genetics as a decision making tool to develop control strategies
Population genetics can be very useful to study the population structure of target populations, particularly their level of isolation with respect to neighbouring populations, which then could inform decisions on which strategy to adopt i.e. suppression versus eradication.329, 331, 332
A new approach combining population genetics, remote sensing and geographic information systems (GIS) was recently proposed to identify natural barriers to tsetse dispersal.33 This approach is based on the concept of landscape resistance to movement (i.e. friction) modelled by predicting tsetse gene flow as influenced by environmental variables such as temperature, human density, etc. Isolated tsetse populations were identified in West Africa (from Burkina Faso in the east to Senegal in the west), by combining friction with the predicted distribution of Glossina pallidipes gambiensis, one of the most important vectors of AAT in this area. Identified tsetse populations that are potentially isolated might then be targeted for an eradication strategy, whilst a suppression strategy might be more appropriate for non-isolated populations. A feasibility study to confirm the status of population isolation will still be necessary before the start of any control effort.
Friction models may also be used to select the most favourable areas to erect artificial barriers that prevent tsetse dispersal, and develop sequential eradication programmes by dividing the target population into partially isolated subunits that would be addressed following a rolling carpet approach.170
Past management tactics that are out of use on environmental grounds
Environmental modification
Human settlements and their effects on tsetse fly populations: In the past there have been many fluctuations in human populations in Africa which were attributed to disease epidemics, tribal conflicts or the colonization of Africa.129 At the time of writing (2017), the continent of Africa is home to 1.2 billion people, up from just 477 million in 1980. Africa is projected by the United Nations Population Division to see a slight acceleration of annual population growth in the immediate future. In 2015, the population of the African continent grew by 30 million. According to the UN, annual increases will exceed 42 million people per year by 2050 or an additional 3.5 million people per month; the total population will have doubled to 2.4 billion. Since the middle of last century, improvements in public health have led to a significant decrease in infant and child mortality, and overall life expectancy has also risen from 37 years in 1955 to 60 in 2016 (https://www.theguardian.com/global-development-professionals-network/2016/jan/11/population-growth-in-africa-grasping-the-scale-of-the-challenge).
The effect of humans on the different subgenera of tsetse flies differs greatly, due to the differences in habitat requirements of the flies. The destruction of the forests of Africa, partly for commercial timber and partly for settlements and agricultural land, is proceeding rapidly, and this process removes the habitat of the fusca group of flies. It is no longer possible to leave cleared land fallow after it has become too depleted in nutrients to support crops, so clearings become permanent. The human population also destroys the food supply of the tsetse flies through hunting, so they disappear. The flies of the fusca groups are likely to survive only in forest reserves in future. In southern Africa, Glossina brevipalpis, which is the most widely distributed member of the fusca groups, is confined to riverine forests and to the few humid forests that occur in the region. It has been assumed that its limited distribution makes it a relatively unimportant species with respect to AAT in KwaZulu-Natal, South Africa but recent data indicated that both Glossina austeni and Glossina brevipalpis play significant roles in trypanosome transmission and it will be essential that any control strategy that aims at the sustainable management of the disease should target both species.97
The influence of humans on the palpalis group of flies is not always adverse, and they can often exist in close contact with people and their domestic stock. Their habitat is riparian vegetation. Agricultural activities tend to be in areas further from the streams in high rainfall areas, and the tree plantations grown there may themselves extend the habitat of the fly. Patches of natural vegetation protected by humans for cultural reasons also occur in many parts of Africa and may provide suitable habitat for palpalis group flies. Members of the subspecies of G. fuscipes can be closely associated with humans and, in western Kenya, they have been able to live in extensive thickets of Lantana camara, which have developed as a result of poor land management.416
It is on the morsitans group of flies that humans have made their greatest impact. Glossina morsitans subspecies, Glossina pallidipes and Glossina austeni are widespread in the characteristic undisturbed savannah and wooded savannah vegetation types, often where wild animals are plentiful. The agricultural activities of humans, and their use of fire, have played an important part in the development of savannah vegetation, so in this sense, humans have been of benefit to the tsetse fly.
But humans often enter tsetse fly-infested savannah country to undertake agricultural activities. If cultivation in these areas is continued on a long-term basis, the vegetation becomes either cropland or grassland, and at the same time the wild animals are killed or move away and the flies ultimately die out. In the past, land was abandoned when its fertility had declined. Such land would, in the course of time, revert to savannah vegetation and the wild animals would return, making the area suitable again for morsitans group flies. Thus the situation was one of long -term rises and falls of fly populations, depending on the activity of human populations. This shifting pattern of agriculture is becoming less frequent in Africa, and more permanent occupation of the land by humans leads to permanent removal of the morsitans group of flies. The density of human settlement needed to achieve this removal of tsetse flies is not known exactly. In West African savannah, densities of 15 to 39 people per km2 reduce morsitans group flies to very low levels, and at human population levels higher than this no tsetse flies exist.269 Some parts of southern Africa have always had a high human population density.342 An account of the waxing and waning of the Glossina population in the Mossurise region of Mozambique, consequent to changes in human population densities in this heavily settled area between 1861 and 1889, was provided by Swynnerton.342
Civil wars have had an impact on the distributon of tsetse flies, such as the one in Zimbabwe in the 1970s. The war prevented control of tsetse flies along the north-east border of the country and over a period of five years the fly (in this case Glossina morsitans) invaded the country rapidly, reached its historical limits, and even progressed slightly further in some places. However, much of the country that was reinvaded was marginal for tsetse flies and their numbers remained low, but the Umfurudzi Safari Area, lying in broken but well-watered country and with a large wild animal population, provided a good habitat and flies built up to large numbers there. This invasion gave rise to cases of AAT in cattle near Shamva, an area that had been free of tsetse flies since the rinderpest pandemic in the 1890s.
In the Sebungwe and Umniati districts of Zimbabwe, outbreaks of HAT, in 1912 and 1934 respectively, caused the authorities of the day to eliminate the problem by moving the human population involved.22 In the long term, this sort of action favours the vector since, in the absence of any fly control measures, the abandoned agricultural lands revert to suitable tsetse fly habitat. Similar events have occurred in East Africa.129
Humans also affect the distribution of tsetse flies indirectly by creating wildlife sanctuaries. If these areas are in tsetse fly-infested country, they serve as a source of the flies to domestic stock grazing on adjacent land, as has occurred in KwaZulu-Natal, South Africa114 and is currently the case in Malawi.282 There is generally resistance to the use of any anti-tsetse fly measure in the sanctuaries. However, if large elephant populations are confined to the sanctuaries and are allowed to increase, they will modify the habitat.
These habitat changes may make the area unsuitable for tsetse flies, and for many other animal species, as has happened in some East African parks.130 Human settlement and agricultural activities disturb the natural vegetation and reduce wild host densities, and can therefore be considered as a sustainable control strategy against tsetse, although it is much more efficient against the morsitans than the palpalis group.387
Rinderpest: This disease entered Egypt from Arabia in the late 1880s, and a pandemic of it swept down Africa in a wide variety of animals. It had reached Lake Malawi by 1892 and crossed the Zambezi in 1895 to 1896 (see Rinderpest). This disease almost eliminated cattle in the infected area and severely depleted the numbers of many wild animal species in southern Africa. Not all wild animals are susceptible, but many of the susceptible animals include the preferred hosts of Glossina morsitans and Glossina pallidipes in particular.303, 410 This was not, of course, known at the time of the rinderpest pandemic, but after the disease had been eradicated from southern Africa, Stevenson-Hamilton338 pointed out that, as not all animal species were equally affected, the tsetse flies must have been associated with those that were most susceptible.
The effect of this disease on the Glossina morsitans and Glossina pallidipes populations was dramatic: (1) in Botswana, tsetse retracted to four small foci, (2) in Zambia, the Kafue tsetse fly belt shrank considerably and tsetse flies disappeared completely from the Western Province,423 (3) in Zimbabwe, Glossina morsitans disappeared from much of its range,181 and disappeared completely in South Africa,132 (4) in Mozambique, Glossina morsitans and Glossina pallidipes106 disappeared from the southern part of their range.132 Although Glossina morsitans numbers did not decline in parts of North Mossurise in Mozambique in spite of heavy mortality from rinderpest among wild animals,342 there can be little doubt that the cause of the tsetse population decline was the removal of its food supply. Flies may have survived in some localities because some herds of susceptible animals remained uninfected, or because of the presence of non-susceptible animals such as rhinoceros and hippopotamus (often very localized in their distribution), which can provide good sources of blood for tsetse flies.
Rhinoceros blood has been found in Glossina morsitans and Glossina pallidipes in Zimbabwe at some localities,303 and Glossina brevipalpis is known to feed readily on hippopotami.411 The exclusion of rhinoceros from the animal host list during the campaign to eliminate tsetse flies by game reduction in the 1930s may have played a considerable part in maintaining the tsetse flies114 and suggests a possible mechanism by which flies were able to survive the rinderpest pandemic in KwaZulu-Natal, South Africa.
The world was officially declared free from rinderpest in 2011 in the course of the 79th OIE General Session. Rinderpest, once the scourge of societies across Asia, Europe and Africa, is only the second infectious disease, after smallpox for humans, to have been eradicated globally thanks to decades of internationally concerted effort (http://www.oie.int/for-the-media/rinderpest/).
Animal reduction as a tsetse fly control measure: The pre-rinderpest retreat of Glossina morsitans in the Limpopo Province of South Africa132 and in north-western Zimbabwe74 was attributed to the activities of big game hunters and the advance of settlers from the south. The effects of rinderpest provided further evidence of the importance of wild animals to Glossina morsitans in particular. As the animal populations in southern Africa recovered after the rinderpest pandemic, so did those of the tsetse fly, and they began to spread to their former limits. The flies soon came into contact with humans and their domestic stock, and the governments of the various countries had to undertake measures to reduce the disease problems associated with tsetse flies. The most obvious way to attack the fly at that time was to remove its source of food by reducing the number of wild animals.
In Zimbabwe, the numbers of wild animals were reduced by hunting between parallel fences 16 km apart.180 A broad spectrum of species was shot, and the method was successful in reclaiming large areas of what became some of Zimbabwe’s most productive farm land. The large-scale destruction of wildlife is abhorrent,183 but at that time it was the only practicable method of controlling tsetse flies, which threatened disaster to the farming community in Zimbabwe. Broad spectrum game reduction campaigns against tsetse fly, either between fences or not, were undertaken in Zambia,120, 422 Botswana95 and KwaZulu-Natal, South Africa,114 but the success achieved was generally not as good as that in Zimbabwe. As alternative methods of tsetse control became available, game reduction campaigns were abandoned in most countries.
Following the demonstration of host preferences of tsetse flies,410 an experiment was conducted in Zimbabwe between 1962 and 1964 which showed that a spectacular decline in Glossina morsitans numbers followed the hunting out of warthog, bushpig, bushbuck and kudu.239 Elephant and buffalo were driven out of the fenced experimental area prior to the start of the experiment because of the damage they do to fences and also because they can be a good source of food when present in an area. No other animal species were hunted. As warthog provided about half the blood meals of Glossina morsitans at the experimental site, the effect of the removal of just warthog was tested.366 It was shown that the flies adjusted rapidly to other preferred hosts in this area, particularly kudu.
The results from these experiments led to the subsequent use in Zimbabwe of selective hunting of the four preferred species between parallel fences to prevent the advance of tsetse flies. The fence immediately adjacent to the fly area was more robust and large animals such as elephant and buffalo were kept away from it by limited hunting. This approach to animal reduction initially enabled the tsetse advances to be contained, but became ineffective during the civil war in the 1970s. Whilst hunting operations were resumed soon after the cessation of hostilities, including those conducted for the protection of fences, these activities were short-lived and by the 1990s had been abandoned completely in favour of other techniques.
Habitat destruction and vegetation modification: Tsetse flies depend in hot weather on vegetation to provide shade in their refuge, resting sites and the sites in which larvae are deposited. Grassland is unsuitable habitat for tsetse flies, and the clearing of woodland to produce grassland could potentially create a barrier to tsetse flies. However, to be effective, the clearing has to be wide and the width is most likely species dependent. Barrier clearings were used in eastern Zimbabwe,183 in Zambia,422 KwaZulu-Natal,114 Botswana95 and Malawi, where clearing was combined with settlement.212 Although barrier clearings have achieved moderate success, they are expensive to create and must be maintained indefinitely, and hence they are not widely used at present. Attempts have been made to use herbicides to control regenerating vegetation,77, 78 but the chemicals are expensive and have deleterious effects on the environment.
Discriminative clearing was used between 1941 and 1942 to eliminate Glossina morsitans near Abercorn in north-eastern Zambia.140 Vegetation was removed only in the shallow valleys at the headwaters of small streams, which formed only a small proportion of the total tsetse fly-infested area. This campaign was one of the few successful attempts at discriminative clearing. In Zimbabwe, extensive discriminative clearing in the south-east of the country was done between 1956 and 1961 in an attempt to arrest advances by both Glossina morsitans and Glossina pallidipes into Zimbabwe from Mozambique.141 Much of this arid area is marginal country for tsetse flies,172 but in some places riverine fringe vegetation and patches of hillslope woodland provided reasonable habitat and abundant wildlife. The clearing certainly helped to limit the flies, but it did not eliminate them. The costs were high, as much of the work was done by bulldozers, and the destruction of riverine vegetation is likely to be permanent in this very harsh part of the country. This form of tsetse fly control has not been used recently, either in Zimbabwe or elsewhere in southern Africa. It can, however, be combined with the deployment of insecticide-impregnated targets to increase their range of attraction, as was recently done in Guinea against Glossina pallidipes gambiensis.196
Pesticides
Residual ground spraying
After the Second World War, the advent of synthetic organic insecticides, and particularly the long-acting organochlorines, led to new methods of tsetse control.
The flies’ hot, dry season habitats (i.e. their essential habitats) can be mapped using remote sensing data and species distribution models (see above). Treating the resting and refuge sites with suitable long-acting insecticides, applied from knapsack sprayers by teams moving through the bush, will ultimately kill the flies when they come into contact with this insecticide. Access tracks must sometimes be developed to apply the insecticide to these places. The insecticide is preferably applied in the cool, dry season, to cover as much territory as possible before the onset of the hot weather. Dieldrin and DDT were the most widely used insecticides for these ground spraying activities, but for environmental reasons, they were more recently replaced by synthetic pyrethroids. These chemicals have a residual life of at least four months in the dry season, and for these reasons, this tactic is rarely used these days.
Ground spraying was used between 1962 and 1974 in southern Africa to halt an advance of tsetse flies in south-eastern Zimbabwe and south-western Mozambique and a large area was reclaimed from the fly.234, 235, 236, 302, 304 Ground spraying was the major method used to eliminate tsetse flies in Zimbabwe from 1960 to 1986 and is still used in this country using pyrethroids as well as in Ethiopia, Burkina Faso and Guinea. It was also used extensively in Nigeria,240 Botswana,95 Zambia,279 the Zambezi (formerly Caprivi strip) region of Namibia and experimentally in Malawi.
Much concern has been expressed over tsetse fly control schemes based on ground spraying using residual insecticides that lead to environmental contamination, i.e. acute poisoning of birds and other wildlife,146 and accumulation of residues of DDT and its metabolites by non-target organisms in sprayed areas.246
A comprehensive four-year study, undertaken in a section of the Zambezi River Valley in Zimbabwe, indicated111 little or no effect of DDT on reptile populations, small effects on some ant species, and deleterious effects on populations of several species of insectivorous birds, i.e. acute DDT poisoning of the white-headed black chat (Thamnolaea arnoti), and marked thinning of the egg shell, a factor associated with chronic poisoning by DDE, a metabolite of DDT, of the African goshawk (Acipter tachiro) and the African fish eagle (Haliaeetus vocifer). However, no detectable effects of DDT were found on soil processes and on the fish species studied.
Deltamethrin used in ground spraying showed that populations of non-target invertebrate species were severely affected in both terrestrial and aquatic habitats. No major effects were seen on other animals, although the large decline in insect populations may have caused reductions of insectivorous species or their move to untreated areas.111
The sequential aerosol technique (SAT)
Aerial spraying of insecticides against tsetse flies was first used in KwaZulu-Natal, South Africa (1945-1948)114 using a coarse spray of atomized liquid droplets that formed toxic residual deposits. Similar approaches were used in West Africa, where helicopters applied the spray.12
The second approach involves the ultra-low volume spraying of insecticides 10-15 m above the tree canopy by fixed wing aircraft or helicopter (in more difficult terrain) in five to six subsequent spraying cycles, separated by 16-18 days depending on the temperature.114 In this form, normally long-acting insecticides such as DDT or hexachlorocyclohexane (formerly known as benzene hexachloride or BHC) have no residual action, and the environment is simply fumigated. The droplet size needs to be sufficiently small to remain suspended long enough in the air rather than sinking to the ground, and large enough to prevent floating upwards. The goal is to kill all adult flies in the first spraying cycle by direct contact and then kill all emerging flies in the subsequent cycles before they can start reproducing.163 A slight wind is an advantage, as the aerosol cloud then drifts through the habitat, but strong winds disperse this cloud and its application has to be stopped until favourable conditions return. Although the technique is delicate (the insecticides have to be applied during periods of temperature inversion i.e. night time, and do not tolerate any delays in the timing of the cycles, it remains a cost-effective and efficient tactic when using GPS-guided navigation and spray systems for effective area-wide tsetse suppression in dense humid forest ecosystems or even eradication in open savannah-type ecosystems.5
The aerial application of insecticides initially developed by Du Toit114 has now become more sophisticated, but the underlying principles are the same.6 The development of rotary atomizers that produce a uniform aerosol droplet size and, in particular, the development of night flying techniques that permit spray aircraft to operate throughout the night, have made the sequential application of insecticide from the air a more practical and economical proposition. Endosulfan, deltamethrin and alphacypermethrin have been the most commonly used insecticides for this technique.423
Large-scale aerial spraying campaigns were conducted at irregular intervals in Zimbabwe between 1953 and 1988.70, 80, 175, 176 Aerial spraying conducted in Zambia has been largely successful.278 As in all insecticide work against tsetse flies, the cleared areas may be reinvaded if they are not developed and settled by humans or the operations are not following the rolling carpet approach.
In Botswana, following successful preliminary aerial spraying trials in 1972, parts of the Okavango Delta were sprayed annually from 1973 to 1991 (excluding 1988). In general, although not exclusively, the annual operations up to 1979 alternated between the western and south-eastern perimeters of the Delta. Thereafter, the operations were aimed at the main body of the infestation.423 Environmental concerns prompted the Government to change tactics and the SAT was replaced with odour-baited targets.384 This change in strategy coincided with the start of a 10-year long drier period, which greatly facilitated the deployment of the 25 000 targets in the area. In addition, the Ramsar Convention on Wetlands came into force for Botswana on 9 April 1997, making the Okavango delta ‘the largest wetland of international importance’, which reinforced the applied strategy. However, in 1999, the rains returned and the regular flooding of the area disrupted the target deployment programme. The tsetse fly reinvaded cleared areas, and cattle residing at the periphery of the Delta became infected again with AAT and by 2000, the tsetse-infested area had expanded to > 11 000 km2. As a result, the Botswana Government decided to revert to aerial spraying, and in 2001 and 2002, 7180 km2 and 8722 km2, respectively were treated with deltamethrin applied at a dose rate of 0.26 g/ha.7 Deltamethrin is an insecticide safe to the environment that does not accumulate in the food chain and kills tsetse after brief contact.231 The spraying was carried out by a South African crop-spraying company, which used four aircraft equipped with efficient GIS-based SATLOCK guidance systems. The total cost of the spraying campaign was estimated at less than USD 270/km2.
The five spraying cycles over a period of approximately eight weeks eliminated Glossina morsitans centralis from the Okavango The last phase of the programme included a monitoring and modellling effort that estimated that each spraying cycle needs to kill at least 98 per cent of adult females to reach eradication.204 Prior to the SAT operations, catches of Glossina mositans centralis using man fly-rounds amounted to 101 per round per day whereas between September 2002 and November 2005, surveys (∼820 daily flyrounds and ∼2050 trap-days) failed to detect any tsetse flies.
The 2001-2002 operations showed that 1) SAT is a rapid, efficient and cost- effective method for tsetse elimination in the open savannah areas of East and southern Africa, 2) SAT does not have any serious lasting negative environmental impact, and 3) it is cost- effective to solicit foreign expertise when it is not available locally.396
Although pesticide application rates are very low in the aerial spray method for tsetse fly control, many aquatic organisms are killed, especially in shallow water. The effects of aerial spraying on non-target organisms have been monitored on several occasions79, 112, 243 and it has been found that generally, if the insecticide is applied at the correct level, and only in one year, no permanent damage to the environment occurs. These conclusions have been supported by the Scientific Environmental Monitoring Group,317 which is part of the Regional Tsetse and Trypanosomiasis Control Programme. The same group also stated that ‘if areas without open water are treated with endosulfan (6 to 24 g/ha), and areas with rivers and lakes are treated with deltamethrin (< 0,3 g/ha), then according to our present knowledge the side-effects seem to be acceptable.268
The above was confirmed by a detailed environmental study undertaken by the UK Department for International Development (DFID) in collaboration with the Harry Oppenheimer Okavango Research Centre of the University of Botswana during the 2001-2002 spraying cycles in Botswana, i.e. both aquatic and terrestrial invertebrates recovered well after the spraying with populations of species of most families returning to pre-spraying abundances after one year.281
More recently, the SAT was used in Ghana in an attempt to eliminate populations of Glossina tachinoides and Glossina pallidipes gambienis from 6745 km2. Low dose aerosols of deltamethrin (0.33-0.35 g a.i./ha) were sprayed using fixed wing aircraft over three main river systems. These SAT operations were followed by an integrated strategy of ground spraying, insecticide-treated targets and cattle to sustain the reduction in population density. The SAT failed to completely eliminate tsetse from the treated areas, but a reduction of 98 per cent was obtained in comparison with pre-treatment population levels, i.e. from 16.7 flies/trap/day to 0.3 flies /trap/day. One year after the SAT, monitoring indicated that both species were still detected but at very low levels (0.10-0.27 flies/trap/day).3
Hot fogging is another method for generating aerosols in which thermal smoke generators have been used to ignite a combustible mixture of sodium chlorate, sugar and DDT to generate an insecticidal smoke.114 Commercially available insecticidal fog generators, such as the Swingfog, have been employed in Zimbabwe and Botswana, and the vehicle-mounted TIFA has been used extensively in Zambia.
Cold foggers use cold spraying methods that in most cases use high air pressure to force out the fogging liquid into tiny particles. The most popular type of cold foggers is the ULV fogger, and most of the time ULV foggers are considered to be the same as cold foggers. The main part of an ULV fogger is an electric motor that is used to spray out the insecticide into small particles. The power of the air flow depends on the power of the motor, which fluctuates between 1 and 4 HP. Some cold foggers use oil-based fogging solution, while others use a water-based solutions and there are foggers that can use both types of solutions.
The use of these methods have mostly been discontinued as aerial application techniques improved.
Ivermectin
The use of ivermectin administered to animals as a means of killing tsetse flies has been investigated.215 Although flies feeding on treated animals died, the cost of ivermectin at the dosage levels required to be effective is considered to be uneconomical. It has been suggested that formulations of ivermectins other than those used may be more suitable, but they have not yet been investigated.413
Insect hormones and growth inhibitors
Insect growth regulators such as diflubenzuron, when applied to adult female tsetse flies, cause the female to produce larvae that do not pupariate.195 Similar effects have been found with insect juvenile hormone analogues.99, 220, 413
Field studies in Zimbabwe with the juvenile hormone mimic pyriproxyfen have demonstrated that it can be used in conjunction with traps, using the system developed by Hall and Langley,156 to, in effect, sterilize the tsetse population.157 The technique was developed further by applying pyriproxyfen in place of conventional insecticides in the widely used odour-baited target system for killing tsetse flies.158 Laboratory studies on triflumuron, a chitin synthesis inhibitor,219 showed that it induced abortion in tsetse flies at higher doses as well as preventing the emergence of adults from puparia at lower doses, thus effectively sterilizing the flies. Triflumuron has been tested on a field scale in Zimbabwe and the preliminary indications were that it would be a suitable candidate material for use in tsetse control by means of odour-baited targets.
Laboratory experiments of pyriproxyfen with Glossina morsitans morsitans proved that doses as low as 0.01 µg in 10 µL oil cm-2 on black cotton cloth targets caused females to produce non-viable offspring for at least two reproductive cycles following exposure.214 It was also possible to contaminate females after mating with contaminated males provided that they were exposed to at least 0.1 µg in 10 µL oil cm-2. This secondary contamination might thus increase the range of actions of targets.
These compounds are available should it become undesirable to use pyrethroids. Their action is, however, delayed, probably reducing their adoption despite their efficiency. It was also proposed to use pyriproxyfen in combination with the sterile insect technique in order to expand induced sterility to already mated females or even females entering into contact with sterile males and refusing mating attempts.29
Traps and targets
The use of traps in attempts to control tsetse flies was first developed in KwaZulu-Natal, South Africa, in 1930.165 Many Glossina pallidipes were killed in this control campaign, but the flies were not eliminated. Three to four per cent or more of the female flies in a population must be eliminated each day for a tsetse population to decline to extinction.161 Despite the large numbers killed in Harris’s traps, this level of elimination could not have been attained, probably because of density-dependent processes limiting the efficiency of traps and targets at low population densities.109 Various traps were designed in the 1930s after the work done by Harris,182, 343 most being based on a visual stimulus to the fly, although Fuller and Mossop131 had shown clearly the importance of odour to Glossina pallidipes.
Since the 1970s intensive attempts have been made to improve traps for tsetse flies both in West Africa68, 213 and in Zimbabwe.127, 160, 374 In West Africa, after the attractiveness of blue cloth had been discovered, the effectiveness of traps for flies in the palpalis group was greatly increased by using traps covered with blue cloth (Figures 9, 12 and 13).
Various patterns of traps have been developed in Africa.86 Traps may be used to monitor tsetse populations and are widely used in Africa to assess the success of control operations against tsetse flies. If the traps are treated with insecticide, traps can be effective in killing a much larger numbers of flies. It was demonstrated that only ~ 20 per cent of Glossina tachinoides, Glossina palpalis gambiensis and Glossina morsitans submorsitans attracted to a trap actually enter it in West Africa299 and the efficiency of insecticide traps is probably five times that of non-impregnated ones. Such traps have been used to break transmission of trypanosomosis in livestock and humans.48, 113, 151, 226
In eastern and southern Africa, phthalogen blue cloth has been incorporated into traps, which originally had a white outer cover, a colour known to be attractive to some tsetse flies. Both blue and black cloth are attractive to Glossina morsitans and Glossina pallidipes,149 but if given a choice, the flies prefer to alight on black cloth. Not all species of flies react in the same way to colour. In KwaZulu-Natal, South Africa, it has been found that Glossina austeni is strongly attracted to phthalogen blue cloth but not to black cloth.202 When given the choice, this tsetse fly alights in roughly equal numbers on black or blue cloth. Traps such as the Epsilon and F3 (Figures 9 and 12) used against Glossina morsitans and Glossina pallidipes have a blue outer cover to attract flies and a section of black material within the trap to induce flies to enter the traps and settle. The same principle was applied for biconical and monoconical traps used to sample riverine tsetse flies in West Africa.68, 225
When ready to move many of the flies on the black cloth fly upwards into the white mosquito net conical canopy, which leads to the retaining device. It is important to have the canopy well illuminated as the upward flight is in response to the light. In KwaZulu-Natal, both Glossina brevipalpis and Glossina austeni can be caught in a low trap with a blue outer cover and black internal crossed screens and with two conical canopies in a lateral position, i.e. the H trap.200 These two fly species tend to fly horizontally when taking off, rather than vertically as is the case with Glossina pallidipes and Glossina morsitans.
Crossed plywood panels painted blue and covered with sticky material have been used to catch Glossina austeni.242 Improved versions of this trap have been used in surveys for both Glossina austeni and Glossina brevipalpis in KwaZulu-Natal199, 201 and to monitor the Glossina austeni eradication campaign on Unguja Island, Zanzibar.393, 401, 403
The performance of traps for Glossina morsitans and Glossinia pallidipes has been greatly improved following the identification of kairomones produced by host animals. Carbon dioxide is known to attract all species of tsetse flies on which it has been tested but it is not cost- effective to use it on a large scale in the field. Kairomones found so far that can be used in the field are acetone, methyl ethyl ketone (butanone), 1-octen- 3ol (octenol), 4-methylphenol, 3-n-propylphenol and 3-methylphenol. Not all of these substances are effective against all flies, and not all need be included in a bait for it to be effective.
Baited Epsilon traps are used to sample both Glossina morsitans and Glossina pallidipes in Zimbabwe but they are considered ten times more efficient for the second species, with trap efficiencies of 0.001 and 0.01, respectively (probability that a single trap catches a fly in one day given that the fly is present in an area of 1 km2 around the trap).71
The kairomones used in the field in Zimbabwe against Glossina morsitans and Glossina pallidipes are ineffective against the palpalis group of flies in West Africa, apart from Glossina tachinoides, which responds to the bait used in Zimbabwe, although different proportions of the constituents are needed. In West Africa Glossina longipalpis (morsitans group) and Glossina medicorum (fusca group) also respond to the bait used in Zimbabwe.336
Despite past and recent extensive research programmes in West Africa, no synthetic odours have been developed that could increase the catches of the palpalis group species by more than two, whereas natural hosts can increase trap efficiency up to five times.125, 126, 133, 253, 300 Natural host odours are generally much more effective than synthetic odours, as evidenced by trap catches of Glossina tachinoides catches that were increased fivefold using odours from cattle but not humans. In contrast, both human and cattle odours increased catches of Glossina pallidipes gambiensis two fold. For Glossina pallidipes palpalis, odours from pigs and humans increased five times the number of tsetse attracted to the vicinity of the odour source but had little effect on landing or trap-entry.300
In KwaZulu-Natal, South Africa, Glossina austeni responded to carbon dioxide but not to any of the other known kairomones. However, on Unguja Island, acetone (300 mg/h) combined with cow urine (60-130 mg/h) increased the catches of male Glossina austeni two- to three-fold in the rainy season, but not in the dry season. There was no effect of octenol used alone or in combination with aceone.401 Glossina brevipalpis responded to carbon dioxide, octenol, 4-methylphenol and acetone (but not to butanone).202 The doses of octenol and 4-methylphenol used in KwaZulu-Natal, South Africa, are higher than those used in Zimbabwe and the use of acetone is only of benefit in the dry season. The reason for this seasonal effect has not yet been determined.
Behaviour of tsetse flies in odour plumes resulting from release of kairomones has been investigated44, 354, 356 and it has been shown that flies may react differently at different doses of odour. Comparison of synthetic odour to the odour from an ox, at the same dose, indicated that further attractant(s) exist in natural ox odour356 for both Glossia morsitans and Glossina pallidipes. A similar conclusion was reached for Glossina brevipalpis, and possibly for Glossia austeni.202 Comparing odour from small to large numbers of cattle with an equivalent dose of the synthetic odour used in Zimbabwe for Glossina morsitans and Glossina pallidipes164 demonstrated a relationship between quantities of odour and fly catch, so that a tenfold increase in odour gave a 2,3- to 2,8-fold increase in catch for Glossina pallidipes and 1,4- fold increase for Glossina morsitans. It was also shown that odour from the cattle was more effective than that from the synthetic odour, again indicating that further attractant(s) remain to be identified in host odour. Skin secretions (sebum) of cattle also affect the behaviour of tsetse flies,407 especially after alighting on a stationary cloth screen.
The way in which tsetse flies move through the habitat has an important bearing on where traps should be positioned so as to be most effective. Tsetse flies of the morsitans group use game trails when following odour in thicket279 and tend to fly round bushes rather than over them. Vale has developed a deterministic model to indicate how flies move through three vegetation types in Zimbabwe.379 When placing traps in the field, they should be clearly visible to a fly but need not be in a large clearing. Provided there are breaks in the vegetation of over a metre wide, a hungry fly will move up an odour trail. In riverine vegetation, traps targeting the palpalis group should be deployed perpendicular to the flying line as this will increase their visibility. There should be no distracting host-sized objects, such as rocks or small dense bushes near the trap, and if the trap is of the single entrance Epsilon or F3 type, the entrance should face downwind. It is particularly important that the canopy of the trap is well illuminated so that flies move up into the collecting device. If a trap is needed in a position that does not meet the requirements, a small amount of trimming of the vegetation can improve the quality of the position and result in better catches. In well-grassed areas, it will be necessary to make a clearing round the trap to protect the trap against fires. Selecting the trap type is crucial when attempting to trap tsetse, as well as knowing the behaviour of the fly in the specific ecosystem that is being sampled. A detailed protocol and an update of the different trap models corresponding to the different groups can be found in32 (http://www.acp-st.eu/sites/all/files/news-views-docs/Attracting_Trapping_insect_vectors.pdf). However, field experience of the field staff remains extremely important and the performance of traps can vary from 1 to 100 between an inexperienced and an expert field technician. This illustrates the importance of sustaining the “know-how” in the existing structures in Africa that are dedicated to tsetse control.
Where tsetse flies are abundant, cattle cannot be easily maintained, but artificial targets can be used against the flies (Figures 10 and 11). These targets correspond to simplified versions of the traps and consist of a cloth screen in a frame mounted on a pole. In some versions the cloth is flanked by bands of black mosquito netting, which is not readily visible to tsetse flies. The screen and the gauze are sprayed with insecticide, often with deltamethrin suspension concentrate. In West Africa, blue screens, which are good visual attractants, are used so that flies come to the target, alight on it and receive a lethal dose of insecticide. Sufficient targets must be deployed to ensure that flies have a high probability of encountering one. Very good results have been achieved using this method in West Africa.224
It has been shown377 that tsetse flies flying in an odour plume are diverted to host-like stationary objects and a variety of visual cues are involved.353 Differences in the degree of response by different species of tsetse flies to host derived stimuli have been demonstrated.407 In Zimbabwe, targets are used in conjunction with the kairomones discussed above. The target first used in routine control operations consisted of a black cloth centre panel, flanked by panels of black mosquito netting, mounted on a pole and pivoted so that the target faced across the prevailing wind (Figure 10). This all-black cloth target was used widely in Zimbabwe in conjunction with kairomones and with deltamethrin as the insecticide applied to the target (Figure 11).
Work in West Africa on the palpalis group of flies showed that bright royal blue targets were better attractants than black targets.151 In Somalia, it was shown that a combination of blue and black cloth targets was more effective than all black targets against Glossina pallidipes.361 Further work in West Africa showed that the best pattern for Glossina palpalis was a phthalogen blue centre panel flanked by two black panels, with both colours being attractive to the fly but the black cloth being the preferred substrate for alighting, enhancing the tendency for this fly to settle on the outer edges of any target. Recent efforts to optimize the efficiency of traps and targets in West Africa focused on parameters like shape, colour and size of the devices, resulting in very efficient devices but with an attractiveness that is mainly based on visual stimuli.298, 299 In particular, adding black mosquito-netting on the sides of the coloured fabrics increased the interception of flies flying around the target without alighting attempts, thus increasing its efficiency.
A detailed study was made of colour combinations and materials that could be used to make targets for Glossina morsitans and Glossina pallidipes in Zimbabwe.378 Although larger targets do perform better, an acceptable target had a central black panel 0,74 m wide, flanked by bright royal blue panels 0,5 m wide. All panels were 1 m high. Both Glossina morsitans and Glossina pallidipes tend to settle near the centre of a target and this trend was enhanced by the black centre panel, which is a preferred alighting stimulus for these species. An additional advantage of this pattern of target is that only the black cloth needs to be treated with insecticide.
Reduced size of blue-black targets , i.e. the so- called “tiny targets” to 0.06 m2 flanked by a panel (0.06 m2) of fine black netting appeared to be efficient to control Glossina fuscipes fuscipes in sleeping sickness foci of Uganda.232 Research conducted on West-African riverine species showed that reducing the target size to 0.25 m2 might allow cost-savings.298 Although the technology proved cost-effective in sleeping sickness foci involving community engagement,321 the entomological index (number of flies killed / item) is important to consider for centralized projects, where the cost of deploying the targets generally exceeds the cost of the fabric.37 In this case, a more efficient cost reduction is obtained by deploying the targets in suitable habitats that will significantly reduce the target density per km2.109
In the original field trial of the odour-baited target system384 the insecticide dosage was low and targets had to be sprayed every four months to ensure high fly mortality. In Zimbabwe, the application of insecticide was done at a central point and, to cater for possible variation in cloth quality, the insecticide was applied to give 2 g of active ingredient on the central black cloth panel (0,75 m2). This blue and black target was the pattern used in Zimbabwe at the time of writing (Figure 15). Most of the recent tsetse control programmes, however, use polyester material impregnated with deltamethrin at the factory. Although it is claimed that this technology results in persistence of the insecticides for up to 1 year, the fading of the colour of the cloth material generally necessitates a target replacement frequency of three months.
Investigations showed that 10 g of the phenols and octenol mixture were needed in a sachet if it was to last a year. It would also be necessary for the sachet to be in a pocket on the blue section of the target for this degree of longevity in full sun. The bait would only last about seven months if held in a pocket on the black cloth.357 A bottle containing 750 ml of acetone or butanone with a 2-mm diameter hole in the lid yields odour for a year.
The original approach to deploying odour-baited targets in Zimbabwe was to aim at a relatively uniform distribution of targets on the ground at an average of three to five per km2.384 This density provided rapid reduction of Glossina morsitans and Glossina pallidipes that could be achieved within a year. This approach has been used extensively, with the modifications in target colour and longer lasting insecticide and baits, with good results in Zimbabwe, Malawi, Namibia and Zambia, provided the guidelines for the method were followed. A modification of the pattern of deployment in Zimbabwe has been followed in the broken country near the Zambezi River Valley escarpment. As was the case at the time that ground spraying was in use, targets were deployed only in the essential hot season habitat of the flies, which was generally associated with drainage lines,as is usually applied against riverine flies in West89 and East Africa.351 In the Niayes area of Senegal, rivers have disappeared and Glossina palpalis gambiensis occurs as a metapopulation occupying a patchy landscape of suitable habitats that represent less than five per cent of the total area. These suitable habitats were accurately identified using distribution models based on remote sensing data. Deployment of insecticide-impregnated targets and traps only in these suitable habitats at a trap density of 1-3.4 per km2 reduced the G. p. gambiensis population by more than 95 per cent.109
In Burkina Faso, targets covered with adhesive film significantly increased trapping efficiency compared with either blue and black biconical or monoconical traps for Glossina tachinoides (up to 14 times), for Glossina palpalis gambiensis (10 times) and Glossina morsitans submorsitans (6.5 times).299 Targets impregnated with pyrethroids are thus commonly used to control tsetse in West Africa. In Burkina Faso, targets deployed at a density of 2.4/km reduced populations of Glossina tachinoides, Glossina palpalis gambiensis and Glossina morsitans submorsitans in an area of 3500 km2.293 In the same country, Glossina tachinoides and Glossina palpalis gambiensis populations were reduced by more than 95 per cent with 10-20 targets deployed per linear km of riverine vegetation in a total area of 40,000 km2.280 On the Loos Islands of Guinea, a Glossina palpalis gambiensis population was reduced by more than 99 per cent in a wet forest environment by deploying 30 targets per km2.196 A density of 15 targets per km2 reduced the population by 80 per cent in the neighbouring continental sleeping sickness focus of Boffa, and as a result the incidence of sleeping sickness was significantly reduced.82 Similar results were obtained against Glossina fuscipes fuscipes in Chad, where insecticide-impregated targets deployed at a density of ~50/km along the river course reduced the apparent density of the tsetse population by more than 99 per cent, which contributed to a reduction of the transmission of sleeping sickness in the Mandoul focus.244 In Uganda, densities of a Glossina fuscipes fuscipes population were reduced by 90 per cent using an overall target density of 5.7 per km2.351 In the Ghibe valley of Ethiopia, Glossina pallidipes densities were reduced by more than 80 per cent using insecticide impregnated targets at a density of 5/km of river.230
The pattern of deployment of the odour-baited targets in the field has been modelled,379, 418 on the basis of known features of tsetse movement. The deployment needed will differ between species, and in different vegetation patterns. At the site of the target in the field, features such as good visibility, absence of distracting objects and pathways through the vegetation379 are important, as is the case for odour baited traps. It is necessary to have a fire break round each target, and the ground round the target should be reasonably level so the target swings easily.
There are factors that adversely affect the use of the odour-baited target system for tsetse control. The prime cause of failure is theft of targets by rural populations. Failure to make adequate fire breaks round targets is another major cause of failure of the target system. Wind damage to targets is another severe effect in many localities, and targets may become ineffective in a matter of weeks after being set up. Alternative materials to cloth may help solve this problem. In some localities, damage by wild animals such as elephants, baboons and hyenas can reach high levels, although in much of the undeveloped country in Zimbabwe damage levels are low. Domestic cattle are also known to destroy odour-baited targets. A major difficulty in operating the system is to ensure maintenance of the targets. Target sites need to be georeferenced using GPS to relocate them. Access to the area being treated is essential, and the supply of the targets and baits at the correct times is a complex logistical problem in much of Africa. The capacity to operate the odour-baited target system does not exist in all the tsetse-infested countries of Africa, so the suppression of the tsetse fly by this means will not be a short-term operation.
Some of these difficulties might be solved if it were possible to convert trees to odour-baited targets. This can be done quite easily for tree stumps, but it has been shown that it is not easy to do for trees,385 and was more expensive to do than using cloth targets.
Attempts have been made to encourage local communities in tsetse-infested areas to run their own tsetse control operations. A successful trial, using traps rather than targets and a simplified bait consisting of cattle urine and acetone, was run at Nguruman in Kenya, and similar systems have been operated in West Africa.113 In Burkina Faso, traps and targets are generally considered as a public good rather than the property of an individual197, 198 and there are no examples of their sustained use by communities in the absence of a centralized programme or a government institute.
Target barriers
Under carefully controlled experimental conditions, odour-baited targets achieved local elimination of Glossina morsitans and Glossina pallidipes in Zimbabwe384 in 12 months if the mean target density was three to five per km2. Trap samples from a traverse across the centre of the main invasion front yielded no flies further into the treated area than 5 km. The targets also exerted a marked reduction of the tsetse population outside the treatment area. These results gave rise to the concept that targets could be used as a barrier to invasion by tsetse flies.
Initially, it was considered that a narrow barrier with a high density of targets would give the best result. It has since been shown267 that such barriers do not prevent some flies crossing them. Using knowledge of fly survival rates, expectation of the net daily movement of the flies and the probability of a fly being killed by an odour-baited target treated with deltamethrin, Hargrove162 has computed the probability of flies crossing barriers of different widths and target densities. An effective barrier width of eight times the expectation of net daily movement of the flies (about 1 km in Zimbabwe), when the target density was the same as for tsetse elimination in 12 months, was proposed. In Zimbabwe, the target density would be a mean of four per km2 and the width about 8 km for Glossina pallidipes and possibly less for Glossina morsitans. The number of targets involved would be less than that of a barrier of four lines of targets 0.5 km apart, with a high density of targets in the lines. The probability of a fly crossing the proposed barrier would be about 0,001. As the barrier exerts an effect beyond its boundary, the fly numbers on the invasion front of the barrier decline as the barrier is approached. The predictions on barrier effectiveness do not take into account game trails, footpaths and roads that cross the barrier. Such features may reduce the effectiveness of the barrier through transport of flies on vehicles or animals.
A trial of the type of barrier proposed by Hargrove162 was conducted on a section of the boundary between Zimbabwe and Mozambique in the Mudzi district.408 The barrier was 40 km long, 8 km wide and target lines were 0.5 km apart with targets spaced at 0.5 km intervals along the line. The country is relatively flat and the target density in this regular pattern was roughly 4 per km2. Locally owned cattle treated with deltamethrin grazed within the barrier and for a distance of about 20 km from the international boundary. The mean density of cattle in the 428 km2 trial area was eight to 12 per km2. The distribution of cattle was not uniform and there was no control over where they grazed.
Monitoring of tsetse flies (mainly Glossina morsitans but with Glossina pallidipes also present) and AAT showed flies to occur in low numbers no further than 2 km from the invasion front and for cases of AAT to be rare. A trial was designed to check whether the insecticide-treated cattle alone would provide a barrier to flies moving into Zimbabwe. Following removal of the targets, there was a rapid increase in the distance into the barrier that flies could be caught, and also a rapid increase in the number of cases of AAT in the cattle, including some fatalities.
Following replacement of the targets, the situation soon returned to the original state. The trial demonstrated that, at this locality, the density of insecticide-treated cattle did not prevent flies from Mozambique entering Zimbabwe. How far into Zimbabwe the flies would penetrate was not determined, as it appeared that they might cross the 20 km-wide belt in which cattle had been treated with deltamethrin, so the targets were re-deployed. It was clearly shown that flies fed on cattle treated with insecticide and that transmission of trypanosomosis occurred. Insecticide-treated cattle can, however, be used to re-inforce the efficiency of target barriers.
In Burkina Faso, a barrier containing insecticide-impregnated targets deployed every 100 m along 7 km of riverine forest efficiently contained Glossina tachinoides and Glossina palpalis gambiensis.85 They are still used at the time of writing within the PATTEC campaign in this country.280
Odour-baited target barriers to invasion by tsetse flies can be effective, but they must be carefully maintained at all times. Attempts are being made to put the maintenance of target barriers out to contract but, as the barriers will have to be maintained for the foreseeable future, it remains to be seen how effective this will be. There are, however, no examples of a target barrier that has been sustained indefinitely. The only strategy to use them is probably as a temporary measure to protect cleared areas in a “rolling carpet” strategy.170
Insecticide treated cattle
The objective, or target, of a hungry tsetse fly is the host animal on which it will feed. If this animal is treated with a suitable residual insecticide, the flies alighting on it or simply touching it will be killed. Cattle dipped in DDT solution were used to eliminate small populations of Glossina pallidipes in KwaZulu-Natal, South Africa, in 1949.114 The same method was investigated in East Africa,414 but was not widely accepted.
The development of synthetic pyrethroid insecticides, such as deltamethrin, which are good acaricides as well as insecticides, has revived interest in using dipped cattle to eliminate tsetse flies in lightly infested areas. In Zimbabwe, a well-developed dipping system against ticks is operated in most parts of the country. In communal or traditional areas bordering on known tsetse fly areas, where cattle numbers are large enough to give good coverage of likely fly habitat, a dip wash containing deltamethrin is being used. Cattle are dipped every 14 days or, in the absence of dipping facilities, treated monthly with pour-on formulations of the dip. Flies alighting on the cattle are killed,350 or knocked down so as to be available to predators, and the reduction in AAT cases in areas under treatment has been dramatic. This approach is very efficient for tsetse fly control where cattle densities are high (>5/km2) and wild fauna is scarce. In terms of area under treatment, deltamethrin dipping is the most widely used anti-tsetse measure in Zimbabwe at present.322 The same approach has also been used in South Africa, where the acaricide (amitraz) usually used to control ticks was replaced temporarily by pyrethroids to manage outbreaks of AAT through the combined control of tsetse and ticks.97
It has been shown that pour-on applications of insecticides do not reduce the attractiveness of cattle to Glossina pallidipes and do not affect the proportion of flies that feed on the animals.21 Disease transmission can thus still occur after the cattle have been treated, and will only cease after the tsetse population has been reduced by the insecticide, and then only if there is no invasion of flies into the area where cattle have been treated with insecticide. Various insecticides for use as pour-on, plunge dipping or spraying of cattle are now available.9
Successes in using insecticide-treated cattle have been reported from southern and eastern Africa,349 as well as from West Africa. In the Stamp Out Sleeping sickness (SOS) campaignagainst Glossina fuscipes fuscipes in Uganda, 9–22 heads of cattle/km2 were treated every three months which suppressed the tsetse population and reduced the transmission of AAT.37, 261 The main goal was to limit the transmission of Trypanosoma brucei rhodesiense from cattle to humans.319 Although quite successful, the method was poorly adopted by the farmers. Pyrethroid pour-ons are also widely used to control tsetse in Ethiopia, their distribution being ensured by the National Institute for Tsetse and Trypanosomosis Control and Eradication (NICETT) that partially subsidizes their use. Recently, a pour on formulation that mixed insecticides with repellents increased the persistence of fly knockdown by 50 per cent and reduced the flies’ rate of engorgement.138 This pour-on also prevented transmission of AAT in northern Cameroon.
In comparative trials in Zimbabwe with three different pour-ons, the active ingredients of which were alphacypermethrin, deltamethrin and cyfluthrin, respectively, the dung of the animal treated with alphacypermethrin, which was dropped about seven hours after the insecticide was applied, was found to be lethal to dung beetles.380 This phenomenon is common to other pour-ons and also dip washes, and their widespread use for tsetse fly and tick control purposes could have considerable environmental implications for soil fertility.367 To reduce this impact, it has been proposed to restrict the application of pyrethroids to the preferred biting sites on the host and to treat only the most attractive animals.39, 40, 360, 362, 382, 386 In West-Africa, insecticide footbaths were used with some success and they reduced the treatment time and cost by more than 90 per cent; however, their use by individual large farms was much more sustainable than their collective use.26 The impact of this method is dual, i.e. both against ticks and tsetse, which remains one of the strong incentives for their use. The impact of the insecticide treatment is, however, more important for ticks than tsetse.
Cattle reared in zero grazing units can also be protected by the use of an insecticide fence placed around the pen (or on most exposed sides).16, 18 This method is not only efficient against tsetse flies but also other biting flies, particularly Stomoxinae, and mosquitoes. It has been tested in Kenya, Burkina Faso, Ghana, Ethiopia and Senegal, and it can also be used to protect pigs in confined areas.196
The sterile insect technique (SIT)
The concept of using sterile insects for pest control is not new and was already conceived by Knipling208 in the 1930s. Knipling’s innovative idea was a radical departure from traditional pest control methods that compromised the survival of the target insect and was based on the realization that insects pests could be managed by compromising their hereditary machinery.392
The SIT requires the production of large numbers of the target insect in dedicated mass-rearing factories, the sterilisation of the male sex using ionizing radiation, and the sustained and systematic release of the sterile males over the target area in numbers large enough to out-compete the wild males for mating with wild females. Virgin, native female insects that have mated with a sterile male will produce no offspring, and the technique therefore is a type of “birth control” of insects. As a result, the population replacement rate will be reduced and the size of the targeted pest population will decline. The sustained release of a constant number of sterile males will result in an increase in the sterile to wild male ratio with each generation, and therefore the technique becomes more efficient at lower population densities. These inverse-density dependent properties make the technique unique, and the SIT should ideally be combined with or preceded by control tactics that are effective at high population densities. If sustained long enough, and in certain ecological settings, the technique can be used to drive insect pest populations to extinction.
The SIT has several advantages: 1) it is non-intrusive to the environment with no adverse effects on non-target organisms; 2) it is species-specific; 3) it can easily be combined with other biological control methods such as parasitoids, predators and pathogens; 4) there is no risk of the development of resistance to the effects of the sterile males (provided that adequate quality assurance is practiced in the production process); and 5) the sterile insects cannot get established in the release areas as with other biological control programmes.394
The SIT also has its limitations: 1) the release of sterile insects is only effective and cost efficient when the target population density is low; 2) it requires detailed knowledge of the biology and ecology of the target pest; 3) the insect should be amenable to mass-rearing at a reasonable cost; 4) there is a delayed effect as the target insect is not killed but sterility introduced; and 5) the technique can only be used for those insects where the development phase that is used for the release (e.g. in most cases the adult insect) is not contributing to the damage. In addition, the SIT necessitates efficient release and monitoring methods, which have to be applied on an area-wide basis.395
The SIT has been used in the last decades in combination with other control tactics with great success to eradicate, suppress, contain or prevent the establishment of pest populations of Diptera, Coleoptera and Lepidoptera.13, 23, 118, 233, 340
The SIT and tsetse flies
Theoretical considerations for applying the SIT for the management of Glossina subspecies date back to 1958.326 The irradiation of puparia as a method for producing male tsetse flies with chromosome abnormalities (and thus effectively sterile if mated to normal females) was developed by Potts.295 The first field trial of the SIT was conducted on an island in Lake Kariba between 1967 and 196993 and chromosomal abnormalities were induced in males by chemical means as the flies emerged from field-collected puparia. The test population was reduced by 98 per cent in this campaign, and eventually reached the stage where no flies could be caught by any of the available sampling methods.
In 1972, field studies in Chad showed the effects of transport, irradiation etc. on the quality of gamma sterilized male flies belonging to the palpalis group. Male Glossina tachinoides, reared in Maison Alfort, France and gamma irradiated with 150 Gy were shipped in isothermic boxes (25 °C) by air mail to Chad for release without any significant negative impact on their quality and behaviour.83, 84
In a later pilot study,88 showed that pre-release suppression of a Glossina p. gambiensis population by application of a non-residual insecticide clearly benefited the outcome of an SIT programme. Following the suppression of the target population, sterile to wild male ratios of 7-10 to 1 were adequate to eradicate the native target population within 19 months. An additional five months were needed to achieve the same in an area without prior insecticide application.
In another release trial, the release of 5,500 sterile male Glossina p. gambiensis in 10 release sessions was preceded by two spraying operations of decamethrin, separated by 14 days on tributaries of the Mouhoun River (then called Volta Noire) in Burkina Faso (1977-1978). Monitoring revealed good dispersal (up to 2 km after 48 hours) and good survival (recapture 20-44 days after release) of the sterile flies. On average 8.7 sterile males were trapped for each wild male.390
The first SIT-based programme of any magnitude was conducted in the Tanga area, United Republic of Tanzania. The native Glossina morsitans morsitans population was reduced by two aerial applications of endosulfan,419 which was followed by the release of sterile male pupae for 15 months, which required the development of efficient and sophisticated pupal handling and irradiation techniques.419 About 135 sterile males were released per km2 resulting in an average ratio of 1.12 sterile to 1 wild male, which was low but enough to keep the indigenous population at the 80 – 95 per cent reduction level obtained after the insecticide applications.420, 421
A combination of trapping techniques, insecticide-impregnated targets and the release of sterile males was used to eradicate Glossina p. gambiensis and Glossina tachinoides from 3,500 km2 of Guinea savannah in Sidéradougou, Burkina Faso in the 1980s. During the dry season 6,500 insecticide-impregnated targets were deployed at intervals of 100 m for 4 months, reducing the population by 91-94 per cent. This drastic reduction in the native fly population reduced the requirement for sterile males to only 20-35 sterile males per linear km to obtain the desired sterile to wild male ratio of 10:1. The main river systems were free of tsetse six months after the release of sterile male flies.293
The same approach was used to eradicate a Glossina. p. palpalis population from 1,500 km2 agro-pastoral land in Southern Plateau State of Nigeria. Insecticide-impregnated targets275 and biconical traps achieved 97-99 per cent reduction of the fly population after three months and weekly releases for 18 months resulted in eradication in the pilot phase of fourisolated forest patches.345 The programme was extended over the entire 1,500 km2 block and in 1988 the fly was declared eradicated. The programme demonstrated that a sterile to wild male ratio of 10:1 was a prerequisite to achieve eradication and that different habitats and different species demanded different release rates and different time frames.
Although the programmes in Burkina Faso and Nigeria managed to eradicate local populations of the target species, the tsetse-free status of the target zones could not be sustained, due to invasion pressure from the adjacent untreated areas. These examples demonstrate the need for an area-wide approach in order to achieve sustainability.
The Glossina austeni eradication programme on Unguja Island, Zanzibar (1994-1997), was the first SIT-based eradication effort that adopted an AW-IPM strategy. Only Glossina austeni was present on the island and was responsible for high prevalence rates of AAT in livestock. The fly population was suppressed using the live-bait technique in the agricultural areas of the northern half of the island that had high cattle densities, and by deploying insecticide-treated blue cotton targets in the dense forested areas mainly in the south. This was followed by the aerial dispersal of sterile male flies over the entire surface area of Unguja Island, including the small offshore islands.399 The last indigenous Glossina austeni fly was trapped in September 1996 and Unguja Island has remained free of tsetse and AAT ever since.314, 398
This programme highlighted that the quality of the released insects is one of the most critical aspects for the SIT to be successful. The released sterile insects must compete with the wild males for mating with wild females, and must therefore intermingle rapidly with the wild insect population after release and mate at the same rate as their wild counterparts.395 The quality of the insects can be impaired by factors in the rearing e.g. crowding, rearing procedures, diets, insect pathogen load, deterioration of the strain, laboratory adaptation, genetic drift etc., or through handling, irradiation, packaging, release methods, etc.325 A detailed analysis of the trapping data of the wild and sterile flies indicated that both types of flies congregated in the same ecological niches, showing that both the sterile and the wild insects responded in a similar way to environmental cues.400 Similar observations were made with Glossina p. gambiensis in Burkina Faso, even after the colony had been cultured for more than 35 years.335
In 2005, the Government of Senegal launched an AW-IPM project that aimed to create a sustainable Glossina palpalis gambiensis free zone in the Niayes, an area located north-east of the capital Dakar. During phase II of the phased conditional approach (PCA), data collection activities showed that there was only one species in the area, with a limited distribution of 1000 km2,38 and genetically isolated from the remainder of the tsetse belt.329 The impact of the removal of AAT was found to be significant, with annual benefits of €2 million for farmers in the tsetse-infested zone.29 The adopted AW-IPM strategy included suppression activities with insecticide-impregnated traps and the use of pour-on for cattle, followed by the release of sterile males to eliminate the remaining relic pockets.
During the pre-operational phase (PCA III) activities were carried out in preparation of the operational phase (IV) such as, establishment of colonies, compatibility and competiveness studies,265 development of long distance shipping procedures,264, 277 aerial release methods,262 and a MaxEnt-based distribution model109 to guide the suppression, sterile male releases and the monitoring of the campaign.
During the operational phase (PCA IV), scientific principles continued to guide project implementation in the three operational blocks of the project. At the time of writing, the fly population was deemed eradicated in Block 1 (last wild fly was trapped on August 9, 2012), and in Block 2 and 3, the apparent fly density was reduced to < 0.001 fly per trap per day.
All these projects demonstrated that 1) the SIT is a powerful and robust control tactic and is very effective against riverine and some savannah tsetse flies in certain ecological settings, 2) tsetse flies can be successfully mass-reared in Africa, 3) a prolonged deployment of traps and targets in dense riverine vegetation does not lead to eradication, and 4) only the Unguja progamme (and later the Senegal project) was implemented according to the area-wide concept and could sustain its tsetse-free status.402
An alternative to rearing flies for sterilization is to sterilize males in a wild population by chemical means (autosterilization). The development of efficient odour-baited traps has opened the way for this approach in Zimbabwe, where a suitable sterilizing device has been developed173 and tested successfully,383 but the very toxic nature of the chemicals used precludes its widespread application. The juvenile hormone mimic and the chitin synthesis inhibitor mentioned above may provide a safe way to autosterilize female tsetse flies effectively. These methods eliminate the need for the costly artificially maintained tsetse fly colonies. It has been suggested that, under some circumstances, the sterilization approach to tsetse fly control may be best216 but, until more accurate information is available on fly population dynamics, the use of population models in predicting the outcome of control measures is unlikely to be accurate. This approach has never been tried on an operational scale.
Hybridisation
The possibility of controlling tsetse populations using hybrid sterility was already proposed in the early 1940s,388 long before the concept of SIT was tested. Despite detailed research and a large volume of publications to unravel the mechanism of hybrid sterility resulting from crosses between closely related morsitans and palpalis species, only two small field tests have been conducted. In 1945, two exotic species, Glossina morsitans morsitans and Glossina fuscipes fuscipes, were introduced in a habitat of Glossina swynnertoni.185 Glossina morsitans morsitans managed to survive and produced a second generation (the habitat of both species overlap and they produce sterile cross matings)129 but G. fuscipes fuscipes failed to do so, probably because they could not find suitable larviposition sites.
Vanderplank (1947)389 conducted a similar experiment that indicated the possibility of eradicating Glossina swynnertoni by releasing large numbers of Glossina m. centralis. Unfortunately, the principle has not been further tested in the field, but it remains an intriguing concept that deserves further experimentation and validation in the field.
Conversely, the possibility of eliminating Glossina morsitans by introducing Glossina swynnertoni into the Glossina morsitans habitat, where it would not become established, was examined.188 These two species cross mate readily in the laboratory and the interspecific cross is sterile. It was hoped that Glossina morsitans would be reduced in number, as many females would mate with male Glossina swynnertoni, but the field experiment was not successful.190
Other suggestions for control based on genetics—such as releasing laboratory-bred strains of tsetse flies, homozygous for translocations, into natural populations90 — have not been tried in the field.
Biological control
Some animals, such as birds, wasps (Bembex spp.), robber flies (Asilidae) and spiders (Hersilia spp.), are known to prey on tsetse flies on occasion.271 As these animals do not feed only on tsetse flies, they are not candidates for elimination of the flies.
A number of insect parasitoids that parasitize Glossina puparia have been discovered.212, 271 Recent taxonomic revisions have altered the old names of many of them.147 One of the parasitoids was released on a large scale in an attempt to control Glossina morsitans in Malawi,211 but it was not successful, possibly because most of the parasitoids are not host specific.
A parasitoid wasp in the family Mutillidae (Chrestomutilla glossinae Turner) is well known in Zimbabwe73, 168, 169 and has also been recorded in Zambia and Malawi. Under natural conditions it appears to parasitize only puparia of the genus Glossina, but in the laboratory it can, with difficulty, be reared from Sarcophaga sp. puparia.73, 167 It has not yet been possible to culture the wasp in the numbers needed to attempt biological control.
A virus, widespread in Africa, that infects Glossina pallidipes and causes hypertrophy of the salivary glands of both sexes and sterility in the males has been reported.273 This virus has no potential for biological control purposes because of its low virulence and because it cannot be produced in vitro. However, this virus represents a major threat to the mass-rearing of tsetse, especially Glossina pallidipes. An integrated strategy including the use of antiviral drugs (valacyclovir) and clean feeding (each holding cage is offered fresh, clean blood) allows proper control of the virus.1, 2
Conclusion and perspectives
AAT will continue to be a major constraint to livestock development in much of Africa for some time to come.
In 1996, a new grouping of international agencies, the Food and Agriculture Organization of the United Nations (FAO), the World Health Organization (WHO), the International Atomic Energy Agency (IAEA) and the African Union/Interafrican Bureau for Animal Resources (AU-IBAR) initiated an international forum called Programme Against African Trypanosomiasis (PAAT). The stated objective was to promote integrated trypanosomosis control through co-ordinated international action so as to improve food security and sustainable agriculture and rural development. One of the notable results of this initiative was the mapping of tsetse distribution, although at a very low resolution,66 and more recently the development of a Progressive Control Pathway for AAT.104
In July 2000, The African Heads of State and Government called for renewed efforts to control tsetse in Africa and launched the Pan African Tsetse and Trypanosomosis Eradication Campaign (PATTEC), at the 36th Ordinary Session of the AU summit meeting in Lomé. The PATTEC initiative was approved at the AU’s meeting in Lusaka, Zambia in 2001 and officially launched in Ouagadougou, Burkina Faso later in the same year.402 The first countries benefiting from this initiative (first phase) were Burkina Faso, Mali, Uganda, Ethiopia, Kenya and Ghana but despite huge investments from the African Bank of Development, none of them succeeded in creating any sustainable tsetse-free zones.10
Whatever the control method (see Table 2), sustaining tsetse reduction always proved difficult, namely because the reduction of the AAT constraint caused the farmers to stop their control efforts. Thus, tsetse elimination at present appears to be the only sustainable strategy for AAT control in Africa, but is generally not applied following an AW-IPM principle, which is conditional for its sustainability. Environmental and socio-economical monitoring is also crucial to ensure the benefits of tsetse control. Moreover, it is necessary to plan land management before starting any tsetse control effort, in order to make sure that a potential increase in cattle density or productivity is compatible with the long-term protection of environmental and cultural resources.
In this chapter, drug treatments, trypanotolerant livestock and attempts to develop a vaccine against trypanosomes are not discussed, although they may be effective strategies for AAT control (see African animal trypanosomoses).
References
- ABD-ALLA, A. M. M., A. G. PARKER, M. J. B. VREYSEN, AND M. BERGOIN. 2011. Tsetse salivary gland hypertrophy virus: hope or hindrance for tsetse control? PloS Negl. Trop. Dis. 5: e1220.
- ABD-ALLA, A. M. M., H. M. KARIITHI, A. H. MOHAMED, E. LAPIZ, A. G. PARKER, AND M. J. B. VREYSEN. 2013. Managing hytrosavirus infections in Glossina pallidipes colonies: feeding regime affects the prevalence of salivary gland hypertrophy syndrome. PLoS One 8: e61875.
- ADAM, Y., G. CECCHI, P. M. KGORI, T. MARCOTTY, C. I. MAHAMA, M. ABAVANA, B. ANDERSON, M. PAONE, R. MATTIOLI, AND J. BOUYER. 2013. The sequential aerosol technique: a major component in an integrated strategy of intervention against riverine tsetse in Ghana. PLoS Negl Trop Dis 7: e2135.
- AKSOY, S., B. WEISS, AND G. ATTARDO. 2008. Paratransgenesis applied for control of tsetse transmitted sleeping sickness, pp. 35-48, Transgenesis and the management of vector-borne disease. Springer.
- ALLSOP, R., AND B. S. HURSEY. 2004. Insecticidal control of tsetse, University of Greenwich.
- ALLSOPP, R., 1984. Control of tsetse flies (Diptera: Glossinidae) using insecticides: A review and future prospects. Bulletin of Entomological Research, 74, 1–23.
- ALLSOPP, R., AND T. K. PHILLEMON-MOTSU. 2002. Tsetse control in Botswana - a reversal in strategy. Pesticide Outlook 13: 73-76.
- ALSAN, M. 2015. The Effect of the TseTse Fly on African Development. Am. Econ. Rev. 105: 382-410.
- ALSOP, N.J., 1993. Training manual for tsetse control personnel. Vol. 5. Insecticides for tsetse and trypanosomiasis control using attractive bait techniques. Food and Agriculture Organisation of the United Nations, Rome.
- AU-PATTEC. 2017. A review of the multinational project for the creation of sustainable tsetse and trypanosomosis free areas in east and west Africa: key results and lessons learned, pp. 47. PATTEC Coordination Office, Department of Rural Economy and Agriculture, Africa Union Commission.
- AUSTEN, E.E., 1903. A monograph of the tsetse flies (Genus Glossina, Westwood) based on the collection in The British Museum. With a chapter on mouth parts by H.J. Hansen, Phil.Doc. British Museum, London.
- BALDRY, D.A.T., EVERTS, J., ROMAN, B., VON OCHSSEE, G.A.B. & LAVEISSIÉRE, C., 1981. The experimental application of insecticides from a helicopter for the control of riverine populations of Glossina tachinoides in West Africa. VIII. The effects of two spray applications of OMS-570 (endosulfan) and OMS-1988 (decamethrin) on G. tachinoides and non-target organisms in Upper Volta. Tropical Pest Management, 27, 83–110.
- BARNES, B. N., J. H. HOFMEYR, S. GROENEWALD, D. E. CONLONG, AND M. WOHLFARTER. 2015. The sterile insect technique in agricultural crops in South Africa: A metamorphosis…. but will it fly? African Entomology 23: 1-18.
- BART, G., J. KNOLS, L. WILLEMSE, S. FLINT, AND A. MATE. 1993. A trial to control the tsetse fly, Glossina morsitans centralis, with low densities of odour-baited targets in west Zambia. Med. Vet. Entomol. 7: 161-169.
- BASSÈNE, M. D., M. T. SECK, S. PAGABELEGUEM, A. G. FALL, M. J. B. VREYSEN, G. GIMONNEAU, AND J. BOUYER. 2017. Competitiveness and survival of two strains of Glossina palpalis gambiensis in an urban area of Senegal. PloS Negl. Trop. Dis. submitted.
- BAUER, B., D. GITAU, F. P. OLOO, AND S. M. KARANJA. 2005. Evaluation of a preliminary trial to protect zero-grazed dairy cattle with insecticide-treated mosquito netting in Western Kenya. Trop. Anim. Health Prod. 38: 31-36.
- BAUER, B., J. FILLEDIER, AND I. KABORE. 1984. Large scale rearing of tsetse flies (Diptera, Glossinidae) in the CRTA Bobo-Dioulasso, Burkina based on in vitro feeding techniques. Rev. Elev. Méd. vét. Pays trop. 37: 9-17.
- BAUER, B., M. MAIA, D. MEHLITZ, P.-H. CLAUSEN, B. HOLZGREFE, A. ABONUUSUM, S. OSEI, T. KRUPPA, J. MAY, R. GARMS, O. DIALL, B. DIARRA, A. SOW, I. SIDIBE, P. LASSANE, C. MAHAMA, C. ABAVANA, B. DJAGMAH, AND E. DOTCHER. 2007. Protection of livestock with insecticide-treated mosquito fences against insects of veterinary and medical relevance. In AU (ed.), 29th Meeting of the International Council for Trypanosomosis Research and Control (ISCTRC), 1.-5.10.2007, Luanda, Angola.
- BAUER, B., S. AMSLER-DELAFOSSE, I. KABORÉ, AND M. KAMUANGA. 1999. Improvement of cattle productivity through rapid alleviation of African Trypanosomosis by integrated disease management practices in the Agropastoral zone of Yalé, Burkina Faso. Trop. Anim. Health Prod. 31: 89-102.
- BAX, S.N., 1937. The senses of smell and sight in Glossina swynnertoni. Bulletin of Entomological Research, 28, 539–582.
- BAYLIS, M., MBWABI, A.L. & STEVENSON, P., 1994. The feeding success of tsetse flies, Glossina pallidipes (Diptera: Glossinidae) on oxen treated with pyrethroid pour-ons at Galana Ranch, Kenya. Bulletin of Entomological Research, 84, 447–452.
- BLAIR, D.M., 1939. Human trypanosomiasis in Southern Rhodesia, 1911–1938. Transactions of the Royal Society of Tropical Medicine and Hygiene, 32, 729–742.
- BLOEM, S., J. E. CARPENTER, A. MCCLUSKEY, R. FUGGER, S. ARTHUR, AND S. WOOD. 2007. Suppression of the codling moth Cydia pomonella in British Columbia, Canada using an area-wide integrated approach with an SIT components. Area-Wide Control of Insect Pests: 591-601.
- BONOMI, A., BASSETTI, F., GABRIELI, P., BEADELL, J., FALCHETTO, M., SCOLARI, F., GOMULSKI, L.M., REGAZZINI, E., OUMA, J.O., CACCONE, A., OKEDI, L.M., ATTARDO, G.M., GUGLIELMINO, C.R., AKSOY, S., & MALACRIDA, A.R., 2011. Polyandry Is a Common Event in Wild Populations of the Tsetse Fly Glossina fuscipes fuscipes and May Impact Population Reduction Measures. PLoS Neglected Tropical Diseases, 5, e1190.
- BOUYER, F., BOUYER, J., SECK, M.T., SALL, B., DICKO, A.H., LANCELOT, R. & CHIA, E., 2015a. Importance of vector-borne infections in different production systems: bovine trypanosomosis and the innovation dynamics of livestock producers in Senegal. Revue scientifique et technique (International Office of Epizootics), 34, 199-212.
- BOUYER, F., HAMADOU, S., ADAKAL, H., LANCELOT, R., STACHURSKI, F., BELEM, A.M.G. & BOUYER, J., 2011. Restricted application of insecticides: a promising tsetse control technique, but what do the farmers think of it? PLoS Neglected Tropical Diseases, 5, e1276.
- BOUYER, F., SECK, M.T., DICKO, A., SALL, B., LO, M., VREYSEN, M., CHIA, E., BOUYER, J. & WANE, A., 2014. Ex-ante cost-benefit analysis of tsetse eradication in the Niayes area of Senegal. PLoS Neglected Tropical Diseases, 8, e3112.
- BOUYER, J. & LANCELOT, R., 2017. Using genetic data to improve species distribution models. Infection, Genetics and Evolution Journal., S1567-1348, 30108-30109.
- BOUYER, J. & LEFRANÇOIS, T., 2014. Boosting the sterile insect technique to control mosquitoes. Trends in Parasitology, 30, 271-273.
- BOUYER, J., BOUYER, F., DONADEU, M., ROWAN, T. & NAPIER, G., 2013c. Community- and farmer-based management of animal African trypanosomosis in cattle. Trends in Parasitology, 29, 519-522.
- BOUYER, J., COX, J.S.H., GUERRINI, L., LANCELOT, R., DICKO, A.H. & VREYSEN, M.J.B., 2018. Use of geographic information systems and spatial modelling in area-wide integrated pest management programmes that integrate the sterile insect technique. In: DYCK, V.A., HENDRICHS, J. & ROBINSON, A.S., (eds.). Sterile insect technique. Principles and practice in area-wide integrated pest management. 2nd edition.
- BOUYER, J., DESQUESNES, M., YONI, W., CHAMISA, A. & GUERRINI, L., 2015b. Attracting and trapping insect vectors. GeosAf Technical guide, 2, 23.
- BOUYER, J., DICKO, A.H., CECCHI, G., RAVEL, S., GUERRINI, L., SOLANO, P., VREYSEN, M.J.B., DE MEEÛS, T. & LANCELOT, R., 2015c. Mapping landscape friction to locate isolated tsetse populations candidate for elimination. Proceedings of the National Academy of Sciences, USA, 112, 14575-14580.
- BOUYER, J., KONÉ, N. & BENGALY, Z., 2013a. Dynamics of tsetse natural infection rates in the Mouhoun river, Burkina Faso, in relation with environmental factors. Special issue (invited paper): New insights in the interactions between African trypanosomes and tsetse flies: Frontiers in Cellular and Infection Microbiology, 47.
- BOUYER, J., PRUVOT, M., BENGALY, Z., GUERIN, P.M. & LANCELOT, R., 2007a. Learning influences host choice in tsetse. Biology Letters, 3, 113-116.
- BOUYER, J., RAVEL, S., VIAL, L., THÉVENON, S., DUJARDIN, J.P., DE MEEUS, T., GUERRINI, L., SIDIBÉ, I. & SOLANO, P., 2007c. Population structuring of Glossina palpalis gambiensis (Diptera: Glossinidae) according to landscape fragmentation in the Mouhoun river, Burkina Faso. Journal of Medical Entomology, 44, 788-795.
- BOUYER, J., SECK, M.T. & SALL, B., 2013b. Misleading guidance for decision making on tsetse eradication: Response to Shaw et al., 2013. Preventive Veterinary Medicine, 112, 443– 446.
- BOUYER, J., SECK, M.T., SALL, B., GUERRINI, L. & VREYSEN, M.J.B., 2010. Stratified entomological sampling in preparation of an area-wide integrated pest management programme: the example of Glossina palpalis gambiensis in the Niayes of Senegal. Journal of Medical Entomology, 47(4), 543-552.
- BOUYER, J., STACHURSKI, F., GOURO, A. & LANCELOT, R., 2009. Control of bovine trypanosomosis by restricted application of insecticides to cattle using footbaths. Veterinary Parasitology, 161, 187-193.
- BOUYER, J., STACHURSKI, F., KABORÉ, I., BAUER, B. & LANCELOT, R., 2007b. Tsetse control in cattle from pyrethroid footbaths. Preventive Veterinary Medicine, 78, 223-238.
- BOYT, W.P., MACKENZIE, P.K. & PILSON, R.D., 1978. The relative attractiveness of donkeys, cattle, sheep and goats to Glossina morsitans morsitans Westwood and G. pallidipes Austen (Diptera: Glossinidae) in a middleveld area of Rhodesia. Bulletin of Entomological Research, 68, 497–500.
- BOYT, W.P., MACKENZIE, P.K. & ROSS, C., 1970. An attempt to demonstrate the natural transmission of bovine trypanosomiasis by agents other than Glossina in the Sabi Valley of Rhodesia. Rhodesian Veterinary Journal, 1, 7–20.
- BOYT, W.P., MACKENZIE, P.K., PILSON, R.D. & LEAVIS, H., 1972. The importance of the donkey (Equus asinus) as a source of food and a reservoir of trypanosomes for Glossina morsitans Westw. Rhodesia Science News, 6, 18–20.
- BRADY, J. & GRIFFITHS, N., 1993. Upwind flight responses of tsetse flies (Glossina, spp.) (Diptera: Glossinidae) to acetone, octenol and phenols in nature: a video study. Bulletin of Entomological Research, 83, 329–333.
- BRADY, J. & SHERENI, W., 1988. Landing responses of the tsetse fly Glossina morsitans morsitans Westwood and the stable fly Stomoxys calcitrans (L.) (Diptera: Glossinidae & Muscidae) to black-and-white patterns: A laboratory study. Bulletin of Entomological Research, 78, 301–311.
- BRADY, J., 1975. Circadian changes in central excitability—the origin of behavioural rhythms in tsetse flies and other animals. Journal of Entomology (A), 50, 79–95.
- BRADY, J., 1975. Hunger in the tsetse fly: The nutritional correlates of behaviour. Journal of Insect Physiology, 21, 807–829.
- BRIGHTWELL, R., DRANSFIELD, R.D., KYORKY, C., GOLDER, T.K., TARIMO, S.A. & MUNGAI, D., 1987. A new trap for Glossina pallidipes. Tropical Pest Management, 33, 151–159.
- BRUCE, D., 1895. Preliminary Report on the Tsetse Fly Disease or Nagana, in KwaZulu Natal. Durban: Bennett and Davis.
- BRUCE, D., HAMERTON, A.E., BATEMAN, H.R. & MACKIE, F.P., 1909. The development of Trypanosoma gambiense in Glossina palpalis. Proceedings of the Royal Society (B), 81, 405–414.
- BUDD, L. 1999. DFID-funded tsetse and trypanosome research and development since 1980. Volume 2: Economic Analysis, Livestock Production Programme, NRInternational, Chatham Maritime.
- BURSELL, E. & TAYLOR, P., 1980. An energy budget for Glossina (Diptera: Glossinidae). Bulletin of Entomological Research, 70, 187–196.
- BURSELL, E., 1958. The water balance of tsetse pupae. Philosophical Transactions of the Royal Society, London (B), 241, 179–210.
- BURSELL, E., 1959. The water balance of tsetse flies. Transactions of the Royal Entomological Society, London, 111, 205–235.
- BURSELL, E., 1960. The effect of humidity and temperature on the extent of abdominal pigmentation in Glossina pallidipes Austen. Bulletin of Entomological Research, 51, 39–46.
- BURSELL, E., 1960. The effect of temperature on the consumption of fat during pupal development in Glossina. Bulletin of Entomological Research, 51, 583–598.
- BURSELL, E., 1966. Aspects of the flight metabolism of tsetse flies (Glossina). Comparative Biochemistry and Physiology, 19, 809–818.
- BURSELL, E., 1977. Synthesis of proline by fat body of the tsetse fly (Glossina orsitans): Metabolic pathways. Insect Biochemistry, 7, 427–434.
- BURSELL, E., 1987. The effect of wind-borne odours on the direction of flight in tsetse flies, Glossina spp. Physiological Entomology, 12, 149–156.
- BUXTON, P. A., 1955. The Natural History of Tsetse Flies. An Account of the Biology of the Genus Glossina (Diptera). Lewis, H.K. & Co Ltd, London, 10.
- CARMICHAEL, J., 1933. The virus of rinderpest and its relation to Glossina morsitans Westw. Bulletin of Entomological Research, 24, 337–342.
- CARMICHAEL, J., 1938. Rinderpest in African game. Journal of Comparative Pathology and Therapeutics, 51, 264–268.
- CARO, T., IZZO, A., REINER, J.R., WALKER, H. & STANKOWICH, T., 2014. The function of zebra stripes. Nature communications 5.
- CARTER, R.M., 1906. Tsetse fly in Arabia. British Medical Journal, 2, 1393–1394.
- CATTAND, P., JANNIN, J. & LUCAS, P., 2001. Sleeping sickness surveillance: an essential step towards elimination. Tropical medicine & international health 6: 348-361.
- CECCHI, G., PAONE, M., HERRERO, R.A., VREYSEN, M.J.B. & MATTIOLI, R.C., 2015. Developing a continental atlas of the distribution and trypanosomal infection of tsetse flies (Glossina species). Parasites & Vectors, 8, 284.
- CHALLIER, A. & LAMBRECHT, F.L., 1985. A new morphological character of superior claspers to differentiate Glossina pallidipes Austen from Glossina longipalpis Widemann (Diptera: Glossinidae). Insect Science and its Application, 6, 719–721.
- CHALLIER, A. & LAVEISSIÉRE, C., 1973. Un nouveau piège pour la capture de glossines (Glossina: Diptera, Muscidae): Description et essais sur le terrain. Cahiers de l’Office de la Recherch Scientifique et Technique Outre-Mer, Série Entomologie médicale et Parasitologie, 11, 251–262.
- CHALLIER, A., 1965. Amélioration de la méthode de détermination de l’âge physiologique des glossines. Bulletin de la Société de Pathologie Exotique, 58, 250–259.
- CHAPMAN, N.G., 1976. Aerial spraying of tsetse flies (Glossina spp.) in Rhodesia with ultra low volumes of endosulfan. Transactions of the Rhodesia Scientific Association, 57, 12–21.
- CHIKOWORE, G., DICKO, A.H., CHINWADA, P., ZIMBA, M., SHERENI, W., ROGER, F., BOUYER, J. & GUERRINI, L., 2017. A pilot study to delimitate tsetse target populations in Zimbabwe. PloS Negl. Trop. Dis. 11: e0005566.
- CHORLEY, J.K., 1929. Experiments in grass fires against Glossina morsitans in Southern Rhodesia. Bulletin of Entomological Research, 20, 377–390.
- CHORLEY, J.K., 1929. The bionomics of Glossina morsitans in the Umniati fly belt, Southern Rhodesia, 1922–1923. Bulletin of Entomological Research, 20, 279–301.
- CHORLEY, J.K., 1938. A short historical account of tsetse in Southern Rhodesia. Proceedings and Transactions of the Rhodesia Scientific Association, 36, 41–51.
- CLAUSEN, P. H., ADEYEMI, I., BAUER, B., BRELOEER, M., SALCHOW, F. & STAAK, C., 1998. Host preferences of tsetse (Diptera : Glossinidae) based on bloodmeal identifications. Med. Vet. Entomol. 12: 169-180.
- CLUTTON-BROCK, T. & LANGLEY, P.A., 1997. Persistent courtship reduces male and female longevity in captive tsetse flies Glossina morsitans morsitans Westwood (Diptera: Glossinidae). Behavioral Ecology 8: 392-395.
- COCKBILL, G.F., 1961. Further investigations into the role of a chemical brush killer in controlling regrowth on the eastern border clearing, Chipinga District. Rhodesia Agricultural Journal, 58, 370–373.
- COCKBILL, G.F., 1961. Preliminary investigations on the use of a chemical brush killer in controlling regrowth on the eastern border clearing— Chipinga Districts. Rhodesia Agricultural Journal, 58, 173–177.
- COCKBILL, G.F., 1979. The effect of ultra-low-volume aerial applications of endosulfan applied against Glossina (Diptera: Glossinidae) on populations of non-target organisms in savanna woodland in Zimbabwe-Rhodesia. Bulletin of Entomological Research, 69, 645–655.
- COCKBILL, G.F., LOVEMORE, D.F. & PHELPS, R.J., 1963. The control of tsetse flies (Glossina: Diptera, Muscidae) in a heavily infested area of Southern Rhodesia by means of insecticide discharged from aircraft, followed by settlement of indigenous people. Bulletin of Entomological Research, 54, 93–106.
- COLVIN, J. & GIBSON, G., 1992. Host-seeking behaviour and management of tsetse. Annual Review of Entomology, 37, 21–40.
- COURTIN, F., CAMARA, M., RAYAISSE, J.B., KAGBADOUNO, M., DAMA, E., CAMARA, O., TRAORÉ, I.S., ROUAMBA, J., PEYLHARD, M., SOMDA, M.B. & LENO, M., 2015. Reducing human-tsetse contact significantly enhances the efficacy of sleeping sickness active screening campaigns: a promising result in the context of elimination. PloS Negl. Trop. Dis. 9: e0003727.
- CUISANCE, D. & ITARD, J., 1973a. Comportement de mâles stériles de Glossina tachinoides West. lâchés dans les conditions naturelles - environs de Fort-Lamy (Tchad). II. Longévité et dispersion. La Revue d'élevage et de médecine vétérinaire des pays tropicaux, 26,169-186.
- CUISANCE, D. & ITARD, J., 1973b. Lâchers de mâles stériles de Glossina tachinoides West. dans un gîte naturel de faible densité (Bas-Logone, Cameroun). La Revue d'élevage et de médecine vétérinaire des pays tropicaux, 26, 405-422.
- CUISANCE, D. & POLITZAR, H., 1983. Etude de l'efficacité contre Glossina palpalis gambiensis et Glossina tachinoides de barrières constituées d'écrans ou de pièges biconiques imprégnés de DDT, de Deltaméthrine ou de Dieldrine. La Revue d'élevage et de médecine vétérinaire des pays tropicaux, 36, 159-168.
- CUISANCE, D., 1989. Le piegeage des tse-tse. Etudes et Syntheses de L’Institute d’elevage et de Médecine Veterinaire des Pays Tropicaux, 32.
- CUISANCE, D., POLITZAR, H., CLAIR, M., SELLIN, E., TAZÉ, Y., BOURDOISEAU, G. & FÉVRIER, J., 1980. La lutte contre Glossina palpalis gambiensis Vanderplank, par lâchers de mâles irradiés en Haute-Volta-Étude de paramètres opérationnels. Proceedings of the Symposium on Isotope and Radiation Research on Animal Diseases and their Vectors, Vienna: International Atomic Energy Agency.
- CUISANCE, D., POLITZAR, H., FEVRIER, J., BOURDOISEAU, G. & SELLIN, E., 1980. Association d'un traitement insecticide avec la méthode du mâle stérile contre Glossina palpalis gambiensis: intérêt de la mise en oeuvre de plusieurs méthodes. La Revue d'élevage et de médecine vétérinaire des pays tropicaux, 33, 127-133.
- CUISANCE, D., POLITZAR, H., MEROT, P. & TAMBOURA, I., 1984. Les lâchers de mâles irradiés dans la campagne de lutte intégrée contre les glossines dans la zone pastorale de Sidéradougou, Burkina Faso. La Revue d'élevage et de médecine vétérinaire des pays tropicaux, 37, 449-468.
- CURTIS, C.F. & HILL, W.G., 1971. Theoretical studies on the use of translocations for the control of tsetse flies and other disease vectors. Theoretical Population Biology, 2, 71–90.
- CURTIS, C.F., 1968. Some observations on reproduction and the effects of radiation on Glossina austeni. Transactions of the Royal Society of Tropical Medicine and Hygiene, 62, 124.
- DA SILVA, J.M. & PIRES, F.A., 1960. Subsidios para o estudo des Glossina do Ultrimar português. Sobre a morfologia das armadura genital da G. pallidipes Austen, 1903 (Diptera). Boletin Sociedade de Estudos, Moçambique, 29, No. 125, Section D, No. 3.
- DAME, D.A. & SCHMIDT, C.H., 1970. The sterile-male technique against tsetse flies. Bulletin of the Entomological Society of America, 16, 24–30.
- DAVIES, J.E. & BOWLES, J., 1979. Effects of large-scale aerial applications of endosulfan on tsetse fly, Glossina morsitans centralis Machado, in Botswana. Centre for Overseas Pest Research, London, Miscellaneous Report, 45.
- DAVIES, J.E., 1980. The History of Tsetse Fly Control in Botswana. Roneoed Report to the Director of Veterinary Services, Botswana.
- DAVIES, J.E., 1983. The distribution of Glossina morsitans in Botswana from historic to recent times. International Scientific Council for Trypanosomiasis Research and Control, Arusha Meeting, 1981.
- DE BEER, C. J., VENTER, G.J., GREEN, K.K., ESTERHUIZEN, J., DE KLERK, D.G., NTSHANGASE, J., VREYSEN, M.J.B., PIENAAR, R., MOTLOANG, M. & NTANTISO, L., 2016. An update of the tsetse fly (Diptera: Glossinidae) distribution and African animal trypanosomosis prevalence in north-eastern KwaZulu-Natal, South Africa. Onderstepoort J. Vet. Res. 83: 1-10.
- DEAN, G.J.W., WILLIAMSON, B.R. & PHELPS, R.J., 1969. Behavioural studies of Glossina morsitans Westw. using tantalum-182. Bulletin of Entomological Research, 58, 763–771.
- DENLINGER, D.L., 1975. Insect hormones as tsetse abortifacients. Nature, 253, 347–348.
- DESQUESNES, M. & DIA, M.L., 2003a. Trypanosoma vivax: mechanical transmission in cattle by one of the most common African tabanids, Atylotus agrestis. Experimental Parasitology, 103, 35-43.
- DESQUESNES, M. & DIA, M.L., 2003b. Mechanical transmission of Trypanosoma congolense in cattle by the African tabanid Atylotus agrestis. Experimental Parasitology, 105, 226-231.
- DESQUESNES, M., 2004. Livestock trypanosomoses and their vectors in Latin America. WAHO/OIE – CIRAD, OIE Paris.
- DESQUESNES, M., BITEAU-COROLLER, F., BOUYER, J., DIA, M.L. & FOIL, L.D., 2009. Development of a mathematical model for mechanical transmission of trypanosomes and other pathogens of cattle transmitted by tabanids. International Journal for Parasitology, 39, 333-346.
- DIALL, O., CECCHI, G., WANDA, G., ARGILÉS-HERRERO, R., VREYSEN, M.J.B., CATTOLI, G., VILJOEN, G.J., MATTIOLI, R. & BOUYER, J., 2017. Developing a Progressive Control Pathway for African Animal Trypanosomosis. Trends in Parasitology,33, 499-509.
- DIAS, J.A. & SANTOS, T., 1956. Descriçao de uma nova subespécie de tsé-tsé de Moçambique Glossina austeni mossurizensis n.ssp. Documentario Moçambique, 88, 51–78.
- DIAS, J.A. & SANTOS, T., 1962. The status of the tsetse fly in Mozambique before 1896. South African Journal of Science, 58, 243–247.
- DIAS, J.A. & SANTOS, T., 1987. Contribuiçao para o estudio da sistemática do genero Glossina Wiedemann, 1830 (Insecta, Brachycera, Cyclorrhapha, Glossinidae) proposta para a criçao de um novo subgénero. Garcia de Orta, Série Zoology, Lisboa, 14, 67–78.
- DIAS, J.A. TRAVASSOS SANTOS, 1987. Consideraçoes sobre o controlo da mosca tsé-tsé em Moçambique. Garcia de Orta, Série Zoology, Lisboa, 14, 61–66.
- DICKO, A. H., LANCELOT, R., SECK, M.T., GUERRINI, L., SALL, B., LO, M., VREYSEN, M.J.B., LEFRANÇOIS, T., WILLIAMS, F., PECK, S.L. & BOUYER, J., 2014. Using species distribution models to optimize vector control: the tsetse eradication campaign in Senegal. Proceedings of the National Academy of Science, USA, 111, 10149-10154.
- DICKO, A. H., PERCOMA, L., SOW, A., ADAM, Y., MAHAMA, C., SIDIBÉ, I., DAYO, G.K., THÉVENON, S., FONTA, W., SANFO, S., DJITEYE, A., SALOU, E., DJOHAN, V., CECCHI, G. & BOUYER, J., 2015. A Spatio-temporal Model of African Animal Trypanosomosis Risk. PLoS Neglected Tropical Diseases, 9, e0003921.
- DOUTHWAITE, R.J. & TINGLE, C.C.D., EDS., 1994. DDT in the tropics. The impact on wildlife in Zimbabwe of ground-spraying for tsetse fly control. Natural Resources Institute, Chatham, UK. ISBN 085954 3641.
- DOUTHWAITE, R.J., FOX, P.J., MATTHIESSEN, P. & RUSSELL-SMITH, A., 1981. Environmental Impact of Aerosols of Endosulfan, Applied for Tsetse Fly Control in the Okavango Delta, Botswana. Final Report of the Endosulfan Monitoring Project, Overseas Development Administration, London.
- DRANSFIELD, R.D. & BRIGHTWELL, R. 1992. Training manual for tsetse control personnel. Vol. 4. Use of attractive devices for tsetse survey and control. Food and Agriculture Organisation of the United Nations, Rome, 4.
- DU TOIT, R., 1954. Trypanosomiasis in KwaZulu Natal and the control of tsetse flies by chemical means. Onderstepoort Journal of Veterinary Research, 26, 317–387.
- DYCK, V. A., PAN, H., KASSIM, S.S., SULEIMAN, F.W., MUSSA, W.A., SALEH, K.M., JUMA, K.G., MKONYI, P.A., HOLLAND, W.G., VAN DER EERDEN, B.J.M. & DWINGER, R.H., 2000. Monitoring the incidence of trypanosomosis in cattle during the release of sterilized tsetse flies on Unguja Island, Zanzibar. La Revue d'élevage et de médecine vétérinaire des pays tropicaux, 53, 239-243.
- ELITH, J., PHILLIPS, S.J., HASTIE, T., DUDÍK, M., CHEE, Y.E. & YATES, C.J., 2011. A statistical explanation of MaxEnt for ecologists. Diversity & Distributions Journal, 17, 43-57.
- ELSEN, P., AMOUDI, M.A. & LECLERCQ, M., 1990. Première découverte de deux espèces de Glossines en Arabie Saoudite. Résumés. Troisième Conférence internationale des Entomoligistes d’expression française. Gembloux, 9–14 Juillet 1990. Commauté française de Belgique, Faculté des sciences agronomique de Gembloux, Gembloux, Belgium.
- ENKERLIN, W., GUTIÉRREZ-RUELAS, J.M., CORTES, A.V., ROLDAN, E.C., MIDGARDEN, D., LIRA, E., LÓPEZ, J.L.Z., HENDRICHS, J., LIEDO, P. & ARRIAGA, F.J.T., 2015. Area Freedom in Mexico from Mediterranean Fruit Fly (Diptera: Tephritidae): A Review of Over 30 Years of a Successful Containment Program Using an Integrated Area-Wide SIT Approach. Florida Entomologist, 98, 665-681.
- ESTERHUIZEN, J., RAYAISSE, J.B., TIRADOS, I., MPIANA, S., SOLANO, P., VALE, G.A., LEHANE, M.J. & TORR, S.J., 2011. Improving the cost-effectiveness of visual devices for the control of riverine tsetse flies, the major vectors of human African trypanosomiasis. PLoS Neglected Tropical Diseases, 5, e1257.
- EVISON, C. & KATHURIA, K.D.S., 1982. A Survey of the Distribution of Glossina spp. and Factors Influencing their Control in the Territory of Northern Rhodesia (Zambia). Ministry of Agriculture and Water Development, Republic of Zambia.
- FARAI, M., 2013. Modelling the distribution of suitable Glossina subspecies habitat in the North Western parts of Zimbabwe using remote sensing and climate data. Geoinformatics & Geostatistics: An Overview, S1, 016.
- FARIKOU, O., NJIOKOU, F., MBIDA, J.A.M., NJITCHOUANG, G.R., DJEUNGA, H.N., ASONGANYI, T., SIMARRO, P.P., CUNY, G. & GEIGER, A., 2010. Tripartite interactions between tsetse flies, Sodalis glossinidius and trypanosomes—an epidemiological approach in two historical human African trypanosomiasis foci in Cameroon. Infection, Genetics & Evolution Journa, 10, 115-121.
- FELDMANN, H. U., LEAK, S. & HENDRICHS, J., 2016. Assessing the feasibility of creating tsetse and trypanosomosis free zones. International Journal of Tropical Insect Science, submitted.
- FELDMANN, U., DYCK, V.A., MATTIOLI, R.C. & JANNIN, J.J., 2005. Potential impact of tsetse fly control involving the sterile insect technique, pp. 701-723. In: Dyck, V.A., Hendrichs, J. & Robinson, A.S., (eds.). Sterile insect technique. Principles and practice in area-wide integrated pest management. Springer, Dordrecht, The Netherlands.
- FILLEDIER, J., & MÉROT, P., 1989. Etude de l'attractivité de solutions isolées par fractionnement de l'urine de bovin Baoulé pour Glossina tachinoides Westwood, 1850 au Burkina Faso. Revue d’élevage et de médecine vétérinaire des pays tropicaux, 42, 453-455.
- FILLEDIER, J., & MEROT, P., 1989. Pouvoir attractif de l'association M-Crésol 1-Octen-3-Ol dans un type de diffuseur pratique pour Glossina tachinoides au Burkina Faso. Revue d’élevage et de médecine vétérinaire des pays tropicaux, 42, 541-544.
- FLINT, S., 1985. A comparison of various traps for Glossina spp. (Glossinidae) and other Diptera. Bulletin of Entomological Research, 37, 529–534.
- FORD, J. & KATONDO, K.M., 1977. Maps of tsetse fly (Glossina) distribution in Africa, 1973 according to sub-generic groups on a scale of 1:5 000 000. Bulletin of Animal Health and Production, 25, 187–193.
- FORD, J. 1971. The role of the trypanosomiases in African ecology. A study of the tsetse fly problem. Oxford: Clarendon Press.
- FORD, J., 1966. The role of elephants in controlling the distribution of tsetse flies. Bulletin of the International Union for the Conservation of Nature, N.S., 19, 6.
- FULLER, C. & MOSSOP, M.C., 1929. Entomological notes on Glossina pallidipes. Union of South Africa, Department of Agriculture, Science Bulletin No. 67.
- FULLER, C., 1923. Tsetse in the Transvaal and surrounding territories. An historical review. Entomology Memoir 1, Department of Agriculture, Union of South Africa.
- GALEY, J. B., MEROT, P., MITTEAULT, A., FILLEDIER, J. & POLITZAR, H., 1986. Efficacité du dioxyde de carbone comme attractif pour Glossina tachinoides en savane humide d'Afrique de l'Ouest. Revue d’élevage et de médecine vétérinaire des pays tropicaux, 39, 351-354.
- GALUN, R. & MARGALIT, J., 1969. Adenine nucleotides as feeding stimulants of the tsetse fly Glossina austeni Newst. Nature, 222, 583–584.
- GASTON, K.A. & RANDOLPH, S.E., 1993. Reproductive under-performance of tsetse flies in the laboratory, related to feeding frequency. Physiological Entomology, 18, 130–136.
- GIBSON, G. & BRADY, J., 1988. Flight behaviour of tsetse flies in host odour plumes: The initial response to leaving or entering odour. Physiological Entomology, 13, 29–42.
- GIBSON, W.C., DUKES, P. & GASHUMBA, J.K., 1988. Species-specific DNA probes for the identification of African trypanosomes in tsetse flies. Parasitology, 97, 63–73.
- GIMONNEAU, G., ALIOUM, Y., ABDOULMOUMINI, M., ZOLI, A., CENE, B., ADAKAL, H. & BOUYER, J., 2016. Insecticide and Repellent Mixture Pour-On Protects Cattle against Animal Trypanosomosis. PLoS Neglected Tropical Diseases, 10, e0005248.
- GLASGOW, J.P., 1963. The Distribution and Abundance of Tsetse. London: Pergamon Press.
- GLOVER, P.E., JACKSON, C.H.N., ROBERTSON, A.G. & THOMSON, W.E.F., 1955. The extermination of the tsetse fly, Glossina morsitans Westw., at Abercorn, Northern Rhodesia. Bulletin of Entomological Research, 46, 57–67.
- GOODIER, R., 1961. Discriminative bush-clearing against tsetse in the south-east of Southern Rhodesia. Rhodesia Agricultural Journal, 58, 262–266.
- GOODING, R.H., 1982. Classification of nine species and subspecies of tsetse flies (Diptera: Glossinidae: Glossina Wiedemann) based on molecular genetics and breeding data. Canadian Journal of Zoology, 60, 2737–2744.
- GOODING, R.H., 1984. Tsetse genetics—A review. Questiones Entomologicae, 20, 89–128.
- GOODING, R.H., 1989. Genetics of two populations of Glossina morsitans centralis (Diptera: Glossinidae) from Zambia. Acta Tropica, 46, 17–22.
- GOVERNMENT OF ZIMBABWE, MINISTRY OF AGRICULTURE AND LANDS., 1999. Final report of the Zimbabwe component of the first and second phases of the Regional Tsetse and Trypanosomiasis Control Programme Malawi, Mozambique, Zambia and Zimbabwe. Consultant’s report by D.F. Lovemore to the Tsetse and Trypanosomiasis Control Branch of the Department of Veterinary Services, Zimbabwe.
- GRAHAM, P., 1964. Destruction of birds and other wildlife by dieldrex spraying against tsetse fly in Bechuanaland. Arnoldia (Rhodesia), 1, 1–4.
- GREATHEAD, D.J., 1980. Biological control of tsetse flies: An assessment of insect parasitoids as control agents. Biocontrol News and Information, 1, 111–123.
- GREEN, C.H. & COSENS, D., 1983. Spectral responses of the tsetse fly, Glossina morsitans morsitans. Journal of Insect Physiology, 29, 795–780
- GREEN, C.H. & FLINT, S., 1986. An analysis of colour effects in the performance of the F2 trap against Glossina pallidipes Austen and G. morsitans morsitans Westwood (Diptera: Glossinidae). Bulletin of Entomological Research, 76, 409–418.
- GREEN, C.H., 1988. The effect of colour on trap- and screen-orientated responses in Glossina palpalis palpalis (Robineau-Desvoidy) (Diptera: Glossinidae). Bulletin of Entomological Research, 78, 591–604.
- GREEN, C.H., 1994. Bait methods for tsetse control. Advances in Parasitology, 34, 229–291.
- GRIFFITHS, N., PAYNTER, Q. & BRADY, J., 1995. Rates of progress up odour plumes by tsetse flies—a mark-release video study of the timing of odour source location by Glossina pallidipes. Physiological Entomology, 20, 100–108.
- GROENENDIJK, C.A., 1996. The behaviour of tsetse flies in an odour plume. PhD thesis. Landbouwuniversiteit Wageningen, Netherlands. ISBN: 90-5485-521-5.
- GUERRINI, L., BORD, J.P., DUCHEYNE, E. & BOUYER, J., 2008. Fragmentation analysis for prediction of suitable habitat for vectors: the example of riverine tsetse flies in Burkina faso. Journal of Medical Entomology, 45, 1180-1186.
- HAESELBARTH, E., SEGERMAN, J. & ZUMPT, F., 1966. The arthropod parasites of vertebrates in Africa south of the Sahara (Ethiopian Region). Vol. III. (Insecta excluding Phthiraptera). Publication of the South African Institute for Medical Research, 13, No. 52, 1–283.
- HALL, M.J.R. & LANGLEY, P.A., 1987. Development of a system for sterilizing tsetse flies, Glossina spp. in the field. Medical and Veterinary Entomology, 1, 201–210.
- HARGROVE, J.W. & LANGLEY, P.A., 1990. Sterilizing tsetse (Diptera: Glossinidae) in the field: a successful trial. Bulletin of Entomological Research, 80, 397–403.
- HARGROVE, J.W. & LANGLEY, P.A., 1993. A field trial of pyriproxyfen-treated targets as an alternative method for controlling tsetse (Diptera: Glossinidae). Bulletin of Entomological Research, 83, 361–368.
- HARGROVE, J.W., 1975. The flight performance of tsetse flies. Journal of Insect Physiology, 21, 1385–1395.
- HARGROVE, J.W., 1977. Some advances in the trapping of tsetse (Glossina spp.) and other flies. Ecological Entomology, 2, 123–137.
- HARGROVE, J.W., 1988. Tsetse: The limits to population growth. Medical and Veterinary Entomology, 2, 203–217.
- HARGROVE, J.W., 1993. Target barriers for tsetse flies (Glossina spp.) (Diptera: Glossinidae): quick estimates of optimal target densities and barrier widths. Bulletin of Entomological Research, 83, 197–200.
- HARGROVE, J.W., 1994. Reproductive rates of tsetse flies in the field in Zimbabwe. Physiological Entomology, 19, 307–318.
- HARGROVE, J.W., HOLLOWAY, M.T.P. & VALE, G.A., 1995. Catches of tsetse (Glossina spp.) (Diptera: Glossinidae) from traps and targets baited with large doses of natural and synthetic host odour.Bulletin of Entomological Research, 85, 215–217.
- HARRIS, R.H.T.P., 1930. Report on the Bionomics of the Tsetse Fly Glossina pallidipes Aust. and a Preliminary Report on a New Method of Control, Presented to the Provincinal Administration of Natal. Pietermaritzburg: Natal Witness Ltd.
- HARRIS, R.H.T.P., 1932. Some facts and figures regarding the attempted control of Glossina pallidipes in KwaZulu Natal. South African Journal of Science, 29, 495–507.
- HEAVERSEDGE, R.C., 1968. Variation in the size of insect parasites of puparia of Glossina spp. Bulletin of Entomological Research, 58, 153–158.
- HEAVERSEDGE, R.C., 1969. Insect parasites of Glossina pallidipes Aust. puparia in Rhodesia. Journal of the Entomological Society of Southern Africa, 32, 225–229.
- HEAVERSEDGE, R.C., 1969. Levels of insect parasitism in puparia of Glossina morsitans orientalis Vanderplank (Diptera) in Rhodesia. Journal of the Entomological Society of Southern Africa, 32, 231–235.
- HENDRICHS, J., VREYSEN, M.J.B., ENKERLIN, W.R. & CAYOL, J.P., 2005. Strategic Options in Using Sterile Insects for Area-Wide Integrated Pest Management. In: DYCK, V.A., HENDRICHS, J. & ROBINSON, A.S., (eds.). Sterile insect technique. Principles and practice in area-wide integrated pest management. Springer, Dordrecht, 563-600.
- HÖRETH-BÖNTGEN, D.W. Control of Glossina austeni and cattle trypanosomiasis in Unguja island by deltamethrin pour-on application to livestock and with stationary targets in cattle-free zones. In: Proceedings of an IAEA/FAO Seminar on Tsetse control, Diagnosis and Chemotherapy using Nuclear Techniques, 11 - 15 February 1991 1992, Muguga, Kenya. IAEA-TECDOC, 213-218.
- HORNBY, H.E., 1947. Report on a survey to ascertain if there be a danger that the ‘Glossina morsitans’ belt associated with the Rio Save may spread south-west to the Rio Limpopo and beyond. Actas da Conferência Inter-Colonial sobre Trypanossomiases. Lorenço Marques, 26th to 31st August, 1946. Lorenço Marques: Imprensa Nacional de Moçambique.
- HOUSE, A.P.R., 1982. Chemosterilization of Glossina morsitans morsitans Westwood and G. pallidipes Austen (Diptera: Glossinidae) in the field. Bulletin of Entomological Research, 72, 65–70.
- HURSEY, B. S. & SLINGENBERGH, J., 1995. The tsetse fly and its effects on agriculture in sub-Saharian Africa. Rev. Mond. Zoot. 84, 67-73.
- HURSEY, B.S. & ALLSOPP, R., 1983. Sequential application of low dosage aerosols from fixed-wing aircraft as a means of eradicating tsetse flies (Glossina subspecies) from rugged terrain in Zimbabwe. Tsetse and Trypanosomiasis Control Branch, Department of Veterinary Services, Zimbabwe.
- HURSEY, B.S. & ALLSOPP, R., 1984. The eradication of tsetse flies (Glossina subspecies) from western Zimbabwe by integrated aerial and ground spraying. Tsetse and Trypanosomiasis Control Branch, Department of Veterinary Services, Zimbabwe.
- HUYTON, P.M. & BRADY, J., 1975. Some effects of light and heat on the feeding and resting behaviour of tsetse flies, Glossina morsitans Westwood. Journal of Entomology (A), 50, 23–30.
- ITARD, J., CUISANCE, D. & TACHER, G., 2003. Trypanosomoses: Historique - Répartition géographique, pp. 1607-1615. In: LEFÈVRE, P.C., BLANCOU, J. & CHERMETTE, R., (eds.), Principales maladies infectieuses et parasitaires du bétail. Europe et Régions chaudes, Lavoisier, Paris, 2.
- JACK, R.W., 1914. Tsetse fly and big game in Southern Rhodesia. Bulletin of Entomological Research, 5, 97–110.
- JACK, R.W., 1926. Tsetse fly in the Lomagundi district. Bulletin No. 587, Department of Agriculture, Government of Southern Rhodesia.
- JACK, R.W., 1927. Some environmental factors relating to the distribution of Glossina morsitans Westw. in Southern Rhodesia. South African Journal of Science, 24, 457–475.
- JACK, R.W., 1933. Experiments with tsetse fly traps against Glossina morsitans in Southern Rhodesia. Bulletin No. 893, Ministry of Mines and Agriculture, Southern Rhodesia.
- JACK, R.W., 1933. The tsetse fly problem in Southern Rhodesia. Bulletin No. 892, Department of Agriculture, Government of Southern Rhodesia.
- JACK, R.W., 1939. Studies in the Physiology and Behaviour of Glossina morsitans Westw. Memoir of the Department of Agriculture 1. Salisbury, Government Printer.
- JACKSON, C. H. N., 1945. Comparative studies of the habitat requirements of tsetse fly species. The Journal of Animal Ecology, 46-51.
- JACKSON, C.H.N., 1933. On a method of marking tsetse flies. Journal of Animal Ecology, 2, 289–290.
- JACKSON, C.H.N., 1940. The analysis of a tsetse fly population. Annals of Eugenics, Cambridge, 10, 332–369.
- JACKSON, C.H.N., 1945. Pairing of Glossina morsitans Westwood with Glossina swynnertoni Austen (Diptera). Proceedings of the Royal Entomological Society, London (A), 20, 106.
- JACKSON, C.H.N., 1946. An artificially isolated generation of tsetse flies (Diptera). Bulletin of Entomological Research, 37, 291–299.
- JACKSON, C.H.N., 1948. Some further isolated generations of tsetse flies. Bulletin of Entomological Research, 39, 441–451.
- JACKSON, C.H.N., 1949. The biology of tsetse flies. Biological Reviews, 24, 174–199.
- JORDAN, A.M., 1958. The mating behaviour of females of Glossina palpalis (R.-D) in captivity. Bulletin of Entomological Research, 49, 35–43.
- JORDAN, A.M., 1976. Tsetse flies as vectors of trypanosomes. Veterinary Parasitology, 2, 143–152.
- JORDAN, A.M., 1986. Trypanosomiasis Control and African Rural Development. London: Longman.
- JORDAN, A.M., TREWERN, M.A., BORKOVEC, A.B. & DEMILO, A.B., 1979. Laboratory studies on the potential of three insect growth regulators for control of the tsetse fly, Glossina morsitans morsitans Westwood (Diptera: Glossinidae). Bulletin of Entomological Research, 69, 55–64.
- KAGBADOUNO, M. S., CAMARA, M., BOUYER, J., COURTIN, F., MORIFASO, O. & SOLANO, P., 2011. Tsetse control in Loos islands, Guinea. Parasites & Vectors, 4, 18.
- KAMUANGA, M., HAMADOU, S., BOUYER, J., YAO, Y., SIDIBÉ, I. & KABORÉ, I., 2005. Comment pérenniser les acquis de la lutte antivectorielle? Santé animale en Afrique de l'Ouest, Recommandations Techniques, CIRDES/CIRAD, 14, 8.
- KAMUANGA, M., SWALLOW, B.M., SIGUÉ, H. & BAUER, B., 2001. Evaluating contingent and actual contributions to a local public good: Tsetse control in the Yale agro-pastoral zone, Burkina Faso. Ecological economics, 39, 115-130.
- KAPPMEIER-GREEN, K. & VENTER, G.J., 2007. Evaluation and improvement of sticky traps as monitoring tools for Glossina austeni and Glossina brevipalpis (Diptera: Glossinidae) in north-eastern KwaZulu-Natal, South Africa. Bulletin of Entomological Research, 97, 545-553.
- KAPPMEIER-GREEN, K. 2000. A newly developped odour-baited "H trap" for the live collection of Glossina brevipalpis and Glossina austeni (Diptera: Glossinidae) in South Africa. Onderstepoort Journal of Veterinary Research, 67, 15-26.
- KAPPMEIER-GREEN, K., POTGIETER, F.T. & VREYSEN, M.J.B., 2007. A strategy for an area-wide control campaign with an SIT component to establish a tsetse-(Glossina austeni and Glossina brevipalpis) free South Africa. Area-wide control of insect pests, 309-323.
- KAPPMEIER, K., 1997. The development of odour-baited targets for the control of Glossina subspecies (Diptera: Glossinidae) in South Africa. MSc thesis, University of Pretoria, South Africa.
- KAPPMEIER, K., NEVILL, E.H. & BAGNALL, A.J., 1998. Review of tsetse flies and trypanosomosis in South Africa. Onderstepoort Journal of Veterinary Research, 65, 195–203.
- KGORI, P.M., MODO, S. & TORR, S.J., 2006. The use of aerial spraying to eliminate tsetse from the Okavango Delta of Botswana. Acta Tropica, 99, 184-199.
- KLASSEN, W., 2005. Area-wide integrated pest management and the sterile insect technique. In: DYCK, V.A., HENDRICHS, J. & ROBINSON, A.S., (eds.), Sterile insect technique. Principles and practice in area-wide integrated pest management. Springer, Dordrecht, The Netherlands, 39-68.
- KLUGE, E.B., 1975. Family Glossinidae. In: HOWELL, C.J., (ed.). Veterinary Entomology in southern Africa. Entomology Memoir 44, Department of Agricultural Technical Services, Republic of South Africa.
- KNIPLING, E. F., 1972. Entomology and the management of man's environment. Austral Entomology, 11, 153-167.
- KNIPLING, E.F., 1979. The basic principles of insect population suppression and management (Agricultural Handbook No. 512). Washington, D.C.: United States Department of Agriculture.
- KNIPLING, E.F., 1982. Present status and future trends of the SIT approach to the control of arthropod pests. Proceedings of the Symposium on the Sterile Insect Control. Vienna: International Atomic Energy Agency.
- KRISTJANSON, P. M., SWALLOW, B.M., RAWLANDS, G.J., KRUSKA, R.L. & LEEUW, P.N., 1999. Measuring the cost of African Animal Trypanosomiasis, the potential benefits of control and returns to research. Agricultural Systems, 59, 79-98.
- LAMBORN, W.A., 1925. An attempt to control Glossina morsitans by means of Syntomosphyrum glossinae Waterston. Bulletin of Entomological Research, 15, 303–310.
- LAMBORN, W.A., 1938. Annual Report of the Medical Entomologist for 1937. Annual Medical and Sanitary Report, Nyasaland, 1938. LANCIEN, J., 1981. Description du piège monoconique utilisé pour l’élimination des golossines en Republique Populaire du Congo. Cahiers de l’Office de la Recherch Scientifique et Technique Outre-Mer, série Entomologie Médicale et Parasitologie, 19, 235–238.
- LANCIEN, J., 1981. Description du piège monoconique utilisé pour l’élimination des golossines en Republique Populaire du Congo. Cahiers de l’Office de la Recherch Scientifique et Technique Outre-Mer, série Entomologie Médicale et Parasitologie, 19, 235–238.
- LANGLEY, P. A., FELTON, T., STAFFORD, K. & OOUCHP, H., 1990. Formulation of pyriproxyfen, a juvenile hormone mimic, for tsetse control. Medical and Veterinary Entomology, 4, 127-133.
- LANGLEY, P.A. & ROE, J.M., 1984. Ivermectin as a possible control agent for the tsetse fly, Glossina morsitans. Entomologia Experimentalis et Applicata, 36, 137–143.
- LANGLEY, P.A. & WEIDHAAS, D., 1986. Trapping as a means of controlling tsetse, Glossina subspecies (Diptera: Glossinidae): The relative merits of killing and of sterilization. Bulletin of Entomological Research, 76, 89–95.
- LANGLEY, P.A., 1968. The effect of feeding the tsetse fly Glossina morsitans Westwood on impala. Bulletin of Entomological Research, 58, 295–298.
- LANGLEY, P.A., 1977. Physiology of tsetse flies (Glossina spp.) (Diptera: Glossinidae): A review. Bulletin of Entomological Research, 64, 523–574.
- LANGLEY, P.A., 1995. Evaluation of the chitin synthesis inhibitor triflumuron for controlling tsetse Glossina morsitans morsitans (Diptera: Glossinidae). Bulletin of Entomological Research, 84, 495–500.
- LANGLEY, P.A., FELTON, T. & OOUCHI, H., 1988. Juvenile hormone mimics as effective sterilants for the tsetse fly Glossina morsitans morsitans. Medical and Veterinary Entomology, 2, 29–35.
- LANGLEY, P.A., HALL, M.J.R., FELTON, T. & CEESAY, M., 1988. Determining the age of tsetse flies, Glossina subspecies (Diptera: Glossinidae): An appraisal of the pteridine fluorescence technique. Bulletin of Entomological Research, 78, 387–395.
- LANGLEY, P.A., HUYTON, P.M. & CARLSON, D.A., 1987. Sex pheromone perception by males of the tsetse fly, Glossina morsitans morsitans. Physiological Entomology, 12, 425–433.
- LANGLEY, P.A., MAUDLIN, I. & LEEDHAM, M.P., 1984. Genetic and behavioural differences between Glossina pallidipes from Uganda and Zimbabwe. Entomologia Experimentalis et Applicata, 35, 55–60.
- LAVEISSIÈRE, C. & COURET, D., 1981. Essai de lutte contre les glossines riveraines à l’aide d’ecrans imprégnés d’insecticide. Cahiers de l’Office de la Recherch Scientifique et Technique Outre-Mer, série Entomologie Médicale et Parasitologie, 19, 271–283.
- LAVEISSIÈRE, C. & GRÉBAUT, P., 1990. Recherches sur les pièges à glossines (Diptera, Glossinidae). Mise au point d'un modèle économique : le piège "Vavoua". Tropical Medicine and Parasitology. 41, 185-192.
- LAVEISSIÈRE, C., COURET, D. & KIÉNON, J.P., 1981. Essai de lutte contre les glossines riveraines à l’aide de pièges biconiques imprégnés d’insecticide, en zone de savane humide. 5. Note de synthèse. Cahiers de l’Office de la Recherch Scientifique et Technique Outre-Mer, série Entomologie Médicale et Parasitologie, 19, 49–54.
- LEAK, S. G. A., 1995. A trial of a cypermethrin ’pour-on’ insecticide to control Glossina pallidipes, Glossina fuscipes fuscipes and Glossina morsitans submorsitans (Diptera: Glossinidae) in south-west Ethiopia. Bulletin of Entomological Research, 85, 241-251.
- LEAK, S. G. A., 1999. Tsetse biology and ecology. Their role in the epidemiology and control of trypanosomosis. Oxford: CABI Publishing.
- LEAK, S. G. A., EJIGU, D. & VREYSEN, M.J.B., 2008. Collection of entomological baseline data for tsetse area-wide integrated pest management programmes. FAO Animal Production and Health Guidelines. Food and Agriculture Organization of the United Nations, Rome, Italy.
- LEAK, S. G. A., PEREGRINE, A.S., MULATU, W., ROWLANDS, G.J. & D’LETEREN, G., 1996. Use of insecticide-impregnated targets for the control of tsetse flies (Glossina spp.) and trypanosomiasis occurring in cattle in an area of south-west Ethiopia with a high prevalence of drug-resistant trypanosomes. Tropical Medicine & International Health, 1, 599-609.
- LEAK, S., 1998. Tsetse biology and ecology. Their role in the epidemiology and control of trypanosomosis. CABI publishing with ILRI, Oxon (UK), 568.
- LINDH, J. M., TORR, S.J., VALE, G.A. & LEHANE, M.J., 2009. Improving the Cost-Effectiveness of Artificial Visual Baits for Controlling the Tsetse Fly Glossina fuscipes fuscipes. PLoS Neglected Tropical Diseases, 3, e474.
- LINDQUIST, D. A., ABUSOWA, M. & HALL, M.J., 1992. The New World screwworm fly in Libya: a review of its introduction and eradication. Tropical Medicine and Parasitology, 6, 2-8.
- LOVEMORE, D.F., 1972. Annual report of the Branch of Tsetse and Trypanosomiasis Control, Department of Veterinary Services, Ministry of Agriculture, Rhodesia, for the year ended 30th September, 1971. Government Printing and Stationery, Salisbury, 65996-2-300.
- LOVEMORE, D.F., 1973. Annual report of the Branch of Tsetse and Trypanosomiasis Control, Department of Veterinary Services, Ministry of Agriculture, Rhodesia, for the year ended 30th September, 1972. Government Printing and Stationery, Salisbury, 68789-6-300.
- LOVEMORE, D.F., 1974. A note on tsetse and trypanosomiasis control in Rhodesia during the year ended 30th September, 1974. Type-written internal report prepared for the Director of Veterinary Services.
- MACHADO, A. DE B., 1954. Revision systématique des Glossines du groupe palpalis (Diptera). Companhia de Diamantes de Angola, Publicaçoes Culturais, 22.
- MACHADO, A. DE B., 1970. Les races géographiques de Glossina morsitans. First International Symposium on Tsetse Fly Breeding under Laboratory Conditions and its Practical Application. 22–23 April 1969. Junta de investigaçoes do ultrimar. Lisboa: Portugal.
- MACKINNON, J., 1964. Report of the Secretary to the Federal Minister of Agriculture for the Year Ended 30th September, 1963. Government Printer, Salisbury.
- MACLENNAN, K.J.R. & KIRBY, W.W., 1958. The eradication of Glossina morsitans submorsitans Newst. in part of a river flood plain in Northern Nigeria by chemical means. Bulletin of Entomological Research, 49, 123–131.
- MACLENNAN, K.J.R., 1975. A review of operations against tsetse flies and trypanosomiasis in Zambia and recommendations for the future. Cyclostyled report. Ministry of Agriculture and Water Development, Republic of Zambia.
- MADUBUNYI, L. C., 1990. Ecological studies of Glossina austeni at Jozani forest, Unguja island, Zanzibar. Insect Science and its Application, 11, 309–313.
- MAGADZA, C. H. D., 1978. Field observations on the environmental effect of large-scale applications of endosulfan in the eradication of Glossina morsitans morsitans Westw. in the western province of Zambia in 1968. Rhodesian Journal of Agricultural Research, 16, 211–220.
- MAHAMAT, M. H., PEKA, M., RAYAISSE, J.B., ROCK, K.S., TOKO, M.A., DARNAS, J., BRAHIM, G.M., ALKATIB, A.B., YONI, W., TIRADOS, I., COURTIN, F., BRAND, S.P.C., NERSY, C., ALFAROUKH, I.O., TORR, S.J, LEHANE, M.J. & SOLANO. P., 2017. Adding tsetse control to medical activities contributes to decreasing transmission of sleeping sickness in the Mandoul focus (Chad). PLoS Neglected Tropical, Diseases. 11, e0005792.
- MARKS, A. D., 1966. Notes on the behaviour of tsetse flies (G. morsitans and G. pallidipes) with regard to impala. Interim report of the Rhodesia Government, Department of Veterinary Services, Tsetse and Trypanosomiasis Control Branch.
- MATTHEISSEN, P., 1985. Contamination of wildlife with DDT insecticide residues in relation to tsetse fly control operations in Zimbabwe. Environmental Pollution (B), 10, 189–211.
- MATTIOLI, R. C., FELDMANN, U., HENDRICKX, G., WINT, W., JANNIN, J. & SLINGENBERGH, J., 2004. Tsetse and trypanosomiasis intervention policies supporting sustainable animal-agricultural development. Journal of Food Agriculture and Environment, 2, 310-314.
- MAUDLIN, I. & ELLIS, D. S., 1985. Association between intracellular rickettsial-like infections of midgut cells and susceptibility to trypanosome infection in Glossina spp. Zeitschrift fu¨r Parasitenkunde, 71, 683–687.
- MAUDLIN, I. & WELBURN, S. C., 1987. Lectin mediated establishment of midgut infections of Trypanosoma congolense and Trypanosoma brucei in Glossina morsitans. Tropical Medicine and Parasitology, 38, 167–170.
- MAUDLIN, I. & WELBURN, S. C., 1988. Tsetse immunity and the transmission of trypanosomiasis. Parasitology Today, 4, 109–111.
- MAUDLIN, I., 1982. Inheritance of susceptibility to Trypanosoma congolense infection in Glossina morsitans. Annals of Tropical Medicine and Parasitology, 76, 225–227.
- MELLANBY, H., 1937. Experimental work on reproduction in the tsetse fly, Glossina palpalis. Parasitology, 29, 131–141.
- MEROT, P., FILLEDIER, J. & MULATO, C., 1988. Pouvoir attractif, pour Glossina tachinoides, de produits chimiques isolés des odeurs animales. Revue d’élevage et de médecine vétérinaire des pays tropicaux, 41, 79-85.
- MEWS, A. R., LANGLEY, P. A., PIMLEY, R. T. W. & FLOOD, M. E. T., 1977. Large-scale rearing of tsetse flies (Glossina spp.) in the absence of a living host. Bulletin of Entomological Research, 67, 119–128.
- MOLOO, S. K. & DAR, F., 1985. Probing by Glossina morsitans centralis infected with pathogenic Trypanosoma species. Transactions of the Royal Society of Tropical Medicine and Hygiene, 79, 119.
- MOLOO, S. K., 1985. Distribution of Glossina species in Africa. Acta Tropica, 42, 275–281.
- MOLOO, S. K., 1993. The distribution of Glossina species in Africa and their natural hosts. International Journal of Tropical Insect Science, 14, 511-527.
- MOLOO, S. K., GROOTENHUIS, J. G., KAR, S. K. & KARSTAD, L., 1988. Survival and reproductive performance of female Glossina morsitans morsitans when maintained on the blood of different species of wild mammals. Medical and Veterinary Entomology 2, 347–350.
- MOLYNEUX, D. H. & JENNI, L., 1981. Mechanoreceptors, feeding behaviour and trypanosome transmission in Glossina. Transactions of the Royal Society of Tropical Medicine and Hygiene, 75, 160–163.
- MOLYNEUX, D. H., 1977. Vector relationships in the Trypanosomatidae. Advances in Parasitology, 15, 1–82.
- MORTON, J., 2010. The innovation trajectory of sleeping sickness control in Uganda: Research knowledge in its context. RIU discussion paper, 8, 43.
- MUBARQUI, R. L., PEREZ, R. C., ANGULO KLADT, R., ZAVALA LOPEZ, J. L., PARKER, A., SECK, M. T., SALL, B. & BOUYER, J., 2014. The smart aerial release machine, a universal system for applying the sterile insect technique. PLoS ONE, 9, e103077.
- MULLIGAN, H. W., 1970. The African Trypanosomiases. London: George Allen and Unwin Ltd.
- MUTIKA, G. N., KABORE, I., PARKER, A. G. & VREYSEN, M. J. B., 2014. Storage of male Glossina palpalis gambiensis pupae at low temperature: effect on emergence, mating and survival. Parasites & vectors, 7, 465.
- MUTIKA, G. N., KABORE, I., SECK, M. T., SALL, B., BOUYER, J., PARKER, A. G. & VREYSEN, M. J. B., 2013. Mating performance of Glossina palpalis gambiensis strains from Burkina Faso, Mali and Senegal. Entomology Experimentalis et Applicata, 146, 177-185.
- MUTURI, C. N., OUMA, J.O., MALELE, I.I., NGURE, R.M., RUTTO, J.J., MITHÖFER, K.M., ENYARU, J.C.K. & MASIGA, D.K., 2011. Tracking the feeding patterns of tsetse flies (Glossina genus) by analysis of bloodmeals using mitochondrial cytochromes genes. PLoS ONE, 6, e17284.
- MUZARI, M. O. & HARGROVE, J. W., 1996. The design of target barriers for tsese flies, Glossina spp. (Diptera: Glossinidae). Bulletin of Entomological Research, 86, 579–583.
- NAGEL, P. A., 1995. Environmental Monitoring Handbook for Tsetse Control Operations. The Scientific Environmental Monitoring Group (SEMG) (ed.). Germany: Margraf. ISBN 3-8236-1249-2.
- NASH, T. A. M., 1948. Tsetse flies in British West Africa. His Majesty’s Stationery Office, London.
- NASH, T. A. M., 1969. Africa's bane: the tsetse fly. Collins, London.
- NASH, T. A. M., 1970. Control by parasites and predators of Glossina. In: MULLIGAN, H.W., (ed.). The African Trypanosomiases. London: George Allen and Unwin Ltd.
- NEVILL, E. M., 1998. A new look at tsetse flies and ngana in South Africa. Paper presented at the South African Veterinary Association Congress. June 1998.
- ODINDO, M. O., 1988. Glossina pallidipes virus: Its potential for use in biological control of tsetse. Insect Science and its Application, 9, 399–403.
- OKOTH, J. O., OKETHI, V. & OGOLA, A., 1991. Control of tsetse and trypanosomiasis transmission in Uganda by applications of lambda-cyhalothrin. Medical and Veterinary Entomology, 5, 121-128.
- OLADUNMADE, M. A., TAKKEN, W., DENGWAT, L. & NDAMS, I., 1985. Studies on insecticide-impregnated targets for the control of riverine Glossina subspecies (Diptera: Glossinidae) in the sub-humid savanna zone of Nigeria. Bulletin of Entomological Research, 75, 275-282.
- OWAGA, M. L. A., 1986. Efficacy of buffarine as a potent attractant for tsetse flies. Bulletin of African Insect Science, 10, 9–10.
- PAGABELEGUEM, S., SECK, M. T., SALL, B., VREYSEN, M. J. B., GIMONNEAU, G., FALL, A. G., BASSENE, M., SIDIBÉ, I., RAYAISSE, J. B., BELEM, A. & BOUYER, J., 2015. Long distance transport of irradiated male Glossina palpalis gambiensis pupae and its impact on sterile male yield. Parasites & Vectors, 8, 259.
- PARK, P. O., GLEDHILL, J. A., ALLSOPP, N. & LEE, C. W., 1972. A large-scale scheme for the eradication of Glossina morsitans morsitans Westw. in the Western Province of Zambia by aerial ultra-low-volume application of endosulfan. Bulletin of Entomological Research, 61, 373–384.
- PAYNTER, Q. & BRADY, J., 1992. Flight behaviour of tsetse flies in thick bush (Glossina pallidipes (Diptera: Glossinidae)). Bulletin of Entomological Research, 82, 513–516.
- PERCOMA, L., SOW, A., PAGABELEGUEM, S., DICKO, A. H., SERDÉBÉOGO, O., OUÉDRAOGO, M., RAYAISSE, J. B., BOUYER, J., BELEM, A. M. G. & SIDIBÉ, I., 2017. Impact of an integrated control campaign on tsetse populations in Burkina Faso. Parasites & Vectors in press.
- PERKINS, J. S. & RAMBERG, L., 2004. Environmental Monitoring of Tsetse Fly Spraying Impacts in the Okavango Delta-2002, Harry Oppenheimer Okavango Research Centre, University of Botswana.
- PETER THOMAS ASSOCIATES CONSULTING SERVICES ZIMBABWE AND MINISTER AGRICULTURE UNITED KINGDOM, 1983. A regional tsetse and trypanosomiasis control study: Malawi, Mozambique, Zambia and Zimbabwe. Peter Thomas Associates Consulting Services (Pty) Ltd., 10 Baker Avenue, Harare, Zimbabwe.
- PHELPS, R. J. & BURROWS, P. M., 1969. Lethal temperatures for puparia of Glossina morsitans orientalis. Entomologia Experimentalis et Applicata, 12, 23–32.
- PHELPS, R. J. & BURROWS, P. M., 1969. Prediction of the pupal duration of Glossina morsitans orientalis Vanderplank under field conditions. Journal of Applied Ecology, 6, 323–337.
- PHELPS, R. J. & BURROWS, P. M., 1969. Puparial duration in Glossina morsitans orientalis under conditions of constant temperature. Entomologia Experimentalis et Applicata, 12, 33–43.
- PHELPS, R. J. & LOVEMORE, D. F., 1990. P.O. Box CH139, Chisipite, Harare, Zimbabwe. Unpublished observations.
- PHELPS, R. J. & MAUDLIN, I., 1987. Glossina morsitans Westwood at Rekomitjie Research Station, Zimbabwe. Transactions of the Zimbabwe Scientific Association, 63, 40–46.
- PHELPS, R.J. & VALE, G.A., 1978. Studies on populations of Glossina morsitans morsitans and Glossina pallidipes (Diptera: Glossinidae) in Rhodesia. Journal of Applied Ecology, 15, 743–760.
- PILSON, R. D. & LEGGATE, B. M., 1962. A diurnal and seasonal study of the resting behaviour of Glossina pallidipes Aust. Bulletin of Entomological Research, 53, 551–562.
- PILSON, R. D. & LEGGATE, B. M., 1967. Behaviour studies of Glossina morsitans Westw. in the field. Bulletin of Entomological Research, 57, 227–257.
- PILSON, R. D., BOYT, W. P. & MACKENZIE, P. K., 1978. The relative attractiveness of cattle, sheep and goats to Glossina morsitans morsitans Westwood and G. pallidipes Austen (Diptera: Glossinidae) in the Zambezi valley of Rhodesia. Bulletin of Entomological Research, 68, 489–495.
- PIRES, F. A., 1960. Uma nova espécie de Glossina do grupo morsitans (Diptera: Muscidae) Glossina borgesi sp. nov. Boletin Sociedade de Estudos, Mozambique 29, No. 125, Section D, No. 4
- POLITZAR, H. & CUISANCE, D., 1984. An integrated campaign against riverine tsetse flies Glossina palpalis gambiensis and Glossina tachinoides by trapping and the release of sterile males. Insect Science and its Application, 5, 439-442.
- PONT, A. C., 1980. Family Glossinidae. In: CROSSKEY, R.W., (ed.). Cataloque of the Diptera of the Afrotropical Region. London: British Museum of Natural History.
- POTTS, W. H., 1958. Sterilization of tsetse flies (Glossina) by gamma radiation. Annals of Tropical Medicine and Parasitology, 52, 484–499.
- POTTS, W. H., 1970. Systematics and identification of Glossina. In: mulligan, h.w., (ed.). The African Trypanosomiases. London: George Allen and Unwin Ltd.
- PROGRAMME AGAINST AFRICAN TRYPANOSOMIASIS, 1977. Tsetse eradication on Zanzibar completed. Report on the third meeting of the programme committee, WHO Headquarters, Geneva.
- RAYAISSE, J. B., ESTERHUIZEN, J., TIRADOS, I., KABA, D., SALOU, E., DIARRASSOUBA, A., VALE, G. A., LEHANE, M. J., TORR, S. J. & SOLANO, P., 2011. Towards an Optimal Design of Target for Tsetse Control: Comparisons of Novel Targets for the Control of Palpalis Group Tsetse in West Africa. PLoS Neglected Tropical Diseases, 5, e1332.
- RAYAISSE, J. B., KRÖBER, T., MCMULLIN, A., SOLANO, P., MIHOK, S. & GUÉRIN, P. M., 2012. Standardizing Visual Control Devices for Tsetse Flies: West African Species Glossina tachinoides, G. palpalis gambiensis and G. morsitans submorsitans. PLoS Neglected Tropical Diseases, 6, e1491.
- RAYAISSE, J. B., TIRADOS, I., KABA, D., DEWHIRST, S. Y., LOGAN, J. G., DIARRASSOUBA, A., SALOU, E., OMOLO, M. O., SOLANO, P., LEHANE, M. J., PICKETT, J. A., VALE, G. A., TORR, S. J. & ESTERHUIZEN, J., 2010. Prospects for the Development of Odour Baits to Control the Tsetse Flies Glossina tachinoides and Glossina palpalis s.l. PLoS Neglected Tropical Diseases, 4, e632.
- RENNISON, B. D., LUMSDEN, W. H. R. & WEBB, C. J., 1959. The Resting sites of Glossina pallidipes at night. Report of the East African Trypanosomiasis Research Organization, 1958.
- ROBERTSON, A. G. & KLUGE, E. B., 1968. The use of insecticide in arresting an advance of Glossina morsitans Westwood in the south-east lowveld of Rhodesia. Proceedings and Transactions of the Rhodesia Scientific Association, 53, 17–33.
- ROBERTSON, A. G., 1983. The feeding habits of tsetse flies in Zimbabwe (formerly Rhodesia) and their relevance to some tsetse control measures. Smithersia, 1, 1–72.
- ROBERTSON, A. G., KLUGE, E. B., KRITZINGER, D. A. & DE SOUSA, A. E., 1972. The use of residual insecticides in the reclamation of the Rhodesia– Mozambique border region between the Sabi/Save and Limpopo rivers from Glossina morsitans Westwood. Proceedings and Transactions of the Rhodesia Scientific Association, 55, 34–62.
- ROBINSON, T., ROGERS, D. & WILLIAMS, B., 1997. Mapping tsetse habitat suitability in the common fly belt of Southern Africa using multivariate analysis of climate and remotely sensed vegetation data. Medical and Veterinary Entomology, 11, 235–245.
- ROBINSON, T., ROGERS, D. & WILLIAMS, B., 1997. Univariate analysis of tsetse habitat in the common fly belt of Southern Africa using climate and remotely sensed vegetation data. Medical and Veterinary Entomology, 11, 223–234.
- ROGERS, D. J. & RANDOLPH, S. E., 1978. Metabolic strategies of male and female tsetse (Diptera: Glossinidae) in the field. Bulletin of Entomological Research, 68, 639–654.
- ROGERS, D. J. & RANDOLPH, S. E., 1985. Population ecology of tsetse. Annual Review of Entomology, 30, 197–216.
- ROGERS, D. J. & RANDOLPH, S. E., 1986. Distribution and abundance of tsetse flies (Glossina spp.). Journal of Animal Ecology, 55, 1007–1025.
- ROGERS, D. J. & RANDOLPH, S. E., 1991. Mortality rates and population density of tsetse flies correlated with satellite imagery. Letters to Nature, Nature, 351 (27th June, 1991.)
- ROGERS, D. J. & RANDOLPH, S. E., 1993. Distribution of tsetse and ticks in Africa: past, present and future. Parasitology Today, 9, 266–271.
- ROGERS, D. J., 1991. Satellite imagery, tsetse and trypanosomiasis in Africa. Preventive Veterinary Entomology, 11, 201–220.
- ROGERS, D., 1979. Tsetse population dynamics and distribution: A new analytical approach. Journal of Animal Ecology, 48, 825–849.
- SALEH, K. M., MUSSA, W. A., JUMA, K. G. & VREYSEN, M. J. B., 1999. Eradication of Glossina austeni from the island of Unguja confirmed: results of 2 years of post-eradication monitoring activities, pp. 231-238. In OAU/IBAR (ed.), 25th meeting of the International Scientific Council for Trypanosomiasis Research and Control, 27 September - 1 October, 1999, Mombasa, Kenya.
- SAUNDERS, D. S., 1960. The ovulation cycle in Glossina morsitans Westwood (Diptera: Muscidae) and a possible method of age determination for female tsetse flies by the examination of their ovaries. Transactions of the Royal Entomological Society, London, 112, 221–238.
- SAUNDERS, D. S., 1964. Effect of site and sampling method on the size and composition of catches of tsetse flies (Glossina) and Tabanidae (Diptera). Bulletin of Entomological Research, 55, 483–497.
- SCIENTIFIC ENVIRONMENTAL MONITORING GROUP, 1987. Impact of aerial spraying and odour-baited targets on treated ecosystems. Institut fu¨r Biogeographie, Universität des Saarlandes, Saarbru¨cken, Federal Republic of Germany.
- SECK, M. T., PAGABELEGUEM, S., BASSENE, M. D., FALL, A. G., DIOUF, T. A. R., SALL, B., VREYSEN, M. J. B., RAYAISSÉ, J. B., TAKAC, P., SIDIBÉ, I., PARKER, A. G., MUTIKA, G. N., BOUYER, J. & GIMONNEAU, G., 2015. Quality of Sterile Male Tsetse after Long Distance Transport as Chilled, Irradiated Pupae. PLoS Neglected Tropical Diseases, 9, e0004229.
- SELBY, R., 2010. Limiting the northerly advance of Trypanosoma brucei rhodesiense in post conflict Uganda. The University of Edinburgh.
- SHAW, A. P. M., 2004. Economics of African trypanosomosis, In: MAUDLIN, I., HOLMES, P. H. & MILES, M. A., (eds.), The trypanosomosis. CABI Publishing, Wallingford, 369-402.
- SHAW, A. P. M., TIRADOS, I., MANGWIRO, C. T. N., ESTERHUIZEN, J., LEHANE, M. J., TORR, S. J. & KOVACIC, V., 2015. Costs of using “tiny targets” to control Glossina fuscipes fuscipes, a vector of gambiense sleeping sickness in Arua District of Uganda. PLoS Neglected Tropical Diseases, 9, e0003624.
- SHERENI, W., 1990. Tsetse and Trypanosomiasis Control Branch, Department of Veterinary Services, P.O. Box 8283, Causeway, Harare, Zimbabwe. Personal communication.
- SIMARRO, P. P., CECCHI, G., FRANCO, J. R., PAONE, M., FÈVRE, E. M., DIARRA, A., RUIZ, J. A., MATTIOLI, R. C. & JANNIN, J. G., 2012. Estimating and mapping the population at risk of sleeping sickness. PLoS Neglected Tropical Diseases, 6, e1859.
- SIMARRO, P. P., DIARRA, A., RUIZ POSTIGO, J. A., FRANCO, J. R. & JANNIN, J. G., 2011. The human African trypanosomiasis control and surveillance programme of the World Health Organization 2000-2009: the way forward. PLoS Neglected Tropical Diseases, 5, e1007.
- SIMMONS, G. S., SUCKLING, D. M., CARPENTER, J. E., ADDISON, M. F., DYCK, V. A. & VREYSEN, M. J. B., 2010. Improved quality management to enhance the efficacy of the sterile insect technique for lepidopteran pests. Journal of Applied Entomology, 134, 261-273.
- SIMPSON, H. R., 1958. The effect of sterilized males on a natural tsetse fly population. Biometrics, 14, 159–173.
- SOLANO, P., BOUYER, J., ITARD, J. & CUISANCE, D., 2010b. Cyclical vectors of trypanosomosis, pp. In: LEFÈVRE, P. C., BLANCOU, J., CHERMETTE, R. & UILENBERG, G., (eds.), Infectious and parasitic diseases of livestock. Éditions Lavoisier (Tec & Doc), Paris, 1, 155-183.
- SOLANO, P., GUEGAN, J. F., REIFENBERG, J. M. & THOMAS, F., 2001. Trying to identify, predict and explain the presence of african trypanosomes in tsetse flies. Journal of Parasitology, 87, 1085-1063.
- SOLANO, P., KABA, D., RAVEL, S., DYER, N., SALL, B., VREYSEN, M. J. B., SECK, M. T., DARBYSHIR, H., GARDES, L., DONNELLY, M. J., DE MEEÛS, T. & BOUYER, J., 2010c. Tsetse population genetics as a tool to choose between suppression and elimination: the case of the Niayes area in Senegal. PLoS Neglected Tropical Diseases, 4, e692.
- SOLANO, P., MICHEL, J. F., LEFRANÇOIS, T., DE LA ROCQUE, S., SIDIBE, I., ZOUNGRANA, A. & CUISANCE, D., 1999. Polymerase Chain reaction as a diagnosis tool for detecting trypanosomes in naturally infected cattle in Burkina Faso. Veterinary Parasitology, 86, 95-103.
- SOLANO, P., RAVEL, S. & DE MEEUS, T., 2010a. How can tsetse population genetics contribute to African trypanosomiasis control? Trends in Parasitology, 26, 255-263.
- SOLANO, P., RAVEL, S., BOUYER, J., CAMARA, M., KAGBADOUNO, M. S., DYER, N., GARDES, L., HERAULT, D., DONNELLY, M. J. & DE MEEÛS, T., 2009. The population structure of Glossina palpalis gambiensis from island and continental locations in coastal Guinea. PLoS Neglected Tropical Diseases, 3, e392.
- SOLANO, P., REIFENBERG, J. M., AMSLER-DELAFOSSE, S., KABORÉ, I., CUISANCE, D. & DUVALLET, G., 1996. Trypanosome characterization by polymerase chain reaction in Glossina palpalis gambiensis and G. tachinoides from Burkina Faso. Medical and Veterinary Entomology, 10, 354-358.
- SOLANO, P., SALOU, E., RAYAISSE, J. B., RAVEL, S., GIMONNEAU, G., TRAORE, I. & BOUYER, B., 2015. Do tsetse flies only feed on blood? Infection, Genetics and Evolution, 36, 184-189.
- SOW, A., SIDIBÉ, I., BENGALY, Z., BANCÉ, Z., SAWADOGO, G. J., SOLANO, P., VREYSEN, M. J. B., LANCELOT, R. & BOUYER, J., 2012. Irradiated male Glossina palpalis gambiensis (Diptera: Glossinidae) from a 40-years old colony are still competitive in a riparian forest in Burkina Faso. PLoS ONE, 7, e37124.
- SPÄTH, J. & KÜPPER, W., 1989. Experiments on olfactory attractants for tsetse flies, Glossina spp. (Diptera: Glossinidae) in Ivory Coast. Twentieth Meeting of the International Scientific Council for Trypanosomiasis Research and Control, Mombasa, Kenya, 10–14 April, 1989.
- STEUBER, S., ABDEL-RADY, A. & CLAUSEN, P. H., 2005. PCR-RFLP analysis: a promising technique for host species identification of blood meals from tsetse flies (Diptera: Glossinidae). Parasitology Research, 97, 247-254.
- STEVENSON-HAMILTON, J., 1912. Animal Life in Africa. London: William Heinemann.
- STUCKENBERG, B. R., 1969. Effective temperature as an ecological factor in southern Africa. Zoologica Africana, 4, 145–197.
- SUCKLING, D. M., BARRINGTON, A. M., CHHAGAN, A., STEPHENS, A. E. A., BURNIP, G. M., CHARLES, J. G. & WEE, S. L., 2007. Eradication of the Australian painted apple moth Teia anartoides in New Zealand: trapping, inherited sterility, and male competitiveness. Area-wide control of insect pests, 603-615.
- SWALLOW, B. M., 1999. Impacts of Trypanosomiasis on African Agriculture, FAO, Rome, 2.
- SWYNNERTON, C. F. M., 1921. An examination of the tsetse problem in north Mosurise, Portuguese East Africa. Bulletin of Entomological Research, 11, 315–385.
- SWYNNERTON, C. F. M., 1933. Some traps for tsetse flies. Bulletin of Entomological Research, 24, 69–102.
- SWYNNERTON, C. F. M., 1936. The tsetse flies of East Africa. Transactions of the Royal Entomological Society, London, 84, 1–579.
- TAKKEN, V., OLADUNMADE, M. A., DENGWAT, L., FELDMANN, H. U., ONAH, J. A., TENABE, S. O. & HAMANN, H. J., 1986. The eradication of Glossina palpalis palpalis (Robineau-Desvoidy) (Diptera: Glossinidae) using traps, insecticide-impregnated targets and the sterile insect technique in central Nigeria. Bulletin of Entomological Research, 76, 275-286.
- TAKKEN, W., 1988. Ecological factors limiting the distribution of Glossina morsitans morsitans and of Glossina pallidipes (Diptera: Glossinidae) in Inhambane Province of Mozambique. Insect Science and its Application, 9, 237–242.
- THAKERSI, H., 1991. Records of the tsetse fly Glossina pallidipes in the north eastern region of Zimbabwe. Transactions of the Zimbabwe Scientific Association, 65, 20–23.
- THAKERSI, H., 1992. New records of tsetse flies in eastern Zimbabwe. Transactions of the Zimbabwe Scientific Association, 66, 30–34.
- THOMPSON, J. W., MITCHELL, M., REES, R. B., SHERENI, W., SCHOENFELD, A. H. & WILSON, A., 1991. Studies on the efficacy of deltamethrin applied to cattle for the control of tsetse flies (Glossina spp.) in southern Africa. Tropical Animal Health and Production, 23, 221–226.
- THOMSON, M. C., 1987. The effect on tsetse flies (Glossina spp.) of deltamethrin applied to cattle either as a spray or incorporated into ear-tags. Tropical Pest Management, 33, 329–335.
- TIRADOS, I., ESTERHUIZEN, J., KOVACIC, V., MANGWIRO, T. N. C., VALE, G. A., HASTINGS, I., SOLANO, P., LEHANE, M. J. & TORR, S. J., 2015. Tsetse control and Gambian sleeping sickness; implications for control strategy. PLoS Neglected Tropical Diseases, 9, e0003822.
- TORR, S. J., 1988. Behaviour of tsetse flies (Glossina) in host odour plumes in the field. Physiological Entomology, 13, 467–486.
- TORR, S. J., 1989. The host-orientated behaviour of tsetse flies (Glossina) and the interaction of visual and olfactory stimuli. Physiological Entomology, 14, 325–340.
- TORR, S. J., 1990. Dose responses of tsetse flies (Glossina) to carbon dioxide, acetone and octenol in the field. Physiological Entomology, 15, 93–108.
- TORR, S. J., 1994. Responses of tsetse flies (Diptera: Glossinidae) to warthog (Phacochoerus aethiopicus Pallas). Bulletin of Entomological Research, 84, 411–419.
- TORR, S. J., HALL, D. R. & SMITH, J. L., 1995. Responses of tsetse flies (Diptera: Glossinidae) to natural and synthetic ox odours. Bulletin of Entomological Research, 88, 157–166.
- TORR, S. J., HALL, D. R., PHELPS, R. J. & VALE, G. A., 1997. Methods for dispensing odour attractants for tsetse flies (Diptera: Glossinidae). Bulletin of Entomological Research, 87, 299–311.
- TORR, S. J., HOLLOWAY, M. T. P. & VALE, G. A., 1992. Improved persistence of insecticide deposits on targets for controlling Glossina pallidipes (Diptera: Glossinidae). Bulletin of Entomological Research, 82, 525–533.
- TORR, S. J., MANGWIRO, T. N. C. & HALL, D. R., 1996. Responses of Glossina pallidipes (Diptera: Glossinidae) to synthetic repellents in the field. Bulletin of Entomological Research, 86, 609-616.
- TORR, S. J., MAUDLIN, I. & VALE, G. A., 2007a. Less is more: restricted application of insecticide to cattle to improve the cost and efficacy of tsetse control. Medical and Veterinary Entomology, 21, 53-64.
- TORR, S. J., PARKER, A. G. & LEIGH-BROWNE, G., 1989. The responses of Glossina pallidipes Austen (Diptera: Glossinidae) to odour-baited traps and targets in Somalia. Bulletin of Entomological Research, 79, 99–108.
- TORR, S. J., PRIOR, A., WILSON, P. J. & SCHOFIELD, S., 2007b. Is there safety in numbers? The effect of cattle herding on biting risk from tsetse flies. Medical and Veterinary Entomology, 21, 301-311.
- TURNER, D. A. & BRIGHTWELL, R., 1986. An evaluation of a sequential aerial spraying operation against Glossina pallidipes Austen (Diptera: Glossinidae) in the Lambwe Valley of Kenya: aspects of post-spray recovery and evidence of natural population regulation. Bulletin of Entomological Research, 76, 331-349.
- TURNER, D. A. & INVEST, J. F., 1973. Laboratory analyses of vision in tsetse flies (Diptera Glossinidae). Bulletin of Entomological Research, 62, 343–357.
- TURNER, D. A., 1981. The colonization by the tsetse, Glossina pallidipes Austen, of a unique habitat—exotic coniferous plantation—with special reference to the Lambwe Valley, Kenya. Insect Science and its Application, 1, 243–248.
- VALE, G. A. & CUMMING, D. H. M., 1976. The effects of selective elimination of hosts on a population of tsetse flies (Glossina morsitans morsitans Westwood (Diptera: Glossinidae). Bulletin of Entomological Research, 66, 713–729.
- VALE, G. A. & GRANT, I. F., 2002. Modelled impact on insecticide-contaminated dung on the abundance and distribution of dung fauna. Bulletin of Entomological Research, 92, 251-263.
- VALE, G. A. & HALL, D. R., 1985. The role of 1-octen-3-ol, acetone and carbon dioxide in the attraction of tsetse flies, Glossina spp. (Diptera: Glossinidae) to ox odour. Bulletin of Entomological Research, 75, 209–217.
- VALE, G. A. & PHELPS, R. J., 1978. Sampling problems with tsetse flies (Diptera: Glossinidae). Journal of Applied Ecology, 15, 715–726.
- VALE, G. A., 1971. Artificial refuges for tsetse flies (Glossina spp.). Bulletin of Entomological Research, 61, 331–350.
- VALE, G. A., 1974. The responses of tsetse flies (Diptera: Glossinidae) to mobile and stationary baits. Bulletin of Entomological Research, 64, 545–588.
- VALE, G. A., 1979. Field responses of tsetse flies (Diptera: Glossinidae) to odours of men, lactic acid and carbon dioxide. Bulletin of Entomological Research, 69, 459–468.
- VALE, G. A., 1980. Field studies on the responses of tsetse flies (Glossinidae) and other Diptera to carbon dioxide, acetone and other chemicals. Bulletin of Entomological Research, 70, 563–570.
- VALE, G. A., 1982. The improvement of traps for tsetse flies (Diptera: Glossinidae). Bulletin of Entomological Research, 72, 95–106.
- VALE, G. A., 1982. The role of the antenna in the availability of tsetse flies (Diptera: Glossinidae) to population sampling techniques. Transactions of the Zimbabwe Scientific Association, 61, 33–40.
- VALE, G. A., 1982. The trap orientated behaviour of tsetse flies (Glossinidae) and other Diptera. Bulletin of Entomological Research, 72, 71–93.
- VALE, G. A., 1984. The responses of Glossina (Glossinidae) and other Diptera to odour plumes in the field. Bulletin of Entomological Research, 74, 143–152.
- VALE, G. A., 1993. Visual Responses of tsetse flies (Diptera: Glossinidae) to odour-baited targets. Bulletin of Entomological Research, 83, 277–289.
- VALE, G. A., 1998. Responses of tsetse flies (Diptera: Glossinidae) to vegetation in Zimbabwe: implications for population distribution and bait siting. Bulletin of Entomological Research, 88, S7–S59.
- VALE, G. A., 1999. Joint tests of insecticides for use on cattle. Final Report. Regional Tsetse and Trypanosomiasis Control Programme, Malawi, Mozambique, Zambia and Zimbabwe. Harare, Zimbabwe.
- VALE, G. A., HALL, D. R. & GOUGH, A. J. E., 1988. The olfactory responses of tsetse flies Glossina subspecies (Diptera: Glossinidae) to phenols and urine in the field. Bulletin of Entomological Research, 78, 293–300.
- VALE, G. A., HARGROVE, J. W., CHAMISA, A., GRANT, I. F. & TORR, S. J., 2015. Pyrethroid treatment of cattle for tsetse control: Reducing its impact on dung fauna. PLoS Neglected Tropical Diseases, 9.
- VALE, G. A., HARGROVE, J. W., COCKBILL, G. F. & PHELPS, R. J., 1986. Field trials of baits to control populations of Glossina morsitans morsitans Westwood and Glossina pallidipes Austen (Diptera: Glossinidae). Bulletin of Entomological Research, 76, 179–193.
- VALE, G. A., LOVEMORE, D. F., FLINT, S. & COCKBILL, G. F., 1988. Odour-baited targets to control tsetse flies, Glossina spp. (Diptera: Glossinidae), in Zimbabwe. Bulletin of Entomological Research, 78, 31-49.
- VALE, G. A., WILCOX, J. & ABSON, J., 1994. Prospects for using odour-baited trees to control tsetse flies. Bulletin of Entomological Research, 84, 123–130.
- VALE, G., 2003. Pyrethroid-treated cattle for tsetse control: effect of restricted application. Newsl. Int. Cont. Path. Tryp. Vect. 7,15-16.
- VAN DEN BOSSCHE, P., DE LA ROCQUE, S., HENDRICKX, G. & BOUYER, J., 2010. A changing environment and the epidemiology of tsetse-transmitted livestock trypanosomiasis. Trends in Parasitology, 26(5), 236-243.
- VAN DER PLANK, F. L., 1944. Hybridization between Glossina species and suggested new method for control of certain species of tsetse. Nature, 154, 607-608.
- VAN DER PLANK, F. L., 1947. Experiments in the hybridisation of tsetse‐flies (glossina, diptera) and the possibility of a new method of control. Ecological Entomology, 98, 1-18.
- VAN DER VLOEDT, A. M. V., BALDRY, D. A. T., POLITZAR, H., KULZER, H. & CUISANCE, D., 1980. Experimental helicopter applications of decamethrin followed by release of sterile males for the control of riverine vectors of trypanosomiasis in upper volta. The international journal of Tropical Insect Science, 1, 105-102.
- VAN SICKLE, J., 1988. Invalid estimates of rate of population increase from Glossina (Diptera: Glossinidae) age distributions. Bulletin of Entomological Research, 78, 155–161.
- VREYSEN, M. & ROBINSON, A. S., 2011. Ionising radiation and area-wide management of insect pests to promote sustainable agriculture. A review. Agronomy for sustainable development, 2, 671-692.
- VREYSEN, M. J. B., & SALEH, K. M., 2001. Long-term sampling of gamma sterilised male Glossina austeni (Diptera: Glossinidae) with sticky panels on Unguja Island (Zanzibar). Acta Tropica, 80, 29-37.
- VREYSEN, M. J. B., 2001. Principles of area-wide integrated tsetse fly control using the Sterile Insect Technique. Journal of Tropical Medicine, 61, 397-411.
- VREYSEN, M. J. B., 2005. Monitoring sterile and wild insects in area-wide integrated pest management programmes. In: DYCK, V.Z., HENDRICHS, J. & ROBINSON, A.S., (eds.). Sterile insect technique. Principles and practice in area-wide integrated pest management. Springer, Dordrecht, The Netherlands, 325-361.
- VREYSEN, M. J. B., 2006. Prospects for area-wide integrated control of tsetse flies (Diptera: Glossinidae) and trypanosomosis in sub-Saharan Africa. Revista de la Sociedad Entomológica Argentina, 65.
- VREYSEN, M. J. B., MEBRATE, A., MENJETA, M., BANCHA, B., WOLDEYES, G., MUSIE, K., BEKELE, K. & ABOSET, G., 1999. The distribution and relative abundance of tsetse flies in the Southern Rift Valley of Ethiopia: preliminary survey results. In: 25th meeting of the International Scientific Council for Trypanosomiasis Research and Control, 27 September - 1 October, 1999, Mombasa, Kenya. OAU/IBAR, 202-213.
- VREYSEN, M. J. B., SALEH, K. M, MRAMBA, F., PARKER, A., FELDMANN, U., DYCK, V. A., MSANGI, A. & BOUYER, J., 2014. Sterile insects to enhance agricultural development: the case of sustainable tsetse eradication on Unguja Island, Zanzibar using an area-wide integrated pest management approach. PLoS Neglected Tropical Diseases, 8, e2857.
- VREYSEN, M. J. B., SALEH, K. M., ALI, M. Y., ABDULLA, A. M., ZHU, Z. R., JUMA, K. G., DYCK, V. A., MSANGI, A. R., MKONYI, P. A. & FELDMANN, H. U., 2000b. Glossina austeni (Diptera: Glossinidae) Eradicated on the Island of Unguja, Zanzibar, Using the Sterile Insect Technique. Journal of Economic Entomology, 93, 123-135.
- VREYSEN, M. J. B., SALEH, K. M., LANCELOT, R. & BOUYER, J., 2011. Factory tsetse flies must behave like wild flies: a prerequisite for the sterile insect technique. PLoS Neglected Tropical Diseases, 5(2), e907.
- VREYSEN, M. J. B., SALEH, K. M., ZHU, Z. R. & SULEIMAN, F. W., 2000a. Responses of Glossina austeni to sticky panels and odours. Medical and Veterinary Entomology. 14, 283-289.
- VREYSEN, M. J. B., SECK, M. T., SALL, B. & BOUYER, J., 2013. Tsetse flies: their biology and control using area-wide integrated pest management approaches. Journal of Invertebrate Pathology, 112, S15–S25.
- VREYSEN, M. J. B., ZHU, Z. R. & SALEH, K. M., 1998. Field responses of Glossina austeni to sticky panels on Unguja Island, Zanzibar. Medical and Veterinary Entomology, 12, 407-416.
- VREYSEN, M., GERARDO-ABAYA, J. & CAYOL, J. P., 2005. Lessons learned from Area-wide Insect Pest Management Programmes with an SIT component: an FAO/IAEA Perspective. In: HENDRICHS, J., KENMORE, P., ROBINSON, A. S. & VREYSEN, M., (eds.), Area-Wide Integrated Pest Management (AW-IPM): Principles, Practice and Prospects. Springer, Dordrecht, The Netherlands, 723-744.
- VREYSEN, M., SALEH, K. M., KHAMIS, I. S. & MRAMBA, F., 1999. An evaluation of insecticide-impregnated screens against Glossina austeni (Diptera: Glossinidae) on Unguja Island of Zanzibar. International Journal of Tropical Insect Science, 19, 75-84.
- WALL, R., 1988. Tsetse mating behaviour: Effects of age and hunger in Glossina morsitans morsitans and G. pallidipes. Physiological Entomology, 13, 479–486.
- WARNES, M. L., 1992. Activation of three species of tsetse (Glossina spp.) in response to host derived stimuli. Medical and Veterinary Entomology, 6, 349–354.
- WARNES, M. L., VAN DEN BOSSCHE, P., CHIHIYA, J., MUDENGE, D., ROBINSON, T. P., SHERENI, W. & CHADENGA, V., 1999. Efficacy of insecticide treated cattle as a barrier to re-invasion of tsetse flies to cleared areas in north-eastern Zimbabwe. Medical and Veterinary Entomology, 13, 177–184.
- WATSON, H. J. C., 1962. The domestic pig as a reservoir of T. gambiense. International Scientific Committee for Trypanosomiasis Research, Ninth Meeting, Conakry.
- WEITZ, B., 1963. The feeding habits of Glossina. Bulletin of the World Health Organization, 28, 711–729.
- WEITZ, B., 1970. Hosts of Glossina. In: mulligan, h.w., (ed.). The African Trypanosomiases. London: George Allen and Unwin Ltd.
- WELLS, E. A., 1972. The importance of mechanical transmission in the epidemiology of nagana: A review. Tropical Animal Health and Production
- WHITEHEAD, D. L., 1985. Use of natural products and their analogues for combating pests of agricultural and public health importance in Africa. In: HEDIN, P.A., (ed.). Bioregulators for Pest Control. American Chemical Society Symposium Series No. 276.
- WHITESIDE, E. F., 1949. An experiment in control of tsetse with DDT-treated oxen. Bulletin of Entomological Research, 40, 123–134.
- WILDE, J. K. H. & FRENCH, M. H., 1945. An experimental study of Trypanosoma rhodesiense infection in Zebu cattle. Journal of Comparative Pathology, 55, 206–219.
- WILLETT, K. C., 1965. Some observations on the recent epidemiology of sleeping sickness in the Nyanza region, Kenya, and its relation to the general epidemiology of Gambian and Rhodesian sleeping sickness in Africa. Transactions of the Royal Society of Tropical Medicine and Hygiene, 59, 374–386.
- WILLIAMS, B., 1994. Models of trap seeking behaviour by tsetse flies and anemotaxis, klinokinesis and edge detection. Journal of Theoretical Biology, 168, 105–115.
- WILLIAMS, B., DRANSFIELD, R. & BRIGHTWELL, R., 1992. The control of tsetse flies in relation to fly movement and trapping efficiency. Journal of Applied Ecology, 29, 163–179.
- WILLIAMSON, D. L., DAME, D. A., GATES, D. B., COBB, P. E., BAKULI, B. & WARNER, P. V., 1983b. Integration of insect sterility and insecticides for control of Glossina morsitans morsitans Westwood (Diptera: Glossinidae) in Tanzania. V. The impact of sequential releases of sterilized tsetse flies. Bulletin of Entomological Research, 73, 391–404.
- WILLIAMSON, D. L., DAME, D. A., GATES, D. B., COBB, P. E., BAKULI, B. & WARNER, P. V., 1983c. Integration of insect sterility and insecticides for control of Glossina morsitans morsitans (Diptera: Glossinidae) in Tanzania: V. The impact of sequential releases of sterilized tsetse flies. Bull. Entomol. Res. 73: 391-404.
- WILLIAMSON, D. L., DAME, D. A., LEE, C. W., GATES, D. B. & COBB, P. E., 1983a. Integration of insect sterility and insecticides for control of Glossina morsitans morsitans (Diptera: Glossinidae) in Tanzania: IV. Application of endosulfan as an aerosol prior to the release of sterile males. Bulletin of Entomological Research, 73, 383-389.
- WILSON, V. J., 1975. Game and tsetse fly in eastern Zambia. Occasional Papers of the National Museum and Monuments of Rhodesia, B, 5, 339–404.
- ZAMBEZI LIVESTOCK AND LANDS LTD., ZAMBIA, IN ASSOCIATION WITH RDP LIVESTOCK SERVICES B.V., THE NETHERLANDS, 1996. Study of the tsetse fly problem common to Angola, Botswana, Namibia and Zambia. Consultants’ report to the Regional Tsetse and Trypanosomiasis Control Programme of Malawi, Mozambique, Zambia and Zimbabwe. P. O. Box A 560, Harare, Zimbabwe.
- ZDÁREK, J. & DENLINGER, D.L., 1993. Metamorphosis behaviour and regulation in tsetse flies (Glossina spp.) (Diptera: Glossinidae): a review. Bulletin of Entomological Research, 83, 447–461.