- Infectious Diseases of Livestock
- Part 2
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Porcine reproductive and respiratory syndrome
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Porcine reproductive and respiratory syndrome (PRRS) — as its name suggests — manifests as both reproductive failure and respiratory disease,55, 63 and is caused by a virus of the genus Arterivirus (family Arteriviridae). Pigs of any age and any parity are affected. A rare but overt clinical sign is the appearance of cyanotic lesions in the extremities, particularly the ears. In uncomplicated infections, a characteristic interstitial pneumonia may be seen microscopically.
The disease was first described in pigs in North Carolina, Minnesota and Iowa, USA, in 1987,43, 47 although retrospective serological studies have revealed evidence of infection since the late 1970s to early 1980s in Canada and the USA,14, 19, 26 and also in countries which had imported pigs from the USA at that time.83, 84 This condition has now been reported in many parts of the world, although some countries are still purportedly free.63 In May 1992, the disease was included in List B (annual reporting) of the Organization International des Epizooties (OIE).8 There is little doubt that countries reporting the disease have subsequently suffered in terms of trade in pigs and semen.
Aetiology
In July 1991, workers at the Central Veterinary Institute at Lelystad, the Netherlands, announced the isolation of a hitherto unknown virus, which they called ‘Lelystad virus’. 102 The virus was isolated using cell cultures of pig alveolar macrophages (PAMs) obtained from a six-week-old specific pathogen-free (SPF) piglet. The virus was highly cytopathic and was noted to be sensitive to chloroform. It did not agglutinate the red blood cells of chickens, guinea pigs, sheep, pigs or humans. The virus had a buoyant density of 1,18g/cm3 in CsCl.
In the USA, another isolate of procine reproductive and respiratory syndrome virus (PRRSV) was also described and designated the ATCC VR 2332 isolate.11, 23 Unlike the European strain, the USA isolation was made in an established monkey kidney cell line CL 2621 (a clone of MA 104).10
A sub-clone of MA 104, called MARC 145, has been prepared, which has enhanced susceptibility to PRRSV and this line is routinely used for isolation of American isolates.48 European isolates of PRRSV do not grow so readily in this cell line, but can be successfully adapted by multiple passage.
Porcine reproductive and respiratory syndrome virus is classified as a member of the genus Arterivirus within the family Arteriviridae, order Nidovirales, whose members have been grouped according to their morphology, genome organization, physicochemical properties and host cell specificity.64, 64, 65 Other members of the family are lactate dehydrogenase elevating virus (LDV) of mice, equine arteritis virus and simian haemorrhagic fever virus. They all have positive sense, single strand ribonucleic acid (RNA) genomes and transcribe multiple subgenomic mRNAs. Although the genomes of the arteriviruses are shorter than those of coronaviruses and their virions differ morphologically, being smaller and having icosahedral rather than helical nucleocapsids, they all possess similar replicative strategies.
Morphologically, the PRRSV virion is round, enveloped and somewhat variable in size, being 45 to 65 nm in diameter, with an icosahedral nucleocapsid of 25 to 35 nm.101 The Arterivirus genome is approximately 15kB and has eight open reading frames (ORFs), arranged as a 3’ co-terminal nested set. The first two ORFs, 1a and 1b, are linked by a ‘slippery knot’ frameshift junction sequence and encode the enzymes involved in replication. Open reading frames 2 to 7 encode structural proteins. Further details of the genome organization of PRRSV can be found in a definitive review by Meulenberg et al.64
Detailed analyses of isolates of PRRSV from America and Europe show them to be genetically and antigenically distinct.70, 99 To a lesser extent, diversity is also seen among isolates from Europe31, 89 and America,7, 12, 56 leading to the conclusion that they represent two distinct genetic groups, with divergent evolution of the two genotypes from some unknown ancestor.70 Emerging evidence indicates that greater diversity than was hitherto expected, may exist among isolates of PRRSV from Eastern Europe and Northern Asia.6 It is likely that the distinction between genotypes may become less straightforward, since evidence of recombination between the two genotypes has recently been reported.107 Such events may have profound effects on the pathogenesis of strains and on the efficacy of vaccines.
Epidemiology
The spread of PRRS suggested an infectious aetiology, yet it was some time before the causative agent was discovered. Many pathogens, both viral and bacterial, were implicated by virtue of their isolation from a high percentage of clinical cases, yet no pathogen was able to reproduce the disease experimentally, either alone or in combination.39, 48, 72 Whilst there was little doubt that these pathogens often contributed to the clinical picture seen in the respiratory form of field cases of PRRS, the lack of consistent isolation of any single agent from cases of the disease suggested that, rather than being the primary agents, their presence was opportunistic, causing secondary infections leading to multifactorial disease. Serological studies failed to identify any single known viral agent as causative of PRRS, nor was the disease experimentally reproducible using these agents either alone or in combination. One group of workers considered that the mycotoxin, fumonisin, produced by Fusarium moniliforme, was involved in the aetiology of PRRS.9
Infection can occur by the respiratory, oral and venereal routes, as well as by intramuscular, intraperitoneal or intravenous inoculation.63, 77, 79
The PRRSV is relatively labile and survives poorly at ambient temperatures above 20 °C.15 It is shed by infected pigs in all secretions, including faeces, and is readily spread between animals by direct contact and between herds by purchased stock. Saliva has been suggested to play a major role in transmission between pigs, being shown to be present for up to 42 days post-infection.103 A remarkable feature of PRRS infections is the speed at which the virus spreads and establishes itself in naive pig populations. Transmission is rapid in areas of high pig density and the virus is generally regarded as being able to be spread by the airborne route, though evidence for this is circumstantial.63 Pork and pork products have been shown to be of little risk in spreading virus.53
Transmission via semen of infected, asymptomatic boars has been well documented and has been implicated in the spread of the virus. Virus can be excreted in semen for up to 21 days post-infection and semen has been shown experimentally to transmit virus to artificially inseminated gilts.21, 77
Depending on the size and management of herds, PRRSV can persist for long periods, especially in large fattening units where all in/out husbandry is not practised.
Pathogenesis
Many early records describe lesions that have subsequently been associated with secondary pathogens rather than with the causative agent of PRRS. The pathogenesis is affected by viral factors and by the age, immune status and breed of pig.
Primary replication after respiratory infection is within the alveolar macrophages, but other cells in the lungs are also susceptible, including the intra-alveolar macrophages and other lymphoid cells, such as monocytes and type II pneumocytes.13, 33 Virus has also been detected in the spleen and Peyer’s patches, and in macrophages in the hepatic sinusoids and adrenal glands.20 Virus can be detected in pulmonary lymph nodes within 24 hours of infection and in the spleen and other body lymph nodes within 84 hours. A viraemia develops within two days and can persist for many weeks especially in young pigs. The long period of viraemia, which in some animals may exceed 100 days, demonstrates the ability of the virus to sustain an active viraemia in the presence of a vigorous immune response. Little, if anything, is known of how and where the virus manages to persist for such periods, although the tonsils and other lymphoid organs are candidate reservoirs of infection. At up to 16 weeks post-infection, virus can be reactivated by corticosteroid treatment.4 Some strains of PRRSV seem to demonstrate a wider tissue tropism, with antigen demonstrable in endothelial cells in the myocardium.40
Infection of cells by PRRSV may not be required for cell death to occur. Apoptotic events have recently been described in cells not infected with the virus,85, 92 suggesting the presence of an indirect mechanism, probably through soluble factors. The product of ORF5, the major envelope glycoprotein gp5, has been shown to cause this effect in vitro.88
Alveolar macrophages are reduced in number during PRRSV infection, but return to normal levels by 28 days postinfection. Interstitial pneumonia occurs, with the histological picture often complicated by the presence of other infections, but it is unclear whether this is the result of immunosuppression of the host by the virus.
The long-term ramifications of congenital infection of piglets is poorly understood. The observation of lymph node damage80 and low survival rate of neonates infected in utero60 suggests some sort of debilitation, but its nature is unknown and warrants further investigation.
Clinical signs
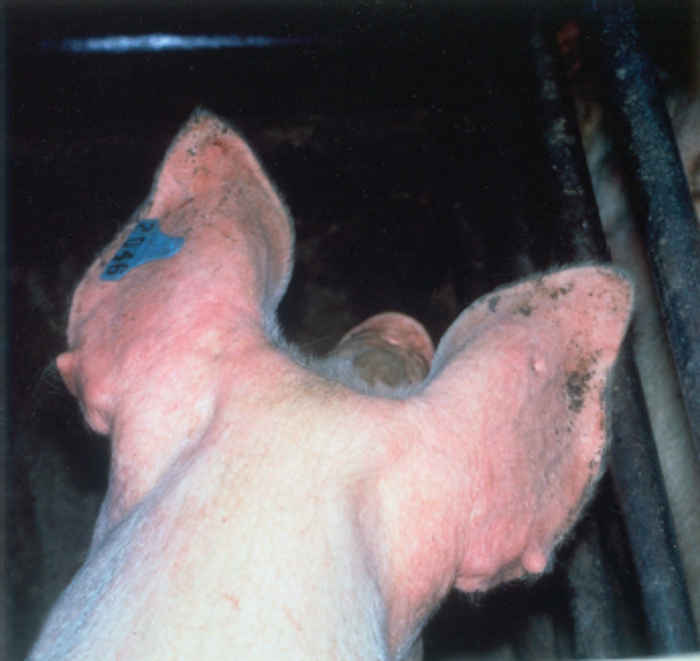
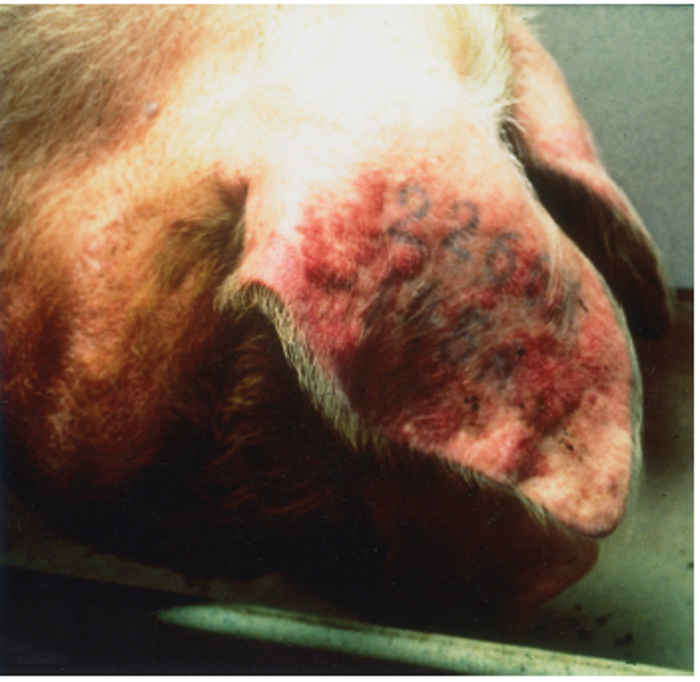
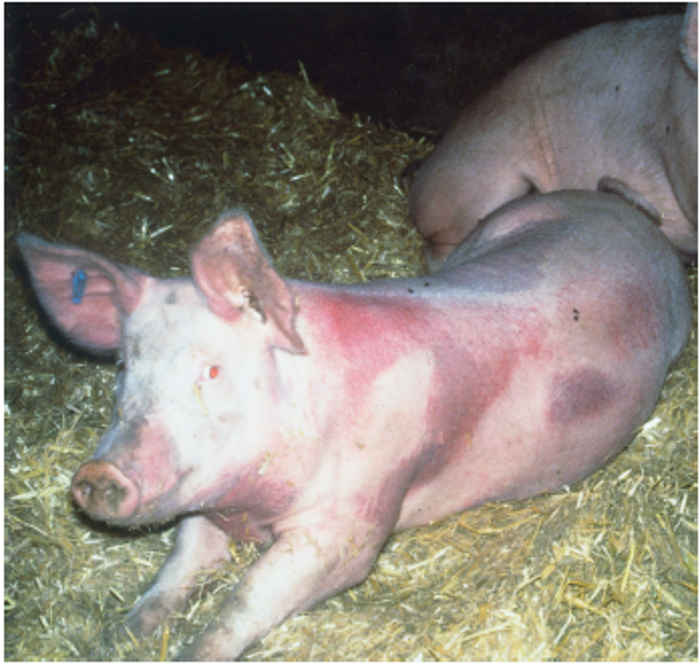
The syndrome manifests as both reproductive failure and respiratory disease,30, 55 and can affect pigs of any age,although more severe respiratory signs are seen in younger animals. The disease among pregnant sows can be dramatic, affecting animals of different parities and causing abortion, stillbirth and high pre-weaning mortality. Infection can result in a number of clinical signs, depending on viral, host and environmental factors. Infected animals are commonly lethargic, with fever, anorexia, agalactia, and sometimes a red/blue discoloration of the skin, particularly at the extremities, most often the ears and vulva (Figure 85.1).
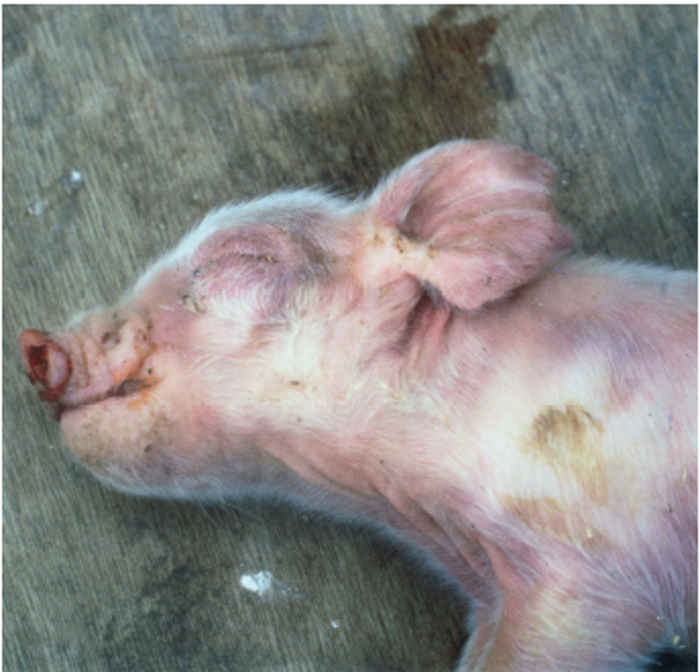
Figure 85.1 Reddish or bluish blush on the ears of an affected pig (a) which may progress to necrotic lesions (b). Blotching is also apparent on the trunk of affected pigs (c). Discoloration also occurs in younger animals and may be accompanied by oedema of eyelids, cranium and snout in piglets infected with the European genotype of PRRS virus (d). (Reproduced by courtesy of M. White, Cotswold Pig Dev. Co., Lincoln, UK)
The sign of ‘blue ear’ usually appears as a reddish or bluish blush, which occasionally results in the development of necrotic lesions and, in severe cases, loss of the ear. Such blotching, which is often transient, can also be apparent on the trunk of affected pigs. There may also be subcutaneous oedema, particularly of the hind limbs. In neonates, oedema of the eyelids and periorbital area, cranium and snout is sometimes seen in those infected with the European genotype of PRRSV (Figure 85.1). Reproductive signs include abortion, which can vary in severity from sporadic to abortion storms raging through the herd (Figure 85.2). A characteristic is the protracted course of the disease episode within herds, which can last for up to six months.4 The most devastating effects are seen following infection of third trimester sows, resulting in abortion or premature birth of mummified, autolysed or stillborn piglets within the same litter. Any surviving piglets are generally weak and undersized, often showing discoloration of the skin, oedema of eyelids and hind leg weakness and ataxia often manifesting as splay-leg and a characteristic sitting posture (Figure 85.2). They may develop a thumping respiration and postnatal mortality can reach 100 per cent.63, 79
In weaned pigs, few overt clinical signs may be seen, apart from fever, lethargy and poor thrift, often resulting in a post-weaning mortality which may be 10 to 15 per cent, or occasionally, as high as 25 per cent.47, 68, 87 Where disease is present, other, concurrent bacterial or viral diseases are commonly implicated in the overall picture.29, 47, 63, 79, 87 In finishing pigs and adults, mortality is rare, and signs may be restricted to a transient fever and inappetance, with a decrease in growth rate, possibly due to secondary infections.27, 47, 68 In boars, a temporary loss of libido is sometimes seen.93
Figure 85.2 Sick, weak, unthrifty piglets are often born together with dead piglets and large, mummified foetuses (a). In litters that are born alive, piglets are sick, with skin discoloration, facial oedema and a characteristic hind leg weakness, often manifesting as splay-leg and a characteristic sitting posture (b). (Reproduced by courtesy of G. Wensvoort, ID-Lelystad, the Netherlands)
Pathology
Infection results in the development of lesions in several organs; in the lungs changes can be evident as early as two days post-infection. Gross lesions in the lungs are variable. The lungs may be slightly more grey in colour and are less likely to collapse when the thoracic cavity is opened, but, in uncomplicated cases, changes may be subtle and difficult to detect on gross examination.27, 29, 59 Under experimental conditions, different strains of PRRSV have been shown to induce varying degrees of multi-focal, tan-coloured areas of consolidation, involving from 6,8 to 54,2 per cent of the lungs.41 In field cases, lesions are usually complicated by concurrent bacterial infections, with Pasteurella multocida and Streptococcus suis most commonly isolated.30 In pure infections of PRRSV alone, the histopathology in the lungs is characterized by mild lesions. Microscopically, the most characteristic finding is an interstitial pneumonia, with a moderate thickening of alveolar septa, mainly by macrophages and some lymphocytes (Figure 85.4 ).28, 79 By day seven post-infection, alveolar spaces may contain necrotic debris and type II pneumocytes proliferation may be present, with interstitial pneumonia persisting for as long as 28 days, but neither bronchioles nor bronchi show significant lesions, although lung damage may still be evident.29, 33
The lymph nodes of piglets infected in utero show marked enlargement by 14 days post-infection, occasionally with small, fluid-filled cystic spaces revealed on gross examination. The germinal centres of affected lymph nodes are enlarged, with many necrotic lymphocytes and increased numbers of sinusoidal macrophages, syncytial giant cells, some of them multinucleated, particularly around cyst-like spaces.33 Such lesions can exist for at least 40 days in affected piglets. A mild rhinitis, lymphocytic encephalitis and lymphocytic myocarditis have also been described.79
In boars, the consequences of PRRSV infection on semen are variable, ranging from no effects to reductions in sperm volume, and motility and morphologic abnormalities, with such variation possibly due to virus strain or breed of pig.21
Diagnosis
Establishing a clinical diagnosis is not easy because of the range and variability of clinical signs, none of which is pathognomonic and because of the complicating effects of secondary infections. Laboratory tests are also required to rule out subclinical infections when certifying animals as PRRS-free. Test protocols for serology and isolation are provided in the OIE Manual of Standards for Diagnostic Tests and Vaccines8 and are reviewed elsewhere.16, 29, 59, 79
In their report of the isolation of PRRSV, Wensvoort et al.100 also described a serological test, the immunoperoxidase monolayer assay (IPMA), which they used to demonstrate seroconversion among experimental and field cases of PRRSV infection. This test, and its fluorescent-based equivalent, the immunofluorescent assay,49 have become the standard assays for antibody to PRRSV throughout the world.16, 29, 59, 63, 79 Though originally using PRRSV-infected and control pig PAM cells as the matrix for the assay, many tests now use cell lines where possible. The IPMA methods used in laboratories now vary somewhat, but essentially follow the original methodology.11 Cross-IPMA tests, employing USA and European isolates of PRRSV, revealed evidence of marked differences between viruses from the two continents. 99 In situations where there is a possibility of either genotype being present, or if it is required to demonstrate absolute freedom from infection, it is prudent to use both genotypes to ensure maximum test sensitivity. Using IPMA, antibodies can be detected within 10 days of infection and titres become very high within a few weeks. Although some pigs remain seropositive for prolonged periods, others revert to a seronegative status within six months. It must also be borne in mind that seropositive animals may still be viraemic.
It is not possible to assay neutralizing antibody using PAM cells due to the phenomenon of antibody-dependent enhancement (ADE), where infection of these cells is actually elevated in the presence of antibody via phagocytosis of virus/antibody complexes by Fc receptors. This phenomenon is thought to contribute to the pathogenesis to varying degrees, depending on the degree of affinity between the antibody and the major envelope glycoprotein, gp5.105, 106 However, serum neutralization can be assayed using the cell line CL 2621.10, 42 Use of these cells has also shown that antibody to gp5, largely correlates with neutralization36, 108 although another envelope glycoprotein, gp4, is also neutralizing, but to a lesser extent.98 Initially it was reported that neutralizing antibodies to PRRSV can only be detected rather later in infection, but with incorporation of complement, 94 neutralizing antibody can be detected as soon as eight days post-infection.
ELISAs have also been used in the serological diagnosis of PRRS. In the first ELISA to be described,2, 3 the supernatant of PAM cells replicating PRRSV was used to produce an antigen, with a control antigen prepared from uninfected cultures. An indirect format was used to measure the binding of antibodies in test sera to positive and negative antigen coated wells. Control positive and negative sera were also included in the test. Conventional conjugate and substrate steps then followed and a sample to positive (S/P) ratio calculated, being derived from sample and control OD values. Improved commercial tests have now been launched, such as HerdChek 78 and Ingenzim (Ingenaza), in which recombinant and/or tissue culture-derived antigens from both genotypes, either as a cocktail or in individual wells, are used. Though not as sensitive as IPMA for the detection of early antibody,31 such tests have real value in rapid, cost-effective diagnosis at the herd level.
Viral isolation is not straightforward, since the virus is fastidious in its requirement for PAMs as the cell of choice.
These cells do not replicate in vitro, so must be harvested from five- to seven-week-old piglets, preferably of SPF status. 8 Not all harvests of PAMs are equally susceptible, so batches must be tested before use.
Serum is a productive source of virus, since viraemia can persist for several weeks, even in the presence of antibody. For post-mortem examination, foetal fluids and organ samples, such as lungs, tonsils and spleen, are the sample of choice for viral isolation.
Examination of lung macrophages obtained by lavage has also been described,62 either by immuno-staining directly, after cultivation, or after cocultivation with cell cultures, although whether the advantages outweigh the difficulties involved in obtaining samples from live pigs is questionable. Since the emergence in the USA of cell lines that support the growth of PRRSV, these too have been employed in virus isolation.

Figure 85.3 Immunoperoxidase staining of PRRSV in alveolar macrophages. Note typical immuno-staining of PRRSV-infected PAMs, achieved using porcine antiserum to PRRSV, rabbit anti-pig horseradish peroxidase (HRPO) and the chromagen amino-ethyl carbazole (AEC). (x100)
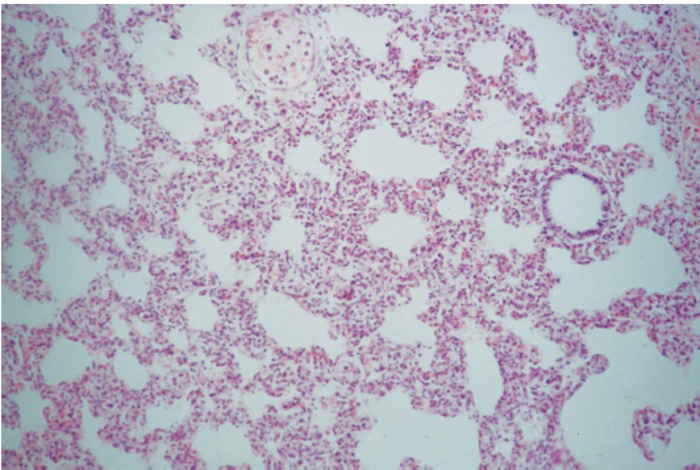
85.4a Peripheral keratitis
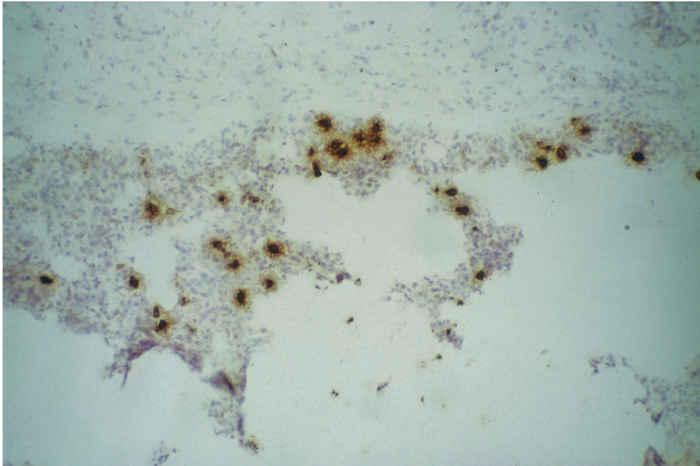
85.4b
Figure 85.4 Mild interstitial pneumonia (a) (x100). (Reproduced by courtesy of S. Done, VLA Weybridge, UK). Immunocytochemistry of a frozen section of lung (b) reveals the presence of infected alveolar macrophages within the lung. Stained with monoclonal antibody WBE 5,32 rabbit anti-mouse HRPO and AEC (x100). (Reproduced by courtesy of J. Pol, ID-Lelystad, the Netherlands)
Generally speaking, European strains grow less readily in cell lines derived from MA 104 than do American strains76 and are therefore the preferred option when attempting isolation from field material.10 The phenomenon of ADE may assist in isolation, hence the productivity of serum as a sample. Antibody has been employed to enhance virus yield by incorporating it into an overlay medium of PAM cultures used for isolation.105
The virus is cytopathic and will rapidly kill PAM cultures, usually within 48 hours. The presence of the virus is confirmed by immuno-staining (Figure 85.3), using the same technique as for IPMA.
The reverse transcriptase-polymerase chain reaction (RT-PCR) has been described to detect viral RNA in a range of specimens,37, 38, 53, 73, 90 and has been used in the screening of semen for artificial insemination.21, 54
Though rarely used in routine diagnosis, it is also possible to demonstrate the presence of PRRSV-infected cells in tissue, by detection of antigen by immunocytochemical staining (Figure 85.4) and detection of viral RNA by in situ hybridization.22, 52, 91
Differential diagnosis
The different manifestations of disease attributable to this virus necessitate a breakdown into a number of clinical scenarios:
Respiratory disease
The diagnosis of the respiratory form of PRRS is compounded by the many other pathogenic agents that may be isolated from clinical cases, and that may cause respiratory disease in their own right. In many herds where clinical signs suggest PRRS, but in which investigations prove to be negative, a positive diagnosis of swine influenza is made.1, 79 A similar disease syndrome—proliferative and necrotizing pneumonia (PNP)—has been described in the USA, in which swine influenza has also been implicated.58 Aujeszky’s disease (pseudorabies) should also be considered, as should porcine respiratory coronavirus infection, particularly in cases of chronic respiratory disease in weaners and growers.
Acute reproductive disease
The most useful laboratory test for investigation of reproductive disease is paired serology or virus isolation. Late-term abortions and premature farrowings particularly are features of PRRSV infection of naive sows, but can also be caused by Aujeszky’s disease virus, porcine parvovirus and Toxoplasma gondi.
Chronic reproductive effects
This is not usually a feature of PRRS. However, PRRS outbreaks may predispose to longer-term problems associated with eperythrozoonosis, leptospirosis and cystitis or pyelonephritis.
Chronic pre-weaning mortality
Many such cases, in which PRRSV was originally implicated, have been found to be due to poor husbandry, poor hygiene or problems with intake of colostrum. Similarly, where acute PRRS is initially diagnosed, chronic disease can be complicated by other agents such as swine influenza. Post-weaning multisystemic wasting syndrome is a common feature of chronically PRRSV-infected herds, and a newly discovered virus, porcine circovirus type 2, has been implicated.5, 67 The nature of any synergy with PRRSV, if any, is unknown.
Cyanosis
This can also be a sign of severe toxaemia, terminal respiratory disease or circulatory failure. Purplish discoloration of the ears may be seen in systemic infections, such as salmonellosis and classical swine fever. A new disease, porcine dermatitis and nephritis syndrome,81 a disease with many features of immune-mediation, also features circular erythematous papules that can become cyanotic and necrotic. Porcine reproductive and respiratory syndrome has been implicated, but not proven to be the causative agent.
Control
Therapeutic strategies
Early attempts to reduce the effects of PRRS in herds focused on responses to the clinical signs, with many reports being descriptions of field syndromes that lacked scientific evidence of their veracity. One early description, however,34 does provide some field data on the effects of a paramunity inducer, ‘Baypamun’ (Bayer). Its use was reported to reduce piglet mortality by 16 per cent and to increase weight gain in piglets, compared to those in untreated control herds. Piglet mortality can also be reduced by providing extra feed, electrolytes, glucose and antibiotic therapy,69 whose contribution is mainly in combating many of the effects of secondary infections. Other indirect strategies involve improving the well-being of piglets through better bedding and warmth. Aspirin has also been suggested as a treatment for sows72 but its efficacy is questionable. Similarly, another antiinflammatory drug, dipyrone, has also given variable results. 14 Anti-inflammatory drugs can be teratogenic in early pregnancy63 and, furthermore, in mice, have been shown to reduce mucosal immunity to the related lactate dehydrogenase elevating virus.18
Colostrum gives some degree of protection4 and there has even been an attempt to confer passive immunity by provision of serum74 with little evidence of success, and with a high inherent risk of passing on infection. An experimental study showed little effect in modulating viraemia, although its onset was delayed.82
Husbandry strategies
The modification of husbandry practices has had the greatest effect in reducing losses due to PRRSV infection. Abortions and litter losses may be followed by failure of sows to come into oestrus.29, 44 This problem can, to some extent, be combated by delaying the re-mating of sows which have lost their litters until the normal time of weaning.63
Early observations of respiratory disease in nursery pigs following the reproductive losses included reports of more persistent respiratory problems.24, 39 The demonstration of high rates of seroconversion in nursery pigs95 and the identification of older piglets, not dams, as being the reservoir of the virus, and hence the source of infection86 led to the development of an ‘all in/all out’ or depopulation strategy for the nursery.35, 45, 46, 104 This strategy has had a great impact in reducing piglet losses and improving weight gains. An allied strategy, involving the placing of weaning piglets into geographically isolated units,25, 87 has also helped to eliminate disease in the nursery unit.
Vaccination strategies
The immunity to PRRSV is unusual in that detectable antibody after recovery from infection is short-lived, with many animals returning to a seronegative status five to seven months after seroconversion.4, 87 This indicates that the virus is a poor inducer of immunological memory. Nevertheless, a previous infection does confer some degree of short-term protection, but re-infection can occur after six months or longer.66 The degree of heterogeneity between strains is likely to have a large influence of subsequent infections,50, 51, 61 and this seems to be especially true where the two different genotypes from America and Europe are involved.97
A number of attempts have been made at vaccination with autogenous vaccines96 and with experimental killed vaccines58, 75 which have shown some ability to protect sows from the reproductive effects of the virus. Interestingly, killed vaccine has no effect on virus excretion in semen.71
The live attenuated vaccines have been shown to be stable and to prevent viraemia and clinical signs following challenge infection and have been widely used in the USA, initially in young pigs, and subsequently in sows. The risks concerned in the use of attenuated virus vaccines in pregnant sows is acknowledged,58 and their use is generally limited to specific times of gestation or to open sows.17 Recent experiences in Denmark have raised questions concerning the use of such vaccines, with particular concern being raised about the ability of the attenuated viruses to spread to non-vaccinated animals and to cross the placenta, resulting in effects that mirror infection with virulent virus.17
References
- ALBINA, E., 1997. Porcine reproductive and respiratory syndrome—10 years of experience (1986–1996) with this undesirable virus-infection. Veterinary Research, 28, 305–352.
- ALBINA, E., LEFORBAN, Y., BARON, T. & PLANA DURAN, J., 1992. Blue-eared pig disease in Brittany: A new test. The Veterinary Record, 130, 83–84.
- ALBINA, E., LEFORBAN, Y., BARON, T., PLANA DURAN, J. & VANNIER, P., 1992. An enzyme linked immunosorbent assay (ELISA) for the detection of antibodies to the porcine reproductive and respiratory syndrome (PRRS) virus. Annales de Recherches Veterinaires, 23, 167–176.
- ALBINA, E., MADEC, F., CARIOLET, R. & TORRISON, J., 1994. Immune response and persistence of the reproductive and respiratory syndrome virus in infected pigs and farm units. The Veterinary Record, 134, 567–573.
- ALLAN G.M., MCNEILLY, F., KENNEDY, S., DAFT, B., CLARKE, E.G., ELLIS, J.A., HAINES, D.M. & MEEHAN, B.M., 1998. Isolation of porcine circovirus-like viruses from pigs with a wasting disease in the USA and Europe. Journal of Veterinary Diagnostic Investigation, 10, 3–10.
- ANDREYEV, V.G., SCHERBAKOV, A.G., PYLNOV, A.A. & GUSEV, A.A., 1999. Genetic heterogeneity of PRRSV in Russia. Proceedings of the 3rd International Symposium on Porcine Reproductive Syndrome and Aujeszky’s Disease, Ploufragan, France, 21–24 July 1999. CNEVA-AFSSA & ISPAIA, France. 211–212.
- ANDREYEV, V.G., WESLEY, R.D., MENGELING, W.L., VORWALD, A.C. & LAGER, K.M., 1997. Genetic variation and phylogenetic relationships of 22 porcine reproductive and respiratory syndrome virus (PRRSV) field strains based on sequence analysis of open reading frame 5. Archives of Virology, 142, 993–1001.
- ANON, 1996. Porcine reproductive and respiratory syndrome. Office International des Epizooties Manual of Standards for Diagnostic Tests and Vaccines. 3rd edition. Paris, France. pp. 694–700.
- BANE, D., NEUMANN, E., HALL, W., HARLIN, K. & SLIFE, L., 1992. PRRS associated with fumosin contamination of feed. American Association of Swine Practitioners Newsletter, 4, 22–23.
- BAUTISTA, E.M., GOYAL, S.M., YOON, I.J., JOO, H.S. & COLLINS, J.E., 1993. Comparison of porcine alveolar macrophages and CL 2621 for the detection of porcine reproductive and respiratory syndrome (PRRS) virus and anti PRRS antibody. Journal of Veterinary Diagnostic Investigation, 5, 163–165.
- BENFIELD, D.A., NELSON, E.A. & CHRISTOPHER-HENNINGS, J., 1994. Porcine reproductive and respiratory syndrome (PRRS) viral proteins and antigenic variation. Proceedings of the 13th International Pig Veterinary Congress, Bangkok, Thailand, 26–30 June 1994. p. 62.
- BENFIELD, D.A., NELSON, E., COLLINS, J.E., HARRIS, L., GOYAL, S.M., ROBISON, D., CHRISTIANSON, W.T., MORRISON, R.B., GORCYCA, D. & CHLADEK, D., 1992. Characterization of swine infertility and respiratory syndrome (SIRS) virus (isolate ATCC VR-2332). Journal of Veterinary Diagnostic Investigation, 4, 127–133.
- BEYER, J., FICHTNER, D., SCHIRRMEIER, H., GRANZOW, H., POLSTER, U., WEILAND, E., BERNDT, A. & WEGE, H., 1998. Arterivirus PRRSV— experimental studies on the pathogenesis of respiratory disease. In: enjuanes, l., siddell, s.g. & spaan, w., (eds). Coronaviruses and Arteriviruses. New York: Plenum Publishing Corporation.
- BILODEAU, R., DEA, S., SAUVAGEAU, R.A. & MARTINEAU, G.P., 1991. Porcine reproductive and respiratory syndrome in Quebec. The Veterinary Record, 129, 102–103.
- BLOEMRAAD, M., DE KLUIJVER, E.P., PETERSEN, A., BURKHARDT, G. & WENSVOORT, G., 1994. Porcine reproductive and respiratory syndrome: Temperature and pH stability of Lelystad virus and its survival in tissue specimens from viraemic pigs. Veterinary Microbiology, 42, 361–371.
- BOTNER, A., 1997. Diagnosis of PRRS. Veterinary Microbiology, 55, 295–301.
- BOTNER, A., STRANDBYGAARD, B., SORENSEN, K.J., HAVE, P., MADSEN, K.G., MADSEN, E.S. & ALEXANDERSEN, S., 1997. Appearance of acute PRRS-like symptoms in sow herds after vaccination with a modified live PRRS vaccine. The Veterinary Record, 141, 497–499.
- CAFRUNY, W.A., DESJARLAIS, S.E., HECHT, M.L., BROEN, J.J. & JAQUA, R.A., 1991. Enhancement of murine susceptibility to oral lactate dehydrogenase elevating virus infection by non-steroidal anti-inflammatory agents, and antagonism by misoprostol. Antiviral Research, 15, 77–83.
- CARMAN, S., SANFORD, S.E. & DEA, S., 1995. Assessment of seropositivity to porcine reproductive and respiratory syndrome (PRRS) virus in swine herds in Ontario—1978 to 1982. Canadian Veterinary Journal—Revue Veterinaire Canadienne, 36, 776–777.
- CHEON, D.S. & CHAE, C., 1999. Distribution of a Korean strain of porcine reproductive and respiratory syndrome virus in experimentally infected pigs, as demonstrated immunohistochemically and by in-situ hybridization. Journal of Comparative Pathology, 120, 79–88.
- CHRISTOPHER-HENNINGS, J., NELSON, E.A., NELSON, J.K., HINES, R.J., SWENSON, S.L., HILL, H.T., ZIMMERMAN, J.J., KATZ, J.B., YAEGER, M.J., CHASE, C.C.L. & BENFIELD, D.A., 1995. Detection of porcine reproductive and respiratory syndrome virus in boar semen by PCR. Journal of Clinical Microbiology, 33, 1730–1734.
- CHUEH, L.L., LEE, K.H., JENG, C.R. & PANG, V.F., 1999. A sensitive fluorescence in situ hybridization technique for detection of porcine reproductive and respiratory syndrome virus. Journal of Virological Methods, 79, 133–140.
- COLLINS, J.E., BENFIELD, D.A., CHRISTIANSON, W.T., HARRIS, L., HENNINGS, J.C., SHAW, D.P., GOYAL, S.M., MCCULLOUGH, S., MORRISON, R.B., JOO, H.S., GORCYCA, D. & CHLADEK, D., 1992. Isolation of swine infertility and respiratory syndrome virus (isolate ATCC VR-2332) in North America and experimental reproduction of the disease in gnotobiotic pigs. Journal of Veterinary Diagnostic Investigation, 4, 117–126.
- DE JONG, M., VANT’VELD, P. & CROMWIJK, W., 1991. The epidemiology of PRRS in the Netherlands. Report of the Second Seminar of the EC on Porcine Reproductive and Respiratory Syndrome (the new pig disease), Brussels, Belgium, 4–5 November 1991. pp. 2–7.
- DEE, S.A., MORRISON, R.B. & JOO, H.S., 1993. Eradication of porcine reproductive and respiratory syndrome (PRRS) virus using multi-site production: successes and failures. Proceedings of the 24th Annual Meeting of the American Association of Swine Practitioners, Kansas City, Missouri, USA. pp. 655–661.
- DEWEY, C., 1998. The porcine reproductive and respiratory syndrome panzootic. Compendium on Continuing Education for the Practicing Veterinarian, 20, S82.
- DONE, S.H. & PATON, D.J., 1995. Porcine reproductive and respiratory syndrome: Clinical disease, pathology and immunosuppression. The Veterinary Record, 136, 32–35.
- DONE, S.H., PATON, D.J., BROWN, I. & COOLEY, W., 1992. Pathology of porcine reproductive and respiratory syndrome in the UK. Proceedings of the 12th Congress of the International Pig Veterinary Society, The Hague, Netherlands, 17–20 August 1992. p. 111.
- DONE, S.H., PATON, D.J. & WHITE, M.E.C., 1996. Porcine reproductive and respiratory syndrome (PRRS)—A review, with emphasis on pathological, virological and diagnostic aspects. British Veterinary Journal, 152, 153–174.
- DONE, S.H., SPENCER, Y., PATON, D.J., DREW, T.W., HANNAM, D., HIGGINS, R., TWIDDY, N., SIMMONS, J.R. & WHITE, M., 1996. The pathology of natural infections with porcine reproductive and respiratory syndrome virus (PRRSV) in the UK. Pig Veterinary Journal, 36, 18–30.
- DREW, T.W., 1995. Comparative serology of porcine reproductive and respiratory syndrome in eight European laboratories, using immunoperoxidase monolayer assay and enzyme-linked immunosorbent assay. Revue Scientifique et Technique de L’Office International Des Epizooties, 14, 761–775.
- DREW, T.W., MEULENBERG, J.J.M., SANDS, J.J. & PATON, D.J., 1995. Production, characterization and reactivity of monoclonal antibodies to porcine reproductive and respiratory syndrome virus. Journal of General Virology, 76, 1361–1369.
- DUAN, X., NAUWYNCK, H.J. & PENSAERT, M.B., 1997. Virus quantification and identification of cellular targets in the lungs and lymphoid-tissues of pigs at different time intervals after inoculation with porcine reproductive and respiratory syndrome virus (PRRSV). Veterinary Microbiology, 56, 9–19.
- EGBERING, O., 1991. Attempts in the treatment of the porcine reproductive and respiratory syndrome (PRRS). Praktische Tierärzt, 72, 851–854.
- FREESE, W.R., JOO, H.S. & SIMONSON, R.R., 1994. A potential spontaneous elimination of porcine epidemic abortion respiratory syndrome (PEARS) virus spread in a commercial swine herd. Proceedings of the 13th International Pig Veterinary Congress, Bangkok, Thailand, 26–30 June 1994. p. 66.
- GONIN, P., PIRZADEH, B., GAGNON, C.A. & DEA, S., 1999. Seroneutralization of porcine reproductive and respiratory syndrome virus correlates with antibody response to the GP(5) major envelope glycoprotein. Journal of Veterinary Diagnostic Investigation, 11, 20–26.
- GUARINO, H., GOYAL, S.M. & MURTAUGH, M.P., 1997. Comparison of four RNA extraction methods for the detection of porcine reproductive and respiratory syndrome virus by RT-PCR. Microbiologica, 20, 319–324.
- GUARINO, H., GOYAL, S.M., MURTAUGH, M.P., MORRISON, R.B. & KAPUR, V., 1999. Detection of porcine reproductive and respiratory syndrome virus by reverse transcription-polymerase chain reaction using different regions of the viral genome. Journal of Veterinary Diagnostic Investigation, 11, 27–33.
- HALBUR, P.G., PAUL, P.S. & ANDREWS, J.J., 1991. Reproduction of respiratory disease with tissue filters for herds experiencing SIRS and endemic pneumonia. Proceedings of the Minnesota Swine Conference for Veterinarians, St Paul, Minnesota, 15 September 1991.
- HALBUR, P.G., PAUL, P.S., FREY, M.L., LANDGRAF, J., EERNISSE, K., MENG, X.J., ANDREWS, J.J., LUM, M.A. & RATHJE, J.A., 1996. Comparison of the antigen distribution of two US porcine reproductive and respiratory syndrome virus isolates with that of the Lelystad virus. Veterinary Pathology, 33, 159–170.
- HALBUR, P.G., PAUL, P.S., FREY, M.L., LANDGRAF, J., EERNISSE, K., MENG, X.J., LUM, M.A., ANDREWS, J.J. & RATHJE, J.A., 1995. Comparison of the pathogenicity of two US porcine reproductive and respiratory syndrome virus isolates with that of the Lelystad virus. Veterinary Pathology, 32, 648–660.
- HARRIS, L., GORCYCA, D., SCHLESINGER, K., CHLADEK, D., SCHULTZ, R., CHRISTIANSON, W.T., COLLINS, J.E. & BENFIELD, D.A., 1992. The development and use of serological assays for the detection of antibody to the porcine epidemic abortion and respiratory syndrome (PEARS). Proceedings of the 12th Congress of the International Pig Veterinary Society, The Hague, Netherlands, 17–20th August, 1992. p. 123.
- HILL, H., 1990. Overview and history of Mystery Swine Disease (Swine Infertility and Respiratory Syndrome). Proceedings of the Mystery Swine Disease Committee Meeting, October 6, 1990, Denver Colorado. Livestock Conservation Institute, Madison, WI, USA.
- HOPPER, S.A., WHITE, M.E.C. & TWIDDY, N., 1992. An outbreak of blue eared pig disease (porcine reproductive and respiratory syndrome) in four pig herds in Great Britain. The Veterinary Record, 131, 140–144.
- JOO, H.S. & DEE, S., 1993. Recurrent PRRS problems in nursery pigs. Proceedings of the Allen D Leman Swine Conference, University of Minnesota, USA. pp. 85–88.
- KEFFABER, K., STEVENSON, G., VAN ALSTINE, W., KANITZ, C., HARRIS, L., GORCYCA, D., SCHLESINNGER, K., SCHULTZ, R., CHADEK, D. & MORRISON, R., 1992. SIRS virus infection in nursery/grower pigs. American Association of Swine Practitioners Newsletter, 4, 38–40.
- KEFFABER, K.K., 1989. Reproductive failure of unknown etiology. American Association of Swine Practitioners Newsletter, 1, 1–10.
- KIM, H., KWANG, J., YOON, I., JOO, H. & FREY, M., 1993. Enhanced replication of porcine reproductive and respiratory syndrome (PRRS) virus in a homogenous subpopulation of MA 104 cells. Archives of Virology. 133, 477–483.
- KONDO, H., KURAMOCHI, T., TAGUCHI, M. & ITO, M., 1997. Serological studies on porcine Pneumocystis carinii pneumonia: Kinetics of the antibody titres in swine herds and the association of porcine reproductive and respiratory syndrome virus infection. Journal of Veterinary Medical Science, 59, 1161–1163.
- LAGER, K.M., MENGELING, W.L. & BROCKMEIER, S.L,. 1995. Limited cross-protection between two strains of porcine reproductive and respiratory syndrome virus in pregnant swine. Abstracts of the Second International Symposium on Porcine Reproductive and Respiratory Syndrome (PRRS), Copenhagen, Denmark, 9–10 August 1995. p. 10.
- LAGER, K.M., MENGELING, W.L. & BROCKMEIER, S.L., 1999. Evaluation of protective immunity in gilts inoculated with the NADC-8 isolate of porcine reproductive and respiratory syndrome virus (PRRSV) and challenge-exposed with an antigenically distinct PRRSV isolate.American Journal of Veterinary Research, 60, 1022–1027.
- LAROCHELLE, R. & MAGAR, R., 1997. Detection of porcine reproductive and respiratory syndrome virus in paraffin-embedded tissues— comparison of immunohistochemistry and in-situ hybridization. Journal of Virological Methods, 63, 227–235.
- LAROCHELLE, R. & MAGAR, R., 1997. Evaluation of the presence of porcine reproductive and respiratory syndrome virus in packaged pig meat using virus isolation and polymerase chain reaction (PCR) method. Veterinary Microbiology, 58, 1–8.
- LEGEAY, O., BOUNAIX, S., DENIS, M., ARNAULD, C., HUTET, E., CARIOLET, R., ALBINA, E. & JESTIN, A., 1997. Development of a RT-PCR test coupled with a microplate colorimetric assay for the detection of a swine Arterivirus (PRRSV) in boar semen. Journal of Virological Methods, 68, 65–80.
- LOULA, T., 1990. Clinical presentation of mystery pig disease in the breeding herd and suckling piglets. Proceedings of the Mystery Swine Disease Committee Meeting, Denver, Colorado, USA, October 1990. Livestock Conservation Institute, Madison WI, USA 29–31.
- MAGAR, R., LAROCHELLE, R., DEA, S., GAGNON, C.A., NELSON, E.A., CHRISTOPHER-HENNINGS, J. & BENFIELD, D.A., 1995. Antigenic comparison of Canadian and US isolates of porcine reproductive and respiratory syndrome virus using monoclonal-antibodies to the nucleocapsid protein. Canadian Journal of Veterinary Research—Revue Canadienne de Recherche Veterinaire, 59, 232–234.
- MARDASSI, H., WILSON, L., MOUNIR, S. & DEA, S., 1994. Detection of porcine reproductive and respiratory syndrome virus and efficient differentiation between Canadian and European strains by reverse transcription and PCR amplification. Journal of Clinical Microbiology, 32, 2197–2203.
- MCGINLEY, M.J., BREVIK, A., CISZEWSKI, D., ZIMMERMAN, J., SWENSON, S., YOON, K.J., WILLS, B., PLATT, K.B., HILL, H.T. & SHIBLEY, G.P., 1994. Prospects for protection against PRRS virus-induced reproductive losses by vaccination. Proceedings of the 25th Annual Meeting of the American Association of Swine Practitioners, Chicago, Illinois, USA 5–8 March 1994. pp. 40–41.
- MENGELING, W.L., LAGER, K.M. & VORWALD, A.C., 1995. Diagnosis of porcine reproductive and respiratory syndrome. Journal of Veterinary Diagnostic Investigation, 7, 3–16.
- MENGELING, W.L., LAGER, K.M. & VORWALD, A.C., 1998. Clinical effects of porcine reproductive and respiratory syndrome virus on pigs during the early postnatal interval. American Journal of Veterinary Research, 59, 52–55.
- MENGELING, W.L., LAGER, K.M. & VORWALD, A.C., 1999. Safety and efficacy of vaccination of pregnant gilts against porcine reproductive and respiratory syndrome. American Journal of Veterinary Research, 60, 796–801.
- MENGELING, W.L., VORWALD, A.C., LAGER, K.M. & BROCKMEIER, S.L., 1996. Diagnosis of porcine reproductive and respiratory syndrome using infected alveolar macrophages collected from live pigs. Veterinary Microbiology, 49, 105–115.
- MEREDITH, M.J., 1995. Porcine reproductive and respiratory syndrome (PRRS). 1. Pig Disease Information Centre, Department of Clinical Veterinary Medicine, University of Cambridge, UK. August 1995: Boehringer Ingelheim Vetmedica GmbH.
- MEULENBERG, J.J.M., DENBESTEN, A.P., DEKLUYVER, E., VANNIEUWSTADT, A. & WENSVOORT, G., 1997. Molecular characterization of Lelystad virus. Veterinary Microbiology, 55, 197–202.
- MEULENBERG, J.J.M., HULST, M.M., DEMEIJER, E.J., MOONEN, P.L.J.M., DEN BESTEN, A., DE KLUYVER, E.P., WENSVOORT, G. & MOORMANN, R.J.M., 1994. Lelystad virus belongs to a new virus family, comprising lactate dehydrogenase-elevating virus, equine arteritis virus, and simian hemorrhagic fever virus. Archives of Virology, Suppl. 9, 441–448.
- MOLITOR, T., 1993. Immune responses to PRRS virus. Proceedings of the Allen D Leman Swine Conference, University of Minnesota, USA. pp. 49–50.
- MOROZOV, I., SIRINARUMITR, T., SORDEN, S.D., HALBUR, P.G., MORGAN, M.K., YOON, K.J. & PAUL, P.S., 1998. Detection of a novel strain of porcine circovirus in pigs with postweaning multisystemic wasting syndrome. Journal of Clinical Microbiology, 36, 2535–2541.
- MORRISON, R.B., 1993. Porcine reproductive and respiratory syndrome in weaned pigs. Proceedings of the 24th Annual Meeting of the American Association of Swine Practitioners, Kansas City, Missouri, USA. pp. 309–311.
- MUIRHEAD, M.R., (1992). Mystery disease, blue ear—Control program cuts losses dramatically. International Pigletter, 12, 9–11.
- NELSEN, C.J., MURTAUGH, M.P. & FAABERG, K.S., 1999. Porcine reproductive and respiratory syndrome virus comparison: Divergent evolution on two continents. Journal of Virology, 73, 270–280.
- NIELSEN, T.L., NIELSEN, J., HAVE, P., BAEKBO, P., HOFFJORGENSEN, R. & BOTNER, A., 1997. Examination of virus shedding in semen from vaccinated and from previously infected boars after experimental challenge with porcine reproductive and respiratory syndrome virus. Veterinary Microbiology, 54, 101–112.
- OHLINGER, V.F., WEILAND, F., HAAS, B., VISSER, N., AHL, R., METTENLEITER, T.C., WEILAND, E., RZIHA, H.J., SAALMULLER, A. & STRAUB, O.C., 1991. Aetiological studies of the porcine reproductive and respiratory syndrome (PRRS). Tierärztliche Umschau, 46, 703–708.
- OLEKSIEWICZ, M.B., BOTNER, A., MADSEN, K.G. & STORGAARD, T., 1998. Sensitive detection and typing of porcine reproductive and respiratory syndrome virus by RT-PCR amplification of whole viral genes. Veterinary Microbiology, 64, 7–22.
- PLAGGE, G., 1993. Administering antibodies to piglets of low birthweight by injection of blood serum does not reduce losses. Praktijkonderzoek Varkenshouderij, 7, 12–13.
- PLANA DURAN, J., BASTONS, M., URNIZA, A., VAYREDA, M., VILA, X. & MANE, H., 1997. Efficacy of an inactivated vaccine for prevention of reproductive failure induced by porcine reproductive and respiratory syndrome virus. Veterinary Microbiology, 55, 361–370.
- POL, J.M.A., WAGENAAR, F. & REUS, J.E.G., 1997. Comparative morphogenesis of three PRRS virus strains. Veterinary Microbiology, 55, 203–208.
- PRIETO, C., SUAREZ, P., SIMARRO, I., GARCIA, C., MARTINRILLO, S. & CASTRO, J.M., 1997. Insemination of susceptible and preimmunized gilts with boar semen containing porcine reproductive and respiratory syndrome virus. Theriogenology, 47, 647–654.
- ROOF, M.B., GINGERICH-FEIL, D., GORCYCA, D., SCHLESINGER, K. & CHLADEK, D., 1995. Serological evaluation of pigs vaccinated with RespPRRSR using the IDEXX PRRS ELISA. Abstracts of the Second International Symposium on Porcine Reproductive and Respiratory Syndrome (PRRS), Copenhagen, Denmark, 9–10 August 1995. p. 50.
- ROSSOW, K.D., 1998. Porcine reproductive and respiratory syndrome. Veterinary Pathology, 35, 1–20.
- ROSSOW, K.D., MORRISON, R.B., GOYAL, S.M., SINGH, G.S. & COLLINS, J.E., 1994. Lymph-node lesions in neonatal pigs congenitally exposed to porcine reproductive and respiratory syndrome virus. Journal of Veterinary Diagnostic Investigation, 6, 368–371.
- SEGALES, J., PIELLA, J., MARCO, E., MATEUDEANTONIO, E.M., ESPUNA, E. & DOMINGO, M., 1998. Porcine dermatitis and nephropathy syndrome in Spain. The Veterinary Record, 142, 483–486.
- SHIBATA, I., MORI, M. & URUNO, K., 1998. Experimental infection of maternally immune pigs with porcine reproductive and respiratory syndrome (PRRS) virus. Journal of Veterinary Medical Science, 60, 1285–1291.
- SHIMIZU, M., YAMADA, S., MURAKAMI, Y., MOROZUMI, T., KOBAYASHI, H., MITANI, K., ITO, N., KUBO, M., KIMURA, K., KOBAYASHI, M., YAMAMOTO, K., MIURA, Y., YAMAMOTO, T. & WATANABE, K., 1994. Isolation of porcine reproductive and respiratory syndrome (PRRS) virus from heko-heko disease of pigs. Journal of Veterinary Medical Science, 56, 389–391.
- SHIN, J.H., KANG, Y.B., KIM, Y.J., YEOM, S.H., KWEON, C.H., LEE, W.Y., JEAN, Y.H., HWANG, E.K., RHEE, J.C., AN, S.H., CHO, I.S., OH, J.S., JOO, H.S., CHOI, C.S. & MOLITOR, T.W., 1993. Sero-epidemiological studies on porcine reproductive and respiratory syndrome in Korea: I. Detection on indirect fluorescent antibodies. Journal of Agricultural Science, 35, 572–576.
- SIRINARUMITR, T., ZHANG, Y.J., KLUGE, J.P., HALBUR, P.G. & PAUL, P.S., 1998. A pneumo-virulent United States isolate of porcine reproductive and respiratory syndrome virus induces apoptosis in bystander cells both in vitro and in vivo. Journal of General Virology, 79, 2989–2995.
- STEVENSON, G.W., VAN ALSTINE, W.G. & KANITZ, C.L., 1993. Endemic PRRS infection in nursery pigs originating from older viraemic pigs and not from dams. Proceedings of the 24th Annual Meeting of the American Association of Swine Practitioners, Kansas City, Missouri, USA. p. 707.
- STEVENSON, G.W., VANALSTINE, W.G. & KANITZ, C.L., 1994. Characterization of infection with endemic porcine reproductive and respiratory syndrome virus in a swine herd. Journal of the American Veterinary Medical Association, 204, 1938–1942.
- SUAREZ, P., DIAZGUERRA, M., PRIETO, C., ESTEBAN, M., CASTRO, J. M., NIETO, A. & ORTIN, J., 1996. Open reading frame 5 of porcine reproductive and respiratory syndrome virus as a cause of virus-induced apoptosis. Journal of Virology, 70, 2876–2882.
- SUAREZ, P., ZARDOYA, R., MARTIN, M.J., PRIETO, C., DOPAZO, J., SOLANA, A. & CASTRO, J.M., 1996. Phylogenetic relationships of European strains of porcine reproductive and respiratory syndrome virus (PRRSV) inferred from DNA sequences of putative ORF-5 and ORF-7 genes. Virus Research, 42, 159–165.
- SUAREZ, P., ZARDOYA, R., PRIETO, C., SOLANA, A., TABARES, E., BAUTISTA, J.M. & CASTRO, J.M., 1994. Direct-detection of the porcine reproductive and respiratory syndrome (PRRS) virus by reverse polymerase chain-reaction (RT-PCR). Archives of Virology, 135, 89–99.
- SUR, J.H., COOPER, V.L., GALEOTA, J.A., HESSE, R.A., DOSTER, A.R. & OSORIO, F.A. 1996. In vivo detection of porcine reproductive and respiratory syndrome virus RNA by in situ hybridization at different times postinfection. Journal of Clinical Microbiology, 34, 2280–2286.
- SUR, J.H., DOSTER, A.R. & OSORIO, F.A., 1998. Apoptosis induced in vivo during acute infection by porcine reproductive and respiratory syndrome virus. Veterinary Pathology, 35, 506–514.
- SWENSON, S.L., HILL, H.T., ZIMMERMAN, J.J., EVANS, L.E., LANDGRAF, J.G., WILLS, R.W., SANDERSON, T.P., MCGINLEY, M.J., BREVIK, A.K., CISZEWSKI, D.K. & FREY, M.L., 1994. Excretion of porcine reproductive and respiratory syndrome virus in semen after experimentally induced infection in boars. Journal of the American Veterinary Medical Association, 204, 1943–1948.
- TAKIKAWA, N., KOBAYASHI, S., IDE, S., YAMANE, Y., TANAKA, Y., HIGASHIHARA, M. & YAMAGISHI, H., 1997. Early serodiagnosis of porcine reproductive and respiratory syndrome virus infection of pigs by detection of slow-reacting and complement-requiring neutralizing antibody. Journal of Veterinary Medical Science, 59, 31–34.
- THACKER, B., 1992. Serological surveys in a herd before,during and after an outbreak of SIRS. American Association of Swine Practitioners Newsletter, 4, 40.
- TRAYER, T.P., 1994. Field impressions of autogenous viral vaccines. Proceedings of the 25th Annual Meeting of the American Association of Swine Practitioners, Chicago, Illinois, USA, 5–8 March 1994. pp. 42–43.
- VANWOENSEL, P.A.M., LIEFKENS, K. & DEMARET, S., 1998. Effect on viraemia of an American and a European serotype PRRSV vaccine after challenge with European wild-type strains of the virus. The Veterinary Record, 142, 510–512.
- WEIAND, E., WIECZOREKKROHMER, M., KOHL, D., CONZELMANN, K.K. & WEILAND, F., 1999. Monoclonal antibodies to the GP(5) of porcine reproductive and respiratory syndrome virus are more effective in virus neutralization than monoclonal antibodies to the GP(4). Veterinary Microbiology, 66, 171–186.
- WENSVOORT, G., DE KLUYVER, E.P., LUIJTZE, E.A., DEN BESTEN, A., HARRIS, L., COLLINS, J.E., CHRISTIANSON, W.T. & CHLADEK, D., 1992. Antigenic comparison of Lelystad virus and swine infertility and respiratory syndrome (SIRS) virus. Journal of Veterinary Diagnostic Investigation, 4, 134–138.
- WENSVOORT, G., TERPSTRA, C., POL, J.M.A., TER LAAK, E.A., BLOEMRAAD, M., DE KLUYVER, E.P., KRAGTEN, C., VAN BUITEN, L., DEN BESTEN, A., WAGENAAR, F., BROEKHUIJSEN, J.M., MOONEN, P.L.J.M., ZETSTRA, T., DE BOER, E.A., TIBBEN, H.J., DE JONG, M.F., VAN’T VELD, P., GROENLAND, G.J.R., VAN GENNEP, J.A., VOETS, M.T., VERHEIJDEN, J.H.M. & BRAAMSKAMP, J., 1991. Mystery swine disease in the Netherlands: the isolation of Lelystad virus. Veterinary Quarterly, 13, 121–130.
- WENSVOORT, G., TERPSTRA, C., POL, J.M.A., TERLAAK, E.A., BLOEMRAAD, M., DE KLUYVER, E.P., KRAGTEN, C., VAN BUITEN, L., DEN BESTEN, A., WAGENAAR, F., BROEKHUIJSEN, J.M., MOONEN, P.L.J.M., ZETSTRA, T., DE BOER, E.A., TIBBEN, H.J., DE JONG, M.F., VAN’T VELD, P., GROENLAND, G.J.R., VAN GENNEP, J.A., VOETS, M.T., VERHEIJDEN, J.H.M. & BRAAMSKAMP, J., 1993. Mystery swine disease in the Netherlands: The isolation of Lelystad virus (Reprinted from the Veterinary Quarterly, 13, 121–130, 1991). Irish Veterinary Journal, 46, 60–68.
- WENSVOORT, G., TERPSTRA, C., POL, J.M.A. & WAGENAAR, F., 1991. Lelystad virus, the cause of Porcine Epidemic Abortion and Respiratory Syndrome (Mystery Swine Disease). Report of the Second Seminar of the EC on Porcine Reproductive and Respiratory Syndrome (the new pig disease), Brussels, Belgium, 4–5 November 1991. pp. 27–35.
- WILLS, R.W., ZIMMERMAN, J.J., YOON, K.J., SWENSON, S.L., HOFFMANN, L.J., MCGINLEY, M.J., HILL, H.T. & PLATT, K.B., 1997. Porcine reproductive and respiratory syndrome virus—routes of excretion. Veterinary Microbiology, 57, 69–81.
- YOON, I.J., JOO, H.S., CHRISTIANSON, W.T., MORRISON, R.B. & DIAL, G.D., 1993. Persistent and contact infection in nursery pigs experimentally infected with porcine reproductive and respiratory syndrome (PRRS) virus. Swine Health and Production, 1, 5–8.
- YOON, K.J., WU, L.L., ZIMMERMAN, J.J., HILL, H.T. & PLATT, K.B., 1996. Antibody-dependent enhancement (ADE) of porcine reproductive and respiratory syndrome virus (PRRSV) infection in pigs. Viral Immunology, 9, 51–63.
- YOON, K.J., WU, L.L., ZIMMERMAN, J.J. & PLATT, K.B., 1997. Field isolates of porcine reproductive and respiratory syndrome virus (PRRSV) vary in their susceptibility to antibody-dependent enhancement (ADE) of infection. Veterinary Microbiology, 55, 277–287.
- YUAN, S.S., NELSEN, C.J., MURTAUGH, M.P., SCHMITT, B.J. & FAABERG, K.S., 1999. Recombination between North American strains of porcine reproductive and respiratory syndrome virus. Virus Research, 61, 87–98.
- ZHANG, Y.J., SHARMA, R.D. & PAUL, P.S., 1998. Monoclonal antibodies against conformationally dependent epitopes on porcine reproductive and respiratory syndrome virus. Veterinary Microbiology, 63, 125–136.