- Infectious Diseases of Livestock
- Part 2
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Suid herpesvirus 2 infection
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Suid herpesvirus 2 infection
N EDINGTON
Introduction
Suid herpesvirus 2 (SHV-2), also called porcine cytomegalovirus (PCMV), is an ubiquitous virus that may cause foetal or neonatal deaths and has been associated with runting, rhinitis and pneumonia in piglets less than three weeks of age, or rhinitis in growing pigs. With the advent of xenotransplantation there is renewed interest in this virus.
Suid herpesvirus 2 was first identified by Done6 as a cause of cytomegalic cells with characteristic basophilic intranuclear inclusion bodies in the mucosal glands of turbinates of pigs. Restriction of viral replication to pigs, localization of the virus in the nasal mucosa, lachrymal and salivary glands, and occasionally in renal tubular epithelium, as well as ultrastructural studies,8, 28 indicated that this was a cytomegalovirus of the pig similar to those described for some other animal species.20, 22, 30 These viruses usually cause clinically silent infections in healthy adults but often induce severe, generalized infections in young animals. Virus may also cross the placenta. Like other herpesviruses, SHV-2 induces latent infection. A cell-associated viraemia can occur in the presence of circulating antibody.
In herds where good management is applied, and in which the virus is endemic, there may be no clinical signs or economic loss.
Suid herpesvirus 2 is likely to be present in countries whose domestic pig populations are of European or Asian origin.
Aetiology
Suid herpesvirus 2 is a beta herpesvirus, with an 80 to 100 nm icosahedral capsid surrounded by an envelope, making the complete virion 120 to 150 nm in diameter. The 45 to 70 nm core is usually elongated and may be oval, rectangular or dumb-bell shaped.8, 28 So far only the sequences of the DNA polymerase gene have been published,31 supporting its classification as a beta virus. The virus is sensitive to chloroform and ether.2
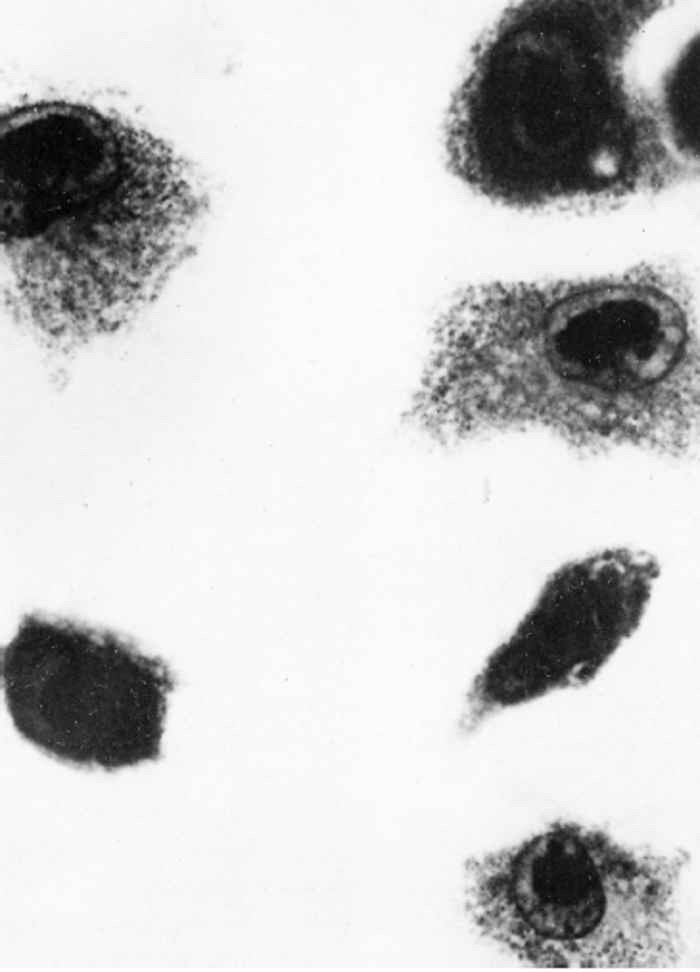
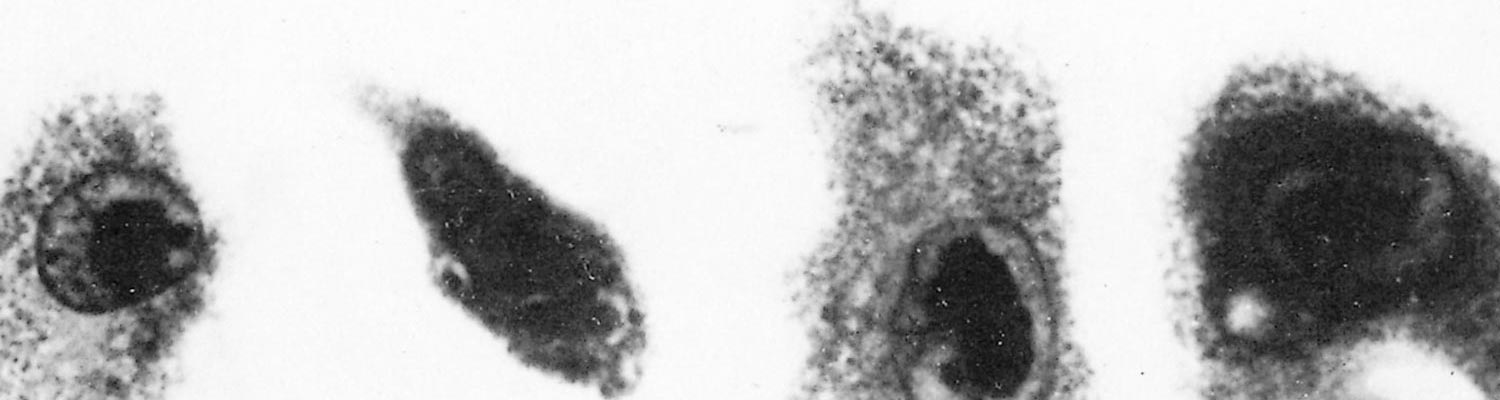
Watt29 found pulmonary macrophage cultures derived from three- to five-week-old piglets to be the most sensitive system for both primary isolation and propagation of virus. Cytomegaly and intranuclear inclusions develop 11 to 14 days after inoculation (Figure 83.1) and infectivity titres of up to 105,5 TCID50 may occur. This culture system has limitations because of the fastidiousness and variability of the cells, and because of residual adventitious viruses. Primary pig lung cells16 and a cell line derived from porcine Fallopian tube 3 also support viral replication, while it has been reported that 12-O-tetradecanoylphorbol 13-acetate accelerates viral replication in this system.15 Suid herpesvirus 2 appears to be host-specific both in vivo and in vitro.
In cultures, infected cells enlarge by up to six times and their mitochondria, endoplasmic reticulum, and Golgi apparatus swell.8 The large, intranuclear inclusions contain the nuclear capsids, often in crystalline array.27 The capsids acquire an electron-dense coat in the nucleus, the envelope derives from the inner nuclear membrane, and the virions come to lie free inside intracytoplasmic cisternae.
Epidemiology
Suid herpesvirus 2 has been detected wherever it has been sought. In the UK a serological survey indicated that over 95 per cent of herds had been exposed to the virus.19 The ubiquity of the infection and the possibility of transplacental infection indicate that even hysterotomy-derived littersmaybe infected.
In infected neonatal piglets the virus is shed predominantly in nasal secretions, but also in urine and ocular secretions. 11, 19 Levels of passive antibody fall initially during the period of virus excretion, but rise when the piglets are between 8 and 11 weeks old as a result of active production. They continue to increase until at least 23 weeks after infection. Susceptible sows show a similar pattern of excretion but, in addition, shedding of virus from the cervix occurs in some pregnant sows.
Recovery of virus from lung macrophages of apparently healthy pigs suggests that the virus either persists at this site or that it may establish a true latent infection. Infection may be reactivated after administration of corticosteroids.11
Pathogenesis
The mucous glands of the nasal mucosa are the primary site of viral replication. Thereafter a cell-associated viraemia develops which is detectable 14 to 16 days after infection in pigs older than three weeks, and from 5 to 19 days after infection in neonatal piglets.11
In animals older than three weeks, the virus occurs most commonly in the glands of the nasal mucosa but it may also be present in the Harderian and lachrymal glands and in the renal tubules. On rare occasions it is found in the mucous glands of the oesophagus and duodenal epithelium.
In foetuses or neonates, the virus has a predilection for reticuloendothelial cells. Inclusions occur in capillary endothelium, the sinus and sinusoidal cells of lymphoid tissues, and in macrophages.
In susceptible sows, isolation of the virus from cervical fluids 30 to 35 days after infection corresponds with the time when viral replication occurs in foetuses. Foetal death generally occurs 60 to 80 days after infection, suggesting that secondary spread of virus within the uterus may occur.12 Congenitally infected animals may be born live but die within 7 to 14 days of birth.
Semen from infected boars has not been shown to contain virus, but nasal secretions and urine are possible sources of infection for susceptible sows.
Clinical signs
Suid herpesvirus 2 infection in foetuses or neonates less than three weeks old results in a generalized disease which may be fatal. In neonates the clinical signs comprise subcutaneous oedema, pallor, dyspnoea, and generalized weakness. Field outbreaks involving litters of piglets have been characterized by shivering, sneezing, and respiratory distress.4, 5 Upto 25 per cent of the litter may die and the remaining pigs show poor weight gains. Uncomplicated infection in pigs older than three weeks is usually not clinically apparent, although rhinitis may ensue and subsequent secondary bacterial infection can result in the development of atrophic rhinitis.10
Pregnant sows may be listless and anorectic during the period when viraemia is present at 14 to 21 days after infection, but do not show pyrexia or any other clinical abnormality during pregnancy. However, transplacental infection is associated with the SMEDI syndrome (still births, mummification, embryonic death and infertility) as well as neonatal deaths and runt piglets.4, 5, 12, 18, 21
Pathology
Lesions in pigs older than three weeks are restricted to epithelial tissues and are scattered. In neonates and foetuses infection is generalized but restricted to reticulo-endothelial tissues.9
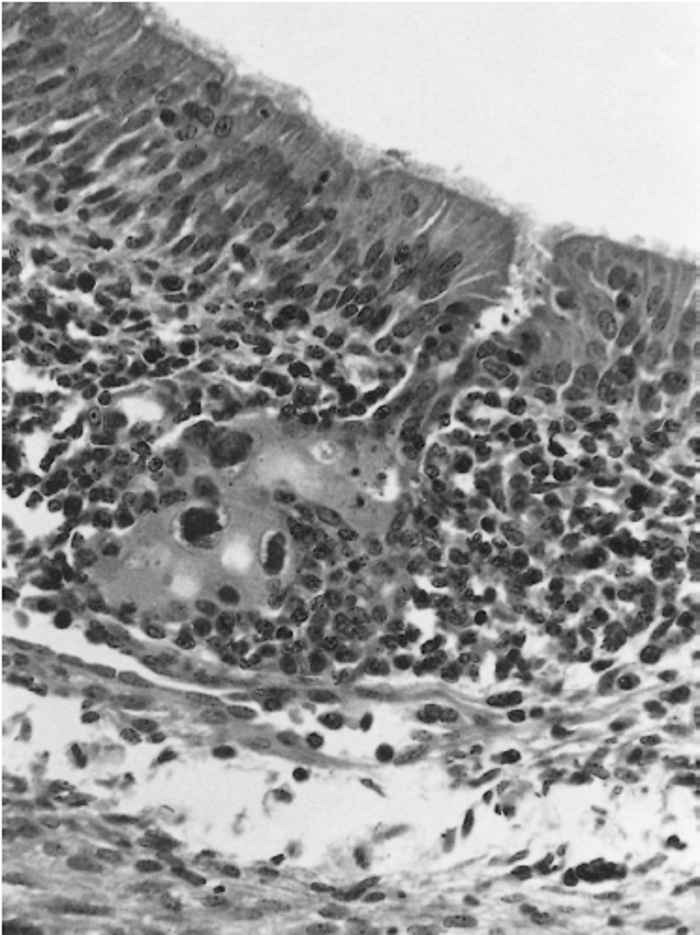
Macroscopic lesions are not usually seen in animals with disseminated infections. Histologically the characteristic cytomegaly and intranuclear inclusion bodies may be observed in the nasal mucous glands (Figure 83.2) as well as in the acini of the Harderian and lachrymal glands, and renal tubular epithelium. Infiltrates of lymphocytes and plasma cells may eventually obliterate the acini. In field infections, secondary bacterial infection with Bordetella bronchiseptica or Pasteurella multocida may induce lesions of atrophic rhinitis; these changes are not seen in pigs experimentally infected with SHV-2 alone.10 Interstitial nephritis may be a sequel to the infection.16
In generalized infections, petechiae and oedema may be widespread. The oedema in neonatal piglets manifests most commonly as an interlobular oedema of the lungs, hydrothorax, ascites, or, occasionally, anasarca. Subcutaneous oedema is most marked around the throat and tarsal joints. Petechiae are most marked in lymph nodes but also occur in the kidneys where subcapsular haemorrhages may result, resembling those associated with hog cholera and African swine fever.
Intranuclear inclusions are found in capillary endothelium and in sinusoidal cells, particularly in lymph nodes and the spleen, and also in the lung, renal glomeruli, choroid plexus, cerebellum and olfactory lobes.23 Monocytes in the lung may also contain inclusions.
Infection of the foetus does not produce characteristic macroscopic lesions and the foetuses are often autolysed.
Diagnosis and differential diagnosis
Indirect immunofluorescence and ELISA tests have been described for the serological detection of SHV-2 antibody.1, 18, 24 The ELISA tests are more sensitive than virus neutralization and have been adapted to distinguish IgG and IgM responses 25 and also demonstrate antigenic diversity.26 Neutralizing antibody titres in serum are rarely greater than one log. However, association of positive serological results with disease may be difficult because the infection is ubiquitous.
Isolation of virus is difficult because of the problems of obtaining and maintaining sensitive cell lines. Detection of virus may be effected by applying either indirect immunofluorescent antibody or special techniques to nasal swabs or scrapings of older pigs,7 or alternatively to tissues such as the lung, liver and central nervous system of aborted foetuses and neonates.12, 13 The characteristic cytomegaly and inclusion bodies may be demonstrated in histological sections of foetal or neonatal tissues. The polymerase chain reaction of the DNA polymerase gene could be used to detect PCMV.31
It is only in the area of reproductive losses that confusion arises. Porcine parvovirus, suid herpesvirus 1 (Aujeszky’s disease), hog cholera, and Leptospira spp. are all associated with reproductive losses. Within the herpesvirus family, PCMV as latent or persistent virus has to be differentiated from PHV-1 and the recently reported lymphotrophic PHV-3 and PHV-4.14
Control
Outbreaks of reproductive losses and rhinitis which have been associated with SHV-2 have generally been self-limiting. 4, 5 Concurrent bacterial infections may be controlled by the use of antibiotics. Virus-free herds can be established by hysterotomy, but the ability of SHV-2 to cross the placenta must always be considered, and offspring should be monitored serologically for at least 70 days after birth to ensure freedom from this infection.
References
- ASSAF, R., BOUILLANT, A.M.O. & DI FRANCO, E., 1982. Enzyme-linked immunosorbent assay (ELISA) for the detection of antibodies to porcine cytomegalovirus. Canadian Journal of Comparative Medicine, 46, 183–185.
- BOOTH, J.C., GOODWIN, R.F.W. & WHITTLESTONE, P., 1967. Inclusion-body rhinitis of pigs: Attempts to grow the causal agent in tissue cultures. Research in Veterinary Science, 8, 338–345.
- BOUILLANT, A.M.P., DULAC, G., WILLIS, N., GIRARD, A., GREIG, A.S. & BOULANGER, P., 1975. Viral susceptibility of a cell line derived from the pig oviduct. Canadian Journal of Comparative Medicine, 39, 450–456.
- CAMERON-STEPHEN, I.D., 1961. Inclusion-body rhinitis of swine. Australian Veterinary Journal, 37, 87–91.
- CORNER, A.H., MITCHELL, D., JULIAN, R.J. & MEADS, E.B., 1964. A generalised disease in piglets associated with the presence of cytomegalic inclusions. Journal of Comparative Pathology, 74, 192–199.
- DONE, J.T., 1955. An ‘inclusion body’ rhinitis of pigs. The Veterinary Record, 67, 525–527.
- DONE, J.T., 1958. Inclusion-body rhinitis of swine; nasal scrapings as an aid to diagnosis. The Veterinary Record, 70, 877–878.
- DUNCAN, J.R., RAMSEY, F.K. & SWITZER, W.P., 1965. Electron microscopy of cytomegalic inclusion disease of swine (inclusion body rhinitis). American Journal of Veterinary Research, 26, 939–946.
- EDINGTON, N., PLOWRIGHT, W. & WATT, R.G., 1976. Generalised cytomegalic inclusion disease: Distribution of cytomegalic cells and virus. Journal of Comparative Pathology, 86, 191–202.
- EDINGTON, N., SMITH, I.M., PLOWRIGHT, W. & WATT, R.G., 1976. Relationship of porcine cytomegalovirus and B. bronchiseptica to atrophic rhinitis in gnotobiotic piglets. The Veterinary Record, 98, 42–45.
- EDINGTON, N., WATT, R.G. & PLOWRIGHT, W., 1976. Cytomegalovirus excretion in gnotobiotic pigs. Journal of Hygiene, 77, 283–290.
- EDINGTON, N., WATT, R.G., PLOWRIGHT, W., WRATHALL, A.E. & DONE, J.T., 1977. Experimental transmission of porcine cytomegalovirus. Journal of Hygiene, 78, 243–251.
- EDINGTON, N., WRATHALL, A.E. & DONE, S., 1988. Porcine cytomegalovirus in early gestation. Veterinary Microbiology, 17, 117–128.
- EHLERS, B., ULRICH, S. & GLOLTZ, M., 1999. Detection of two novel herpesviruses with similarity to gammaherpesviruses. Journal of General Virology, 80, 971–978.
- KAWAMURA, H. & MATSUZAKI, S. 1996. Influence of 12-O-tetradecanoylphorbol 13-acetate on replication of porcine cytomegalovirus in the 19PFT cell line. Journal of Veterinary Medical Science, 58. 263–265.
- KELLY, D.F., 1967. Pathology of extranasal lesions in experimental inclusion body rhinitis of pigs. Research in Veterinary Science, 8, 472–478.
- L’ECUYER, C. & CORNER, A.H., 1966. Propagation of porcine cytomegalic inclusion disease virus in cell cultures. Preliminary report. Canadian Journal of Comparative Medicine, 30, 321–326.
- L’ECUYER, C., CORNER, A.H. & RANDALL, G.C.B., 1972. Porcine cytomegalic inclusion disease. Transplacental transmission. Proceedings of the 2nd International Congress of the Pig Veterinary Society, Hannover.
- PLOWRIGHT, W., EDINGTON, N. & WATT, R.G., 1976. The behaviour of porcine cytomegalovirus in commercial pig herds. Journal of Hygiene, 75, 125–135.
- PLUMMER, G., 1973. Cytomegaloviruses of man and animals. Progress in Medical Virology, 15, 92–125.
- RAC, R., 1961. Infectious rhinitis in pigs: Laboratory aspects. Australian Veterinary Journal, 37, 91–93.
- SMITH, M.G., 1959. The salivary gland viruses of man and animals. Progress in Medical Virology, 2, 171–202.
- STEPHANO HORNEDO, A. & EDINGTON, N., 1987. Encefalitis experimental por citomegalovirus porcino en cerdos gnotobióticos: Estudio histopatológico. Veterinaria Mexico, 18, 189–202.
- TAJIMA, T., HIRONAO, T., KAJIKAWA, T. & KAWAMURA, H., 1993. Application of ELISA for the seroepizootiological survey of antibodies against porcine cytomegalovirus. Journal of Veterinary Medical Science, 55. 421–424.
- TAJIMA, T., HIRONAO, T., KAJIKAWA, T., SUZUKI, Y. & KAWAMURU, H., 1994. Detection of antibodies against porcine cytomegalovirus from whole blood collected on the blood sampling paper. Journal of Veterinary Medical Science, 56, 189–190.
- TAJIMA, T. & KAWAMURU, H., 1998. Serological relationship among porcine cytomegalovirus Japanese isolates and an UK isolate. Journal of Veterinary Medical Science, 60, 107–109.
- VALICEK, L., SMID, B. & MEUSIK, J., 1973. Electron microscopy of porcine cytomegalovirus. Archiv Gesamte Virusforschung, 41, 344–353.
- VALICEK, L., SMID, B., PLEVAV & MEUSIK, J., 1970. Porcine cytomegalic inclusion disease virus. Electron microscopic study of the nasal mucosa. Archiv Gesamte Virusforschung, 32, 19–30.
- WATT, R.G., PLOWRIGHT, W., SABO, A. & EDINGTON, N., 1973. A sensitive cell culture system for the virus of porcine inclusion body rhinitis (cytomegalic inclusion disease). Research in Veterinary Science, 14, 119–121.
- WELLER, T.H., HANSHAW, J.B. & SCOTT, D.E., 1960. Serologic differentiation of viruses responsible for cytomegalic inclusion disease. Virology, 12, 130–132.
- WIDEN, B.F., LOWINGS, J.P., BELAK, S. & BANKS, M., 1999. Development of a PCR system for porcine cytomegalovirus detection and determination of the putative partial sequence of its DNA polymerase gene. Epidemiology & Infections,123, 177–180.